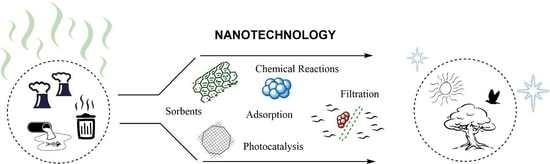
Nanotechnology for Environmental Remediation: Materials and Applications
Abstract
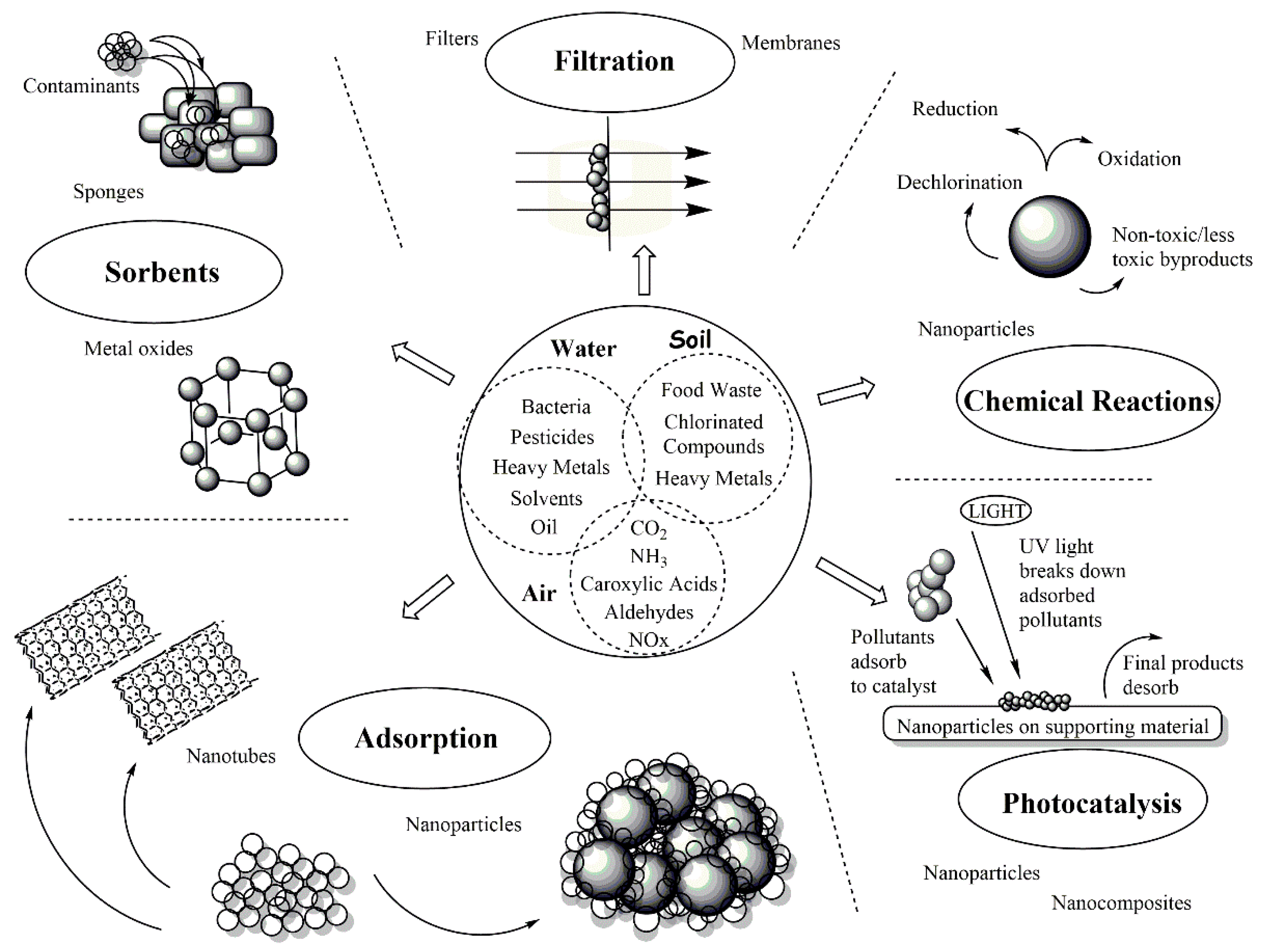
1. Introduction
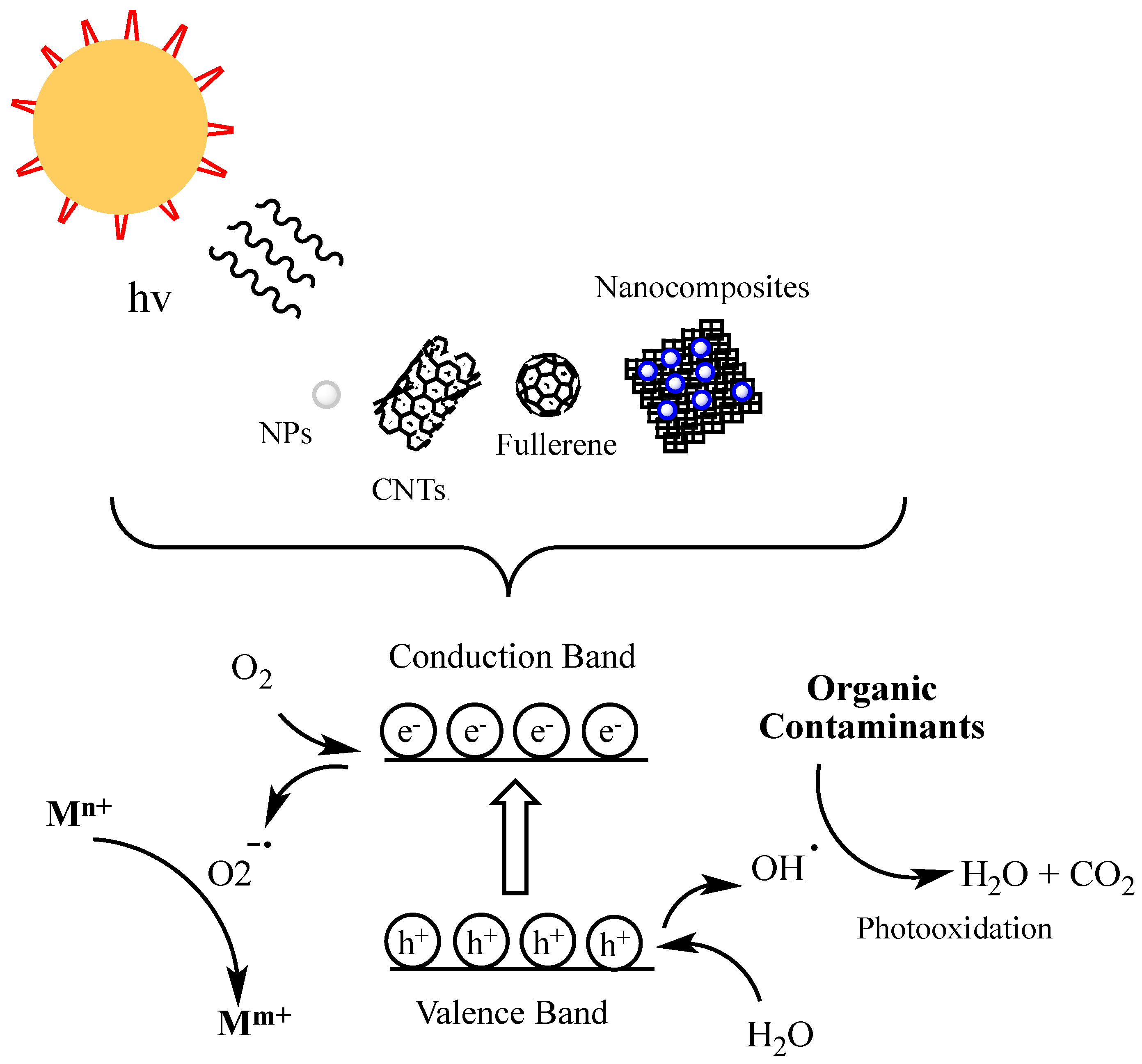
2. Inorganic Nanomaterials
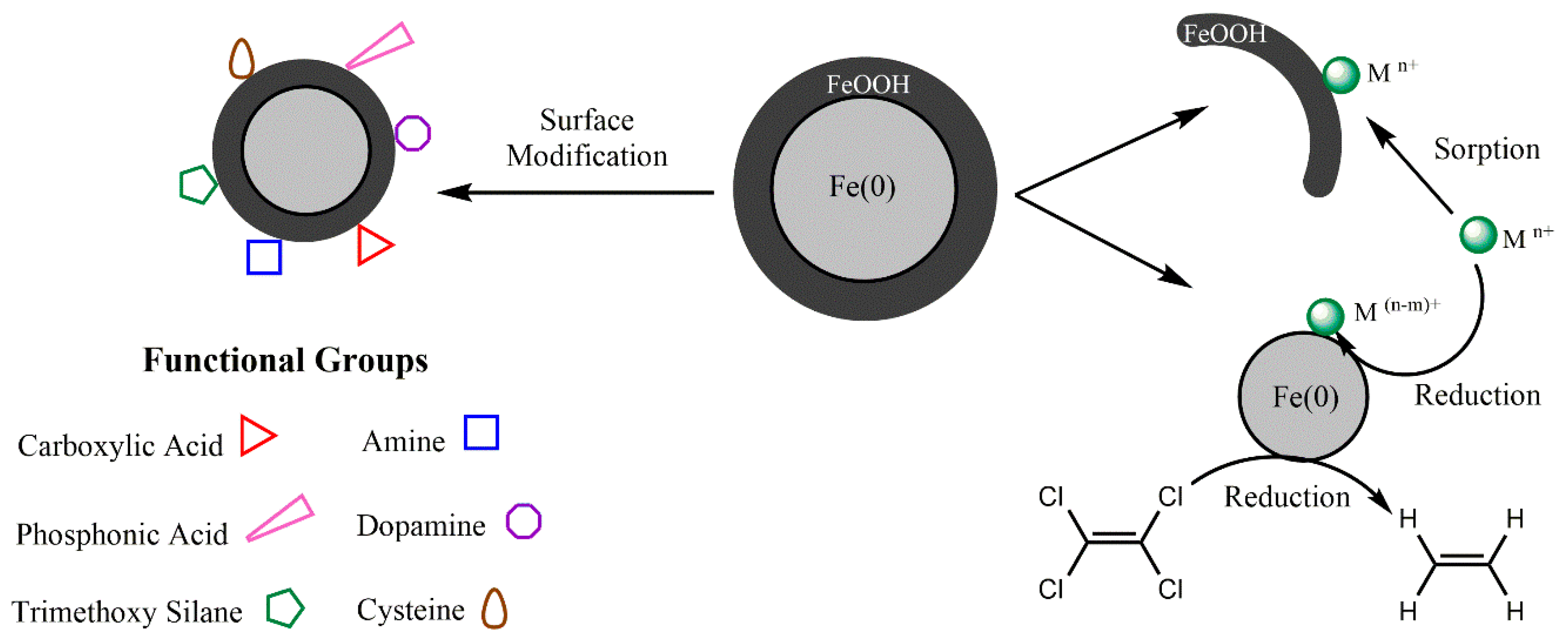
2.1. Metal- and Metal Oxide-Based Nanomaterials
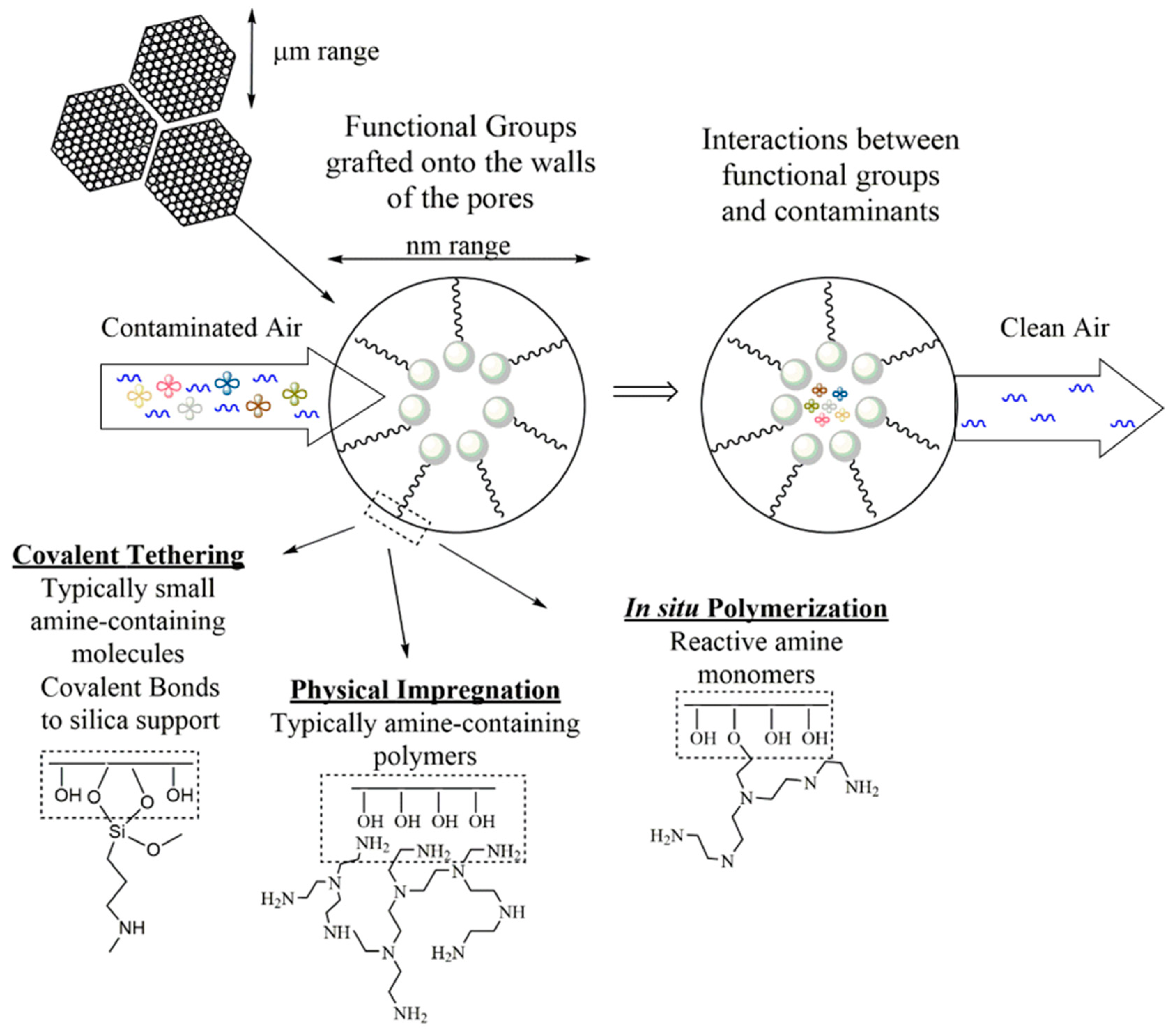
2.2. Silica Nanomaterials
3. Carbon-Based Nanomaterials
3.1. Graphene Materials
3.2. Carbon Nanotubes (CNTs)
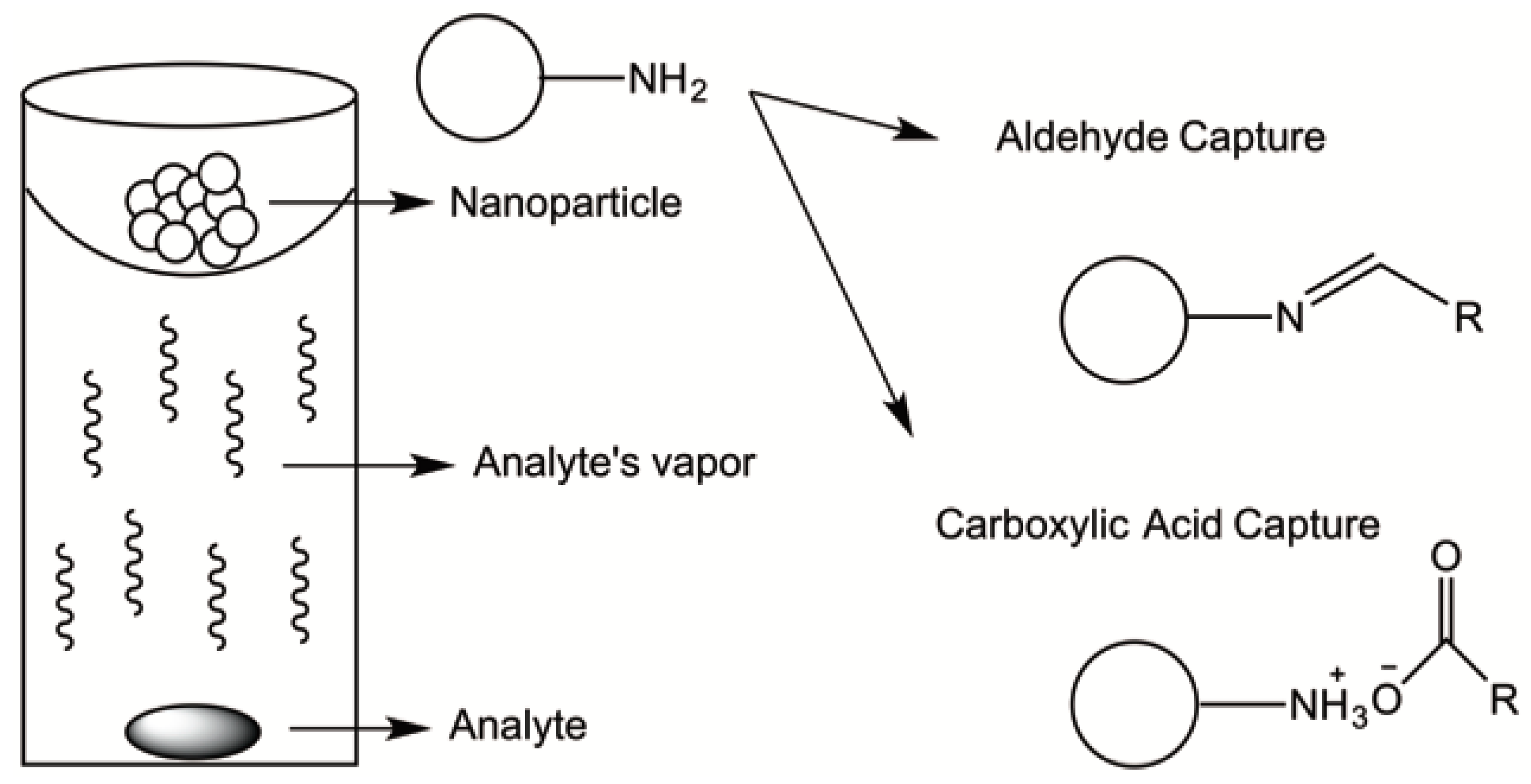
4. Polymer-Based Nanomaterials
5. Conclusions
Funding
Acknowledgments
Conflicts of Interest
References
- Masciangoli, T.; Zhang, W. Environmental Technologies. Environ. Sci. Technol. 2003, 37, 102–108. [Google Scholar]
- Vaseashta, A.; Vaclavikova, M.; Vaseashta, S.; Gallios, G.; Roy, P.; Pummakarnchana, O. Nanostructures in environmental pollution detection, monitoring, and remediation. Sci. Technol. Adv. Mater. 2007, 8, 47–59. [Google Scholar] [CrossRef]
- Khan, F.I.; Ghoshal, A.K. Removal of Volatile Organic Compounds from polluted air. J. Loss Prev. Process Ind. 2000, 13, 527–545. [Google Scholar] [CrossRef]
- Tratnyek, P.G.; Johnson, R.L. Nanotechnologies for environmental cleanup. Nano Today 2006, 1, 44–48. [Google Scholar] [CrossRef]
- Guerra, F.D.; Smith, G.D.; Alexis, F.; Whitehead, D.C. A Survey of VOC Emissions from Rendering Plants. Aerosol Air Qual. Res. 2017, 17, 209–217. [Google Scholar] [CrossRef]
- Guerra, F.D.; Campbell, M.L.; Whitehead, D.C.; Alexis, F. Tunable Properties of Functional Nanoparticles for Efficient capture of VOCs. ChemistrySelect 2017, 2, 9889–9894. [Google Scholar] [CrossRef]
- Campbell, M.L.; Guerra, F.D.; Dhulekar, J.; Alexis, F.; Whitehead, D.C. Target-Specific Capture of Environmentally Relevant Gaseous Aldehydes and Carboxylic Acids with Functional Nanoparticles. Chem. A Eur. J. 2015, 21, 14834–14842. [Google Scholar] [CrossRef] [PubMed]
- Shah, K.J.; Imae, T. Selective gas capture ability of gas-adsorbent-incorporated cellulose nanofiber films. Biomacromolecules 2016, 17, 1653–1661. [Google Scholar] [CrossRef] [PubMed]
- Ojea-Jiménez, I.; López, X.; Arbiol, J.; Puntes, V. Citrate-coated gold nanoparticles as smart scavengers for mercury(II) removal from polluted waters. ACS Nano 2012, 6, 2253–2260. [Google Scholar] [CrossRef] [PubMed]
- Kamat, P.V.; Meisel, D. Nanoscience opportunities in environmental remediation. C. R. Chim. 2003, 6, 999–1007. [Google Scholar] [CrossRef]
- Pandey, B.; Fulekar, M.H. Nanotechnology: Remediation Technologies to Clean Up the Environmental Pollutants. Res. J. Chem. Sci. 2012, 2, 90–96. [Google Scholar]
- Tong, H.; Ouyang, S.; Bi, Y.; Umezawa, N.; Oshikiri, M.; Ye, J. Nano-photocatalytic materials: Possibilities and challenges. Adv. Mater. 2012, 24, 229–251. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Lv, L.; Pan, B.; Zhang, W.; Zhang, S.; Zhang, Q. Polymer-supported nanocomposites for environmental application: A review. Chem. Eng. J. 2011, 170, 381–394. [Google Scholar] [CrossRef]
- Mauter, M.; Elimelech, M. Environmental applications of carbon-based nanomaterials. Environ. Sci. Technol. 2008, 42, 5843–5859. [Google Scholar] [CrossRef] [PubMed]
- Sharma, Y.C.; Srivastava, V.; Singh, V.K.; Kaul, S.N.; Weng, C.H. Nano-adsorbents for the removal of metallic pollutants from water and wastewater. Environ. Technol. 2009, 30, 583–609. [Google Scholar] [CrossRef] [PubMed]
- Gui, X.; Wei, J.; Wang, K.; Cao, A.; Zhu, H.; Jia, Y.; Shu, Q.; Wu, D. Carbon nanotube sponges. Adv. Mater. 2010, 22, 617–621. [Google Scholar] [CrossRef] [PubMed]
- Ng, L.Y.; Mohammad, A.W.; Leo, C.P.; Hilal, N. Polymeric membranes incorporated with metal/metal oxide nanoparticles: A comprehensive review. Desalination 2013, 308, 15–33. [Google Scholar] [CrossRef]
- Santhosh, C.; Velmurugan, V.; Jacob, G.; Jeong, S.K.; Grace, A.N.; Bhatnagar, A. Role of nanomaterials in water treatment applications: A review. Chem. Eng. J. 2016, 306, 1116–1137. [Google Scholar] [CrossRef]
- Das, S.; Sen, B.; Debnath, N. Recent trends in nanomaterials applications in environmental monitoring and remediation. Environ. Sci. Pollut. Res. 2015, 22, 18333–18344. [Google Scholar] [CrossRef] [PubMed]
- Chou, K.-S.; Lu, Y.-C.; Lee, H.-H. Effect of alkaline ion on the mechanism and kinetics of chemical reduction of silver. Mater. Chem. Phys. 2005, 94, 429–433. [Google Scholar] [CrossRef]
- Gupta, A.; Silver, S. Molecular genetics: Silver as a biocide: Will resistance become a problem? Nat. Biotechnol. 1998, 16, 888. [Google Scholar] [CrossRef] [PubMed]
- Bosetti, M.; Masse, A.; Tobin, E.; Cannas, M. Silver coated materials for external fixation devices: In vitro biocompatibility and genotoxicity. Biomaterials 2002, 23, 887–892. [Google Scholar] [CrossRef]
- Xiu, Z.; Zhang, Q.; Puppala, H.L.; Colvin, V.L.; Alvarez, P.J.J. Negligible particle-specific antibacterial activity of silver nanoparticles. Nano Lett. 2012, 12, 4271–4275. [Google Scholar] [CrossRef] [PubMed]
- Pal, S.; Tak, Y.K.; Song, J.M. Does the antibacterial activity of silver nanoparticles depend on the shape of the nanoparticle? A study of the gram-negative bacterium Escherichia coli. Appl. Environ. Microbiol. 2007, 73, 1712–1720. [Google Scholar] [CrossRef] [PubMed]
