Study on the Polar Extracts of Dendrobium nobile, D. officinale, D. loddigesii, and Flickingeria fimbriata: Metabolite Identification, Content Evaluation, and Bioactivity Assay
Abstract
:1. Introduction
2. Results and Discussion
2.1. Water-Soluble Metabolite Structure
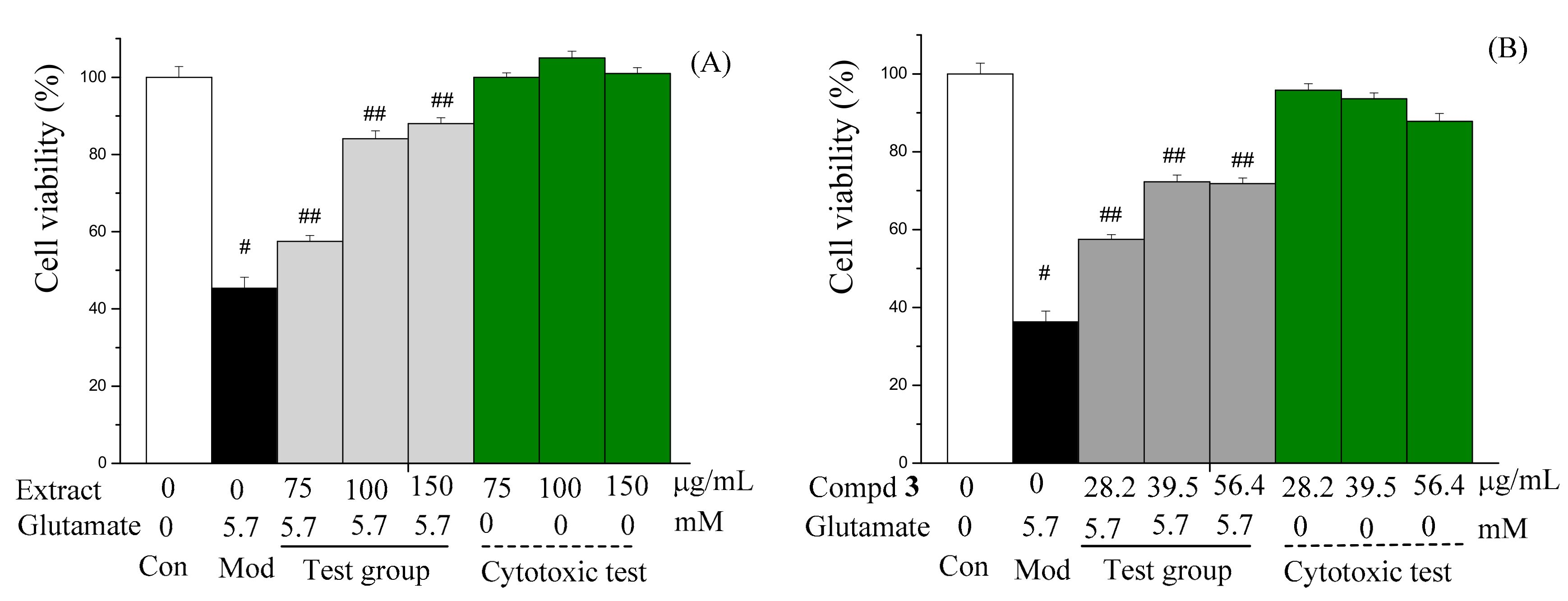
2.2. Content and Bioactivity
3. Materials and Methods
3.1. General Experimental Procedures
3.2. Plant Sample
3.3. Extraction
3.4. Polar Extract Preparation
3.5. qNMR Analysis
3.6. Water-Soluble Metabolite Isolation
3.7. Spectral Data
3.8. Biological Assay
Supplementary Materials
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Hossain, M.M. Therapeutic orchids: Traditional uses and recent advances—An overview. Fitoterapia 2011, 82, 102–140. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.B.; Feng, J.; Zhao, X.B.; Guo, K.X.; Peng, X.X.; Wu, W.J. Analysis on potential of TCM cultural tourism product of Dendrobium officinale exploited in Hunan Province. Chin. J. Inf. Tradit. Chin. Med. 2014, 21, 8–9. [Google Scholar]
- China Pharmacopoeia Committee. Chinese Pharmacopoeia; China Medical Science Press: Beijing, China, 2010; pp. 85–86. [Google Scholar]
- Zhang, J.P.; Zheng, X.L.; Hong, J.Z.; Chen, J.C.; Zheng, Y.Y.; Xin, J.Z.; Wang, Q.Y.; Zhu, K.D.; Wang, X.N.; Shi, H. Dendrobium compound in treating 90 case of type 2 diabetes memtus. J. Fujian. Univ. TCM 2011, 21, 6–10. [Google Scholar]
- Qu, X.Y.; Wu, Y.X.; Zhang, J.L. Research advances on chemical constituents and pharmacological activities of Dendrobium. Tianjin Agric. Sci. 2015, 21, 19–32. [Google Scholar]
- Chen, Y.G.; Wang, H.Y. Advance on the chemical and pharmacological studies on plants of Flickingeria genus. Lishizhen Med. Mater. Med. Res. 2005, 16, 725–727. [Google Scholar]
