Analysis of Menaquinone-7 Content and Impurities in Oil and Non-Oil Dietary Supplements
Abstract
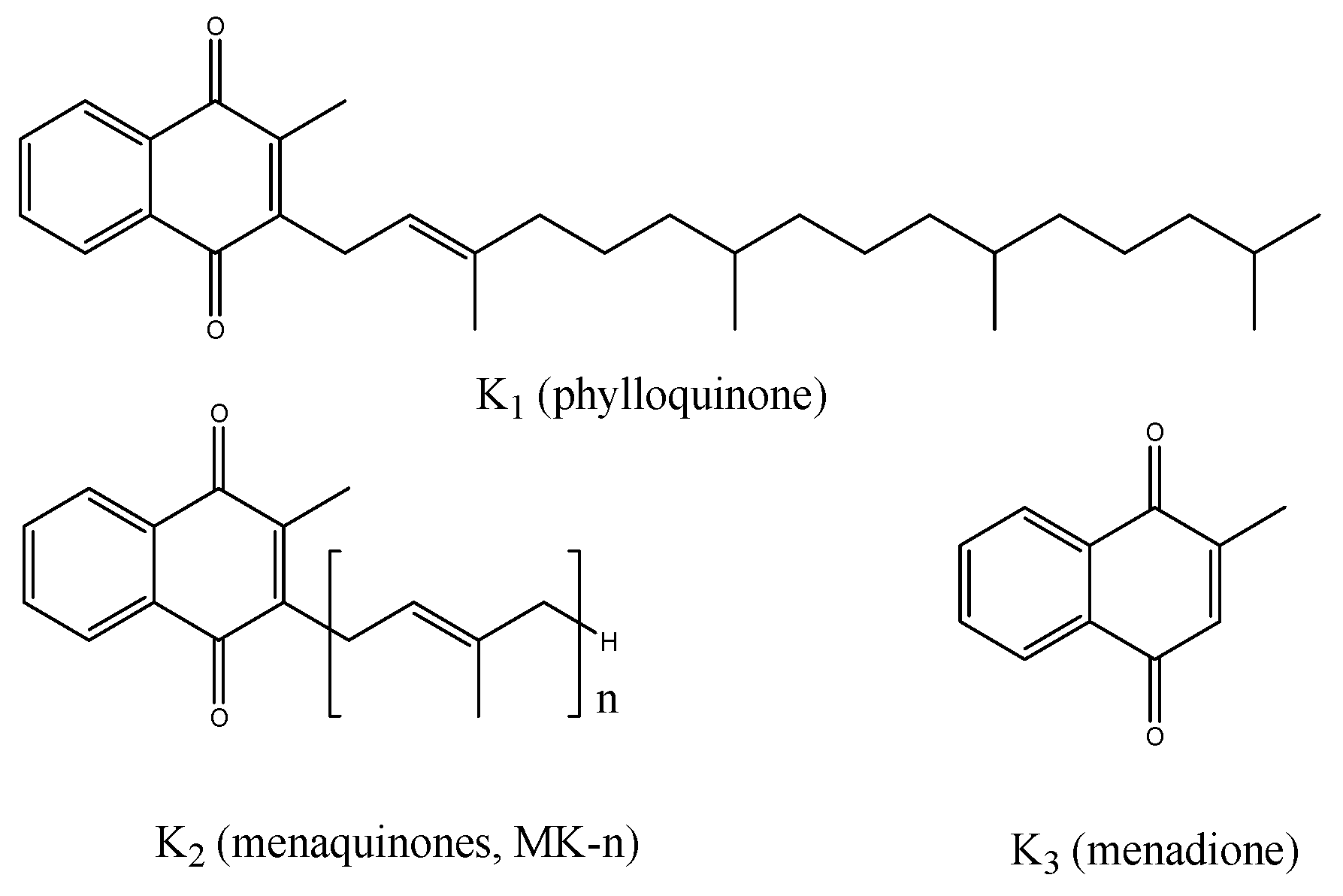
1. Introduction
2. Materials and Methods
2.1. Research Material
2.2. Chemicals
2.3. Preparing a Sample—Hard Tablets
2.4. Preparing a Sample—Gelatine Capsules
2.5. Fractioning
2.6. Chromatographic Analysis, DAD, and HRMS-QTOF
2.7. Method Validation
2.7.1. Preparation of Stock, Spiking Solutions, and LOD and LOQ Calculation
2.7.2. Recovery
2.7.3. Matrix Effect (ME)
2.7.4. Precision and Accuracy
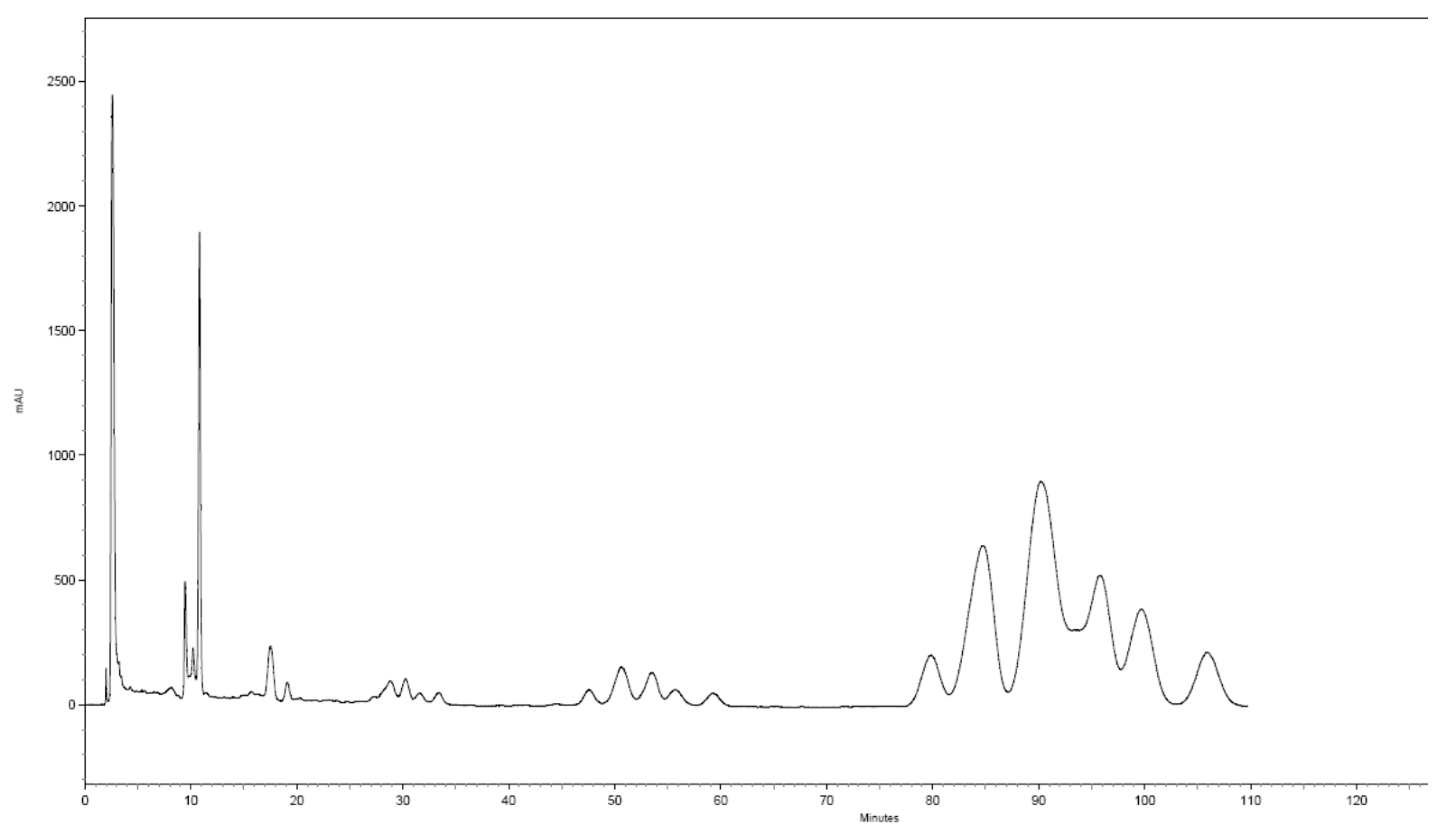
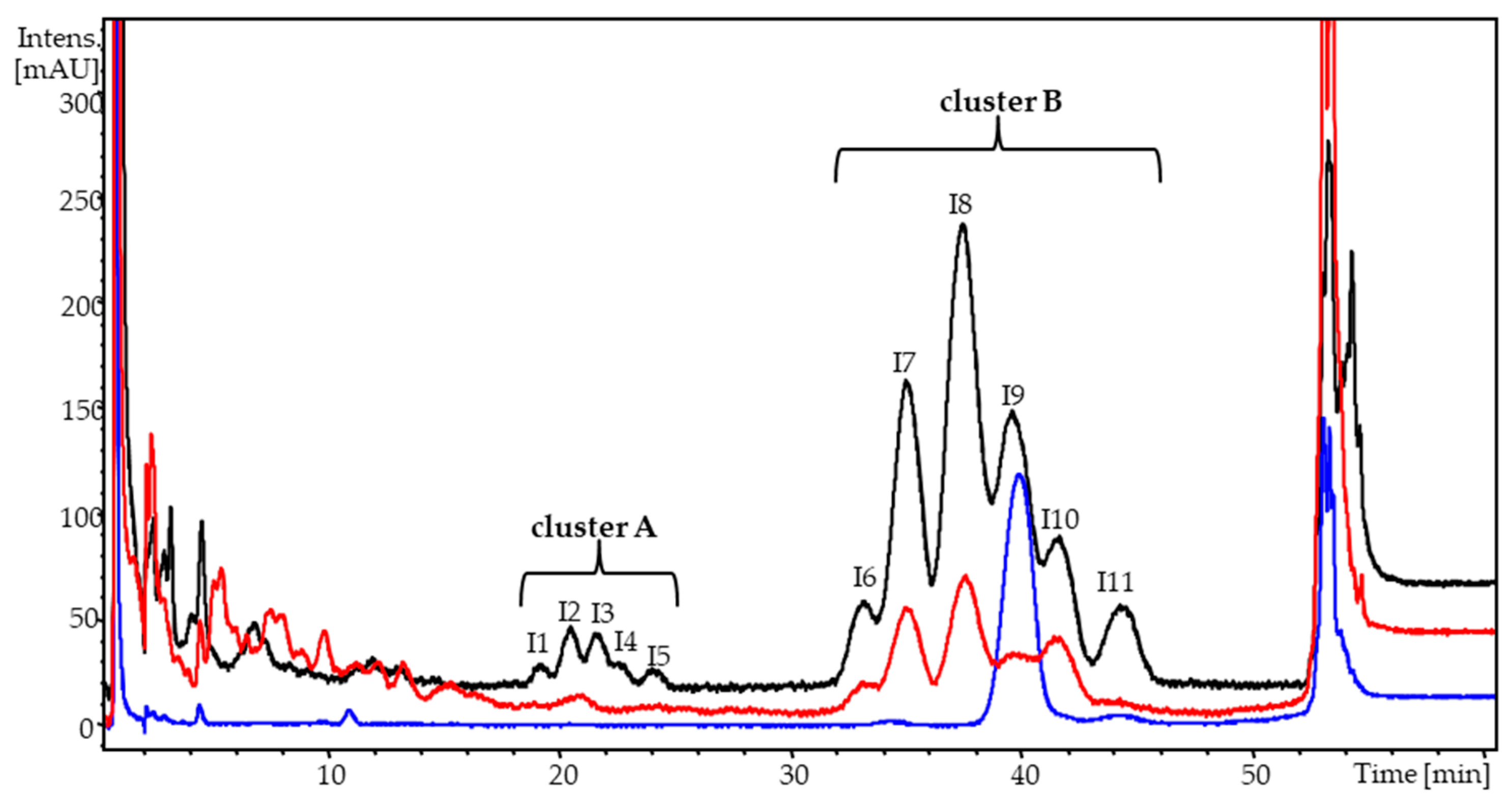
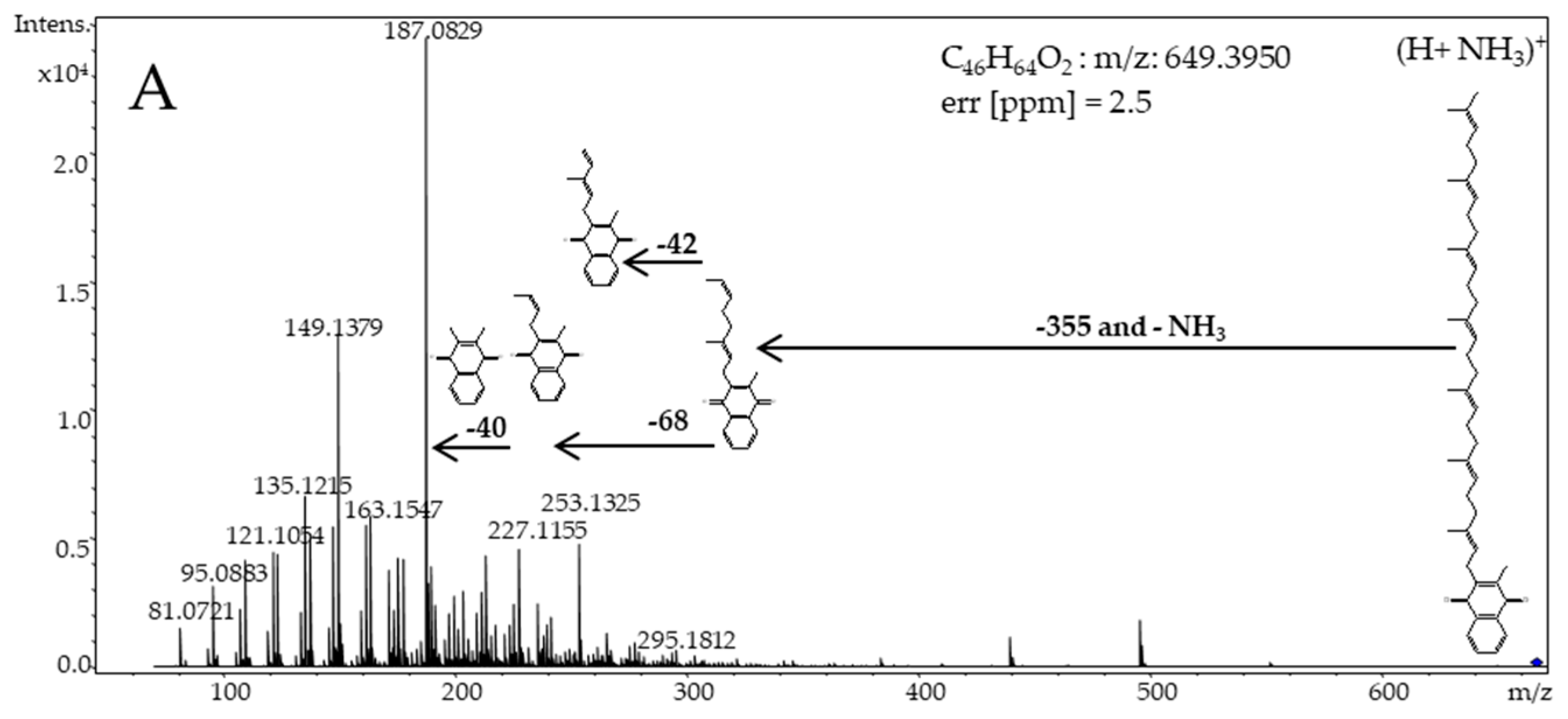
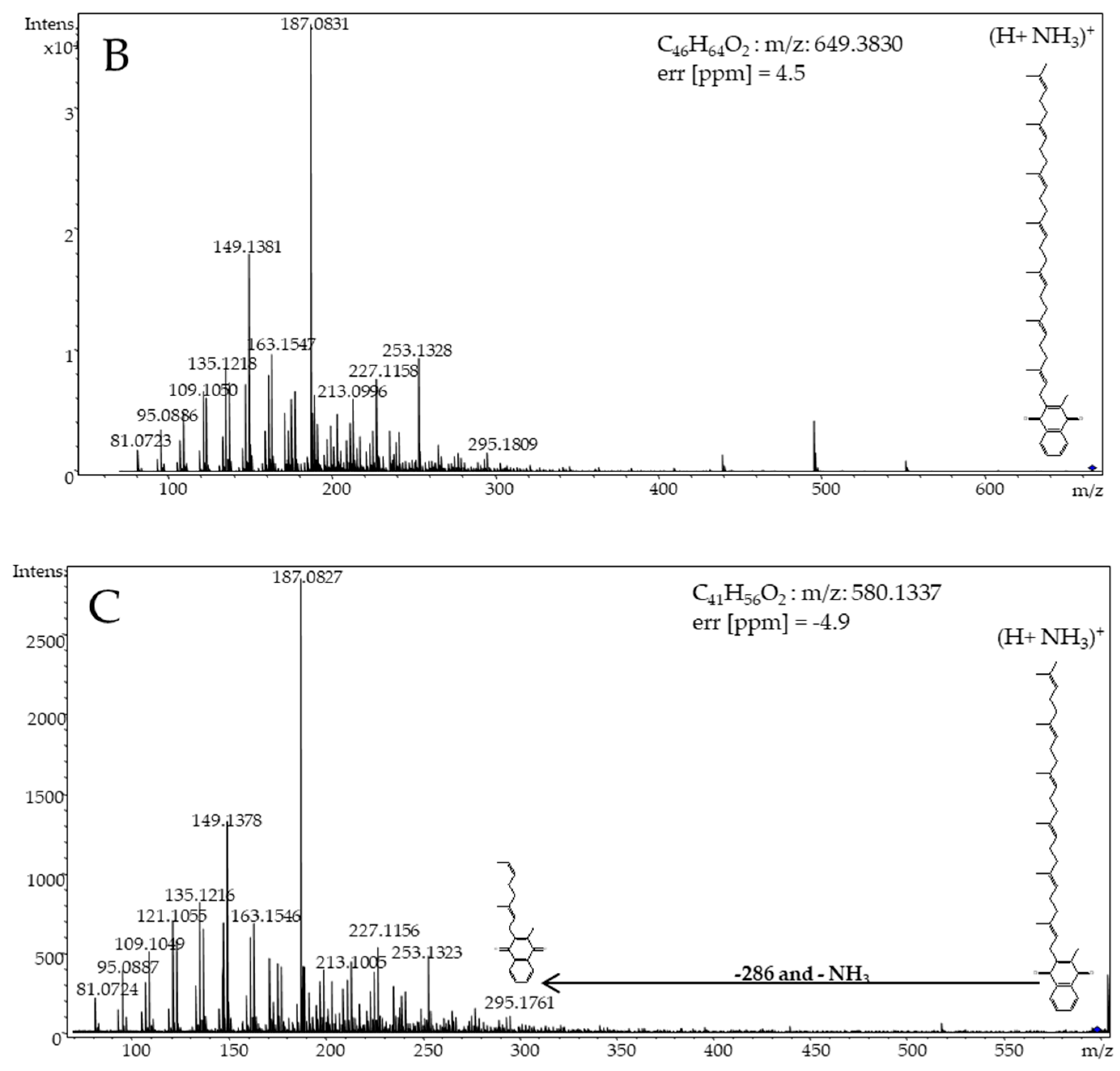
3. Results
4. Discussion
5. Concluding Remarks
Supplementary Materials
Author Contributions
Conflicts of Interest
References
- Shea, M.K.; Booth, S.L. Role of vitamin K in the regulation of calcification. Int. Congr. Ser. 2007, 1297, 165–178. [Google Scholar] [CrossRef]
- Szterk, A.; Zmysłowski, A.; Bus, K. Identification of cis/trans isomers of menaquinone-7 in food as exemplified by dietary supplements. Food Chem. 2018, 243, 403–409. [Google Scholar] [CrossRef] [PubMed]
