Synthesis and Evaluation of the Tumor Cell Growth Inhibitory Potential of New Putative HSP90 Inhibitors
Abstract
:1. Introduction
2. Results and Discussion
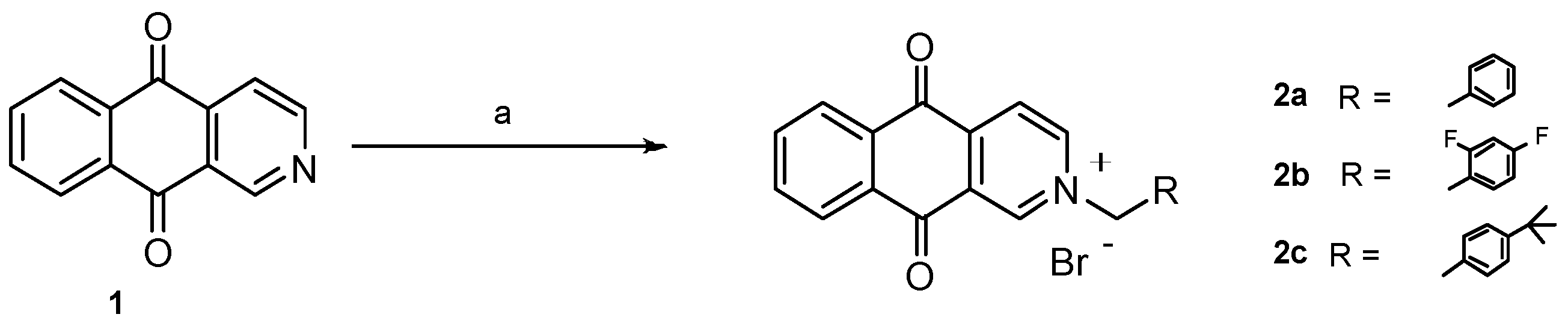