- Cho, M.; Chung, H.; Choi, W.; Yoon, J. Different inactivation behaviors of MS-2 phage and Escherichia coli in TiO2 photocatalytic disinfection. Appl. Environ. Microbiol. 2005, 71, 270–275. [Google Scholar] [CrossRef] [PubMed]
- Alizadeh Fard, M.; Aminzadeh, B.; Vahidi, H. Degradation of petroleum aromatic hydrocarbons using TiO2 nanopowder film. Environ. Technol. 2013, 34, 1183–1190. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, M.B.; Abrantes, N.; Nogueira, V.; Gonçalves, F.; Pereira, R. TiO2 nanoparticles for the remediation of eutrophic shallow freshwater systems: Efficiency and impacts on aquatic biota under a microcosm experiment. Aquat. Toxicol. 2016, 178, 58–71. [Google Scholar] [CrossRef] [PubMed]
- Gu, J.; Dong, D.; Kong, L.; Zheng, Y.; Li, X. Photocatalytic degradation of phenanthrene on soil surfaces in the presence of nanometer anatase TiO2 under UV-light. J. Environ. Sci. 2012, 24, 2122–2126. [Google Scholar] [CrossRef]
- Li, D.; Cui, F.; Zhao, Z.; Liu, D.; Xu, Y.; Li, H.; Yang, X. The impact of titanium dioxide nanoparticles on biological nitrogen removal from wastewater and bacterial community shifts in activated sludge. Biodegradation 2014, 25, 167–177. [Google Scholar] [CrossRef] [PubMed]
- Park, J.-Y.; Lee, I.-H. Photocatalytic degradation of 2-chlorophenol using Ag-doped TiO2 nanofibers and a near-UV light-emitting diode system. J. Nanomater. 2014, 2014, 250803. [Google Scholar] [CrossRef]
- Bao, Q.; Zhang, D.; Qi, P. Synthesis and characterization of silver nanoparticle and graphene oxide nanosheet composites as a bactericidal agent for water disinfection. J. Colloid Interface Sci. 2011, 360, 463–470. [Google Scholar] [CrossRef] [PubMed]
- Sreeja, S.; Vidya Shetty, K. Microbial disinfection of water with endotoxin degradation by photocatalysis using Ag@TiO2 core shell nanoparticles. Environ. Sci. Pollut. Res. 2016, 23, 18154–18164. [Google Scholar] [CrossRef]
- Xiong, Z.; Ma, J.; Ng, W.J.; Waite, T.D.; Zhao, X.S. Silver-modified mesoporous TiO2 photocatalyst for water purification. Water Res. 2011, 45, 2095–2103. [Google Scholar] [CrossRef] [PubMed]
- Younas, H.; Qazi, I.A.; Hashmi, I.; Ali Awan, M.; Mahmood, A.; Qayyum, H.A. Visible light photocatalytic water disinfection and its kinetics using Ag-doped titania nanoparticles. Environ. Sci. Pollut. Res. 2014, 21, 740–752. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Cen, C.; Tang, Z.; Zeng, W.; Chen, D.; Fang, P.; Chen, Z. The key role of pH value in the synthesis of titanate nanotubes-loaded manganese oxides as a superior catalyst for the selective catalytic reduction of NO with NH3. J. Nanomater. 2013, 2013, 871528. [Google Scholar] [CrossRef]
- Rasalingam, S.; Peng, R.; Koodali, R.T. Removal of hazardous pollutants from wastewaters: Applications of TiO2-SiO2 mixed oxide materials. J. Nanomater. 2014, 2014, 617405. [Google Scholar] [CrossRef]
- Hooshyar, Z.; Rezanejade Bardajee, G.; Ghayeb, Y. Sonication enhanced removal of nickel and cobalt ions from polluted water using an iron based sorbent. J. Chem. 2013, 2013, 786954. [Google Scholar] [CrossRef]
- Ebrahim, S.E.; Sulaymon, A.H.; Saad Alhares, H. Competitive removal of Cu2+, Cd2+, Zn2+, and Ni2+ ions onto iron oxide nanoparticles from wastewater. Desalin. Water Treat. 2016, 57, 20915–20929. [Google Scholar] [CrossRef]
- Poguberović, S.S.; Krčmar, D.M.; Maletić, S.P.; Kónya, Z.; Pilipović, D.D.T.; Kerkez, D.V.; Rončević, S.D. Removal of As(III) and Cr(VI) from aqueous solutions using “green” zero-valent iron nanoparticles produced by oak, mulberry and cherry leaf extracts. Ecol. Eng. 2016, 90, 42–49. [Google Scholar] [CrossRef]
- Guo, M.; Weng, X.; Wang, T.; Chen, Z. Biosynthesized iron-based nanoparticles used as a heterogeneous catalyst for the removal of 2,4-dichlorophenol. Sep. Purif. Technol. 2017, 175, 222–228. [Google Scholar] [CrossRef]
- Han, Y.; Yan, W. Reductive Dechlorination of Trichloroethene by Zero-valent Iron Nanoparticles: Reactivity Enhancement through Sulfidation Treatment. Environ. Sci. Technol. 2016, 50, 12992–13001. [Google Scholar] [CrossRef] [PubMed]
- Li, X.Q.; Zhang, W.X. Iron nanoparticles: The core–shell structure and unique properties for Ni(II) sequestration. Langmuir 2006, 22, 4638–4642. [Google Scholar] [CrossRef] [PubMed]
- Mystrioti, C.; Xanthopoulou, T.D.; Tsakiridis, P.; Papassiopi, N.; Xenidis, A. Comparative evaluation of five plant extracts and juices for nanoiron synthesis and application for hexavalent chromium reduction. Sci. Total Environ. 2016, 539, 105–113. [Google Scholar] [CrossRef] [PubMed]
- Lien, H.-L.; Zhang, W. Hydrodechlorination of Chlorinated Ethanes by Nanoscale Pd/Fe Bimetallic Particles. J. Environ. Eng. 2005, 131, 4–10. [Google Scholar] [CrossRef]
- Chen, C.; Wang, X.; Chang, Y.; Liu, H. Dechlorination of disinfection by-product monochloroacetic acid in drinking water by nanoscale palladized iron bimetallic particle. J. Environ. Sci. 2008, 20, 945–951. [Google Scholar] [CrossRef]
