2.1. Phytochemical Analysis
Colorimetric determinations highlighted the presence of total polyphenols and flavonoids in all the samples of DR2 aubergine. Particularly, the amounts of polyphenols and flavonoids were found increased in DR2C with respect to DR2B extract (amount about doubled and tripled for polyphenols and flavonoids respectively) (
Table 1).
Eggplant is known to contain high levels of phenolic compounds, mainly phenolic acids (chlorogenic acid, caffeic acid,
p-coumaric acid) in pulp and anthocyanins, such as NAS and delphinidin conjugates in the peel [
37,
38]. Total phenolic content has been found varied in different extracts, with acidified ethanol being the most frequently used extraction solvent. Fresh eggplant peels were found to contain high levels of total phenols, with major differences due to the extraction method [
39,
40]. Lower amounts were reported in the extracts of whole fresh fruits [
41] and in those from dried peels [
42]. Several factors have been shown to affect the phenolic amount of eggplant, among which, the harvesting period [
43,
44].
Under our experimental conditions, the ratio between the total amounts of polyphenols and flavonoids, calculated as chlorogenic acid equivalents, was about 21.7 and 12.6 for DR2B and DR2C, respectively: this suggests that physiological ripeness markedly increases the flavonoid levels in DR2 eggplant peel. Our results agree with literature that reported flavonoids generally representing about 10–15% of total phenolics in eggplant [
45].
On the basis of the drug extract ratio (DER) values obtained for each sample (i.e., DR2B, 23:1; DR2C, 25:1), the total flavonoid amount was estimated to be 140 mg/kg (0.014%
w/
w) and 392 mg/kg (0.039 %
w/
w) in the raw material from DR2B and DR2C aubergine berries, respectively. According to Peterson and Dwyer [
46], who classified the flavonoid concentration in foods as low (0.1–39.9 mg/kg), moderate (40–99.9 mg/kg), and high (>100 mg/kg), the peels of DR2B and DR2C aubergine berries produced samples with high flavonoid contents. Similar results were reported by Ji et al. [
47], which found a total flavonoid amount of 116.4 mg/kg of fresh eggplant peel, in spite of undetectable levels in the pulp. Lower amounts of flavonoids and tannins were found in ethanolic extract from dried eggplant peels [
40,
42].
The HPTLC analysis of DR2B and DR2C extracts revealed the presence of several polyphenols, mainly visualized as fluorescent spots at 366 nm after derivatization with NPR (
Figure S1). HPTLC analysis was also performed on the extract from the pulp and the edible part of DR2 aubergine berries at both ripening stages. The pulp represented the mesocarp of the fruit, obtained after peel separation, while the edible part was the pericarp after removing the calix. Also, control aubergine berries from a 67/3 variety of
S. melongena obtained from the same experimental fields, was included in the analysis (
Figure S1). The edible part exhibited a polyphenolic fingerprint similar to that of the relative peel, with lower amounts as evidenced by the fluorescence intensity of the spots; conversely, in pulp samples, several compounds disappeared, thus suggesting that peel represents the mainly phenolic-enriched part of aubergine berries. Comparing the control eggplant and DR2, a similar trend was found at different ripening stages, with some differences in the upper part of chromatogram (
Figure S1). The HPTLC chromatograms highlighted the presence of rutin, chlorogenic acid, caffeic acid, and epicatechin in both DR2B and DR2C samples (
Figure S2). Conversely, apigenin was a peculiar compound of DR2C extract (
Figure S2,
Table 2).
Among phytochemicals identified and determined by densitometric analysis, chlorogenic acid and rutin were found to be ubiquitous, with a significantly increased content in DR2B extract respect to DR2C (
Figure S2;
Table 2). The control 67/3C eggplant sample contained chlorogenic acid, rutin, apigenin, while it was lacking epicatechin and caffeic acid (
Figure S1). Regarding anthocyanins, D3R was found to be the most abundant compound at both ripening stages, with low amounts of NAS and delphinidin-3-
O-β-
d-glucoside: the ratio between D3R and NAS was about 18 and 19 for DR2B and DR2C, respectively; this confirms that it represents the characteristic anthocyanin of DR2 eggplant variety, in agreement with what reported by Mennella et al. [
33].
Anthocyanins were found abundant in these extracts from aubergine, with a total amount of 0.53 ± 0.012 mg cyanidin-3-glucoside equivalent and 76.44 ± 3.82 mg of D3R equivalent per 100 g of fresh fruit [
39,
41]. The acidified ethanolic extract from eggplant peel was found to contain 62.92 ± 0.15 mg delphinidin-3-glucoside equivalent/100 g of extract, with a ratio between anthocyanins and flavonoids of about 4 [
42].
Although for our samples the total anthocyanin amount was not determined, taking into account that DR2 eggplant mainly produces delphinidin-3-glucoside as a peculiar anthocyanin [
33], we estimated the ratio between anthocyanins and flavonoids was about 35 and 9 for DR2B and DR2C, respectively. This suggests that ripening increases the flavonoid amount, although anthocyanins were important constituents of eggplant peel. DR2B and DR2C extracts were also characterized by the presence of chlorogenic acid as a representative hydroxycinnamic acid, with a significant lowered amount (about 2.4-fold lower than DR2B) for ripening stage C. The seasonal variations of phenolic compounds have been also reported for other Solanaceae species: a decreased concentration of chlorogenic acid was found in long eggplants (about 70% lower levels), as well as in other species such as tomato (about a 43% reduced levels) with rising temperatures due to the summer season [
43,
48]. The levels of phenolic acids have also been found increased in relation to the organic growth, thus suggesting that they can be produced as a plant response to environmental stressors [
49].
