β-Glucans: Multi-Functional Modulator of Wound Healing
Abstract
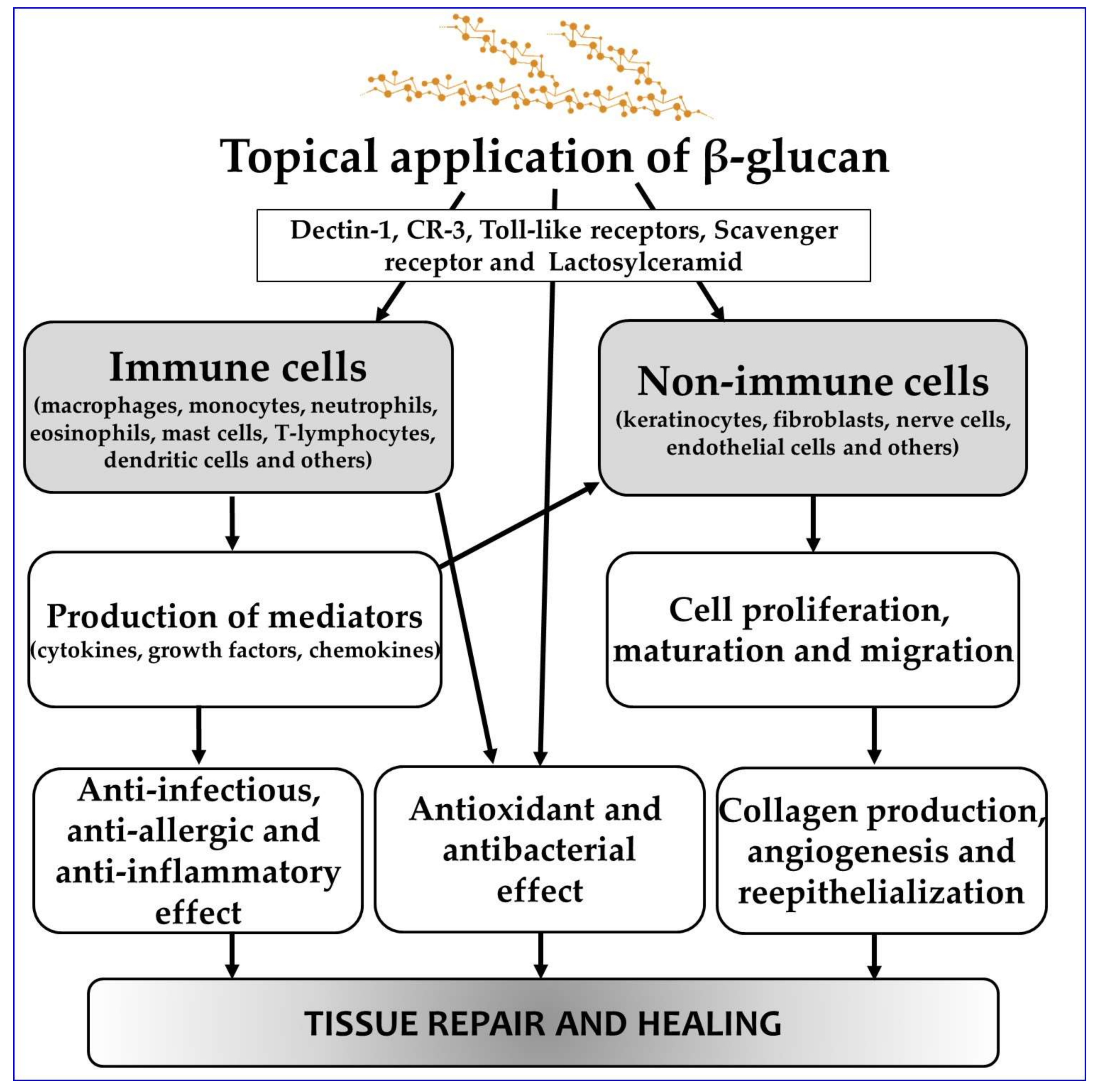
1. Introduction
2. Wound Healing Activity of β-Glucans: In Vitro Experiments
3. Wound Healing Activity of β-Glucans: Animal Studies
4. Clinical Evidence for the Use of β-Glucans in the Management of Human Non-Healing Wounds and Burns
5. Sterilization of β-Glucans and Their Impact on β-Glucan Bioactivity
Gamma Irradiation
6. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Bohn, J.A.; BeMiller, J.N. (1→3)-b-d-Glucans as biological response modifiers: A review of structure-functional activity relationships. Carbohydr. Polym. 1995, 28, 3–14. [Google Scholar] [CrossRef]
- Pretus, H.A.; Ensley, H.E.; McNamee, R.B.; Jones, E.L.; Browder, I.W.; Williams, D.L. Isolation, physicochemical characterization and preclinical efficacy evaluation of soluble scleroglucan. J. Pharmacol. Exp. Ther. 1991, 257, 500–510. [Google Scholar] [PubMed]
- Williams, J.D.; Mueller, A.; Browder, W. Glucan-based macrophage stimulators. A review of their anti-infective potential. Clin. Immunother. 1996, 5, 392–399. [Google Scholar] [CrossRef]
- Browder, W.; Williams, D.; Pretus, H.; Olivero, G.; Enrichens, F.; Mao, P.; Franchello, A. Beneficial effect of enhanced macrophage function in trauma patient. Ann. Surg. 1990, 211, 605–613. [Google Scholar] [PubMed]
- Sherwood, E.R.; Williams, D.L.; DiLuzio, N.R. Glucan stimulates production of antitumor cytolitic/cytostatic factor(s) by macrophages. J. Biol. Response Mod. 1986, 5, 504–526. [Google Scholar] [PubMed]
- Sherwood, E.R.; Williams, D.L.; McNamee, R.B.; Jones, E.L.; Browder, I.W.; DiLuzio, N.R. In vitro tumoricidal activity of resting and glucan-activated Kupffer cells. J. Leukoc. Biol. 1987, 42, 69–75. [Google Scholar] [CrossRef] [PubMed]
- Stier, H.; Ebbeskotte, V.; Gruenwald, J. Immune-modulatory effects of dietary Yeast Beta-1,3/1,6-D-glucan. Nutr. J. 2014, 13, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Sheng, X.; Shi, A.; Hu, H.; Yang, Y.; Liu, L.; Fei, L.; Liu, H. β-Glucans: Relationships between modification, conformation and functional activities. Molecules 2017, 22, 257. [Google Scholar] [CrossRef] [PubMed]
- Bloebaum, R.M.; Grant, J.A.; Sur, S. Immunomodulation: The future of allergy and asthma treatment. Curr. Opin. Allergy Clin. Immunol. 2004, 4, 63–67. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.; Zhao, Y. The biological role of dectin-1 in immune response. Int. Rev. Immunol. 2007, 26, 349–364. [Google Scholar] [CrossRef] [PubMed]
- Kanlayavattanakul, M.; Lourith, N. Carboxymethylglucan in cosmetics. Thai Pharm. Health Sci. J. 2008, 3, 378–382. [Google Scholar]
- Gautier, S.; Xhauflaire-Uhoda, E.; Gonry, P.; Pierard, G.E. Chitin-glucan, a natural cell scaffold for skin moisturization and rejuvenation. Int. J. Cosmet. Sci. 2008, 30, 459–469. [Google Scholar] [CrossRef] [PubMed]
- Jesenak, M.; Urbancek, M.; Majtan, J.; Banovcin, P.; Hercogova, J. β-Glucan-based cream (containing pleuran isolated from Pleurotus ostreatus) in supportive treatment of mild-to-moderate atopic dermatitis. J. Dermatol. Treat. 2016, 27, 351–354. [Google Scholar] [CrossRef] [PubMed]
- Jantova, S.; Bakos, D.; Birosova, L.; Matejov, P. Biological properties of a novel coladerm-beta glucan membrane. In vitro assessment using human fibroblasts. Biomed. Pap. Med. Fac. Univ. Palacky Olomouc 2015, 159, 67–76. [Google Scholar]
- Singer, A.J.; Clark, R.A. Cutaneous wound healing. N. E. J. Med. 1999, 341, 738–746. [Google Scholar] [CrossRef] [PubMed]
- Falanga, V. Growth factors and wound healing. J. Dermatol. Surg. Oncol. 1993, 19, 711–714. [Google Scholar] [CrossRef] [PubMed]
