Melatonin: A Versatile Protector against Oxidative DNA Damage
Abstract
1. Introduction
2. Oxidative Damage to DNA
2.1. Induction by Free Radicals.
2.2. Other Causes
2.3. Consequences for Human Health
3. Antioxidant Protection
3.1. Free radical Scavenging Activity
3.2. Inhibiting Metal-induced Oxidation
- (i)
- sequestering metal ions from reductants, i.e., inhibiting the reduction of metal ions thus their reduced forms are not available for Fenton-like reactions; or
- (ii)
- deactivating •OH after being produced via Fenton-like reactions. In this case the •OH radicals are still formed, but they are rapidly scavenged by the organic ligands in the metal chelates.
3.3. Counteracting the Effects of Other OD Triggers
3.4. Metabolic Derivatives
3.5. Other Protection Mechanisms
3.5.1. Activating Antioxidative Enzymes
3.5.2. Inhibiting Pro-oxidative Enzymes
3.5.3. Boosting DNA Repair Machinery
4. Concluding Remarks
Acknowledgments
Conflicts of Interest
References
- Lindahl, T. Instability and decay of the primary structure of DNA. Nature 1993, 362, 709–715. [Google Scholar] [CrossRef] [PubMed]
- Pham-Huy, L.A.; He, H.; Pham-Huy, C. Free radicals, antioxidants in disease and health. Int. J. Biomed. Sci. 2008, 4, 89–96. [Google Scholar] [PubMed]
- Pacher, P.; Beckman, J.S.; Liaudet, L. Nitric oxide and peroxynitrite in health and disease. Physiol. Rev. 2007, 87, 315–424. [Google Scholar] [CrossRef] [PubMed]
- Genestra, M. Oxyl radicals, redox-sensitive signalling cascades and antioxidants. Cell. Signal. 2007, 19, 1807–1819. [Google Scholar] [CrossRef] [PubMed]
- Valko, M.; Leibfritz, D.; Moncol, J.; Cronin, M.T.D.; Mazur, M.; Telser, J. Free radicals and antioxidants in normal physiological functions and human disease. Int. J. Biochem. Cell Biol. 2007, 39, 44–84. [Google Scholar] [CrossRef] [PubMed]
- Valko, M.; Rhodes, C.J.; Moncol, J.; Izakovic, M.; Mazur, M. Free radicals, metals and antioxidants in oxidative stress-induced cancer. Chem. Biol. Interact. 2006, 160, 1–40. [Google Scholar] [CrossRef] [PubMed]
- Dröge, W. Free radicals in the physiological control of cell function. Physiol. Rev. 2002, 82, 47–95. [Google Scholar] [CrossRef] [PubMed]
- Young, I.S.; Woodside, J.V. Antioxidants in health and disease. J. Clin. Pathol. 2001, 54, 176–186. [Google Scholar] [CrossRef] [PubMed]
- Halliwell, B. Biochemistry of oxidative stress. Biochem. Soc. Trans. 2007, 35, 1147–1150. [Google Scholar] [CrossRef] [PubMed]
- Willcox, J.K.; Ash, S.L.; Catignani, G.L. Antioxidants and prevention of chronic disease. Crit. Rev. Food Sci. Nutr. 2004, 44, 275–295. [Google Scholar] [CrossRef] [PubMed]
- Parthasarathy, S.; Santanam, N.; Ramachandran, S.; Meilhac, O. Oxidants and antioxidants in atherogenesis: An appraisal. J. Lipid Res. 1999, 40, 2143–2157. [Google Scholar] [PubMed]
- Lowe, F.J.; Cemeli, E. Biomarkers of oxidative stress and the relationship to cigarette smoking. Mini Rev. Org. Chem. 2011, 8, 377–386. [Google Scholar] [CrossRef]
- Valko, M.; Morris, H.; Cronin, M.T.D. Metals, toxicity and oxidative stress. Curr. Med. Chem. 2005, 12, 1161–1208. [Google Scholar] [CrossRef] [PubMed]
- Reiter, R.J.; Tan, D.X.; Sainz, R.M.; Mayo, J.C.; Lopez-Burillo, S. Melatonin: Reducing the toxicity and increasing the efficacy of drugs. J. Pharm. Pharmacol. 2002, 54, 1299–1321. [Google Scholar] [CrossRef] [PubMed]
- Reiter, R.J.; Manchester, L.C.; Tan, D.X. Neurotoxins: Free radical mechanisms and melatonin protection. Curr. Neuropharmacol. 2010, 8, 194–210. [Google Scholar] [CrossRef] [PubMed]
- Tan, D.X.; Manchester, L.C.; Reiter, R.J.; Qi, W.B.; Karbownik, M.; Calvo, J.R. Significance of melatonin in antioxidative defense system: Reactions and products. Biol. Signals Recept. 2000, 9, 137–159. [Google Scholar] [CrossRef] [PubMed]
- Reiter, R.J.; Tan, D.X.; Galano, A. Melatonin: Exceeding expectations. Physiology (Bethesda) 2014, 29, 325–333. [Google Scholar] [CrossRef] [PubMed]
- Galano, A.; Medina, M.E.; Tan, D.X.; Reiter, R.J. Melatonin and its metabolites as copper chelating agents and their role in inhibiting oxidative stress: A physicochemical analysis. J. Pineal Res. 2015, 58, 107–116. [Google Scholar] [CrossRef] [PubMed]
- Álvarez-Diduk, R.; Galano, A.; Tan, D.X.; Reiter, R.J. N-Acetylserotonin and 6-hydroxymelatonin against oxidative stress: Implications for the overall protection exerted by melatonin. J. Phys. Chem. B 2015, 119, 8535–8543. [Google Scholar] [CrossRef] [PubMed]
- Tan, D.X.; Reiter, R.J.; Manchester, L.C.; Yan, M.T.; El-Sawi, M.; Sainz, R.M.; Mayo, J.C.; Kohen, R.; Allegra, M.; Hardeland, R. Chemical and physical properties and potential mechanisms: melatonin as a broad spectrum antioxidant and free radical scavenger. Curr. Top. Med. Chem. 2002, 2, 181–197. [Google Scholar] [CrossRef] [PubMed]
- Hardeland, R. Antioxidative protection by melatonin: Multiplicity of mechanisms from radical detoxification to radical avoidance. Endocrine 2005, 27, 119–130. [Google Scholar] [CrossRef]
- Galano, A.; Tan, D.X.; Reiter, R.J. Melatonin as a natural ally against oxidative stress: A physicochemical examination. J. Pineal Res. 2011, 51, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Miller, E.; Walczak, A.; Majsterek, I.; Kedziora, J. Melatonin reduces oxidative stress in the erythrocytes of multiple sclerosis patients with secondary progressive clinical course. J. Neuroimmunol. 2013, 257, 97–101. [Google Scholar] [CrossRef] [PubMed]
- Manchester, L.C.; Coto-Montes, A.; Boga, J.A.; Andersen, L.P.H.; Zhou, Z.; Galano, A.; Vriend, J.; Tan, D.X.; Reiter, R.J. Melatonin: an ancient molecule that makes oxygen metabolically tolerable. J. Pineal Res. 2015, 59, 403–419. [Google Scholar] [CrossRef] [PubMed]
- Rosales-Corral, S.A.; Reiter, R.J.; Tan, D.X.; Manchester, L.C.; Liu, X. Antioxidant and anti-inflammatory role of melatonin in Alzheimer’s neurodegeneration. Aging 2014, 177–193. [Google Scholar] [CrossRef]
- Yang, Y.; Sun, Y.; Yi, W.; Li, Y.; Fan, C.; Xin, Z.; Jiang, S.; Di, S.; Qu, Y.; Reiter, R.J.; Yi, D. A review of melatonin as a suitable antioxidant against myocardial ischemia-reperfusion injury and clinical heart diseases. J. Pineal Res. 2014, 57, 357–366. [Google Scholar] [CrossRef] [PubMed]
- Galano, A.; Tan, D.X.; Reiter, R.J. Cyclic 3-hydroxymelatonin, a key metabolite enhancing the peroxyl radical scavenging activity of melatonin. RSC Adv. 2014, 4, 5220–5227. [Google Scholar] [CrossRef]
- Tan, D.X.; Hardeland, R.; Manchester, L.C.; Galano, A.; Reiter, R.J. Cyclic-3-hydroxymelatonin (C3HOM), a potent antioxidant, scavenges free radicals and suppresses oxidative reactions. Curr. Med. Chem. 2014, 21, 1557–1565. [Google Scholar] [CrossRef] [PubMed]
- Matuszak, Z.; Reszka, K.J.; Chignell, C.F. Reaction of melatonin and related indoles with hydroxyl radicals: EPR and spin trapping investigations. Free Radic. Biol. Med. 1997, 23, 367–372. [Google Scholar] [CrossRef]
- Stasica, P.; Ulanski, P.; Rosiak, J.M. Melatonin as a hydroxyl radical scavenger. J. Pineal Res. 1998, 25, 65–66. [Google Scholar] [CrossRef] [PubMed]
- Velkov, Z.A.; Velkov, Y.Z.; Galunska, B.T.; Paskalev, D.N.; Tadjer, A.V. Melatonin: Quantum-chemical and biochemical investigation of antioxidant activity. Eur. J. Med. Chem. 2009, 44, 2834–2839. [Google Scholar] [CrossRef] [PubMed]
- Galano, A. On the direct scavenging activity of melatonin towards hydroxyl and a series of peroxyl radicals. Phys. Chem. Chem. Phys. 2011, 13, 7178–7188. [Google Scholar] [CrossRef] [PubMed]
- Scaiano, J.C. Exploratory laser flash photolysis study of free radical reactions and magnetic field effects in melatonin chemistry. J. Pineal Res. 1995, 19, 189–195. [Google Scholar] [CrossRef] [PubMed]
- Ressmeyer, A.R.; Mayo, J.C.; Zelosko, V.; Sáinz, R.M.; Tan, D.X.; Poeggeler, B.; Antolín, I.; Zsizsik, B.K.; Reiter, R.J.; Hardeland, R. Antioxidant properties of the melatonin metabolite N1-acetyl-5-methoxykynuramine (AMK): Scavenging of free radicals and prevention of protein destruction. Redox Rep. 2003, 8, 205–213. [Google Scholar] [CrossRef] [PubMed]
- Guenther, A.L.; Schmidt, S.I.; Laatsch, H.; Fotso, S.; Ness, H.; Ressmeyer, A.R.; Poeggeler, B.; Hardeland, R. Reactions of the melatonin metabolite AMK (N1-acetyl-5-methoxykynuramine) with reactive nitrogen species: Formation of novel compounds, 3-acetamidomethyl-6-methoxycinnolinone and 3-nitro-AMK. J. Pineal Res. 2005, 39, 251–260. [Google Scholar] [CrossRef] [PubMed]
- Hardeland, R.; Backhaus, C.; Fadavi, A. Reactions of the NO redox forms NO+, •NO and HNO (protonated NO-) with the melatonin metabolite N1-acetyl-5-methoxykynuramine. J. Pineal Res. 2007, 43, 382–388. [Google Scholar] [CrossRef] [PubMed]
- Tan, D.X.; Chen, L.D.; Poeggeler, B.; Manchester, L.C.; Reiter, R.J. Melatonin: a potent, endogenous hydroxyl radical scavenger. Endocr. J. 1993, 1, 57–60. [Google Scholar]
- Matuszak, Z.; Bilska, M.A.; Reszkat, K.J.; Chignell, C.F.; Bilski, P. Interaction of Singlet Molecular Oxygen with Melatonin and Related Indoles. Photochem. Photobiol. 2003, 78, 449–455. [Google Scholar] [CrossRef]
- Behrends, A.; Hardeland, R.; Ness, H.; Grube, S.; Poeggeler, B.; Haldar, C. Photocatalytic actions of the pesticide metabolite 2-hydroxyquinoxaline: Destruction of antioxidant vitamins and biogenic amines-Implications of organic redox cycling. Redox Rep. 2004, 9, 279–288. [Google Scholar] [CrossRef] [PubMed]
- Kuesel, J.T.; Hardeland, R.; Pfoertner, H.; Aeckerle, N. Reactions of the melatonin metabolite N1-acetyl-5-methoxykynuramine with carbamoyl phosphate and related compounds. J. Pineal Res. 2010, 48, 47–54. [Google Scholar] [CrossRef] [PubMed]
- Mayo, J.C.; Tan, D.X.; Sainz, R.M.; Natarajan, M.; Lopez-Burillo, S.; Reiter, R.J. Protection against oxidative protein damage induced by metal-catalyzed reaction or alkylperoxyl radicals: Comparative effects of melatonin and other antioxidants. Biochim. Biophys. Acta 2003, 1620, 139–150. [Google Scholar] [CrossRef]
- Park, S.; Lee, D.E.; Jang, H.; Byeon, Y.; Kim, Y.S.; Back, K. Melatonin-rich transgenic rice plants exhibit resistance to herbicide-induced oxidative stress. J. Pineal Res. 2013, 54, 258–263. [Google Scholar] [CrossRef] [PubMed]
- Maharaj, D.S.; Limson, J.L.; Daya, S. 6-Hydroxymelatonin converts Fe (III) to Fe (II) and reduces iron-induced lipid peroxidation. Life Sci. 2003, 72, 1367–1375. [Google Scholar] [CrossRef]
- Maharaj, D.S.; Maharaj, H.; Daya, S.; Glass, B.D. Melatonin and 6-hydroxymelatonin protect against iron-induced neurotoxicity. J. Neurochem. 2006, 96, 78–81. [Google Scholar] [CrossRef] [PubMed]
- García, J.J.; Reiter, R.J.; Karbownik, M.; Calvo, J.R.; Ortiz, G.G.; Tan, D.X.; Martínez-Ballarín, E.; Acua-Castroviejo, D. N-Acetylserotonin suppresses hepatic microsomal membrane rigidity associated with lipid peroxidation. Eur. J. Pharmacol. 2001, 428, 169–175. [Google Scholar] [CrossRef]
- Parmar, P.; Limson, J.; Nyokong, T.; Daya, S. Melatonin protects against copper-mediated free radical damage. J. Pineal Res. 2002, 32, 237–242. [Google Scholar] [CrossRef] [PubMed]
- Seeger, H.; Mueck, A.O.; Lippert, T.H. Effect of melatonin and metabolites on copper-mediated oxidation of low density lipoprotein. Br. J. Clin. Pharmacol. 1997, 44, 283–284. [Google Scholar] [CrossRef]