For eggplant varieties containing D3R, Mennella et al. [
33] reported a 1.5-fold reduction of chlorogenic acid and total polyphenols ratio at the physiological ripening stage, with respect to the commercial one. For our samples, we found that the chlorogenic acid and total polyphenols ratio was about 1.5 and 0.4 for DR2B and DR2C extracts, respectively, with a reduction of 3.7-fold: this can be explained on the basis of the lower amounts of chlorogenic acid and the high anthocyanin levels in eggplant peel with respect to the pulp.
The phenolic fingerprint of the extracts was also detected by HPLC-PDA analysis [
50], showing a different pattern for DR2B and DR2C samples (expressed as μg/mg of dry extract) (
Table 3;
Figure S3). Collectively, DR2B extract was characterized by a predominant presence of chlorogenic acid and a low amount of gallic acid, while DR2C displayed appreciable amounts of chlorogenic acid,
t-ferulic acid, and
t-cinnamic acid, along with small quantities of gallic acid, vanillic acid, naringin, syringic acid, 3-OH-4-MeO benzaldehyde, and 2,3-diMeO benzoic acid (
Table 3).
García-Salas et al. [
43] previously found the whole long eggplant to contain the flavonols quercetin 3,4′-
O-diglucoside and kaempferol-3-
O-rutinoside, hydroxycinnamic acids, homovanillic acid hexose, and delphinidin-3-
O-rutinoside. Accordingly, Singh et al. [
51] reported eggplant contains chlorogenic acids, with traces of quercetin and myricetin glucosides. Hydroxycinnamic acid conjugates were peculiar to the whole aubergine berry and pulp [
44]; however, chlorogenic acid, ferulic acid, and caffeic acid were also found in the eggplant peel [
38,
52]. A complex flavonol profile was described for the eggplant skins [
49], in spite of a low amount for the pulp [
51]. Our results on the peel extracts from DR2 aubergine berries partly agree with literature evidence, mainly regarding the hydroxycinnamic acid composition, while the flavones apigenin and naringenin were firstly identified. Naringenin was also found to be transported and incorporated in the outer cuticle matrix [
53], while apigenin was reported abundant in eggplant leaves [
54].
2.2. DR2C Extract Exerts Cytoprotective Effect against Oxidative Cell Damage
Considering the characteristic polyphenolic composition, DR2B and DR2C were assayed for their ability to interfere with the oxidative injury induced by the known oxidative agent
tert-butyl hydroperoxide (tBOOH) in Caco2 cells, which represent a sensitive biological model to oxidant injury and have been widely used to study the effect of reactive oxygen metabolites and antioxidant agents [
34]. The effect was evaluated under both normoxia and CoCl
2-induced hypoxia, during which respiratory distress and oxidative stress occur as a result of the altered oxygen homeostasis [
55]. In mammalian systems, CoCl
2 is known to mimic hypoxia through stabilization of HIF-1α [
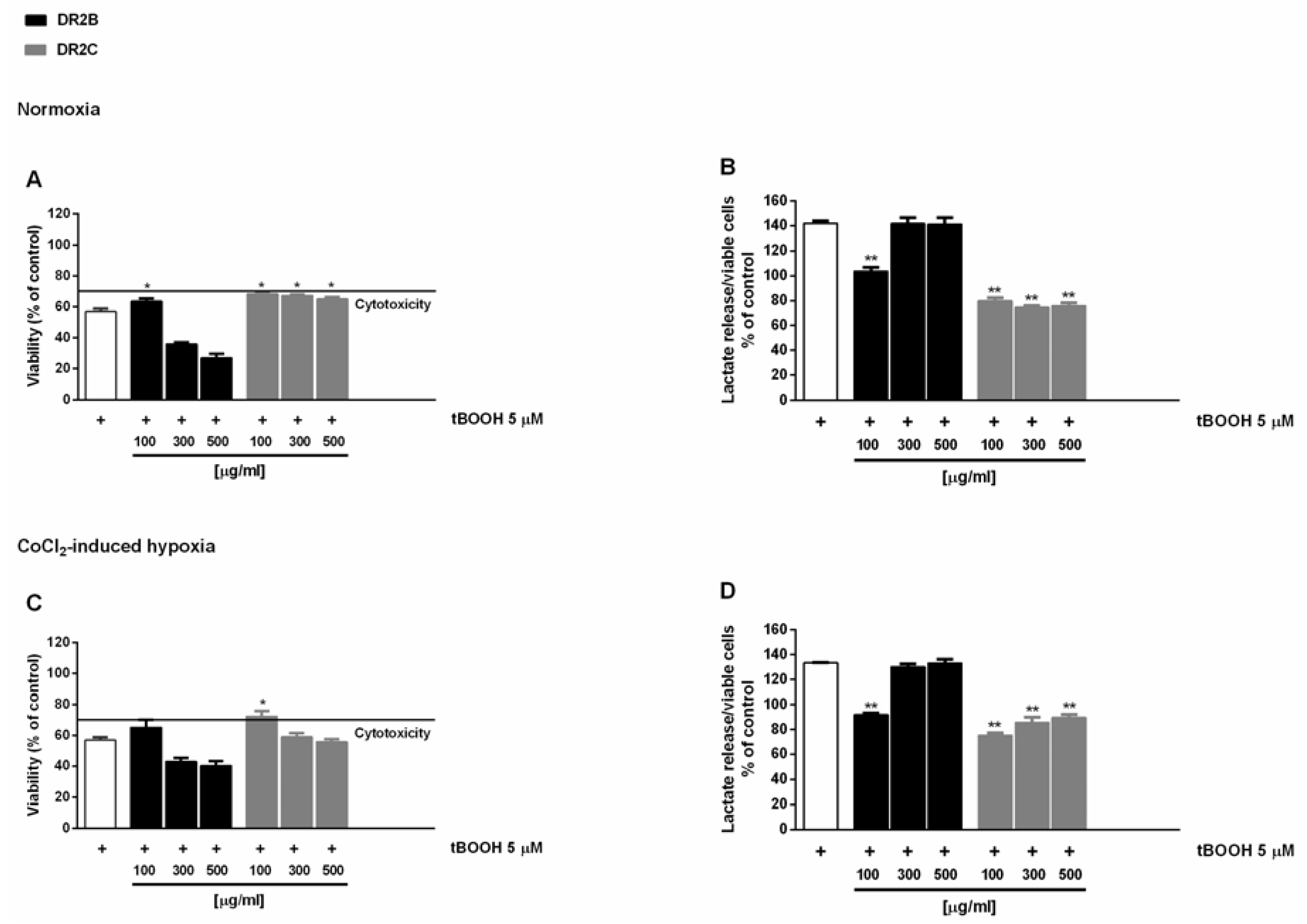
56]. Preliminary experiments showed that DR2B-induced cytotoxicity signs starting from the concentration of 300 μg/mL, with a maximum inhibition of cell viability of about 30% with respect to control (
Figure S4). Conversely, DR2C extract was nontoxic under both normoxic and hypoxic conditions, and slightly reduced the lactate levels extruded by treated cells (
Figure S4).