- Geleijnse, J.M.; Vermeer, C.; Grobbee, D.E.; Schurgers, L.J.; Knapen, M.H.; van der Meer, I.M.; Hofman, A.; Witteman, J.C. Dietary intake of menaquinone is associated with a reduced risk of coronary heart disease: The Rotterdam Study. J. Nutr. 2004, 134, 3100–3105. [Google Scholar] [CrossRef] [PubMed]
- Beulens, J.W.; Bots, M.L.; Atsma, F.; Bartelink, M.L.; Prokop, M.; Geleijnse, J.M.; Witteman, J.C.; Grobbee, D.E.; van der Schouw, Y.T. High dietary menaquinone intake is associated with reduced coronary calcification. Atherosclerosis 2009, 203, 489–493. [Google Scholar] [CrossRef] [PubMed]
- Beulens, J.W.J.; Booth, S.L.; van den Heuvel, E.G.H.M.; Stoecklin, E.; Baka, A.; Vermeer, C. The role of menaquinones (vitamin K2) in human health. Br. J. Nutr. 2013, 110, 1357–1368. [Google Scholar] [CrossRef] [PubMed]
- Vermeer, C. Vitamin K: The effect on health beyond coagulation—An overview. Food Nutr. Res. 2012, 56. [Google Scholar] [CrossRef] [PubMed]
- Plaza, S.M.; Lamson, D.W. Vitamin K2 in bone metabolism and osteoporosis. Altern. Med. Rev. 2005, 10, 24–35. [Google Scholar] [PubMed]