- Gozzo, A.; Lesieur, D.; Duriez, P.; Fruchart, J.C.; Teissier, E. Structure-activity relationships in a series of melatonin analogues with the low-density lipoprotein oxidation model. Free Radic. Biol. Med. 1999, 26, 1538–1543. [Google Scholar] [CrossRef]
- Tan, D.X.; Hardeland, R.; Manchester, L.C.; Paredes, S.D.; Korkmaz, A.; Sainz, R.M.; Mayo, J.C.; Fuentes-Broto, L.; Reiter, R.J. The changing biological roles of melatonin during evolution: From an antioxidant to signals of darkness, sexual selection and fitness. Biol. Rev. Camb. Philos. Soc. 2010, 85, 607–623. [Google Scholar] [CrossRef] [PubMed]
- Davanipour, Z.; Poulsen, H.E.; Weimann, A.; Sobel, E. Endogenous melatonin and oxidatively damaged guanine in DNA. BMC Endocr. Disord. 2009, 9, 22. [Google Scholar] [CrossRef] [PubMed]
- Rosen, J.; Than, N.N.; Koch, D.; Poeggeler, B.; Laatsch, H.; Hardeland, R. Interactions of melatonin and its metabolites with the ABTS cation radical: Extension of the radical scavenger cascade and formation of a novel class of oxidation products, C2-substituted 3-indolinones. J. Pineal Res. 2006, 41, 374–381. [Google Scholar] [CrossRef] [PubMed]
- Tan, D.X.; Manchester, L.C.; Terron, M.P.; Flores, L.J.; Reiter, R.J. One molecule, many derivatives: A never-ending interaction of melatonin with reactive oxygen and nitrogen species? J. Pineal Res. 2007, 42, 28–42. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.; Cui, Y.; Niedernhofer, L.J.; Wang, Y. Occurrence, Biological Consequences, and Human Health Relevance of Oxidative Stress-Induced DNA Damage. Chem. Res. Toxicol. 2016, 29, 2008–2039. [Google Scholar] [CrossRef] [PubMed]
- Wiseman, H.; Halliwell, B. Damage to DNA by reactive oxygen and nitrogen species: Role in inflammatory disease and progression to cancer. Biochem. J 1996, 313, 17–29. [Google Scholar] [CrossRef] [PubMed]
- Minko, I.G.; Kozekov, I.D.; Harris, T.M.; Rizzo, C.J.; Lloyd, R.S.; Stone, M.P. Chemistry and biology of DNA containing 1,N2-deoxyguanosine adducts of the α,β-unsaturated aldehydes acrolein, crotonaldehyde, and 4-hydroxynonenal. Chem. Res. Toxicol. 2009, 22, 759–778. [Google Scholar] [CrossRef] [PubMed]
- Chatgilialoglu, C.; D’Angelantonio, M.; Guerra, M.; Kaloudis, P.; Mulazzani, Q.G. A reevaluation of the ambident reactivity of the guanine moiety towards hydroxyl radicals. Angew. Chem. Int. Ed. Engl. 2009, 48, 2214–2217. [Google Scholar] [CrossRef] [PubMed]
- Candeias, L.P.; Steenken, S. Reaction of HO• with guanine derivatives in aqueous solution: Formation of two different redox-active OH-adduct radicals and their unimolecular transformation reactions. Properties of G(-H)•. Chem. Eur. J. 2000, 6, 475–484. [Google Scholar] [CrossRef]
- Pryor, W.A. Why is the hydroxyl radical the only radical that commonly adds to DNA? Hypothesis: It has a rare combination of high electrophilicity, high thermochemical reactivity, and a mode of production that can occur near DNA. Free Radic. Biol. Med. 1988, 4, 219–223. [Google Scholar] [CrossRef]
- Galano, A.; Alvarez-Idaboy, J.R. Guanosine + OH radical reaction in aqueous solution: A reinterpretation of the UV-vis data based on thermodynamic and kinetic calculations. Org. Lett. 2009, 11, 5114–5117. [Google Scholar] [CrossRef] [PubMed]
- Steenken, S.; Jovanovic, S.V. How easily oxidizable is DNA? One-electron reduction potentials of adenosine and guanosine radicals in aqueous solution. J. Am. Chem. Soc. 1997, 119, 617–618. [Google Scholar] [CrossRef]
- Cadet, J.; Douki, T.; Ravanat, J.-L. Oxidatively generated damage to the guanine moiety of DNA: Mechanistic aspects and formation in cells. Acc. Chem. Res. 2008, 41, 1075–1083. [Google Scholar] [CrossRef] [PubMed]
- Galano, A.; Alvarez-Idaboy, J.R. On the evolution of one-electron-oxidized deoxyguanosine in damaged DNA under physiological conditions: A DFT and ONIOM study on proton transfer and equilibrium. Phys. Chem. Chem. Phys. 2012, 14, 12476–12484. [Google Scholar] [CrossRef] [PubMed]
- Balasubramanian, B.; Pogozelski, W.K.; Tullius, T.D. DNA strand breaking by the hydroxyl radical is governed by the accessible surface areas of the hydrogen atoms of the DNA backbone. Proc. Natl. Acad. Sci. USA 1998, 95, 9738–9743. [Google Scholar] [CrossRef] [PubMed]
- Malone, M.E.; Cullis, P.M.; Symons, M.C.R.; Parker, A.W. Biphotonic photoionization of cytosine and its derivatives with UV radiation at 248 nm: An EPR study in low-temperature perchlorate glasses. J. Phys. Chem. 1995, 99, 9299–9308. [Google Scholar] [CrossRef]
- Tronche, C.; Goodman, B.K.; Greenberg, M.M. DNA damage induced via independent generation of the radical resulting from formal hydrogen atom abstraction from the C1′-position of a nucleotide. Chem. Biol. 1998, 5, 263–271. [Google Scholar] [CrossRef]
- Pogozelski, W.K.; Tullius, T.D. Oxidative Strand Scission of Nucleic Acids: Routes Initiated by Hydrogen Abstraction from the Sugar Moiety. Chem. Rev. 1998, 98, 1089–1108. [Google Scholar] [CrossRef] [PubMed]
- Dedon, P.C. The Chemical Toxicology of 2-Deoxyribose Oxidation in DNA. Chem. Res. Toxicol. 2007, 21, 206–219. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Diao, L.; Liao, X.; Kou, L.; Lu, W. DFT study on addition reaction mechanism of guanine-cytosine base pair with OH radical. J. Phys. Org. Chem. 2015, 28, 437–444. [Google Scholar] [CrossRef]
- Valavanidis, A.; Vlachogianni, T.; Fiotakis, C. 8-Hydroxy-2′-deoxyguanosine (8-OHdG): A critical biomarker of oxidative stress and carcinogenesis. J. Environ. Sci. Health C Environ. Carcinog. Ecotoxicol. Rev. 2009, 27, 120–139. [Google Scholar] [CrossRef] [PubMed]
- Roszkowski, K.; Jozwicki, W.; Blaszczyk, P.; Mucha-Malecka, A.; Siomek, A. Oxidative damage DNA: 8-oxogua and 8-oxodG as molecular markers of cancer. Med. Sci. Monit. 2011, 17, CR329–CR333. [Google Scholar] [CrossRef] [PubMed]
- Steenken, S.; Jovanovic, S.V.; Bietti, M.; Bernhard, K. The trap depth (in DNA) of 8-oxo-7,8-dihydro-2′deoxyguanosine as derived from electron-transfer equilibria in aqueous solution. J. Am. Chem. Soc. 2000, 122, 2373–2374. [Google Scholar] [CrossRef]
- Cui, L.; Ye, W.; Prestwich, E.G.; Wishnok, J.S.; Taghizadeh, K.; Dedon, P.C.; Tannenbaum, S.R. Comparative analysis of four oxidized guanine lesions from reactions of DNA with peroxynitrite, singlet oxygen, and γ-radiation. Chem. Res. Toxicol. 2013, 26, 195–202. [Google Scholar] [CrossRef] [PubMed]
- Fleming, A.M.; Muller, J.G.; Ji, I.; Burrows, C.J. Characterization of 2′-deoxyguanosine oxidation products observed in the Fenton-like system Cu(ii)/H2O2/reductant in nucleoside and oligodeoxynucleotide contexts. Org. Biomol. Chem. 2011, 9, 3338–3348. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, T. Formation of spiroiminodihydantoin nucleoside from 8-oxo-7,8-dihydro-2′-deoxyguanosine by nitric oxide under aerobic conditions. Bioorg. Med. Chem. Lett. 2009, 19, 4944–4947. [Google Scholar] [CrossRef] [PubMed]
- Niles, J.C.; Wishnok, J.S.; Tannenbaum, S.R. Spiroiminodihydantoin and guanidinohydantoin are the dominant products of 8-oxoguanosine oxidation at low fluxes of peroxynitrite: Mechanistic studies with 18O. Chem. Res. Toxicol. 2004, 17, 1510–1519. [Google Scholar] [CrossRef] [PubMed]
- Luo, W.; Muller, J.G.; Rachlin, E.M.; Burrows, C.J. Characterization of hydantoin products from one-electron oxidation of 8-oxo-7,8-dihydroguanosine in a nucleoside model. Chem. Res. Toxicol. 2001, 14, 927–938. [Google Scholar] [CrossRef] [PubMed]
- Dizdaroglu, M.; Kirkali, G.; Jaruga, P. Formamidopyrimidines in DNA: Mechanisms of formation, repair, and biological effects. Free Radic. Biol. Med. 2008, 45, 1610–1621. [Google Scholar] [CrossRef] [PubMed]
- Dizdaroglu, M.; Jaruga, P. Mechanisms of free radical-induced damage to DNA. Free Radic. Res. 2012, 46, 382–419. [Google Scholar] [CrossRef] [PubMed]
- Wagner, J.R.; Cadet, J. Oxidation reactions of cytosine DNA components by hydroxyl radical and one-electron oxidants in aerated aqueous solutions. Acc. Chem. Res. 2010, 43, 564–571. [Google Scholar] [CrossRef] [PubMed]
- Box, H.C.; Budzinski, E.E.; Dawidzik, J.B.; Gobey, J.S.; Freund, H.G. Free radical-induced tandem base damage in DNA oligomers. Free Radic. Biol. Med. 1997, 23, 1021–1030. [Google Scholar] [CrossRef]
- Bellon, S.; Ravanat, J.L.; Gasparutto, D.; Cadet, J. Cross-linked thymine-purine base tandem lesions: Synthesis, characterization, and measurement in γ-irradiated isolated DNA. Chem. Res. Toxicol. 2002, 15, 598–606. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Wang, Y. Independent generation of 5-(2′-deoxycytidinyl)methyl radical and the formation of a novel cross-link lesion between 5-methylcytosine and guanine. J. Am. Chem. Soc. 2003, 125, 12795–12802. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Y.; Wang, Y. UVB-induced formation of intrastrand cross-link products of DNA in MCF-7 cells treated with 5-bromo-2′-deoxyuridine. Biochemistry 2007, 46, 8189–8195. [Google Scholar] [CrossRef] [PubMed]
- Crean, C.; Uvaydov, Y.; Geacintov, N.E.; Shafirovich, V. Oxidation of single-stranded oligonucleotides by carbonate radical anions: Generating intrastrand cross-links between guanine and thymine bases separated by cytosines. Nucleic Acids Res. 2008, 36, 742–755. [Google Scholar] [CrossRef] [PubMed]
- Uvaydov, Y.; Geacintov, N.E.; Shafirovich, V. Generation of guanine-amino acid cross-links by a free radical combination mechanism. Phys. Chem. Chem. Phys. 2014, 16, 11729–11736. [Google Scholar] [CrossRef] [PubMed]
- Leinisch, F.; Mariotti, M.; Rykaer, M.; Lopez-Alarcon, C.; Hägglund, P.; Davies, M.J. Peroxyl radical- and photo-oxidation of glucose 6-phosphate dehydrogenase generates cross-links and functional changes via oxidation of tyrosine and tryptophan residues. Free Radic. Biol. Med. 2017, 112, 240–252. [Google Scholar] [CrossRef] [PubMed]
- Asahi, T.; Kondo, H.; Masuda, M.; Nishino, H.; Aratani, Y.; Naito, Y.; Yoshikawa, T.; Hisaka, S.; Kato, Y.; Osawa, T. Chemical and immunochemical detection of 8-halogenated deoxyguanosines at early stage inflammation. J. Biol. Chem. 2010, 285, 9282–9291. [Google Scholar] [CrossRef] [PubMed]
- Kang Jr, J.I.; Sowers, L.C. Examination of hypochlorous acid-induced damage to cytosine residues in a CpG dinucleotide in DNA. Chem. Res. Toxicol. 2008, 21, 1211–1218. [Google Scholar] [CrossRef] [PubMed]
- Kawai, Y.; Morinaga, H.; Kondo, H.; Miyoshi, N.; Nakamura, Y.; Uchida, K.; Osawa, T. Endogenous formation of novel halogenated 2′-deoxycytidine: Hypohalous acid-mediated DNA modification at the site of inflammation. J. Biol. Chem. 2004, 279, 51241–51249. [Google Scholar] [CrossRef] [PubMed]
- Badouard, C.; Masuda, M.; Nishino, H.; Cadet, J.; Favier, A.; Ravanat, J.L. Detection of chlorinated DNA and RNA nucleosides by HPLC coupled to tandem mass spectrometry as potential biomarkers of inflammation. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2005, 827, 26–31. [Google Scholar] [CrossRef] [PubMed]