- Davis, S.C.; Perez, R. Cosmeceuticals and natural products: Wound healing. Clin. Dermatol. 2009, 27, 502–506. [Google Scholar] [CrossRef] [PubMed]
- Lundahl, M.L.E.; Scanlan, E.M.; Lavelle, E.C. Therapeutic potential of carbohydrates as regulators of macrophage activation. Biochem. Pharmacol. 2017, 146, 23–41. [Google Scholar] [CrossRef] [PubMed]
- Wei, D.; Zhang, L.; Williams, D.L.; Browder, W. Glucan stimulates human dermal fibroblast collagen biosynthesis through a nuclear factor-1 dependent mechanism. Wound Repair Regen. 2002, 10, 161–168. [Google Scholar] [CrossRef] [PubMed]
- Son, H.J.; Han, D.-W.; Baek, H.S.; Lim, H.R.; Lee, M.H.; Woo, Y.I.; Park, J.-C. Stimulated TNF-alpha release in macrophage and enhanced migration of dermal fibroblast by β-glucan. Curr. Appl. Phys. 2007, 7 (Suppl. 1), 33–36. [Google Scholar] [CrossRef]
- Shin, M.S.; Lee, S.; Lee, K.Y.; Lee, H.G. Structural and biological characterization of aminated-derivatized oat beta-glucan. J. Agric. Food Chem. 2005, 53, 5554–5558. [Google Scholar] [CrossRef] [PubMed]
- Chamidah, A.; Hardoko; Prihanto, A.A.; American Institute of Physics. Antibacterial activities of β–glucan (laminaran) against gram-negative and grampositive bacteria. AIP Conf. Proc. 2017, 1844, 020011. [Google Scholar] [CrossRef]
- Chinnu, K.; Muthukumaran, M.; Mukund, S.; Sivasubramanian, V. Antimicrobial and antifungal activity of isolated betaglucan from chroococcus turgidus. Indian J. Pharm. Sci. Res. 2014, 4, 217–220. [Google Scholar]
- Estrada, A.; Yun, C.H.; Van Kessel, A.; Li, B.; Hauta, S.; Laarveld, B. Immunomodulatory activities of oat beta-glucan in vitro and in vivo. Microbiol. Immunol. 1997, 41, 991–998. [Google Scholar] [CrossRef] [PubMed]
- Hetland, G.; Ohno, N.; Aaberge, I.S.; Lovik, M. Protective effect of beta-glucan against systemic Streptococcus pneumoniae infection in mice. FEMS Immunol. Med. Microbiol. 2000, 27, 111–116. [Google Scholar] [PubMed]
- Basha, R.Y.; Sampath Kumar, T.S.; Doble, M. Electrospun nanofibers of curdlan (β-1,3 glucan) blend as a potential skin scaffold material. Macromol. Mater. Eng. 2017, 302, 1600417. [Google Scholar] [CrossRef]
- Wu, C.; Chen, T.; Xin, Y.; Zhang, Z.; Ren, Z.; Lei, J.; Chu, B.; Wang, Y.; Tang, S. Nanofibrous asymmetric membranes self-organized from chemically heterogeneous electrospun mats for skin tissue engineering. Biomed. Mater. 2016, 11, 035019. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.-S.; Kim, J.W.; Jung, G.-W.; Moon, S.-B.; Cho, H.-R.; Sung, S.H.; Jung, J.J.; Kwon, Y.S.; Ku, S.K.; Sohn, J.-H. Effect of a β-glucan from Aureobasidium on TGF-β1-modulated in vitro dermal wound repair. Toxicol. Environ. Health Sci. 2016, 8, 12–18. [Google Scholar] [CrossRef]
- Nair, A.V.; Raman, M.; Doble, M. Cyclic β-(1→3) (1→6) glucan/carrageenan hydrogels for wound healing applications. RSC Adv. 2016, 6, 98545–98553. [Google Scholar] [CrossRef]
- Van den Berg, L.M.; Zijlstra-Willems, E.M.; Richters, C.D.; Ulrich, M.M.W.; Geijtenbeek, T.B.H. Dectin-1 activation induces proliferation and migration of human keratinocytes enhancing wound re-epithelialization. Cell. Immunol. 2014, 289, 49–54. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.A.; Oh, T.-H.; Choi, J.-S.; Chang, D.-J.; Joo, C.-K. Impact of β-1,3-glucan isolated from Euglena gracilis on corneal epithelial cell migration and on wound healing in a rat alkali burn model. Curr. Eye Res. 2013, 38, 1207–1213. [Google Scholar] [CrossRef] [PubMed]
- Roy, S.; Dickerson, R.; Khanna, S.; Collard, E.; Gnyawali, U.; Gordillo, G.M.; Sen, C.K. Particulate β-glucan induces TNF-α production in wound macrophages via a redox-sensitive NF-κβ-dependent pathway. Wound Repair Regen. 2011, 19, 411–419. [Google Scholar] [CrossRef] [PubMed]