- Xu, J.; Han, Q.B.; Li, S.L.; Chen, X.J.; Wang, X.N.; Zhao, Z.Z.; Chen, H.B. Chemistry, bioactivity and quality control of Dendrobium, a commonly used tonic herb in traditional Chinese medicine. Phytochem. Rev. 2013, 12, 341–367. [Google Scholar] [CrossRef]
- Yang, L.; Wang, Z.; Xu, L. Simultaneous determination of phenols (bibenzyl, phenanthrene, and fluorenone) in Dendrobium species by high-performance liquid chromatography with diode array detection. J. Chromatogr. A 2006, 1104, 230–237. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.C.; Ren, J.; Zhu, S.; Xie, Z.S.; Ye, J.H.; Wei, G. Analysis of HPLC characteristic spectrum of Hera Flickingeriae. Trad. Chin. Drug Res. Clin. Pharmcol. 2015, 26, 658–664. [Google Scholar]
- Lam, Y.; Ng, T.B.; Yao, R.M.; Shi, J.; Xu, K.; Sze, S.C.; Zhang, K.Y. Evaluation of chemical constituents and important mechanism of pharmacological biology in Dendrobium plants. Evid. Based Complement. Altern. Med. 2015. [Google Scholar] [CrossRef] [PubMed]
- Li, M.Y.; Deng, W.; Tang, X.; Yang, W.; Yan, T.; Yan, T.L.; Gu, X. Research status on functional ingredients and health care food of Dendrobium nobile Lindl. J. Food Sci. Biotechnol. 2014, 33, 1233–1238. [Google Scholar]
- Chen, S.H.; Yan, M.Q.; Lü, G.Y.; Liu, X. Development of Dendrobium officinale Kimura et Migo releted health foods. Chin. Pharm. 2013, 48, 1625–1628. [Google Scholar]
- Li, H.; Zhao, J.J.; Chen, J.L.; Zhu, L.P.; Wang, D.M.; Jiang, L.; Yang, D.P.; Zhao, Z.M. Diterpenoids from aerial parts of Flickingeria fimbriata and their nuclear factor-kappaB inhibitory activities. Phytochemistry 2015, 117, 400–409. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.Y.; Cheng, C.W.; Tien, Y.C.; Kuo, C.L.; Lo, S.F.; Peng, W.H. Anti-oxidative and hepatoprotective activities of Dendrobium Tosaense and Ephemerantha Fimbriata in carbon tetrachloride-induced acute liver injury. J. Chin. Med. 2011, 22, 47–63. [Google Scholar]
