Application of Docking Analysis in the Prediction and Biological Evaluation of the Lipoxygenase Inhibitory Action of Thiazolyl Derivatives of Mycophenolic Acid
Abstract
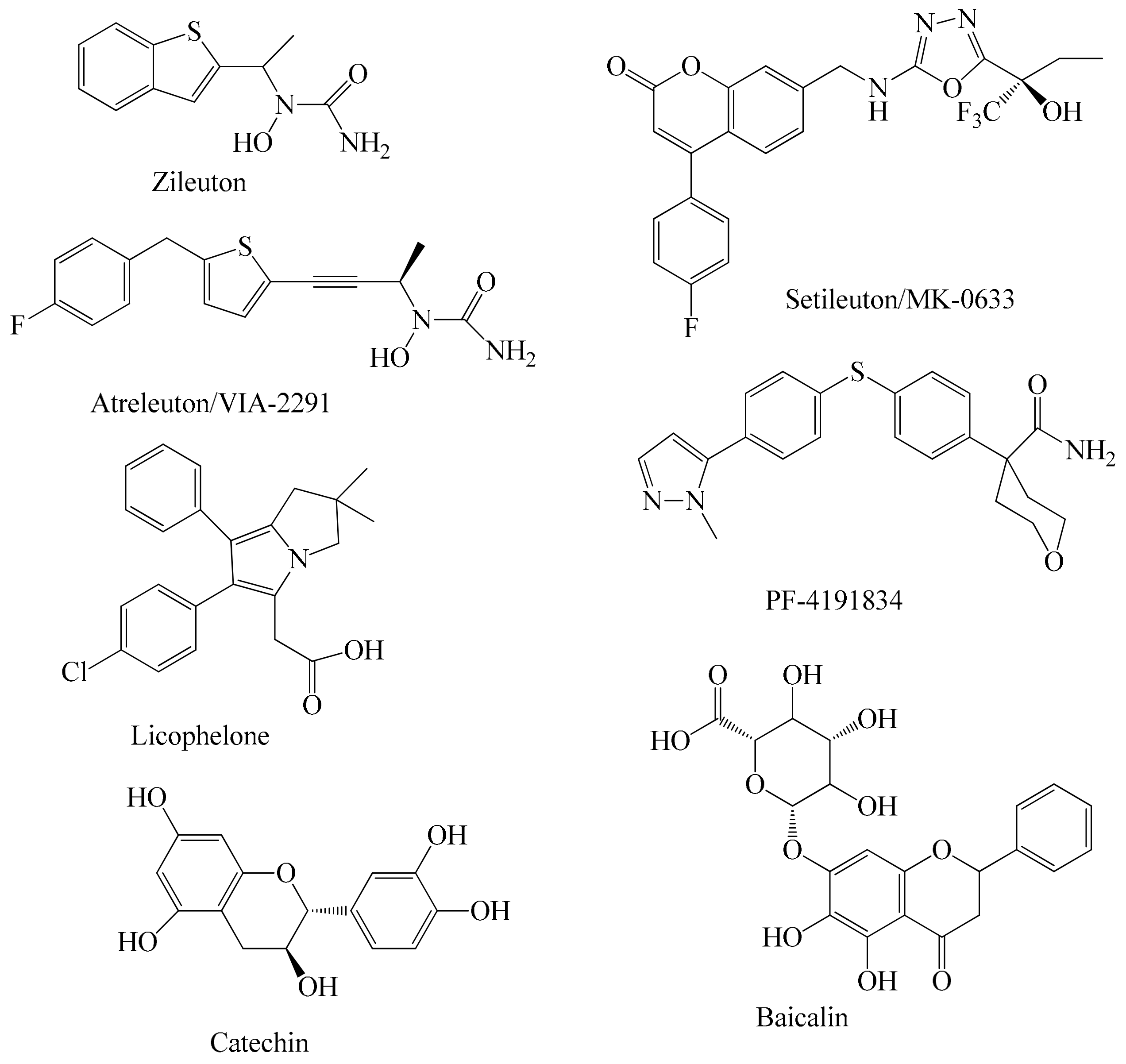
1. Introduction
2. Results and Discussion
2.1. Prediction of Biological Activity Spectra of Compounds by PASS
2.2. Prediction of Toxicity
- Category I: LD50 ≤ 5 mg/kg
- Category II: 5 < LD50 ≤ 50 mg/kg
- Category III: 50 < LD50 ≤ 300 mg/kg
- Category IV: 300 < LD50 ≤ 2000 mg/kg
- Category V: 2000 < LD50 ≤ 5000 mg/kg
- Category VI: LD50 > 5000 mg/kg
2.3. Docking-Aided Prediction of the Inhibitory Action
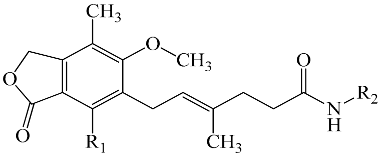
2.4. Chemistry
2.5. Biological Evaluation
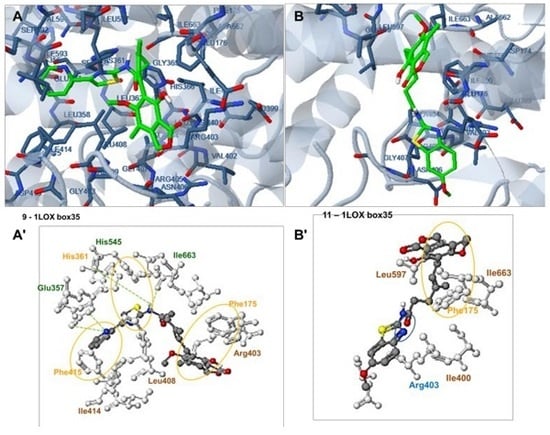
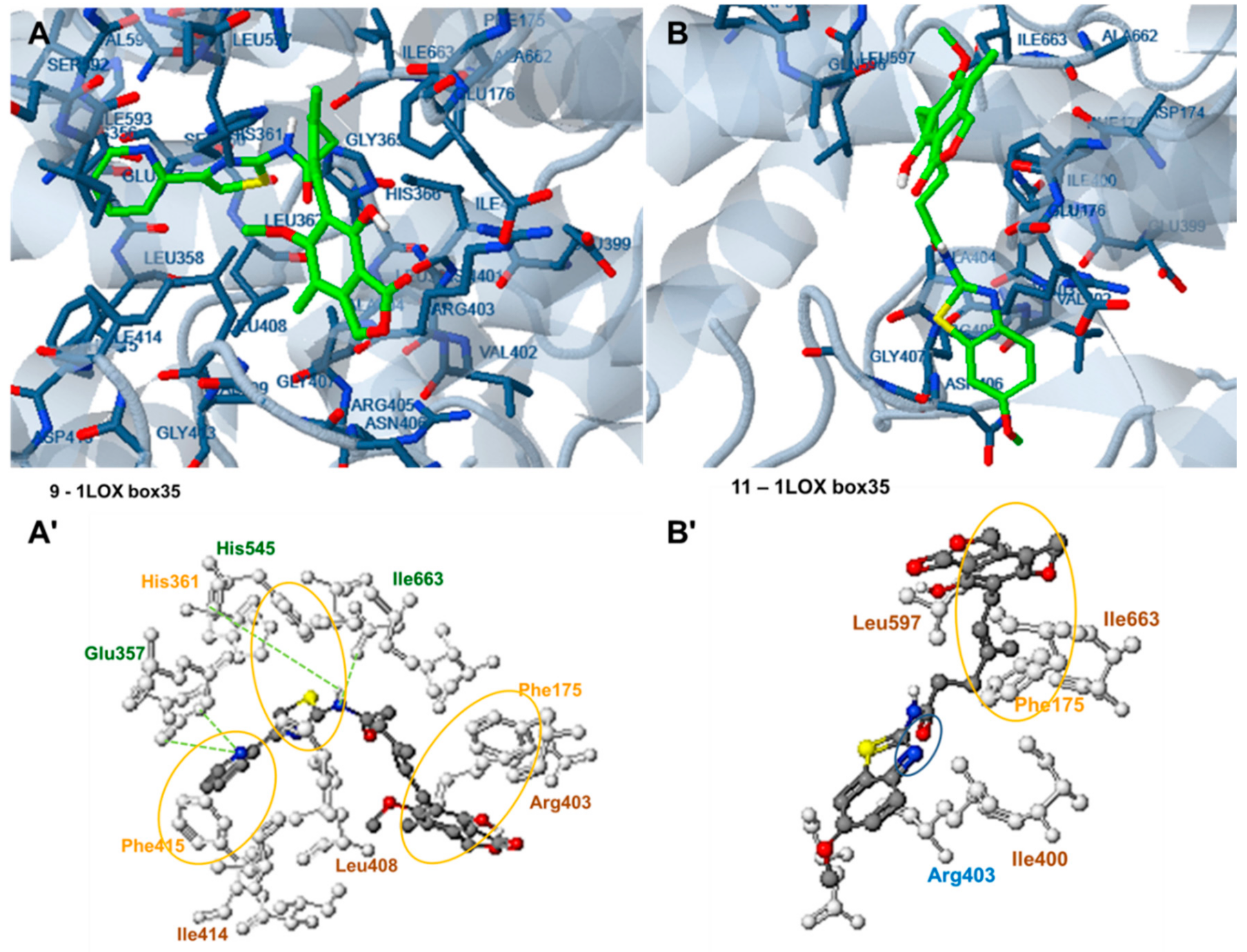
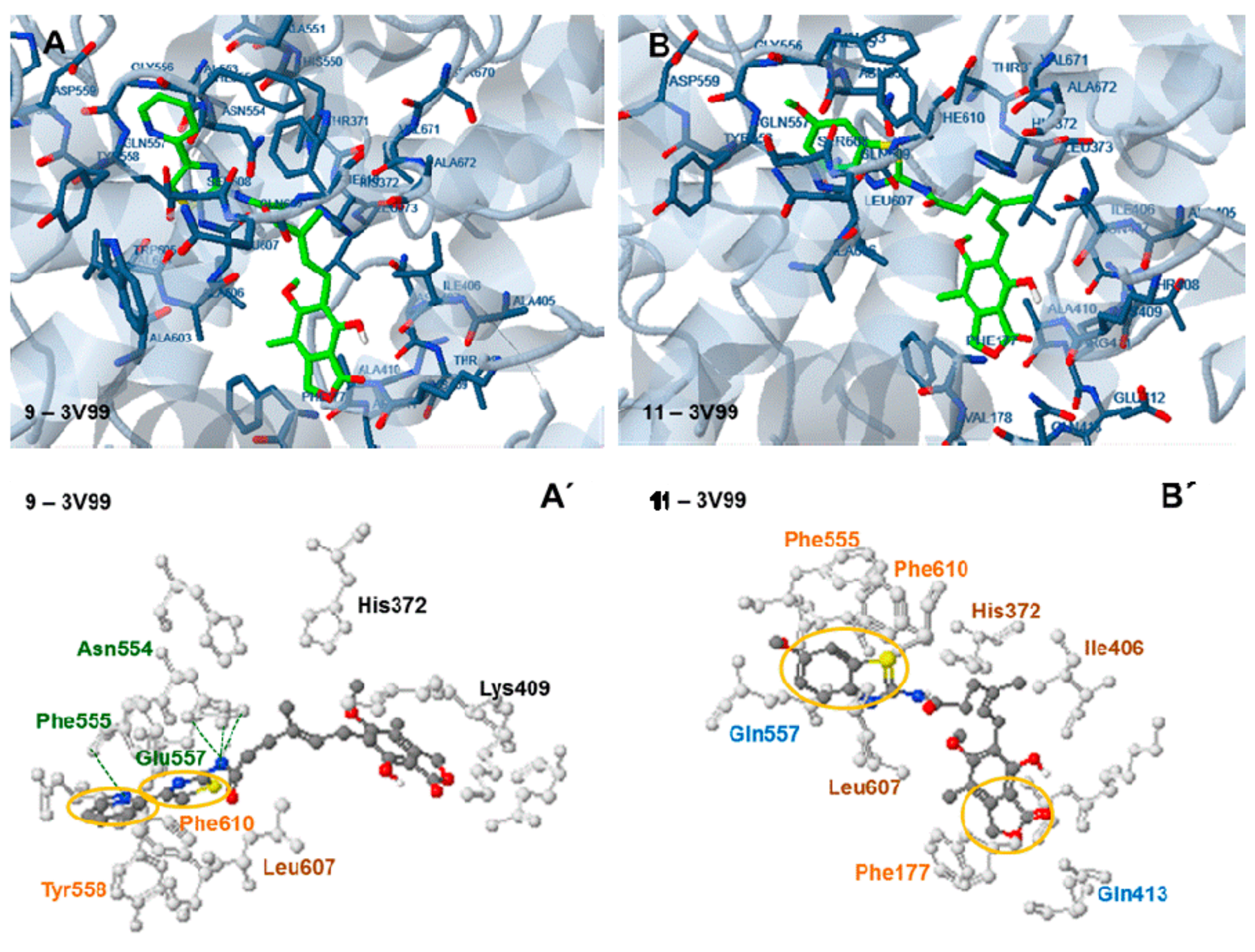
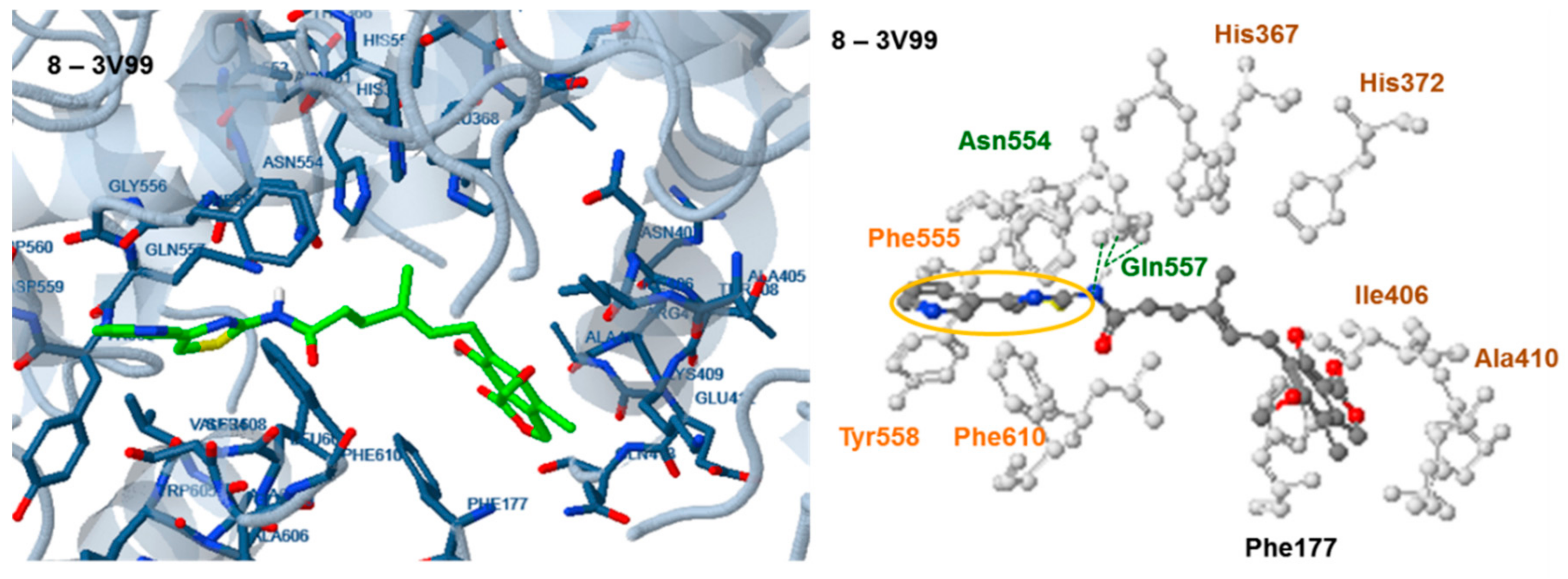
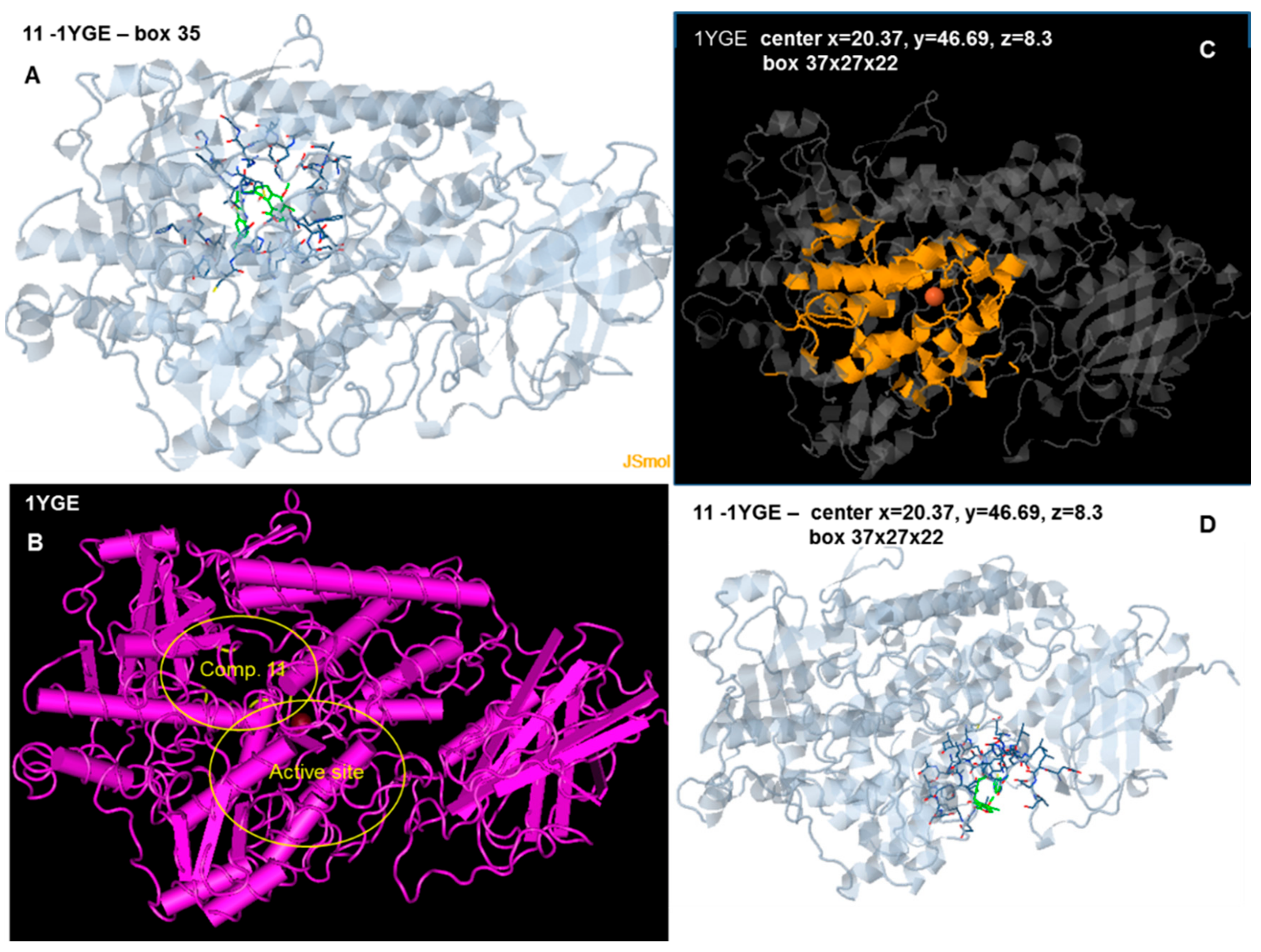
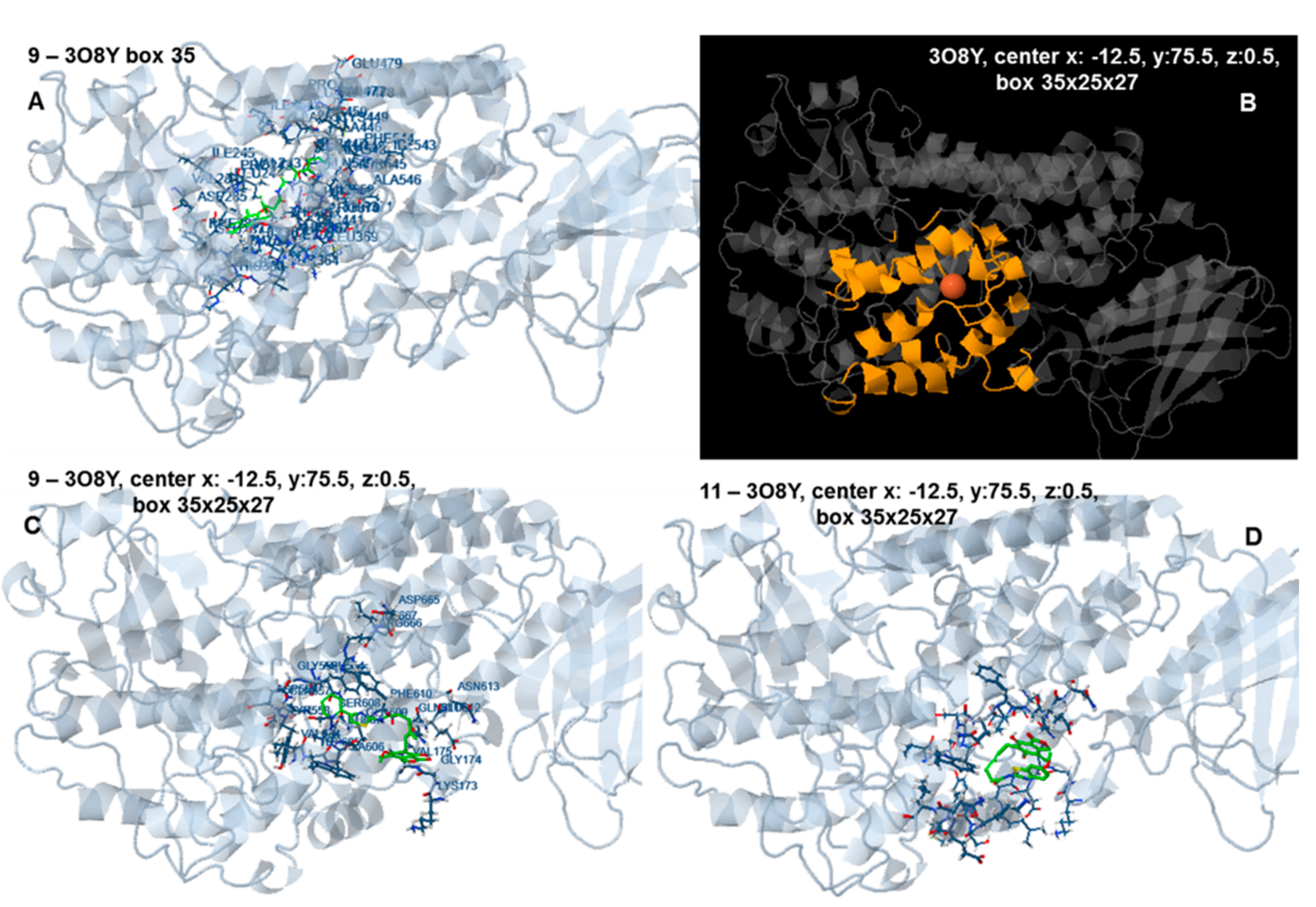
2.6. Docking Analysis-Assisted Justification of Inhibitory Action
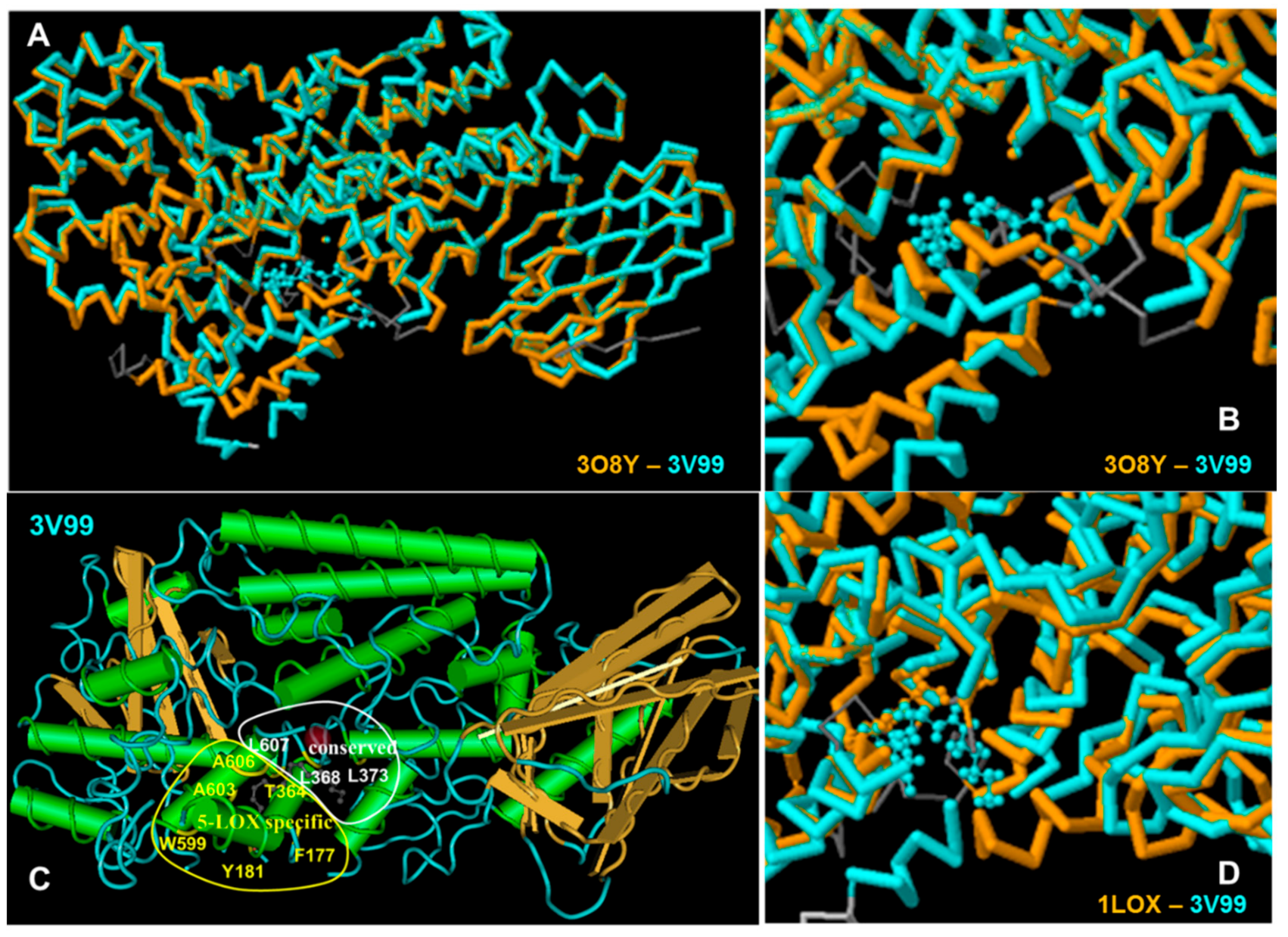
2.7. Evaluation of Docking Analysis Efficiency
3. Materials and Methods
3.1. General Information
3.2. Biological Activity Spectra Prediction
3.3. Docking Analysis
3.4. Chemistry
General Method for the Synthesis of Mycophenolic Acid Derivatives
3.5. Biological Assay
Soybean Lipoxygenase Inhibition Study In Vitro
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hirata, F.; Axelrod, J. Phospholipid methylation and biological signal transmission. Science 1980, 209, 1082–1090. [Google Scholar] [CrossRef] [PubMed]
- Roberts, L.J. Introduction: Lipids as regulators of cell function. Cell. Mol. Life Sci. 2002, 59, 727–728. [Google Scholar] [CrossRef]
- Kuhn, H. Lipoxygenases. In Prostaglandins, Leukotrienes and Other Eicosanoids; Marks, F., Fustenberger, G., Eds.; Wiley-VCH: Weinheim, Germany, 1999; p. 109. ISBN 978-3-527-61363-2. [Google Scholar]
- Radmark, O.; Samuelsson, B. 5-Lipoxygenase: Mechanisms of regulation. J. Lipid Res. 2009, 50, S40–S45. [Google Scholar] [CrossRef] [PubMed]
- Schneider, C.; Pratt, D.A.; Porter, N.A.; Brash, A.R. Control of oxygenation in lipoxygenase and cyclooxygenase catalysis. Chem. Biol. 2007, 14, 473–488. [Google Scholar] [CrossRef] [PubMed]
- Kuhn, H. Mammalian arachidonate 15-lipoxygenases structure, function, and biological implications. Prostaglandins Other Lipid Mediat. 2002, 68–69, 263–290. [Google Scholar] [CrossRef]
- Hussain, H.; Shornick, L.P.; Shannon, V.R.; Wilson, J.D.; Funk, C.D.; Pentland, A.P.; Holtzman, M.J. Epidermis contains platelet-type 12-lipoxygenase that is overexpressed in germinal layer keratinocytes in psoriasis. Am. J. Physiol. 1994, 266, C243–C253. [Google Scholar] [CrossRef] [PubMed]
- Yoo, H.; Jeon, B.; Jeon, M.-S.; Lee, H.; Kim, T.-Y. Reciprocal regulation of 12 and 15 lipoxygenases by UV-irradiation in human keratinocytes. FEBS Lett. 2008, 582, 3249–3253. [Google Scholar] [CrossRef] [PubMed]