- Knutson, C.G.; Mangerich, A.; Zeng, Y.; Raczynski, A.R.; Liberman, R.G.; Kang, P.; Ye, W.; Prestwich, E.G.; Lu, K.; Wishnok, J.S.; et al. Chemical and cytokine features of innate immunity characterize serum and tissue profiles in inflammatory bowel disease. Proc. Natl. Acad. Sci. USA 2013, 110, E2332–E2341. [Google Scholar] [CrossRef] [PubMed]
- Lonkar, P.; Dedon, P.C. Reactive species and DNA damage in chronic inflammation: Reconciling chemical mechanisms and biological fates. Int. J. Cancer 2011, 128, 1999–2009. [Google Scholar] [CrossRef] [PubMed]
- Huie, R.E.; Padmaja, S. The reaction of no with superoxide. Free Radic. Res. 1993, 18, 195–199. [Google Scholar] [CrossRef]
- Dedon, P.C.; Tannenbaum, S.R. Reactive nitrogen species in the chemical biology of inflammation. Arch. Biochem. Biophys. 2004, 423, 12–22. [Google Scholar] [CrossRef] [PubMed]
- Cantoni, O.; Guidarelli, A. Peroxynitrite damages U937 cell DNA via the intermediate formation of mitochondrial oxidants. IUBMB Life 2008, 60, 753–756. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.; Venkatarangan, L.; Wishnok, J.S.; Tannenbaum, S.R. Quantitation of four guanine oxidation products from reaction of DNA with varying doses of peroxynitrite. Chem. Res. Toxicol. 2005, 18, 1849–1857. [Google Scholar] [CrossRef] [PubMed]
- Burney, S.; Niles, J.C.; Dedon, P.C.; Tannenbaum, S.R. DNA damage in deoxynucleosides and oligonucleotides treated with peroxynitrite. Chem. Res. Toxicol. 1999, 12, 513–520. [Google Scholar] [CrossRef] [PubMed]
- Blair, I.A. DNA adducts with lipid peroxidation products. J. Biol. Chem. 2008, 283, 15545–15549. [Google Scholar] [CrossRef] [PubMed]
- Stonez, M.P.; Cho, Y.J.; Huang, H.; Kim, H.Y.; Kozekov, I.D.; Kozekova, A.; Wang, H.; Minko, I.G.; Lloyd, R.S.; Harris, T.M.; et al. Interstrand DNA cross-links induced by α,β-unsaturated aldehydes derived from lipid peroxidation and environmental sources. Acc. Chem. Res. 2008, 41, 793–804. [Google Scholar] [CrossRef] [PubMed]
- Karbownik, M.; Reiter, R.J.; Cabrera, J.; Garcia, J.J. Comparison of the protective effect of melatonin with other antioxidants in the hamster kidney model of estradiol-induced DNA damage. Mutat. Res. 2001, 474, 87–92. [Google Scholar] [CrossRef]
- Chen, L.J.; Gao, Y.Q.; Li, X.J.; Shen, D.H.; Sun, F.Y. Melatonin protects against MPTP/MPP+-induced mitochondrial DNA oxidative damage in vivo and in vitro. J. Pineal Res. 2005, 39, 34–42. [Google Scholar] [CrossRef] [PubMed]
- Karbownik, M.; Tan, D.X.; Reiter, R.J. Melatonin reduces the oxidation of nuclear DNA and membrane lipids induced by the carcinogen δ-aminolevulinic acid. Int. J. Cancer 2000, 88, 7–11. [Google Scholar] [CrossRef]
- Lebda, M.A.; Sadek, K.M.; Abouzed, T.K.; Tohamy, H.G.; El-Sayed, Y.S. Melatonin mitigates thioacetamide-induced hepatic fibrosis via antioxidant activity and modulation of proinflammatory cytokines and fibrogenic genes. Life Sci. 2018, 192, 136–143. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.J.; Liu, C.; Duan, W.X.; Xu, S.C.; He, M.D.; Chen, C.H.; Wang, Y.; Zhou, Z.; Yu, Z.P.; Zhang, L.; et al. Melatonin ameliorates bisphenol A-induced DNA damage in the germ cells of adult male rats. Mutat. Res. 2013, 752, 57–67. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.; Fu, A.; Hoffman, A.E.; Zheng, T.; Zhu, Y. Melatonin enhances DNA repair capacity possibly by affecting genes involved in DNA damage responsive pathways. BMC Cell Biol. 2013, 14, 1. [Google Scholar] [CrossRef] [PubMed]
- Pappolla, M.A.; Chyan, Y.J.; Poeggeler, B.; Bozner, P.; Ghiso, J.; LeDoux, S.P.; Wilson, G.L. Alzheimer β protein mediated oxidative damage of mitochondrial DNA: Prevention by melatonin. J. Pineal Res. 1999, 27, 226–229. [Google Scholar] [CrossRef] [PubMed]
- Sokolovic, D.; Djordjevic, B.; Kocic, G.; Stoimenov, T.J.; Stanojkovic, Z.; Sokolovic, D.M.; Veljkovic, A.; Ristic, G.; Despotovic, M.; Milisavljevic, D.; et al. The effects of melatonin on oxidative stress parameters and DNA fragmentation in testicular tissue of rats exposed to microwave radiation. Adv. Clin. Exp. Med. 2015, 24, 429–436. [Google Scholar] [CrossRef] [PubMed]
- Das, B.; Bennett, P.V.; Cutter, N.C.; Sutherland, J.C.; Sutherland, B.M. Melatonin protects human cells from clustered DNA damages, killing and acquisition of soft agar growth induced by X-rays or 970 MeV/n Fe ions. Int. J. Radiat. Biol. 2011, 87, 545–555. [Google Scholar] [CrossRef] [PubMed]
- Ündeǧer, Ü.; Giray, B.; Zorlu, A.F.; Öge, K.; Baçaran, N. Protective effects of melatonin on the ionizing radiation induced DNA damage in the rat brain. Exp. Toxicol. Pathol. 2004, 55, 379–384. [Google Scholar] [CrossRef] [PubMed]
- Wakatsuki, A.; Okatani, Y.; Izumiya, C.; Ikenoue, N. Melatonin protects against ischemia and reperfusion-induced oxidative lipid and DNA damage in fetal rat brain. J. Pineal Res. 1999, 26, 147–152. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Zhou, F.; Dou, Y.; Tian, X.; Liu, C.; Li, H.; Shen, H.; Chen, G. Melatonin Alleviates Intracerebral Hemorrhage-Induced Secondary Brain Injury in Rats via Suppressing Apoptosis, Inflammation, Oxidative Stress, DNA Damage, and Mitochondria Injury. Transl. Stroke Res. 2017, 9, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Morioka, N.; Okatani, Y.; Wakatsuki, A. Melatonin protects against age-related DNA damage in the brains of female senescence-accelerated mice. J. Pineal Res. 1999, 27, 202–209. [Google Scholar] [CrossRef] [PubMed]
- Evans, M.D.; Dizdaroglu, M.; Cooke, M.S. Oxidative DNA damage and disease: Induction, repair and significance. Mutat. Res. 2004, 567, 1–61. [Google Scholar] [CrossRef] [PubMed]
- Cooke, M.S.; Evans, M.D.; Dizdaroglu, M.; Lunec, J. Oxidative DNA damage: Mechanisms, mutation, and disease. FASEB J. 2003, 17, 1195–1214. [Google Scholar] [CrossRef] [PubMed]
- Kawanishi, S.; Ohnishi, S.; Ma, N.; Hiraku, Y.; Oikawa, S.; Murata, M. Nitrative and oxidative DNA damage in infection-related carcinogenesis in relation to cancer stem cells. Genes Environ. 2016, 38, 26. [Google Scholar] [CrossRef] [PubMed]
- Murata, M.; Thanan, R.; Ma, N.; Kawanishi, S. Role of nitrative and oxidative DNA damage in inflammation-related carcinogenesis. J. Biomed. Biotechnol. 2012, 2012. [Google Scholar] [CrossRef] [PubMed]
- Kryston, T.B.; Georgiev, A.B.; Pissis, P.; Georgakilas, A.G. Role of oxidative stress and DNA damage in human carcinogenesis. Mutat. Res. 2011, 711, 193–201. [Google Scholar] [CrossRef] [PubMed]
- Markkanen, E. Not breathing is not an option: How to deal with oxidative DNA damage. DNA Repair 2017, 59, 82–105. [Google Scholar] [CrossRef] [PubMed]
- Talhaoui, I.; Matkarimov, B.T.; Tchenio, T.; Zharkov, D.O.; Saparbaev, M.K. Aberrant base excision repair pathway of oxidatively damaged DNA: Implications for degenerative diseases. Free Radic. Biol. Med. 2017, 107, 266–277. [Google Scholar] [CrossRef] [PubMed]
- Kasprzak, K.S. Oxidative DNA and protein damage in metal-induced toxicity and carcinogenesis. Free Radic. Biol. Med. 2002, 32, 958–967. [Google Scholar] [CrossRef]
- Kasai, H. What causes human cancer? Approaches from the chemistry of DNA damage. Genes Environ 2016, 38, 19. [Google Scholar] [CrossRef] [PubMed]
- Smith, J.A.; Park, S.; Krause, J.S.; Banik, N.L. Oxidative stress, DNA damage, and the telomeric complex as therapeutic targets in acute neurodegeneration. Neurochem. Int. 2013, 62, 764–775. [Google Scholar] [CrossRef] [PubMed]
- Santos, R.X.; Correia, S.C.; Zhu, X.; Lee, H.G.; Petersen, R.B.; Nunomura, A.; Smith, M.A.; Perry, G.; Moreira, P.I. Nuclear and mitochondrial DNA oxidation in Alzheimer’s disease. Free Radic. Res. 2012, 46, 565–576. [Google Scholar] [CrossRef] [PubMed]
- Mao, P.; Reddy, P.H. Aging and amyloid beta-induced oxidative DNA damage and mitochondrial dysfunction in Alzheimer’s disease: Implications for early intervention and therapeutics. Biochim. Biophys. Acta 2011, 1812, 1359–1370. [Google Scholar] [CrossRef] [PubMed]
- Markesbery, W.R.; Lovell, M.A. DNA oxidation in Alzheimer’s disease. Antioxid. Redox Signal. 2006, 8, 2039–2045. [Google Scholar] [CrossRef] [PubMed]
- Isobe, C.; Abe, T.; Terayama, Y. Levels of reduced and oxidized coenzyme Q-10 and 8-hydroxy-2′-deoxyguanosine in the CSF of patients with Alzheimer’s disease demonstrate that mitochondrial oxidative damage and/or oxidative DNA damage contributes to the neurodegenerative process. J. Neurol. 2010, 257, 399–404. [Google Scholar] [CrossRef] [PubMed]
- Isobe, C.; Abe, T.; Terayama, Y. Levels of reduced and oxidized coenzymeQ-10 and 8-hydroxy-2′-deoxyguanosine in the cerebrospinal fluid of patients with living Parkinson’s disease demonstrate that mitochondrial oxidative damage and/or oxidative DNA damage contributes to the neurodegenerative process. Neurosci. Lett. 2010, 469, 159–163. [Google Scholar] [PubMed]
- Gackowski, D.; Rozalski, R.; Siomek, A.; Dziaman, T.; Nicpon, K.; Klimarczyk, M.; Araszkiewicz, A.; Olinski, R. Oxidative stress and oxidative DNA damage is characteristic for mixed Alzheimer disease/vascular dementia. J. Neurol. Sci. 2008, 266, 57–62. [Google Scholar] [CrossRef] [PubMed]
- Murata, T.; Ohtsuka, C.; Terayama, Y. Increased mitochondrial oxidative damage and oxidative DNA damage contributes to the neurodegenerative process in sporadic amyotrophic lateral sclerosis. Free Radic. Res. 2008, 42, 221–225. [Google Scholar] [CrossRef] [PubMed]
- Gmitterová, K.; Gawinecka, J.; Heinemann, U.; Valkovič, P.; Zerr, I. DNA versus RNA oxidation in Parkinson’s disease: Which is more important? Neurosci. Lett. 2018, 662, 22–28. [Google Scholar] [CrossRef] [PubMed]
- Malik, Q.; Herbert, K.E. Oxidative and non-oxidative DNA damage and cardiovascular disease. Free Radic. Res. 2012, 46, 554–564. [Google Scholar] [CrossRef] [PubMed]
- Satoh, M.; Ishikawa, Y.; Takahashi, Y.; Itoh, T.; Minami, Y.; Nakamura, M. Association between oxidative DNA damage and telomere shortening in circulating endothelial progenitor cells obtained from metabolic syndrome patients with coronary artery disease. Atherosclerosis 2008, 198, 347–353. [Google Scholar] [CrossRef] [PubMed]
- Mondal, N.K.; Sorensen, E.; Hiivala, N.; Feller, E.; Griffith, B.; Wu, Z.J. Oxidative stress, DNA damage and repair in heart failure patients after implantation of continuous flow left ventricular assist devices. Int. J. Med. Sci. 2013, 10, 883–893. [Google Scholar] [CrossRef] [PubMed]
- Kono, Y.; Nakamura, K.; Kimura, H.; Nishii, N.; Watanabe, A.; Banba, K.; Miura, A.; Nagase, S.; Sakuragi, S.; Kusano, K.F.; et al. Elevated levels of oxidative DNA damage in serum and myocardium of patients with heart failure. Circ. J. 2006, 70, 1001–1005. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.; Guerrero, C.R.; Liu, S.; Amato, N.J.; Sharma, Y.; Gupta, S.; Wang, Y. Comprehensive assessment of oxidatively induced modifications of DNA in a rat model of human Wilson’s disease. Mol. Cell. Proteom. 2016, 15, 810–817. [Google Scholar] [CrossRef] [PubMed]
- Mansouri, A.; Gaou, I.; Fromenty, B.; Berson, A.; Letteron, P.; Degott, C.; Erlinger, S.; Pessayre, D. Premature oxidative aging of hepatic mitochondrial DNA in Wilson’s disease. Gastroenterology 1997, 113, 599–605. [Google Scholar] [CrossRef] [PubMed]