- Nagpal, V.; Bokare, A.D.; Chikate, R.C.; Rode, C.V.; Paknikar, K.M. Reductive dechlorination of γ-hexachlorocyclohexane using Fe-Pd bimetallic nanoparticles. J. Hazard. Mater. 2010, 175, 680–687. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Shen, Q.; Cissoko, N.; Wo, J.; Xu, X. Catalytic dechlorination of 2,4-dichlorophenol by Pd/Fe bimetallic nanoparticles in the presence of humic acid. J. Hazard. Mater. 2010, 182, 252–258. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Chen, C.; Chang, Y.; Liu, H. Dechlorination of chlorinated methanes by Pd/Fe bimetallic nanoparticles. J. Hazard. Mater. 2009, 161, 815–823. [Google Scholar] [CrossRef] [PubMed]
- Xie, Y.; Fang, Z.; Cheng, W.; Tsang, P.E.; Zhao, D. Remediation of polybrominated diphenyl ethers in soil using Ni/Fe bimetallic nanoparticles: Influencing factors, kinetics and mechanism. Sci. Total Environ. 2014, 485–486, 363–370. [Google Scholar] [CrossRef] [PubMed]
- Tee, Y.H.; Grulke, E.; Bhattacharyya, D. Role of Ni/Fe nanoparticle composition on the degradation of trichloroethylene from water. Ind. Eng. Chem. Res. 2005, 44, 7062–7070. [Google Scholar] [CrossRef]
- Fang, Z.; Qiu, X.; Chen, J.; Qiu, X. Debromination of polybrominated diphenyl ethers by Ni/Fe bimetallic nanoparticles: Influencing factors, kinetics, and mechanism. J. Hazard. Mater. 2011, 185, 958–969. [Google Scholar] [CrossRef] [PubMed]
- Zhu, N.; Luan, H.; Yuan, S.; Chen, J.; Wu, X.; Wang, L. Effective dechlorination of HCB by nanoscale Cu/Fe particles. J. Hazard. Mater. 2010, 176, 1101–1105. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Z.; Yuan, S.; Liu, Y.; Lu, X.; Wan, J.; Wu, X.; Chen, J. Reductive dechlorination of hexachlorobenzene by Cu/Fe bimetal in the presence of nonionic surfactant. J. Hazard. Mater. 2009, 170, 895–901. [Google Scholar] [CrossRef] [PubMed]
- Willis, A.L.; Turro, N.J.; O’Brien, S. Spectroscopic characterization of the surface of iron oxide nanocrystals. Chem. Mater. 2005, 17, 5970–5975. [Google Scholar] [CrossRef]
- Cushing, B.L.; Kolesnichenko, V.L.; O’Connor, C.J. Recent advances in the liquid-phase syntheses of inorganic nanoparticles. Chem. Rev. 2004, 104, 3893–3946. [Google Scholar] [CrossRef] [PubMed]
- Gogoi, S.K.; Gopinath, P.; Paul, A.; Ramesh, A.; Ghosh, S.S.; Chattopadhyay, A. Green Fluorescent Protein-Expressing Escherichia coli as a Model System for Investigating the Antimicrobial Activities of Silver Nanoparticles. Langmuir 2006, 22, 9322–9328. [Google Scholar] [CrossRef] [PubMed]
- Adesina, A.A. Industrial exploitation of photocatalysis: Progress, perspectives and prospects. Catal. Surv. Asia 2004, 8, 265–273. [Google Scholar] [CrossRef]
- Li, Q.; Mahendra, S.; Lyon, D.Y.; Brunet, L.; Liga, M.V.; Li, D.; Alvarez, P.J.J. Antimicrobial nanomaterials for water disinfection and microbial control: Potential applications and implications. Water Res. 2008, 42, 4591–4602. [Google Scholar] [CrossRef] [PubMed]
- Zan, L.; Fa, W.; Peng, T.; Gong, Z. Photocatalysis effect of nanometer TiO2 and TiO2-coated ceramic plate on Hepatitis B virus. J. Photochem. Photobiol. B Biol. 2007, 86, 165–169. [Google Scholar] [CrossRef] [PubMed]
- Kharisov, B.I.; Rasika Dias, H.V.; Kharissova, O.V.; Manuel Jiménez-Pérez, V.; Olvera Pérez, B.; Muñoz Flores, B. Iron-containing nanomaterials: Synthesis, properties, and environmental applications. RSC Adv. 2012, 2, 9325–9358. [Google Scholar] [CrossRef]
- Khin, M.M.; Nair, A.S.; Babu, V.J.; Murugan, R.; Ramakrishna, S. A review on nanomaterials for environmental remediation. Energy Environ. Sci. 2012, 5, 8075–8109. [Google Scholar] [CrossRef]
- Tao, F. Synthesis, catalysis, surface chemistry and structure of bimetallic nanocatalysts. Chem. Soc. Rev. 2012, 41, 7977–7979. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Ritchie, S.M.C. Removal of trichloroethylene from water by cellulose acetate supported bimetallic Ni/Fe nanoparticles. Chemosphere 2006, 63, 285–292. [Google Scholar] [CrossRef] [PubMed]
- Karn, B.; Kuiken, T.; Otto, M. Nanotechnology and in situ remediation: A review of the benefits and potential risks. Environ. Health Perspect. 2009, 117, 1813–1831. [Google Scholar] [CrossRef] [PubMed]
- Machado, S.; Pinto, S.L.; Grosso, J.P.; Nouws, H.P.A.; Albergaria, J.T.; Delerue-Matos, C. Green production of zero-valent iron nanoparticles using tree leaf extracts. Sci. Total Environ. 2013, 445–446, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Ding, S.; Zhao, L.; Qi, Y.; Lv, Q. Preparation and characterization of lecithin-nano Ni/Fe for effective removal of PCB77. J. Nanomater. 2014, 2014, 678489. [Google Scholar] [CrossRef]
- Su, C.; Puls, R.W.; Krug, T.A.; Watling, M.T.; O’Hara, S.K.; Quinn, J.W.; Ruiz, N.E. A two and half-year-performance evaluation of a field test on treatment of source zone tetrachloroethene and its chlorinated daughter products using emulsified zero valent iron nanoparticles. Water Res. 2012, 46, 5071–5084. [Google Scholar] [CrossRef] [PubMed]
- Mackenzie, K.; Bleyl, S.; Kopinke, F.D.; Doose, H.; Bruns, J. Carbo-Iron as improvement of the nanoiron technology: From laboratory design to the field test. Sci. Total Environ. 2016, 563–564, 641–648. [Google Scholar] [CrossRef] [PubMed]
- O’Hara, S.; Krug, T.; Quinn, J.; Clausen, C.; Geiger, C. Field and laboratory evaluation of the treatment of DNAPL source zones using emulsified zero-valent iron. Remediation 2006, 16, 35–56. [Google Scholar] [CrossRef]