- Wang, H.; Zhang, J.X.; Wu, K.L.; Zeng, S.J.; Duan, J. Comparative analysis of the biological characteristics main components of different Dendrobii. Guangdong Agric. Sci. 2012, 39, 44–59. [Google Scholar]
- Huang, M.; Cai, T.; Liu, Y. Effects of polysaccharides from Dendrobium candidum on white blood cells and lymph cell moving inhibition factor of mice. Nat. Prod. Res. Dev. 1996, 8, 39–41. [Google Scholar]
- Yuan, H.Q.; Liang, C.Y.; Liang, J.; Hou, S.Z.; Huang, S.; Lai, X.P. Protection of Dendrobium Officinale against acute alcoholic liver injury in mice. J. Jinan Univ. 2016, 37, 384–388. [Google Scholar]
- Zhang, W.; Wu, Q.; Lu, Y.L.; Gong, Q.H.; Zhang, F.; Shi, J.S. Protective effects of Dendrobium nobile Lindl. alkaloids on amyloid beta (25–35)-induced neuronal injury. Neural Regen. Res. 2017, 12, 1131–1136. [Google Scholar] [PubMed]
- Long, Y.; Wei, X.Y.; Zhan, Y.J.; Li, X.C.; Xu, C.L.; Xu, Q. In-vitro experimental study on the extract of Dendrobium nobile Lindl. in counteracting cataract in rats. J. Guangzhou Univ. Tradit. Chin. Med. 2008, 25, 345–349. [Google Scholar]
- Li, C.Y.; Lu, Y.; Chen, Y.; Zheng, J.W.; Wang, J. Chemical components of Dendrobium loddigesii. Acta Sci. Nat. Univ. Sunyatseni 2013, 52, 73–76. [Google Scholar]
- Lu, Y.; Kuang, M.; Hu, G.P.; Wu, R.B.; Wang, J.; Liu, L.; Lin, Y.C. Loddigesiinols G–J: α-glucosidase inhibitors from Dendrobium loddigesii. Molecules 2014, 19, 8544–8555. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.C.; Wu, L.G.; Ko, F.N.; Teng, C.M. Antiplatelet aggregation principles of Dendrobium loddigesii. J. Nat. Prod. 1994, 57, 1271–1274. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.L.; Zhao, Z.M.; Xue, X.; Tang, G.H.; Zhu, L.P.; Yang, D.P.; Jiang, L. Bioactive norditerpenoids from Flickingeria fimbriata. RSC Adv. 2014, 4, 14447–14456. [Google Scholar] [CrossRef]
