Scutellarin Mitigates Aβ-Induced Neurotoxicity and Improves Behavior Impairments in AD Mice
Abstract
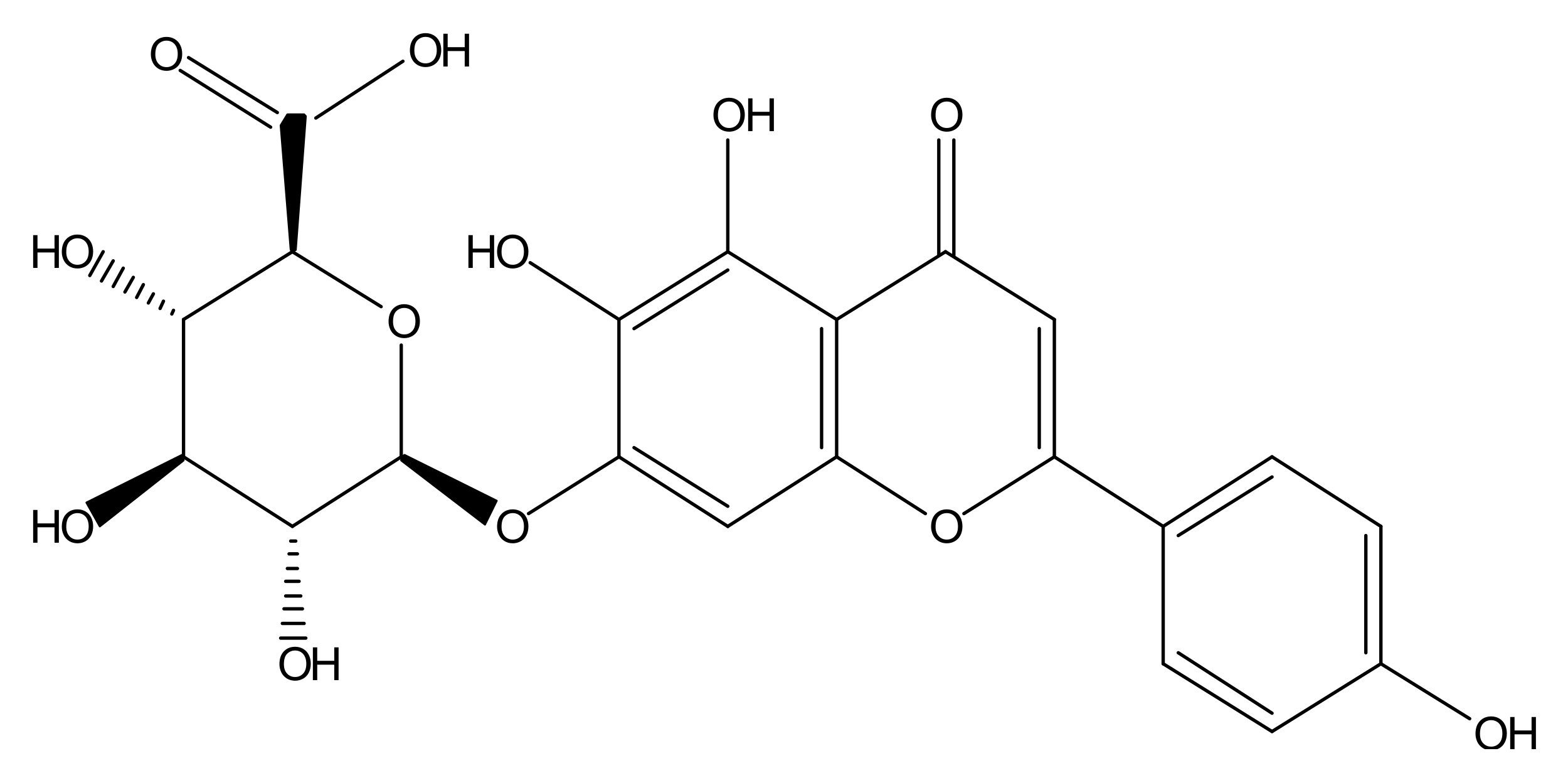
:1. Introduction
2. Materials and Methods
2.1. Aβ Disaggregation Assay
2.1.1. Aβ Preparation
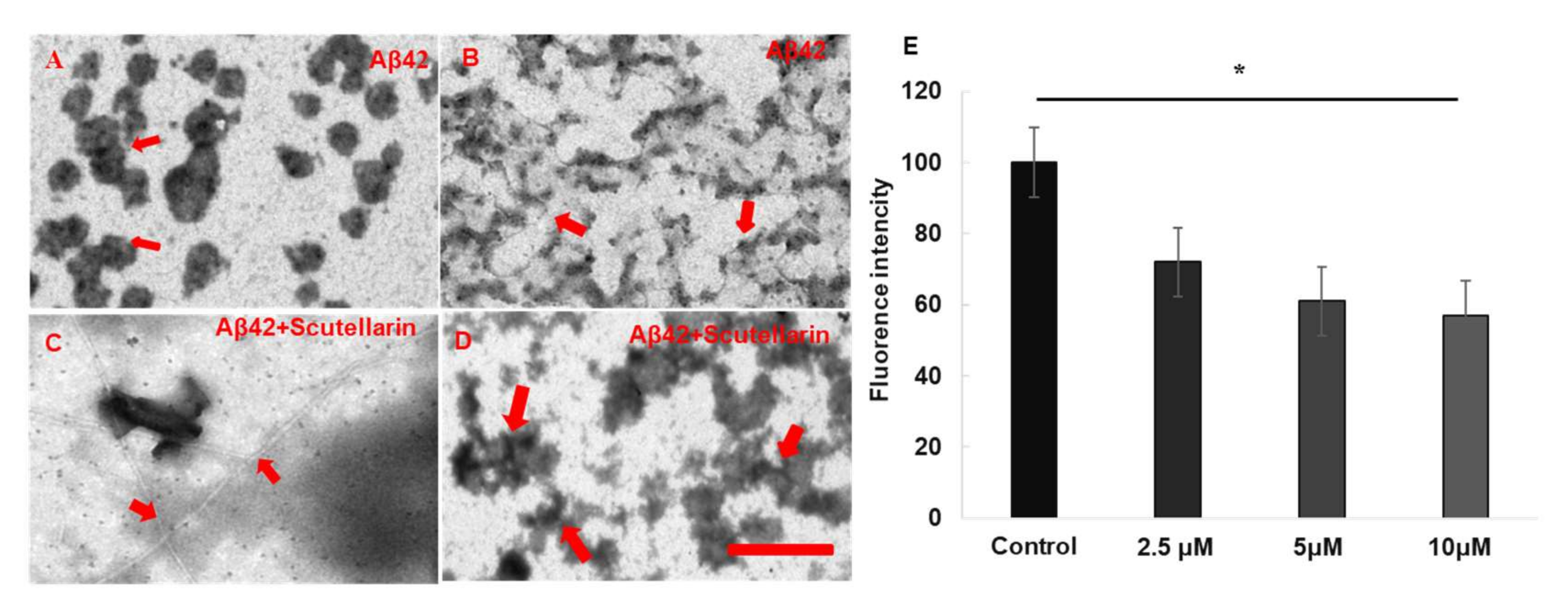
2.1.2. Thioflavin T Fluorescence Assay
2.1.3. Electron Microscopy (EM) Assay
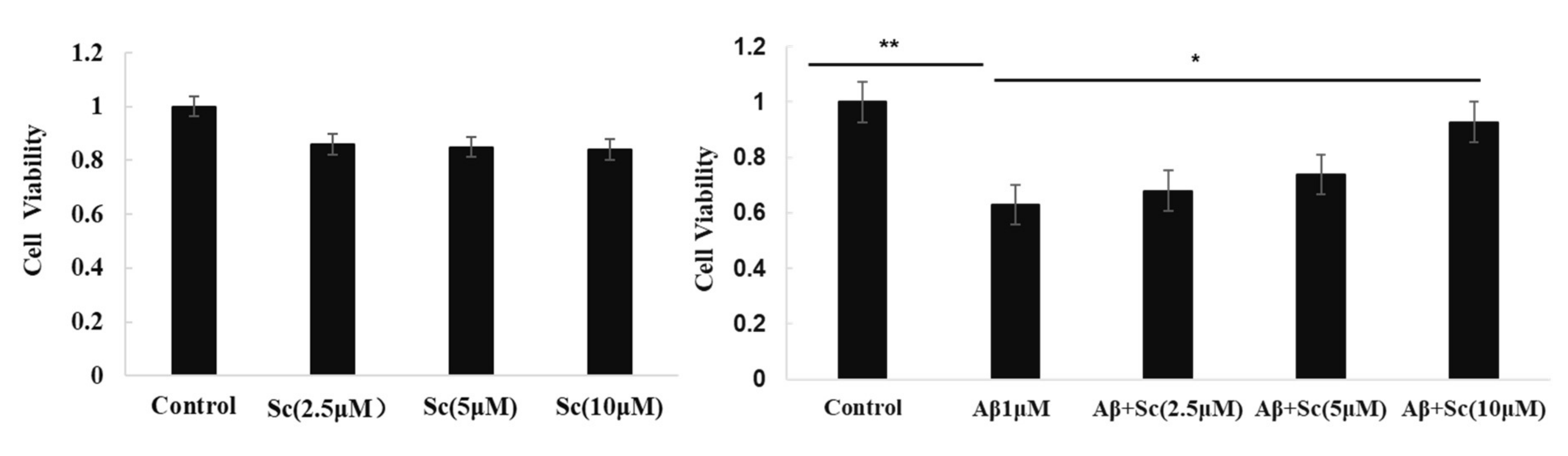
2.2. SH-SY5Y Cell Culture and Viability Assay
2.3. Animals
2.4. Diet Treatment
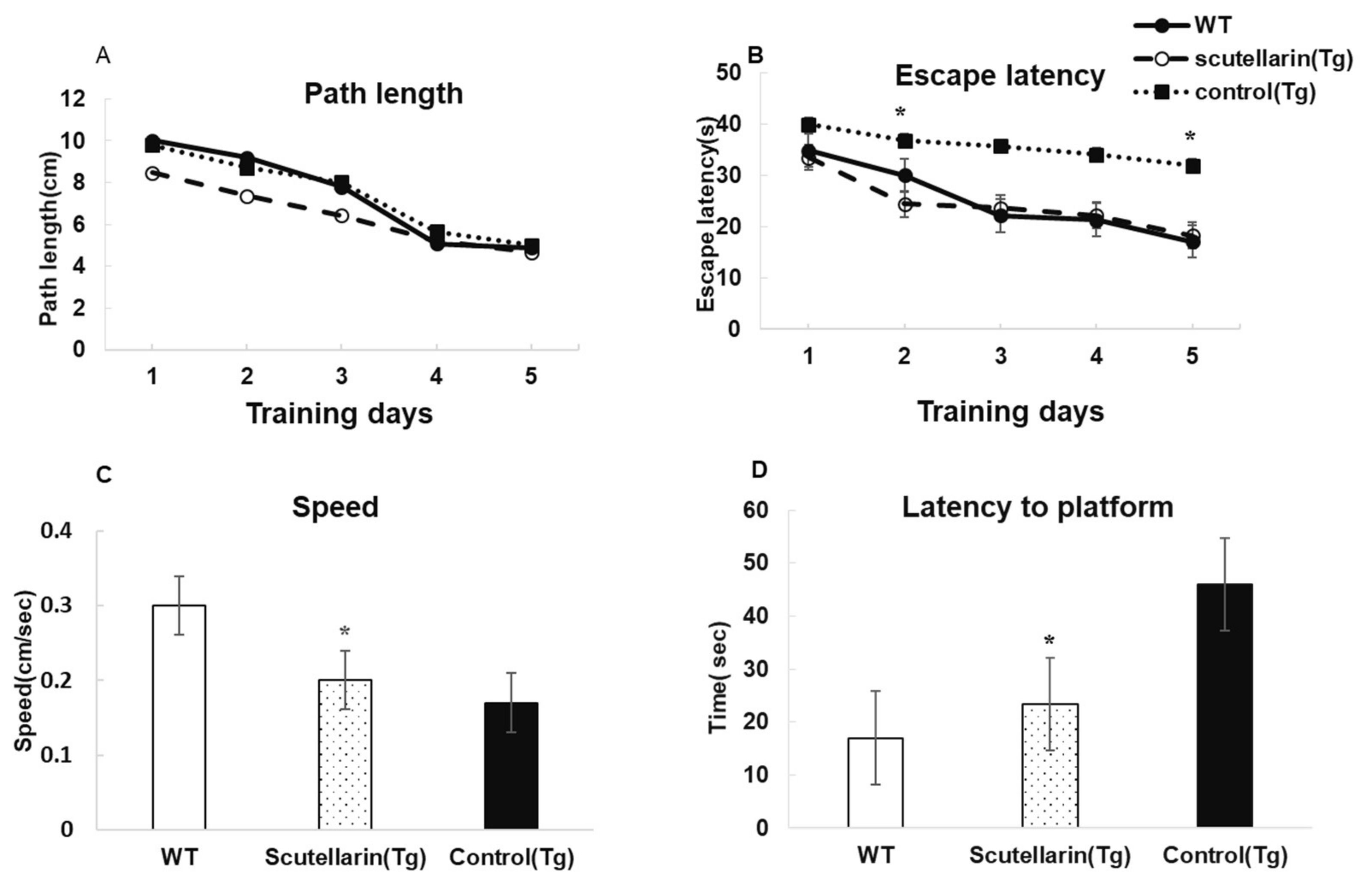
2.5. Behavioral Procedures
2.6. AD Type Pathology and Bioanalysis
2.6.1. Tissue Processing
2.6.2. Histological Staining and Imagine Analysis
2.6.3. ELISA Assay for Soluble and Insoluble Aβ
2.6.4. Quantification of Inflammatory Cytokines in the Mouse Plasma by ELISA
2.7. Statistical Analysis
3. Results
3.1. Protective Effect of Scutellarin on the Cytotoxicity of Aβ42
3.2. Effects of the Scutellarin On Aβ42 Oligomerization and Fibrillation
3.3. Scutellarin Improves Behavioral Impairment after Nine Months of Treatment
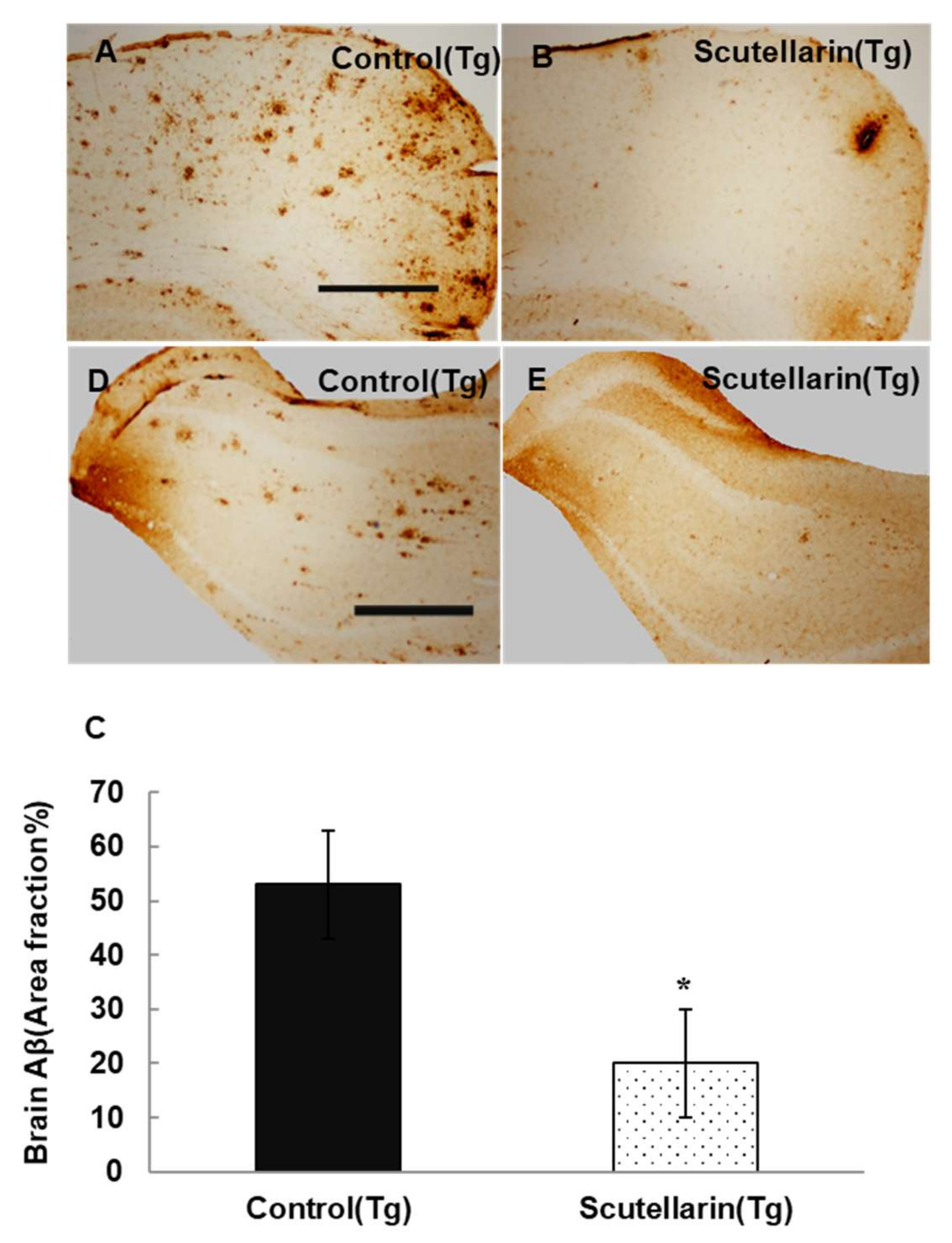
