An Ethnopharmacological, Phytochemical and Pharmacological Review on Lignans from Mexican Bursera spp.
Abstract
1. Introduction
2. Genus Bursera
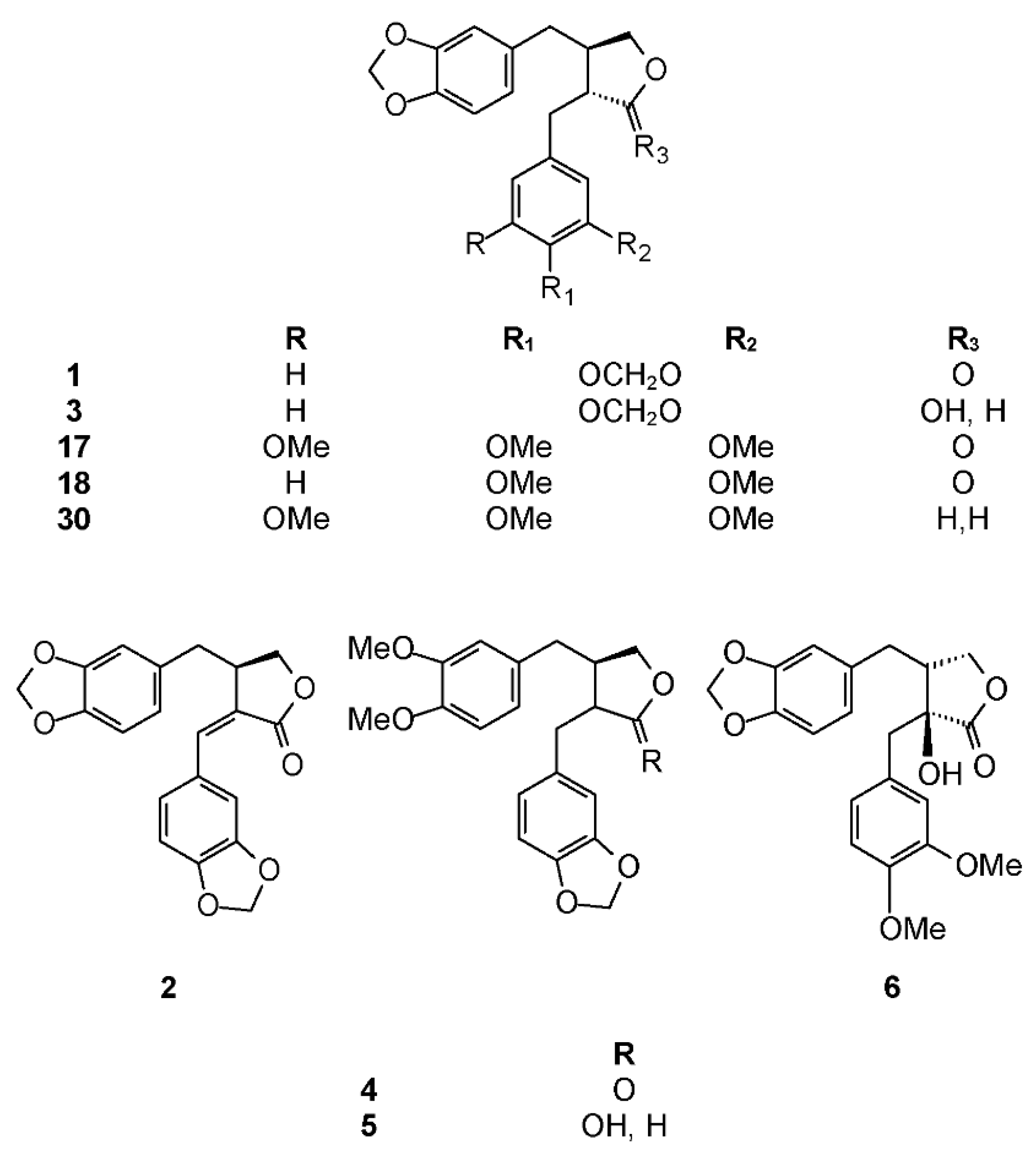
2.1. Traditional Uses, Phytochemistry, and Biological Activities
2.2. Subgenus Elaphrium
2.2.1. Bursera citronella McVaugh and Rzed.
2.2.2. Bursera cuneata (Schltdl.) Engl.
2.2.3. Bursera excelsa (Kunth) Engl.
2.2.4. Bursera graveolens (Kunth) Triana and Planch.
2.2.5. Bursera penicillata (Sessé and Moç. ex DC.) Engl.
2.2.6. Bursera submoniliformis Engl.
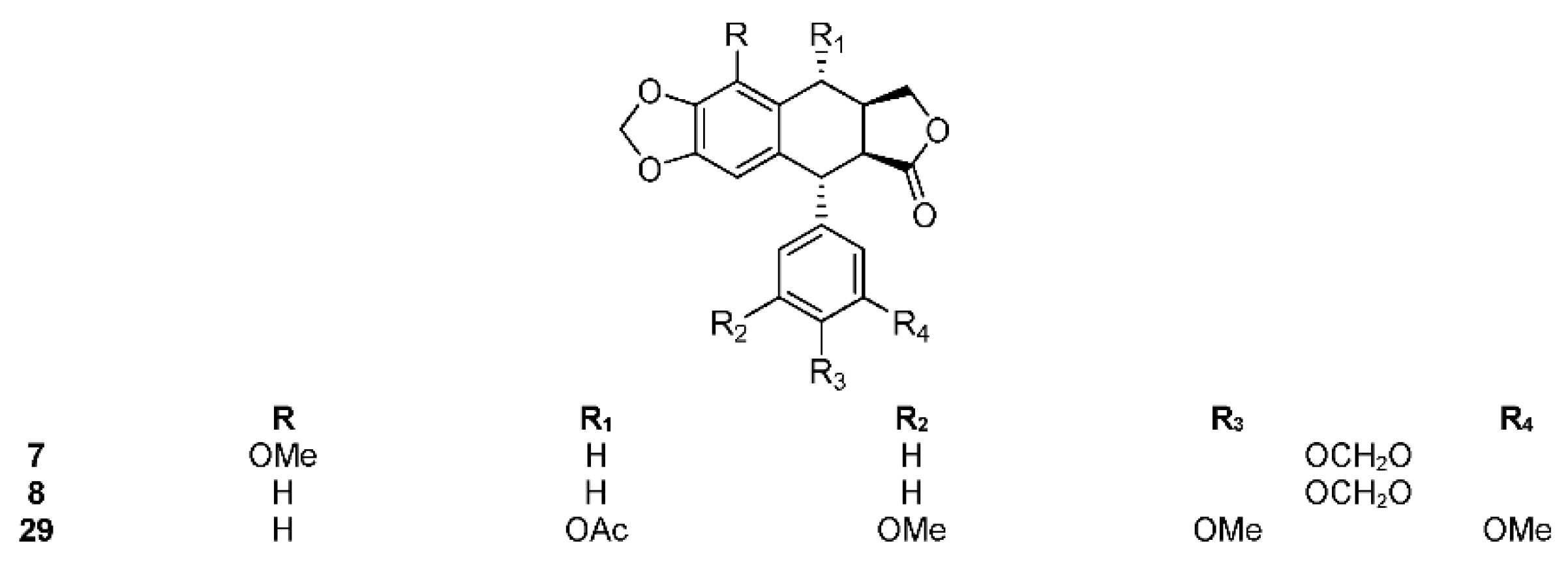
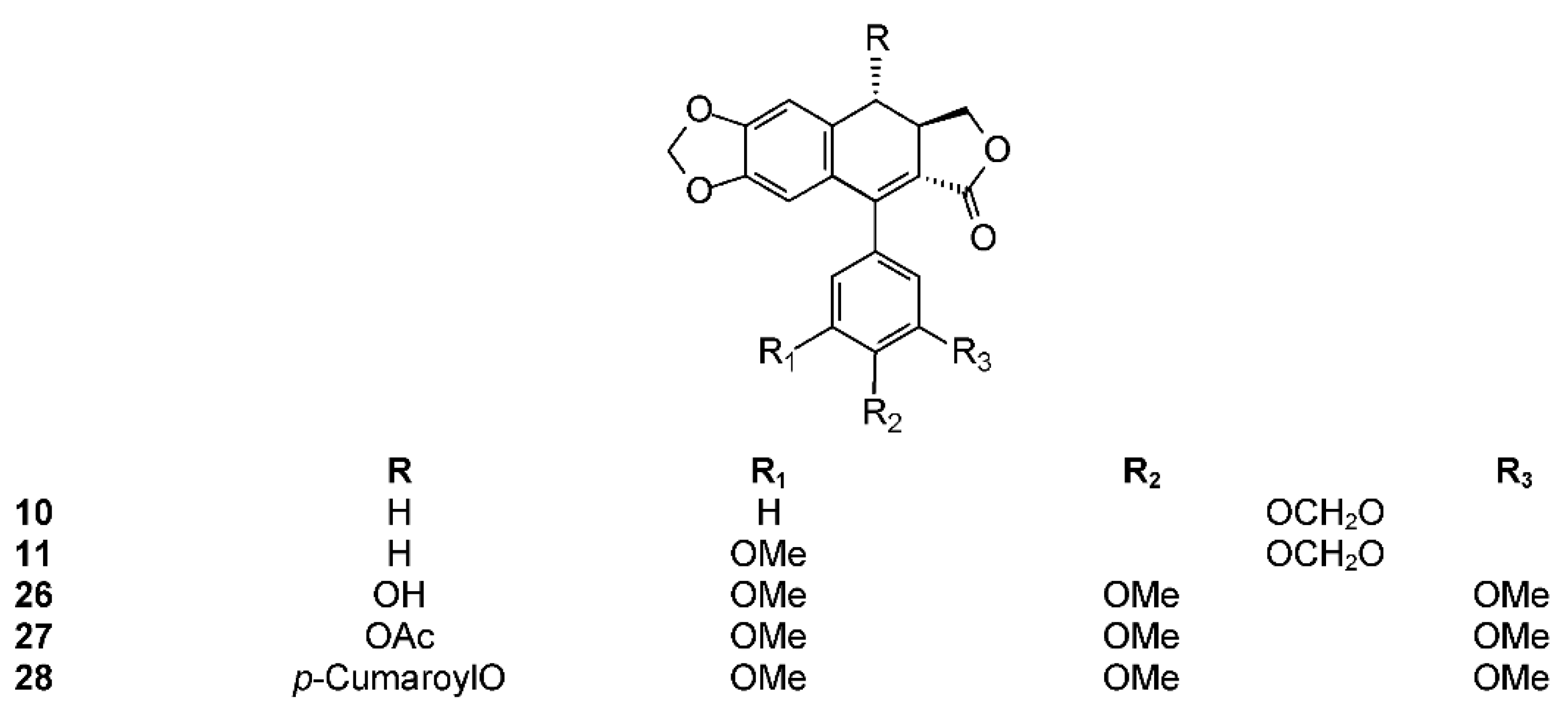
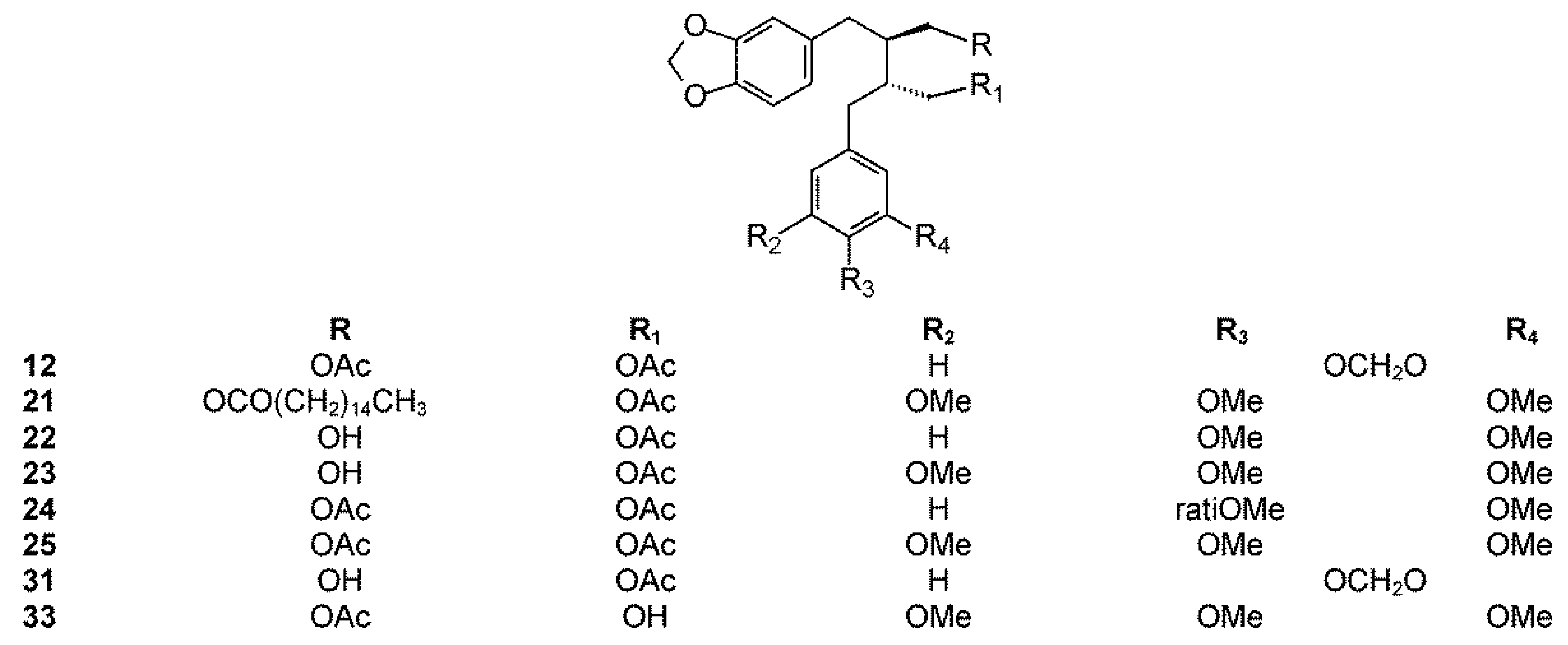
2.3. Section Bursera
2.3.1. Bursera aptera Ramirez
2.3.2. Bursera arida (Rose) Standl.
2.3.3. Bursera ariensis (Kunth) McVaugh and Rzed.
2.3.4. Bursera fagaroides (H.B.K.) Engl.
2.3.5. Bursera microphylla A. Gray
2.3.6. Bursera morelensis Ramírez
2.3.7. Bursera roseana Rzed., Calderón & Medina
2.3.8. Bursera schlechtendalii Engl.
2.3.9. Bursera simaruba (L.) Sarg.
3. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Becerra, J.X.; Noge, K.; Olivier, S.; Venable, D.L. The monophyly of Bursera and its impact for divergence times of Burseraceae. Taxon 2012, 61, 333–343. [Google Scholar]
- Rzedowski, J.; Medina Lemos, R.; Calderón de Rzedowski, G. Inventario del conocimiento taxonómico, así como de la diversidad y del endemismo regionales de las especies mexicanas de Bursera (Burseraceae). Acta Bot. Mex. 2005, 70, 75–111. [Google Scholar] [CrossRef]
- Becerra, J.X.; Venable, D.L.; Evans, P.H.; Bowers, W.S. Interactions between chemical and mechanical defenses in the plant genus Bursera and their implications for herbivores. Am. Zool. 2001, 41, 865–876. [Google Scholar]
- Gigliarelli, G.; Becerra, J.X.; Curini, M.; Marcotullio, M.C. Chemical composition and biological activities of fragrant Mexican copal (Bursera spp.). Molecules 2015, 20, 22383–22394. [Google Scholar] [CrossRef] [PubMed]
- Becerra, J.X. Evolution of Mexican Bursera (Burseraceae) inferred from ITS, ETS, and 5S nuclear ribosomal DNA sequences. Mol. Phylogenet. Evol. 2003, 26, 300–309. [Google Scholar] [CrossRef]
- Becerra, J.X.; Noge, K.; Venable, D.L. Macroevolutionary chemical escalation in an ancient plant-herbivore arms race. Proc. Natl. Acad. Sci. USA 2009, 106, 18062–18066. [Google Scholar] [CrossRef] [PubMed]