- Wang, C.Z.; Yu, D.Q. Lignan and acetylenic glycosides from Aster auriculatus. Phytochemistry 1998, 48, 711–717. [Google Scholar] [CrossRef]
- Leander, K.; Luning, B. Studies on Orchidaceae alkaloids. VIII. Animidazolium salt from Dendrobium anosmum and Dendrobium parishii. Tetrahedron Lett. 1968, 8, 905–908. [Google Scholar] [CrossRef]
- D’Abrosca, B.; Scognamiglio, M.; Fiumano, V.; Esposito, A.; Choi, Y.H.; Verpoorte, R.; Fiorentino, A. Plant bioassay to assess the effects of allelochemicals on the metabolome of the target species Aegilops geniculata by an NMR-based approach. Phytochemistry 2013, 93, 27–40. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Liu, G.M.; Ren, Y.L.; He, H.P.; Hao, X.J. Chemical constituents from Glochidion puberum. Nat. Prod. Res. Dev. 2008, 20, 447–449. [Google Scholar]
- Goux, W.J. Complex isomerization of ketoses: A 13C-NMR study of the base- catalyzed ring-opening and ring-closing rates of D-fructose isomers in aqueous solution. J. Am. Chem. Soc. 1985, 107, 4320–4327. [Google Scholar] [CrossRef]
- Kitajima, J.; Ishikawa, T.; Tanaka, Y. Water-soluble constituents of Fennel. I. alkyl glycosides. Chem. Pharm. Bull. 1998, 46, 1643–1646. [Google Scholar] [CrossRef]
- Klyne, W. The configuration of the anomeric carbon atoms in some cardiac glycosides. Biochem. J. 1950, 47, Xii–Xiii. [Google Scholar]
- Devkota, H.P.; Watanabe, M.; Watanabe, T.; Yahara, S. Phenolic compounds from the aerial parts of Diplomorpha canescens. Chem. Pharm. Bull. 2012, 60, 554–556. [Google Scholar] [CrossRef] [PubMed]
- Tapiolas, D.M.; Raina, J.B.; Lutz, A.; Willis, B.L.; Motti, C.A. Direct measurement of dimethylsulfoniopropionate (DMSP) in reef-building corals using quantitative nuclear magnetic resonance (qNMR) spectroscopy. J. Exp. Mar. Biol. Ecol. 2013, 443, 85–89. [Google Scholar] [CrossRef]
