Anticancer Profiling for Coumarins and Related O-Naphthoquinones from Mansonia gagei against Solid Tumor Cells In Vitro
Abstract
:1. Introduction
2. Results and Discussion
2.1. Isolation and Structural Identification of O-Naphthoquinones and Coumarins
2.2. Cytotoxicity Assessment of Some O-Naphthoquinones and Coumarins
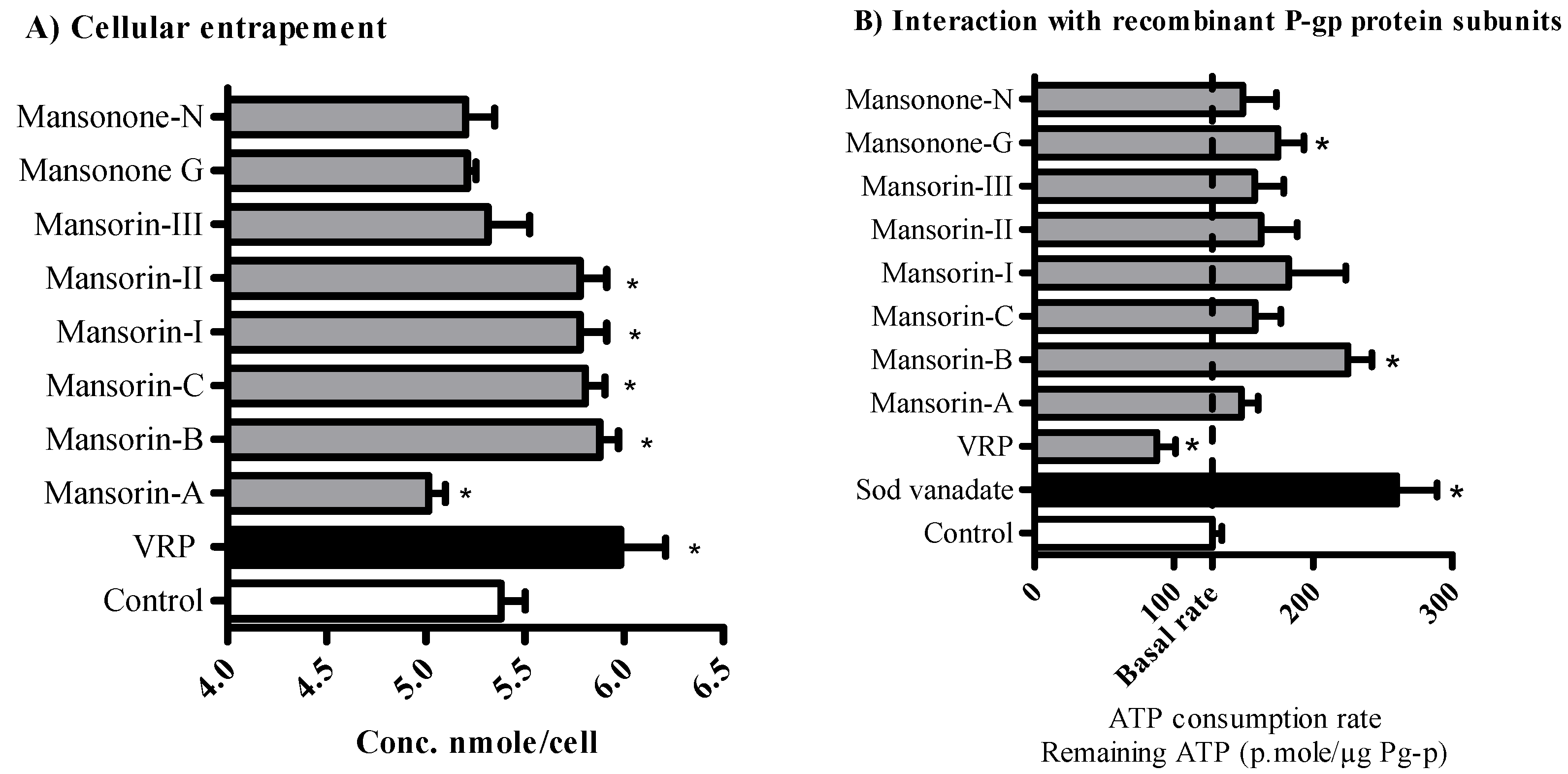
2.3. The Influence of O-Naphthoquinones and Related Coumarins on the Cellular Pharmacokinetics within Tumor Cells
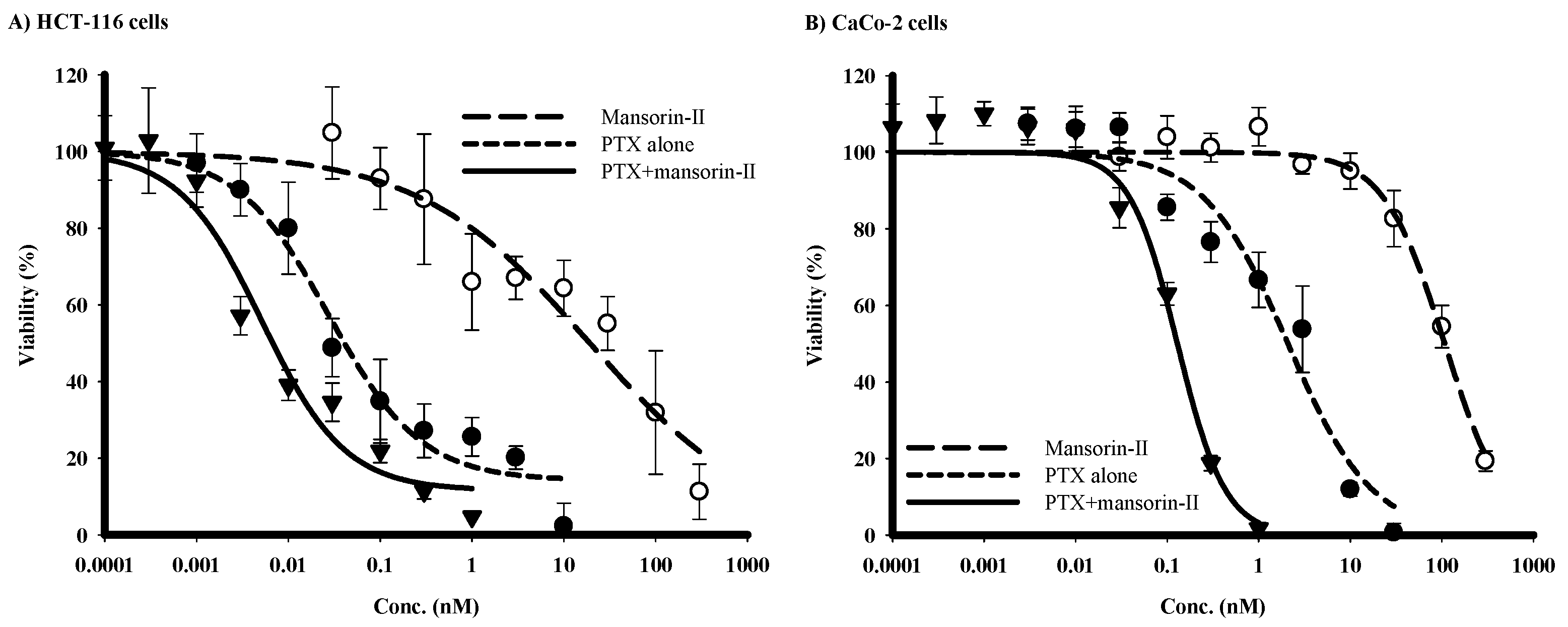
2.4. Chemomodulatory Effect of Mansorin-II to Paclitaxel (PTX) against Colorectal Cancer Cells
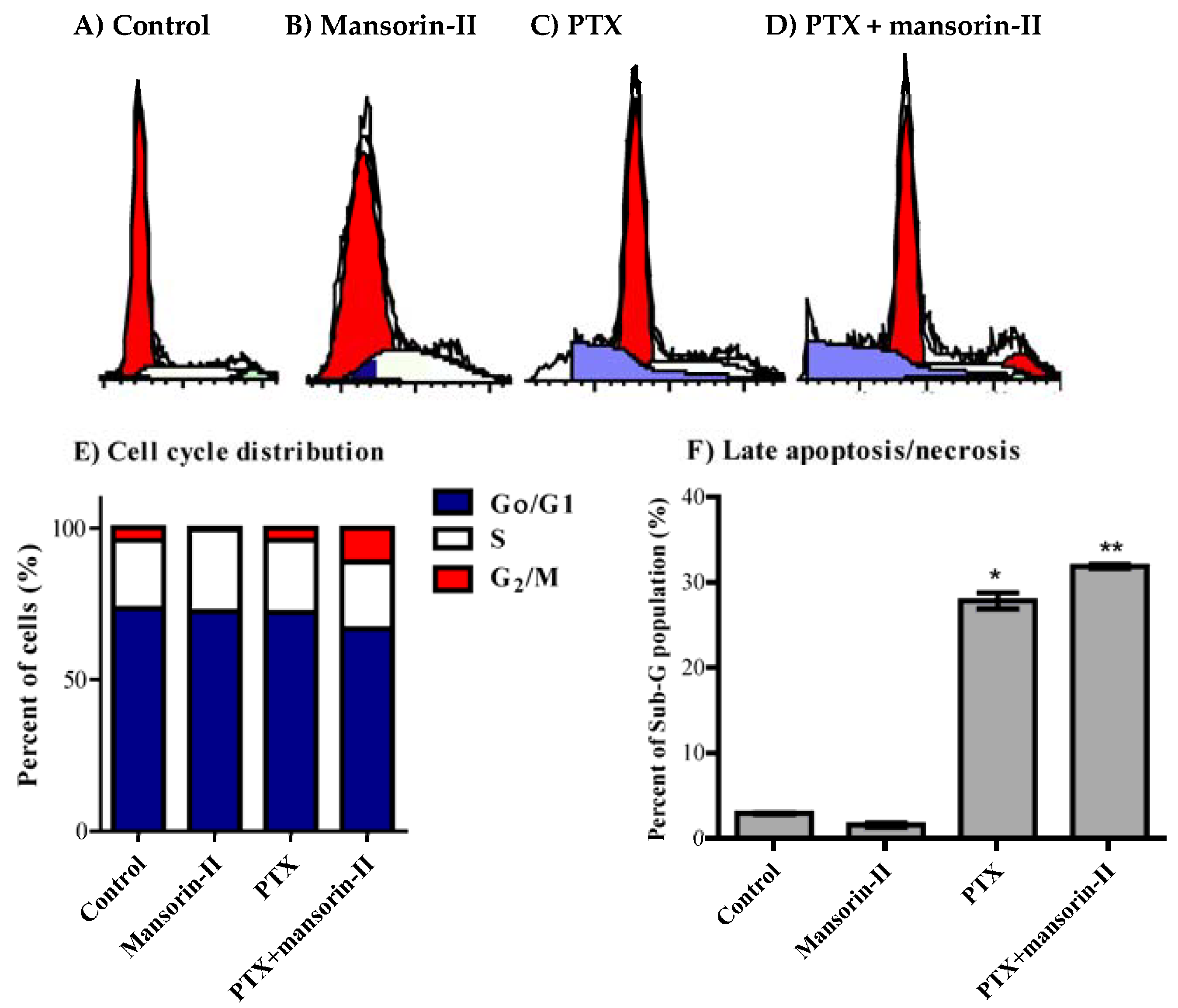
2.5. Cell Cycle Distribution Analysis of Colorectal Cancer Cells
3. Materials and Methods
3.1. Drugs and Chemicals
3.2. General Experimental Procedures
3.3. Plant Material
3.4. Extraction and Isolation of Compounds from A. melegueta
3.5. Preparation of Compound 2
3.6. Cell Culture
3.7. Cytotoxicity Assessment
3.8. Data Analysis
3.9. The Influence of Mansorin-II and Other O-Naphthoquinones/Coumarins on the Cellular Pharmacokinetics
3.10. Determining Sub-Molecular Interaction Characteristics between P-gp Protein and O-Naphthoquinones/Coumarins
3.11. Chemomodulatory Effect of Mansorin-II to Paclitaxel within Colorectal Cancer Cells
3.12. Analysis of Cell Cycle Distribution
3.13. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Acknowledgments
Conflicts of Interest
Abbreviations
| CI-value | Combination Index |
| DOX | Doxorubicin |
| FBS | Fetal bovine serum |
| P-gp | P-glycoprotein |
| PBS | Phosphate buffer saline |
| PTX | Paclitaxel |
| SRB | Sulforhodamine-B |
| VRP | Verapamil |
References
- Hook, I.; Mills, C.; Sheridan, H. Bioactive naphthoquinones from higher plants. In Studies in Natural Products Chemistry; Elsevier: New York, NY, USA, 2014; Volume 41, pp. 119–160. ISBN 1572-5995. [Google Scholar]