- Vetvicka, V.; Vetvickova, J. β (1-3)-d-glucan affects adipogenesis, wound healing and inflammation. Orient. Pharm. Exp. Med. 2011, 11, 169–175. [Google Scholar] [CrossRef]
- Woo, Y.I.; Park, B.J.; Kim, H.-L.; Lee, M.H.; Kim, J.; Yang, Y.-I.; Kim, J.K.; Tsubaki, K.; Han, D.-W.; Park, J.-C. The biological activities of (1,3)-(1,6)-β-d-glucan and porous electrospun PLGA membranes containing β-glucan in human dermal fibroblasts and adipose tissue-derived stem cells. Biomed. Mater. 2010, 5, 044109. [Google Scholar] [CrossRef] [PubMed]
- Majtan, J.; Kumar, P.; Koller, J.; Dragunova, J.; Gabriz, J. Induction of metalloproteinase 9 secretion from human keratinocytes by pleuran (β-glucan from Pleurotus ostreatus). Z. Naturforsch. C Biosci. 2009, 64, 597–600. [Google Scholar] [CrossRef]
- Son, H.J.; Bae, H.C.; Kim, H.J.; Lee, D.H.; Han, D.-W.; Park, J.-C. Effects of β-glucan on proliferation and migration of fibroblasts. Curr. Appl. Phys. 2005, 5, 468–471. [Google Scholar] [CrossRef]
- Wei, D.; Williams, D.; Browder, W. Activation of AP-1 and SP1 correlates with wound growth factor gene expression in glucan-treated human fibroblasts. Int. Immunopharmacol. 2002, 2, 1163–1172. [Google Scholar] [CrossRef]
- Kougias, P.; Wei, D.; Rice, P.J.; Ensley, H.E.; Kalbfleisch, J.; Williams, D.L.; Browder, I.W. Normal human fibroblasts express pattern recognition receptors for fungal (1→3)-Beta-d-glucans. Infect. Immun. 2001, 69, 3933–3938. [Google Scholar] [CrossRef] [PubMed]
- Zulli, F.; Suter, F. Improving skin function with CM-glucan, a biological response modifier from yeast. Int. J. Cosmet. Sci. 1998, 20, 79–86. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.-W.; Kwon, Y.-S.; Jang, K.-H. Effect of β-glucan originated from Aureobasidium on infected dermal wound healing of the normal nude mouse. J. Vet. Clin. 2012, 29, 464–469. [Google Scholar]
- Gwon, H.-J.; Ann, S.-J.; Jeong, J.-O.; Kim, S.-M.; Park, J.-S.; Jeong, S.I.; Nho, Y.-C.; Lim, Y.-M. Preparation and characterization of radiation fabricated β-glucan Hydrogels for wound dressing. Polymer (Korea) 2017, 41, 586–891. [Google Scholar] [CrossRef]
- Schmidt, J.G.; Andersen, E.W.; Ersbøll, B.K.; Nielsen, M.E. Muscle wound healing in rainbow trout (Oncorhynchus mykiss). Fish Shellfish Immunol. 2016, 48, 273–284. [Google Scholar] [CrossRef] [PubMed]
- Yun, S.; Ku, S.-K.; Kwon, Y.-S. Effect of β-glucan from Aureobasidium on dermal wound healing in diabetic C57BL/KsJ-db/db mouse model. J. Biomed. Res. 2015, 16, 140–145. [Google Scholar] [CrossRef]
- Przybylska-Diaz, D.A.; Schmidt, J.G.; Vera-Jiménez, N.I.; Steinhagen, D.; Nielsen, M.E. β-glucan enriched bath directly stimulates the wound healing process in common carp (Cyprinus carpio L.). Fish Shellfish Immunol. 2013, 35, 998–1006. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, K.; Kimura, T. Orally and topically administered Sparassis crispa (hanabiratake) improved healing of skin wounds in mice with streptozotocin-induced diabetes. Biosci. Biotechnol. Biochem. 2013, 77, 1303–1305. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.-L.; Lee, J.-H.; Lee, M.H.; Kwon, B.J.; Park, J.-C. Evaluation of electrospun (1,3)-(1,6)-β-d-glucans/biodegradable polymer as artificial skin for full-thickness wound healing. Tissue Eng. 2012, 18, 2315–2322. [Google Scholar] [CrossRef] [PubMed]
- Berdal, M.; Appelbom, H.I.; Eikrem, J.H.; Lund, A.; Busund, L.-T.; Hanes, R.; Seljelid, R.; Jenssen, T. Aminated β-1,3-D-glucan has a dose-dependent effect on wound healing in diabetic db/db mice. Wound Repair Regen. 2011, 19, 579–587. [Google Scholar] [CrossRef] [PubMed]