- Montuschi, P. Role of Leukotrienes and Leukotriene Modifiers in Asthma. Pharmaceuticals 2010, 3, 1792–1811. [Google Scholar] [CrossRef] [PubMed]
- Mashima, R.T. The role of lipoxygenases in pathophysiology; new insights and future perspectives. Redox Biol. 2015, 6, 297–310. [Google Scholar] [CrossRef] [PubMed]
- Bray, M.A.; Ford-Hutchinson, A.W.; Smith, M.J. Leukotriene B4: An inflammatory mediator in vivo. Prostaglandins 1981, 22, 213–222. [Google Scholar] [CrossRef]
- Peskar, B.M. Role of leukotriene C4 in mucosal damage caused by necrotizing agents and indomethacin in the rat stomach. Gastroenterology 1991, 100, 619–626. [Google Scholar] [CrossRef]
- Zhao, L.; Funk, C.D. Lipoxygenase pathways in atherogenesis. Trends Cardiovasc. Med. 2004, 14, 191–195. [Google Scholar] [CrossRef] [PubMed]
- Xu, S.; Ogura, S.; Chen, J.; Little, P.J.; Moss, J.; Liu, P. LOX-1 in atherosclerosis: Biological functions and pharmacological modifiers. Cell Mol. Life Sci. 2013, 70, 2859–2872. [Google Scholar] [CrossRef] [PubMed]
- Park, Y.B.; Ahn, C.W.; Choi, H.K.; Lee, S.H.; In, B.H.; Lee, H.C.; Nam, C.M.; Lee, S.K. Atherosclerosis in rheumatoid arthritis: Morphologic evidence obtained by carotid ultrasound. Arthritis Rheum. 2002, 46, 1714–1719. [Google Scholar] [CrossRef] [PubMed]
- Radmark, O.; Samuelsson, B. 5-lipoxygenase: Regulation and possible involvement in atherosclerosis. Prostagl. Lipid Mediat. 2007, 83, 162–174. [Google Scholar] [CrossRef] [PubMed]
- Czapski, G.A.; Czubowicz, K.; Strosznajder, J.B.; Strosznajder, R.P. The Lipoxygenases: Their Regulation and Implication in Alzheimer’s Disease. Neurochem. Res. 2016, 41, 243–257. [Google Scholar] [CrossRef] [PubMed]
- Nithipatikom, K.; Campbell, W. Roles of eicosanoids in prostate cancer. Future Lipidol. 2008, 3, 453–467. [Google Scholar] [CrossRef] [PubMed]
- Wei, B.; Zhou, X.; Liang, C.; Zheng, X.; Lei, P.; Fang, J.; Han, X.; Wang, L.; Qi, C.; Wei, H. Human colorectal cancer progression correlates with LOX-induced ECM stiffening. Int. J. Biol. Sci. 2017, 13, 1450–1457. [Google Scholar] [CrossRef] [PubMed]
- Anwar, Y.; Sabir, J.S.; Qureshi, M.I.; Saini, K.S. 5-lipoxygenase: A promising drug target against inflammatory diseases-biochemical and pharmacological regulation. Curr. Drug Targets 2014, 15, 410–422. [Google Scholar] [CrossRef] [PubMed]
- Van Leyen, K. Lipoxygenase: An Emerging Target for Stroke Therapy. CNS Neurol. Disord. Drug Targets 2013, 12, 191–199. [Google Scholar] [CrossRef] [PubMed]
- Mehta, J.L.; Khaidakov, M.; Hermonat, P.L.; Mitra, S.; Wang, X.; Novelli, G.; Sawamura, T. LOX-1: A new target for therapy for cardiovascular diseases. Cardiovasc. Drugs Ther. 2011, 25, 495–500. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Yokomizo, T. The Role of Leukotriene B4 in Allergic Diseases. Allergol. Int. 2015, 64, 17–26. [Google Scholar] [CrossRef] [PubMed]