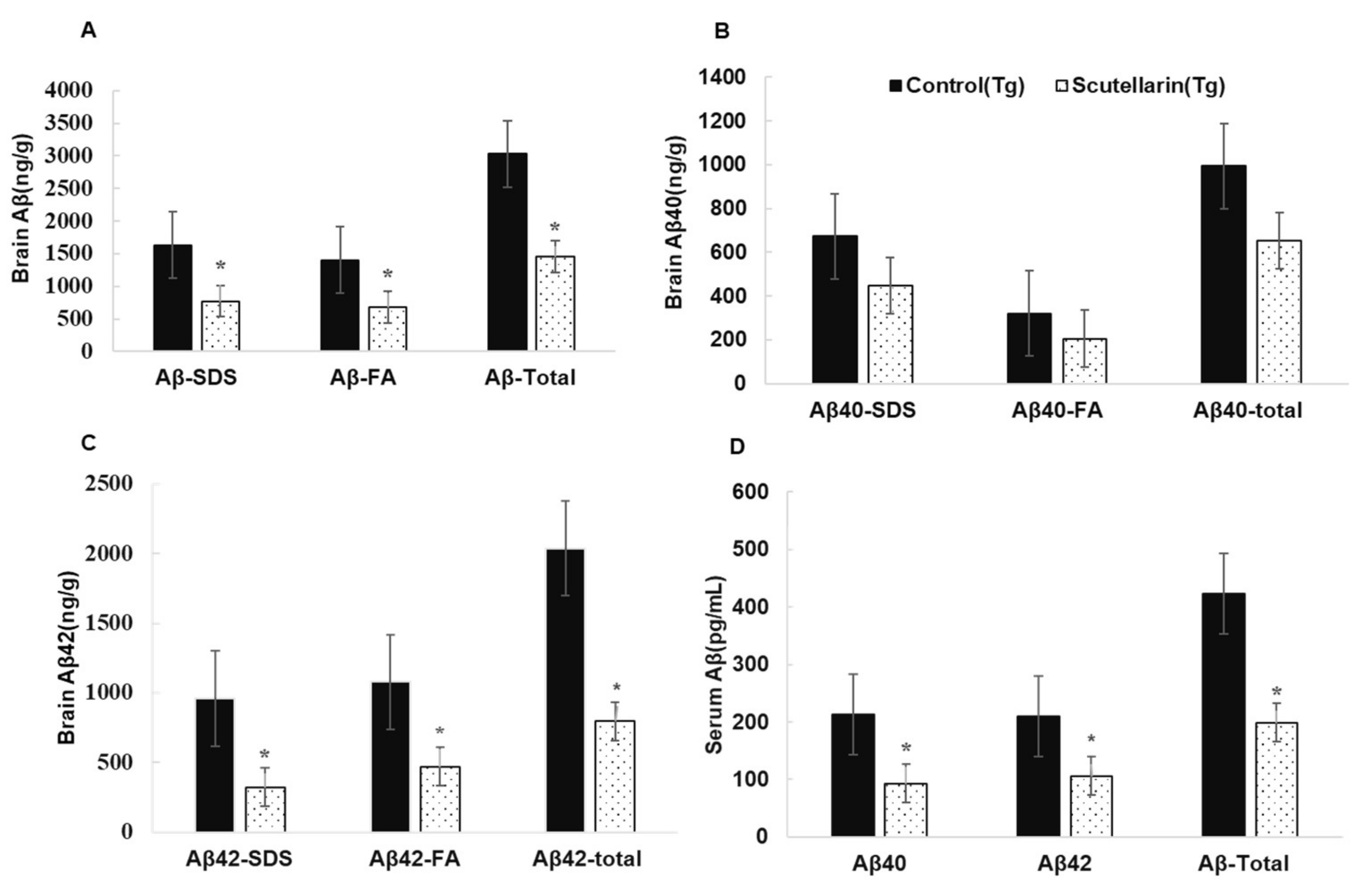
3.4. Scutellarin Reduces Aβ Burden in APP/PS1 Mice
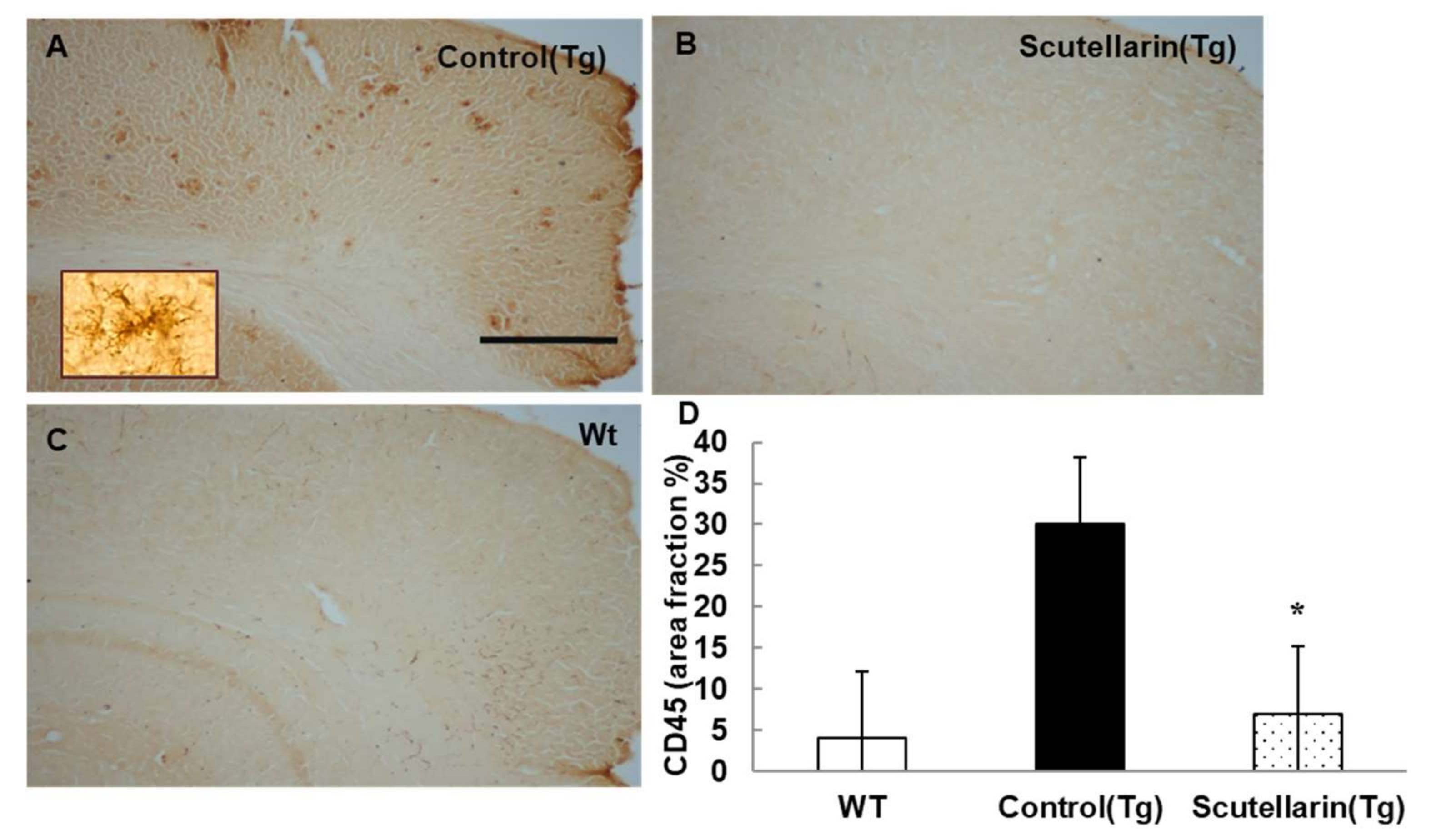
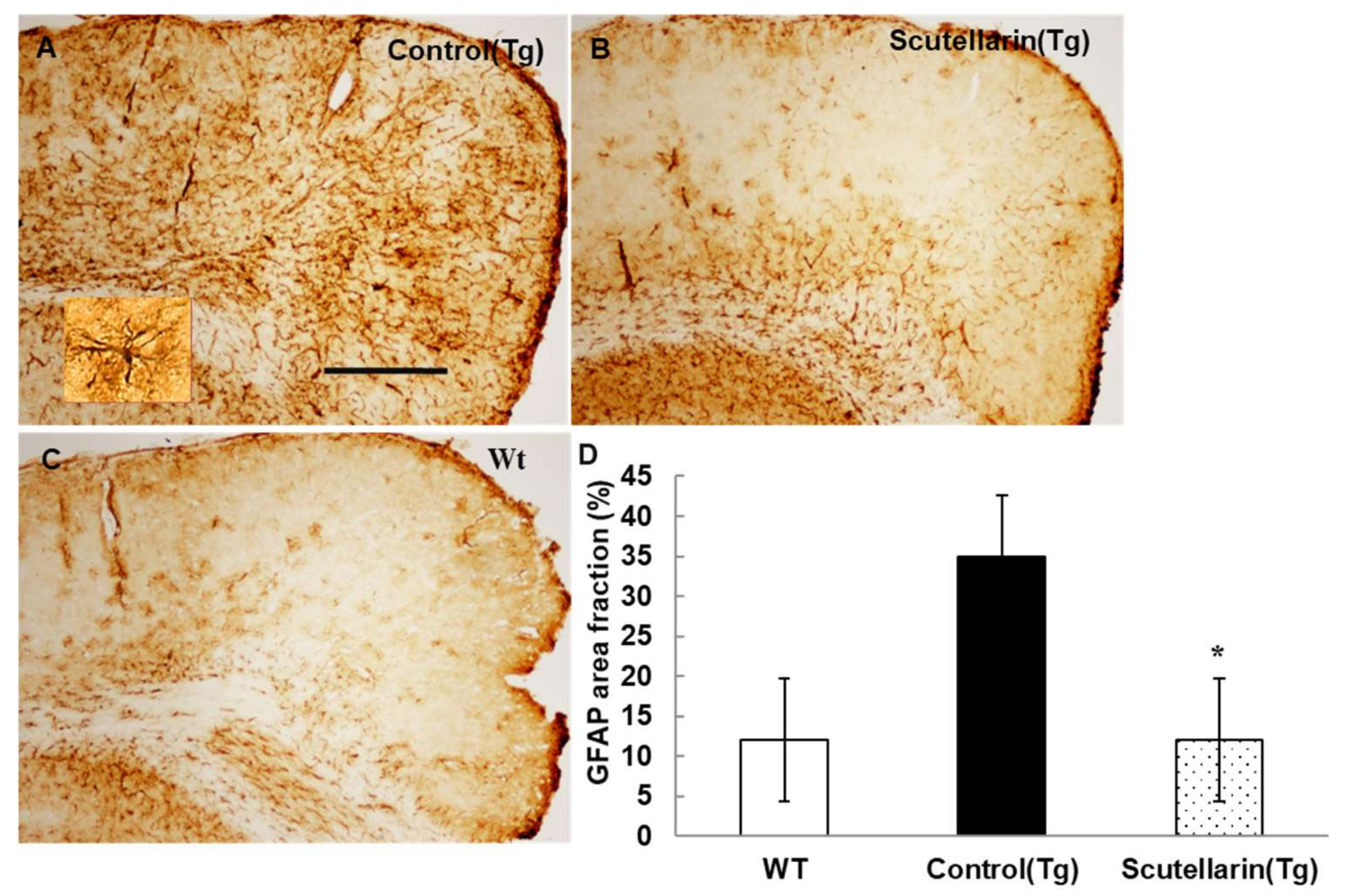
3.5. Scutellarin Decreases Reactive Gliosis and Proinflammatory Cytokines in APP/PS1mice
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Querfurth, H.W.; Laferla, F.M. Alzheimer’s disease. N. Engl. J. Med. 2010, 362, 329–344. [Google Scholar] [CrossRef] [PubMed]
- Serrano-Pozo, A.; Frosch, M.P.; Masliah, E.; Hyman, B.T. Neuropathological alterations in Alzheimer disease. Cold Spring Harb. Perspect. Med. 2011, 1, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Lakey-Beitia, J.; Berrocal, R.; Rao, K.S.; Durant, A.A. Polyphenols as therapeutic molecules in Alzheimer’s disease through modulating amyloid pathways. Mol. Neurobiol. 2015, 51, 466–479. [Google Scholar] [CrossRef] [PubMed]
- Huang, D.Y.; Li, H.X.; Zhang, L.N.; Lv, Y.H.; Cui, H.D.; Zheng, J.H. Scutellarin promotes in vitro angiogenesis in human umbilical vein endothelial cells. Biochem. Biophys. Res. Commun. 2010, 400, 151–156. [Google Scholar]
