Dual Effects of Alpha-Hydroxy Acids on the Skin
Abstract
:1. Introduction
2. Role of AHAs in the Biological Responses of Skin Cells
2.1. The Effectiveness of AHAs
2.2. The Safety of AHA
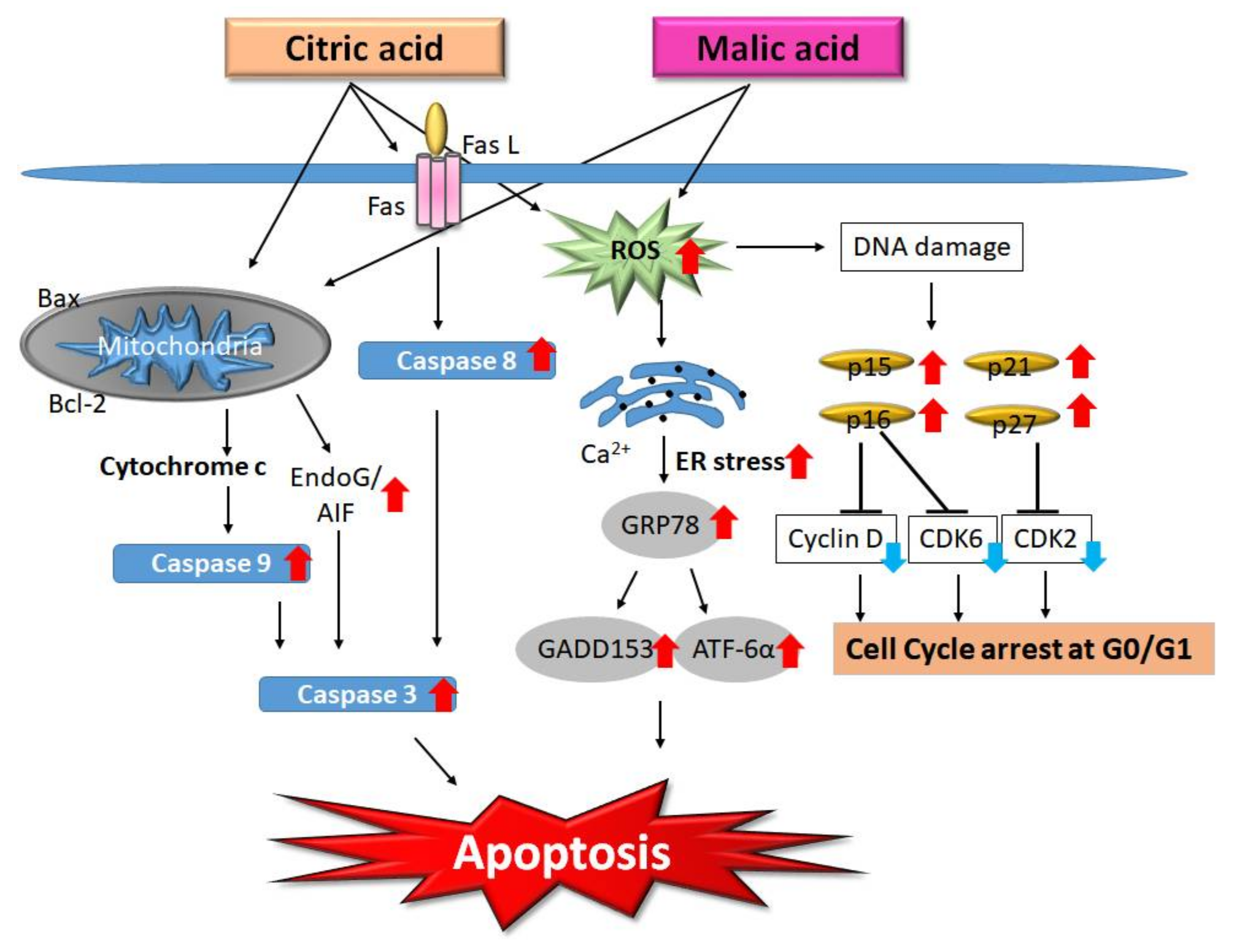
2.3. AHA-Induced Apoptosis
2.4. Lactic Acid and Skin Microbiota
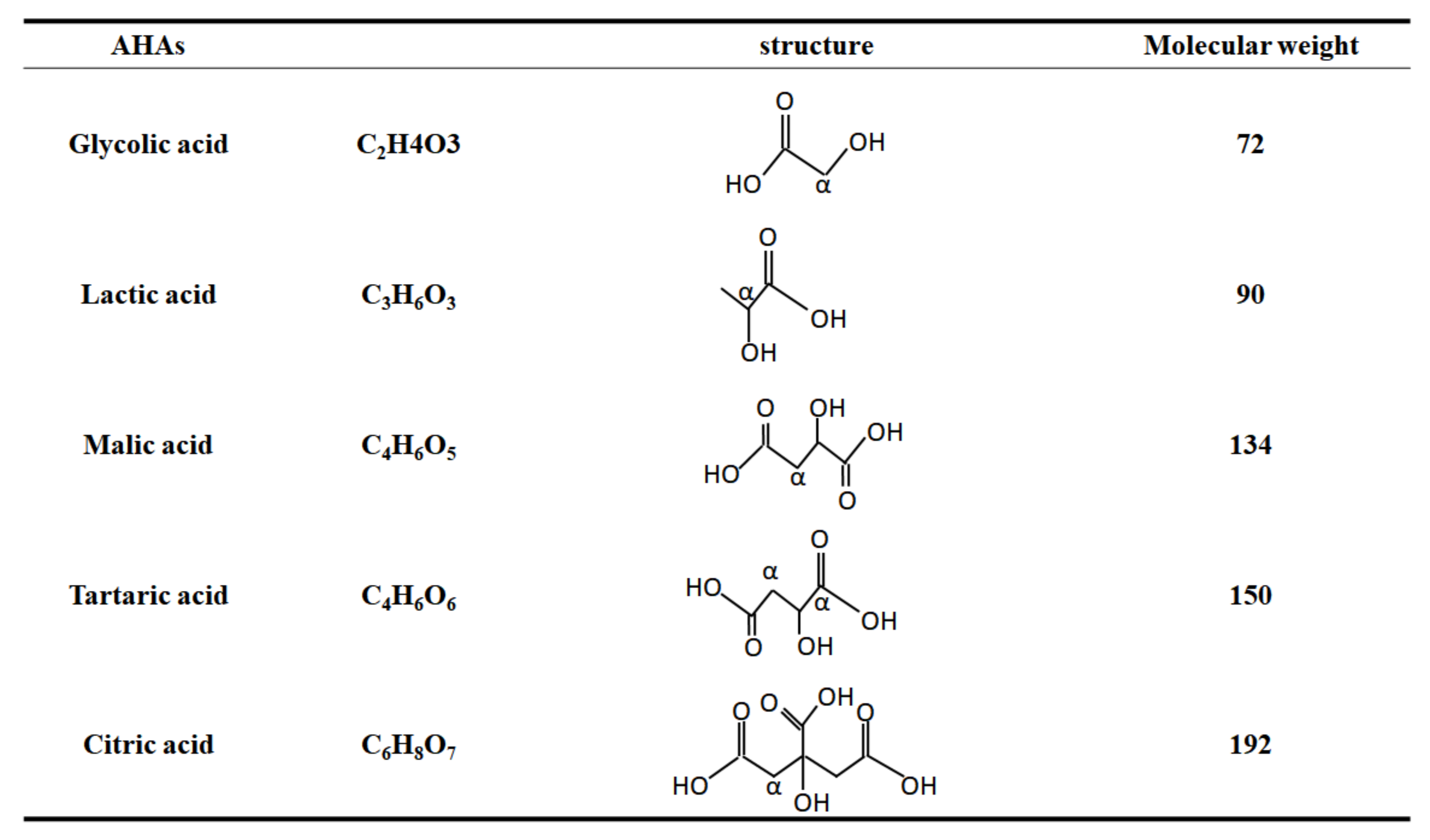
2.5. The Forms of AHAs
3. AHAs, Peeling, and UV Irradiation
3.1. Clinical Peeling Concentration of AHAs
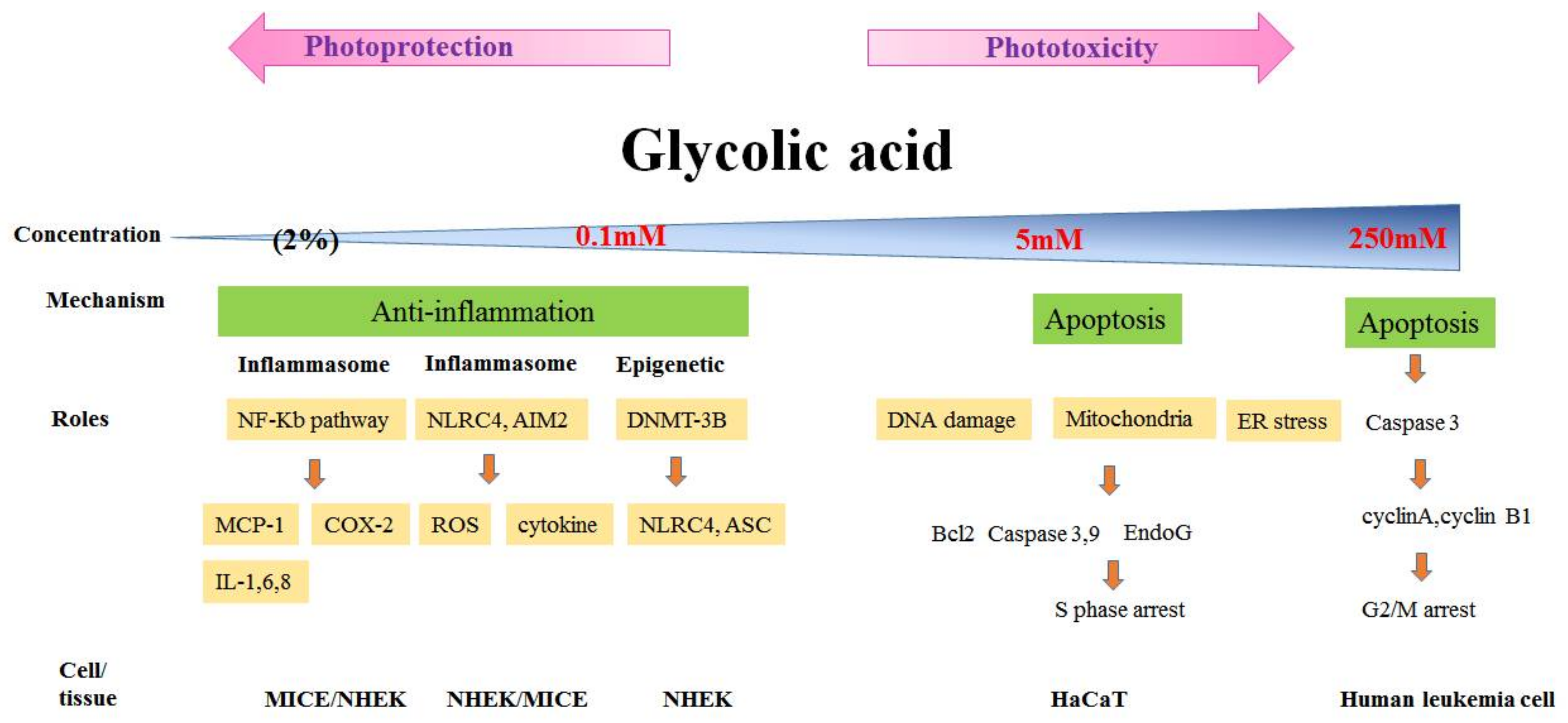
3.2. Phototoxicity and Photoprotection of GA
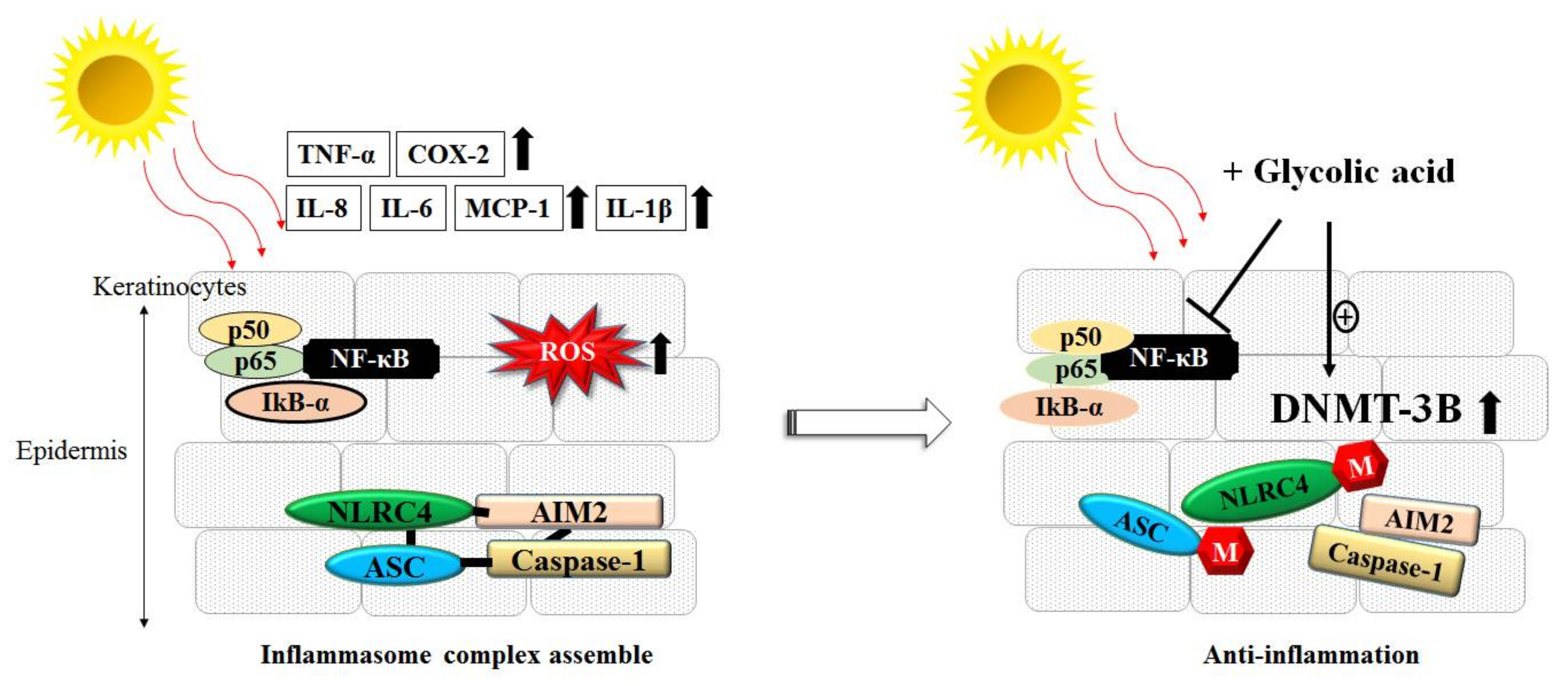
3.3. Inflammation, ROS and AHAs
3.4. GA and in Vivo Study
4. Future Prospects
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| AD | atopic dermatitis |
| AHAs | alpha-hydroxy acids |
| BID | BH3-interacting domain death agonist protein |
| CA | citric acid |
| ELISA | enzyme-linked immunosorbent assay |
| Endo G | endonuclease G |
| GA | glycolic acid |
| GM-CSF | granulocyte-macrophage colony-stimulating factor |
| IL | interleukin |
| LA | lactic acid |
| MA | malic acid |
| MCP-1 | monocyte chemoattractant protein-1 |
| MED | minimal erythemal dose |
| MMP | matrix metalloproteinase |
| NF-kB | nuclear factor-kappa B |
| NHEK | normal human epidermal keratinocyte |
| ROS | reactive oxygen species |
| SBCs | sunburn cells |
| SP | Substance P |
| TNF | tumor necrosis factor |
| UVB | ultraviolet radiation B |
References
- Andersen, F.A. Final report on the safety assessment of glycolic acid, ammonium, calcium, potassium, and sodium glycolates, methyl, ethyl, propyl, and butyl glylates, and lactic acid, ammonium, calcium, potassium, sodium, and TEA-lactates, methyl, ethyl, isopropyl, and butyl lactates, and lauryl, myristyl, and cetyllactates. Int. J. Toxicol. 1998, 17, 1–241. [Google Scholar]