- Li, C.Y.; Tsai, S.I.; Damu, A.G.; Wu, T.S. A rapid and simple determination of protoberberine alkaloids in Rhizoma Coptidis by 1H NMR and its application for quality control of commercial prescriptions. J. Pharm. Biomed. Anal. 2009, 49, 1272–1276. [Google Scholar] [CrossRef] [PubMed]
- Frank, O.; Kreissl, J.K.; Daschner, A.; Hofmann, T. Accurate determination of reference materials and natural isolates by means of quantitative 1H-NMR spectroscopy. J. Agric. Food Chem. 2014, 62, 2506–2515. [Google Scholar] [CrossRef] [PubMed]
- Akoka, S.; Trierweiler, M. Improvement of the ERETIC method by digital synthesis of the signal and addition of a broadband antenna inside the NMR probe. Instrum. Sci. Technol. 2002, 30, 21–29. [Google Scholar] [CrossRef]
- Fei, J.D.; Chen, L.; Ding, G.; Zhang, H.W.; Li, R.T.; Yu, C.Y.; Zou, Z.M. Isolation, identification and quantitative analysis of vitexin 2″-O-β-d-glucopyranoside in the whole herb of Flickingeria fimbriata. Chin. J. Exp. Tradit. Med. Form. 2013, 19, 64–67. [Google Scholar]
- Shimada, K.; Fujikawa, K.; Yahara, K.; Nakamura, T. Antioxidative properties of xanthin on autoxidation of soybean oil in cyclodextrin emulsion. J. Agric. Food Chem. 1992, 40, 945–948. [Google Scholar] [CrossRef]
- Sun, Y.; Yang, K.; Cao, Q.; Sun, J.; Xia, Y.; Wang, Y.; Li, W.; Ma, C.; Liu, S. Homogenate-assisted Vacuum-powered Bubble Extraction of Moso Bamboo Flavonoids for On-line Scavenging Free Radical Capacity Analysis. Molecules 2017, 22, 1156. [Google Scholar] [CrossRef] [PubMed]
- Harasit, K.M.; Tapas, M.; Ambikesh, M. Kinetics of the basic hydrolysis of tris (1,10-phenenthroline) Fe (II): Influence of polymer-surfactant interactions. Colloid Surf. A Physicochem. Eng. Asp. 2011, 380, 300–307. [Google Scholar]