- Abdelazeem, A.H.; El-Saadi, M.T; Safi El-Din, A.G.; El-Moghazy, S.M. Design, synthesis and biological evaluation of novel diphenylthiazole-thiazolidin-4-one-based derivatives as anti-inflammatory/analgesic agents. J. Chem. Pharm. Res. 2015, 7, 1073–1079. [Google Scholar]
- Kouatly, O.; Eleftheriou, P.; Petrou, A.; Hadjipavlou-Litina, D.; Geronikaki, A. Docking assisted design of novel 4-adamantanyl-2-thiazolylimino-5-arylidene-4-thiazolidinones as potent NSAIDs. SAR QSAR Environ. Res. 2018, 29, 83–101. [Google Scholar] [CrossRef] [PubMed]
- Eleftheriou, P.; Geronikaki, A.; Hadjipavlou-Litina, D.; Vicini, P.; Filz, O.; Filimonov, D.; Poroikov, V.; Chaudhaery, S.S.; Roy, K.K.; Saxena, A.K. Fragment-based design, docking, synthesis, biological evaluation and structure-activity relationships of 2-benzo/benzisothiazolimino-5-aryliden-4-thiazolidinones as cycloxygenase/lipoxygenase inhibitors. Eur. J. Med. Chem. 2012, 47, 111–124. [Google Scholar] [CrossRef] [PubMed]
- Lagunin, A.A.; Geronikak, A.; Eleftheriou, P.T.; Hadjipavlou-Litina, D.I.; Filimonov, D.I.; Poroikov, V.V. Computer-aided discovery of potential anti-inflammatory thiazolidinones with dual 5-LOX/COX inhibition. J. Med. Chem. 2008, 51, 1601–1609. [Google Scholar]
- Joshi, E.M.; Healsley, B.H.; Macdonald, T.L. 2-ABT-S-oxide detoxification by glutathione s-transferases A1-1, M1-1 and P1-1; implications for toxicity associated with zileuton. Xenobiotica 2009, 39, 197–204. [Google Scholar] [CrossRef] [PubMed]
- Braeckman, R.A.; Granneman, G.R.; Locke, C.S.; Machinisi, J.M.; Cavannaugh, J.H.; Awni, W.M. The pharmacokinetics of zileuton in healthy young and elderly volunteers. Clin. Pharmacokine 1995, 29, 42–48. [Google Scholar] [CrossRef]
- Tardif, J.C.; L’allier, P.L.; Ibrahim, R.; Grégoire, J.C.; Nozza, A.; Cossette, M.; Kouz, S.; Lavoie, M.A.; Paquin, J.; Brotz, T.M.; et al. Treatmnet with 5-lipoxygenase inhibitor VIA-2291(Altreleuton) in patients with recent acute coronary syndrome. Circ. Cardiovas. Imaging 2010, 2, 298–307. [Google Scholar] [CrossRef] [PubMed]
- Wasfi, Y.S.; Villaran, C.; de Tilleghem, C.B.; Smugar, S.S.; Hanley, W.D.; Reiss, T.F.; Knor, B.A. The efficacy and tolerability of MK-0633, a 5-lipoxygenase inhibitor, in chronic asthma. Respir. Med. 2012, 106, 34–46. [Google Scholar] [CrossRef] [PubMed]
- Kulkarni, S.K.; Singh, V.P. Licofelone: The answer to unmet needs in osteoarthritis therapy? Curr. Reumatol. Rep. 2008, 10, 43–48. [Google Scholar] [CrossRef]
- Alvaro-Gracia, J.M. Licofelone-clinical update on a novel LOX/COX inhibitor for the treatment of osteoarthritis. Rheumatology 2004, 43, 21–25. [Google Scholar] [CrossRef] [PubMed]