- Wang, S.; Wang, H.; Guo, H.; Kang, L.; Gao, X.; Hu, L. Neuroprotection of scutellarin is mediated by inhibition of microglial inflammatory activation. Neuroscience 2011, 185, 150–160. [Google Scholar] [CrossRef] [PubMed]
- Guo, L.L.; Guan, Z.Z.; Wang, Y.L. Scutellarin protects against abeta-induced learning and memory deficits in rats: Involvement of nicotinic acetylcholine receptors and cholinesterase. Acta Pharmacol. Sin. 2011, 32, 1446–1453. [Google Scholar] [CrossRef] [PubMed]
- Dahlgren, K.N.; Manelli, A.M.; Stine, W.B.; Baker, L.K.; Krafft, G.A.; LaDu, M.J. Oligomeric and fibrillar species of amyloid-beta peptides differentially affect neuronal viability. J. Biol. Chem. 2002, 277, 32046–32053. [Google Scholar] [CrossRef] [PubMed]
- Hudson, S.A.; Ecroyd, H.; Kee, T.W.; Carver, J.A. The thioflavin t fluorescence assay for amyloid fibril detection can be biased by the presence of exogenous compounds. FEBS J. 2009, 276, 5960–5972. [Google Scholar] [CrossRef] [PubMed]