- Elliott, D.W.; Zhang, W.X. Field assessment of nanoscale bimetallic particles for groundwater treatment. Environ. Sci. Technol. 2001, 35, 4922–4926. [Google Scholar] [CrossRef]
- Quinn, J.; Geiger, C.; Clausen, C.; Brooks, K.; Coon, C.; O’Hara, S.; Krug, T.; Major, D.; Yoon, W.S.; Gavaskar, A.; et al. Field demonstration of DNAPL dehalogenation using emulsified zero-valent iron. Environ. Sci. Technol. 2005, 39, 1309–1318. [Google Scholar] [CrossRef] [PubMed]
- Bennett, P.; He, F.; Zhao, D.; Aiken, B.; Feldman, L. In situ testing of metallic iron nanoparticle mobility and reactivity in a shallow granular aquifer. J. Contam. Hydrol. 2010, 116, 34–46. [Google Scholar] [CrossRef] [PubMed]
- Wei, Y.T.; Wu, S.C.; Chou, C.M.; Che, C.H.; Tsai, S.M.; Lien, H.L. Influence of nanoscale zero-valent iron on geochemical properties of groundwater and vinyl chloride degradation: A field case study. Water Res. 2010, 44, 131–140. [Google Scholar] [CrossRef] [PubMed]
- Kocur, C.M.D.; Lomheim, L.; Molenda, O.; Weber, K.P.; Austrins, L.M.; Sleep, B.E.; Boparai, H.K.; Edwards, E.A.; O’Carroll, D.M. Long-Term Field Study of Microbial Community and Dechlorinating Activity Following Carboxymethyl Cellulose-Stabilized Nanoscale Zero-Valent Iron Injection. Environ. Sci. Technol. 2016, 50, 7658–7670. [Google Scholar] [CrossRef] [PubMed]
- Chowdhury, A.I.A.; Krol, M.M.; Kocur, C.M.; Boparai, H.K.; Weber, K.P.; Sleep, B.E.; O’Carroll, D.M. NZVI injection into variably saturated soils: Field and modeling study. J. Contam. Hydrol. 2015, 183, 16–28. [Google Scholar] [CrossRef] [PubMed]
- Fagerlund, F.; Illangasekare, T.H.; Phenrat, T.; Kim, H.J.; Lowry, G.V. PCE dissolution and simultaneous dechlorination by nanoscale zero-valent iron particles in a DNAPL source zone. J. Contam. Hydrol. 2012, 131, 9–28. [Google Scholar] [CrossRef] [PubMed]
- Jiang, D.; Zeng, G.; Huang, D.; Chen, M.; Zhang, C.; Huang, C.; Wan, J. Remediation of contaminated soils by enhanced nanoscale zero valent iron. Environ. Res. 2018, 163, 217–227. [Google Scholar] [CrossRef] [PubMed]
- Tsai, C.H.; Chang, W.C.; Saikia, D.; Wu, C.E.; Kao, H.M. Functionalization of cubic mesoporous silica SBA-16 with carboxylic acid via one-pot synthesis route for effective removal of cationic dyes. J. Hazard. Mater. 2016, 309, 236–248. [Google Scholar] [CrossRef] [PubMed]
- Son, W.-J.; Choi, J.-S.; Ahn, W.-S. Adsorptive removal of carbon dioxide using polyethyleneimine-loaded mesoporous silica materials. Microporous Mesoporous Mater. 2008, 113, 31–40. [Google Scholar] [CrossRef]
- Leal, O.; Bolívar, C.; Ovalles, C.; García, J.J.; Espidel, Y. Reversible adsorption of carbon dioxide on amine surface-bonded silica gel. Inorg. Chim. Acta 1995, 240, 183–189. [Google Scholar] [CrossRef]
- Huang, H.Y.; Yang, R.T.; Chinn, D.; Munson, C.L. Amine-Grafted MCM-48 and Silica Xerogel as Superior Sorbents for Acidic Gas Removal from Natural Gas. Ind. Eng. Chem. Res. 2003, 42, 2427–2433. [Google Scholar] [CrossRef]
- Xu, X.; Song, C.; Andresen, J.M.; Miller, B.G.; Scaroni, A.W. Preparation and characterization of novel CO2 “molecular basket” adsorbents based on polymer-modified mesoporous molecular sieve MCM-41. Microporous Mesoporous Mater. 2003, 62, 29–45. [Google Scholar] [CrossRef]
- Knofel, C.; Descarpentries, J.; Benzaouia, A.; Zelenak, V.; Mornet, S.; Llewellyn, P.L.; Hornebecq, V. Functionalised micro-/mesoporous silica for the adsorption of carbon dioxide. Microporous Mesoporous Mater. 2007, 99, 79–85. [Google Scholar] [CrossRef]
- Satyapal, S.; Filburn, T.; Trela, J.; Strange, J. Performance and Properties of a Solid Amine Sorbent for Carbon Dioxide Removal in Space Life Support Applications. Energy Fuels 2001, 15, 250–255. [Google Scholar] [CrossRef]
- Mandal, B.P.; Bandyopadhyay, S.S. Simultaneous absorption of carbon dioxide and hydrogen sulfide into aqueous blends of 2-amino-2-methyl-1-propanol and diethanolamine. Chem. Eng. Sci. 2005, 60, 6438–6451. [Google Scholar] [CrossRef]
- Franchi, R.S.; Harlick, P.J.E.; Sayari, A. Applications of pore-expanded mesoporous silica. 2. Development of a high-capacity, water-tolerant adsorbent for CO2. Ind. Eng. Chem. Res. 2005, 44, 8007–8013. [Google Scholar] [CrossRef]
- Harlick, P.J.E.; Sayari, A. Applications of pore-expanded mesoporous silicas. 3. Triamine silane grafting for enhanced CO2 adsorption. Ind. Eng. Chem. Res. 2006, 45, 3248–3255. [Google Scholar] [CrossRef]
- Kim, S.; Ida, J.; Guliants, V.V.; Lin, J.Y.S. Tailoring pore properties of MCM-48 silica for selective adsorption of CO2. J. Phys. Chem. B 2005, 109, 6287–6293. [Google Scholar] [CrossRef] [PubMed]
- Afkhami, A.; Saber-Tehrani, M.; Bagheri, H. Simultaneous removal of heavy-metal ions in wastewater samples using nano-alumina modified with 2,4-dinitrophenylhydrazine. J. Hazard. Mater. 2010, 181, 836–844. [Google Scholar] [CrossRef] [PubMed]
- Brigante, M.; Pecini, E.; Avena, M. Magnetic mesoporous silica for water remediation: Synthesis, characterization and application as adsorbent of molecules and ions of environmental concern. Microporous Mesoporous Mater. 2016, 230, 1–10. [Google Scholar] [CrossRef]
- Nomura, A.; Jones, C.W. Amine-functionalized porous silicas as adsorbents for aldehyde abatement. ACS Appl. Mater. Interfaces 2013, 5, 5569–5577. [Google Scholar] [CrossRef] [PubMed]