- Masferrer, J.L.; Zweifel, B.S.; Hardy, M.; Anderson, G.D.; Dufield, D.; Cortes-Burgos, L.; Pufahl, R.A.; Graneto, M. Pharmac ology of PF-4191834, a novel selective non-redox 5-lipoxygenase inhibitor effective in inflammation and pain. J. Pharmacol. Exp. Ther. 2010, 334, 294–301. [Google Scholar] [CrossRef] [PubMed]
- Levi, R.M.; Khohlov, A.; Kopenkin, S.; Bart, B.; Ermolova, T.; Kantemirova, R.; Mazurov, V.; Bell, M.; Caldron, P.; Pillai, L.; et al. Efficacy and safety of flavocoxid, a novel therapeutic, compared with naproxen: A randomized multicenter controlled trial in subjects with osteoarthritisof the knee. Adv. Ther. 2010, 27, 731–742. [Google Scholar] [CrossRef] [PubMed]
- Saura, P.; Maréchal, J.-D.; Masgrau, L.; Lluch, J.M.; González-Lafont, A. Computational insight into the catalytic implication of head/tail-first orientation of arachidonic acid in human 5-lipoxygenase: Consequences for the positional specificity of oxygenation. Phys. Chem. Chem. Phys. 2016, 18, 23017–23035. [Google Scholar] [CrossRef] [PubMed]
- Lü, J.M.; Nurko, J.; Weakley, S.M.; Jiang, J.; Kougias, P.; Lin, P.H.; Yao, Q.; Chen, C. Molecular mechanisms and clinical applications of nordihydroguaiaretic acid (NDGA) and its derivatives: An update. Med. Sci. Monit. 2010, 16, RA93–R1001. [Google Scholar] [PubMed]
- Lapenna, D.; Ciofani, G.; Pierdomenico, S.D.; Neri, M.; Cuccurullo, C.; Giamberardino, M.A.; Cuccurullo, F. Inhibitory activity of salicylic acid on lipoxygenase-dependent lipid peroxidation. Biochim. Biophys. Acta 2009, 1790, 25–30. [Google Scholar] [CrossRef] [PubMed]
- Tehran, M.B.; Emami, S.; Asadi, M.; Saeedi, M.; Mirzahekmati, M.; Ebrahimi, S.M.; Mohammad, M.; Nadri, H.; Moradi, A.; Moghadam, F.H.; et al. Imidazo[2,1-b]thiazole derivatives as new inhibitors of15-lipoxygenase. Eur. J. Med. Chem. 2014, 87, 759–764. [Google Scholar] [CrossRef] [PubMed]
- Sharma, R.N.; Xavier, F.P.; Vasu, K.K.; Chaturvedi, S.C.; Pancholi, S.S. Synthesis of 4-benzyl-1,3-thiazole derivatives as potential anti-inflammatory agents: An analogue-based drug design approach. J. Enzyme Inhib. Med. Chem. 2009, 24, 890–897. [Google Scholar] [CrossRef] [PubMed]
- Suh, J.; Yum, E.K.; Cheon, H.G.; Cho, Y.S. Synthesis and Biological Evaluation of N-aryl-4-aryl-1,3-Thiazole-2-Amine Derivatives as Direct 5-Lipoxygenase Inhibitors. Chem. Biol. Drug Des. 2012, 80, 89–98. [Google Scholar] [CrossRef] [PubMed]
- Assadieskandar, A.; Amini, M.; Salehi, M.; Sadeghian, H.; Alimardani, M.; Sakhteman, A.; Nadri, H.; Shafiee, A. Synthesis and SAR study of 4,5-diaryl-1H-imidazole-2(3H)-thione derivatives, as potent 15-lipoxygenase inhibitors. Bioorg. Med. Chem. 2012, 20, 7160–7166. [Google Scholar] [CrossRef] [PubMed]
- Aslam, M.; Anis, I.; Afza, N.; Iqbal, L.; Iqbal, S.; Hussain, A.; Mehmood, R.; Hussain, M.T.; Khalid, M.; Nawaz, H. Biological evaluation of potent antioxidant, lipoxygenase inhibitor and antibacterial: Acomparative study. J. Saudi Chem. Soc. 2016, 20, 45–48. [Google Scholar] [CrossRef]