In the antioxidative cytoprotective assay, the pro-oxidant agent tBOOH reduced cell viability by about 40% with respect to the control under both normoxia and hypoxia (
Figure 1A,C), with an increased lactate metabolism with respect to control of about 40% and 30%, respectively (
Figure 1B,D).
The DR2C extract displayed cytoprotective effects by inhibiting the tBOOH-induced cytotoxicity of about 21% and 23% under normoxia and hypoxia (
Figure 1A,C): under these experimental conditions, lactate levels induced by tBOOH were found to be markedly reduced by DR2C (
Figure 1B,D), reaching a maximum inhibition of 67% and 58% under normoxia and hypoxia.
A slight but significant cytoprotection (lower than 10% increase of cell viability), with a 42% reduction of tBOOH-induced lactate metabolism, was also found at the lowest concentration of DR2B under normoxic conditions (
Figure 1A,B). These results are remarkable, and highlight that the tested extracts can counteract the oxidative damage, thus blocking the tBOOH-induced upregulation of Warburg effect.
Caco2 cells, along with several types of cancer cells, are characterized by peculiar energetic requirements, due to their need to grow quickly with respect to normal cells. Therefore, they give a modified metabolism with increased aerobic glycolysis and lactate production. This glycolytic phenotype, known as the Warburg effect, leads to an increased nontoxic oxidative stress with a loss of antioxidant capabilities and seems to be responsible for a highly resistant and malignant behavior of cancer cells [
57]. When Caco2 and other glycolytic cancer cells are subjected to an exogenous oxidative stressor, ROS increase over tolerable levels; therefore, resistant cells markedly upregulate their glycolytic metabolism to overcome oxidative injury [
58]: as a consequence, high lactate levels are extruded by cells with an increased steady-state ROS condition [