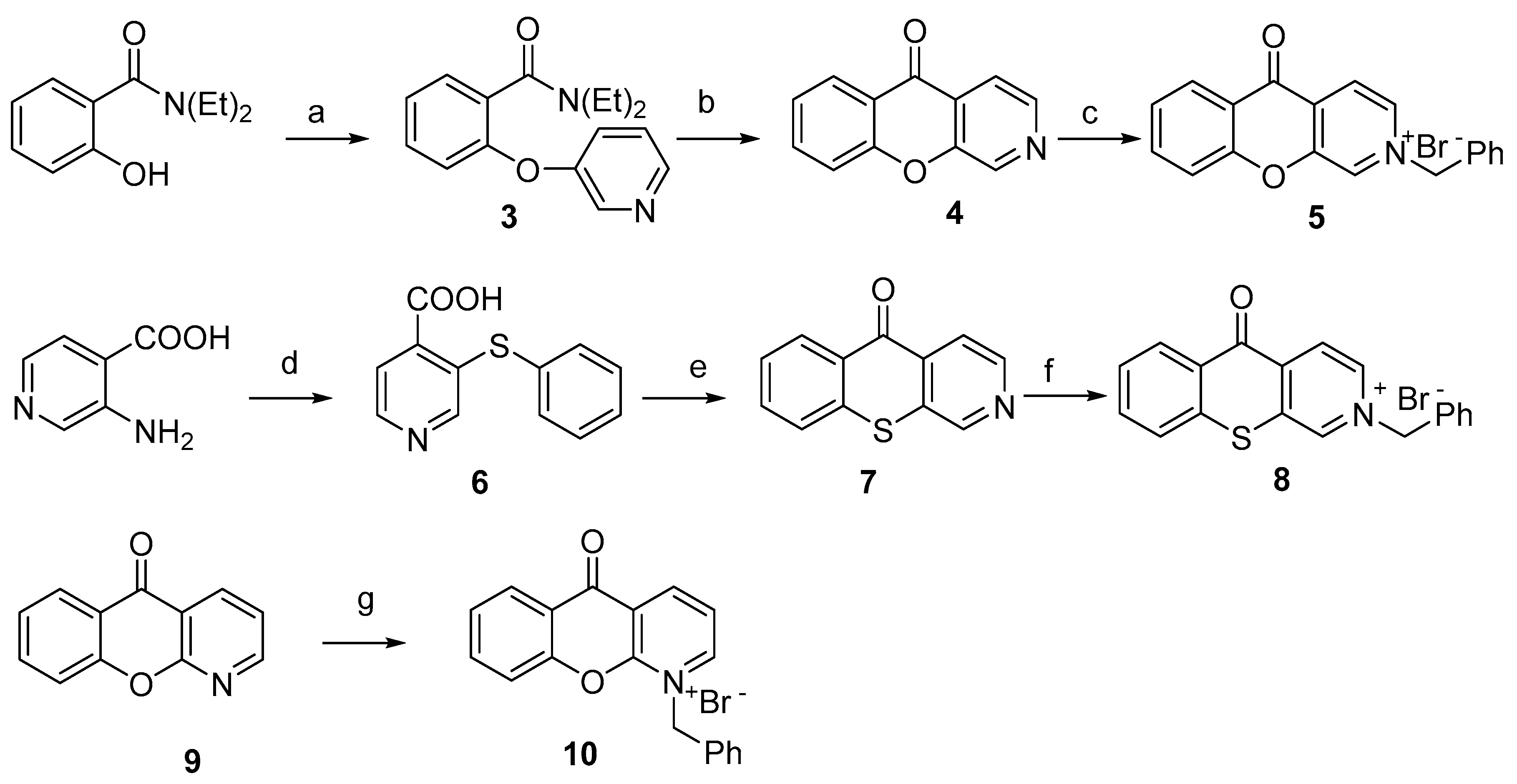
2.1. Chemistry
2.2. Binding Affinity to HSP90, Cell Growth Inhibitory Activity and Structure–Activity Relationship Studies of the Newly Synthesized Compounds
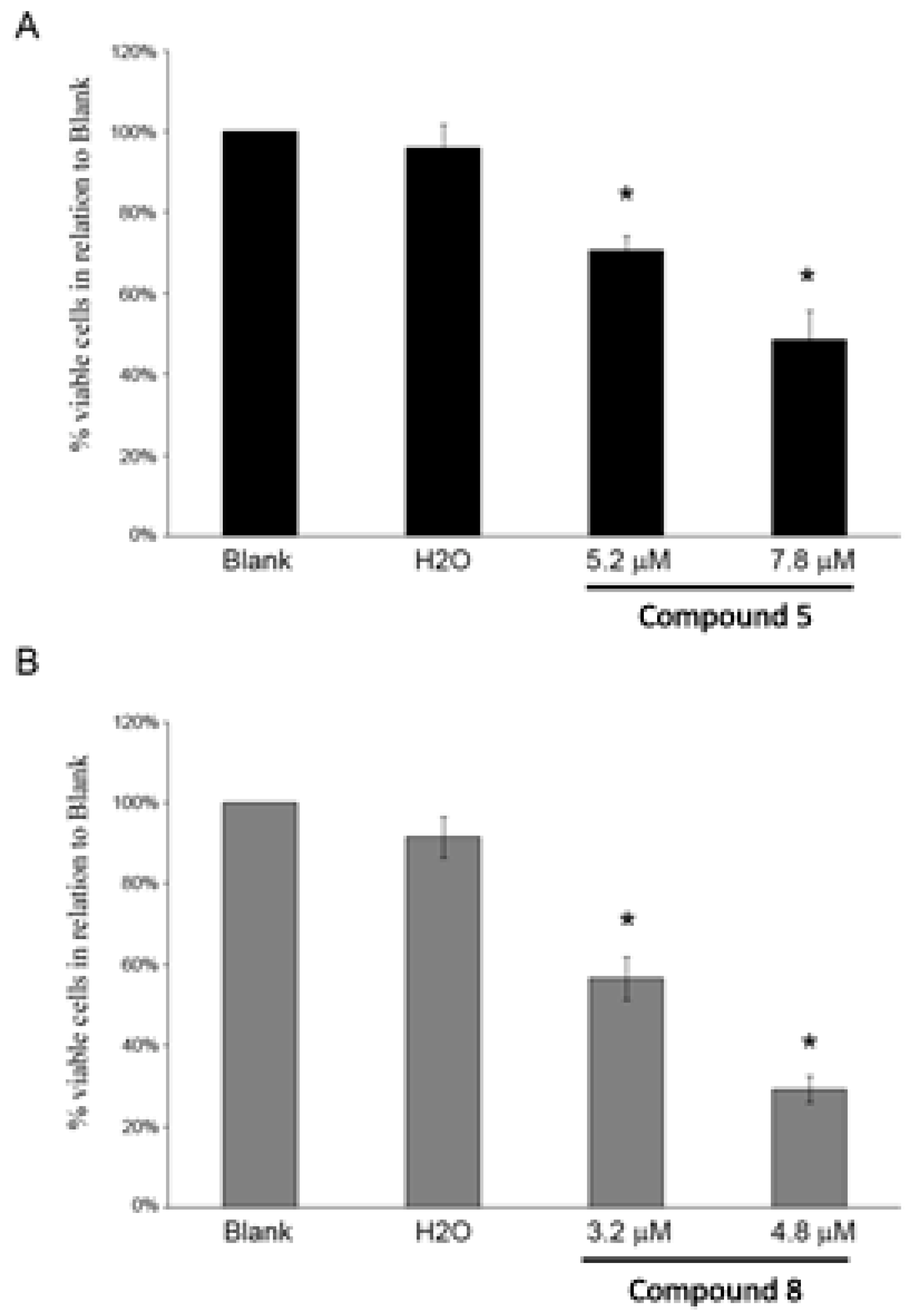