- Gundberg, C.M.; Lian, J.B.; Booth, S.L. Vitamin K-dependent carboxylation of osteocalcin: Friend or foe? Adv. Nutr. 2013, 3, 149–157. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, M. Role of nutritional factor menaquinone-7 in bone homeostasis and osteoporosis prevention. Integr. Mol. Med. 2014, 1, 1–6. [Google Scholar] [CrossRef]
- Spronk, H.M.; Soute, B.A.; Schurgers, L.J.; Thijssen, H.H.; de Mey, J.G.; Vermeer, C. Tissuespecific utilization of menaquinone-4 results in the prevention of arterial calcification in warfarin-treated rats. J. Vasc. Res. 2003, 40, 531–537. [Google Scholar] [CrossRef] [PubMed]
- Weber, P. Vitamin K and bone health. Nutrition 2001, 17, 880–887. [Google Scholar] [CrossRef]
- Booth, S.L. Vitamin K: Food composition and dietary intakes. Food Nutr. Res. 2012, 56, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Hamidi, M.S.; Cheung, A.M. Vitamin K and musculoskeletal health in postmenopausal women. Mol. Nutr. Food Res. 2014, 58, 1647–1657. [Google Scholar] [CrossRef] [PubMed]
- Scheiber, D.; Veulemans, V.; Horn, P.; Chatrou, M.L.; Potthoff, S.A.; Kelm, M.; Westenfeld, L.J.S.R. High-dose menaquinone-7 supplementation reduces cardiovascular calcification in a murine model of extraosseous calcification. Nutrients 2015, 7, 6991–7011. [Google Scholar] [CrossRef] [PubMed]
- Bresson, J.L.; Flynn, A.; Heinonen, M.; Hulshof, K.; Korhonen, H.; Lagiou, P.; Løvik, M.; Marchelli, R.; Martin, A.; Moseley, B.; et al. Vitamin K2 added for nutritional purpose in foods for particular nutritional uses, food supplements and foods intended for the general population and Vitamin K2 as a source of vitamin K added for nutritional purposes to foodstuffs, in the context of Regulation (EC) N° 258/97—Scientific Opinion of the Panel on Dietetic Products, Nutrition and Allergies. EFSA J. 2008, 822, 1–31. [Google Scholar]
- Schurgers, L.J.; Vermeer, C. Determination of phylloquinone and menaquinones in food. Effect of food matrix on circulating vitamin K concentrations. Haemostasis 2000, 30, 298–307. [Google Scholar] [PubMed]
- Nimptsch, K.; Rohrmann, S.; Linseisen, J. Dietary intake of vitamin K and risk of prostate cancer in the Heidelberg cohort of the European Prospective Investi- gation into Cancer and Nutrition (EPIC-Heidelberg). Am. J. Clin. Nutr. 2008, 87, 985–992. [Google Scholar] [CrossRef] [PubMed]