- Liu, Y.Y.; Yang, Q.; Xia, G.P.; Huang, H.B.; Li, H.X.; Ma, L.; Lu, Y.J.; He, L.; Xia, X.K.; She, Z.G. Polyketides with α-glucosidase inhibitory activity from a mangrove endophytic fungus, Penicillium sp. HN29-3B1. J. Nat. Prod. 2015, 78, 1816–1822. [Google Scholar] [CrossRef] [PubMed]
- Khlifi, D.; Sghaier, R.M.; Laouni, D.; Hayouni, A.A.; Hamdi, M.; Bouajila, J. Anti-inflammatory and acetylcholinesterase inhibition activities of Globularia Alypum. JOMB 2013, 2, 232–237. [Google Scholar] [CrossRef]
- Fang, L.; Li, J.; Zhou, J.; Wang, X.; Guo, L. Isolation and purification of three ecdysteroids from the stems of Diploclisia glaucescens by high-speed countercurrent chromatography and their anti-inflammatory activities in vitro. Molecules 2017, 22, 1310. [Google Scholar] [CrossRef] [PubMed]
- Song, J.H.; Kang, K.S.; Choi, Y.K. Protective effect of casuarinin against glutamate-induced apoptosis in HT22 cells through inhibition of oxidative stress-mediated MAPK phosphorylation. Bioorg. Med. Chem. Lett. 2017, 27, 5109–5113. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Yin, Q.M.; Zhang, X.W.; Wang, W.; Dong, X.Y.; Yan, X.; Hu, R. Bioactivity-guided isolation of biphenanthrenes from Liparis nervosa. Fitoterapia 2016, 115, 15–18. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are not available from the authors. |
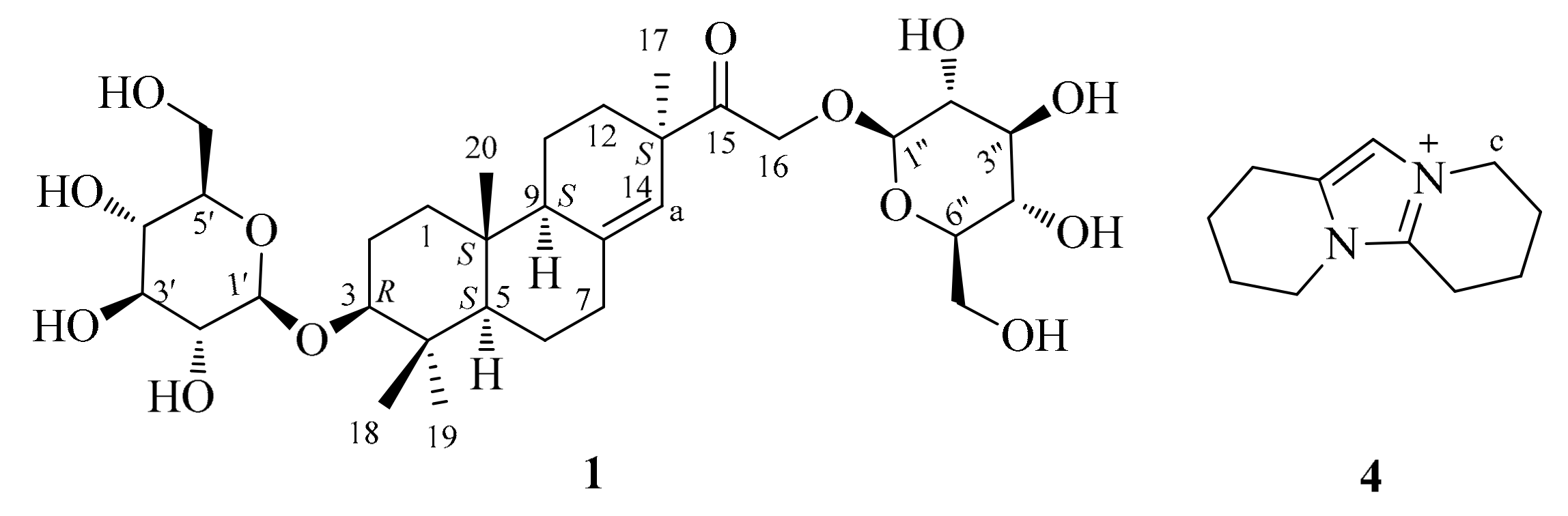
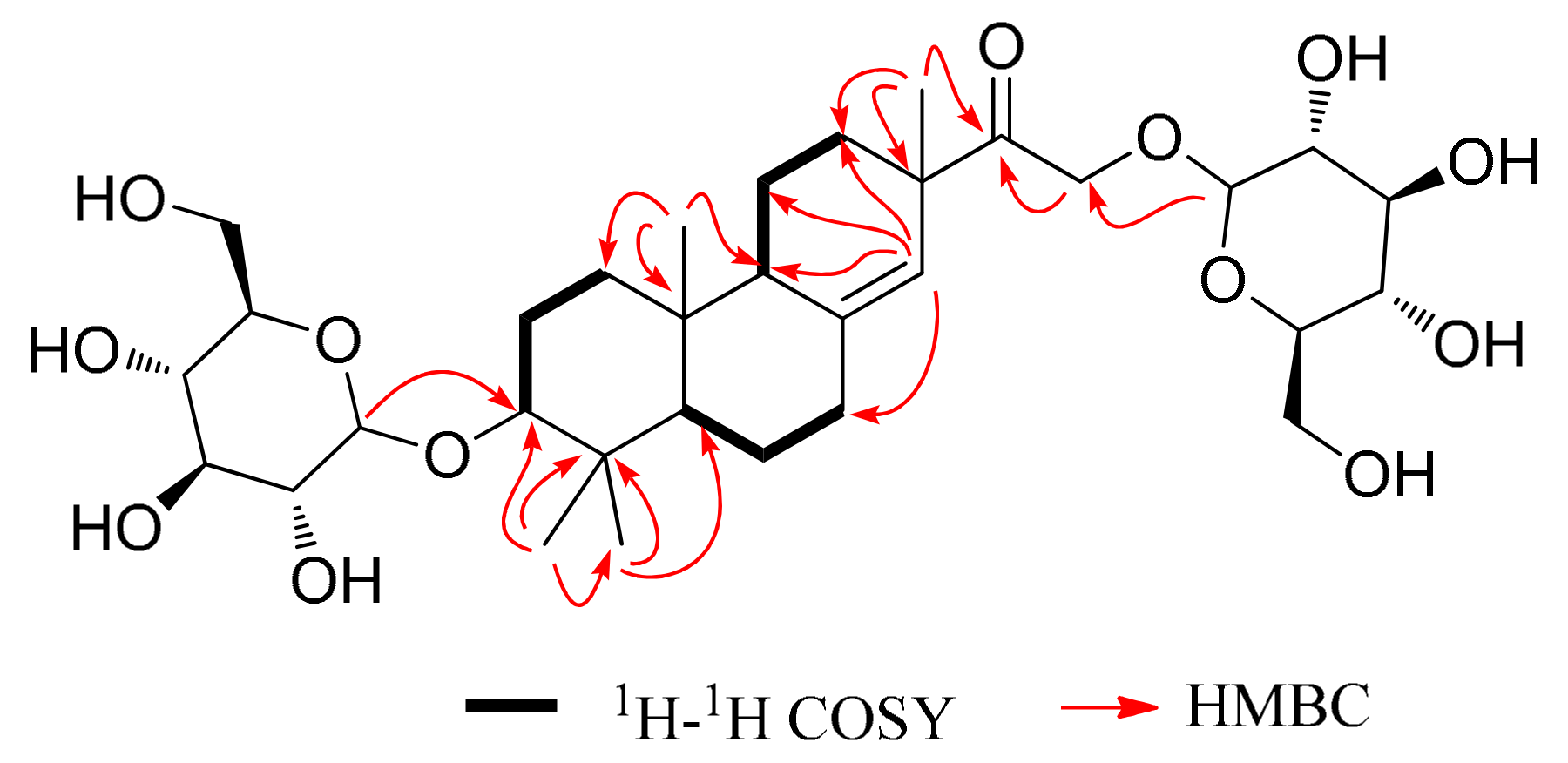
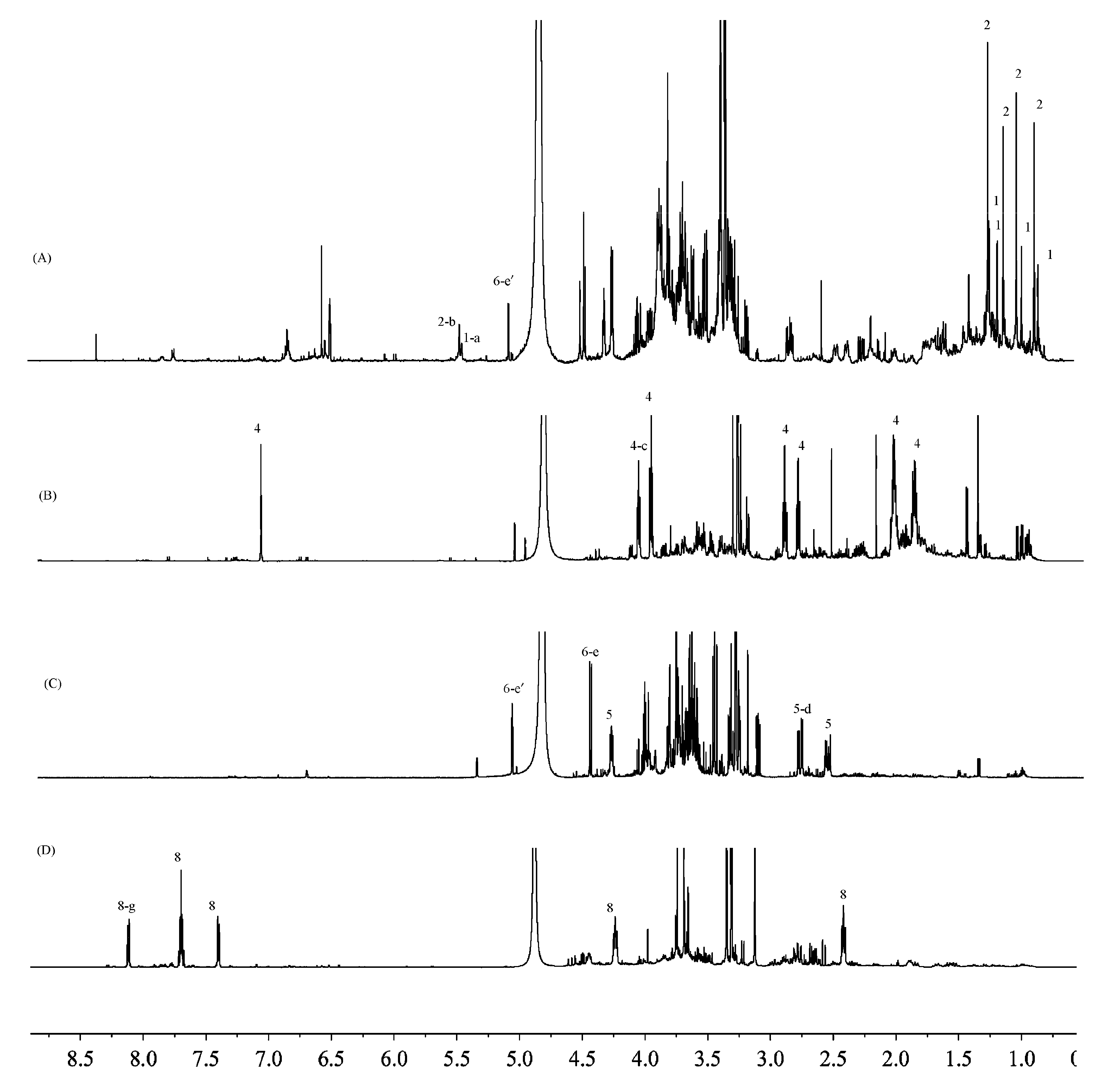
| Position | δC | δH | Position | δC | δH | Position | δC | δH |
|---|---|---|---|---|---|---|---|---|
| 1 | 37.7 t | 1.68 m, 1.16 m | 11 | 23.2 t | 1.61 m, 1.46 m | 1′ | 101.9 d | 4.33 d (7.6) |
| 2 | 24.1 t | 1.79 m, 1.55 m | 12 | 33.6 t | 2.29 m, 1.14 m | 2′ | 75.1 d | 3.26–3.18 m |