- Kofuji, K.; Huang, Y.; Tsubaki, K.; Kokido, F.; Nishikawa, K.; Isobe, T.; Murata, Y. Preparation and evaluation of a novel wound dressing sheet comprised of β-glucan-chitosan complex. React. Funct. Polym. 2010, 70, 784–789. [Google Scholar] [CrossRef]
- Cerci, C.; Yildirim, M.; Ceyhan, M.; Bozkurt, S.; Doguc, D.; Gokicimen, A. The effects of topical and systemic beta glucan administration on wound healing impaired by corticosteroids. Wounds 2008, 20, 341–346. [Google Scholar] [PubMed]
- Huang, M.-H.; Yang, M.-C. Evaluation of glucan/poly(vinyl alcohol) blend wound dressing using rat models. Int. J. Pharm. 2008, 346, 38–46. [Google Scholar] [CrossRef] [PubMed]
- Berdal, M.; Appelbom, H.I.; Eikrem, J.H.; Lund, A.; Zykova, S.N.; Busund, L.-T.; Seljelid, R.; Jenssen, T. Aminated β-1,3-d-glucan improves wound healing in diabetic db/db mice. Wound Repair Regen. 2007, 15, 825–832. [Google Scholar] [CrossRef] [PubMed]
- LeBlanc, B.W.; Albina, J.E.; Reichner, J.S. The effect of PGG-β-glucan on neutrophil chemotaxis in vivo. J. Leukoc. Biol. 2006, 79, 667–675. [Google Scholar] [CrossRef] [PubMed]
- Dinc, S.; Durmus, E.; Gulcelik, M.A.; Kuru, B.; Ustun, H.; Renda, N.; Alagol, H. Effects of beta-d-glucan on steroid-induced impairment of colonic anastomotic healing. Acta Chir. Belg. 2006, 106, 63–67. [Google Scholar] [CrossRef] [PubMed]
- Medeiros, S.D.; Cordeiro, S.L.; Cavalcanti, J.E.; Melchuna, K.M.; Lima, A.M.; Filho, I.A.; Medeiros, A.C.; Rocha, K.B.; Oliveira, E.M.; Faria, E.D.; et al. Effects of purified Saccharomyces cerevisiae (1→3)-β-glucan on venous ulcer healing. Int. J. Mol. Sci. 2012, 13, 8142–8158. [Google Scholar] [CrossRef] [PubMed]
- Zykova, S.N.; Balandina, K.A.; Vorokhobina, N.V.; Kuznetsova, A.V.; Engstad, R.; Zykova, T.A. Macrophage stimulating agent soluble yeast β-1,3/1,6-glucan as a topical treatment of diabetic foot and leg ulcers: A randomized, double blind, placebo-controlled phase II study. J. Diabetes Investig. 2014, 5, 392–399. [Google Scholar] [CrossRef] [PubMed]
- Karaaslan, O.; Kankaya, Y.; Sungur, N.; Kocer, U.; Cuzdan, S.; Sahin, B.; Uysal, A. Case series of topical and orally administered β-glucan for the treatment of diabetic wounds: Clinical study. J. Cutan. Med. Surg. 2012, 16, 180–186. [Google Scholar] [CrossRef] [PubMed]
- King, B.; Barrett, S.; Cutting, K.F. Clinical evaluation of a bioactive beta-glucan gel in the treatment of ‘hard-to-heal’ wounds. J. Wound Care 2017, 26, 58–63. [Google Scholar] [CrossRef] [PubMed]
- Duckova, K.; Kucera, J.; Kuniak, L.; Augustin, J. Some results of preclinical and clinical evaluation of topical preparations containing glucan. In Proceedings of the 1st World Meeting APGI/APV, Budapest, Hungary, 9–11 May 1995; pp. 729–730. [Google Scholar]
- Delatte, S.J.; Evans, J.; Hebra, A.; Adamson, W.; Othersen, H.B.; Tagge, E.P. Effectiveness of beta-glucan collagen for treatment of partial-thickness burns in children. J. Pediatr. Surg. 2001, 36, 113–118. [Google Scholar] [CrossRef] [PubMed]
- Cutting, K.F. The cost-effectiveness of a novel soluble beta-glucan gel. J. Wound Care 2017, 26, 228–234. [Google Scholar] [CrossRef] [PubMed]
- Munarin, F.; Bozzini, S.; Visai, L.; Tanzi, M.C.; Petrini, P. Sterilization treatments on polysaccharides: Effects and side effects on pectin. Food Hydrocoll. 2013, 31, 74–84. [Google Scholar] [CrossRef]
- Khan, A.A.; Gani, A.; Masoodi, F.A.; Amin, F.; Wani, I.A.; Khanday, F.A.; Gani, A. Structural, thermal, functional, antioxidant & antimicrobial properties of β-d-glucan extracted from baker’s yeast (Saccharomyces cereviseae)-Effect of γ-irradiation. Carbohydr. Polym. 2016, 140, 442–450. [Google Scholar] [PubMed]