- Moy, L.S.; Murad, H.; Moy, R.L. Glycolic acid peels for the treatment of wrinkles and photoaging. J. Dermatol. Surg. Oncol. 1993, 19, 243–246. [Google Scholar] [CrossRef] [PubMed]
- Sharad, J. Glycolic acid peel therapy—A current review. Clin. Cosmet. Investig. Dermatol. 2013, 6, 281–288. [Google Scholar] [CrossRef] [PubMed]
- Ditre, C.M.; Griffin, T.D.; Murphy, G.F.; Sueki, H.; Telegan, B.; Johnson, W.C.; Yu, R.J.; Van Scott, E.J. Effects of alpha-hydroxy acids on photoaged skin: A pilot clinical, histologic, and ultrastructural study. J. Am. Acad. Dermatol. 1996, 34 Pt 1, 187–195. [Google Scholar] [CrossRef]
- National Toxicology Program. Photocarcinogenesis study of glycolic acid and salicylic acid (CAS Nos. 79-14-1 and 69-72-7) in SKH-1 mice (simulated solar light and topical application study). Natl. Toxicol. Progr. Tech. Rep. Ser. 2007, 524, 1–242. [Google Scholar]
- Morreale, M.; Livrea, M.A. Synergistic effect of glycolic acid on the antioxidant activity of alpha-tocopherol and melatonin in lipid bilayers and in human skin homogenates. Biochem. Mol. Biol. Int. 1997, 42, 1093–1102. [Google Scholar] [PubMed]
- Kaidbey, K.; Sutherland, B.; Bennett, P.; Wamer, W.G.; Barton, C.; Dennis, D.; Kornhauser, A. Topical glycolic acid enhances photodamage by ultraviolet light. Photodermatol. Photoimmunol. Photomed. 2003, 19, 21–27. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.H.; Chou, C.C.; Cheng, Y.W.; Sheen, L.Y.; Chou, M.C.; Yu, H.S.; Wei, Y.H.; Chung, J.G. Effects of glycolic acid on the induction of apoptosis via caspase-3 activation in human leukemia cell line (HL-60). Food Chem. Toxicol. 2004, 42, 1777–1784. [Google Scholar] [CrossRef] [PubMed]
- Lai, W.W.; Hsiao, Y.P.; Chung, J.G.; Wei, Y.H.; Cheng, Y.W.; Yang, J.H. Synergistic phototoxic effects of glycolic acid in a human keratinocyte cell line (HaCaT). J. Dermatol. Sci. 2011, 64, 191–198. [Google Scholar] [CrossRef] [PubMed]