57]. In this context, affecting the glycolytic phenotype of Caco2 cells can represent a measure of the antioxidant power of the tested samples.
Literature evidence suggests a possible involvement of anthocyanins in the registered effects, since both NAS and D3R displayed protective properties in osteoclasts against the tBOOH-induced oxidative damage, likely by scavenging free radicals. This effect could be ascribed to the –OH moieties on the B ring, to the number of OH– moieties in total, or to the type and extent of glycosylation and acylation [
59,
60]. Along with anthocyanins, the tested samples are important source of phenylpropanoids, mainly chlorogenic acid (
Table 3).
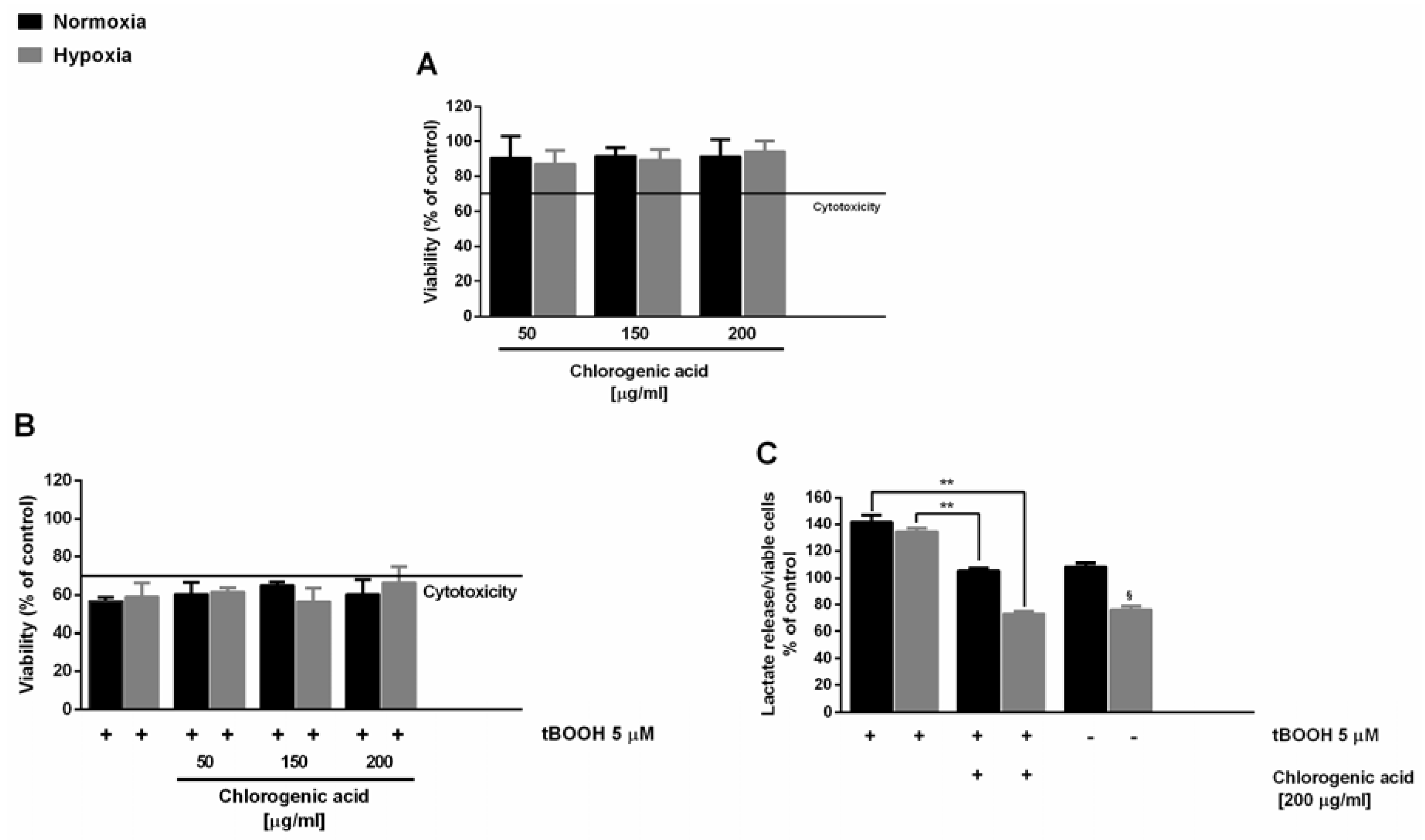
In this context, here, we also evaluated the antioxidant cytoprotective activity of chlorogenic acid, in order to highlight its involvement in the activity of the extracts. Under our experimental conditions, chlorogenic acid induced nontoxic effects up to the highest concentration revealed in the DR2C extract (about 200 μg/mL), with a significant reduction of lactate levels under hypoxia (
Figure 2A). When assessed for cytoprotection, the substance induced only a slight inhibition of the tBOOH-induced cytotoxicity (
Figure 2B), in spite of a remarkable lowering of t-BOOH-induced lactate secretion (about 40% and 60% reduction under normoxia and hypoxia, respectively) (
Figure 2C). This suggests that this compound can partly contribute to the activity of the extracts, although it does not represent the only bioactive constituent.
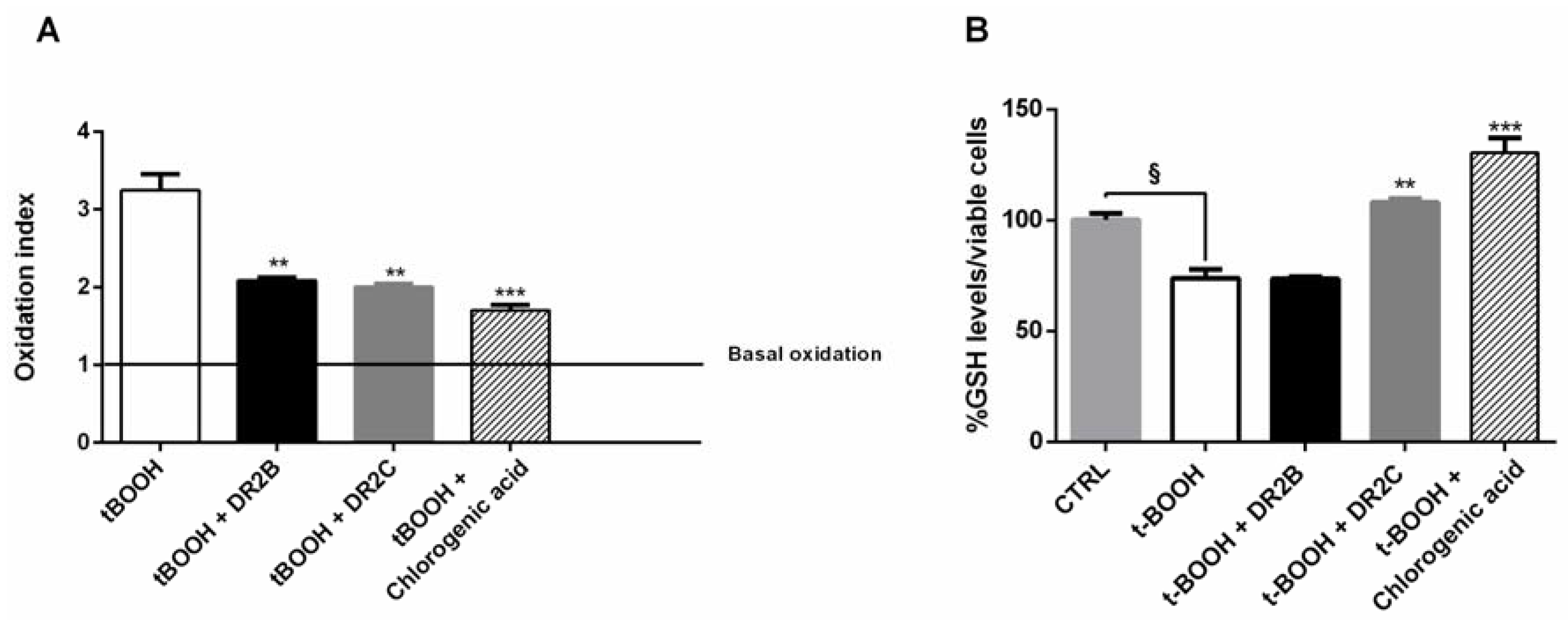
In order to better characterize the antioxidant cytoprotective effects of the tested samples, we also measured if the treatments may affect the intracellular content of ROS and GSH. In spite of a significant increase of ROS level induced by tBOOH (about 3-fold increase), both DR2B and DR2C extracts, and chlorogenic acid, significantly lowered the tBOOH-induced ROS levels, with a reduction of about 32%, 35%, and 45% respectively (
Figure 3A).