- Bollini, P.; Didas, S.A.; Jones, C.W. Amine-oxide hybrid materials for acid gas separations. J. Mater. Chem. 2011, 21, 15100–15120. [Google Scholar] [CrossRef]
- Drese, J.H.; Talley, A.D.; Jones, C.W. Aminosilica materials as adsorbents for the selective removal of aldehydes and ketones from simulated bio-oil. ChemSusChem 2011, 4, 379–385. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.; Drese, J.H.; Eisenberger, P.M.; Jones, C.W. Application of amine-tethered solid sorbents for direct CO2 capture from the ambient air. Environ. Sci. Technol. 2011, 45, 2420–2427. [Google Scholar] [CrossRef] [PubMed]
- Hicks, J.C.; Drese, J.H.; Fauth, D.J.; Gray, M.L.; Qi, G.; Jones, C.W. Designing adsorbents for CO2 capture from flue gas-hyperbranched aminosilicas capable of capturing CO2 reversibly. J. Am. Chem. Soc. 2008, 130, 2902–2903. [Google Scholar] [CrossRef] [PubMed]
- Nomura, A.; Jones, C.W. Enhanced Formaldehyde-Vapor Adsorption Capacity of Polymeric Amine-Incorporated Aminosilicas. Chem. A Eur. J. 2014, 20, 6381–6390. [Google Scholar] [CrossRef] [PubMed]
- Qi, G.; Wang, Y.; Estevez, L.; Duan, X.; Anako, N.; Park, A.-H.A.; Li, W.; Jones, C.W.; Giannelis, E.P. High efficiency nanocomposite sorbents for CO2 capture based on amine-functionalized mesoporous capsules. Energy Environ. Sci. 2011, 4, 444–452. [Google Scholar] [CrossRef]
- Kuwahara, Y.; Kang, D.Y.; Copeland, J.R.; Brunelli, N.A.; Didas, S.A.; Bollini, P.; Sievers, C.; Kamegawa, T.; Yamashita, H.; Jones, C.W. Dramatic enhancement of CO2 uptake by poly(ethyleneimine) using zirconosilicate supports. J. Am. Chem. Soc. 2012, 134, 10757–10760. [Google Scholar] [CrossRef] [PubMed]
- Kuwahara, Y.; Kang, D.Y.; Copeland, J.R.; Bollini, P.; Sievers, C.; Kamegawa, T.; Yamashita, H.; Jones, C.W. Enhanced CO2 adsorption over polymeric amines supported on heteroatom-incorporated SBA-15 silica: Impact of heteroatom type and loading on sorbent structure and adsorption performance. Chem. A Eur. J. 2012, 18, 16649–16664. [Google Scholar] [CrossRef] [PubMed]
- Bruzzoniti, M.C.; Prelle, A.; Sarzanini, C.; Onida, B.; Fiorilli, S.; Garrone, E. Retention of heavy metal ions on SBA-15 mesoporous silica functionalised with carboxylic groups. J. Sep. Sci. 2007, 30, 2414–2420. [Google Scholar] [CrossRef] [PubMed]
- Yan, Z.; Tao, S.; Yin, J.; Li, G. Mesoporous silicas functionalized with a high density of carboxylate groups as efficient absorbents for the removal of basic dyestuffs. J. Mater. Chem. 2006, 16, 2347–2353. [Google Scholar] [CrossRef]
- Wang, S.; Wang, K.; Dai, C.; Shi, H.; Li, J. Adsorption of Pb2+ on amino-functionalized core–shell magnetic mesoporous SBA-15 silica composite. Chem. Eng. J. 2015, 262, 897–903. [Google Scholar] [CrossRef]
- Hernández-Morales, V.; Nava, R.; Acosta-Silva, Y.J.; MacÍas-Sánchez, S.A.; Pérez-Bueno, J.J.; Pawelec, B. Adsorption of lead (II) on SBA-15 mesoporous molecular sieve functionalized with –NH2 groups. Microporous Mesoporous Mater. 2012, 160, 133–142. [Google Scholar] [CrossRef]
- Da’na, E.; Sayari, A. Adsorption of copper on amine-functionalized SBA-15 prepared by co-condensation: Equilibrium properties. Chem. Eng. J. 2011, 166, 445–453. [Google Scholar] [CrossRef]
- Zhao, Y.; Gao, Q.; Tang, T.; Xu, Y.; Wu, D. Effective NH2-grafting on mesoporous SBA-15 surface for adsorption of heavy metal ions. Mater. Lett. 2011, 65, 1045–1047. [Google Scholar] [CrossRef]
- Nakanishi, K.; Tomita, M.; Kato, K. Synthesis of amino-functionalized mesoporous silica sheets and their application for metal ion capture. J. Asian Ceram. Soc. 2015, 3, 70–76. [Google Scholar] [CrossRef]
- Repo, E.; Warchoł, J.K.; Bhatnagar, A.; Mudhoo, A.; Sillanpää, M. Aminopolycarboxylic acid functionalized adsorbents for heavy metals removal from water. Water Res. 2013, 47, 4812–4832. [Google Scholar] [CrossRef] [PubMed]
- Arencibia, A.; Aguado, J.; Arsuaga, J.M. Regeneration of thiol-functionalized mesostructured silica adsorbents of mercury. Appl. Surf. Sci. 2010, 256, 5453–5457. [Google Scholar] [CrossRef]
- Rostamian, R.; Najafi, M.; Rafati, A.A. Synthesis and characterization of thiol-functionalized silica nano hollow sphere as a novel adsorbent for removal of poisonous heavy metal ions from water: Kinetics, isotherms and error analysis. Chem. Eng. J. 2011, 171, 1004–1011. [Google Scholar] [CrossRef]
- Hakami, O.; Zhang, Y.; Banks, C.J. Thiol-functionalised mesoporous silica-coated magnetite nanoparticles for high efficiency removal and recovery of Hg from water. Water Res. 2012, 46, 3913–3922. [Google Scholar] [CrossRef] [PubMed]
- Walcarius, A.; Delacôte, C. Mercury(II) binding to thiol-functionalized mesoporous silicas: Critical effect of pH and sorbent properties on capacity and selectivity. Anal. Chim. Acta 2005, 547, 3–13. [Google Scholar] [CrossRef]
- Vunain, E.; Mishra, A.K.; Mamba, B.B. Dendrimers, mesoporous silicas and chitosan-based nanosorbents for the removal of heavy-metal ions: A review. Int. J. Biol. Macromol. 2016, 86, 570–586. [Google Scholar] [CrossRef] [PubMed]
- Lee, B.; Kim, Y.; Lee, H.; Yi, J. Synthesis of functionalized porous silicas via templating method as heavy metal ion adsorbents: The introduction of surface hydrophilicity onto the surface of adsorbents. Microporous Mesoporous Mater. 2001, 50, 77–90. [Google Scholar] [CrossRef]