- Tang, S.C.; Yeh, J.I.; Hung, S.J.; Hsiao, Y.P.; Liu, F.T.; Yang, J.H. Glycolic Acid Silences Inflammasome Complex Genes, NLRC4 and ASC, by Inducing DNA Methylation in HaCaT Cells. DNA Cell Biol. 2016, 35, 124–134. [Google Scholar] [CrossRef] [PubMed]
- Hung, S.J.; Tang, S.C.; Liao, P.Y.; Ge, J.S.; Hsiao, Y.P.; Yang, J.H. Photoprotective Potential of Glycolic Acid by Reducing NLRC4 and AIM2 Inflammasome Complex Proteins in UVB Radiation-Induced Normal Human Epidermal Keratinocytes and Mice. DNA Cell Biol. 2017, 36, 177–187. [Google Scholar] [CrossRef] [PubMed]
- Tang, S.C.; Liao, P.Y.; Hung, S.J.; Ge, J.S.; Chen, S.M.; Lai, J.C.; Hsiao, Y.P.; Yang, J.H. Topical application of glycolic acid suppresses the UVB induced IL-6, IL-8, MCP-1 and COX-2 inflammation by modulating NF-kappaB signaling pathway in keratinocytes and mice skin. J. Dermatol. Sci. 2017, 86, 238–248. [Google Scholar] [CrossRef] [PubMed]
- Lo, C.; Lai, T.Y.; Yang, J.H.; Yang, J.S.; Ma, Y.S.; Weng, S.W.; Chen, Y.Y.; Lin, J.G.; Chung, J.G. Gallic acid induces apoptosis in A375.S2 human melanoma cells through caspase-dependent and -independent pathways. Int. J. Oncol. 2010, 37, 377–385. [Google Scholar] [PubMed]
- Lo, C.; Lai, T.Y.; Yang, J.S.; Yang, J.H.; Ma, Y.S.; Weng, S.W.; Lin, H.Y.; Chen, H.Y.; Lin, J.G.; Chung, J.G. Gallic acid inhibits the migration and invasion of A375.S2 human melanoma cells through the inhibition of matrix metalloproteinase-2 and Ras. Melanoma Res. 2011, 21, 267–273. [Google Scholar] [CrossRef] [PubMed]
- Ying, T.H.; Chen, C.W.; Hsiao, Y.P.; Hung, S.J.; Chung, J.G.; Yang, J.H. Citric acid induces cell-cycle arrest and apoptosis of human immortalized keratinocyte cell line (HaCaT) via caspase- and mitochondrial-dependent signaling pathways. Anticancer Res. 2013, 33, 4411–4420. [Google Scholar] [PubMed]