- Sadeghian, H.; Jabbari, A. 15-LOX inhibitors: A patent erview. Expert Opin. Ther. Patents 2016, 26, 65–88. [Google Scholar] [CrossRef] [PubMed]
- Duque, A.L.; Pinto, M.D.C.; Macias, P. Lipoxygenase inhibition by red wine phenolics compounds. J. Food Biochem. 2011, 35, 542–555. [Google Scholar] [CrossRef]
- Grzegorz, A.; Cholewiński, G.; Garwolińska, D.; Olejnik, A.; JanuszRachoń, J.; Chojnacki, J. The synthesis and structure of a potential immunosuppressant: N-mycophenoyl malonic acid dimethyl ester. J. Mol. Struct. 2018, 1151, 218–222. [Google Scholar]
- Iwaszkiewicz-Grzes, D.; Cholewinski, G.; Kot-Wasikasik, A.; Trzonkowski, P.; Dzierzbicka, K. Synthesis and biological activity of mycophenolic acid-amino acid derivatives. Eur. J. Med. Chem. 2013, 69, 863–871. [Google Scholar] [CrossRef] [PubMed]
- Warren, W.; Epinette, M.D.; Colleen, M.; Parker, M.D.; Linn, E.; Jones, M.D.; Mary, C.; Greist, M.D. Mycophenolic acid for psoriasis: A review of pharmacology, long-term efficacy, and safety. J. Am. Acad. Dermatol. 1987, 17, 962–971. [Google Scholar]
- Marinari, R.; Fleischmajer, R.; Schragger, A.H.; Rosenthal, A.L. Mycophenolic acid in the treatment of psoriasis: Long-term administration. Arch. Dermatol. 1977, 113, 930–932. [Google Scholar] [CrossRef] [PubMed]
- Akhyani, M.; Toosi, S.; Robati, R.M. Treatment of chronic plaque type psoriasis with systemic mycophenolate mofetil. Acta Med. Iran. 2006, 44, 383–386. [Google Scholar]
- Fallah, A.S.; Waalboer, S.R.; Nijsten, T.; Neumann, H.A.; Thio, B. Enteric-coated mycophenolate sodium in psoriasis vulgaris: An open pilot study. J. Dermatol. Treat. 2014, 25, 46–49. [Google Scholar] [CrossRef] [PubMed]
- ChemNavigator. Available online: http://www. Chemnavigator.com/cnc/products/iRL.asp (accessed on 5 May 2018).
- Morphy, R.; Rankovic, Z. Designed multiple ligands. An emerging drug discovery paradigm. J. Med. Chem. 2005, 48, 6523–6543. [Google Scholar] [CrossRef] [PubMed]
- Kapetanovic, I.M. Computer-aided drug discovery and development (CADDD): In silico-chemico-biological approach. Chem. Biol. Interact. 2008, 171, 165–176. [Google Scholar] [CrossRef] [PubMed]
- Ganou, C.A.; Eleftheriou, P.T.; Theodosis-Nobelos, P.; Geronikaki, A.; Lialiaris, T.; Rekka, E.A. Docking analysis targeted to the whole enzyme: An application to the prediction of inhibition of PTP1B by thiomorpholine and thiazolyl derivatives SAR QSAR Environ. Res. 2018, 29, 133–149. [Google Scholar] [CrossRef] [PubMed]
- OpenTox. Available online: http://www.opentox.org/toxicity-prediction (accessed on 5 May 2018).
- ToxPredict. Available online: https://apps.ideaconsult.net/ToxPredict (accessed on 11 May 2018).
- PROTOX. Available online: http://tox.charite.de/tox (accessed on 11 May 2018).
- GHS-Unece. Available online: http://www.unece.org/trans/danger/publi/ghs/ghs_welcome_e.html (accessed on 11 May 2018).