- Aguado, J.; Arsuaga, J.M.; Arencibia, A.; Lindo, M.; Gascón, V. Aqueous heavy metals removal by adsorption on amine-functionalized mesoporous silica. J. Hazard. Mater. 2009, 163, 213–221. [Google Scholar] [CrossRef] [PubMed]
- Da’na, E.; Sayari, A. Optimization of copper removal efficiency by adsorption on amine-modified SBA-15: Experimental design methodology. Chem. Eng. J. 2011, 167, 91–98. [Google Scholar] [CrossRef]
- Da’na, E.; Sayari, A. Adsorption of heavy metals on amine-functionalized SBA-15 prepared by co-condensation: Applications to real water samples. Desalination 2012, 285, 62–67. [Google Scholar] [CrossRef]
- Lei, D.; Zheng, Q.; Wang, Y.; Wang, H. Preparation and evaluation of aminopropyl-functionalized manganese-loaded SBA-15 for copper removal from aqueous solution. J. Environ. Sci. 2015, 28, 118–127. [Google Scholar] [CrossRef] [PubMed]
- Iwamoto, M. Fast and Quantitative Dehydration of Lower Alcohols to Corresponding Olefins on Mesoporous Silica Catalyst. Chem. Lett. 2011, 1, 614–616. [Google Scholar] [CrossRef]
- Ren, X.; Chen, C.; Nagatsu, M.; Wang, X. Carbon nanotubes as adsorbents in environmental pollution management: A review. Chem. Eng. J. 2011, 170, 395–410. [Google Scholar] [CrossRef]
- Theron, J.; Walker, J.A.; Cloete, T.E. Nanotechnology and Water Treatment: Applications and Emerging Opportunities. Crit. Rev. Microbiol. 2008, 34, 43–69. [Google Scholar] [CrossRef] [PubMed]
- Di Paola, A.; García-López, E.; Marcì, G.; Palmisano, L. A survey of photocatalytic materials for environmental remediation. J. Hazard. Mater. 2012, 211–212, 3–29. [Google Scholar] [CrossRef] [PubMed]
- Kharisov, B.I.; Rasika Dias, H.V.; Kharissova, O.V. Nanotechnology-based remediation of petroleum impurities from water. J. Pet. Sci. Eng. 2014, 122, 705–718. [Google Scholar] [CrossRef]
- Zhang, Y.; Tang, Z.-R.; Fu, X.; Xu, Y.-J. TiO2 Graphene Nanocomposites for Gas-Phase Photocatalytic Degradation of Volatile Aromatic Pollutant: Is TiO2 Graphene Truly Different from Other TiO2 Carbon Composite Materials? ACS Nano 2010, 4, 7303–7314. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Pan, L.; Lv, T.; Lu, T.; Zhu, G.; Sun, Z.; Sun, C. Microwave-assisted synthesis of ZnO-graphene composite for photocatalytic reduction of Cr(vi). Catal. Sci. Technol. 2011, 1, 1189–1193. [Google Scholar] [CrossRef]
- Chowdhury, S.; Balasubramanian, R. Recent advances in the use of graphene-family nanoadsorbents for removal of toxic pollutants from wastewater. Adv. Colloid Interface Sci. 2014, 204, 35–56. [Google Scholar] [CrossRef] [PubMed]
- Yang, M.; Zhang, N.; Xu, Y. Synthesis of Fullerene-, Carbon Nanotube-, and Graphene-TiO2 Nanocomposite Photocatalysts for Selective Oxidation: A Comparative Study. ACS Appl. Mater. Interfaces 2013, 5, 1156–1164. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Zhang, P.; Du, Q.; Peng, X.; Liu, T.; Wang, Z.; Xia, Y.; Zhang, W.; Wang, K.; Zhu, H.; et al. Adsorption of fluoride from aqueous solution by graphene. J. Colloid Interface Sci. 2011, 363, 348–354. [Google Scholar] [CrossRef] [PubMed]
- Seredych, M.; Bandosz, T.J. Removal of ammonia by graphite oxide via its intercalation and reactive adsorption. Carbon 2007, 45, 2130–2132. [Google Scholar] [CrossRef]
- Song, B.; Zhang, C.; Zeng, G.; Gong, J.; Chang, Y.; Jiang, Y. Antibacterial properties and mechanism of graphene oxide-silver nanocomposites as bactericidal agents for water disinfection. Arch. Biochem. Biophys. 2016, 604, 167–176. [Google Scholar] [CrossRef] [PubMed]
- Gao, P.; Ng, K.; Sun, D.D. Sulfonated graphene oxide-ZnO-Ag photocatalyst for fast photodegradation and disinfection under visible light. J. Hazard. Mater. 2013, 262, 826–835. [Google Scholar] [CrossRef] [PubMed]
- Deng, C.H.; Gong, J.L.; Zhang, P.; Zeng, G.M.; Song, B.; Liu, H.Y. Preparation of melamine sponge decorated with silver nanoparticles-modified graphene for water disinfection. J. Colloid Interface Sci. 2017, 488, 26–38. [Google Scholar] [CrossRef] [PubMed]
- Zhang, N.; Yang, M.-Q.; Tang, Z.-R.; Xu, Y.-J. CdS-graphene nanocomposites as visible light photocatalyst for redox reactions in water: A green route for selective transformation and environmental remediation. J. Catal. 2013, 303, 60–69. [Google Scholar] [CrossRef]
- Wang, S.; Sun, H.; Ang, H.M.; Tadé, M.O. Adsorptive remediation of environmental pollutants using novel graphene-based nanomaterials. Chem. Eng. J. 2013, 226, 336–347. [Google Scholar] [CrossRef]
- An, X.; Yu, J.C. Graphene-based photocatalytic composites. RSC Adv. 2011, 1, 1426–1434. [Google Scholar] [CrossRef]
- Lithoxoos, G.P.; Labropoulos, A.; Peristeras, L.D.; Kanellopoulos, N.; Samios, J.; Economou, I.G. Adsorption of N2, CH4, CO and CO2 gases in single walled carbon nanotubes: A combined experimental and Monte Carlo molecular simulation study. J. Supercrit. Fluids 2010, 55, 510–523. [Google Scholar] [CrossRef]
- Yang, Q.H.; Hou, P.X.; Cheng, H.M. Adsorption and capillarity of nitrogen in inside channel of carbon nanotubes. Abstr. Pap. Am. Chem. Soc. 2001, 221, U581. [Google Scholar]
- Li, Y.-H.; Luan, Z.; Di, Z.; Zhu, Y.; Xu, C.; Wu, D.; Wei, B.; Ding, J. Competitive adsorption of Pb, Cu and Cd ions from aqueous solutions by multiwalled carbon nanotubes. Carbon 2003, 41, 2787–2792. [Google Scholar] [CrossRef]