- Evans, P.H.; Becerra, J.X. Non-terpenoid essential oils from Bursera chemapodicta. Flavour Fragr. J. 2006, 21, 616–618. [Google Scholar] [CrossRef]
- Noge, K.; Becerra, J.X. Germacrene D, a Common Sesquiterpene in the Genus Bursera (Burseraceae). Molecules 2009, 14, 5289–5297. [Google Scholar] [CrossRef] [PubMed]
- Hernandez-Hernandez, J.D.; Garcia-Gutierrez, H.A.; Roman-Marin, L.U.; Torres-Blanco, Y.I.; Cerda-Garcia-Rojas, C.M.; Joseph-Nathan, P. Absolute configuration of cembrane diterpenoids from Bursera multijuga. Nat. Prod. Commun. 2014, 9, 1249–1252. [Google Scholar] [PubMed]
- Hernández-Hernández, J.D.; Román-Marín, L.U.; Cerda-García-Rojas, C.M.; Joseph-Nathan, P. Verticillane Derivatives from Bursera suntui and Bursera kerberi. J. Nat. Prod. 2005, 68, 1598–1602. [Google Scholar] [CrossRef] [PubMed]
- Messina, F.; Curini, M.; Di Sano, C.; Zadra, C.; Gigliarelli, G.; Rascon-Valenzuela, L.A.; Robles Zepeda, R.E.; Marcotullio, M.C. Diterpenoids and triterpenoids from the resin of Bursera microphylla and their cytotoxic activity. J. Nat. Prod. 2015, 78, 1184–1188. [Google Scholar] [CrossRef] [PubMed]
- Lucero-Gómez, P.; Mathe, C.; Vieillescazes, C.; Bucio, L.; Belio, I.; Vega, R. Analysis of Mexican reference standards for Bursera spp. resins by Gas Chromatography–Mass Spectrometry and application to archaeological objects. J. Archaeol. Sci. 2014, 41, 679–690. [Google Scholar] [CrossRef]
- Hernandez, J.D.; Garcia, L.; Hernandez, A.; Alvarez, R.; Roman, L.U. Luteolin and myricetin glycosides of Burseraceae. Rev. Soc. Quim. Mex. 2002, 46, 295–300. [Google Scholar]
- Nakanishi, T.; Inatomi, Y.; Arai, S.; Yamada, T.; Fukatsu, H.; Murata, H.; Inada, A.; Matsuura, N.; Ubukata, M.; Murata, J.; et al. New luteolin 3′-O-acylated rhamnosides from leaves of Bursera graveolens. Heterocycles 2003, 60, 2077–2083. [Google Scholar]
- Souza, M.P.; Machado, M.I.L.; Braz-Filho, R. Six flavonoids from Bursera leptophloeos. Phytochemistry 1989, 28, 2467–2470. [Google Scholar] [CrossRef]
- Cunha, W.R.; Andrade e Silva, M.L.; Sola Veneziani, R.C.; Ambrósio, S.R.; Kenupp Bastos, J. Lignans: Chemical and biological properties. In Phytochemicals-A Global Perspective of Their Role in Nutrition and Health; Rao, V., Ed.; InTech: Vienna, Austria, 2012. [Google Scholar] [CrossRef]
- The Plant List A Working List of All Plant Species. Available online: http://www.theplantlist.org/1.1/browse/A/Burseraceae/Bursera/ (accessed on 12 February 2015).