- Wu, P.-L.; Wu, T.-S.; He, C.-X.; Su, C.-H.; Lee, K.-H. Constituents from the stems of Hibiscus taiwanensis. Chem. Pharm. Bull. 2005, 53, 56–59. [Google Scholar] [CrossRef] [PubMed]
- Wu, W.-B.; Ou, J.-B.; Huang, Z.-H.; Chen, S.-B.; Ou, T.-M.; Tan, J.-H.; Li, D.; Shen, L.-L.; Huang, S.-L.; Gu, L.-Q. Synthesis and evaluation of mansonone F derivatives as topoisomerase inhibitors. Eur. J. Med. Chem. 2011, 46, 3339–3347. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.-F.; Liu, Z.-P.; Wang, F.-P. Natural sesquiterpenoids as cytotoxic anticancer agents. Mini Rev. Med. Chem. 2011, 11, 1153–1164. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Xia, M.; Cui, Z.; Tashiro, S.; Onodera, S.; Ikejima, T. Cytotoxic effects of mansonone E and F isolated from Ulmus pumila. Biol. Pharm. Bull. 2004, 27, 1025–1030. [Google Scholar] [CrossRef] [PubMed]
- Klenkar, J.; Molnar, M. Natural and synthetic coumarins as potential anticancer agents. J. Chem. Pharm. Res. 2015, 7, 1223–1238. [Google Scholar]
- Amin, K.M.; Eissa, A.A.M.; Abou-Seri, S.M.; Awadallah, F.M.; Hassan, G.S. Synthesis and biological evaluation of novel coumarin–pyrazoline hybrids endowed with phenylsulfonyl moiety as antitumor agents. Eur. J. Med. Chem. 2013, 60, 187–198. [Google Scholar] [CrossRef] [PubMed]
- Nasr, T.; Bondock, S.; Youns, M. Anticancer activity of new coumarin substituted hydrazide–hydrazone derivatives. Eur. J. Med. Chem. 2014, 76, 539–548. [Google Scholar] [CrossRef] [PubMed]
- Abdallah, H.M.; Al-Abd, A.M.; El-Dine, R.S.; El-Halawany, A.M. P-glycoprotein inhibitors of natural origin as potential tumor chemo-sensitizers: A review. J. Adv. Res. 2015, 6, 45–62. [Google Scholar] [CrossRef] [PubMed]