- Neau, D.B.; Gilbert, N.C.; Bartlett, S.G.; Boeglin, W.; Brash, A.R.; Newcomer, M.E. The 1.85 Å structure of an 8R-Lipoxygenase suggests a general model for lipoxygenase product specificity. Biochemistry 2009, 48, 7906–7915. [Google Scholar] [CrossRef] [PubMed]
- Gilbert, N.C.; Bartlett, S.G.; Waight, M.T.; Neau, D.B.; Boeglin, W.E.; Brash, A.R.; Newcomer, M.E. The structure of human 5-lipoxygenase. Science 2011, 331, 217–219. [Google Scholar] [CrossRef] [PubMed]
- Taraporewala, I.B.; Kauffman, J.M. Synthesis and structure-activity relationships of anti-inflammatory 9,10-dihydro-9-oxo-2-acridinealkanoic acids and 4-(2-carboxyphenyl)aminobenzenealkanoic acids. J. Pharm. Sci. 1990, 79, 173–178. [Google Scholar] [CrossRef] [PubMed]
- Somvanshi, R.K.; Singh, A.K.; Saxena, M.; Mishra, B.; Dey, S. Development of novel peptide inhibitor of lipoxygenase based on biochemical and BIAcore evidences. Biochim. Biophys. Acta 2008, 1784, 1812–1817. [Google Scholar] [CrossRef] [PubMed]
- Eleftheriou, P.; Petrou, A.; Geronikaki, A.; Liaras, K.; Dirnali, S.; Anna, M. Prediction of enzyme inhibition and mode of inhibitory action based on calculation of distances between hydrogen bond donor/acceptor groups of the molecule and docking analysis: An application on the discovery of novel effective PTP1B inhibitors. SAR QSAR Environ. Res. 2015, 26, 557–576. [Google Scholar] [CrossRef] [PubMed]
- Feinstein, W.P.; Brylinski, M. Calculating an optimal box size for ligand docking and virtual screening against experimental and predicted binding pockets. J. Cheminform. 2015, 7, 18. [Google Scholar] [CrossRef] [PubMed]
- Minor, W.; Steczko, J.; Stec, B.; Otwinowski, Z.; Bolin, J.T.; Walter, R.; Axelrod, B. Crystal structure of soybean lipoxygenase L-1 at 1.4 Å resolution. Biochemistry 1996, 35, 10687–10701. [Google Scholar] [CrossRef] [PubMed]
- Skrzypczak-Jankun, E.; Zhou, K.; Jankun, J. Inhibition of lipoxygenase by (−)-epigallocatechin gallate: X-ray analysis at 2.1 A reveals degradation of EGCG and shows soybean LOX-3 complex with EGC instead. Int. J. Mol. Med. 2003, 12, 415–420. [Google Scholar] [CrossRef] [PubMed]
- Thomsen, R.; Christensen, M.H. MolDock: A new technique for high-accuracy molecular docking. J. Med. Chem. 2006, 49, 3315–3321. [Google Scholar] [CrossRef] [PubMed]
- Gilbert, N.C.; Rui, Z.; Neau, D.B.; Waight, M.T. Bartlett, S.G.; Boeglin, W.E.; Brash, A.R.; Newcomer, M.E. Conversion of human 5-lipoxygenase to a 15-lipoxygenase by a point mutation to mimic phosphorylation at Serine-663. FASEB J. 2012, 26, 3222–3229. [Google Scholar] [CrossRef] [PubMed]
- Gillmor, S.A.; Villasenor, A.; Fletterick, R.; Sigal, E.; Browner, M.F. The structure of mammalian 15-lipoxygenase reveals similarity to the lipases and the determinants of substrate specificity. Nat. Struct. Biol. 1997, 4, 1003–1009. [Google Scholar] [CrossRef] [PubMed]
- PASS Online. Available online: http://www.way2drug.com/passonline (accessed on 10 May 2018).
- Bikadi, Z.; Hazai, E. Application of the PM6 semi-empirical method to modeling proteins enhances docking accuracy of AutoDock. J. Cheminf. 2009, 1, 15. [Google Scholar] [CrossRef] [PubMed]
- Sashidhara, K.V.; Avula, S.R.; Doharey, P.K.; Singh, L.R.; Balaramnavar, V.M.; Gupta, J.; Misra-Bhattacharya, S.; Rathaur, S.; Saxena, A.K.; Saxena, J.K. Designing, synthesis of selective and high-affinity chalcone-benzothiazole hybrids as Brugia malayi thymidylate kinase inhibitors: In vitro validation and docking studies. Eur. J. Med. Chem. 2015, 103, 418–428. [Google Scholar] [CrossRef] [PubMed]
- Saxena, A.; Balaramnavar, V.M.; Hohlfeld, T.; Saxena, A.K. Drug/drug interaction of common NSAIDs with antiplatelet effect of aspirin in human platelets. Eur. J. Pharmacol. 2013, 721, 215–224. [Google Scholar] [CrossRef] [PubMed]
- Sunohara, K.; Mitsuhashi, S.; Shigetomi, K.; Ubukata, M. Discovery of N-(2,3,5-triazoyl)mycophenolic amide and mycophenolic epoxyketone as novel inhibitors of human IMPDH. Bioorg. Med. Chem. Lett. 2013, 23, 5140–5144. [Google Scholar] [CrossRef] [PubMed]
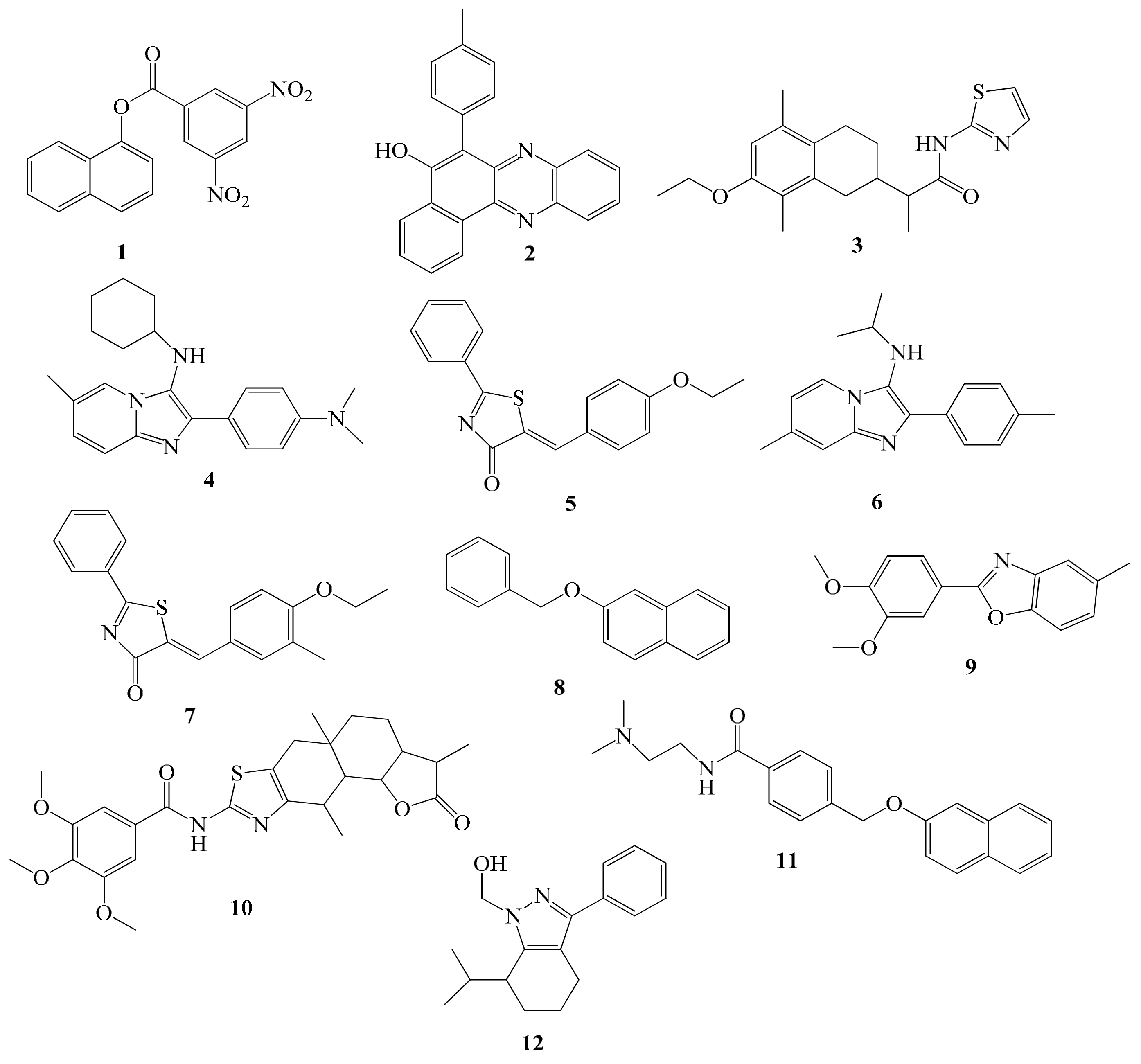





| Comp. | R1 | R2 | Comp. | R1 | R2 |
|---|---|---|---|---|---|
| 1 | -OH | 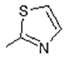 | 9 | -OH | 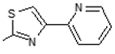 |
| 2 | -OH | 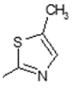 | 10 | -OH | 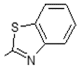 |
| 3 | -OH | 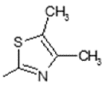 | 11 | -OH | 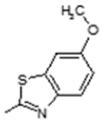 |
| 4 | -OH | 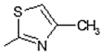 | 12 | -OH | 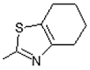 |
| 5 | -OH | 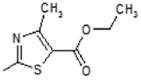 | 13 | -OH | 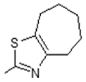 |
| 6 | -OH |  | 14 | -OH |  |
| 7 | -OH | 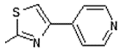 | 15 | -OCH3 |  |
| 8 | -OH | 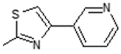 | 16 | -OCH3 | 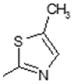 |
| No. | R2 | Predicted Activity | Pa |
|---|---|---|---|
| 1 |  | Systemic lupus erythematosus treatment | 0.905 |
| Lipoxygenase inhibitor | 0.846 | ||
| Transcription factor inhibitor | 0.768 | ||
| Immunosuppressant | 0.727 | ||
| Lipid peroxidase inhibitor | 0.682 | ||
| Antiulcerative | 0.674 | ||
| 2 | 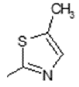 | Lipoxygenase inhibitor | 0.879 |
| Systemic lupus erythematosus treatment | 0.72 | ||
| Immunosuppressant | 0.726 | ||
| Antineoplastic | 0.696 | ||
| Antiinflammatory | 0.645 | ||
| Lipid peroxidase inhibitor… | 0.6 | ||
| 3 | 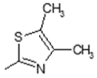 | Antiulcerative | 0.748 |
| Lipoxygenase inhibitor | 0.743 | ||