- Hsiao, Y.P.; Lai, W.W.; Wu, S.B.; Tsai, C.H.; Tang, S.C.; Chung, J.G.; Yang, J.H. Triggering apoptotic death of human epidermal keratinocytes by malic Acid: Involvement of endoplasmic reticulum stress- and mitochondria-dependent signaling pathways. Toxins 2015, 7, 81–96. [Google Scholar] [CrossRef] [PubMed]
- Hsiao, Y.P.; Huang, H.L.; Lai, W.W.; Chung, J.G.; Yang, J.H. Antiproliferative effects of lactic acid via the induction of apoptosis and cell cycle arrest in a human keratinocyte cell line (HaCaT). J. Dermatol. Sci. 2009, 54, 175–184. [Google Scholar] [CrossRef] [PubMed]
- Parker, E.T.; Cleaves, H.J., 2nd; Bada, J.L.; Fernandez, F.M. Quantitation of alpha-hydroxy acids in complex prebiotic mixtures via liquid chromatography/tandem mass spectrometry. Rapid Commun. Mass Spectrom. 2016, 30, 2043–2051. [Google Scholar] [CrossRef] [PubMed]
- Van Scott, E.J.; Yu, R.J. Hyperkeratinization, corneocyte cohesion, and alpha hydroxy acids. J. Am. Acad. Dermatol. 1984, 11 Pt 1, 867–879. [Google Scholar] [CrossRef]
- Gupta, R.R.; Mahajan, B.B.; Garg, G. Chemical peeling—Evaluation of glycolic acid in varying concentrations and time intervals. Indian J. Dermatol. Venereol. Leprol. 2001, 67, 28–29. [Google Scholar] [PubMed]
- De Gruijl, F.R.; van Kranen, H.J.; Mullenders, L.H. UV-induced DNA damage, repair, mutations and oncogenic pathways in skin cancer. J. Photochem. Photobiol. B Biol. 2001, 63, 19–27. [Google Scholar] [CrossRef]
- Decker, R.H. Nature and regulation of energy metabolism in the epidermis. J. Investig. Dermatol. 1971, 57, 351–363. [Google Scholar] [CrossRef] [PubMed]