- Braidy, N.; Poljak, A.; Jayasena, T.; Sachdev, P. Natural Plant-Derived Acetylcholinesterase Inhibitors: Relevance for Alzheimer’s Disease. Nat. Prod. Target. Clin. Relevant Enzym. 2017, 297–318. [Google Scholar] [CrossRef]
- Al Muqarrabun, L.M.R.; Ahmat, N. Medicinal uses, phytochemistry and pharmacology of family Sterculiaceae: A review. Eur. J. Med. Chem. 2015, 92, 514–530. [Google Scholar] [CrossRef] [PubMed]
- Tiew, P.; Takayama, H.; Kitajima, M.; Aimi, N.; Kokpol, U.; Chavasiri, W. A novel neolignan, mansoxetane, and two new sesquiterpenes, mansonones R and S, from Mansonia gagei. Tetrahedron Lett. 2003, 44, 6759–6761. [Google Scholar] [CrossRef]
- Tiew, P.; Puntumchai, A.; Kokpol, U.; Chavasiri, W. Coumarins from the heartwoods of Mansonia gagei Drumm. Phytochemistry 2002, 60, 773–776. [Google Scholar] [CrossRef]
- Tiew, P.; Ioset, J.; Kokpol, U.; Chavasiri, W.; Hostettmann, K. Antifungal, antioxidant and larvicidal activities of compounds isolated from the heartwood of Mansonia gagei. Phytother. Res. 2003, 17, 190–193. [Google Scholar] [CrossRef] [PubMed]
- Tiew, P.; Ioset, J.-R.; Kokpol, U.; Schenk, K.; Jaiboon, N.; Chaichit, N.; Chavasiri, W.; Hostettmann, K. Four New Sesquiterpenoid Derivatives from the Heartwood of Mansonia g agei. J. Nat. Prod. 2002, 65, 1332–1335. [Google Scholar] [CrossRef] [PubMed]
- El-Halawany, A.M.; Chung, M.H.; Ma, C.-M.; Komatsu, K.; Nishihara, T.; Hattori, M. Anti-estrogenic Activity of Mansorins and Mansonones from the Heartwood of Mansonia gagei DRUMM. Chem. Pharm. Bull. 2007, 55, 1332–1337. [Google Scholar] [CrossRef] [PubMed]
- Galeffi, C.; delle Monache, E.M.; Casinovi, C.G.; Bettolo, G.B.M. A new quinone from the heartwood of Mansonia Altissima chev: Mansonone L. Tetrahedron Lett. 1969, 10, 3583–3584. [Google Scholar] [CrossRef]