- Sato, T.; Yamada, Y.; Ohtani, Y.; Mitsui, N.; Murasawa, H.; Araki, S. Production of menaquinone (vitamin K2)-7 by Bacillus subtilis. J. Biosci. Bioeng. 2001, 91, 16–20. [Google Scholar] [CrossRef]
- Sato, K.; Inoue, S.; Saito, S. A new synthesis of vitamin K via π-allylnickel intermediates. J. Chem. Soc. Perkin Trans. 1973, 10, 2289–2293. [Google Scholar] [CrossRef]
- Snyder, C.D.; Rapoport, H. Synthesis of menaquinones. J. Am. Chem. Soc. 1974, 96, 8046–8054. [Google Scholar] [CrossRef] [PubMed]
- Daines, A.M.; Payne, R.J.; Humphries, M.E.; Abell, A.D. The synthesis of naturally occurring vitamin K and vitamin K analogues. Curr. Org. Chem. 2003, 7, 1–15. [Google Scholar] [CrossRef]
- Baj, A.; Wałejko, P.; Kutner, A.; Kaczmarek, Ł.; Morzycki, J.W.; Witkowski, S. Convergent synthesis of menaquinone-7 (MK-7). Org. Process. Res. Dev. 2016, 20, 1026–1033. [Google Scholar] [CrossRef]