- Pereira, C.; Coelho, R.; Grácio, D.; Dias, C.; Silva, M.; Peixoto, A.; Lopes, P.; Costa, C.; Teixeira, J.P.; Macedo, G.; et al. DNA damage and oxidative DNA damage in inflammatory bowel disease. J. Crohn’s Colitis 2016, 10, 1316–1323. [Google Scholar] [CrossRef] [PubMed]
- Sohn, J.J.; Schetter, A.J.; Yfantis, H.G.; Ridnour, L.A.; Horikawa, I.; Khan, M.A.; Robles, A.I.; Hussain, S.P.; Goto, A.; Bowman, E.D.; et al. Macrophages, nitric oxide and microRNAs are associated with DNA damage response pathway and senescence in inflammatory bowel disease. PLoS ONE 2012, 7, e44156. [Google Scholar] [CrossRef] [PubMed]
- Tatsch, E.; Bochi, G.V.; Piva, S.J.; De Carvalho, J.A.M.; Kober, H.; Torbitz, V.D.; Duarte, T.; Signor, C.; Coelho, A.C.; et al. Association between DNA strand breakage and oxidative, inflammatory and endothelial biomarkers in type 2 diabetes. Mutat. Res. 2012, 732, 16–20. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Gan, W.; Zou, Y.; Yang, B.; Su, Z.; Deng, J.; Wang, L.; Cai, J. Elevated levels of urinary markers of oxidative DNA and RNA damage in type 2 diabetes with complications. Oxid. Med. Cell. Longev. 2016, 2016. [Google Scholar] [CrossRef] [PubMed]
- Tatsch, E.; Carvalho, J.A.; Hausen, B.S.; Bollick, Y.S.; Torbitz, V.D.; Duarte, T.; Scolari, R.; Duarte, M.M.M.F.; Londero, S.W.K.; Vaucher, R.A.; et al. Oxidative DNA damage is associated with inflammatory response, insulin resistance and microvascular complications in type 2 diabetes. Mutat. Res. 2015, 782, 17–22. [Google Scholar] [CrossRef] [PubMed]
- Tabak, O.; Gelisgen, R.; Erman, H.; Erdenen, F.; Muderrisoglu, C.; Aral, H.; Uzun, H. Oxidative lipid, protein, and DNA damage as oxidative stress markers in vascular complications of diabetes mellitus. Clin. Investig. Med. 2011, 34, E163–E171. [Google Scholar] [CrossRef]
- Olinski, R.; Gackowski, D.; Foksinski, M.; Rozalski, R.; Roszkowski, K.; Jaruga, P. Oxidative DNA damage: Assessment of the role in carcinogenesis, atherosclerosis, and acquired immunodeficiency syndrome. Free Radic. Biol. Med. 2002, 33, 192–200. [Google Scholar] [CrossRef]
- Kolgiri, V.; Patil, V.W.; Nagar, V. Correlation of total antioxidant status (TAS) with DNA damage in HIV/AIDS patients. Int. J. Pharm. Pharm. Sci. 2016, 8, 240–244. [Google Scholar]
- De Luca, G.; Russo, M.T.; Degan, P.; Tiveron, C.; Zijno, A.; Meccia, E.; Ventura, I.; Mattei, E.; Nakabeppu, Y.; Crescenzi, M.; et al. A role for oxidized DNA precursors in Huntington’s disease-like striatal neurodegeneration. PLoS Genet. 2008, 4, e1000266. [Google Scholar] [CrossRef] [PubMed]
- Ayala-Peña, S. Role of oxidative DNA damage in mitochondrial dysfunction and Huntington’s disease pathogenesis. Free Radic. Biol. Med. 2013, 62, 102–110. [Google Scholar] [CrossRef] [PubMed]
- Seven, A.; Güzel, S.; Aslan, M.; Hamuryudan, V. Lipid, protein, DNA oxidation and antioxidant status in rheumatoid arthritis. Clin. Biochem. 2008, 41, 538–543. [Google Scholar] [CrossRef] [PubMed]
- Altindag, O.; Karakoc, M.; Kocyigit, A.; Celik, H.; Soran, N. Increased DNA damage and oxidative stress in patients with rheumatoid arthritis. Clin. Biochem. 2007, 40, 167–171. [Google Scholar] [CrossRef] [PubMed]
- Hajizadeh, S.; DeGroot, J.; TeKoppele, J.M.; Tarkowski, A.; Collins, L.V. Extracellular mitochondrial DNA and oxidatively damaged DNA in synovial fluid of patients with rheumatoid arthritis. Arthritis Res. Ther. 2003, 5, R234–240. [Google Scholar] [CrossRef] [PubMed]
- Neofytou, E.; Tzortzaki, E.G.; Chatziantoniou, A.; Siafakas, N.M. DNA damage due to oxidative stress in chronic obstructive pulmonary disease (COPD). Int. J. Mol. Sci. 2012, 13, 16853–16864. [Google Scholar] [CrossRef] [PubMed]
- Galano, A. Free radicals induced oxidative stress at a molecular level: The current status, challenges and perspectives of computational chemistry based protocols. J. Mex. Chem. Soc. 2015, 59, 231–262. [Google Scholar]
- Alvarez-Idaboy, J.R.; Galano, A. On the chemical repair of DNA radicals by glutathione: Hydrogen vs. electron transfer. J. Phys. Chem. B 2012, 116, 9316–9325. [Google Scholar] [CrossRef] [PubMed]
- Wiechmann, A.F.; Sherry, D.M. Role of melatonin and its receptors in the vertebrate retina. Int. Rev. Cell Mol. Biol. 2013, 300, 211–242. [Google Scholar] [PubMed]
- Kim, T.K.; Kleszczynśki, K.; Janjetovic, Z.; Sweatman, T.; Lin, Z.; Li, W.; Reiter, R.J.; Fischer, T.W.; Slominski, A.T. Metabolism of melatonin and biological activity of intermediates of melatoninergic pathway in human skin cells. FASEB J. 2013, 27, 2742–2755. [Google Scholar] [CrossRef] [PubMed]
- Pinato, L.; da Silveira Cruz-Machado, S.; Franco, D.G.; Campos, L.M.G.; Cecon, E.; Fernandes, P.A.C.M.; Bittencourt, J.C.; Markus, R.P. Selective protection of the cerebellum against intracerebroventricular LPS is mediated by local melatonin synthesis. Brain Struct. Funct. 2013, 220, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Lahiri, D.K.; Ge, Y.W.; Sharman, E.H.; Bondy, S.C. Age-related changes in serum melatonin in mice: Higher levels of combined melatonin and 6-hydroxymelatonin sulfate in the cerebral cortex than serum, heart, liver and kidney tissues. J. Pineal Res. 2004, 36, 217–223. [Google Scholar] [CrossRef] [PubMed]
- Cruz, M.H.C.; Leal, C.L.V.; Cruz, J.F.; Tan, D.X.; Reiter, R.J. Essential actions of melatonin in protecting the ovary from oxidative damage. Theriogenology 2014, 82, 925–932. [Google Scholar] [CrossRef] [PubMed]
- Peschke, E. Melatonin, endocrine pancreas and diabetes. J. Pineal Res. 2008, 44, 26–40. [Google Scholar] [CrossRef] [PubMed]
- Acuna-Castroviejo, D.; Lowenstein, P.R.; Rosenstein, R.; Cardinali, D.P. Diurnal variations of benzodiazepine binding in rat cerebral cortex: Disruption by pinealectomy. J. Pineal Res. 1986, 3, 101–109. [Google Scholar] [CrossRef] [PubMed]
- Reiter, R.J. The melatonin rhythm: both a clock and a calendar. Experientia 1993, 49, 654–664. [Google Scholar] [CrossRef] [PubMed]
- Reiter, R.J. Circannual reproductive rhythms in mammals related to photoperiod and pineal function: A review. Chronobiologia 1974, 1, 365–395. [Google Scholar] [PubMed]
- Reiter, R.J.; Tan, D.X.; Fuentes-Broto, L. Melatonin: A multi-tasking molecule. Prog. Brain Res. 2010, 181, 127–151. [Google Scholar] [PubMed]
- Jung, K.H.; Hong, S.W.; Zheng, H.M.; Lee, H.S.; Lee, H.; Lee, D.H.; Lee, S.Y.; Hong, S.S. Melatonin ameliorates cerulein-induced pancreatitis by the modulation of nuclear erythroid 2-related factor 2 and nuclear factor-kappaB in rats. J. Pineal Res. 2010, 48, 239–250. [Google Scholar] [CrossRef] [PubMed]
- Chahbouni, M.; Escames, G.; Venegas, C.; Sevilla, B.; García, J.A.; López, L.C.; Muñoz-Hoyos, A.; Molina-Carballo, A.; Acuna-Castroviejo, D. Melatonin treatment normalizes plasma pro-inflammatory cytokines and nitrosative/oxidative stress in patients suffering from Duchenne muscular dystrophy. J. Pineal Res. 2010, 48, 282–289. [Google Scholar] [CrossRef] [PubMed]
- Carrillo-Vico, A.; Guerrero, J.M.; Lardone, P.J.; Reiter, R.J. A review of the multiple actions of melatonin on the immune system. Endocrine 2005, 27, 189–200. [Google Scholar] [CrossRef]
- Jou, M.J.; Peng, T.I.; Hsu, L.F.; Jou, S.B.; Reiter, R.J.; Yang, C.M.; Chiao, C.C.; Lin, Y.F.; Chen, C.C. Visualization of melatonin’s multiple mitochondrial levels of protection against mitochondrial Ca2+-mediated permeability transition and beyond in rat brain astrocytes. J. Pineal Res. 2010, 48, 20–38. [Google Scholar] [CrossRef] [PubMed]
- Paradies, G.; Petrosillo, G.; Paradies, V.; Reiter, R.J.; Ruggiero, F.M. Melatonin, cardiolipin and mitochondrial bioenergetics in health and disease. J. Pineal Res. 2010, 48, 297–310. [Google Scholar] [CrossRef] [PubMed]
- Milczarek, R.; Hallmann, A.; Sokołowska, E.; Kaletha, K.; Klimek, J. Melatonin enhances antioxidant action of α-tocopherol and ascorbate against NADPH- and iron-dependent lipid peroxidation in human placental mitochondria. J. Pineal Res. 2010, 49, 149–155. [Google Scholar] [CrossRef] [PubMed]
- García, J.J.; Lõpez-Pingarrõn, L.; Almeida-Souza, P.; Tres, A.; Escudero, P.; García-Gil, F.A.; Tan, D.X.; Reiter, R.J.; Ramírez, J.M.; et al. Protective effects of melatonin in reducing oxidative stress and in preserving the fluidity of biological membranes: A review. J. Pineal Res. 2014, 56, 225–237. [Google Scholar] [CrossRef] [PubMed]
- Reiter, R.J.; Rosales-Corral, S.; Tan, D.X.; Jou, M.J.; Galano, A.; Xu, B. Melatonin as a mitochondria-targeted antioxidant: one of evolution’s best ideas. Cell. Mol. Life Sci. 2017, 74, 3863–3881. [Google Scholar] [CrossRef] [PubMed]
- Majidinia, M.; Sadeghpour, A.; Mehrzadi, S.; Reiter, R.J.; Khatami, N.; Yousefi, B. Melatonin: A pleiotropic molecule that modulates DNA damage response and repair pathways. J. Pineal Res. 2017, 63, e12416. [Google Scholar] [CrossRef] [PubMed]
- Galano, A.; Castañeda-Arriaga, R.; Pérez-González, A.; Tan, D.X.; Reiter, R.J. Phenolic melatonin-related compounds: Their role as chemical protectors against oxidative stress. Molecules 2016, 21, 1442. [Google Scholar] [CrossRef] [PubMed]
- Galano, A.; Tan, D.-X.; Reiter, R.J. Melatonin and related compounds: chemical insights into their protective effects against oxidative stress. Curr. Org. Chem. 2016, 21, 1–19. [Google Scholar] [CrossRef]
- Sliwinski, T.; Rozej, W.; Morawiec-Bajda, A.; Morawiec, Z.; Reiter, R.; Blasiak, J. Protective action of melatonin against oxidative DNA damage-Chemical inactivation versus base-excision repair. Mutat. Res. 2007, 634, 220–227. [Google Scholar] [CrossRef] [PubMed]
- George, A. The Role of Indoleamines in Reducing Free Radical Damage and Oxidative Stress: A Physicochemical Perspective. In Indoleamines: Sources, Role in Biological Processes and Health Effects; Nova Science Publishers, Inc.: New York, NY, USA, 2015; pp. 1–41. ISBN 978-1-63482-098-1. [Google Scholar]
- Reiter, R.J.; Tan, D.X.; Rosales-Corral, S.; Manchester, L.C. The universal nature, unequal distribution and antioxidant functions of melatonin and its derivatives. Mini-Rev. Med. Chem. 2013, 13, 373–384. [Google Scholar] [PubMed]
- Hevia, D.; González-Menéndez, P.; Quiros-González, I.; Miar, A.; Rodríguez-García, A.; Tan, D.X.; Reiter, R.J.; Mayo, J.C.; Sainz, R.M. Melatonin uptake through glucose transporters: A new target for melatonin inhibition of cancer. J. Pineal Res. 2015, 58, 234–250. [Google Scholar] [CrossRef] [PubMed]
- Andersen, L.P.H.; Gögenur, I.; Rosenberg, J.; Reiter, R.J. The safety of melatonin in humans. Clin. Drug Investig. 2016, 36, 169–175. [Google Scholar] [CrossRef] [PubMed]
- Arendt, J. Safety of melatonin in long-term use (?). J. Biol. Rhythms 1997, 12, 673–681. [Google Scholar] [CrossRef] [PubMed]
- Gringras, P.; Nir, T.; Breddy, J.; Frydman-Marom, A.; Findling, R.L. Efficacy and safety of pediatric prolonged-release melatonin for insomnia in children with autism spectrum disorder. J. Am. Acad. Child Adolesc. Psychiatry 2017, 56, 948–957. [Google Scholar] [CrossRef] [PubMed]
- Vijayalaxmi; Meltz, M.L.; Reiter, R.J.; Herman, T.S. Melatonin and protection from genetic damage in blood and bone marrow: Whole-body irradiation studies in mice. J. Pineal Res. 1999, 27, 221–225. [Google Scholar]