- El-Araby, M.E.; Omar, A.M.; Khayat, M.T.; Assiri, H.A.; Al-Abd, A.M. Molecular Mimics of Classic P-Glycoprotein Inhibitors as Multidrug Resistance Suppressors and Their Synergistic Effect on Paclitaxel. PLoS ONE 2017, 12, e0168938. [Google Scholar] [CrossRef] [PubMed]
- Thiebaut, F.; Tsuruo, T.; Hamada, H.; Gottesman, M.M.; Pastan, I.; Willingham, M.C. Cellular localization of the multidrug-resistance gene product P-glycoprotein in normal human tissues. Proc. Natl. Acad. Sci. USA 1987, 84, 7735–7738. [Google Scholar] [CrossRef] [PubMed]
- Sell, S.; Leffert, H.L. Liver cancer stem cells. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2008, 26, 2800. [Google Scholar] [CrossRef] [PubMed]
- Gottesman, M.M. Mechanisms of cancer drug resistance. Annu. Rev. Med. 2002, 53, 615–627. [Google Scholar] [CrossRef] [PubMed]
- Chuang, J.-Y.; Huang, Y.-F.; Lu, H.-F.; Ho, H.-C.; Yang, J.-S.; Li, T.-M.; Chang, N.-W.; Chung, J.-G. Coumarin induces cell cycle arrest and apoptosis in human cervical cancer HeLa cells through a mitochondria-and caspase-3 dependent mechanism and NF-κB down-regulation. In Vivo 2007, 21, 1003–1009. [Google Scholar] [PubMed]
- Raju, G.; Srinivas, R.; Reddy, M.D.; Reddy, C.R.; Nagesh, N. Studies on non-covalent interaction of coumarin attached pyrimidine and 1-methyl indole 1, 2, 3 triazole analogues with intermolecular telomeric G-quadruplex DNA using ESI-MS and spectroscopy. Nucleosides Nucleotides Nucleic Acids 2014, 33, 489–506. [Google Scholar] [CrossRef] [PubMed]
- Al-Abd, A.M.; Aljehani, Z.K.; Gazzaz, R.W.; Fakhri, S.H.; Jabbad, A.H.; Alahdal, A.M.; Torchilin, V.P. Pharmacokinetic strategies to improve drug penetration and entrapment within solid tumors. J. Control. Release 2015, 219, 269–277. [Google Scholar] [CrossRef] [PubMed]
- Khaleel, S.A.; Al-Abd, A.M.; Ali, A.A.; Abdel-Naim, A.B. Didox and resveratrol sensitize colorectal cancer cells to doxorubicin via activating apoptosis and ameliorating P-glycoprotein activity. Sci. Rep. 2016, 6. [Google Scholar] [CrossRef] [PubMed]