- El-Sonbaty, S.M.; Ismail, A.F.M.A.F.M.; Nabeel, A.I. Irradiated β-glucan enhances immune response to bacterial infection through CD4 and CD8 T-lymphocytes. Int. J. Radiat. Res. 2013, 11, 35–42. [Google Scholar]
- Shah, A.; Ahmad, M.; Ashwar, B.A.; Gani, A.; Masoodi, F.A.; Wani, I.A.; Wani, S.M.; Gani, A. Effect of γ-irradiation on structure and nutraceutical potential of β-d-glucan from barley (Hordeum vulgare). Int. J. Biol. Macromol. 2015, 72, 1168–1175. [Google Scholar] [CrossRef] [PubMed]
- Byun, E.-B.; Park, S.-H.; Jang, B.-S.; Sung, N.-Y.; Byun, E.-H. Gamma-irradiated β-glucan induces immunomodulation and anticancer activity through MAPK and NF-κB pathways. J. Sci. Food Agric. 2016, 96, 695–702. [Google Scholar] [CrossRef] [PubMed]
- El-sonbaty, S.M.; Zahran, W.E.; Moawed, F.S.M.F.S.M. Gamma-irradiated β-glucan modulates signaling molecular targets of hepatocellular carcinoma in rats. Tumor Biol. 2017, 39, 1–10. [Google Scholar]
| Type of β-Glucan/Source | Structure of β-Glucan | Target Cell Type | Actions | Ref. |
|---|---|---|---|---|
| W-S Curdlan, (7%)/Agrobacterium sp. | Linear (1,3;1,6) β-glucan | Swiss-3T3 fibroblast cells | Fibroblasts exposed to a curdlan/polyvinyl alcohol (PVA) blend showed significantly higher spreading rates in the scratch wound assay after 24 h when compared to control and pure PVA. | [26] |
| W-INS (2%)/Saccharomyces cerevisiae | Linear (1,3) β-glucan | 3T3 fibroblast cells, HaCaTs | β-glucan-based nanofibrous membranes promoted the adhesion and proliferation of fibroblasts and keratinocytes. | [27] |
| W-S (2.5%)/Aureobasidium pullulans | Branched (1,3;1,6) β-glucan | Human fetal dermal fibroblast cell line FW20-2, primary human dermal fibroblasts | β-glucan consistently stimulated dermal fibroblast proliferation and migration and modulated the effect of transforming growth factor-β1. β-glucan l did not affect the procollagen production from fibroblasts. | [28] |
| W-S (0.75%)/Bradyrhizobium japonicum | Cyclic (1,3;1,6) β-glucan | Swiss-3T3 fibroblast cells | Cyclic β-glucan incorporated in hydrogels increased hydrogel porosity and enhanced cell attachment, proliferation and migration activity. | [29] |
| W-S Curdlan, (10 μg/mL)/Alcaligenes faecalis | Linear (1,3) β-glucan | Primary human keratinocytes | Curdlan enhanced migration, proliferation and wound closure of primary human keratinocytes in a Dectin-1 concentration-dependent manner. | [30] |
| W-S Laminarin (0.2 mg/mL)/Laminaria sp. W-INS Paramylon (0.2 mg/mL)/Euglena gracilis | Linear (1,3) β-glucan with (1,6)-linked β-glucosyl Linear (1,3) β-glucan | Immortalized human corneal epithelial cells | Both β-glucan types promoted epithelial migration in a concentration-dependent manner. This effect was enhanced when β-glucan was conjugated with hyaluronic acid to increase solubility. | [31] |
| W-INS (1 mg/mL)/Saccharomyces cerevisiae | Branched (1,3;1,6) β-glucan | Human and mouse wound macrophages | β-glucan potently induced TNF-α production in wound macrophages via activation of the Dectin-1 and TLR2 signaling pathways. Interestingly, it also induced phosphorylation of the macrophage-colony stimulating factor. | [32] |
| W-INS, W-S (1 μg/mL)/various sources | Linear (1,3;1,4) β-glucanBranched (1,3;1,6) β-glucan Linear (1,3) β-glucan | HaCaTs, 3T3-Li preadipocytes | β-glucans of different origin, solubility and purity were tested for inhibition of adipogenic differentiation, wound healing and skin irritation. Significant differences in efficacy between β-glucan preparations were shown. The most active was yeast-derived insoluble β-glucan. | [33] |