- Medina Lemos, R.; Ramos Rivera, P. El Género Bursera en México. Parte II; Version 1.3; Comisión Nacional Para el Conocimiento y uso de la Biodiversidad: Mexico City, Mexico, 2017. [Google Scholar]
- CONABIO Enciclovida CONABIO. Available online: http://www.enciclovida.mx/busquedas/resultados?utf8=%E2%9C%93&busqueda=basica&id=&nombre=bursera&button= (accessed on 29 July 2018).
- Bullock, L.L. Notes on the Mexican species of the genus Bursera. Bull. Misc. Inf. Kew 1936, 347–387. [Google Scholar]
- McVaugh, R.; Rzedowski, J. Synopsis of the genus Bursera L. in western Mexico, with notes on the material of Bursera collected by Sessè & Mocino. Kew Bull. 1965, 18, 317–382. [Google Scholar]
- Becerra, J.X.; Venable, D.L. Nuclear ribosomal DNA phylogeny and its implications for evolutionary trends in Mexican Bursera (Burseraceae). Am. J. Bot. 1999, 86, 1047–1057. [Google Scholar] [CrossRef] [PubMed]
- Rzedowki, J. Notas sobre el genero Bursera (Burseraceae) en el Estado de Guerrero, (Mexico). Am. Esc Nac Cienc. Biol. Mex. 1968, 7, 17–36. [Google Scholar]
- Toledo, C.A. El Genero Bursera en el Estado Guerrero; Universidad Nacional Autónoma de México: Mexico City, Mexico, 1982. [Google Scholar]
- Gillet, J. Commiphora (Burseraceae) in South America and its relationship to Bursera. Kew Bull. 1980, 34, 569–587. [Google Scholar] [CrossRef]
- Rzedowki, J.; Kruse, H. Algunas tendencias evolutivas en Bursera (Burseraceae). Taxon 1979, 28, 103–116. [Google Scholar] [CrossRef]
- Hernández, F. Historia de las Plantas de Nueva España; Imprenta Universitaria: Mexico City, México, 1942. [Google Scholar]
- Tripplett, K.J. The Ethnobotany of Plant Resins in the Maya Cultural Region of Southern Mexico and Central America; University of Texas: Austin, TX, USA, 1999. [Google Scholar]
- Acevedo, M.; Nuñez, P.; Gónzalez-Maya, L.; CardosoTaketa, A.; Villarreal, M.L. Cytotoxic and anti-inflammatory activities of Bursera species from Mexico. J. Clin. Toxicol. 2015, 5, 1–8. [Google Scholar]
- Alonso-Castro, A.J.; Villarreal, M.L.; Salazar-Olivo, L.A.; Gomez-Sanchez, M.; Dominguez, F.; Garcia-Carranca, A. Mexican medicinal plants used for cancer treatment: Pharmacological, phytochemical and ethnobotanical studies. J. Ethnopharmacol. 2011, 133, 945–972. [Google Scholar] [CrossRef] [PubMed]
- CONABIO Biodiversidad Mexicana. Available online: http://www.biodiversidad.gob.mx/usos/copales/almarciga.html (accessed on 8 May 2018).