| Immunosuppressant | 0.72 | ||
| Systemic lupus erythematosus treatment | 0.662 | ||
| Antiinflammatory | 0.632 | ||
| Lipid peroxidase inhibitor | 0.601 | ||
| Antineoplastic… | 0.639 | ||
| 4 | 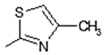 | Lipoxygenase inhibitor | 0.808 |
| Antiulcerative | 0.794 | ||
| Transcription factor inhibitor | 0.76 | ||
| Systemic lupus erythematosus treatment | 0.734 | ||
| Immunosuppressant | 0.706 | ||
| Lipid peroxidase inhibitor | 0.696 | ||
| 5 | 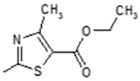 | Immunosuppressant | 0.745 |
| Lipoxygenase inhibitor | 0.716 | ||
| Antiulcerative | 0.689 | ||
| Systemic lupus erythematosus treatment | 0.631 | ||
| Antiinflammatory | 0.616 | ||
| Transcription factor inhibitor | 0.594 | ||
| Antineoplastic… | 0.581 | ||
| 6 | 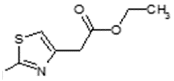 | Lipoxygenase inhibitor | 0.875 |
| Antiulcerative | 0.799 | ||
| Transcription factor inhibitor | 0.753 | ||
| Immunosuppressant | 0.731 | ||
| Antiinflammatory | 0.692 | ||
| Systemic lupus erythematosus treatment | 0.587 | ||
| Antineoplastic | 0.609 | ||
| 7 | 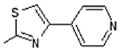 | Antiulcerative | 0.765 |
| Lipoxygenase inhibitor | 0.726 | ||
| Transcription factor inhibitor | 0.726 | ||
| Immunosuppressant | 0.716 | ||
| Systemic lupus erythematosus treatment | 0.699 | ||
| Mediator release inhibitor | 0.642 | ||
| 8 | 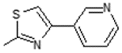 | Transcription factor inhibitor | 0.751 |
| Antiulcerative | 0.709 | ||
| Immunosuppressant | 0.7 | ||
| Systemic lupus erythematosus treatment | 0.689 | ||
| Mediator release inhibitor | 0.662 | ||
| Lipoxygenase inhibitor | 0.661 | ||
| 9 | 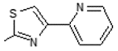 | Antiulcerative | 0.825 |
| Lipoxygenase inhibitor | 0.716 | ||
| Transcription factor inhibitor | 0.7 | ||
| Immunosuppressant | 0.688 | ||
| Systemic lupus erythematosus treatment | 0.615 | ||
| Mediator release inhibitor | 0.609 | ||
| 10 | 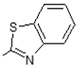 | Lipoxygenase inhibitor | 0.774 |
| Systemic lupus erythematosus treatment | 0.746 | ||
| Lipid peroxidase inhibitor | 0.722 | ||
| Immunosuppressant | 0.7 | ||
| Antiulcerative | 0.679 | ||
| Transcription factor inhibitor | 0.673 | ||
| 11 | 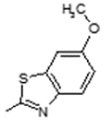 | Lipoxygenase inhibitor | 0.809 |
| Lipid peroxidase inhibitor | 0.698 | ||
| Systemic lupus erythematosus treatment | 0.688 | ||
| Immunosuppressant | 0.687 | ||
| Antineoplastic | 0.674 | ||
| Antiulcerative | 0.63 | ||
| Apoptosis agonist... | 0.615 | ||
| 12 | 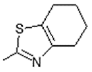 | Antiulcerative | 0.748 |
| Lipoxygenase inhibitor | 0.743 | ||
| Immunosuppressant | 0.72 | ||
| Systemic lupus erythematosus treatment | 0.662 | ||
| Antiinflammatory | 0.632 | ||
| Lipid peroxidase inhibitor | 0.601 | ||
| Antineoplastic... | 0.639 | ||
| 13 | 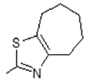 | Antiulcerative | 0.748 |
| Lipoxygenase inhibitor | 0.743 | ||
| Immunosuppressant | 0.72 | ||
| Systemic lupus erythematosus treatment | 0.662 | ||
| Antiinflammatory | 0.632 | ||
| Lipid peroxidase inhibitor | 0.601 | ||
| Antineoplastic... | 0.639 | ||
| 14 | 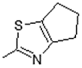 | Antiulcerative | 0.748 |
| Lipoxygenase inhibitor | 0.743 | ||
| Immunosuppressant | 0.72 | ||
| Systemic lupus erythematosus treatment | 0.662 | ||
| Antiinflammatory | 0.632 | ||
| Lipid peroxidase inhibitor | 0,601 | ||
| Antineoplastic... | 0.639 | ||
| 15 | 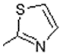 | Systemic lupus erythematosus treatment | 0.791 |
| Immunosuppressant | 0.715 | ||
| Lipoxygenase inhibitor | 0.697 | ||
| Transcription factor inhibitor | 0.699 | ||
| Antiinflammatory | 0.656 | ||
| Antineoplastic | 0.664 | ||
| Antiulcerative | 0.625 | ||
| 16 | 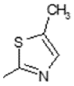 | Lipoxygenase inhibitor | 0.732 |
| Immunosuppressant | 0.714 | ||
| Antineoplastic | 0.692 | ||
| Antiinflammatory | 0.625 | ||
| Systemic lupus erythematosus treatment | 0.583 | ||
| Apoptosis agonist | 0.576 | ||
| Antiulcerative | 0.501 |
| No. | DDStox Carcinogenic Potency DBS MultiCellCall | DDStox Carcinogenic Potency DBS Mouse | DDStox Carcinogenic Potency DBS Rat | DDStox Carcinogenic Potency DBS SingleCellcall | DDStox Carcinogenic Potency DBS Hamster | DSSTox Carcinogenic Potency DBS Mutagenicity | Kazius-Bursi Salmonella Mutagenicity | FDA v3b Maximum Recommended Daily Dose mmol |
|---|---|---|---|---|---|---|---|---|
| Non Carcinogen Confidence: | Non Carcinogen Confidence: | Non Carcinogen Confidence: | Non Carcinogen Confidence: | Non Carcinogen Confidence: | Non Mutagenic Confidence: | Non Mutagenic Confidence: | 0.162 mmol Confidence: | |
| 1 | 0.084 | 0.132 | 0.082 | 0.0296 | 0.155 | 0.0345 | 0.0774 | 0.165 |
| 2 | 0.058 | 0.241 | 0.0541 | 0.0035 | 0.099 | 0.0801 | 0.0756 | 0.0735 mmol |
| 3 | 0.139 | 0.252 | 0.0282 | 0.0108 | 0.084 | 0.0374 | 0.0791 | 0.0641 mmol |
| 4 | 0.057 | 0.223 | 0.0342 | 0.0156 | 0.082 | 0.0567 | 0.0784 | 0.0724 mmol |
| 5 | 0.120 | 0.253 | 0.0274 | 0.0245 | 0.096 | 0.0357 | 0.0698 | 0.188 mmol |
| 6 | 0.086 | 0.142 | 0.0670 | 0.0276 | 0.081 | 0.0340 | 0.0768 | 0.0723 mmol |
| 7 | 0.056 | 0.250 | 0.0567 | 0.0145 | 0.099 | 0.0786 | 0.0712 | 0.145 mmol |
| 8 | 0.050 | 0.261 | 0.0022 | 0.0111 | 0.098 | 0.1051 | 0.0062 | 0.109 mmol |
| 9 | 0.059 | 0.254 | 0.0459 | 0.0179 | 0.087 | 0.1131 | 0.0566 | 0.133 mmol |
| 10 | 0.122 | 0.199 | 0.0677 | 0.0186 | 0.097 | 0.1291 | 0.0734 | 0.107 mmol |
| 11 | 0.045 | 0.252 | 0.0141 | 0.0187 | 0.217 | 0.0146 | 0.0112 | 0.117 mmol |