GSH levels were found reduced by tBOOH by about 25% with respect to the control; DR2B extract did not affect the tBOOH-induced GSH levels, while in the presence of DR2C and chlorogenic acid, the GSH content was significantly restored with respect to tBOOH treatment (about 1.5- and 1.8-fold, respectively) (
Figure 3B). These results suggest that DR2C extract and chlorogenic acid not only counteract the oxidative damage of tBOOH, but also restore the antioxidative cell defenses, which are known to be downregulated in cancer cells, and modulate lactate production: altogether, these mechanisms can contribute to their antioxidative cytoprotective effects. However, the involvement of other specific factors or signaling pathways cannot be excluded.
The present results agree with a previous study, in which eggplant stalks were found to possess cytoprotective, antioxidative, and anti-inflammatory properties, thus suggesting possible further interest for eggplant product waste as a source of nutraceuticals and new pharmaceutical agents [
61].
2.3. Antioxidant Activity Assays
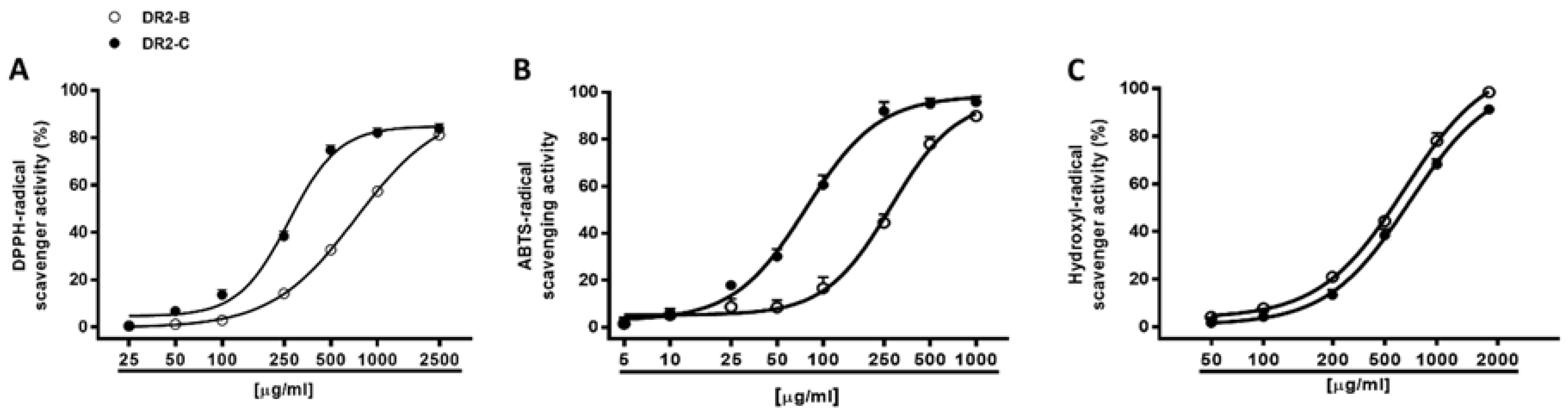
Different antioxidant mechanisms, including radical scavenging and chelating activity, were also evaluated for the DR2B and DR2C extracts by spectrophotometric assays. The scavenging activity was evaluated against both synthetic DPPH• and ABTS•+ radicals and against the hydroxyl radical.
Under our experimental conditions, both DR2B and DR2C extracts (5–2500 μg/mL) inhibited, in a significant and concentration dependent manner, the DPPH
• and ABTS
•+ radicals, with a higher potency of DR2C (
Figure 4A,B): the IC
50 value of DR2C was about 2.7- and 3.6-fold lower than that of DR2B (
Table 4). The positive control Trolox was about 54- and 39-fold more effective than DR2C against DPPH and ABTS, respectively.
According to the Pearson analysis, the scavenger activities against DPPH and ABTS radicals were significantly correlated for both DR2B and DR2C extracts (
Table 5 and
Table 6), although they exhibited higher potency against ABTS with respect to DPPH radical (2.6- and 3.4-fold respectively). DPPH is a pre-existing radical which can be neutralized by electron or hydrogen transfer, thus forming a stable diamagnetic molecule; conversely, ABTS cation, generated just before the experiments through different activators, requires an electron-transfer process to be scavenged, and is more reactive than DPPH [
62]. These important chemical features can lead to different affinities and kinetics reactions, and in the scavenging potencies. Our results suggest a major involvement of electronic transfer as scavenging mechanism. Taking into account ABTS requires an aqueous reaction media; in spite of the methanolic or ethanolic environment of DPPH, we hypothesize that radical scavenging effects of DR2B and DR2C extracts are mainly due to polar bioactive constituents.
The extracts were also assayed for their ability to neutralize hydroxyl radical, thus resulting in both being able to scavenge this ROS metabolite, in a significant and concentration-dependent manner, with DR2B being slightly more potent than DR2C (
Figure 4C;
Table 4). The positive control Trolox resulted at least 10-fold more potency than the tested extracts (
Table 4).