- Jankowsky, J.L.; Slunt, H.H.; Ratovitski, T.; Jenkins, N.A.; Copeland, N.G.; Borchelt, D.R. Co-expression of multiple transgenes in mouse cns: A comparison of strategies. Biomol. Eng. 2001, 17, 157–165. [Google Scholar] [CrossRef]
- Lin, L.L.; Liu, A.J.; Liu, J.G.; Yu, X.H.; Qin, L.P.; Su, D.F. Protective effects of scutellarin and breviscapine on brain and heart ischemia in rats. J. Cardiovasc. Pharmacol. 2007, 50, 327–332. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Y.Q.; Wang, Y.J.; Zhou, X.F. Effects of (-)Epicatechin on the Pathology of APP/PS1 Transgenic Mice. Front Neurol. 2014, 5, 69. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.J.; Pollard, A.; Zhong, J.H.; Dong, X.Y.; Wu, X.B.; Zhou, H.D.; Zhou, X.F. Intramuscular delivery of a single chain antibody gene reduces brain abeta burden in a mouse model of Alzheimer’s disease. Neurobiol. Aging 2009, 30, 364–376. [Google Scholar] [CrossRef] [PubMed]
- Griffin, W.S.T. Inflammation and neurodegenerative diseases. Am. J. Clin. Nutr. 2006, 83, 5960–5972. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, M.; Davis, J.; Aucoin, D.; Sato, T.; Ahuja, S.; Aimoto, S.; Elliott, J.I.; Van Nostrand, W.E.; Smith, S.O. Structural conversion of neurotoxic amyloid-beta(1-42) oligomers to fibrils. Nat. Struct. Mol. Biol. 2010, 17, 561–567. [Google Scholar] [CrossRef] [PubMed]