| W-S (1 mg/mL)/Aureobasidium pullulans | Branched (1,3;1,6) β-glucan | Human dermal fibroblast, adipose tissue-derived stem cells | β-glucan enhanced cellular response, proliferation and migration of both human dermal fibroblasts and adipose tissue-derived stem cells. | [34] |
| W-INS Pleuran, (0.2 mg/mL)/Pleurotus ostreatus | Branched (1,3;1,6) β-glucan | Primary human keratinocytes | β-glucan induced the production of matrix metalloproteinase-9 from human keratinocytes. | [35] |
| W-S (0.5–1 mg/mL)/Aureobasidium sp. | Branched (1,3;1,6) β-glucan | Mouse macrophage cell line RAW264.7, human dermal fibroblasts | β-glucan accelerated wound healing by enhancing fibroblast migration and collagen synthesis via glucan receptors on fibroblasts, as well as by activating macrophages to release more TNF-α. | [20] |
| W-S (0.5 and 5 mg/mL)/Lentinula edodes | Linear (1,3) β-glucan with (1,6)-linked- β-glucosyl | Mouse connective tissue L-929 cells | β-glucan treatment of L-929 cells resulted in a dose-dependent increase in cell proliferation. No significant difference in the migration speed between the glucan-treated and non-treated cell was documented. | [36] |
| W-S (1 μg/mL)/Saccharomyces cerevisiae | Branched (1,3;1,6) β-glucan | Human dermal fibroblasts | Derivatized β-glucan increased nuclear factor-1 binding capacity and stimulated collagen biosynthesis in human dermal fibroblasts. | [19] |
| W-S (1 μg/mL)/Saccharomyces cerevisiae | Branched (1,3;1,6) β-glucan | Normal human dermal fibroblasts | Derivatized β-glucan stimulated the expression of cytokine and procollagen genes through the transcription factors activator protein-1 and specificity protein-1 in normal human dermal fibroblasts. It also stimulated expression of various growth factors participating in wound repair. | [37] |
| W-S (1 mg/mL)/Saccharomyces cerevisiae | Branched (1,3;1,6) β-glucan | Normal human dermal fibroblasts | β-glucan stimulated fibroblast NF-kB nuclear binding activity and interleukin 6 gene expression in a time-dependent manner. It can directly modulate the functional activity of human dermal fibroblasts. At least two binding sites for β-glucan on fibroblasts were identified. | [38] |
| W-S (0.01%)/Saccharomyces cerevisiae | Branched (1,3;1,6) β-glucan | Porcine keratinocytes | Carboxymethyl β-glucan at a concentration of 0.005 and 0.01% stimulated keratinocytes proliferation within 120 h of incubation in culture media. | [39] |
| Type of β-Glucan/Source | Structure of β-glucan | Animal Model | Actions | Ref. |
|---|---|---|---|---|
| W-INS (4%)/Schizophyllum commune | Branched (1,3;1,6) β-glucan | Sprague-Dawley rat | Hydrogels incorporating β-glucan and sterilized with gamma radiation accelerated wound repair while releasing of β-glucan molecules from the hydrogel. | [41] |
| W-INS (2%)/Saccharomyces cerevisiae | Linear (1,3) β-glucan | Kunming mouse | β-glucan-based nanofibrous membranes significantly reduced wound size compared to control groups. On Day 14, 83% wound size reduction was observed (26% in the control group). All wounds were completely covered with epidermis by Day 14, but the β-glucan group exhibited substantially more epithelialization. | [27] |
| W-S (0.75%)/Bradyrhizobium japonicum | Cyclic (1,3) β-glucan | Wistar rat | Nanofibrous asymmetric membranes containing β-glucan significantly accelerated wound healing by promoting reepithelialization, tissue remodeling and collagen deposition. Collagen deposition was more organized than in the control group. The levels of pro-inflammatory cytokines at Day 8 were significantly lower in the β-glucan group compared to the control group. | [29] |