| 3 | 85.8 d | 3.38 m | 13 | 48.6 s | 3′ | 78.2 d | 3.37–3.27 m | |
| 4 | 39.4 s | 14 | 124.8 d | 5.52 s | 4′ | 71.9 d | 3.29 m | |
| 5 | 55.9 d | 1.15 m | 15 | 213.7 s | 5′ | 77.6 d | 3.37–3.27 m | |
| 6 | 21.2 t | 1.63 m, 1.23 m | 16 | 72.4 t | 4.86 d (18.4) | 6′ | 62.9 t | 3.86 m, 3.67 m |
| 7 | 36.7 t | 2.04 m, 2.11 m | 4.50 d (18.4) | 1″ | 104.2 d | 4.25 d (7.6) | ||
| 8 | 143.5 s | 17 | 27.5 q | 1.13 s | 2″ | 74.9 d | 3.26–3.18 m | |
| 9 | 51.9 d | 1.78 m | 18 | 17.3 q | 0.87 s | 3″ | 78.1 d | 3.37–3.27 m |
| 10 | 40.4 s | 19 | 29.1 q | 1.06 s | 4″ | 71.4 d | 3.29 m | |
| 20 | 15.1 q | 0.73 s | 5″ | 77.7 d | 3.37–3.27 m | |||
| 6″ | 62.7 t | 3.86 m, 3.67 m |
| Plant | Compound | Target Signal | Concentration (mg/g) |
|---|---|---|---|
| δ (ppm, Multiplicity Hz) | |||
| F. fimbriata | flifimdioside A (1) | H-a/5.51 (s, 1H) | 0.97 |
| flickinflimoside B (2) | H-b/5.54 (s, 1H) | 1.63 | |
| syringaresinol-4′-O-d-glucopyranoside (3) | / | / | |
| 3-O-β-d-galactopyranosyl-β-d-galactopyranose (6) | H-e′/5.12 (d, 2.4, 1H) | 0.96 | |
| D. nobile | anosmine (4) | H-c/4.11 (t, 3.6, 2H) | 3.79 |
| D. officinale | malic acid (5) | H-d/2.78 (dd, 10.8, 3.6, 1H) | 1.38 |
| 3-O-β-d-galactopyranosyl-β-d-galactopyranose (6) | H-e/4.48 (d, 5.2, 1H) | 2.42 | |
| β-pyranose (7) | C-f/99.2 (s) | about 1.09 | |
| β-furanose (7) | C-f′/103.1 (s) | about 0.32 | |
| α-furanose (7) | C-f″/105.3 (s) | about 0.069 | |
| D. loddigesii | shihunine (8) | H-g/8.11 (dd, 4.4, 1.2, 1H) | 2.27 |
| DPPH | OH | α-glucosidase | NO | ||
|---|---|---|---|---|---|
| Sample | polar extract of F. fimbriata | 131.8 | 274.7 | 396.7 | a.a a |
| 1 | a.a | a.a | 40.3 | a.a | |
| 2 | a.a | a.a | 45.8 | a.a | |
| 3 | 48.1 | a.a | 40.4 | a.a | |
| polar extract of D. nobile | 1278.9 | a.a | 396.7 | 122 | |