- Zuttel, A.; Mauron, P.; Kiyobayashi, T.; Emmenegger, C.; Schlapbach, L. Hydrogen storage in carbon nanostructures. Int. J. Hydrog. Energy 2002, 27, 203–212. [Google Scholar] [CrossRef]
- Tungittiplakorn, W.; Lion, L.W.; Cohen, C.; Kim, J.Y. Engineered Polymeric Nanoparticles for Soil Remediation. Environ. Sci. Technol. 2004, 38, 1605–1610. [Google Scholar] [CrossRef] [PubMed]
- Diallo, M.S.; Christie, S.; Swaminathan, P.; Johnson, J.H.; Goddard, W.A. Dendrimer enhanced ultrafiltration. 1. Recovery of Cu(II) from aqueous solutions using PAMAM dendrimers with ethylene diamine core and terminal NH2 groups. Environ. Sci. Technol. 2005, 39, 1366–1377. [Google Scholar] [CrossRef] [PubMed]
- Guerra, F.D.; Campbell, M.L.; Whitehead, D.C.; Alexis, F. Capture of Aldehyde VOCs Using a Series of Amine-Functionalized Cellulose Nanocrystals. ChemistrySelect 2018, 3, 5495–5501. [Google Scholar] [CrossRef]
- Khare, P.; Yadav, A.; Ramkumar, J.; Verma, N. Microchannel-embedded metal–carbon–polymer nanocomposite as a novel support for chitosan for efficient removal of hexavalent chromium from water under dynamic conditions. Chem. Eng. J. 2016, 293, 44–54. [Google Scholar] [CrossRef]
- Mittal, H.; Maity, A.; Ray, S.S. Synthesis of co-polymer-grafted gum karaya and silica hybrid organic–inorganic hydrogel nanocomposite for the highly effective removal of methylene blue. Chem. Eng. J. 2015, 279, 166–179. [Google Scholar] [CrossRef]
- Sun, X.-F.; Liu, B.; Jing, Z.; Wang, H. Preparation and adsorption property of xylan/poly (acrylic acid) magnetic nanocomposite hydrogel adsorbent. Carbohydr. Polym. 2015, 118, 16–23. [Google Scholar] [CrossRef] [PubMed]
- Ge, F.; Li, M.-M.; Ye, H.; Zhao, B.-X. Effective removal of heavy metal ions Cd2+, Zn2+, Pb2+, Cu2+ from aqueous solution by polymer-modified magnetic nanoparticles. J. Hazard. Mater. 2012, 211, 366–372. [Google Scholar] [CrossRef] [PubMed]
- Brinkmann, N.; Giebel, D.; Lohmer, G.; Reetz, M.T.; Kragl, U. Allylic substitution with dendritic palladium catalysts in a continuously operating membrane reactor. J. Catal. 1999, 183, 163–168. [Google Scholar] [CrossRef]
- Biricova, V.; Laznickova, A. Dendrimers: Analytical characterization and applications. Bioorg. Chem. 2009, 37, 185–192. [Google Scholar] [CrossRef] [PubMed]
- Trujillo-Reyes, J.; Peralta-Videa, J.R.; Gardea-Torresdey, J.L. Supported and unsupported nanomaterials for water and soil remediation: Are they a useful solution for worldwide pollution? J. Hazard. Mater. 2014, 280, 487–503. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Bhattacharyya, D. Membrane-based bimetallic nanoparticles for environmental remediation: Synthesis and reactive properties. Environ. Prog. 2005, 24, 358–366. [Google Scholar] [CrossRef]
- Kim, J.; Van Der Bruggen, B. The use of nanoparticles in polymeric and ceramic membrane structures: Review of manufacturing procedures and performance improvement for water treatment. Environ. Pollut. 2010, 158, 2335–2349. [Google Scholar] [CrossRef] [PubMed]
- Lakouraj, M.M.; Mojerlou, F.; Zare, E.N. Nanogel and superparamagnetic nanocomposite based on sodium alginate for sorption of heavy metal ions. Carbohydr. Polym. 2014, 106, 34–41. [Google Scholar] [CrossRef] [PubMed]
- Rieger, K.A.; Cho, H.J.; Yeung, H.F.; Fan, W.; Schiffman, J.D. Antimicrobial activity of silver ions released from zeolites immobilized on cellulose nanofiber mats. ACS Appl. Mater. Interfaces 2016, 8, 3032–3040. [Google Scholar] [CrossRef] [PubMed]
- Elechiguerra, J.L.; Burt, J.L.; Morones, J.R.; Camacho-Bragado, A.; Gao, X.; Lara, H.H.; Yacaman, M.J. Interaction of silver nanoparticles with HIV-1. J. Nanobiotechnol. 2005, 3, 6. [Google Scholar] [CrossRef] [PubMed]
- Nair, A.S.; Binoy, N.P.; Ramakrishna, S.; Kurup, T.R.R.; Chan, L.W.; Goh, C.H.; Islam, M.R.; Utschig, T.; Pradeep, T. Organic-Soluble Antimicrobial Silver Nanoparticle-Polymer Composites in Gram Scale by One-Pot Synthesis. ACS Appl. Mater. Interfaces 2009, 1, 2413–2419. [Google Scholar] [CrossRef] [PubMed]
- Jin, G.; Prabhakaran, M.P.; Nadappuram, B.P.; Singh, G.; Kai, D.; Ramakrishna, S. Electrospun poly (l-lactic acid)-co-poly (ϵ-caprolactone) nanofibres containing silver nanoparticles for skin-tissue engineering. J. Biomater. Sci. Polym. Ed. 2012, 23, 2337–2352. [Google Scholar] [PubMed]
- Zaman, J.; Chakma, A. Inorganic membrane reactors. J. Memb. Sci. 1994, 92, 1–28. [Google Scholar] [CrossRef]
- Rengaraj, S.; Li, X.Z. Enhanced photocatalytic activity of TiO2 by doping with Ag for degradation of 2,4,6-trichlorophenol in aqueous suspension. J. Mol. Catal. A Chem. 2006, 243, 60–67. [Google Scholar] [CrossRef]
- Srisitthiratkul, C.; Pongsorrarith, V.; Intasanta, N. The potential use of nanosilver-decorated titanium dioxide nanofibers for toxin decomposition with antimicrobial and self-cleaning properties. Appl. Surf. Sci. 2011, 257, 8850–8856. [Google Scholar] [CrossRef]
- Sa, J.; Agüera, C.A.; Gross, S.; Anderson, J.A. Photocatalytic nitrate reduction over metal modified TiO2. Appl. Catal. B Environ. 2009, 85, 192–200. [Google Scholar] [CrossRef]