- Minderman, H.; O’Loughlin, K.L.; Pendyala, L.; Baer, M.R. VX-710 (biricodar) increases drug retention and enhances chemosensitivity in resistant cells overexpressing P-glycoprotein, multidrug resistance protein, and breast cancer resistance protein. Clin. Cancer Res. 2004, 10, 1826–1834. [Google Scholar] [CrossRef] [PubMed]
- Walker, J.; Martin, C.; Callaghan, R. Inhibition of P-glycoprotein function by XR9576 in a solid tumour model can restore anticancer drug efficacy. Eur. J. Cancer 2004, 40, 594–605. [Google Scholar] [CrossRef] [PubMed]
- Boesch, D.; Gaveriaux, C.; Jachez, B.; Pourtier-Manzanedo, A.; Bollinger, P.; Loor, F. In vivo circumvention of P-glycoprotein-mediated multidrug resistance of tumor cells with SDZ PSC 833. Cancer Res. 1991, 51, 4226–4233. [Google Scholar] [PubMed]
- Newman, M.J.; Rodarte, J.C.; Benbatoul, K.D.; Romano, S.J.; Zhang, C.; Krane, S.; Moran, E.J.; Uyeda, R.T.; Dixon, R.; Guns, E.S.; et al. Discovery and characterization of OC144-093, a novel inhibitor of P-glycoprotein-mediated multidrug resistance. Cancer Res. 2000, 60, 2964–2972. [Google Scholar] [PubMed]
- Jang, S.H.; Wientjes, M.G.; Au, J.L. Kinetics of P-glycoprotein-mediated efflux of paclitaxel. J. Pharmacol. Exp. Ther. 2001, 298, 1236–1242. [Google Scholar] [PubMed]
- Zhang, Y.; Zhang, Y.K.; Wang, Y.J.; Vispute, S.G.; Jain, S.; Chen, Y.; Li, J.; Youssef, D.T.A.; El Sayed, K.A.; Chen, Z.S. Esters of the marine-derived triterpene sipholenol a reverse P-GP-mediated drug resistance. Mar. Drugs 2015, 13, 2267–2286. [Google Scholar] [CrossRef] [PubMed]
- Quan, F.; Pan, C.; Ma, Q.; Zhang, S.; Yan, L. Reversal effect of resveratrol on multidrug resistance in KBv200 cell line. Biomed. Pharmacother. 2008, 62, 622–629. [Google Scholar] [CrossRef] [PubMed]