| 4 | a.a | a.a | 17.7 | 16.1 | |
| polar extract of D. officinale | 1032.0 | 457.5 | 161.7 | a.a | |
| polar extract of D. loddigesii | 317 | 486.3 | 682.2 | 130 | |
| 8 | a.a | a.a | 21.9 | 11.5 | |
| Positive control | trans-resveratrol | n.t b | n.t | 7.7 | n.t |
| L-NG-monomethyl arginine citrate | n.t | n.t | n.t | 7.2 | |
| 2,6-ditertbutyl-4-methyl-phenol | 5.7 | n.t | n.t | n.t | |
| vitamin C | n.t | 6.3 | n.t | n.t |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, H.; Li, X.; Xu, Y.; Lo, K.; Zheng, H.; Hu, H.; Wang, J.; Lin, Y. Study on the Polar Extracts of Dendrobium nobile, D. officinale, D. loddigesii, and Flickingeria fimbriata: Metabolite Identification, Content Evaluation, and Bioactivity Assay. Molecules 2018, 23, 1185. https://doi.org/10.3390/molecules23051185
Chen H, Li X, Xu Y, Lo K, Zheng H, Hu H, Wang J, Lin Y. Study on the Polar Extracts of Dendrobium nobile, D. officinale, D. loddigesii, and Flickingeria fimbriata: Metabolite Identification, Content Evaluation, and Bioactivity Assay. Molecules. 2018; 23(5):1185. https://doi.org/10.3390/molecules23051185
Chicago/Turabian StyleChen, Huiping, Xuewen Li, Yongli Xu, Kakei Lo, Huizhen Zheng, Haiyan Hu, Jun Wang, and Yongcheng Lin. 2018. "Study on the Polar Extracts of Dendrobium nobile, D. officinale, D. loddigesii, and Flickingeria fimbriata: Metabolite Identification, Content Evaluation, and Bioactivity Assay" Molecules 23, no. 5: 1185. https://doi.org/10.3390/molecules23051185
APA StyleChen, H., Li, X., Xu, Y., Lo, K., Zheng, H., Hu, H., Wang, J., & Lin, Y. (2018). Study on the Polar Extracts of Dendrobium nobile, D. officinale, D. loddigesii, and Flickingeria fimbriata: Metabolite Identification, Content Evaluation, and Bioactivity Assay. Molecules, 23(5), 1185. https://doi.org/10.3390/molecules23051185