- Waizel, J.; Waizel-Hayat, S. Antitussive plants used in Mexican traditional medicine. Pharmacogn. Rev. 2009, 3, 29–43. [Google Scholar]
- Koulman, A. Podophyllotoxin: A Study of the Biosynthesis, Evolution, Function and Use of Podophyllotoxin and Related Lignans; University of Groningen: Groningen, The Netherlands, 2003. [Google Scholar]
- Marcotullio, M.C.; Pelosi, A.; Curini, M. Hinokinin, an emerging bioactive lignan. Molecules 2014, 19, 14862–14878. [Google Scholar] [CrossRef] [PubMed]
- Ikeda, R.; Nagao, T.; Okabe, H.; Nakano, Y.; Matsunaga, H.; Katano, M.; Mori, M. Antiproliferative constituents in Umbelliferae plants. IV. Constituents in the fruits of Anthriscus sylvestris Hoffm. Chem. Pharm. Bull. 1998, 46, 875–878. [Google Scholar] [CrossRef] [PubMed]
- Chang, S.T.; Wang, D.S.Y.; Wu, C.L.; Shiah, S.G.; Kuo, Y.H.; Chang, C.J. Cytotoxicity of extractives from Taiwania cryptomerioides heartwood. Phytochemistry 2000, 55, 227–232. [Google Scholar] [CrossRef]
- Chen, J.-J.; Huang, H.-Y.; Duh, C.-Y.; Chen, I.-S. Cytotoxic constituents from the stem bark of Zanthoxylum pistaciiflorum. J. Chin. Chem. Soc. 2004, 51, 659–663. [Google Scholar] [CrossRef]
- Lin, R.-W.; Tsai, I.-L.; Duh, C.-Y.; Lee, K.-H.; Chen, I.-S. New lignans and cytotoxic constituents from Wikstroemia lanceolata. Planta Med. 2004, 70, 234–238. [Google Scholar] [PubMed]
- Chen, J.-J.; Chung, C.-Y.; Hwang, T.-L.; Chen, J.-F. Amides and benzenoids from Zanthoxylum ailanthoides with inhibitory activity on superoxide generation and elastase release by neutrophils. J. Nat. Prod. 2009, 72, 107–111. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.-Y.; Seo, K.-H.; Jeong, R.-H.; Lee, S.-M.; Kim, G.-S.; Noh, H.-J.; Kim, S.-Y.; Kim, G.-W.; Kim, J.-Y.; Baek, N.-I. Anti-inflammatory lignans from the fruits of Acanthopanax sessiliflorus. Molecules 2013, 18, 41–49. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, R.; de Souza, G.H.B.; da Silva, A.A.; de Souza, V.A.; Pereira, A.C.; Royo, V.D.A.; e Silva, M.L.A.; Donate, P.M.; de Matos Araujo, A.L.S.; Carvalho, J.C.T.; et al. Synthesis and biological activity evaluation of lignan lactones derived from (−)-cubebin. Bioorg. Med. Chem. Lett. 2005, 15, 1033–1037. [Google Scholar] [CrossRef] [PubMed]
- Lima, T.S.C.; Lucarini, R.; Volpe, A.C.; de Andrade, C.Q.J.; Souza, A.M.P.; Pauletti, P.M.; Januário, A.H.; Símaro, G.V.; Bastos, J.K.; Cunha, W.R.; et al. In vivo and in silico anti-inflammatory mechanism of action of the semisynthetic (−)-cubebin derivatives (−)-hinokinin and (−)-O-benzylcubebin. Bioorg. Med. Chem. Lett. 2017, 27, 176–179. [Google Scholar] [CrossRef] [PubMed]
- Ramos, F.; Takaishi, Y.; Kawazoe, K.; Osorio, C.; Duque, C.; Acuna, R.; Fujimoto, Y.; Sato, M.; Okamoto, M.; Oshikawa, T.; et al. Immunosuppressive diacetylenes, ceramides and cerebrosides from Hydrocotyle leucocephala. Phytochemistry 2006, 67, 1143–1150. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Yao, Z.; Zhang, Y.W.; Zhang, X.X.; Takaishi, Y.; Duan, H.Q. Immunosuppressive sesquiterpenes from Buddleja daviddi. Planta Med. 2010, 76, 1882–1887. [Google Scholar] [CrossRef] [PubMed]
- Silva, M.L.A.; Coimbra, H.S.; Pereira, A.C.; Almeida, V.A.; Lima, T.C.; Costa, E.S.; Vinholis, A.H.C.; Royo, V.A.; Silva, R.; Filho, A.A.S.; et al. Evaluation of Piper cubeba extract, (−)-cubebin and its semi-synthetic derivatives against oral pathogens. Phytoth. Res. 2007, 21, 420–422. [Google Scholar] [CrossRef] [PubMed]
- Huang, R.-L.; Huang, Y.-L.; Ou, J.-C.; Chen, C.-C.; Hsu, F.-L.; Chang, C. Screening of 25 compounds isolated from Phyllanthus species for anti-human Hepatitis B virus in vitro. Phytother. Res. 2003, 17, 449–453. [Google Scholar] [CrossRef] [PubMed]
- Zhang, G.; Shimokawa, S.; Mochizuki, M.; Kumamoto, T.; Nakanishi, W.; Watanabe, T.; Ishikawa, T.; Matsumoto, K.; Tashima, K.; Horie, S.; et al. Chemical constituents of Aristolochia constricta: Antispasmodic effects of its constituents in guinea-pig ileum and isolation of a diterpeno-lignan hybrid. J. Nat. Prod. 2008, 71, 1167–1172. [Google Scholar] [CrossRef] [PubMed]
- Kuroyanagi, M.; Ikeda, R.; Gao, H.Y.; Muto, N.; Otaki, K.; Sano, T.; Kawahara, N.; Nakane, T. Neurite outgrowth-promoting active constituents of the Japanese cypress (Chamaecyparis obtusa). Chem. Pharm. Bull. 2008, 56, 60–63. [Google Scholar] [CrossRef] [PubMed]
- Adfa, M.; Rahmad, R.; Ninomiya, M.; Yudha, S.; Tanaka, K.; Koketsu, M. Antileukemic activity of lignans and phenylpropanoids of Cinnamomum parthenoxylon. Bioorg. Med. Chem. Lett. 2016, 26, 761–764. [Google Scholar] [CrossRef] [PubMed]
- Cunha, N.L.; Teixeira, G.M.; Martins, T.S.D.; Souza, A.R.; Oliveira, P.F.; Símaro, G.V.N.; Rezende, K.C.S.; Gonçalves, N.L.D.S.; Souza, D.G.; Tavares, D.C.; et al. (−)-Hinokinin induces G2/M arrest and contributes to the antiproliferative effects of doxorubicin in breast cancer cells. Planta Med. 2016, 82, 530–538. [Google Scholar] [CrossRef] [PubMed]
- Yoon, J.S.; Koo, K.A.; Ma, C.J.; Sung, S.H.; Kim, Y.C. Neuroprotective lignans from Biota orientalis leaves. Nat. Prod. Sci. 2008, 14, 167–170. [Google Scholar]
- De Souza, V.A.; da Silva, R.; Pereira, A.C.; Royo Vde, A.; Saraiva, J.; Montanheiro, M.; de Souza, G.H.; da Silva Filho, A.A.; Grando, M.D.; Donate, P.M.; et al. Trypanocidal activity of (−)-cubebin derivatives against free amastigote forms of Trypanosoma cruzi. Bioorg. Med. Chem. Lett. 2005, 15, 303–307. [Google Scholar] [CrossRef] [PubMed]
- Esperandim, V.R.; da Silva Ferreira, D.; Saraiva, J.; Silva, M.L.A.; Costa, E.S.; Pereira, A.C.; Bastos, J.K.; de Albuquerque, S. Reduction of parasitism tissue by treatment of mice chronically infected with Trypanosoma cruzi with lignano lactones. Parasitol. Res. 2010, 107, 525–530. [Google Scholar] [CrossRef] [PubMed]
- Esperandim, V.R.; da Silva Ferreira, D.; Rezende, K.C.; Cunha, W.R.; Saraiva, J.; Bastos, J.K.; e Silva, M.L.; de Albuquerque, S. Evaluation of the in vivo therapeutic properties of (−)-cubebin and (−)-hinokinin against Trypanosoma cruzi. Experim. Parasitol. 2013, 133, 442–446. [Google Scholar] [CrossRef] [PubMed]