- Jahnke, G.; Marr, M.; Myers, C.; Wilson, R.; Travlos, G.; Price, C. Maternal and developmental toxicity evaluation of melatonin administered orally to pregnant Sprague-Dawley rats. Toxicol. Sci. 1999, 50, 271–279. [Google Scholar] [CrossRef] [PubMed]
- Vijayalaxmi; Meltz, M.L.; Reiter, R.J.; Herman, T.S.; Kumar K, S. Melatonin and protection from whole-body irradiation: Survival studies in mice. Mutat. Res. 1999, 425, 21–27. [Google Scholar] [CrossRef]
- Kaya, H.; Delibas, N.; Serteser, M.; Ulukaya, E.; Özkaya, O. The effect of melatonin on lipid peroxidation during radiotherapy in female rats. Strahlenther. Onkol. 1999, 175, 285–288. [Google Scholar] [CrossRef] [PubMed]
- Nordlund, J.J.; Lerner, A.B. The effects of oral melatonin on skin color and on the release of pituitary hormones. J. Clin. Endocrinol. Metab. 1977, 45, 768–774. [Google Scholar] [CrossRef] [PubMed]
- Sharman, E.H.; Bondy, S.C. Melatonin: A safe nutraceutical and clinical agent. Nutraceuticals 2016, 501–509. [Google Scholar] [CrossRef]
- Yoshida, M.; Fukuda, A.; Hara, M.; Terada, A.; Kitanaka, Y.; Owada, S. Melatonin prevents the increase in hydroxyl radical-spin trap adduct formation caused by the addition of cisplatin in vitro. Life Sci. 2003, 72, 1773–1780. [Google Scholar] [CrossRef]
- Reiter, R.J.; Acuña-Castroviejo, D.; Tan, D.X.; Burkhardt, S. Free radical-mediated molecular damage: Mechanisms for the protective actions of melatonin in the central nervous system. Ann. N. Y. Acad. Sci. 2001, 939, 200–215. [Google Scholar] [CrossRef] [PubMed]
- Escames, G.; Guerrero, J.M.; Reiter, R.J.; Garcia, J.J.; Munoz-Hoyos, A.; Ortiz, G.G.; Oh, C.S. Melatonin and vitamin E limit nitric oxide-induced lipid peroxidation in rat brain homogenates. Neurosci. Lett. 1997, 230, 147–150. [Google Scholar] [CrossRef]
- Siu, A.W.; Ortiz, G.G.; Benitez-King, G.; To, C.H.; Reiter, R.J. Effect of melatonin on the nitric oxide treated retina. Br. J. Ophthalmol. 2004, 88, 1078–1081. [Google Scholar] [CrossRef] [PubMed]
- Zavodnik, I.B.; Domanski, A.V.; Lapshina, E.A.; Bryszewska, M.; Reiter, R.J. Melatonin directly scavenges free radicals generated in red blood cells and a cell-free system: Chemiluminescence measurements and theoretical calculations. Life Sci. 2006, 79, 391–400. [Google Scholar] [CrossRef] [PubMed]
- Tan, D.X.; Manchester, L.C.; Reiter, R.J.; Plummer, B.F.; Limson, J.; Weintraub, S.T.; Qi, W. Melatonin directly scavenges hydrogen peroxide: A potentially new metabolic pathway of melatonin biotransformation. Free Radic. Biol. Med. 2000, 29, 1177–1185. [Google Scholar] [CrossRef]
- Romero, M.P.; Osuna, C.; García-Pergañeda, A.; Carrillo-Vico, A.; Guerrero, J.M. The pineal secretory product melatonin reduces hydrogen peroxide- induced DNA damage in U-937 cells. J. Pineal Res. 1999, 26, 227–235. [Google Scholar] [CrossRef] [PubMed]
- Reiter, R.J.; Tan, D.X.; Osuna, C.; Gitto, E. Actions of melatonin in the reduction of oxidative stress: A review. J. Biomed. Sci. 2000, 7, 444–458. [Google Scholar] [CrossRef] [PubMed]
- Gurer-Orhan, H.; Suzen, S. Melatonin, its metabolites and its synthetic analogs as multi-faceted compounds: Antioxidant, prooxidant and inhibitor of bioactivation reactions. Curr. Med. Chem. 2015, 22, 490–499. [Google Scholar] [CrossRef] [PubMed]
- Suzen, S. Melatonin and synthetic analogs as antioxidants. Curr. Drug Del. 2013, 10, 71–75. [Google Scholar] [CrossRef]
- Ateş-Alagöz, Z.; Coban, T.; Suzen, S. A comparative study: Evaluation of antioxidant activity of melatonin and some indole derivatives. Med. Chem. Res. 2005, 14, 169–179. [Google Scholar] [CrossRef]
- Suzen, S.; Bozkaya, P.; Coban, T.; Nebioǧlu, D. Investigation of the in vitro antioxidant behaviour of some 2-phenylindole derivatives: Discussion on possible antioxidant mechanisms and comparison with melatonin. J. Enzyme Inhib. Med. Chem. 2006, 21, 405–411. [Google Scholar] [CrossRef] [PubMed]
- Shirinzadeh, H.; Eren, B.; Gurer-Orhan, H.; Suzen, S.; Özden, S. Novel indole-based analogs of melatonin: Synthesis and in vitro antioxidant activity studies. Molecules 2010, 15, 2187–2202. [Google Scholar] [CrossRef] [PubMed]
- Yilmaz, A.D.; Coban, T.; Suzen, S. Synthesis and antioxidant activity evaluations of melatonin-based analogue indole-hydrazide/hydrazone derivatives. J. Enzyme Inhib. Med. Chem. 2012, 27, 428–436. [Google Scholar] [CrossRef] [PubMed]
- Gürkök, G.; Coban, T.; Suzen, S. Melatonin analogue new indole hydrazide/hydrazone derivatives with antioxidant behavior: Synthesis and structureactivity relationships. J. Enzyme Inhib. Med. Chem. 2009, 24, 506–515. [Google Scholar] [CrossRef] [PubMed]
- Suzen, S.; Cihaner, S.S.; Coban, T. Synthesis and comparison of antioxidant properties of indole-based melatonin analogue indole amino acid derivatives. Chem. Biol. Drug Des. 2012, 79, 76–83. [Google Scholar] [CrossRef] [PubMed]
- Liang, S.; Jin, Y.X.; Yuan, B.; Zhang, J.B.; Kim, N.H. Melatonin enhances the developmental competence of porcine somatic cell nuclear transfer embryos by preventing DNA damage induced by oxidative stress. Sci. Rep. 2017, 7, 11114. [Google Scholar] [CrossRef] [PubMed]
- Erenberk, U.; Dundaroz, R.; Gok, O.; Uysal, O.; Agus, S.; Yuksel, A.; Yilmaz, B.; Kilic, U. Melatonin attenuates phenytoin sodium-induced DNA damage. Drug Chem. Toxicol. 2014, 37, 233–239. [Google Scholar] [CrossRef] [PubMed]
- Sekkin, S.; İpek, E.D.; Boyacioğlu, M.; Kum, C.; Karademir, Ü.; Yalinkilinç, H.S.; Ak, M.O.; Başaloğlu, H. DNA protective and antioxidative effects of melatonin in streptozotocin-induced diabetic rats. Turkish J. Biol. 2015, 39, 932–940. [Google Scholar] [CrossRef]
- Shokrzadeh, M.; Naghshvar, F.; Ahmadi, A.; Chabra, A.; Jeivad, F. The potential ameliorative effects of melatonin against cyclophosphamide-induced DNA damage in murine bone marrow cells. Eur. Rev. Med. Pharmacol. Sci. 2014, 18, 605–611. [Google Scholar] [PubMed]
- Espino, J.; Bejarano, I.; Ortiz, A.; Lozano, G.M.; García, J.F.; Pariente, J.A.; Rodríguez, A.B. Melatonin as a potential tool against oxidative damage and apoptosis in ejaculated human spermatozoa. Fertil. Steril. 2010, 94, 1915–1917. [Google Scholar] [CrossRef] [PubMed]
- Bejarano, I.; Monllor, F.; Marchena, A.M.; Ortiz, A.; Lozano, G.; Jiménez, M.I.; Gaspar, P.; García, J.F.; Pariente, J.A.; Rodríguez, A.B.; Espino, J. Exogenous melatonin supplementation prevents oxidative stress-evoked DNA damage in human spermatozoa. J. Pineal Res. 2014, 57, 333–339. [Google Scholar] [CrossRef] [PubMed]
- Reiter, R.J.; Rosales-Corral, S.A.; Manchester, L.C.; Tan, D.X. Peripheral reproductive organ health and melatonin: Ready for prime time. Int. J. Mol. Sci. 2013, 14, 7231–7272. [Google Scholar] [CrossRef] [PubMed]
- Vargas, A.; Bustos-Obregón, E.; Hartley, R. Effects of hypoxia on epididymal sperm parameters and protective role of ibuprofen and melatonin. Biol. Res. 2011, 44, 161–167. [Google Scholar] [CrossRef] [PubMed]
- Liang, S.; Guo, J.; Choi, J.W.; Kim, N.H.; Cui, X.S. Effect and possible mechanisms of melatonin treatment on the quality and developmental potential of aged bovine oocytes. Reprod. Fertil. Dev. 2017, 29, 1821–1831. [Google Scholar] [CrossRef] [PubMed]
- He, C.; Wang, J.; Zhang, Z.; Yang, M.; Li, Y.; Tian, X.; Ma, T.; Tao, J.; Zhu, K.; Song, Y.; et al. Mitochondria synthesize melatonin to ameliorate its function and improve mice oocyte’s quality under in vitro conditions. Int. J. Mol. Sci. 2016, 17, 939. [Google Scholar] [CrossRef] [PubMed]
- Kulms, D.; Zeise, E.; Pöppelmann, B.; Schwarz, T. DNA damage, death receptor activation and reactive oxygen species contribute to ultraviolet radiation-induced apoptosis in an essential and independent way. Oncogene 2002, 21, 5844–5851. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, N.U.; Ueda, M.; Nikaido, O.; Osawa, T.; Ichihashi, M. High levels of 8-hydroxy-2′-deoxyguanosine appear in normal human epidermis after a single dose of ultraviolet radiation. Br. J. Dermatol. 1999, 140, 226–231. [Google Scholar] [CrossRef] [PubMed]
- Fischer, T.W.; Kleszczyński, K.; Hardkop, L.H.; Kruse, N.; Zillikens, D. Melatonin enhances antioxidative enzyme gene expression (CAT, GPx, SOD), prevents their UVR-induced depletion, and protects against the formation of DNA damage (8-hydroxy-2′-deoxyguanosine) in ex vivo human skin. J. Pineal Res. 2013, 54, 303–312. [Google Scholar] [CrossRef] [PubMed]
- Al-Jassabi, S.; Khalil, A.M. Microcystin-induced 8-hydroxydeoxyguanosine in DNA and its reduction by melatonin, vitamin C, and vitamin e in mice. Biochemistry 2006, 71, 1115–1119. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, H.A.; Mohanan, P.V. In vivo and in vitro effects of melatonin or ganglioside GT1B on l-cysteine-induced brain mitochondrial DNA damage in mice. Toxicol. Sci. 2003, 73, 416–422. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, H.A.; Mohanan, P.V. Ganglioside GT1B and melatonin inhibit brain mitochondrial DNA damage and seizures induced by kainic acid in mice. Brain Res. 2003, 964, 100–106. [Google Scholar] [CrossRef]
- Mohanan, P.V.; Yamamoto, H.A. Preventive effect of melatonin against brain mitochondria DNA damage, lipid peroxidation and seizures induced by kainic acid. Toxicol. Lett. 2002, 129, 99–105. [Google Scholar] [CrossRef]
- Yamamoto, H.; Mohanan, P.V. Melatonin attenuates brain mitochondria DNA damage induced by potassium cyanide in vivo and in vitro. Toxicology 2002, 179, 29–36. [Google Scholar] [CrossRef]
- Yamamoto, H.A.; Mohanan, P.V. Preventive effect of melatonin against DNA damage induced by cyanide, kainate, glutathione/Fe3+/O2, or H2O2/Fe2+. J. Pineal Res. 2001, 31, 314–319. [Google Scholar] [CrossRef] [PubMed]
- Hsu, C.H.; Chi, B.C.; Casida, J.E. Melatonin reduces phosphine-induced lipid and DNA oxidation in vitro and in vivo in rat brain. J. Pineal Res. 2002, 32, 53–58. [Google Scholar] [CrossRef] [PubMed]
- Bagchi, M.; Balmoori, J.; Ye, X.; Bagchi, D.; Ray, S.D.; Stohs, S.J. Protective effect of melatonin on naphthalene-induced oxidative stress and DNA damage in cultured macrophage J774A.1 cells. Mol. Cell. Biochem. 2001, 221, 49–55. [Google Scholar] [CrossRef] [PubMed]
- Lai, H.; Singh, N.P. Melatonin and a spin-trap compound block radiofrequency electromagnetic radiation-induced DNA strand breaks in rat brain cells. Bioelectromagnetics 1997, 18, 446–454. [Google Scholar] [CrossRef]
- Shaikh, A.Y.; Xu, J.; Wu, Y.; He, L.; Hsu, C.Y. Melatonin protects bovine cerebral endothelial cells from hyperoxia- induced DNA damage and death. Neurosci. Lett. 1997, 229, 193–197. [Google Scholar] [CrossRef]
- Miche, H.; Brumas, V.; Berthon, G. Copper(II) interactions with nonsteroidal antiinflammatory agents. II. Anthranilic acid as a potential OH-inactivating ligand. J. Inorg. Biochem. 1997, 68, 27–38. [Google Scholar] [CrossRef]
- Gaubert, S.; Bouchaut, M.; Brumas, V.; Berthon, G. Copper-ligand interactions and physiological free radical processes. Part 3. Influence of histidine, salicylic acid and anthranilic acid on copper-driven Fenton chemistry in vitro. Free Radic. Res. 2000, 32, 451–461. [Google Scholar] [CrossRef] [PubMed]
- Berthon, G. Is copper pro- or anti-inflammatory? A reconciling view and a novel approach for the use of copper in the control of inflammation. Agents Actions 1993, 39, 210–217. [Google Scholar] [CrossRef] [PubMed]