As estimated by the Pearson analysis, for all the samples, DPPH, ABTS, and hydroxyl radical scavenger activities were significantly correlated, with a correlation coefficient from 0.91 to 0.99 (
Table 5 and
Table 6). The correlation of DPPH and hydroxyl radical scavenger activity could be due to their common high reactivity, and suggests the presence in the extracts of some constituents able to counteract both species, among which includes phenolic compounds with multiple hydroxyl groups (i.e., caffeic acid and gallic acid) [
63]. However, due to the low levels of these compounds in the extracts (
Table 3), high concentrations of the samples are required for exerting the scavenger activity. Taking into account the phytochemical composition (
Table 2 and
Table 3), the contribution of phenolic acids, flavonoids (or flavonoid glycosides), and anthocyanins to the radical scavenger activity of the extracts is expected. In fact, numerous naturally occurring flavonoids have been highlighted to possess hydroxyl radical scavenging properties, and to interfere with other ROS species, thus suggesting their important protective role for health [
64].
Hydroxyl radical is a strongly reactive radical which can cause serious damage to biomolecules, such as lipids, proteins, and nucleic acids. Due to its extremely high reaction rate, reducing its generation, for instance, by blocking the Fenton reaction, appears to be a suitable antioxidant strategy for preventing its damage. ROS species production can be facilitated by elemental species, such as iron: ferrous ions induce metal-catalyzed oxidation and participate in hydroxyl radical-generating Fenton type reactions; ferric ions also produce radicals from peroxides, although the rate is 10-fold less than that of ferrous ion [
65]. Here, we assessed the ability of DR2B and DR2C extracts to indirectly affect ROS generation by reducing and/or chelating iron through the formation of ferrous/ferrozine complex [
66]. Under our experimental conditions, both samples exerted a weak ferric reducing activity, thus hindering evaluation of IC
50 (
Table 4). Conversely, they were effective as chelators of both ferrous and ferric ions, with almost 3-fold high potency of DR2C as chelators of ferric ions (
Figure 5A), in spite of a similar potency against ferrous ions (
Figure 5B). As estimated by the Pearson analysis, the chelating and radical scavenging activities of the extracts appeared to be significantly correlated among them (
Table 5 and
Table 6).
The antioxidant capacity of eggplant is ranked in the top ten of 120 different vegetables, although some differences can occur due to variety, fruit shape, and size and methodology [
25]. Jung et al. [
40] and Nisha et al. [
41] reported the DPPH radical scavenging and metal chelating properties of eggplant peel, while Kaneyuki et al. [
67] found strong scavenging effects against hydroxyl radical generation. Different extracts from fresh aubergine berry peel produced radical scavenging effects against DPPH and hydrogen peroxide, and metal chelation, which were mainly related to phenolics [
42]. Singh et al. [
49] also described the ability of skin eggplant to inhibit the cupric ion-mediated lipoperoxidation. Likewise, aubergine berries exhibited radical scavenging and chelating activities [
26,
68]. Mennella et al. [
33] also suggest the involvement of several bioactive constituents to the scavenging activity of NAS-type (containing nasunin as major anthocyanin) and D3R-type (containing delphinidin-3-rutinoside as major anthocyanin) eggplant varieties. Higher antioxidant capacities were also reported for semi-purified peel extracts from D3R-type and NAS-type aubergine berries respect to the purified D3R and NAS anthocyanins [
69], thus confirming that other phytochemicals besides anthocyanins are involved in the antioxidant activity of the phytocomplex. Accordingly, our results suggest that several components of the DR2B and DR2C phytocomplex can contribute to the described antioxidant activity, acting by direct (by neutralizing DPPH, ABTS, and hydroxyl radical) and/or indirect (as chelators of ferrous and ferric ions) mechanisms. Among phytochemicals, the antioxidant properties of chlorogenic acid and the esters formed between caffeic and quinic acids are widely reported [
70]. On the other hand, the radical scavenging and chelating effects of different polyphenols and anthocyanins have been published [
28,
29,
37,
71]. Particularly, delphinidin glucosides have shown to possess the strongest scavenging activity against superoxide anion and peroxynitrite, and NAS has been demonstrated to be a potent superoxide anion radical scavenger [
69]. However, along with polyphenols and anthocyanins, a possible role of other unidentified phytochemicals in the biological activity of DR2 eggplant peel cannot be excluded.