- Kamao, M.; Suhara, Y.; Tsugawa, N.; Uwano, M.; Yamaguchi, N.; Uenishi, K.; Ishida, H.; Sasaki, S.; Okano, T. Vitamin K content of foods and dietary vitamin K intake in Japanese young women. J. Nutr. Sci. Vitaminol. 2007, 53, 464–470. [Google Scholar] [CrossRef] [PubMed]
- Walther, B.; Karl, J.P.; Booth, S.L.; Boyaval, P. Menaquinones, Bacteria, and the food supply: The relevance of dairy and fermented food products to Vitamin K requirements. Adv. Nutr. 2013, 4, 463–473. [Google Scholar] [CrossRef] [PubMed]
- Lowenthal, J.; Rivera, G.M.V. Comparison of the activity of the cis and trans isomer of vitamin K1 in vitamin K—Deficient and coumarin anticoagulant p retreated rats. J. Pharmacol. Exp. Ther. 1979, 209, 330–333. [Google Scholar] [PubMed]
- Knauer, T.E.; Siegfried, C.; Willingham, A.K.; Matschiner, J.T. Metabolism and biological activity of cis- and trans-phylloquinone in the rat. J. Nutr. 1975, 105, 1519–1524. [Google Scholar] [CrossRef] [PubMed]
- SCCS. Scientific Committee on Consumer Safety OPINION ON Vitamin K1 (Phytonadione). The SCCS Adopted This Opinion at 6th Plenary Meeting of 23 March 2010. Available online: https://ec.europa.eu/health/scientific_committees/consumer_safety/docs/sccs_o_014.pdf (accessed on 12 March 2018).