- Falchi, M.; Bertelli, A.; Lo Scalzo, R.; Morassut, M.; Morelli, R.; Das, S.; Cui, J.; Das, D.K. Comparison of cardioprotective abilities between the flesh and skin of grapes. J. Agric. Food Chem. 2006, 54, 6613–6622. [Google Scholar] [CrossRef] [PubMed]
- Fiume, M.M.; Heldreth, B.A.; Bergfeld, W.F.; Belsito, D.V.; Hill, R.A.; Klaassen, C.D.; Liebler, D.C.; Marks, J.G., Jr.; Shank, R.C.; Slaga, T.J.; et al. Safety Assessment of Citric Acid, Inorganic Citrate Salts, and Alkyl Citrate Esters as Used in Cosmetics. Int. J. Toxicol. 2014, 33 (Suppl. S2), 16S–46S. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, Y.; Uede, K.; Yonei, N.; Kishioka, A.; Ohtani, T.; Furukawa, F. Effects of alpha-hydroxy acids on the human skin of Japanese subjects: The rationale for chemical peeling. J. Dermatol. 2006, 33, 16–22. [Google Scholar] [CrossRef] [PubMed]
- Yu, R.J.; Van Scott, E.J. Alpha-hydroxyacids and carboxylic acids. J. Cosmet. Dermatol. 2004, 3, 76–87. [Google Scholar] [CrossRef] [PubMed]
- Eberle, J.; Fecker, L.F.; Forschner, T.; Ulrich, C.; Rowert-Huber, J.; Stockfleth, E. Apoptosis pathways as promising targets for skin cancer therapy. Br. J. Dermatol. 2007, 156 (Suppl. S3), 18–24. [Google Scholar] [CrossRef] [PubMed]
- Sousa, L.Q.; Machado, K.D.; Oliveira, S.F.; Araujo, L.D.; Moncao-Filho, E.D.; Melo-Cavalcante, A.A.; Vieira-Junior, G.M.; Ferreira, P.M. Bufadienolides from amphibians: A promising source of anticancer prototypes for radical innovation, apoptosis triggering and Na+/K+-ATPase inhibition. Toxicon 2017, 127, 63–76. [Google Scholar] [CrossRef] [PubMed]
- Boukamp, P.; Petrussevska, R.T.; Breitkreutz, D.; Hornung, J.; Markham, A.; Fusenig, N.E. Normal keratinization in a spontaneously immortalized aneuploid human keratinocyte cell line. J. Cell Biol. 1988, 106, 761–771. [Google Scholar] [CrossRef] [PubMed]