- Saraiva, J.; Vega, C.; Rolon, M.; da Silva, R.; Andrade, E.S.M.L.; Donate, P.M.; Bastos, J.K.; Gomez-Barrio, A.; de, A.S. In vitro and in vivo activity of lignan lactones derivatives against Trypanosoma cruzi. Parasitol. Res. 2007, 100, 791–795. [Google Scholar] [CrossRef] [PubMed]
- Saraiva, J.; Lira, A.A.M.; Esperandim, V.R.; da Silva Ferreira, D.; Ferraudo, A.S.; Bastos, J.K.; e Silva, M.L.A.; de Gaitani, C.M.; de Albuquerque, S.; Marchetti, J.M. (−)-Hinokinin-loaded poly(D,L-lactide-co-glycolide) microparticles for Chagas disease. Parasitol. Res. 2010, 106, 703–708. [Google Scholar] [CrossRef] [PubMed]
- Sartorelli, P.; Carvalho, C.S.; Reimao, J.Q.; Lorenzi, H.; Tempone, A.G. Antitrypanosomal activity of a diterpene and lignans isolated from Aristolochia cymbifera. Planta Med. 2010, 76, 1454–1456. [Google Scholar] [CrossRef] [PubMed]
- Haribabu, K.; Ajitha, M.; Mallavadhani, U.V. Quantitative estimation of (−)-hinokinin, a trypanosomicidal marker in Piper cubeba, and some of its commercial formulations using HPLC-PDA. J. Pharm. Anal. 2015, 5, 130–136. [Google Scholar] [CrossRef] [PubMed]
- Andrade e Silva, M.L.; Cicarelli, R.M.; Pauletti, P.M.; Luz, P.P.; Rezende, K.C.; Januario, A.H.; da Silva, R.; Pereira, A.C.; Bastos, J.K.; de Albuquerque, S.; et al. Trypanosoma cruzi: Evaluation of (−)-cubebin derivatives activity in the messenger RNAs processing. Parasitol. Res. 2011, 109, 445–451. [Google Scholar] [CrossRef] [PubMed]
- Resende, F.A.; Tomazella, I.M.; Barbosa, L.C.; Ponce, M.; Furtado, R.A.; Pereira, A.C.; Bastos, J.K.; Andrade, E.S.M.L.; Tavares, D.C. Effect of the dibenzylbutyrolactone lignan (−)-hinokinin on doxorubicin and methyl methanesulfonate clastogenicity in V79 Chinese hamster lung fibroblasts. Mutat. Res. 2010, 700, 62–66. [Google Scholar] [CrossRef] [PubMed]
- Resende, F.A.; Barbosa, L.C.; Tavares, D.C.; de Camargo, M.S.; de Souza Rezende, K.C.; ML, E.S.; Varanda, E.A. Mutagenicity and antimutagenicity of (−)-hinokinin a trypanosomicidal compound measured by Salmonella microsome and comet assays. BMC Complement. Altern. Med. 2012, 12, 203. [Google Scholar] [CrossRef] [PubMed]
- Masumura, M.; Okumura, F.S. Identity of hibalactone and savinin. J. Am. Chem. Soc. 1955, 77, 1906. [Google Scholar] [CrossRef]
- Hartwell, J.L.; Johnson, J.M.; Fitzgerald, D.B.; Belkin, M. Podophyllotoxin from Juniperus species; savinin. J. Am. Chem. Soc. 1953, 75, 235–236. [Google Scholar] [CrossRef]
- Woo, K.W.; Choi, S.U.; Park, J.C.; Lee, K.R. A new lignan glycoside from Juniperus rigida. Arch. Pharm. Res. 2011, 34, 2043–2049. [Google Scholar] [CrossRef] [PubMed]
- Badawi, M.M.; Seida, A.A.; Kinghorn, A.D.; Cordell, G.A.; Farnsworth, N.R. Potential anticancer agents. XVIII. Constituents of Amyris pinnata (Rutaceae). J. Nat. Prod. 1981, 44, 331–334. [Google Scholar] [CrossRef]
- Mansoor, T.A.; Borralho, P.M.; Luo, X.; Mulhovo, S.; Rodrigues, C.M.P.; Ferreira, M.-J.U. Apoptosis inducing activity of benzophenanthridine-type alkaloids and 2-arylbenzofuran neolignans in HCT116 colon carcinoma cells. Phytomedicine 2013, 20, 923–929. [Google Scholar] [CrossRef] [PubMed]
- Cho, J.Y.; Park, J.; Kim, P.S.; Yoo, E.S.; Baik, K.U.; Park, M.H. Savinin, a lignan from Pterocarpus santalinus inhibits tumor necrosis factor-α production and T cell proliferation. Biol. Pharm. Bull. 2001, 24, 167–171. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Ban, H.S.; Kim, Y.P.; Kim, B.-K.; Cho, S.H.; Ohuchi, K.; Shin, K.H. Lignans from Acanthopanax chiisanensis having an inhibitory activity on prostaglandin E2 production. Phytother. Res. 2005, 19, 103–106. [Google Scholar] [CrossRef] [PubMed]
- Mbaze, L.M.A.; Lado, J.A.; Wansi, J.D.; Shiao, T.C.; Chiozem, D.D.; Mesaik, M.A.; Choudhary, M.I.; Lacaille-Dubois, M.-A.; Wandji, J.; Roy, R.; et al. Oxidative burst inhibitory and cytotoxic amides and lignans from the stem bark of Fagara heitzii (Rutaceae). Phytochemistry 2009, 70, 1442–1447. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, T.L.S.; Leite, K.C.D.S.; de Macêdo, I.Y.L.; de Morais, S.R.; Costa, E.A.; de Paula, J.R.; de Souza Gil, E. Electrochemical behavior and antioxidant activity of hibalactone. Int. J. Electrochem. Sci. 2017, 12, 7956–7964. [Google Scholar] [CrossRef]
- Oliveira, T.L.S.; Morais, S.R.; Sá, S.; Oliveira, M.G.; Florentino, I.F.; Silva, D.M.D.; Carvalho, V.V.; Silva, V.B.D.; Vaz, B.G.; Sabino, J.R.; et al. Antinociceptive, anti-inflammatory and anxiolytic-like effects of the ethanolic extract, fractions and Hibalactone isolated from Hydrocotyle umbellata L. (Acaricoba)—Araliaceae. Biomed. Pharmacother. 2017, 95, 837–846. [Google Scholar] [CrossRef] [PubMed]
- Rzedowski, J.; Guevara-Féfer, F. Flora del Bajio y de Regiones Adyacentes; Instituto de Ecologia: Xalapa, Mexico, 1992; Volume 3. [Google Scholar]
- Chatterjee, A.; Basa, S.C.; Ray, A.B. Spectral properties of the lignin cubebin. J. Indian Chem. Soc. 1968, 45, 723–725. [Google Scholar]
- Bastos, J.K.; Albuquerque, S.; Silva, M.L.A. Evaluation of the trypanocidal activity of lignans isolated from the leaves of Zanthoxylum naranjillo. Planta Med. 1999, 65, 541–544. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, M.T.M.; Rezende, K.C.S.; Evora, P.R.B.; Bastos, J.K.; Cunha, W.R.; Andrade e Silva, M.L.; Celotto, A.C. The lignan (−)-Cubebin inhibits vascular contraction and induces relaxation via nitric oxide activation in isolated rat aorta. Phytother. Res. 2013, 27, 1784–1789. [Google Scholar] [CrossRef] [PubMed]
- Niwa, A.M.; Marcarini, J.C.; Sartori, D.; Maistro, E.L.; Mantovani, M.S. Effects of (−)-cubebin (Piper cubeba) on cytotoxicity, mutagenicity and expression of p38 MAP kinase and GSTa2 in a hepatoma cell line. J. Food Compos. Anal. 2013, 30, 1–5. [Google Scholar] [CrossRef]
- De Rezende, A.A.A.; Silva, M.L.A.; Tavares, D.C.; Cunha, W.R.; Rezende, K.C.S.; Bastos, J.K.; Lehmann, M.; de Andrade, H.H.R.; Guterres, Z.R.; Silva, L.P.; et al. The effect of the dibenzylbutyrolactolic lignan (−)-cubebin on doxorubicin mutagenicity and recombinogenicity in wing somatic cells of Drosophila melanogaster. Food Chem. Toxicol. 2011, 49, 1235–1241. [Google Scholar] [CrossRef] [PubMed]