- Goure, W.F.; Krafft, G.A.; Jerecic, J.; Hefti, F. Targeting the proper amyloid-beta neuronal toxins: A path forward for Alzheimer’s disease immunotherapeutics. Alzheimer Res. Ther. 2014, 6, 42. [Google Scholar] [CrossRef] [PubMed]
- Minter, M.R.; Taylor, J.M.; Crack, P.J. The contribution of neuroinflammation to amyloid toxicity in Alzheimer’s disease. J. Neurochem. 2016, 136, 457–474. [Google Scholar] [CrossRef] [PubMed]
- Heneka, M.T.; Carson, M.J.; Khoury, J.E.; Landreth, G.E.; Brosseron, F.; Feinstein, D.L. Neuroinflammation in Alzheimer’s disease. Lancet Neurol. 2015, 14, 388–405. [Google Scholar] [CrossRef]
- Qu, J.; Wang, Y.; Luo, G. Determination of scutellarin in erigeron breviscapus extract by liquid chromatography–tandem mass spectrometry. J. Chromatogr. A 2001, 919, 437–441. [Google Scholar] [CrossRef]
- Dajas, F.; Juan Andres, A.C.; Florencia, A.; Carolina, E.; Felicia, R.M. Neuroprotective actions of flavones and flavonols: Mechanisms and relationship to flavonoid structural features. Cent. Nerv. Syst. Agents Med. Chem. 2013, 13, 30–35. [Google Scholar] [CrossRef] [PubMed]
- Guo, L.L.; Guan, Z.Z.; Huang, Y.; Wang, Y.L.; Shi, J.S. The neurotoxicity of beta-amyloid peptide toward rat brain is associated with enhanced oxidative stress, inflammation and apoptosis, all of which can be attenuated by scutellarin. Exp. Toxicol. Pathol. 2013, 65, 579–584. [Google Scholar] [CrossRef] [PubMed]
- Yuan, Y.R.P.; Kan, E.M. Scutellarin regulates the notch pathway and affects the migration and morphological transformation of activated microglia in experimentally induced cerebral ischemia in rats and in activated bv-2 microglia. J. Neuroinflamm. 2015, 12, 11. [Google Scholar] [CrossRef] [PubMed]
- Xu, S.L.; Bi, C.W.; Choi, R.C.; Zhu, K.Y.; Miernisha, A.; Dong, T.T.; Tsim, K.W. Flavonoids induce the synthesis and secretion of neurotrophic factors in cultured rat astrocytes: A signaling response mediated by estrogen receptor. Evid. Based Complement. Altern. Med. 2013, 2013. [Google Scholar] [CrossRef] [PubMed]