- Zhan, Z.; Scala, S.; Monks, A.; Hose, C.; Bates, S.; Fojo, T. Resistance to paclitaxel mediated by P-glycoprotein can be modulated by changes in the schedule of administration. Cancer Chemother. Pharmacol. 1997, 40, 245–250. [Google Scholar] [CrossRef] [PubMed]
- Kuh, H.J.; Nakagawa, S.; Usuda, J.; Yamaoka, K.; Saijo, N.; Nishio, K. A computational model for quantitative analysis of cell cycle arrest and its contribution to overall growth inhibition by anticancer agents. Jpn. J. Cancer Res. 2000, 91, 1303–1313. [Google Scholar] [CrossRef] [PubMed]
- Pucci, B.; Kasten, M.; Giordano, A. Cell cycle and apoptosis. Neoplasia 2000, 2, 291–299. [Google Scholar] [CrossRef] [PubMed]
- Kostova, I. Synthetic and natural coumarins as cytotoxic agents. Curr. Med. Chem. Agents 2005, 5, 29–46. [Google Scholar] [CrossRef]
- Kostova, I.; Bhatia, S.; Grigorov, P.; Balkansky, S.; Parmar, V.S.; Prasad, A.K.; Saso, L. Coumarins as antioxidants. Curr. Med. Chem. 2011, 18, 3929–3951. [Google Scholar] [CrossRef] [PubMed]
- Boonsri, S.; Karalai, C.; Ponglimanont, C.; Chantrapromma, S.; Kanjana-opas, A. Cytotoxic and antibacterial sesquiterpenes from Thespesia populnea. J. Nat. Prod. 2008, 71, 1173–1177. [Google Scholar] [CrossRef] [PubMed]
- El-Halawany, A.M.; El Dine, R.S.; Hattori, M. Anti-estrogenic activity of mansonone G and mansorin A derivatives. Pharm. Biol. 2013, 51, 948–954. [Google Scholar] [CrossRef] [PubMed]
- Skehan, P.; Storeng, R.; Scudiero, D.; Monks, A.; McMahon, J.; Vistica, D.; Warren, J.T.; Bokesch, H.; Kenney, S.; Boyd, M.R. New colorimetric cytotoxicity assay for anticancer-drug screening. J. Natl. Cancer Inst. 1990, 82, 1107–1112. [Google Scholar] [CrossRef] [PubMed]