- Da Rocha Pissurno, A.P.; De Laurentiz, R.D.S. Cubebin: A small molecule with great potential. Rev. Virtual Quim. 2017, 9, 656–671. [Google Scholar] [CrossRef]
- Cook, S. The Forest of the Lacandon Maya: An Ethnobotanical Guide; Springer: New York, NY, USA, 2016. [Google Scholar]
- Takaoka, D.; Imooka, M.; Hiroi, M. Studies of lignoids in Lauraceae. III. A new lignan from the heart wood of Cinnamomum camphora Sieb. Bull. Chem. Soc. Jpn. 1977, 50, 2821–2822. [Google Scholar] [CrossRef]
- Lopes, L.M.X.; Yoshida, M.; Gottlieb, O.R. The chemistry of Brazilian Myristicaceae. Part XIX. Dibenzylbutyrolactone lignans from Virola sebifera. Phytochemistry 1983, 22, 1516–1518. [Google Scholar] [CrossRef]
- Kato, M.J.; Yoshida, M.; Gottlieb, O.R. Lignoids and arylalkanones from fruits of Virola elongata. Phytochemistry 1990, 29, 1799–1810. [Google Scholar] [CrossRef]
- Gozler, T.; Gozler, B.; Patra, A.; Leet, J.E.; Freyer, A.J.; Shamma, M. Konyanin: A new lignan from Haplophyllum vulcanicum. Tetrahedron 1984, 40, 1145–1150. [Google Scholar] [CrossRef]
- Evcim, U.; Gozler, B.; Freyer, A.J.; Shamma, M. Haplomyrtin and (−)-haplomyrfolin: Two lignans from Haplophyllum myrtifolium. Phytochemistry 1986, 25, 1949–1951. [Google Scholar] [CrossRef]
- Sheriha, G.M.; Abouamer, K.; Elshtaiwi, B.Z.; Ashour, A.S.; Abed, F.A.; Alhallaq, H.H. Quinoline alkaloids and cytotoxic lignans from Haplophyllum tuberculatum. Phytochemistry 1987, 26, 3339–3341. [Google Scholar] [CrossRef]
- Rifai, Y. A new method for fast isolation of GLI inhibitory compounds. Int. J. Pharma Res. Rev. 2012, 1, 28–30. [Google Scholar]
- Ogungbe, I.V.; Erwin, W.R.; Setzer, W.N. Antileishmanial phytochemical phenolics: Molecular docking to potential protein targets. J. Mol. Gr. Modell. 2014, 48, 105–117. [Google Scholar] [CrossRef] [PubMed]
- Sriwiriyajan, S.; Sukpondma, Y.; Srisawat, T.; Madla, S.; Graidist, P. (−)-Kusunokinin and piperloguminine from Piper nigrum: An alternative option to treat breast cancer. Biomed. Pharmacother. 2017, 92, 732–743. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, A.G.; Estevez-Reyes, R.; Mato, C.; Estevez-Braun, A.M. Three lignans from Bupleurum salicifolium. Phytochemistry 1990, 29, 1981–1983. [Google Scholar] [CrossRef]
- Espinosa Organista, D.; Ramos Rivera, P.; Careaga Olvera, S.A. El Género Bursera en México. Parte II, 1.3 ed.; CONABIO: México City, México, 2017. [Google Scholar]
- Nakanishi, T.; Inatomi, Y.; Murata, H.; Shigeta, K.; Iida, N.; Inada, A.; Murata, J.; Farrera, M.A.P.; Iinuma, M.; Tanaka, T.; et al. A new and known cytotoxic aryltetralin-type lignans from stems of Bursera graveolens. Chem. Pharm. Bull. 2005, 53, 229–231. [Google Scholar] [CrossRef] [PubMed]
- Provan, G.J.; Waterman, P.G. Picropolygamain: A new lignan from Commiphora incisa resin. Planta Med. 1985, 51, 271–272. [Google Scholar] [CrossRef]
- Peraza-Sanchez, S.R.; Pena-Rodriguez, L.M. Isolation of picropolygamain from the resin of Bursera simaruba. J. Nat. Prod. 1992, 55, 1768–1771. [Google Scholar] [CrossRef] [PubMed]
- Mi, Q.; Pezzuto, J.M.; Farnsworth, N.R.; Wani, M.C.; Kinghorn, A.D.; Swanson, S.M. Use of the in Vivo Hollow Fiber Assay in Natural Products Anticancer Drug Discovery. J. Nat. Prod. 2009, 72, 573–580. [Google Scholar] [CrossRef] [PubMed]
- Felger, R.S.; Johnson, M.B.; Wilson, M.F. The Trees of Sonora, Mexico; Oxford University Press: New York, NY, USA, 2001. [Google Scholar]
- Gentry, H.S. Rio Mayo Plants; Carnegie Institution: Washington, DC, USA, 1942. [Google Scholar]
- Garcia Martinez, L.E. Aspectos Socio-Ecologicos Para el Manejo Sustenable del Coal en el Ejido de Acateyahualco, Gro; UNAM: Morelia, Mexico, 2012. [Google Scholar]
- Montúfar, A. Los Copales Mexicanos y la Resina Sagrada del Templo Mayor de Tenochtitlan; Instituto Nacional de Antropología e Historia: Mexico City, Mexico, 2007.
- Dávila Aranda, P.; Ramos Rivera, P. La Flora útil de dos Comunidades Indígenas del Valle de Tehuacán-Cuicatlán: Coxcatlán y Zapotitlán de las Salinas, Puebla; Version 1.3; Comisión Nacional Para el Conocimiento y uso de la Biodiversidad: Mexico City, Mexico, 2017.
- Nieto-Yañez, O.J.; Resendiz-Albor, A.A.; Ruiz-Hurtado, P.A.; Rivera-Yañez, N.; Rodriguez-Canales, M.; Rodriguez-Sosa, M.; Juarez-Avelar, I.; Rodriguez-Lopez, M.G.; Canales-Martinez, M.M.; Rodriguez-Monroy, M.A. In vivo and in vitro antileishmanial effects of methanolic extract from bark of Bursera aptera. Afr. J. Tradit. Complement. Altern. Med. 2017, 14, 188–197. [Google Scholar] [CrossRef] [PubMed]
- Rzedowski, J.; Medina Lemos, R.; Calderón de Rzedowski, G. Las especies de Bursera (Burseraceae) en la cuenca superior del rio Papaloapan (Mexico). Acta Bot. Mex. 2004, 66, 23–151. [Google Scholar] [CrossRef]
- Canales, M.; Hernández, T.; Caballero, J.; Vivar, A.R.D.; Avila, G.; Duran, A.; Lira, R. Informant consensus factor and antibacterial activity of the medicinal plants used by the people of San Rafael Coxcatlán, Puebla, México. J. Ethnopharmacol. 2005, 97, 429–439. [Google Scholar] [CrossRef] [PubMed]
- Gorgua Jiménez, G.; Nieto Yañez, O.; Ruiz Hurtado, P.A.; Rivera Yáñez, N.; Rodríguez Monroy, M.A.; Canales Martínez, M.M. Bursera arida. Evaluacion de su actividad antiparasitaria y caracterizacion fitoquimica del extracto metanolico. In 11 Reunion Internacional de Investigacion en Productos Naturales; Navarrete, A., Ed.; Revista Latinoamericana de Quimica: San Carlos, Mexico, 2015. [Google Scholar]
- Ionescu, F. Phytochemical Investigation of Bursera Arida Family Burseraceae; The University of Arizona: Tucson, Arizona, 1974. [Google Scholar]
- Hernández, J.D.; Román, L.U.; Espiñeira, J.; Joseph-Nathan, P. Ariensin, a New Lignan from Bursera ariensis. Planta Med. 1983, 47, 215–217. [Google Scholar] [CrossRef] [PubMed]
- LLIFLE Encyclopedias of Living Form. Available online: http://www.llifle.com/Encyclopedia/ (accessed on 5 August 2018).