- Limson, J.; Nyokong, T.; Daya, S. The interaction of melatonin and its precursors with aluminium, cadmium, copper, iron, lead, and zinc: An adsorptive voltammetric study. J. Pineal Res. 1998, 24, 15–21. [Google Scholar] [CrossRef] [PubMed]
- López-Burillo, S.; Tan, D.X.; Mayo, J.C.; Sainz, R.M.; Manchester, L.C.; Reiter, R.J. Melatonin, xanthurenic acid, resveratrol, EGCG, vitamin C and α-lipoic acid differentially reduce oxidative DNA damage induced by Fenton reagents: A study of their individual and synergistic actions. J. Pineal Res. 2003, 34, 269–277. [Google Scholar] [CrossRef] [PubMed]
- Zatta, P.; Tognon, G.; Carampin, P. Melatonin prevents free radical formation due to the interaction between β-amyloid peptides and metal ions [Al(III), Zn(II), Cu(II), Mn(II), Fe(II)]. J. Pineal Res. 2003, 35, 98–103. [Google Scholar] [CrossRef] [PubMed]
- Romero, A.; Ramos, E.; De Los Ríos, C.; Egea, J.; Del Pino, J.; Reiter, R.J. A review of metal-catalyzed molecular damage: Protection by melatonin. J. Pineal Res. 2014, 56, 343–370. [Google Scholar] [CrossRef] [PubMed]
- Qi, W.; Reiter, R.J.; Tan, D.X.; Manchester, L.C.; Calvo, J.R. Melatonin prevents δ-aminolevulinic acid-induced oxidative DNA damage in the presence of Fe2+. Mol. Cell. Biochem. 2001, 218, 87–92. [Google Scholar] [CrossRef] [PubMed]
- Onuki, J.; Almeida, E.A.; Medeiros, M.H.G.; Di Mascio, P. Inhibition of 5-aminolevulinic acid-induced DNA damage by melatonin, N1-acetyl-N2-formyl-5-methoxykynuramine, quercetin or resveratrol. J. Pineal Res. 2005, 38, 107–115. [Google Scholar] [CrossRef] [PubMed]
- Qi, W.; Reiter, R.J.; Tan, D.X.; Manchester, L.C.; Kim, S.J.; Garcia, J.J. Inhibitory effects of melatonin on ferric nitrilotriacetate-induced lipid peroxidation and oxidative DNA damage in the rat kidney. Toxicology 1999, 139, 81–91. [Google Scholar] [CrossRef]
- Jajte, J.; Zmyślony, M.; Palus, J.; Dziubałtowska, E.; Raikowska, E. Protective effect of melatonin against in vitro iron ions and 7 mT 50 Hz magnetic field-induced DNA damage in rat lymphocytes. Mutat. Res. 2001, 483, 57–64. [Google Scholar] [CrossRef]
- Aruoma, O.I.; Halliwell, B.; Gajewski, E.; Dizdaroglu, M. Copper-ion-dependent damage to the bases in DNA in the presence of hydrogen peroxide. Biochem. J. 1991, 273, 601–604. [Google Scholar] [CrossRef] [PubMed]
- Aiyar, J.; Berkovits, H.J.; Floyd, R.A.; Wetterhahn, K.E. Reaction of chromium(VI) with hydrogen peroxide in the presence of glutathione: reactive intermediates and resulting DNA damage. Chem. Res. Toxicol. 1990, 3, 595–603. [Google Scholar] [CrossRef] [PubMed]
- Susa, N.; Ueno, S.; Furukawa, Y.; Ueda, J.; Sugiyama, M. Potent protective effect of melatonin on chromium(VI)-induced DNA single-strand breaks, cytotoxicity, and lipid peroxidation in primary cultures of rat hepatocytes. Toxicol. Appl. Pharmacol. 1997, 144, 377–384. [Google Scholar] [CrossRef] [PubMed]
- Qi, W.; Reiter, R.J.; Tan, D.X.; Garcia, J.J.; Manchester, L.C.; Karbownik, M.; Calvo, J.R. Chromium(III)-induced 8-hydroxydeoxyguanosine in DNA and its reduction by antioxidants: Comparative effects of melatonin, ascorbate, and vitamin E. Environ. Health Perspect. 2000, 108, 399–403. [Google Scholar] [CrossRef] [PubMed]
- Cabrera, J.; Burkhardt, S.; Tan, D.X.; Manchester, L.C.; Karbownik, M.; Reiter, R.J. Autoxidation and toxicant-induced oxidation of lipid and DNA in monkey liver: Reduction of molecular damage by melatonin. Pharmacol. Toxicol. 2001, 89, 225–230. [Google Scholar] [CrossRef]
- Beyersmann, D.; Hartwig, A. Carcinogenic metal compounds: Recent insight into molecular and cellular mechanisms. Arch. Toxicol. 2008, 82, 493–512. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Alfaro, M.; Hernández-Cortés, D.; Wrobel, K.; Cruz-Jiménez, G.; Rivera-Leyva, J.C.; Piña-Zentella, R.M.; Cárabez Trejo, A. Effect of melatonin administration on DNA damage and repair responses in lymphocytes of rats subchronically exposed to lead. Mutat. Res. 2012, 742, 37–42. [Google Scholar] [CrossRef] [PubMed]
- Xu, S.C.; He, M.D.; Lu, Y.H.; Li, L.; Zhong, M.; Zhang, Y.W.; Wang, Y.; Yu, Z.P.; Zhou, Z. Nickel exposure induces oxidative damage to mitochondrial DNA in Neuro2a cells: The neuroprotective roles of melatonin. J. Pineal Res. 2011, 51, 426–433. [Google Scholar] [CrossRef] [PubMed]
- Hengstler, J.G.; Bolm-Audorff, U.; Faldum, A.; Janssen, K.; Reifenrath, M.; Götte, W.; Jung, D.; Mayer-Popken, O.; Fuchs, J.; Gebhard, S.; et al. Occupational exposure to heavy metals: DNA damage induction and DNA repair inhibition prove co-exposures to cadmium, cobalt and lead as more dangerous than hitherto expected. Carcinogenesis 2003, 24, 63–73. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Hazra, T.K.; Mitra, S.; Lee, H.M.; Englander, E.W. Mitochondrial DNA damage and a hypoxic response are induced by CoCl2 in rat neuronal PC12 cells. Nucleic Acids Res. 2000, 28, 2135–2140. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Yang, X.; Wang, W.; Wu, X.; Zhu, H.; Liu, F. Melatonin counteracts cobalt nanoparticle-induced cytotoxicity and genotoxicity by deactivating reactive oxygen species-dependent mechanisms in the NRK cell line. Mol. Med. Report. 2017, 16, 4413–4420. [Google Scholar] [CrossRef] [PubMed]
- Şener, G.; Şehirli, A.Ö.; Ayanoǧlu-Dülger, G. Melatonin protects against mercury(II)-induced oxidative tissue damage in rats. Pharmacol. Toxicol. 2003, 93, 290–296. [Google Scholar] [CrossRef] [PubMed]
- Purohit, A.R.; Rao, M.V. Mitigative role of melatonin and α-tocopherol against mercury-induced genotoxicity. Drug Chem. Toxicol. 2014, 37, 221–226. [Google Scholar] [CrossRef] [PubMed]
- Cebulska-Wasilewska, A.; Panek, A.; Zabiński, Z.; Moszczyński, P.; Au, W.W. Occupational exposure to mercury vapour on genotoxicity and DNA repair. Mutat. Res. 2005, 586, 102–114. [Google Scholar] [CrossRef] [PubMed]
- Rao, M.V.; Sharma, P.S.N. Protective effect of vitamin E against mercuric chloride reproductive toxicity in male mice. Reprod. Toxicol. 2001, 15, 705–712. [Google Scholar] [CrossRef]
- Kim, C.Y.; Nakai, K.; Kameo, S.; Kurokawa, N.; Liu, Z.M.; Satoh, H. Protective effect of melatonin on methylmercury-induced mortality in mice. Tohoku J. Exp. Med. 2000, 191, 241–246. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Rossman, T.G. Mechanism of arsenic carcinogenesis: An integrated approach. Mutat. Res. 2003, 533, 37–65. [Google Scholar] [CrossRef] [PubMed]
- Pant, H.H.; Rao, M.V. Evaluation of in vitro anti-genotoxic potential of melatonin against arsenic and fluoride in human blood cultures. Ecotoxicol. Environ. Saf. 2010, 73, 1333–1337. [Google Scholar] [CrossRef] [PubMed]
- Aydemir, S.; Akgun, S.G.; Beceren, A.; Yuksel, M.; Kumas, M.; Erdogan, N.; Sardas, S.; Omurtag, G.Z. Melatonin ameliorates oxidative DNA damage and protects against formaldehyde-induced oxidative stress in rats. Int. J. Clin. Exp. Med. 2017, 10, 6250–6261. [Google Scholar]
- Hare, D.J.; Adlard, P.A.; Doble, P.A.; Finkelstein, D.I. Metallobiology of 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine neurotoxicity. Metallomics 2013, 5, 91–109. [Google Scholar] [CrossRef] [PubMed]
- Nicotra, A.; Parvez, S.H. Apoptotic molecules and MPTP-induced cell death. Neurotoxicol. Teratol. 2002, 24, 599–605. [Google Scholar] [CrossRef]
- Przedborski, S.; Vila, M. MPTP: A review of its mechanisms of neurotoxicity. Clin. Neurosci. Res. 2001, 1, 407–418. [Google Scholar] [CrossRef]
- Cadenas, S.; Barja, G. Resveratrol, melatonin, vitamin E, and PBN protect against renal oxidative DNA damage induced by the kidney carcinogen KBrO3. Free Radic. Biol. Med. 1999, 26, 1531–1537. [Google Scholar] [CrossRef]
- Spencer, J.P.E.; Whiteman, M.; Jenner, A.; Halliwell, B. Nitrite-induced deamination and hypochlorite-induced oxidation of DNA in intact human respiratory tract epithelial cells. Free Radic. Biol. Med. 2000, 28, 1039–1050. [Google Scholar] [CrossRef]
- Zavodnik, I.B.; Lapshina, E.A.; Zavodnik, L.B.; Łabieniec, M.; Bryszewska, M.; Reiter, R.J. Hypochlorous acid-induced oxidative stress in Chinese hamster B14 cells: Viability, DNA and protein damage and the protective action of melatonin. Mutat. Res. 2004, 559, 39–48. [Google Scholar] [CrossRef] [PubMed]
- Bozner, P.; Grishko, V.; LeDoux, S.P.; Wilson, G.L.; Chyan, Y.C.; Pappolla, M.A. The amyloid 13 protein induces oxidative damage of mitochondrial DNA. J. Neuropathol. Exp. Neurol. 1997, 56, 1356–1362. [Google Scholar] [CrossRef] [PubMed]
- Rezapoor, S.; Shirazi, A.; Abbasi, S.; Bazzaz, J.; Izadi, P.; Rezaeejam, H.; Valizadeh, M.; Soleimani-Mohammadi, F.; Najafi, M. Modulation of radiation-induced base excision repair pathway gene expression by melatonin. J. Med. Phys. 2017, 42, 245–250. [Google Scholar] [PubMed]
- Karbownik, M.; Reiter, R.J.; Qi, W.; Garcia, J.J.; Tan, D.X.; Manchester, L.C.; Vijayalaxmi. Protective effects of melatonin against oxidation of guanine bases in DNA and decreased microsomal membrane fluidity in rat liver induced by whole body ionizing radiation. Mol. Cell. Biochem. 2000, 211, 137–144. [Google Scholar] [CrossRef] [PubMed]
- Sun, C.K.; Chen, C.H.; Chang, C.L.; Chiang, H.J.; Sung, P.H.; Chen, K.H.; Chen, Y.L.; Chen, S.Y.; Kao, G.S.; Chang, H.W.; et al. Melatonin treatment enhances therapeutic effects of exosomes against acute liver ischemia-reperfusion injury. Am. J. Transl. Res. 2017, 9, 1543–1560. [Google Scholar] [PubMed]
- Nagai, R.; Watanabe, K.; Wakatsuki, A.; Hamada, F.; Shinohara, K.; Hayashi, Y.; Imamura, R.; Fukaya, T. Melatonin preserves fetal growth in rats by protecting against ischemia/reperfusion-induced oxidative/nitrosative mitochondrial damage in the placenta. J. Pineal Res. 2008, 45, 271–276. [Google Scholar] [CrossRef] [PubMed]
- Cetin, N.; Suleyman, H.; Sener, E.; Demirci, E.; Gundogdu, C.; Akcay, F. The prevention of ischemia/reperfusion induced oxidative damage by venous blood in rabbit kidneys monitored with biochemical, histopatological and immunohistochemical analysis. J. Physiol. Pharmacol. 2014, 65, 383–392. [Google Scholar] [PubMed]
- Chen, H.H.; Chen, Y.T.; Yang, C.C.; Chen, K.H.; Sung, P.H.; Chiang, H.J.; Chen, C.H.; Chua, S.; Chung, S.Y.; Chen, Y.L.; et al. Melatonin pretreatment enhances the therapeutic effects of exogenous mitochondria against hepatic ischemia–reperfusion injury in rats through suppression of mitochondrial permeability transition. J. Pineal Res. 2016, 61, 52–68. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, T.; Keep, R.F.; Hua, Y.; Hoff, J.T.; Xi, G. Oxidative DNA injury after experimental intracerebral hemorrhage. Brain Res. 2005, 1039, 30–36. [Google Scholar] [CrossRef] [PubMed]
- Young, I.M.; Leone, R.M.; Francis, P.; Stovell, P.; Silman, R.E. Melatonin is metabolized to N-acetyl serotonin and 6-hydroxymelatonin in man. J. Clin. Endocrinol. Metab. 1985, 60, 114–119. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.; Wang, J.; Jiang, J.; Stavrovskaya, I.G.; Li, M.; Li, W.; Wu, Q.; Zhang, X.; Luo, C.; Zhou, S.; et al. N-Acetyl-serotonin offers neuroprotection through inhibiting mitochondrial death pathways and autophagic activation in experimental models of ischemic injury. J. Neurosci. 2014, 34, 2967–2978. [Google Scholar] [CrossRef] [PubMed]