- Diallo, M.S.; Falconer, K.; Johnson, J.H.; Goddard, W.A., III. Dendritic anion hosts: Perchlorate uptake by G5-NH2 poly (propyleneimine) dendrimer in water and model electrolyte solutions. Environ. Sci. Technol. 2007, 41, 6521–6527. [Google Scholar] [CrossRef] [PubMed]
- Kouketsu, T.; Duan, S.; Kai, T.; Kazama, S.; Yamada, K. PAMAM dendrimer composite membrane for CO2 separation: Formation of a chitosan gutter layer. J. Memb. Sci. 2007, 287, 51–59. [Google Scholar] [CrossRef]
- Yew, S.P.; Tang, H.Y.; Sudesh, K. Photocatalytic activity and biodegradation of polyhydroxybutyrate films containing titanium dioxide. Polym. Degrad. Stab. 2006, 91, 1800–1807. [Google Scholar] [CrossRef]
- Sugunan, A.; Thanachayanont, C.; Dutta, J.; Hilborn, J.G. Heavy-metal ion sensors using chitosan-capped gold nanoparticles. Sci. Technol. Adv. Mater. 2005, 6, 335–340. [Google Scholar] [CrossRef]
- Baybas, D.; Ulusoy, U. The use of polyacrylamide-aluminosilicate composites for thorium adsorption. Appl. Clay Sci. 2011, 51, 138–146. [Google Scholar] [CrossRef]
- Li, Y.-H.; Wang, S.; Cao, A.; Zhao, D.; Zhang, X.; Xu, C.; Luan, Z.; Ruan, D.; Liang, J.; Wu, D.; et al. Adsorption of fluoride from water by amorphous alumina supported on carbon nanotubes. Chem. Phys. Lett. 2001, 350, 412–416. [Google Scholar] [CrossRef]
- Lu, C.; Chiu, H. Adsorption of zinc (II) from water with purified carbon nanotubes. Chem. Eng. Sci. 2006, 61, 1138–1145. [Google Scholar] [CrossRef]
| Material | Application | Reference |
|---|---|---|
| Ag NPs/Ag ions | Water disinfectant—E. coli | [20,21,22,23,24] |
| TiO2 NPs | Water disinfectant, soil—MS-2 phage, E. coli, hepatitis B virus, aromatic hydrocarbons, biological nitrogen, phenanthrene | [25,26,27,28,29] |
| Metal-doped TiO2 | Water contaminants—2-chlorophenol, endotoxin, E. coli, Rhodamine B, Staphylococcus aureus | [30,31,32,33,34] |
| Titanate nanotubes | Gaseous—Nitric oxide | [35] |
| Binary mixed oxide | Water—Methylene blue dye | [36] |
| Iron-based | Water—Heavy metals, chlorinated organic solvents | [37,38,39,40,41,42,43] |
| Bimetallic NPs | Water, soil—Chlorinated and brominated contaminants | [44,45,46,47,48,49,50,51,52,53] |
| Material | Application | Reference |
|---|---|---|
| Amine-modified xerogels | Gaseous—CO2, H2S | [81] |
| Amine-modified aluminosilicates and porous silica | Gaseous—CO2, aldehydes, ketones | [91,92,93,94,95,96,97,98,99] |
| Carboxylic acid-functionalized mesoporous silica | Wastewater—Cationic dyes, heavy metals | [78,100,101] |
| Amino-functionalized mesoporous silica | Wastewater—Heavy metals | [102,103,104,105,106,107] |
| Thiol-functionalized mesoporous silica | Wastewater—Heavy metals | [108,109,110,111] |
| Material | Application | Reference |
|---|---|---|
| Pristine graphene | Water—Fluoride | [127] |
| Graphene oxide | Water/Gaseous—SOx, H2, NH3, heavy metals, pesticides, pharmaceuticals | [31,128,129,130,131] |
| ZnO-graphene/CdS-graphene | Water—Heavy metals | [132] |
| TiO2-graphene | Gaseous—Benzene | [123,126] |
| Material | Application | Reference |
|---|---|---|
| Amphiphilic polyurethane NPs | Soil—Polynuclear aromatic hydrocarbons | [139] |
| PAMAM dendrimers | Wastewater—Heavy metals | [140] |
| Amine-modified PDLLA-PEG | Gaseous—VOCs | [6,7] |
| Polyamine-modified Cellulose | Gaseous—VOCs | [141] |
| Polymer nanocomposites (PNCs) | Water—Metal ions, dyes, microorganisms | [142,143,144,145] |
| Type of Nanoparticles | Removal Target | Reference |
|---|---|---|
| Ag-doped TiO2 | 2,4,6-Trichlorophenol | [157] |
| Ag-doped TiO2 nanofibers | Methylene blue dye | [158] |
| Cu/Fe/Ag-doped TiO2 | Nitrate (NO3−) | [159] |
| Silica nanoparticles prepared by mixing salicylic acid and hyper-branched poly (propylene imine) | Removal of polycyclic aromatic hydrocarbons (PAH), such as pyrene and phenanthrene, and Pb2+, Hg2+, Cd2+, Cr2O72− from contaminated aqueous solutions | [160] |
| PAMAM dendrimer composite membrane consisting of chitosan and a dendrimer | Separation of CO2 from a feed gas mixture of CO2 and N2 on porous substrates | [161] |
| Fe0 coated with carboxymethyl cellulose polymer matrix | Hexavalent chromium (Cr6+) from aqueous solutions | [162] |
| Gold coated with chitosan polymer | Zn2+, Cu2+ form aqueous solutions | [163] |
| Poly (methacrylic acid)-grafted chitosan/bentonite | Th4+ | [164] |
| Carbon nanotubes/Al2O3 nanocomposite | Fluoride | [165] |
| Multiwall carbon nanotube (MWCTs) | Zn2+ | [166] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guerra, F.D.; Attia, M.F.; Whitehead, D.C.; Alexis, F. Nanotechnology for Environmental Remediation: Materials and Applications. Molecules 2018, 23, 1760. https://doi.org/10.3390/molecules23071760
Guerra FD, Attia MF, Whitehead DC, Alexis F. Nanotechnology for Environmental Remediation: Materials and Applications. Molecules. 2018; 23(7):1760. https://doi.org/10.3390/molecules23071760
Chicago/Turabian StyleGuerra, Fernanda D., Mohamed F. Attia, Daniel C. Whitehead, and Frank Alexis. 2018. "Nanotechnology for Environmental Remediation: Materials and Applications" Molecules 23, no. 7: 1760. https://doi.org/10.3390/molecules23071760
APA StyleGuerra, F. D., Attia, M. F., Whitehead, D. C., & Alexis, F. (2018). Nanotechnology for Environmental Remediation: Materials and Applications. Molecules, 23(7), 1760. https://doi.org/10.3390/molecules23071760