- Chai, L.; Guo, H.; Li, H.; Wang, S.; Wang, Y.L.; Shi, F.; Hu, L.M.; Liu, Y.; Adah, D. Scutellarin and caffeic acid ester fraction, active components of dengzhanxixin injection, upregulate neurotrophins synthesis and release in hypoxia/reoxygenation rat astrocytes. J. Ethnopharmacol. 2013, 150, 100–107. [Google Scholar] [CrossRef] [PubMed]
- Mori, T.; Koyama, N.; Guillot-Sestier, M.V.; Tan, J.; Town, T. Ferulic acid is a nutraceutical beta-secretase modulator that improves behavioral impairment and Alzheimer-like pathology in transgenic mice. PLoS ONE 2013, 8, e55774. [Google Scholar] [CrossRef] [PubMed]
- Currais, A.; Prior, M.; Dargusch, R.; Armando, A.; Ehren, J.; Schubert, D.; Quehenberger, O.; Maher, P. Modulation of p25 and inflammatory pathways by fisetin maintains cognitive function in Alzheimer’s disease transgenic mice. Aging Cell 2014, 13, 379–390. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Cui, L.; Duan, X.; Ma, B.; Zhong, D. Pharmacokinetics and metabolism of the flavonoid scutellarin in humans after a single oral administration. Drug Metab. Dispos. 2006, 34, 1345–1352. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are available from the authors. |

© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zeng, Y.-Q.; Cui, Y.-B.; Gu, J.-H.; Liang, C.; Zhou, X.-F. Scutellarin Mitigates Aβ-Induced Neurotoxicity and Improves Behavior Impairments in AD Mice. Molecules 2018, 23, 869. https://doi.org/10.3390/molecules23040869
Zeng Y-Q, Cui Y-B, Gu J-H, Liang C, Zhou X-F. Scutellarin Mitigates Aβ-Induced Neurotoxicity and Improves Behavior Impairments in AD Mice. Molecules. 2018; 23(4):869. https://doi.org/10.3390/molecules23040869
Chicago/Turabian StyleZeng, Yue-Qin, Yin-Bo Cui, Juan-Hua Gu, Chen Liang, and Xin-Fu Zhou. 2018. "Scutellarin Mitigates Aβ-Induced Neurotoxicity and Improves Behavior Impairments in AD Mice" Molecules 23, no. 4: 869. https://doi.org/10.3390/molecules23040869
APA StyleZeng, Y.-Q., Cui, Y.-B., Gu, J.-H., Liang, C., & Zhou, X.-F. (2018). Scutellarin Mitigates Aβ-Induced Neurotoxicity and Improves Behavior Impairments in AD Mice. Molecules, 23(4), 869. https://doi.org/10.3390/molecules23040869