- Huang, B.; Zheng, F.; Fu, S.; Yao, J.; Tao, B.; Ren, Y. UPLC-ESI-MS/MS for determining trans- and cis-vitamin K1 in infant formulas: method and applications. Eur. Food Res. Technol. 2012, 235, 873–879. [Google Scholar] [CrossRef]
- Yilmaz, B.; Kadioglu, Y.; Meral, K.; Onganer, Y. Determination of human growth hormone in pure and pharmaceutical dosage form by spectrofluorometry and high performance liquid chromatography. Chem. Ind. Chem. Eng. Q. 2012, 18, 399–405. [Google Scholar] [CrossRef]
- Booth, S.L.; Davidson, K.W.; Sadowski, J.A. Evaluation of an HPLC method for the determination of phylloquinone (Vitamin K1) in various food matrices. J. Agric. Food Chem. 1994, 42, 295–300. [Google Scholar] [CrossRef]
- Booth, S.L.; Sadowski, J.A.; Penningtont, J.A.T. Phylloquinone (Vitamin K1) content of foods in the U.S. food and drug administration’s total diet study. J. Agric. Food Chem. 1995, 43, 1574–1579. [Google Scholar] [CrossRef]
- Ferreira, D.W.; Haytowitz, D.B.; Tassinari, M.A.; Peterson, J.W.; Booth, S.L. Vitamin K contents of grains, cereals, fast-food breakfasts, and baked goods. J. Food Sci. 2006, 71, S66–S70. [Google Scholar] [CrossRef]
- Huang, N.; Siegel, M.M. Automation of a fourier transform ion cyclotron resonance mass spectrometer for acquisition, analysis, and e-mailing of high- resolution exact-mass electrospray ionization mass spectral data. J. Am. Soc. Mass Spectr. 1999, 10, 1166–1173. [Google Scholar] [CrossRef]
- Fouquet, T.; Humbel, S.; Charles, L. Tandem mass spectrometry of trimethylsilyl-terminated poly (dimethylsiloxane) ammonium adducts generated by electrospray ionization. J. Am. Soc. Mass Spectr. 2011, 22, 649–658. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Carlone, G.M.; Anet, F.L. Detection of menaquinone-6 and a novel methyl-substituted menaquinone-6 in cmpylobacter jejuni and cmpylobacter fetus subsp. J. Gen. Microbiol. 1983, 129, 3385–3393. [Google Scholar] [PubMed]
- Pucaj, K.; Rasmussen, H.; Møller, M.; Preston, T. Safety and toxicological evaluation of a synthetic vitamin K2, menaquinone-7. Toxicol. Mech. Method 2011, 21, 520–532. [Google Scholar] [CrossRef] [PubMed]
- Weber, F.; Wiss, O. Die reaktivierung der bernsteinsaure-cytochrom-c-reduktase durch die vitamine K1 und K2 und deren Isoprenologen. Helvetica 1959, 42, 217–225. [Google Scholar] [CrossRef]
- Yamada, Y.; Aoki, K.; Tahara, Y. The structure of the hexahydrogenated isoprenoid side-chain menaquinone with nine isoprene units isolated from actinomadura madurae. J. Gen. Appl. Microbiol. 1982, 28, 321–329. [Google Scholar] [CrossRef]
- Centi, A.J.; Brown-Ramos, M.; Haytowitz, D.B.; Booth, S.L. Changes in the content and forms of vitamin K in processed foods. J. Food Compos. Anal. 2015, 4, 42–44. [Google Scholar] [CrossRef]
Sample Availability: Samples of the dietary supplements are available from the authors. |