2.4. DR2C Extract Affects HSV-1 Replication
Several studies have reported that viral infections are often associated with an imbalance in the intracellular redox state of host cell, that shifts towards pro-oxidant conditions [
72,
73]. Redox alterations are useful for the virus, since many pathways involved in the regulation of viral replication and host responses are highly responsive to even transient changes in the redox state of the cytoplasmic environment [
74,
75]. For this reason, several antioxidant molecules, both natural and synthesized, have been reported to exert antiviral activity [
5,
13,
17,
76,
77,
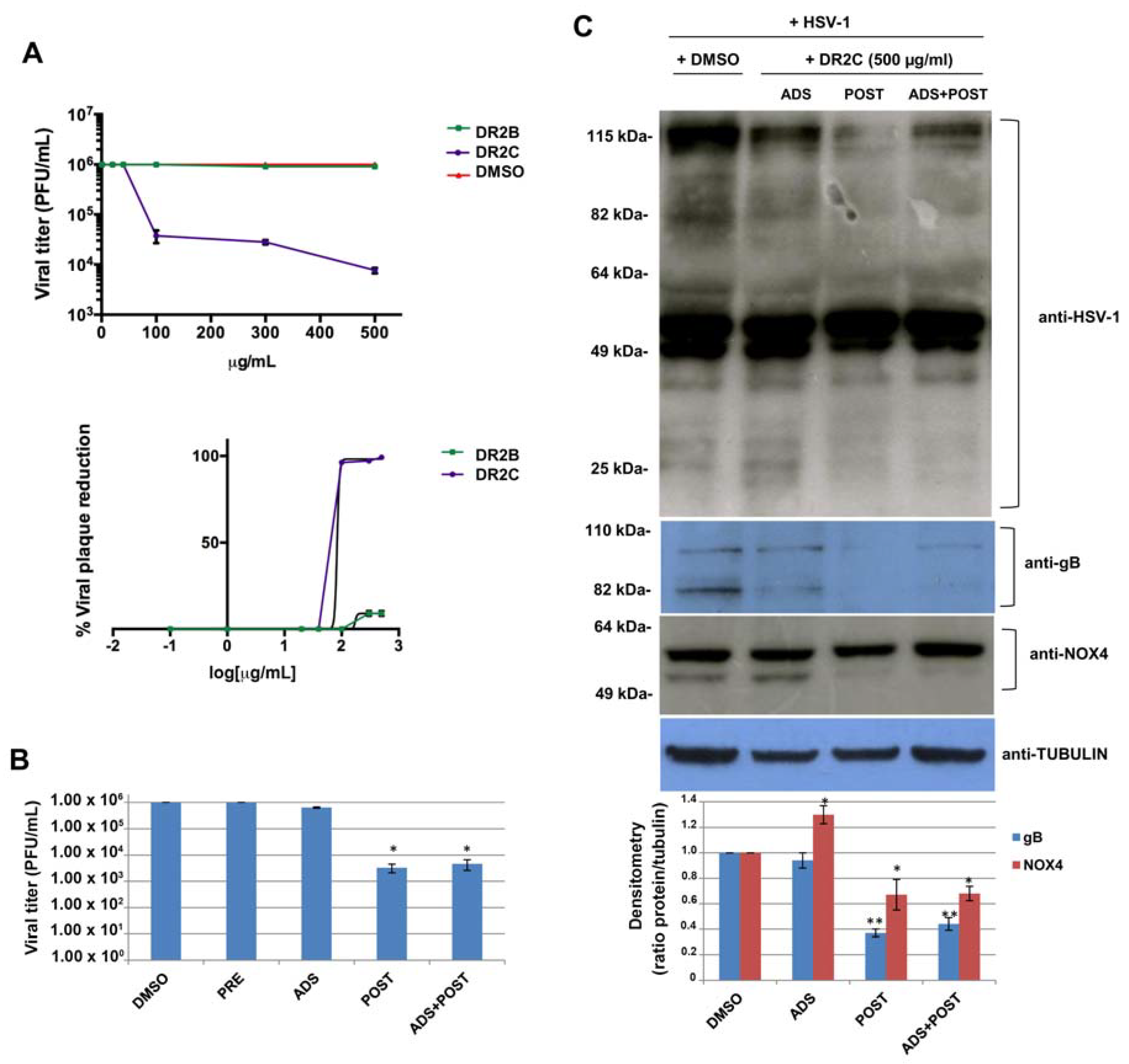
78]. Therefore, based on the above results relating to antioxidant properties of DR2B and DR2C extracts, their potential antiviral activity against HSV-1 was studied. To this aim, a plaque reduction assay was performed: confluent monolayers of VERO cells were infected with HSV-1 at multiplicity of infection (m.o.i.) of 1 for 24 h in the presence of increasing concentrations of both the extracts, ranging from 1 μg/mL to 500 μg/mL. DMSO-treated HSV-1-infected cells were used as control. Plaque formation was scored two days later. The dose–response curves, shown in
Figure 6A, demonstrate that DR2C inhibited HSV-1 plaque formation in a dose-dependent manner with respect to DMSO-treated cells. In particular, the higher inhibition of viral replication was reached with 500 μg/mL of the extract (about 2.5 log inhibition compared with untreated infected cells). On the contrary, HSV-1 replication was not affected by DR2B treatment. Next, to demonstrate that the antiviral effect of DR2C was not a consequence of cytotoxicity, VERO cells were incubated with different concentrations (ranging from 1 to 500 μg/mL) of DR2C, and cell viability was evaluated by the trypan blue dye exclusion assay. As control, VERO cells were treated with DMSO. After 24 h of incubation, no significant alteration of cellular morphology and viability was detected by light microscope observations up to the concentrations of 500 μg/mL. Cell count of DR2C treated cells was compared to DMSO-treated and untreated cells, and no significant differences were observed (data not shown). Although the 50% inhibitory concentration (IC
50) value for DR2C was 83.4 μg/mL (
Figure 6A), the following experiments were performed using the extract at 500 μg/mL, as the highest antiviral activity was achieved with that dose, which was not cytotoxic.