- Bachurin, S.; Oxenkrug, G.F.; Lermontova, N.; Afanasiev, A.; Beznosko, B.; Vankin, G.; Shevtzova, E.; Mukhina, T.; Serkova, T. N-Acetylserotonin, melatonin and their derivatives improve cognition and protect against β-amyloid-induced neurotoxicity. Ann. N. Y. Acad. Sci. 1999, 890, 155–166. [Google Scholar] [CrossRef] [PubMed]
- Oxenkrug, G.; Requintina, P.; Bachurin, S. Antioxidant and antiaging activity of N-acetylserotonin and melatonin in the in vivo models. Ann. N. Y. Acad. Sci. 2001, 939, 190–199. [Google Scholar] [CrossRef] [PubMed]
- Qi, W.; Reiter, R.J.; Tan, D.X.; Manchester, L.C.; Siu, A.W.; Garcia, J.J. Increased levels of oxidatively damaged DNA induced by chromium(III) and H2O2: Protection by melatonin and related molecules. J. Pineal Res. 2000, 29, 54–61. [Google Scholar] [CrossRef] [PubMed]
- Jiang, J.; Yu, S.; Jiang, Z.; Liang, C.; Yu, W.; Li, J.; Du, X.; Wang, H.; Gao, X.; Wang, X. N-Acetyl-serotonin protects HepG2 cells from oxidative stress injury induced by hydrogen peroxide. Oxid. Med. Cell. Longev. 2014, 2014, 310504. [Google Scholar] [CrossRef] [PubMed]
- Reiter, R.J.; Mayo, J.C.; Tan, D.X.; Sainz, R.M.; Alatorre-Jimenez, M.; Qin, L. Melatonin as an antioxidant: under promises but over delivers. J. Pineal Res. 2016, 61, 253–278. [Google Scholar] [CrossRef] [PubMed]
- Janjetovic, Z.; Jarrett, S.G.; Lee, E.F.; Duprey, C.; Reiter, R.J.; Slominski, A.T. Melatonin and its metabolites protect human melanocytes against UVB-induced damage: Involvement of NRF2-mediated pathways. Sci. Rep. 2017, 7, 1274. [Google Scholar] [CrossRef] [PubMed]
- Slominski, A.T.; Kleszczyński, K.; Semak, I.; Janjetovic, Z.; Żmijewski, M.A.; Kim, T.K.; Slominski, R.M.; Reiter, R.J.; Fischer, T.W. Local melatoninergic system as the protector of skin integrity. Int. J. Mol. Sci. 2014, 15, 17705–17732. [Google Scholar] [CrossRef] [PubMed]
- Ximenes, V.F.; Padovan, C.Z.; Carvalho, D.A.; Fernandes, J.R. Oxidation of melatonin by taurine chloramine. J. Pineal Res. 2010, 49, 115–122. [Google Scholar] [CrossRef] [PubMed]
- De Oliveira Silva, S.; Ximenes, V.F.; Catalani, L.H.; Campa, A. Myeloperoxidase-catalyzed oxidation of melatonin by activated neutrophils. Biochem. Biophys. Res. Commun. 2000, 279, 657–662. [Google Scholar] [CrossRef] [PubMed]
- Semak, I.; Korik, E.; Antonova, M.; Wortsman, J.; Slominski, A. Metabolism of melatonin by cytochrome P450s in rat liver mitochondria and microsomes. J. Pineal Res. 2008, 45, 515–523. [Google Scholar] [CrossRef] [PubMed]
- Horstman, J.A.; Wrona, M.Z.; Dryhurst, G. Further insights into the reaction of melatonin with hydroxyl radical. Bioorg. Chem. 2002, 30, 371–382. [Google Scholar] [CrossRef]
- Hardeland, R.; Poeggeler, B.; Niebergall, R.; Zelosko, V. Oxidation of melatonin by carbonate radicals and chemiluminescence emitted during pyrrole ring cleavage. J. Pineal Res. 2003, 34, 17–25. [Google Scholar] [CrossRef] [PubMed]
- De Almeida, E.A.; Martinez, G.R.; Klitzke, C.F.; De Medeiros, M.H.G.; Di Mascio, P. Oxidation of melatonin by singlet molecular oxygen (O2(1Δg)) produces N1-acetyl-N2-formyl-5-methoxykynurenine. J. Pineal Res. 2003, 35, 131–137. [Google Scholar] [CrossRef] [PubMed]
- Fischer, T.W.; Sweatman, T.W.; Semak, I.; Sayre, R.M.; Wortsman, J.; Slominski, A. Constitutive and UV-induced metabolism of melatonin in keratinocytes and cell-free systems. FASEB J. 2006, 20, E897–E907. [Google Scholar] [CrossRef] [PubMed]
- Bonnefont-Rousselot, D.; Collin, F.; Jore, D.; Gardès-Albert, M. Reaction mechanism of melatonin oxidation by reactive oxygen species in vitro. J. Pineal Res. 2011, 50, 328–335. [Google Scholar] [CrossRef] [PubMed]
- Tan, D.X.; Manchester, L.C.; Burkhardt, S.; Sainz, R.M.; Mayo, J.C.; Kohen, R.; Shohami, E.; Huo, Y.S.; Hardeland, R.; Reiter, R.J. N1-acetyl-N2-formyl-5-methoxykynuramine, a biogenic amine and melatonin metabolite, functions as a potent antioxidant. FASEB J. 2001, 15, 2294–2296. [Google Scholar] [CrossRef] [PubMed]
- Burkhardt, S.; Reiter, R.J.; Tan, D.X.; Hardeland, R.; Cabrera, J.; Karbownik, M. DNA oxidatively damaged by chromium(III) and H2O2 is protected by the antioxidants melatonin, N1-acetyl-N2-formyl-5-methoxykynuramine, resveratrol and uric acid. Int. J. Biochem. Cell Biol. 2001, 33, 775–783. [Google Scholar] [CrossRef]
- Manda, K.; Ueno, M.; Anzai, K. AFMK, a melatonin metabolite, attenuates X-ray-induced oxidative damage to DNA, proteins and lipids in mice. J. Pineal Res. 2007, 42, 386–393. [Google Scholar] [CrossRef] [PubMed]
- Galano, A.; Tan, D.X.; Reiter, R.J. On the free radical scavenging activities of melatonin’s metabolites, AFMK and AMK. J. Pineal Res. 2013, 54, 245–257. [Google Scholar] [CrossRef] [PubMed]
- Manda, K.; Ueno, M.; Anzai, K. Space radiation-induced inhibition of neurogenesis in the hippocampal dentate gyrus and memory impairment in mice: Ameliorative potential of the melatonin metabolite, AFMK. J. Pineal Res. 2008, 45, 430–438. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Chen, Z.; Chua, C.C.; Ma, Y.S.; Youngberg, G.A.; Hamdy, R.; Chua, B.H.L. Melatonin as an effective protector against doxorubicin-induced cardiotoxicity. Am. J. Physiol. Heart Circ. Physiol. 2002, 283, H254–H263. [Google Scholar] [CrossRef] [PubMed]
- Hardeland, R.; Tan, D.X.; Reiter, R.J. Kynuramines, metabolites of melatonin and other indoles: The resurrection of an almost forgotten class of biogenic amines. J. Pineal Res. 2009, 47, 109–126. [Google Scholar] [CrossRef] [PubMed]
- Schaefer, M.; Hardeland, R. The melatonin metabolite N1-acetyl-5-methoxykynuramine is a potent singlet oxygen scavenger. J. Pineal Res. 2009, 46, 49–52. [Google Scholar] [CrossRef] [PubMed]
- Maharaj, D.S.; Anoopkumar-Dukie, S.; Glass, B.D.; Antunes, E.M.; Lack, B.; Walker, R.B.; Daya, S. The identification of the UV degradants of melatonin and their ability to scavenge free radicals. J. Pineal Res. 2002, 32, 257–261. [Google Scholar] [CrossRef] [PubMed]
- León, J.; Escames, G.; Rodríguez, M.I.; López, L.C.; Tapias, V.; Entrena, A.; Camacho, E.; Carrión, M.D.; Gallo, M.A.; Espinosa, A.; et al. Inhibition of neuronal nitric oxide synthase activity by N1-acetyl-5-methoxykynuramine, a brain metabolite of melatonin. J. Neurochem. 2006, 98, 2023–2033. [Google Scholar] [CrossRef] [PubMed]
- Hardeland, R.; Backhaus, C.; Fadavi, A.; Hess, M. N1-acetyl-5-methoxykynuramine contrasts with other tryptophan metabolites by a peculiar type of NO scavenging: Cyclization to a cinnolinone prevents formation of unstable nitrosamines. J. Pineal Res. 2007, 43, 104–105. [Google Scholar] [CrossRef] [PubMed]
- Tapias, V.; Escames, G.; López, L.C.; López, A.; Camacho, E.; Carrión, M.D.; Entrena, A.; Gallo, M.A.; Espinosa, A.; Acuña-Castroviejo, D. Melatonin and its brain metabolite N1-acetyl-5-methoxykynuramine prevent mitochondrial nitric oxide synthase induction in Parkinsonian mice. J. Neurosci. Res. 2009, 87, 3002–3010. [Google Scholar] [CrossRef] [PubMed]
- Tan, D.X.; Manchester, L.C.; Reiter, R.J.; Plummer, B.F.; Hardies, L.J.; Weintraub, S.T.; Vijayalaxmi; Shepherd, A.M.M. A novel melatonin metabolite, cyclic 3-hydroxymelatonin: A biomarker of in vivo hydroxyl radical generation. Biochem. Biophys. Res. Commun. 1998, 253, 614–620. [Google Scholar] [CrossRef] [PubMed]
- Tan, D.X.; Hardeland, R.; Manchester, L.C.; Poeggeler, B.; Lopez-Burillo, S.; Mayo, J.C.; Sainz, R.M.; Reiter, R.J. Mechanistic and comparative studies of melatonin and classic antioxidants in terms of their interactions with the ABTS cation radical. J. Pineal Res. 2003, 34, 249–259. [Google Scholar] [CrossRef] [PubMed]
- López-Burillo, S.; Tan, D.X.; Rodriguez-Gallego, V.; Manchester, L.C.; Mayo, J.C.; Sainz, R.M.; Reiter, R.J. Melatonin and its derivatives cyclic 3-hydroxymelatonin, N1-acetyl-N2-formyl-5-methoxykynuramine and 6-methoxymelatonin reduce oxidative DNA damage induced by Fenton reagents. J. Pineal Res. 2003, 34, 178–184. [Google Scholar] [CrossRef]
- Kim, T.K.; Lin, Z.; Tidwell, W.J.; Li, W.; Slominski, A.T. Melatonin and its metabolites accumulate in the human epidermis in vivo and inhibit proliferation and tyrosinase activity in epidermal melanocytes in vitro. Mol. Cell. Endocrinol. 2015, 404, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Maharaj, D.S.; Maharaj, H.; Antunes, E.M.; Maree, D.M.; Nyokong, T.; Glass, B.D.; Daya, S. 6-Hydroxymelatonin protects against quinolinic-acid-induced oxidative neurotoxicity in the rat hippocampus. J. Pharm. Pharmacol. 2005, 57, 877–881. [Google Scholar] [CrossRef] [PubMed]
- Pierrefiche, G.; Topall, G.; Courboin, G.; Henriet, I.; Laborit, H. Antioxidant activity of melatonin in mice. Res. Commun. Chem. Pathol. Pharmacol. 1993, 80, 211–224. [Google Scholar] [PubMed]
- Maharaj, D.S.; Walker, R.B.; Glass, B.D.; Daya, S. 6-Hydroxymelatonin protects against cyanide induced oxidative stress in rat brain homogenates. J. Chem. Neuroanat. 2003, 26, 103–107. [Google Scholar] [CrossRef]
- Janjetovic, Z.; Nahmias, Z.P.; Hanna, S.; Jarrett, S.G.; Kim, T.K.; Reiter, R.J.; Slominski, A.T. Melatonin and its metabolites ameliorate ultraviolet B-induced damage in human epidermal keratinocytes. J. Pineal Res. 2014, 57, 90–102. [Google Scholar] [CrossRef] [PubMed]
- Dellegar, S.M.; Murphy, S.A.; Bourne, A.E.; Dicesare, J.C.; Purser, G.H. Identification of the factors affecting the rate of deactivation of hypochlorous acid by melatonin. Biochem. Biophys. Res. Commun. 1999, 257, 431–439. [Google Scholar] [CrossRef] [PubMed]
- Agozzino, P.; Avellone, G.; Bongiorno, D.; Ceraulo, L.; Filizzola, F.; Natoli, M.C.; Livrea, M.A.; Tesoriere, L. Melatonin: Structural characterization of its non-enzymatic mono-oxygenate metabolite. J. Pineal Res. 2003, 35, 269–275. [Google Scholar] [CrossRef] [PubMed]
- Abbaszadeh, A.; Haddadi, G.H.; Haddadi, Z. Melatonin role in ameliorating radiation-induced skin damage: From theory to practice (A review of literature). J. Biomed. Phys. Eng. 2017, 7, 127–136. [Google Scholar] [PubMed]
- Mayo, J.C.; Sainz, R.M.; Antolín, I.; Rodriguez, C. Ultrastructural confirmation of neuronal protection by melatonin against the neurotoxin 6-hydroxydopamine cell damage. Brain Res. 1999, 818, 221–227. [Google Scholar] [CrossRef]
- Wu, Z.H.; Zhang, H.; Wang, X.Y.; Yang, R.; Liu, B.; Liu, Y.; Zhao, W.P.; Feng, H.Y.; Xue, L.G.; Hao, J.F.; Niu, B.T.; Wang, Z.H. Protective effects of melatonin against12C6+beam irradiation-induced oxidative stress and DNA injury in the mouse brain. Adv. Space Res. 2012, 49, 196–203. [Google Scholar] [CrossRef]
- Laothong, U.; Pinlaor, P.; Hiraku, Y.; Boonsiri, P.; Prakobwong, S.; Khoontawad, J.; Pinlaor, S. Protective effect of melatonin against Opisthorchis viverrini-induced oxidative and nitrosative DNA damage and liver injury in hamsters. J. Pineal Res. 2010, 49, 271–282. [Google Scholar] [CrossRef] [PubMed]
- Bekyarova, G.; Tzaneva, M.; Hristova, M. Melatonin protects against burn-induced hepatic oxidative injury by inducing HO-1 via the Nrf2 pathway. Vet. Med. (Praha) 2015, 60, 621–628. [Google Scholar] [CrossRef]