- Mahmoud, A.M.; Al-Abd, A.M.; Lightfoot, D.A.; El-Shemy, H.A. Anti-cancer characteristics of mevinolin against three different solid tumor cell lines was not solely p53-dependent. J. Enzyme Inhib. Med. Chem. 2012, 27. [Google Scholar] [CrossRef] [PubMed]
- Al-Abd, A.M.; Mahmoud, A.M.; El-Sherbiny, G.A.; El-Moselhy, M.A.; Nofal, S.M.; El-Latif, H.A.; El-Eraky, W.I.; El-Shemy, H.A. Resveratrol enhances the cytotoxic profile of docetaxel and doxorubicin in solid tumour cell lines in vitro. Cell Prolif. 2011, 44. [Google Scholar] [CrossRef] [PubMed]
- Chou, T.C.; Talalay, P. Quantitative analysis of dose-effect relationships: The combined effects of multiple drugs or enzyme inhibitors. Adv. Enzym. Regul. 1984, 22, 27–55. [Google Scholar] [CrossRef]
- Nawwar, M.A.; Swilam, N.F.; Hashim, A.N.; Al-Abd, A.M.; Abdel-Naim, A.B.; Lindequist, U. Cytotoxic isoferulic acidamide from Myricaria germanica (Tamaricaceae). Plant Signal. Behav. 2013, 8. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, G.A.; Al-Abd, A.M.; El-Halawany, A.M.; Abdallah, H.M.; Ibrahim, S.R.M. New xanthones and cytotoxic constituents from Garcinia mangostana fruit hulls against human hepatocellular, breast, and colorectal cancer cell lines. J. Ethnopharmacol. 2017, 198, 302–312. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the all compounds mentioned in the manuscript are available from the authors. |
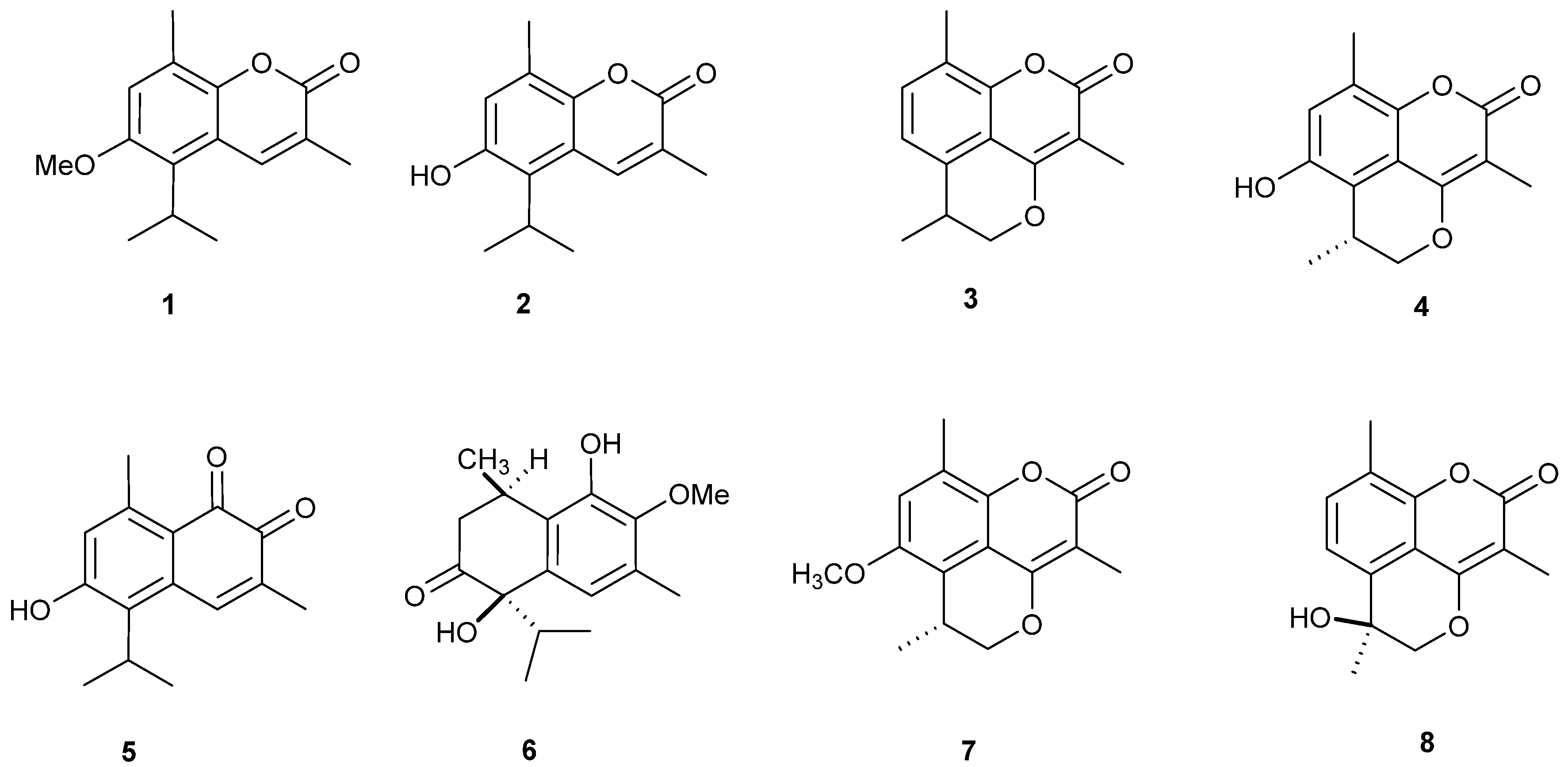

| Compound | HCT-116 | HepG2 | MCF-7 | HeLa | ||||
|---|---|---|---|---|---|---|---|---|
| IC50 (µM) | R-Fraction (%) | IC50 (µM) | R-Fraction (%) | IC50 (µM) | R-Fraction (%) | IC50 (µM) | R-Fraction (%) | |
| Mansorin-A (1) | 11.2 | 0.0 | 3.9 | 46.5 | 2.1 | 90.5 | 12.3 | 1.5 |
| Mansorin-B (2) | 5.7 | 26.3 | 21.9 | 0.0 | 5.0 | 78.9 | 38.7 | 1.3 |
| Mansorin-C (3) | 8.6 | 49.9 | 12.1 | 31.3 | 3.1 | 77.2 | 1.0 | 3.5 |
| Mansorin-I (4) | 11.1 | 0.0 | 35.3 | 0.0 | 23.8 | 0.0 | 3.95 | 0.0 |