| W-S (0.1 mg/L)/Saccharomyces cerevisiae | Branched (1,3;1,6) β-glucan | Rainbow trout | Wound healing progressed, at least partially, due to a low temperature of about 8.5 °C: β-glucan was shown to have a very limited effect on wound healing in trout, when in a bath. Complete healing was achieved only by Day 100. | [42] |
| W-S (2.5%)/Aureobasidium pullulans | Branched (1,3;1,6) β-glucan | Diabetic C57BL/KsJ-db/db mouse | Treatment with β-glucan caused a significant decrease in wound size compared to the vehicle control (distilled water). Increased inflammatory cells in the granulation tissues of wound areas were also significantly less compared to the vehicle control, showing increased numbers of micro-vessels and fibroblasts, as well as reepithelialization. | [43] |
| W-S Laminarin (0.2 mg/mL)/Laminaria sp. | Linear (1,3) β-glucan with (1,6)-linked β-glucosyl Linear (1,3) β-glucan | Sprague-Dawley rat | β-glucan and β-glucan conjugated with hyaluronic acid suppressed acute inflammatory reactions in the rat corneal alkali burn model. Decreased corneal edema and less polymorphonuclear leukocytes infiltrates were observed in the β-glucan-treated wound. | [31] |
| W-S MacroGrad, (0.1 μg/mL)/Saccharomyces cerevisiae | Branched (1,3;1,6) β-glucan | Cyprinus carpio L (carp) | β-glucan promoted the wound healing process in common carp compared to the group treated with 6.3 kDa oat fiber and the control fish group. The positive effect of the β-glucan preparation was related to the high branching levels due to fish being bathed in β-glucan-supplemented water, showing a higher wound closure ratio compared to a 6.3-kDa-supplemented bath. | [44] |
| W-S (1 mg/mL)/Sparassis crispa | Branched (1,3;1,6) β-glucan | ICR mouse | Fungal β-glucan significantly increased the rate of wound healing due to, at least partially, local stimulation of collagen synthesis. | [45] |
| W-S (2.5%)/Aureobasidium pullulans | Branched (1,3;1,6) β-glucan | ICR nu/nu mouse | β-glucan promoted wound healing of full-thickness wounds infected by S. aureus, S. pyogenes and P. aeruginosa. β-glucan did not show any direct antibacterial activity, though it was able to heal the wounds. | [40] |
| W-S (50%)/Aureobasidium pullulans | Branched (1,3;1,6) β-glucan | BALB/c nude mouse | Poly-(lactic-co-glycolic acid) (PLGA), a constituent of the membranes containing 50% β-glucan, enhanced the wound’s interaction with the surrounding cells, proliferation and angiogenesis compared to the ones from the membranes without PLGA. A PLGA membrane with incorporated β-glucan may be applied as a skin substitute to accelerate wound healing. | [46] |
| W-S Curdlan, (11.1 mg/mL)/Alcaligenes faecalis | Linear (1,3) β-glucan | Diabetic C57Bl/KsBom-db/db | β-glucan exerted its primary effects in the early phase of wound healing and was coherent with an effect on macrophage functions in the wound. A higher dosage frequency showed a significant improvement in wound closure compared to a low dosage frequency. | [47] |
| W-S (1%)/Aureobasidium pullulans | Branched (1,3;1,6) β-glucan | ddY mouse | A β-glucan and chitosan complex was used for wound healing. The complex sheet did not dissolve during the application period, did not adhere to the wound and was easy to remove. The complex accelerated wound repair by activating macrophages and cytokines’ release and accelerated reepithelialization of the skin wound. | [48] |
| W-INS Imuneks, (5% cream or 50 mg/kg)/Saccharomyces cerevisiae | Branched (1,3;1,6) β-glucan | Wistar rat | Systemic and local administration of β-glucan stimulated wound contraction, increased incision tensile strength and improved epithelialization. Systemic administration was more effective than topical administration. | [49] |