| DAD Detector | QTOF Detector (SIM, m/z = 666.5 ± 0.5 m/z) | |
|---|---|---|
| LOD (ng mL−1) | 290.3 | 200.9 |
| % CV | 9.1 | 9.8 |
| LOQ (ng mL−1) | 957.9 | 603.7 |
| % CV | 0.3 | 9.7 |
| Recovery: | ||
| Spiked level 1: 10 µg mL−1 (%) | 85.6 | 81.4 |
| SD | 1.7 | 6.1 |
| Spiked level 2: 20 µg mL−1 (%) | 89.3 | 90.5 |
| SD | 2.3 | 6.2 |
| Spiked level 3: 30 µg mL−1 (%) | 93.8 | 94.6 |
| SD | 0.9 | 4.7 |
| Matrix effect (ME) (%) | <5 | <5 |
| SD | - | - |
| Linear (range in µg mL−1 and R2) | 0.9–250 µg mL−1 R2 = 0.997 | Very low, for example: 2–4 µg mL−1 R2 = 0.991, but for 2–20 µg mL−1 R2 = 0.860 |
| Precision and accuracy: | ||
| Intra-day precision in % CV | 0.8 | 6.1 |
| Intra-day accuracy | 2.9 | 5.8 |
| Inter-day precision in % CV | 4.1 | 11.3 |
| Inter-day accuracy | −2.9 | −5.0 |
| Sample | Average Weight of Pill (mg) | SD | Declared Amount of MK-7 Vitamin by Producer (µg pill−1) | all trans MK-7 (µg pill−1) | SD | Sum of cis/trans Isomers of Vitamin MK-7 (µg pill−1) | SD | Sum of cis/trans Isomers of Vitamin MK-6 (µg pill−1) | SD | |
|---|---|---|---|---|---|---|---|---|---|---|
| Hard pills | Switzerland producer | 394.2 | 2.9 | 75 (from natto) | 80.7 | 11.0 | 218.7 | 28.5 | 16.9 | 2.3 |
| British producer 1 | 548.2 | 5.1 | 100 (from natto) | 87.7 | 3.4 | <LOD | - | <LOD | - | |
| British producer 2 | 300.8 | 6.7 | 100 (no data) | 22.7 | 0.8 | <LOD | - | <LOD | - | |
| Polish producer | 232.2 | 2.2 | 75 (no data) | 42.5 | 1.6 | <LOD | - | <LOD | - | |
| Soft pills (gelatine pills) | Polish producer 1 | 427.8 | 25.7 | 75 (no data) | 26.0 | 0.4 | 70.9 | 1.1 | 5.5 | 0.1 |
| Polish producer 2 | 425.5 | 23.8 | 50 (from natto) | 223.8 | 35.6 | <LOD | - | <LOD | - | |
| Polish producer 3 | 972.9 | 15.0 | 50 (from natto) | 224.8 | 23.4 | <LOD | - | <LOD | - | |
| Polish producer 4 | 431.9 | 11.4 | 75 (from natto) | 373.8 | 17.7 | <LOD | - | <LOD | - |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Szterk, A.; Bus, K.; Zmysłowski, A.; Ofiara, K. Analysis of Menaquinone-7 Content and Impurities in Oil and Non-Oil Dietary Supplements. Molecules 2018, 23, 1056. https://doi.org/10.3390/molecules23051056
Szterk A, Bus K, Zmysłowski A, Ofiara K. Analysis of Menaquinone-7 Content and Impurities in Oil and Non-Oil Dietary Supplements. Molecules. 2018; 23(5):1056. https://doi.org/10.3390/molecules23051056
Chicago/Turabian StyleSzterk, Arkadiusz, Katarzyna Bus, Adam Zmysłowski, and Karol Ofiara. 2018. "Analysis of Menaquinone-7 Content and Impurities in Oil and Non-Oil Dietary Supplements" Molecules 23, no. 5: 1056. https://doi.org/10.3390/molecules23051056
APA StyleSzterk, A., Bus, K., Zmysłowski, A., & Ofiara, K. (2018). Analysis of Menaquinone-7 Content and Impurities in Oil and Non-Oil Dietary Supplements. Molecules, 23(5), 1056. https://doi.org/10.3390/molecules23051056