- Wilson, G.; Davis, S.S.; Illum, L.; Zweibaum, A. (Eds.) Human Keratinocyte Cell Lines. In Pharmaceutical Applications of Cell and Tissue Culture to Drug Transport; Springer: New York, NY, USA, 1991; pp. 283–284. [Google Scholar]
- Sporl, F.; Schellenberg, K.; Blatt, T.; Wenck, H.; Wittern, K.P.; Schrader, A.; Kramer, A. A circadian clock in HaCaT keratinocytes. J. Investig. Dermatol. 2011, 131, 338–348. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Fandos, E.; Herrera, B. Efficacy of malic acid against Listeria monocytogenes attached to poultry skin during refrigerated storage. Poultry Sci. 2013, 92, 1936–1941. [Google Scholar] [CrossRef] [PubMed]
- Smith, W.P. Comparative effectiveness of alpha-hydroxy acids on skin properties. Int. J. Cosmet. Sci. 1996, 18, 75–83. [Google Scholar] [CrossRef] [PubMed]
- Gueniche, A.; Bastien, P.; Ovigne, J.M.; Kermici, M.; Courchay, G.; Chevalier, V.; Breton, L.; Castiel-Higounenc, I. Bifidobacterium longum lysate, a new ingredient for reactive skin. Exp. Dermatol. 2010, 19, e1–e8. [Google Scholar] [CrossRef] [PubMed]
- Ai, C.; Ma, N.; Zhang, Q.; Wang, G.; Liu, X.; Tian, F.; Chen, P.; Chen, W. Immunomodulatory Effects of Different Lactic Acid Bacteria on Allergic Response and Its Relationship with In Vitro Properties. PLoS ONE 2016, 11, e0164697. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, M.; Ebata, T.; Hirooka, J.; Hosoya, R.; Inoue, N.; Itami, S.; Tsuji, K.; Yaginuma, T.; Muramatsu, K.; Nakamura, A.; Fujita, A.; Nagakura, T. Antipruritic effects of the probiotic strain LKM512 in adults with atopic dermatitis. Ann. Allergy Asthma Immunol. 2014, 113, 209–216. [Google Scholar] [CrossRef] [PubMed]