| W-INS/Aureobasidium pullulans | Branched (1,3;1,6) β-glucan | Wistar rat | An aqueous mixture of β-glucan and polyvinyl alcohol increased the wound contraction ratio by 83% after 11 days, while when treating with cotton gauze, an 85% contraction was observed, but only after 21 days. Additionally, healing time was significantly reduced by 48% using the β-glucan mixture. The accelerating effect of wound healing might be attributed to the release of β-glucan. | [50] |
| W-S Curdlan, (11.1 mg/mL)/Alcaligenes faecalis | Linear (1,3) β-glucan | Diabetic/non-diabetic C57Bl/KsBom mouse | Significantly higher wound closure rates were observed in diabetic mice given topical applications of β-glucan compared to the placebo-treated mice. A more cell-rich and vascularized granulation tissue and an increase in the reepithelialization were observed. | [51] |
| W-S Betafectin, (2 mg/kg i.v.)/Saccharomyces cerevisiae | Branched (1,3;1,6) β-glucan | Fisher-344 rat | Systemic β-glucan treatment resulted in enhanced migration of neutrophils to the site of inflammation and improved antimicrobial function. Polymorphonuclear cells obtained from β-glucan-treated animals showed a heightened respiratory burst and a reduced bacterial load in a pulmonary model of infection. | [52] |
| Imuneks (100 mg/kg p.o.)/Saccharomyces cerevisiae | Branched (1,3;1,6) β-glucan | Sprague-Dawley rat | An oral administration of β-glucan improved impaired anastomotic wound healing in rats treated with corticosteroids over a long period of time. Increased macrophages and fibroblast population were observed in samples from β-d-glucan-treated animals. | [53] |
| Type of β-Glucan (Source)/Placebo | Structure of β-Glucan | Ulcer Type | No. of Participants β-Glucan/Placebo | Treatment Duration | Result | Ref. |
|---|---|---|---|---|---|---|
| 3% W-INS (Saccharomyces cerevisiae)/- | Linear (1,3) β-glucan | venous | 12/- | 90 d | A 55.2% reduction of the ulcer area was documented at the 90th day. β-glucan increased epithelial hyperplasia, as well as increased inflammatory cells, angiogenesis and fibroblast proliferation. | [54] |
| 2% W-S (Saccharomyces cerevisiae)/methylcellulose | Branched (1,3;1,6) β-glucan | diabetic | 27/27 | 12 w | An 87% reduction of the ulcer was documented in the β-glucan group compared to a 56% reduction in the control group. No serious adverse effects in the β-glucan group were recorded. | [55] |
| W-S (Saccharomyces cerevisiae)/- | Branched (1,3;1,6) β-glucan | leg | 26/- | 12 w | An average wound surface area reduction of 41% was observed; one wound healed; 20 decreased in size; four remained static; and two increased in size. | [56] |
| W-S (Saccharomyces cerevisiae)/- | Branched (1,3;1,6) β-glucan | diabetic | 22/- | 20 w | The time for complete healing averaged 10.8 weeks (range 6–20 weeks). No adverse effects were recorded during the treatment. | [57] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Majtan, J.; Jesenak, M. β-Glucans: Multi-Functional Modulator of Wound Healing. Molecules 2018, 23, 806. https://doi.org/10.3390/molecules23040806
Majtan J, Jesenak M. β-Glucans: Multi-Functional Modulator of Wound Healing. Molecules. 2018; 23(4):806. https://doi.org/10.3390/molecules23040806
Chicago/Turabian StyleMajtan, Juraj, and Milos Jesenak. 2018. "β-Glucans: Multi-Functional Modulator of Wound Healing" Molecules 23, no. 4: 806. https://doi.org/10.3390/molecules23040806
APA StyleMajtan, J., & Jesenak, M. (2018). β-Glucans: Multi-Functional Modulator of Wound Healing. Molecules, 23(4), 806. https://doi.org/10.3390/molecules23040806