- Avendaño Reyes, S.; Acosta Rosado, I. Plantas utilizadas como cercas vivas en el estado de Veracruz. Madera Bosques 2000, 6, 55–71. [Google Scholar] [CrossRef]
- Velazquez-Jimenez, R.; Torres-Valencia, J.M.; Cerda-Garcia-Rojas, C.M.; Hernandez-Hernandez, J.D.; Roman-Marin, L.U.; Manriquez-Torres, J.J.; Gomez-Hurtado, M.A.; Valdez-Calderon, A.; Motilva, V.; Garcia-Maurino, S.; et al. Absolute configuration of podophyllotoxin related lignans from Bursera fagaroides using vibrational circular dichroism. Phytochemistry 2011, 72, 2237–2243. [Google Scholar] [CrossRef] [PubMed]
- Jolad, S.D.; Wiedhopf, R.M.; Cole, J.R. Cytotoxic agents from Bursera morelensis (Burseraceae): Deoxypodophyllotoxin and a new lignan, 5′-desmethoxydeoxypodophyllotoxin. J. Pharm. Sci. 1977, 66, 892–893. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.J.; Ishikawa, T.; Duh, C.Y.; Tsai, I.L.; Chen, I.S. New dimeric aporphine alkaloids and cytotoxic constituents of Hernandia nymphaeifolia. Planta Med. 1996, 62, 528–533. [Google Scholar] [CrossRef] [PubMed]
- Novelo, M.; Cruz, J.G.; Hernández, L.; Pereda-Miranda, R.; Chai, H.; Mar, W.; Pezzuto, J.M. Cytotoxic Constituents from Hyptis verticillata. J. Nat. Prod. 1993, 56, 1728–1736. [Google Scholar] [CrossRef] [PubMed]
- Donoso-Fierro, C.S.; Tiezzi, A.; Ovidi, E.; Ceccarelli, D.; Triggiani, D.; Mastrogiovanni, F.; Taddei, A.R.; Pérez, C.; Becerra, J.; Silva, M.; Passarella, D. Antiproliferative activity of yatein isolated from Austrocedrus chilensis against murine myeloma cells: Cytological studies and chemical investigations. Pharm. Biol. 2015, 53, 378–385. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Young, K.; Rakotondraibe, L.H.; Brodie, P.J.; Wiley, J.D.; Cassera, M.B.; Callmander, M.W.; Rakotondrajaona, R.; Rakotobe, E.; Rasamison, V.E.; Tendyke, K.; et al. Antiproliferative compounds from Cleistanthus boivinianus from the Madagascar dry forest. J. Nat. Prod. 2015, 78, 1543–1547. [Google Scholar] [CrossRef] [PubMed]
- Mosmann, T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef]
- Wu, X.-D.; Wang, S.-Y.; Wang, L.; He, J.; Li, G.-T.; Ding, L.-F.; Gong, X.; Dong, L.-B.; Song, L.-D.; Li, Y.; et al. Labdane diterpenoids and lignans from Calocedrus macrolepis. Fitoterapia 2013, 85, 154–160. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.J.; Hung, H.C.; Sung, P.J.; Chen, I.S.; Kuo, W.L. Aporphine alkaloids and cytotoxic lignans from the roots of Illigera luzonensis. Phytochemistry 2011, 72, 523–532. [Google Scholar] [CrossRef] [PubMed]
- Doussot, J.; Mathieu, V.; Colas, C.; Molinié, R.; Corbin, C.; Montguillon, J.; Moreno, Y.; Banuls, L.; Renouard, S.; Lamblin, F.; et al. Investigation of the Lignan Content in Extracts from Linum, Callitris and Juniperus Species in Relation toTheir In Vitro Antiproliferative Activities. Planta Med. 2017, 83, 574–581. [Google Scholar] [PubMed]
- Kuo, Y.-C.; Kuo, Y.-H.; Lin, Y.-L.; Tsai, W.-J. Yatein from Chamaecyparis obtusa suppresses herpes simplex virus type 1 replication in HeLa cells by interruption the immediate-early gene expression. Antivir. Res. 2006, 70, 112–120. [Google Scholar] [CrossRef] [PubMed]
- Usia, T.; Watabe, T.; Kadota, S.; Tezuka, Y. Metabolite-cytochrome P450 complex formation by methylenedioxyphenyl lignans of Piper cubeba: Mechanism-based inhibition. Life Sci. 2005, 76, 2381–2391. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.J.; Chang, Y.L.; Teng, C.M.; Chen, I.S. Anti-platelet aggregation alkaloids and lignans from Hernandia nymphaeifolia. Planta Med. 2000, 66, 251–256. [Google Scholar] [CrossRef] [PubMed]
- Picking, D.; Chambers, B.; Barker, J.; Shah, I.; Porter, R.; Naughton, D.P.; Delgoda, R. Inhibition of cytochrome P450 activities by extracts of Hyptis verticillata Jacq.: Assessment for potential HERB-drug interactions. Molecules 2018, 23, 430. [Google Scholar] [CrossRef] [PubMed]
- Rojas-Sepúlveda, A.M.; Mendieta-Serrano, M.; Mojica, M.Y.A.; Salas-Vidal, E.; Marquina, S.; Villarreal, M.L.; Puebla, A.M.; Delgado, J.I.; Alvarez, L. Cytotoxic podophyllotoxin type-lignans from the steam bark of Bursera fagaroides var. fagaroides. Molecules 2012, 17, 9506. [Google Scholar] [CrossRef] [PubMed]
- Puebla-Pérez, A.M.; Huacuja-Ruiz, L.; Rodríguez-Orozco, G.; Villaseñor-García, M.M.; Miranda-Beltrán, M.D.L.L.; Celis, A.; Sandoval-Ramírez, L. Cytotoxic and antitumour activity from Bursera fagaroides ethanol extract in mice with L5178Y lymphoma. Phytother. Res. 1998, 12, 545–548. [Google Scholar] [CrossRef]
- Guerram, M.; Jiang, Z.-Z.; Zhang, L.-Y. Podophyllotoxin, a medicinal agent of plant origin: Past, present and future. Chin. J. Nat. Med. 2012, 10, 161–169. [Google Scholar] [CrossRef]
- Acton, Q.A. Advances in Podophyllotoxin Research and Application: 2012 Edition: ScholarlyBrief; Scholarly Editions: Atlanta, GA, USA, 2012. [Google Scholar]
- Kamal, A.; Ali Hussaini, S.M.; Malik, M.S. Recent Developments Towards Podophyllotoxin Congeners as Potential Apoptosis Inducers. Anti-Cancer Agents Med. Chem. 2015, 15, 565–574. [Google Scholar] [CrossRef]
- Lee, K.-H.; Xiao, Z. Podophyllotoxins and Analogs; CRC Press: Boca Raton, FL, USA, 2012; pp. 95–122. [Google Scholar]
- Ionkova, I. Podophyllotoxin and Related Lignans: Biotechnological Production by In Vitro Plant Cell Cultures; CAB International: Wallingford, Oxfordshire, UK, 2010; pp. 138–155. [Google Scholar]
- Khaled, M.; Jiang, Z.-Z.; Zhang, L.-Y. Deoxypodophyllotoxin: A promising therapeutic agent from herbal medicine. J. Ethnopharmacol. 2013, 149, 24–34. [Google Scholar] [CrossRef] [PubMed]
- Guerram, M.; Jiang, Z.-Z.; Sun, L.; Zhu, X.; Zhang, L.-Y. Antineoplastic effects of deoxypodophyllotoxin, a potent cytotoxic agent of plant origin, on glioblastoma U-87 MG and SF126 cells. Pharmacol. Rep. 2015, 67, 245–252. [Google Scholar] [CrossRef] [PubMed]