At first, time-of-addition assays were performed. Vero cells were pre-incubated (PRE) with the extract for 3 h at 37 °C before the viral challenge (i.e., before virus adsorption phase), in order to analyze its possible interference with some cellular receptors and co-receptors used by HSV-1 to bind the host cell. Next, DR2C was added to VERO cells during the HSV-1 adsorption phase (ADS) (i.e., during the very early phase of virus lifecycle). Finally, the extract was administrated to the cellular monolayer after the viral adsorption phase (POST) for 24 h (i.e., during the early and late phases of the virus lifecycle). Furthermore, DR2C was also tested as a double dose during, and after, the HSV-1 adsorption period (ADS+POST). DMSO alone was administered as control. In all the experimental conditions, 24 h (post infection) p.i., supernatants of infected cells were collected and used to determine viral titer by standard plaque assay. As shown in
Figure 6B, no difference in HSV-1 replication was observed in DR2C pre-treated cells compared to control (about 10
6 pfu/mL in both cases). Similarly, DR2C treatment during viral adsorption did not significantly impair HSV-1 replication (6.5 × 10
5 pfu/ml vs. 10
6 pfu/mL). On the contrary, DR2C maintained in cellular culture medium for 24 h after viral adsorption significantly inhibited viral replication, confirming the trend observed in the previous plaque reduction assays (3.3 × 10
3 pfu/mL vs. 10
6 pfu/mL, i.e., about 2.6-log inhibition). Interestingly, the same HSV-1 titer reduction was observed in samples treated with a double dose of DR2C, confirming that the extract was not effective during the virus challenge and showing that the extract affected only the post adsorption phase of the virus lifecycle, including the synthesis of viral DNA and proteins. Thus, to check whether DR2C treatment altered viral protein synthesis, we performed a Western blot by using an antibody directed against the main HSV-1 proteins (ICPs and late). As shown in
Figure 6C, DR2C administration to cells for 24 h after viral adsorption was able to reduce the expression of many viral proteins, particularly proteins with high and low molecular weight (over 80 kDa and lower 40 kDa, respectively). Furthermore, in the same experimental condition, the viral glycoprotein B (gB, late protein) expression was also strongly reduced, as confirmed by densitometric analysis (about 60% reduction compared to DMSO-treated infected cells) (
Figure 6C).
The results obtained in HSV-1-infected cells treated with DR2C are in agreement with previous studies showing that several stages of HSV-1 replication, favored by oxidative conditions, are affected by modulation of intracellular redox state [
17,
79]. To note that although DR2B was able to partially reduce ROS production induced by tBOOH, it did not restore the GSH levels (see
Figure 3B). Since reducing conditions in infected host cells are important for the control of viral replication, the inability of DR2B in restoring the imbalance in the redox state could, in part, explain its inefficacy against HSV-1 replication. On the contrary, according to the Pearson analysis, the anti-HSV1 activity of DR2C appears to be significantly correlated with both the radical scavenging and chelating activities (
Table 7), thus suggesting that the samples can counteract the redox imbalance induced by virus in host cell.
One of the main intracellular sources of ROS is represented by the NADPH (nicotinamide adenine dinucleotide phosphate) oxidase (NOX) family, constituted by seven enzymes generating superoxide anion (O
2−) or hydrogen peroxide (H
2O
2), from molecular oxygen [
80], that regulate specific cellular processes. However, they are also activated during acute or chronic viral infections, i.e., influenza virus and HCV, playing an important role in the regulation of immune response to infections (isoform NOX2) [
81,
82] as well as viral replication (isoform NOX4) [
75,
83,
84,
85]. Therefore, we evaluated the effect of the DR2C on the expression level of NOX4 during HSV-1 infection. To this aim, HSV-1-infected VERO cells, treated with DR2C at different time of infection, were analyzed by Western blotting to evaluate NOX-4 expression. As shown in
Figure 6C, the anti-NOX4 antibody recognized two NOX4 isoforms, and the one with a lower molecular weight was completely reduced by DR2C treatment after viral challenge (POST). It is known that NOX4 is highly expressed in the kidney [
86], and that in the human, there are many NOX4 isoforms produced as splice-variants [
87], some of which may have different functions in ROS-related cell signaling. Unfortunately, scientific literature does not report evidence about NOX4 isoforms in
Cercopithecus aethiops (from which Vero cells are derived); however, we would mention as evidence that 227 organisms have orthologs with human gene
NOX4 (as reported in gene bank HomoloGene:
41065). Further studies are needed to clarify this aspect.
Among the phytoconstituents of DR2C extracts, as highlighted at phytochemical analysis (
Table 2 and
Table 3), several phenylpropanoids (i.e., chlorogenic acid, caffeic acid, and vanillic acid) and flavonoids have been reported to possess antiviral properties [
88,
89,
90,
91]. Along with phenylpropanoids, D3R is one of the most represented compounds in the DR2C extract, as found by HPTLC densitometric analysis (
Table 2). This compound has been reported to possess several antioxidant properties, and to be able to protect osteoblastic cells against oxidative damage induced by
tert-butyl hydroperoxide [
29,
69]. Therefore, we tested whether the anti-HSV-1 activity of DR2C was mainly due to this compound. To this aim, D3R was added to VERO cells at concentration of 5 µg/mL (corresponding to 1% (
w/
w) of the first effective DR2C concentration against HSV-1) after HSV-1 adsorption for the following 24 h; cellular supernatant was collected to perform a standard plaque assay. The treatment with D3R did not inhibit HSV-1 replication (4 × 10
5 PFU/mL treated vs. 3.8 × 10
5 PFU/mL not-treated), suggesting that DR2C anti-HSV-1 activity depends on the whole phytochemical complex.
Altogether, our results indicate that DR2C is able to significantly impair HSV-1 propagation and viral protein expression in an in vitro model of VERO cells. To date, the mechanisms underlying the inhibition are not well defined, however, it is possible to speculate a control by DR2C of NOX4 activity and redox-regulated pathways during HSV-1 infection.
Importantly, DR2C treatment could also affect viral replication by raising the intracellular GSH levels in HSV-1-infected VERO cells. In fact, we demonstrated, in the cytoprotective assay above-described, that DR2C is able to restore the GSH intracellular content in tBOOH-treated Caco2 cells (
Figure 3B). It is known that different acute and chronic viral infections, including that of Herpes virus, induce a drop of GSH levels in infected cells, which favors viral propagation [
17,
18]. The administration of GSH or GSH derivative strongly inhibits virus replication by affecting specific steps of virus lifecycle and restores the GSH content in infected cells. However, further studies are in progress in our lab, aimed at clarifying this aspect. The modulation of redox state, through the re-establishment of reducing conditions in infected cells, may represent a good weapon to contrast viral infection with the advantage that the virus could have a lower probability to develop resistance.