- Mijouin, L.; Hillion, M.; Ramdani, Y.; Jaouen, T.; Duclairoir-Poc, C.; Follet-Gueye, M.L.; Lati, E.; Yvergnaux, F.; Driouich, A.; Lefeuvre, L.; et al. Effects of a skin neuropeptide (substance p) on cutaneous microflora. PLoS ONE 2013, 8, e78773. [Google Scholar] [CrossRef] [PubMed]
- Margolis, K.G.; Gershon, M.D. Neuropeptides and inflammatory bowel disease. Curr. Opin. Gastroenterol. 2009, 25, 503–511. [Google Scholar] [CrossRef] [PubMed]
- Biaggini, K.; Borrel, V.; Szunerits, S.; Boukherroub, R.; N’Diaye, A.; Zebre, A.; Bonnin-Jusserand, M.; Duflos, G.; Feuilloley, M.; Drider, D.; et al. Substance P enhances lactic acid and tyramine production in Enterococcus faecalis V583 and promotes its cytotoxic effect on intestinal Caco-2/TC7 cells. Gut Pathog. 2017, 9, 20. [Google Scholar] [CrossRef] [PubMed]
- Middleton, J.D. Sodium lactate as a moisturiser. Cosmet. Toiletries 1978, 93, 85–86. [Google Scholar]
- Lask, G.; Fournier, N.; Trelles, M.; Elman, M.; Scheflan, M.; Slatkine, M.; Naimark, J.; Harth, Y. The utilization of nonthermal blue (405–425 nm) and near infrared (850–890 nm) light in aesthetic dermatology and surgery—A multicenter study. J. Cosmet. Laser Ther. 2005, 7, 163–170. [Google Scholar] [CrossRef] [PubMed]
- Davidson, T.M.; Wolfe, D.P. Sunscreens, Skin Cancer, and Your Patient. Phys. Sportsmed. 1986, 14, 65–79. [Google Scholar] [CrossRef] [PubMed]
- Antoniou, C.; Kosmadaki, M.G.; Stratigos, A.J.; Katsambas, A.D. Photoaging: Prevention and topical treatments. Am. J. Clin. Dermatol. 2010, 11, 95–102. [Google Scholar] [CrossRef] [PubMed]
- Kurtzweil, P. Alpha hydroxy acids for skin care. FDA Consum. 1998, 32, 30–35. [Google Scholar] [PubMed]
- Taiwan Ministry of Health and Welfare Commercially Available Alpha-Hydroxy Acids-Containing Cosmetic Ingredients Have Complete Management Practices. Available online: http://cosmetic-recall.fda.gov.tw (accessed on 9 September 2014).