- Deng, Y.; Zhu, J.; Mi, C.; Xu, B.; Jiao, C.; Li, Y.; Xu, D.; Liu, W.; Xu, Z. Melatonin antagonizes Mn-induced oxidative injury through the activation of keap1-Nrf2-ARE signaling pathway in the striatum of mice. Neurotox. Res. 2014, 27, 156–171. [Google Scholar] [CrossRef] [PubMed]
- Ding, K.; Wang, H.; Xu, J.; Li, T.; Zhang, L.; Ding, Y.; Zhu, L.; He, J.; Zhou, M. Melatonin stimulates antioxidant enzymes and reduces oxidative stress in experimental traumatic brain injury: The Nrf2-ARE signaling pathway as a potential mechanism. Free Radic. Biol. Med. 2014, 73, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Sun, J.; Li, T.; Zhang, Q.; Bu, S.; Wang, Q.; Lai, D. Melatonin ameliorates restraint stress-induced oxidative stress and apoptosis in testicular cells via NF-κB/iNOS and Nrf2/ HO-1 signaling pathway. Sci. Rep. 2017, 7, 9599. [Google Scholar] [CrossRef] [PubMed]
- Jumnongprakhon, P.; Govitrapong, P.; Tocharus, C.; Pinkaew, D.; Tocharus, J. Melatonin protects methamphetamine-induced neuroinflammation through NF-κB and Nrf2 pathways in glioma cell line. Neurochem. Res. 2015, 40, 1448–1456. [Google Scholar] [CrossRef] [PubMed]
- Jumnongprakhon, P.; Govitrapong, P.; Tocharus, C.; Tocharus, J. Melatonin promotes blood-brain barrier integrity in methamphetamine-induced inflammation in primary rat brain microvascular endothelial cells. Brain Res. 2016, 1646, 182–192. [Google Scholar] [CrossRef] [PubMed]
- Jumnongprakhon, P.; Govitrapong, P.; Tocharus, C.; Tocharus, J. Melatonin improves methamphetamine-induced blood brain barrier impairment through NADPH oxidase-2 in primary rat brain microvascular endothelium cells. Brain Res. 2016, 1646, 393–401. [Google Scholar] [CrossRef] [PubMed]
- Kleszczyński, K.; Zillikens, D.; Fischer, T.W. Melatonin enhances mitochondrial ATP synthesis, reduces reactive oxygen species formation, and mediates translocation of the nuclear erythroid 2-related factor 2 resulting in activation of phase-2 antioxidant enzymes (γ-GCS, HO-1, NQO1) in ultraviolet radiation-treated normal human epidermal keratinocytes (NHEK). J. Pineal Res. 2016, 61, 187–197. [Google Scholar]
- Chen, L.Y.; Renn, T.Y.; Liao, W.C.; Mai, F.D.; Ho, Y.J.; Hsiao, G.; Lee, A.W.; Chang, H.M. Melatonin successfully rescues hippocampal bioenergetics and improves cognitive function following drug intoxication by promoting Nrf2- ARE signaling activity. J. Pineal Res. 2017, 63, e12417. [Google Scholar] [CrossRef] [PubMed]
- Negi, G.; Kumar, A.; Sharma, S.S. Melatonin modulates neuroinflammation and oxidative stress in experimental diabetic neuropathy: Effects on NF-κB and Nrf2 cascades. J. Pineal Res. 2011, 50, 124–131. [Google Scholar] [CrossRef] [PubMed]
- Parada, E.; Buendia, I.; León, R.; Negredo, P.; Romero, A.; Cuadrado, A.; López, M.G.; Egea, J. Neuroprotective effect of melatonin against ischemia is partially mediated by alpha-7 nicotinic receptor modulation and HO-1 overexpression. J. Pineal Res. 2014, 56, 204–212. [Google Scholar] [CrossRef] [PubMed]
- Santofimia-Castaño, P.; Clea Ruy, D.; Garcia-Sanchez, L.; Jimenez-Blasco, D.; Fernandez-Bermejo, M.; Bolaños, J.P.; Salido, G.M.; Gonzalez, A. Melatonin induces the expression of Nrf2-regulated antioxidant enzymes via PKC and Ca2+ influx activation in mouse pancreatic acinar cells. Free Radic. Biol. Med. 2015, 87, 226–236. [Google Scholar] [CrossRef] [PubMed]
- Shang, B.; Shi, H.; Wang, X.; Guo, X.; Wang, N.; Wang, Y.; Dong, L. Protective effect of melatonin on myenteric neuron damage in experimental colitis in rats. Fundam. Clin. Pharmacol. 2016, 30, 117–127. [Google Scholar] [CrossRef] [PubMed]
- Tao, R.R.; Huang, J.Y.; Shao, X.J.; Ye, W.F.; Tian, Y.; Liao, M.H.; Fukunaga, K.; Lou, Y.J.; Han, F.; Lu, Y.M. Ischemic injury promotes Keap1 nitration and disturbance of antioxidative responses in endothelial cells: A potential vasoprotective effect of melatonin. J. Pineal Res. 2013, 54, 271–281. [Google Scholar] [CrossRef] [PubMed]
- Tripathi, D.N.; Jena, G.B. Effect of melatonin on the expression of Nrf2 and NF-κB during cyclophosphamide-induced urinary bladder injury in rat. J. Pineal Res. 2010, 48, 324–331. [Google Scholar] [CrossRef] [PubMed]
- Trivedi, P.P.; Jena, G.B.; Tikoo, K.B.; Kumar, V. Melatonin modulated autophagy and Nrf2 signaling pathways in mice with colitis-associated colon carcinogenesis. Mol. Carcinog. 2016, 55, 255–267. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Ma, C.; Meng, C.J.; Zhu, G.Q.; Sun, X.B.; Huo, L.; Zhang, J.; Liu, H.X.; He, W.C.; Shen, X.M.; et al. Melatonin activates the Nrf2-ARE pathway when it protects against early brain injury in a subarachnoid hemorrhage model. J. Pineal Res. 2012, 53, 129–137. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Ni, L.; Wang, J.; Lu, C.; Ren, M.; Han, W.; Liu, C. The protective effect of melatonin on smoke-induced vascular injury in rats and humans: A randomized controlled trial. J. Pineal Res. 2016, 60, 217–227. [Google Scholar] [CrossRef] [PubMed]
- Yu, G.M.; Kubota, H.; Okita, M.; Maeda, T. The anti-inflammatory and antioxidant effects of melatonin on LPS-stimulated bovine mammary epithelial cells. PLoS ONE 2017, 12, e0178525. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Guo, S.; Wang, J.; Shen, Y.; Zhang, J.; Wu, Q. Nrf2/HO-1 mediates the neuroprotective effect of mangiferin on early brain injury after subarachnoid hemorrhage by attenuating mitochondria-related apoptosis and neuroinflammation. Sci. Rep. 2017, 7, 11883. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.C.; Lu, K.C.; Lin, G.J.; Hsieh, H.Y.; Chu, P.; Lin, S.H.; Sytwu, H.K. Melatonin enhances endogenous heme oxygenase-1 and represses immune responses to ameliorate experimental murine membranous nephropathy. J. Pineal Res. 2012, 52, 460–469. [Google Scholar] [CrossRef] [PubMed]
- Ortiz-Franco, M.; Planells, E.; Quintero, B.; Acuña-Castroviejo, D.; Rusanova, I.; Escames, G.; Molina-López, J. Effect of melatonin supplementation on antioxidant status and DNA damage in high intensity trained athletes. Int. J. Sports Med. 2017, 38, 1117–1125. [Google Scholar] [CrossRef] [PubMed]
- Reiter, R.J.; Tan, D.X.; Manchester, L.C.; Pilar Terron, M.; Flores, L.J.; Koppisepi, S. Medical implications of melatonin: receptor-mediated and receptor-independent actions. Adv. Med. Sci. 2007, 52, 11–28. [Google Scholar] [PubMed]
- Tomás-Zapico, C.; Coto-Montes, A. A proposed mechanism to explain the stimulatory effect of melatonin on antioxidative enzymes. J. Pineal Res. 2005, 39, 99–104. [Google Scholar] [CrossRef] [PubMed]
- Barlow-Walden, L.R.; Reiter, R.J.; Abe, M.; Pablos, M.; Menendez-Pelaez, A.; Chen, L.D.; Poeggeler, B. Melatonin stimulates brain glutathione peroxidase activity. Neurochem. Int. 1995, 26, 497–502. [Google Scholar] [CrossRef]
- Pablos, M.I.; Agapito, M.T.; Gutierrez, R.; Recio, J.M.; Reiter, R.J.; Barlow-Walden, L.; Acuña-Castroviejo, D.; Menendez-Pelaez, A. Melatonin stimulates the activity of the detoxifying enzyme glutathione peroxidase in several tissues of chicks. J. Pineal Res. 1995, 19, 111–115. [Google Scholar] [CrossRef] [PubMed]
- Fischer, T.W.; Slominski, A.; Zmijewski, M.A.; Reiter, R.J.; Paus, R. Melatonin as a major skin protectant: From free radical scavenging to DNA damage repair. Exp. Dermatol. 2008, 17, 713–730. [Google Scholar] [CrossRef] [PubMed]
- Djordjevic, B.; Sokolovic, D.; Kocic, G.; Veljkovic, A.; Despotovic, M.; Basic, J.; Jevtovic-Stoimenov, T.; Sokolovic, D.M. The effect of melatonin on the liver of rats exposed to microwave radiation. Bratisl. Lek. Listy 2015, 116, 96–100. [Google Scholar] [CrossRef] [PubMed]
- Koyu, A.; Naziroǧlu, M.; Ozguner, F.; Yilmaz, H.R.; Uz, E.; Cesur, G. Caffeic acid phenethyl ester modulates 1800 MHz microwave-induced oxidative stress in rat liver. Electromagn. Biol. Med. 2005, 24, 135–142. [Google Scholar] [CrossRef]
- Meena, R.; Kumari, K.; Kumar, J.; Rajamani, P.; Verma, H.N.; Kesari, K.K. Therapeutic approaches of melatonin in microwave radiations-induced oxidative stress-mediated toxicity on male fertility pattern of wistar rats. Electromagn. Biol. Med. 2014, 33, 81–91. [Google Scholar] [CrossRef] [PubMed]
- Sokolovic, D.; Djordjevic, B.; Kocic, G.; Veljkovic, A.; Marinkovic, M.; Basic, J.; Jevtovic-Stoimenov, T.; Stanojkovic, Z.; Sokolovic, D.M.; Pavlovic, V.; et al. Melatonin protects rat thymus against oxidative stress caused by exposure to microwaves and modulates proliferation/apoptosis of thymocytes. Gen. Physiol. Biophys. 2013, 32, 79–90. [Google Scholar] [CrossRef] [PubMed]
- De Souza-Pinto, N.C.; Eide, L.; Hogue, B.A.; Thybo, T.; Stevnsner, T.; Seeberg, E.; Klungland, A.; Bohr, V.A. Repair of 8-oxodeoxyguanosine lesions in mitochondrial DNA depends on the oxoguanine DNA glycosylase (OGG1) gene and 8-oxoguanine accumulates in the mitochondrial DNA of OGG1-defective mice. Cancer Res. 2001, 61, 5378–5381. [Google Scholar] [PubMed]
- Mol, C.D.; Hosfield, D.J.; Tainer, J.A. Abasic site recognition by two apurinic/apyrimidinic endonuclease families in DNA base excision repair: The 3′ ends justify the means. Mutat. Res. 2000, 460, 211–229. [Google Scholar] [CrossRef]
- Brem, R.; Hall, J. XRCC1 is required for DNA single-strand break repair in human cells. Nucleic Acids Res. 2005, 33, 2512–2520. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, S.G.; Peliciari-Garcia, R.A.; Takahashi-Hyodo, S.A.; Rodrigues, A.C.; Amaral, F.G.; Berra, C.M.; Bordin, S.; Curi, R.; Cipolla-Neto, J. Effects of melatonin on DNA damage induced by cyclophosphamide in rats. Braz. J. Med. Biol. Res. 2013, 46, 278–286. [Google Scholar] [CrossRef] [PubMed]
- Valizadeh, M.; Shirazi, A.; Izadi, P.; Tavakkoly Bazzaz, J.; Rezaeejam, H. Expression levels of two dna repair-related genes under 8 gy ionizing radiation and 100 mg/kg melatonin delivery in rat peripheral blood. J. Biomed. Phys. Eng. 2017, 7, 27–36. [Google Scholar] [PubMed]
- Santoro, R.; Marani, M.; Blandino, G.; Muti, P.; Strano, S. Melatonin triggers p53 Ser phosphorylation and prevents DNA damage accumulation. Oncogene 2012, 31, 2931–2942. [Google Scholar] [CrossRef] [PubMed]
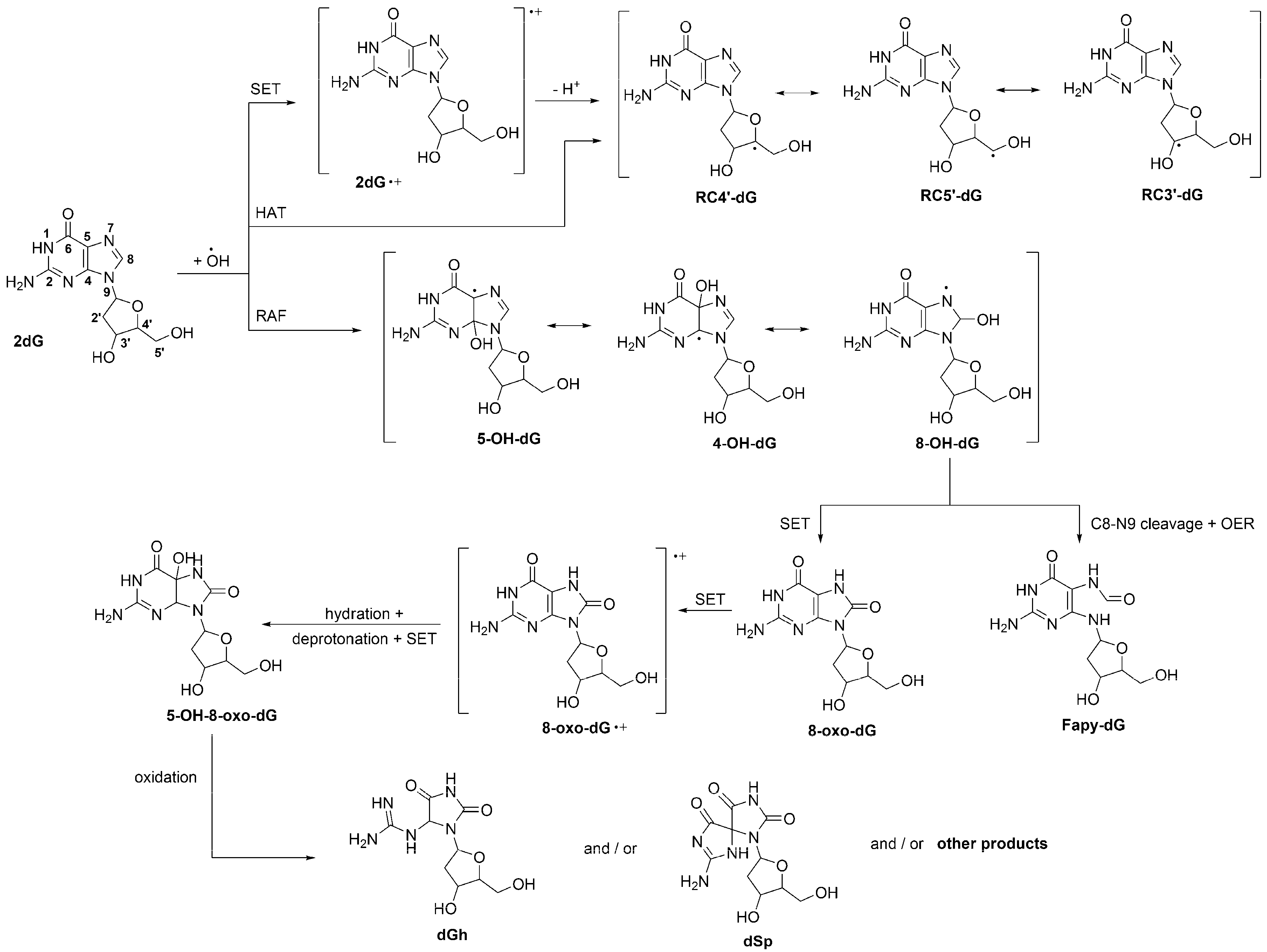
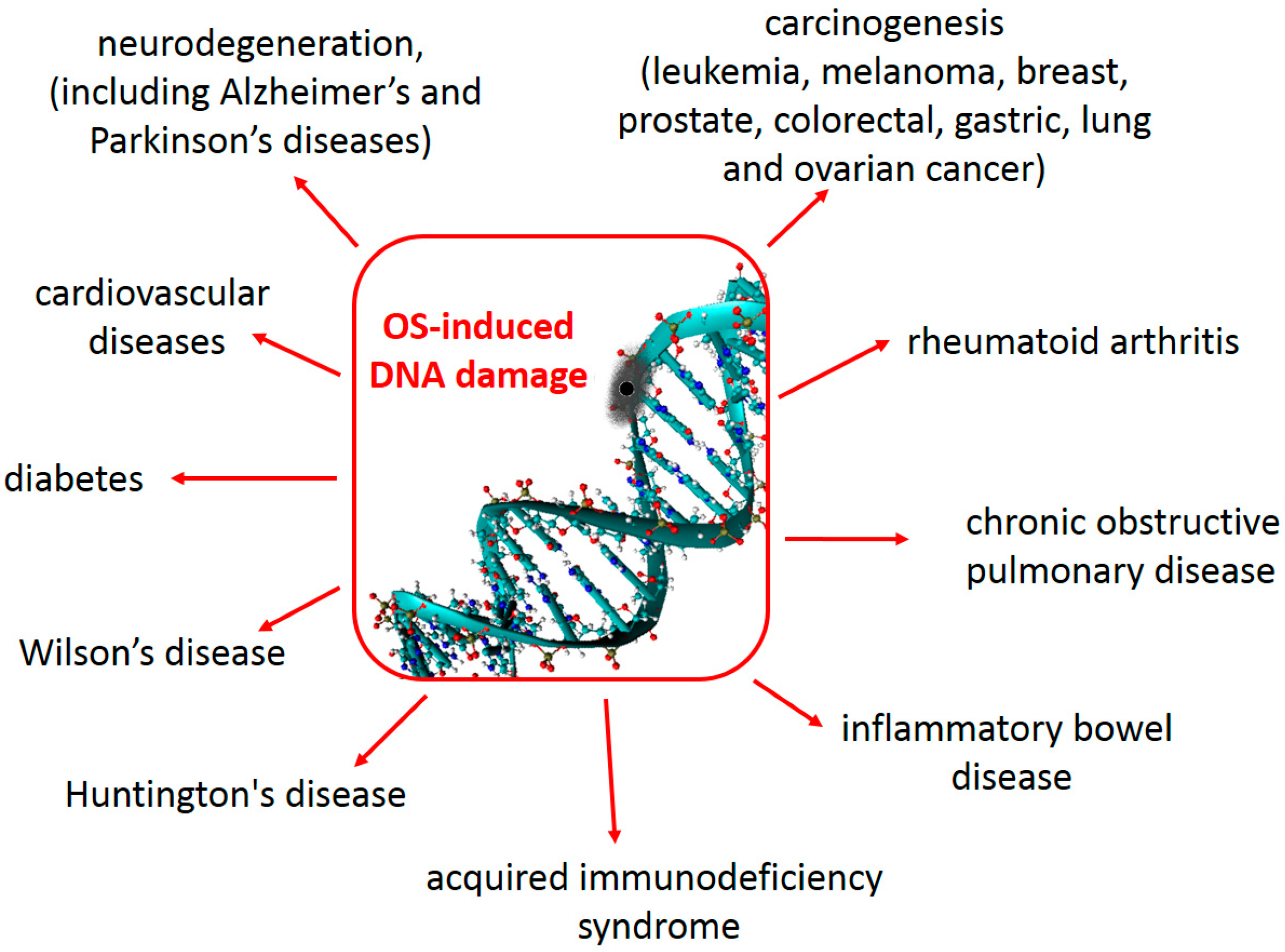
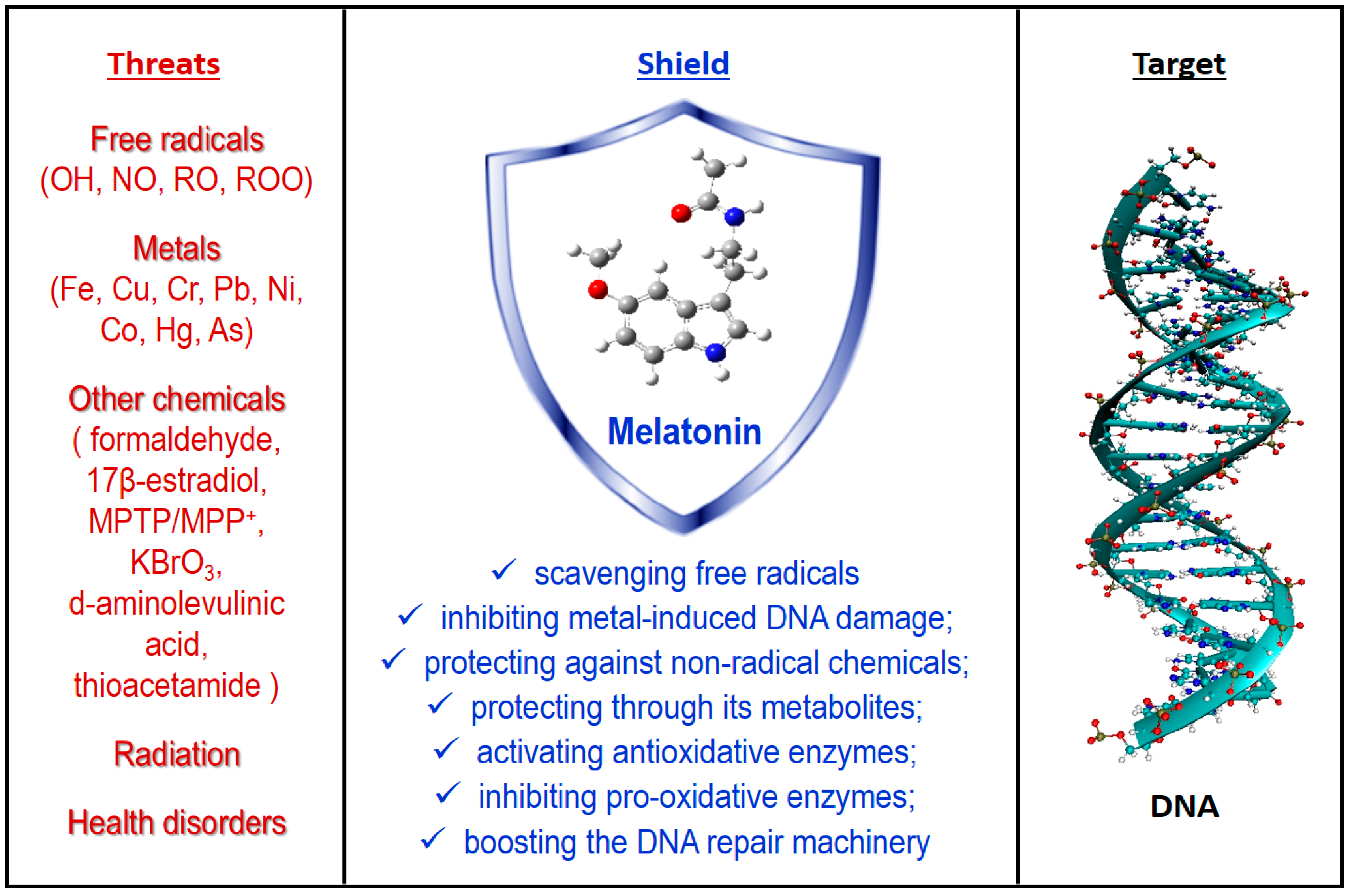
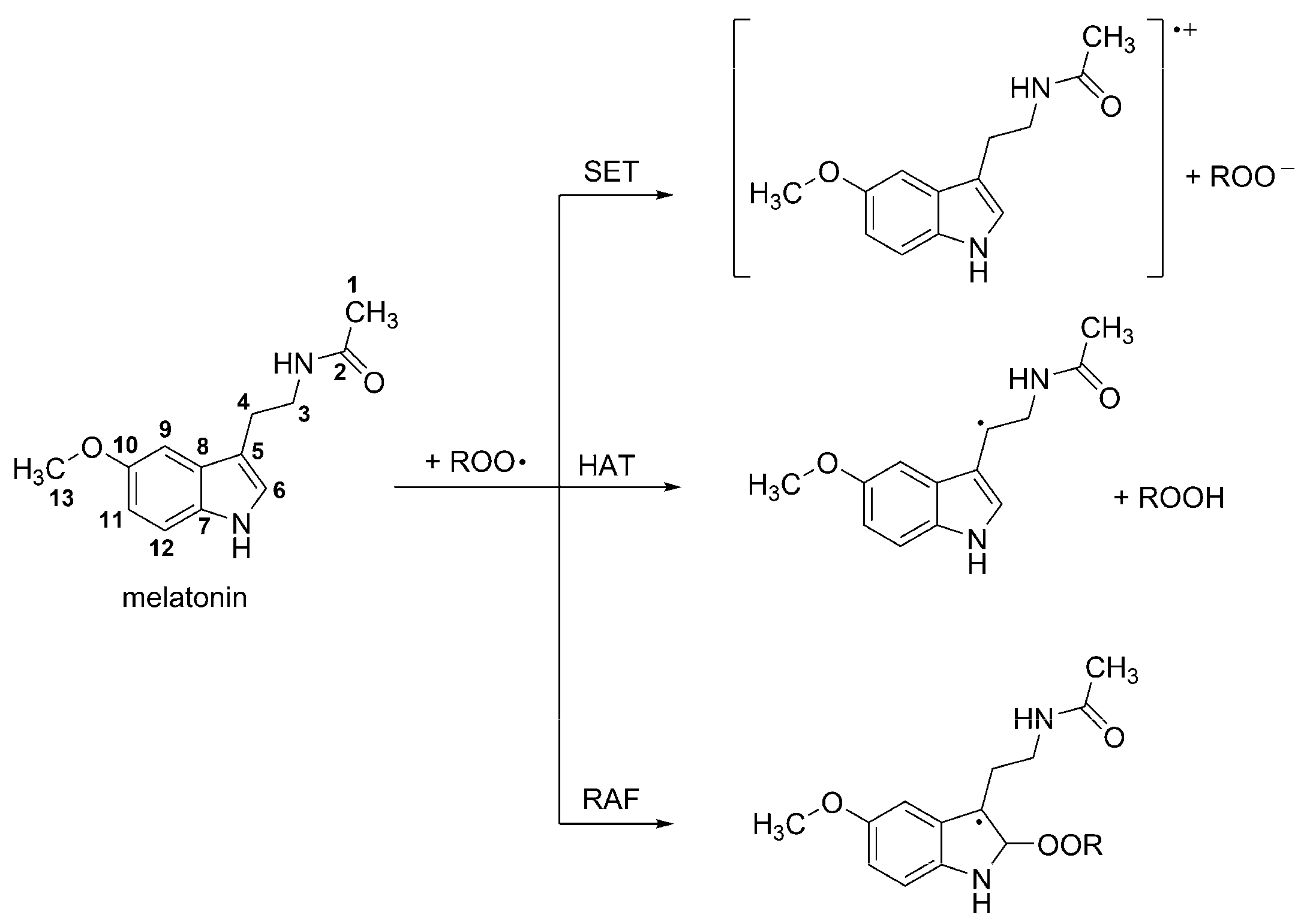
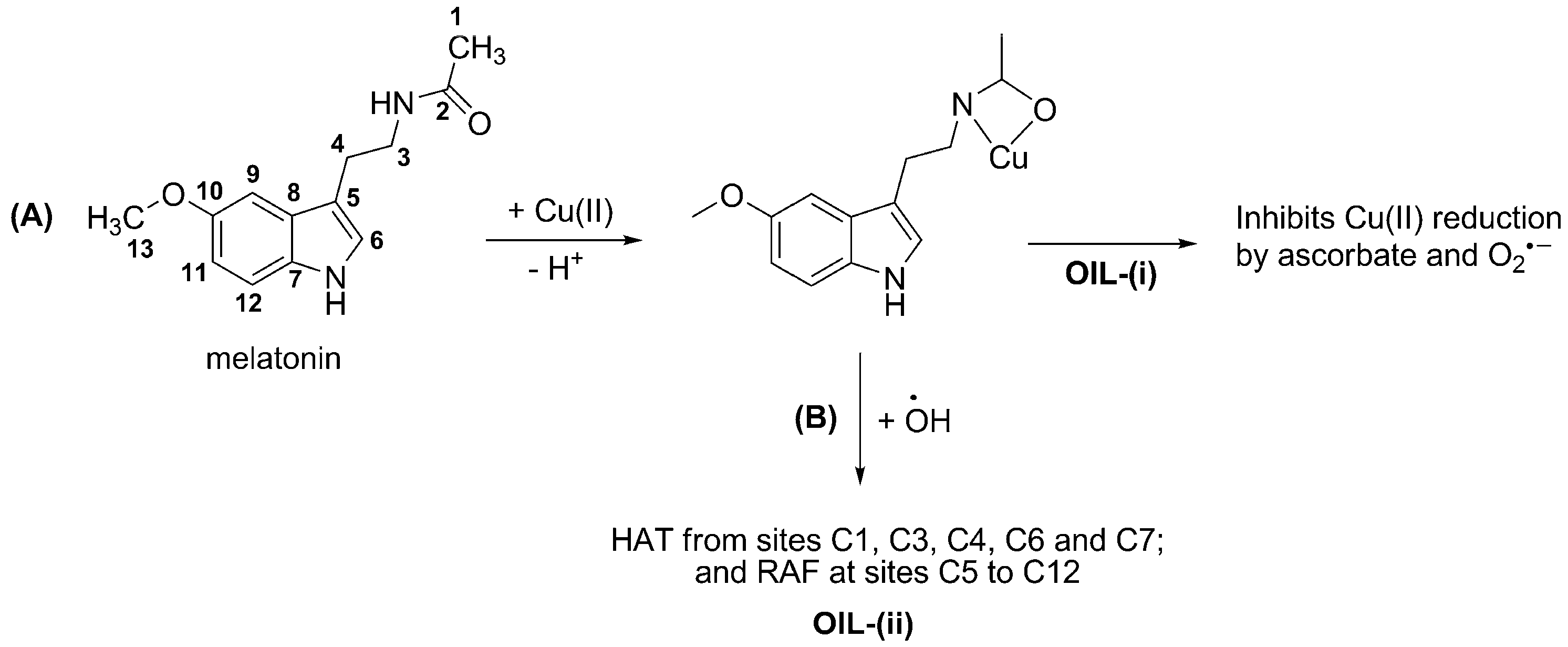
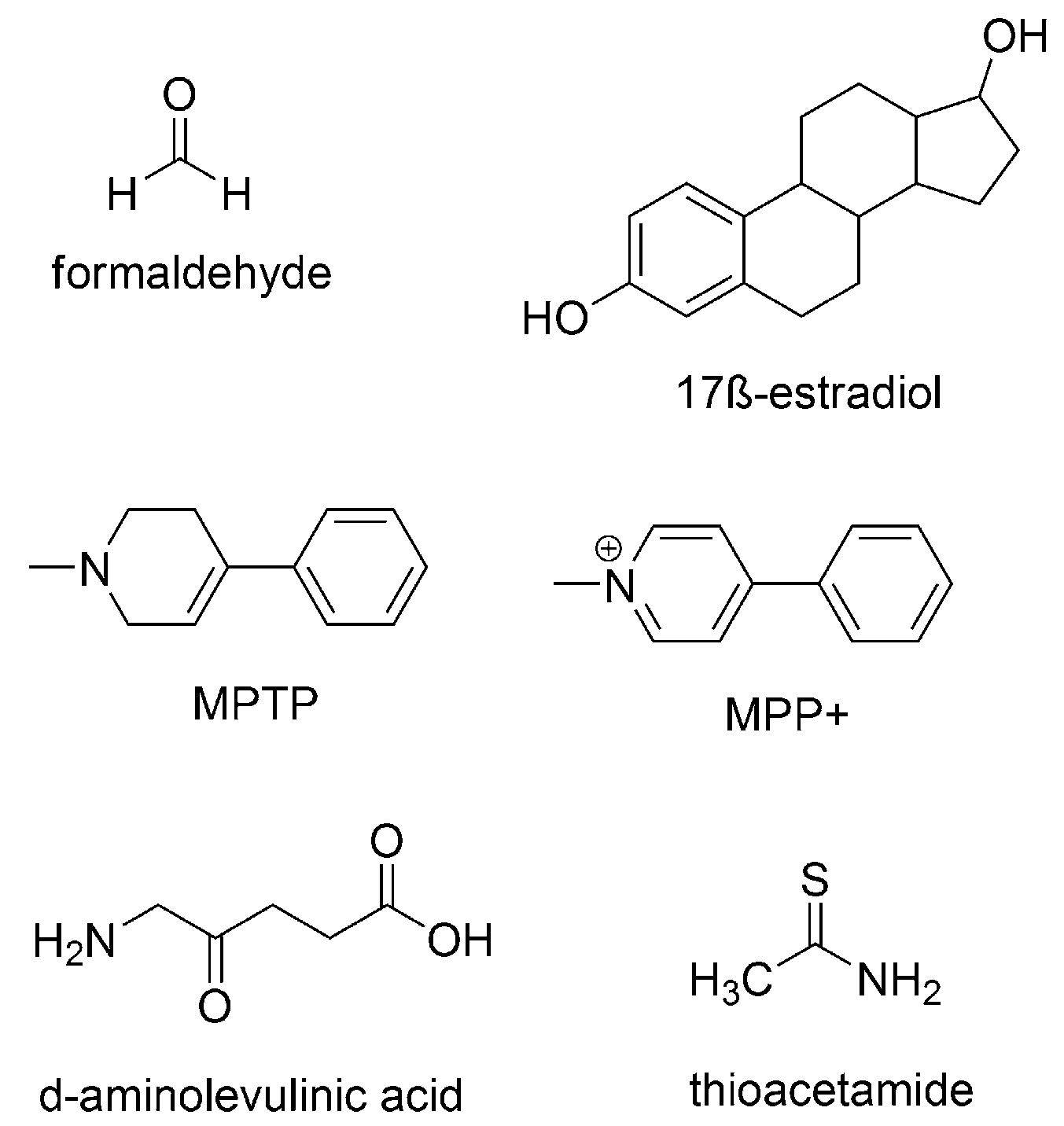
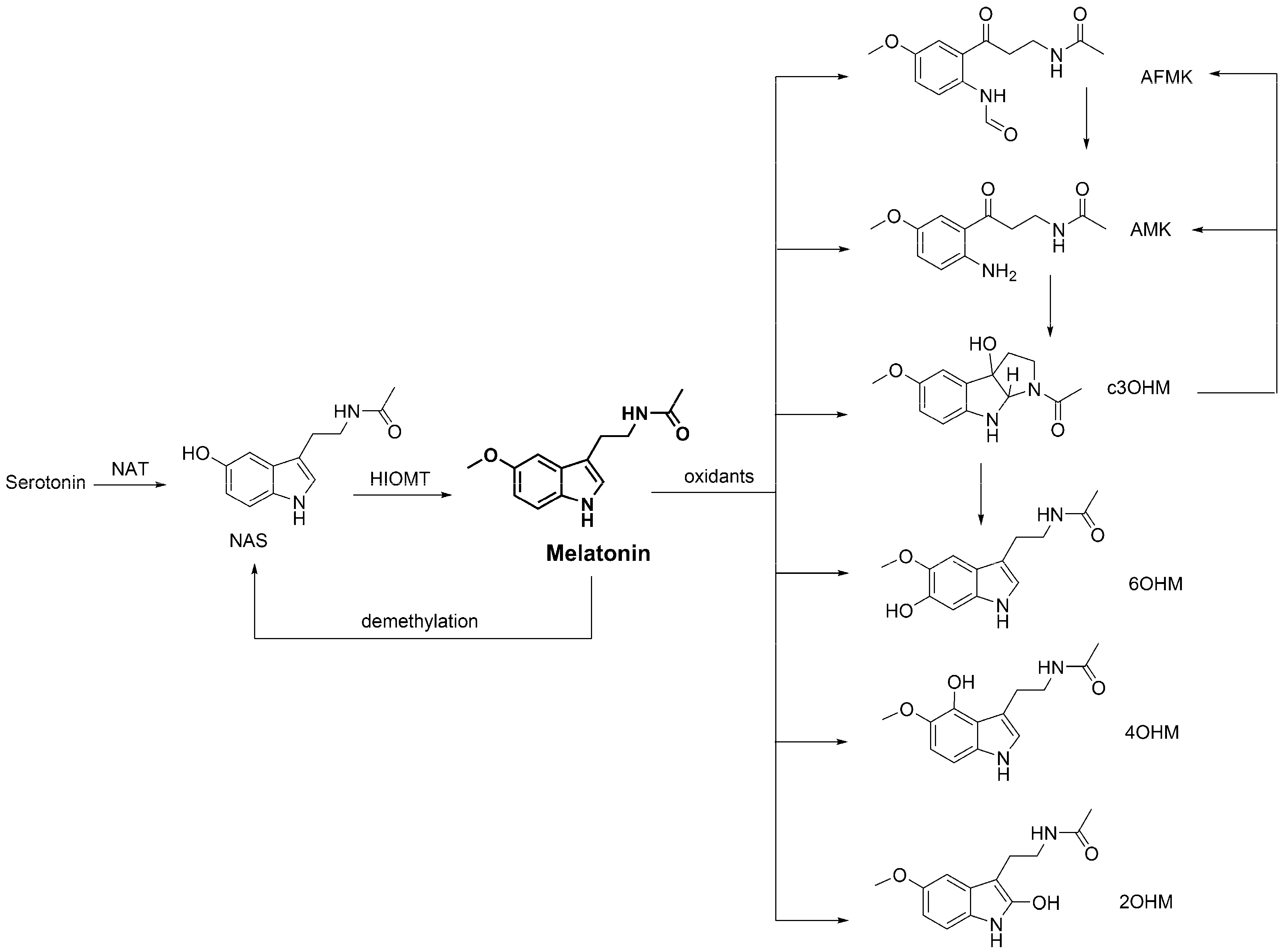
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Galano, A.; Tan, D.-X.; Reiter, R.J. Melatonin: A Versatile Protector against Oxidative DNA Damage. Molecules 2018, 23, 530. https://doi.org/10.3390/molecules23030530
Galano A, Tan D-X, Reiter RJ. Melatonin: A Versatile Protector against Oxidative DNA Damage. Molecules. 2018; 23(3):530. https://doi.org/10.3390/molecules23030530
Chicago/Turabian StyleGalano, Annia, Dun-Xian Tan, and Russel J. Reiter. 2018. "Melatonin: A Versatile Protector against Oxidative DNA Damage" Molecules 23, no. 3: 530. https://doi.org/10.3390/molecules23030530
APA StyleGalano, A., Tan, D.-X., & Reiter, R. J. (2018). Melatonin: A Versatile Protector against Oxidative DNA Damage. Molecules, 23(3), 530. https://doi.org/10.3390/molecules23030530