| 12 | 0.078 | 0.234 | 0.0781 | 0.0234 | 0.239 | 0.0358 | 0.0689 | 0.189 mmol |
| 13 | 0.109 | 0.287 | 0.0678 | 0.0189 | 0.221 | 0.0347 | 0.0765 | 0.179 mmol |
| 14 | 0.167 | 0.178 | 0.0443 | 0.0179 | 0.187 | 0.0156 | 0.0651 | 0.123 mmol |
| 15 | 0.058 | 0.257 | 0.0448 | 0.0177 | 0.084 | 0.1132 | 0.0559 | 0.131 mmol |
| 16 | 0.132 | 0.187 | 0.0657 | 0.0184 | 0.087 | 0.1275 | 0.0744 | 0.110 mmol |
| 17 | 0.035 | 0.241 | 0.0124 | 0.0182 | 0.213 | 0.0191 | 0.0123 | 0.113 mmol |
| Comp. | Predicted LD50 | Predicted Toxicity Class | Hepatotoxicity | Carcinogenicity | Mutagenicity | Cytotoxicity |
|---|---|---|---|---|---|---|
| 1 | 500 mg/kg | 4 | Inactive 0.64 | Inactive 0.55 | Inactive 0.63 | Inactive 0.63 |
| 2 | 500 mg/kg | 4 | Inactive 0.64 | Inactive 0.54 | Inactive 0.63 | Inactive 0.63 |
| 3 | 1000 mg/kg | 4 | Inactive 0.64 | Inactive 0.55 | Inactive 0.63 | Inactive 0.63 |
| 4 | 1000 mg/kg | 4 | Inactive 0.64 | Inactive 0.55 | Inactive 0.63 | Inactive 0.63 |
| 5 | 300 mg/kg | 3 | Inactive 0.64 | Inactive 0.55 | Inactive 0.63 | Inactive 0.63 |
| 6 | 500 mg/kg | 4 | Inactive 0.64 | Inactive 0.54 | Inactive 0.63 | Inactive 0.63 |
| 7 | 1000 mg/kg | 4 | Inactive 0.64 | Inactive 0.55 | Inactive 0.63 | Inactive 0.63 |
| 8 | 1000 mg/kg | 4 | Inactive 0.64 | Inactive 0.55 | Inactive 0.63 | Inactive 0.63 |
| 9 | 1000 mg/kg | 4 | Inactive 0.64 | Inactive 0.55 | Inactive 0.63 | Inactive 0.63 |
| 10 | 500 mg/kg | 4 | Inactive 0.64 | Inactive 0.54 | Inactive 0.63 | Inactive 0.63 |
| 11 | 1000 mg/kg | 4 | Inactive 0.64 | Inactive 0.55 | Inactive 0.63 | Inactive 0.63 |
| 12 | 500 mg/kg | 4 | Inactive 0.64 | Inactive 0.55 | Inactive 0.63 | Inactive 0.63 |
| 13 | 300 mg/kg | 3 | Inactive 0.64 | Inactive 0.55 | Inactive 0.63 | Inactive 0.63 |
| 14 | 300 mg/kg | 3 | Inactive 0.64 | Inactive 0.55 | Inactive 0.63 | Inactive 0.63 |
| 15 | 500 mg/kg | 4 | Inactive 0.64 | Inactive 0.55 | Inactive 0.63 | Inactive 0.63 |
| 16 | 500 mg/kg | 4 | Inactive 0.64 | Inactive 0.55 | Inactive 0.63 | Inactive 0.63 |
| Comp. | Free Binding Energy (kcal/mole) | Docking Scores | IC50 (µM) | pIC50 | Length | |||
|---|---|---|---|---|---|---|---|---|
| Rat 15-LOX 1LOX | Human 5-LOX 3V99 | 1JNQ | ||||||
| Box 30 | Box 35 | Box 30 | Moldock | Rerank | ||||
| 1 | −5.24 | −117.175 | −87.9702 | 26.25 | −1.419 | 14.82 | ||
| 2 | −6.68 | −128.445 | −72.0312 | 22.50 | −1.352 | 15.72 | ||
| 3 | −126.37 | −75.89 | 43.75 | −1.641 | ||||
| 4 | −7.50 | −128.45 | −82.031 | 31.25 | −1.495 | 16.29 | ||
| 5 | −7.76 | −6.43 | −136.803 | −83.9406 | 22.50 | −3.343 | 18.79 | |
| 6 | −116.026 | −57.1526 | 30.00 | −1.477 | 15.23 | |||
| 7 | −85.017 | −61.202 | 78.75 | −1.896 | ||||
| 8 | −9.01 | −119.09 | −90.416 | 90.00 | −1.954 | 18.22 | ||
| 9 | −9.27 | −8.29 | −10.00 | −142.373 | −105.8901 | 2.50 | −0.398 | 17.71 |
| 10 | −61.93 | −71.647 | 90.00 | −1.954 | ||||
| 11 | −9.25 | −6.71 | −7.49 | −62.19 | −59.631 | 156.25 | −2.194 | 19.61 |
| 12 | 61.25 | −1.787 | ||||||
| 13 | −142.186 | −89.631 | 25.00 | −1.398 | 16.90 | |||
| 14 | −136.03 | −77.1526 | 31.25 | −1.498 | ||||
| 15 | −6.27 | −147.28 | −47.554 | 96.25 | −1.983 | 15.30 | ||
| 16 | −152.14 | −80.727 | 90.00 | −1.954 | ||||
| OPPA | −8.05 | −8.09 | ||||||
| A.A | −6.46 | |||||||
| Free Binding Energy (Kcal/mole) | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Comp. | Soybean sLOX 1YGE | Human 5-LOX 3O8Y | IC50 | ||||||||
| Center: x: 26.37, y: 42.69, z: 12.36 | Center: x: 20.37, y: 46.69, z: 8.36 | Center: x: 8.59, y: 22.65, z: −1.02 | Center: x: −3.57, y: 17.66, z: 8.36 | Center: x: −12.50, y: 75.50, z: 0.50 | |||||||
| Box 20 | Box 30 | Box 35 | Box 37 × 27 × 22 | Box 36 × 20 × 20 | Box 20 | Box 30 | Box 35 | Box 20 × 25 × 20 | Box 35 × 25 × 27 | ||
| 2 | −7.11 | −7.42 | 22.50 | ||||||||
| 4 | −7.00 | 31.25 | |||||||||
| 5 | +90.68 | −8.20 | −5.84 | 22.50 | |||||||
| 8 | −7.83 | 90.00 | |||||||||
| 9 | +90.14 | −6.34 | −8.26 | −5.68 | −1.08 | −8.28 | −7.77 | −8.25 | −5.49 | −6.09 | 2.50 |
| 11 | −95.16 | −6.41 | −6.24 | −6.01 | +5.37 | −8.09 | −7.77 | −6.51 | −5.11 | −6.63 | 156.25 |
| 13 | −7.75 | −7.57 | −6.75 | 25.00 | |||||||
| 14 | −7.09 | −8.51 | 31.25 | ||||||||
| 15 | −7.32 | 96.25 | |||||||||
| NDHDA | −7.00 | ||||||||||
| A.A | −6.63 | −5.04 | −4.37 | −3.31 | −6.72 | −4.58 | −4.97 | ||||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tsolaki, E.; Eleftheriou, P.; Kartsev, V.; Geronikaki, A.; Saxena, A.K. Application of Docking Analysis in the Prediction and Biological Evaluation of the Lipoxygenase Inhibitory Action of Thiazolyl Derivatives of Mycophenolic Acid. Molecules 2018, 23, 1621. https://doi.org/10.3390/molecules23071621
Tsolaki E, Eleftheriou P, Kartsev V, Geronikaki A, Saxena AK. Application of Docking Analysis in the Prediction and Biological Evaluation of the Lipoxygenase Inhibitory Action of Thiazolyl Derivatives of Mycophenolic Acid. Molecules. 2018; 23(7):1621. https://doi.org/10.3390/molecules23071621
Chicago/Turabian StyleTsolaki, Evangelia, Phaedra Eleftheriou, Victor Kartsev, Athina Geronikaki, and Anil K. Saxena. 2018. "Application of Docking Analysis in the Prediction and Biological Evaluation of the Lipoxygenase Inhibitory Action of Thiazolyl Derivatives of Mycophenolic Acid" Molecules 23, no. 7: 1621. https://doi.org/10.3390/molecules23071621
APA StyleTsolaki, E., Eleftheriou, P., Kartsev, V., Geronikaki, A., & Saxena, A. K. (2018). Application of Docking Analysis in the Prediction and Biological Evaluation of the Lipoxygenase Inhibitory Action of Thiazolyl Derivatives of Mycophenolic Acid. Molecules, 23(7), 1621. https://doi.org/10.3390/molecules23071621