- Bernstein, E.F.; Lee, J.; Brown, D.B.; Yu, R.; Van Scott, E. Glycolic acid treatment increases type I collagen mRNA and hyaluronic acid content of human skin. Dermatol. Surg. 2001, 27, 429–433. [Google Scholar] [PubMed]
- Ahn, K.S.; Park, K.S.; Jung, K.M.; Jung, H.K.; Lee, S.H.; Chung, S.Y.; Yang, K.H.; Yun, Y.P.; Pyo, H.B.; Park, Y.K.; et al. Inhibitory effect of glycolic acid on ultraviolet B-induced c-fos expression, AP-1 activation and p53–p21 response in a human keratinocyte cell line. Cancer Lett. 2002, 186, 125–135. [Google Scholar] [CrossRef]
- Watson, R.E.; Gibbs, N.K.; Griffiths, C.E.; Sherratt, M.J. Damage to skin extracellular matrix induced by UV exposure. Antioxid. Redox Signal. 2014, 21, 1063–1077. [Google Scholar] [CrossRef] [PubMed]
- Kennedy Crispin, M.; Fuentes-Duculan, J.; Gulati, N.; Johnson-Huang, L.M.; Lentini, T.; Sullivan-Whalen, M.; Gilleaudeau, P.; Cueto, I.; Suarez-Farinas, M.; Lowes, M.A.; et al. Gene profiling of narrowband UVB-induced skin injury defines cellular and molecular innate immune responses. J. Investig. Dermatol. 2013, 133, 692–701. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.; He, Y.Y. Ultraviolet radiation-induced non-melanoma skin cancer: Regulation of DNA damage repair and inflammation. Genes Dis. 2014, 1, 188–198. [Google Scholar] [CrossRef] [PubMed]
- Nishisgori, C. Current concept of photocarcinogenesis. Photochem. Photobiol. Sci. 2015, 14, 1713–1721. [Google Scholar] [CrossRef] [PubMed]
- Cueni, L.N.; Detmar, M. New insights into the molecular control of the lymphatic vascular system and its role in disease. J. Investig. Dermatol. 2006, 126, 2167–2177. [Google Scholar] [CrossRef] [PubMed]
- Hong, J.T.; Kim, E.J.; Ahn, K.S.; Jung, K.M.; Yun, Y.P.; Park, Y.K.; Lee, S.H. Inhibitory effect of glycolic acid on ultraviolet-induced skin tumorigenesis in SKH-1 hairless mice and its mechanism of action. Mol. Carcinog. 2001, 31, 152–160. [Google Scholar] [CrossRef] [PubMed]
- Moran, M.M. TRP Channels as Potential Drug Targets. Annu. Rev. Pharmacol. Toxicol. 2018, 58, 309–330. [Google Scholar] [CrossRef] [PubMed]
- Cao, X.; Yang, F.; Zheng, J.; Wang, K. Intracellular proton-mediated activation of TRPV3 channels accounts for the exfoliation effect of alpha-hydroxyl acids on keratinocytes. J. Biol. Chem. 2012, 287, 25905–25916. [Google Scholar] [CrossRef] [PubMed]
- Hara-Chikuma, M.; Verkman, A.S. Aquaporin-3 functions as a glycerol transporter in mammalian skin. Biol. Cell 2005, 97, 479–486. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tang, S.-C.; Yang, J.-H. Dual Effects of Alpha-Hydroxy Acids on the Skin. Molecules 2018, 23, 863. https://doi.org/10.3390/molecules23040863
Tang S-C, Yang J-H. Dual Effects of Alpha-Hydroxy Acids on the Skin. Molecules. 2018; 23(4):863. https://doi.org/10.3390/molecules23040863
Chicago/Turabian StyleTang, Sheau-Chung, and Jen-Hung Yang. 2018. "Dual Effects of Alpha-Hydroxy Acids on the Skin" Molecules 23, no. 4: 863. https://doi.org/10.3390/molecules23040863
APA StyleTang, S.-C., & Yang, J.-H. (2018). Dual Effects of Alpha-Hydroxy Acids on the Skin. Molecules, 23(4), 863. https://doi.org/10.3390/molecules23040863