- Morales-Serna, J.A.; Cruz-Galicia, E.; Garcia-Rios, E.; Madrigal, D.; Gavino, R.; Cardenas, J.; Salmon, M. Three new diarylbutane lignans from the resin of Bursera fagaroides. Nat. Prod. Res. 2013, 27, 824–829. [Google Scholar] [CrossRef] [PubMed]
- Mojica, M.A.; Leon, A.; Rojas-Sepulveda, A.M.; Marquina, S.; Mendieta-Serrano, M.A.; Salas-Vidal, E.; Villarreal, M.L.; Alvarez, L. Aryldihydronaphthalene-type lignans from Bursera fagaroides var. fagaroides and their antimitotic mechanism of action. RSC Adv. 2016, 6, 4950–4959. [Google Scholar] [CrossRef]
- Gu, J.-Q.; Park, E.J.; Totura, S.; Riswan, S.; Fong, H.H.S.; Pezzuto, J.M.; Kinghorn, A.D. Constituents of the twigs of Hernandia ovigera that inhibit the transformation of JB6 murine epidermal cells. J. Nat. Prod. 2002, 65, 1065–1068. [Google Scholar] [CrossRef] [PubMed]
- Rosas-Arreguin, P.; Arteaga-Nieto, P.; Reynoso-Orozco, R.; Villagomez-Castro, J.C.; Sabanero-Lopez, M.; Puebla-Perez, A.M.; Calvo-Mendez, C. Bursera fagaroides, effect of an ethanolic extract on ornithine decarboxylase (ODC) activity in vitro and on the growth of Entamoeba histolytica. Exp. Parasitol. 2008, 119, 398–402. [Google Scholar] [CrossRef] [PubMed]
- Gutierrez-Gutierrez, F.; Puebla-Perez, A.M.; Gonzalez-Pozos, S.; Hernandez-Hernandez, J.M.; Perez-Rangel, A.; Alvarez, L.P.; Tapia-Pastrana, G.; Castillo-Romero, A. Antigiardial Activity of Podophyllotoxin-Type Lignans from Bursera fagaroides var. fagaroides. Molecules 2017, 22, 799. [Google Scholar] [CrossRef] [PubMed]
- Cole, J.R.; Bianchi, E.; Trumbull, E.R. Antitumor agents from Bursera microphylla (Burseraceae) II: Isolation of a new lignan—Burseran. J. Pharm. Sci. 1969, 58, 175–176. [Google Scholar] [CrossRef] [PubMed]
- Tomioka, K.; Ishiguro, T.; Mizuguchi, H.; Komeshima, N.; Koga, K.; Tsukagoshi, S.; Tsuruo, T.; Tashiro, T.; Tanida, S.; Kishi, T. Stereoselective reactions. XVII. Absolute structure-cytotoxic activity relationships of steganacin congeners and analogs. J. Med. Chem. 1991, 34, 54–57. [Google Scholar] [CrossRef] [PubMed]
- Tomioka, K.; Koga, K. Stereoselective total synthesis of optically active trans- and cis-burseran. Determination of the stereochemistry of natural antitumor burseran. Heterocycles 1979, 12, 1523–1528. [Google Scholar] [CrossRef]
- Gigliarelli, G.; Zadra, C.; Cossignani, L.; Robles Zepeda, R.E.; Rascon-Valenzuela, L.A.; Velazquez-Contreras, C.A.; Marcotullio, M.C. Two new lignans from the resin of Bursera microphylla A. gray and their cytotoxic activity. Nat. Prod. Res. 2017, 18, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Tian, X.; Fan, P.C.; Zhan, Y.J.; Shen, D.W.; Jin, Y. Separation, determination and identification of the diastereoisomers of podophyllotoxin and its esters by high-performance liquid chromatography/tandem mass spectrometry. J. Chromatogr. A 2008, 1210, 168–177. [Google Scholar] [CrossRef] [PubMed]
- Alina, C.-M.C.; Rocio, R.-L.; Aurelio, R.-M.M.; Margarita, C.-M.M.; Angelica, R.-G.; Ruben, J.-A. Chemical composition and in vivo anti-inflammatory activity of Bursera morelensis Ramirez essential oil. J. Essent. Oil-Bear. Plants 2014, 17, 758–768. [Google Scholar] [CrossRef]
- Rzedowski, J.; Medina Lemos, R.; Calderón de Rzedowski, G. Segunda restauración de Bursera ovalifolia y nombre nuevo para otro componente del complejo de B. simaruba (Burseraceae). Acta Bot. Mex. 2007, 81, 45–70. [Google Scholar] [CrossRef]
- Buckingham, J. Dictionary of Natural Products; Taylor & Francis: Abingdon, UK, 1993. [Google Scholar]
- Chang, C.-C.; Lien, Y.-C.; Liu, K.C.S.C.; Lee, S.-S. Lignans from Phyllanthus urinaria. Phytochemistry 2003, 63, 825–833. [Google Scholar] [CrossRef]
- Ito, C.; Itoigawa, M.; Ogata, M.; Mou, X.Y.; Tokuda, H.; Nishino, H.; Furukawa, H. Lignans as anti-tumor-promoter from the seeds of Hernandia ovigera. Planta Med. 2001, 67, 166–168. [Google Scholar] [CrossRef] [PubMed]
- Murakami, A.; Ohigashi, H.; Koshimizu, K. Anti-tumor promotion with food phytochemicals: A strategy for cancer chemoprevention. Biosci. Biotechnol. Biochem. 1996, 60, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Pettit, G.R.; Meng, Y.; Gearing, R.P.; Herald, D.L.; Pettit, R.K.; Doubek, D.L.; Chapuis, J.-C.; Tackett, L.P. Antineoplastic Agents. 522. Hernandia peltata (Malaysia) and Hernandia nymphaeifolia (Republic of Maldives). J. Nat. Prod. 2004, 67, 214–220. [Google Scholar] [CrossRef] [PubMed]
- Noguera, B.; Diaz, E.; Garcia, M.V.; Feliciano, A.S.; Lopez-Perez, J.L.; Israel, A. Anti-inflammatory activity of leaf extract and fractions of Bursera simaruba (L.) Sarg (Burseraceae). J. Ethnopharmacol. 2004, 92, 129–133. [Google Scholar] [CrossRef] [PubMed]
- Maldini, M.; Montoro, P.; Piacente, S.; Pizza, C. Phenolic compounds from Bursera simaruba Sarg. bark: Phytochemical investigation and quantitative analysis by tandem mass spectrometry. Phytochemistry 2009, 70, 641–649. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are not available from the authors. |

© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Marcotullio, M.C.; Curini, M.; Becerra, J.X. An Ethnopharmacological, Phytochemical and Pharmacological Review on Lignans from Mexican Bursera spp. Molecules 2018, 23, 1976. https://doi.org/10.3390/molecules23081976
Marcotullio MC, Curini M, Becerra JX. An Ethnopharmacological, Phytochemical and Pharmacological Review on Lignans from Mexican Bursera spp. Molecules. 2018; 23(8):1976. https://doi.org/10.3390/molecules23081976
Chicago/Turabian StyleMarcotullio, Maria Carla, Massimo Curini, and Judith X. Becerra. 2018. "An Ethnopharmacological, Phytochemical and Pharmacological Review on Lignans from Mexican Bursera spp." Molecules 23, no. 8: 1976. https://doi.org/10.3390/molecules23081976
APA StyleMarcotullio, M. C., Curini, M., & Becerra, J. X. (2018). An Ethnopharmacological, Phytochemical and Pharmacological Review on Lignans from Mexican Bursera spp. Molecules, 23(8), 1976. https://doi.org/10.3390/molecules23081976