| Mansorin-II (7) | 19.3 | 0.36 | 26.8 | 0.0 | 36.0 | 0.0 | 0.74 | 5.7 |
| Mansorin III (8) | >100 | 0.0 | 7.2 | 69.6 | >100 | 0.0 | 5.2 | 39.8 |
| Mansonone-G (5) | 63.4 | 0.5 | 49.4 | 1.1 | 23.0 | 5.1 | 18.8 | 1.8 |
| Mansonone-N (6) | >100 | 0.0 | >100 | 5.2 | >100 | 97.2 | >100 | 0.0 |
| HCT-116 | CaCo-2 | |||
|---|---|---|---|---|
| IC50 | R-Value (%) | IC50 | R-Value (%) | |
| PTX | 27.9 ± 10.2 nM | 14.2 ± 5.3 | 2.1 ± 0.8 µM | 3.1 ± 0.37 |
| Mansorin-II | 19.3 ± 3.7 µM | 0.36 ± 0.007 | 107.9 ± 6.4 µM | 0.34 ± 0.004 |
| PTX with mansorin-II | 5.1 ± 1.9 nM | 11.6 ± 6.3 | 0.13 ± 0.03 µM | 1.8 ± 0.25 |
| CI-index/CI-value | Synergism/0.44 | Synergism/0.18 | ||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Baghdadi, M.A.; Al-Abbasi, F.A.; El-Halawany, A.M.; Aseeri, A.H.; Al-Abd, A.M. Anticancer Profiling for Coumarins and Related O-Naphthoquinones from Mansonia gagei against Solid Tumor Cells In Vitro. Molecules 2018, 23, 1020. https://doi.org/10.3390/molecules23051020
Baghdadi MA, Al-Abbasi FA, El-Halawany AM, Aseeri AH, Al-Abd AM. Anticancer Profiling for Coumarins and Related O-Naphthoquinones from Mansonia gagei against Solid Tumor Cells In Vitro. Molecules. 2018; 23(5):1020. https://doi.org/10.3390/molecules23051020
Chicago/Turabian StyleBaghdadi, Mohammed A., Fahad A. Al-Abbasi, Ali M. El-Halawany, Ali H. Aseeri, and Ahmed M. Al-Abd. 2018. "Anticancer Profiling for Coumarins and Related O-Naphthoquinones from Mansonia gagei against Solid Tumor Cells In Vitro" Molecules 23, no. 5: 1020. https://doi.org/10.3390/molecules23051020
APA StyleBaghdadi, M. A., Al-Abbasi, F. A., El-Halawany, A. M., Aseeri, A. H., & Al-Abd, A. M. (2018). Anticancer Profiling for Coumarins and Related O-Naphthoquinones from Mansonia gagei against Solid Tumor Cells In Vitro. Molecules, 23(5), 1020. https://doi.org/10.3390/molecules23051020