2.3. Compounds 5 and 8 Induced Cytotoxicity in Different Tumor Cell Lines
2.4. Apoptotic Induction in NCI-H460 Cells after Treatment with Compound 8
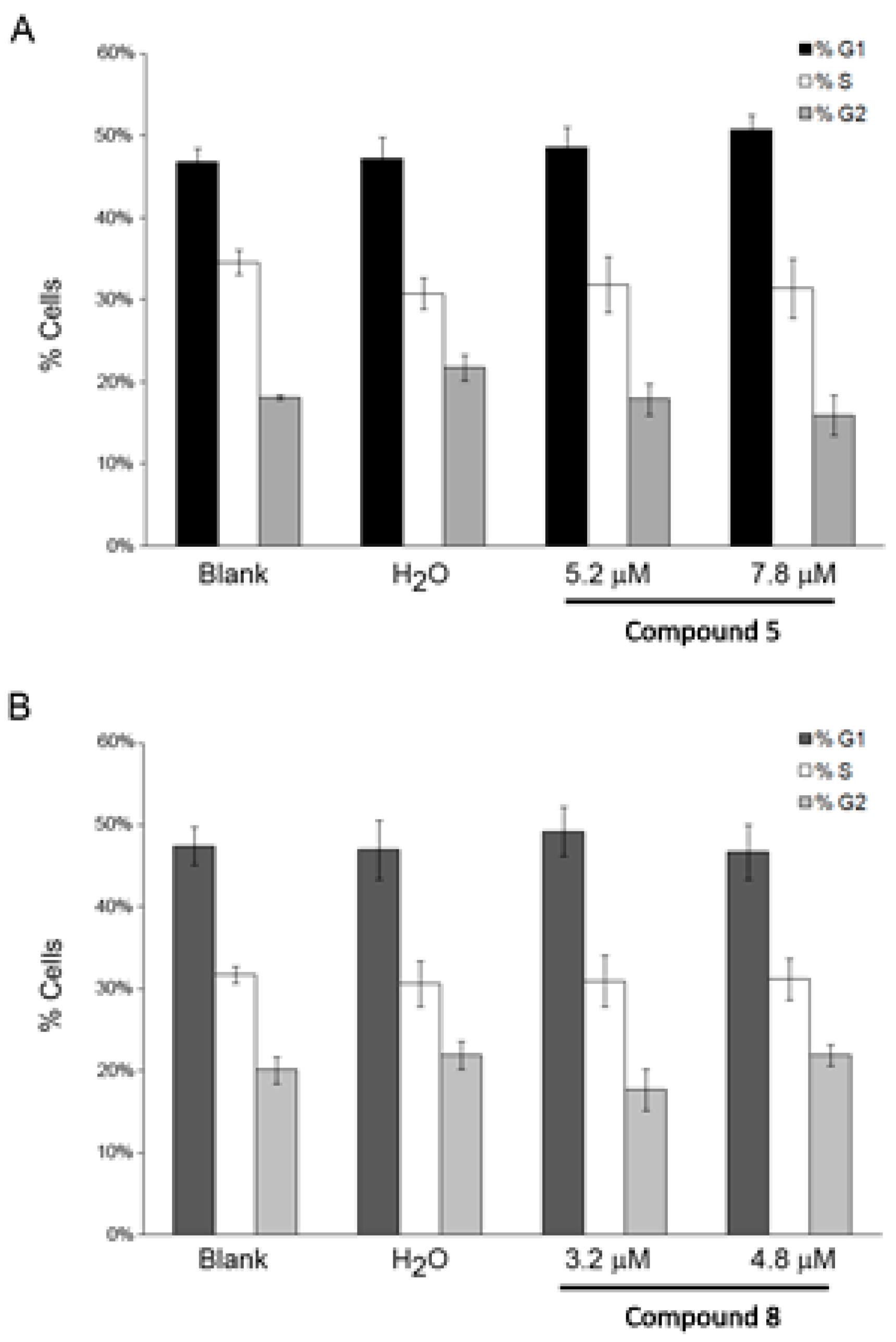
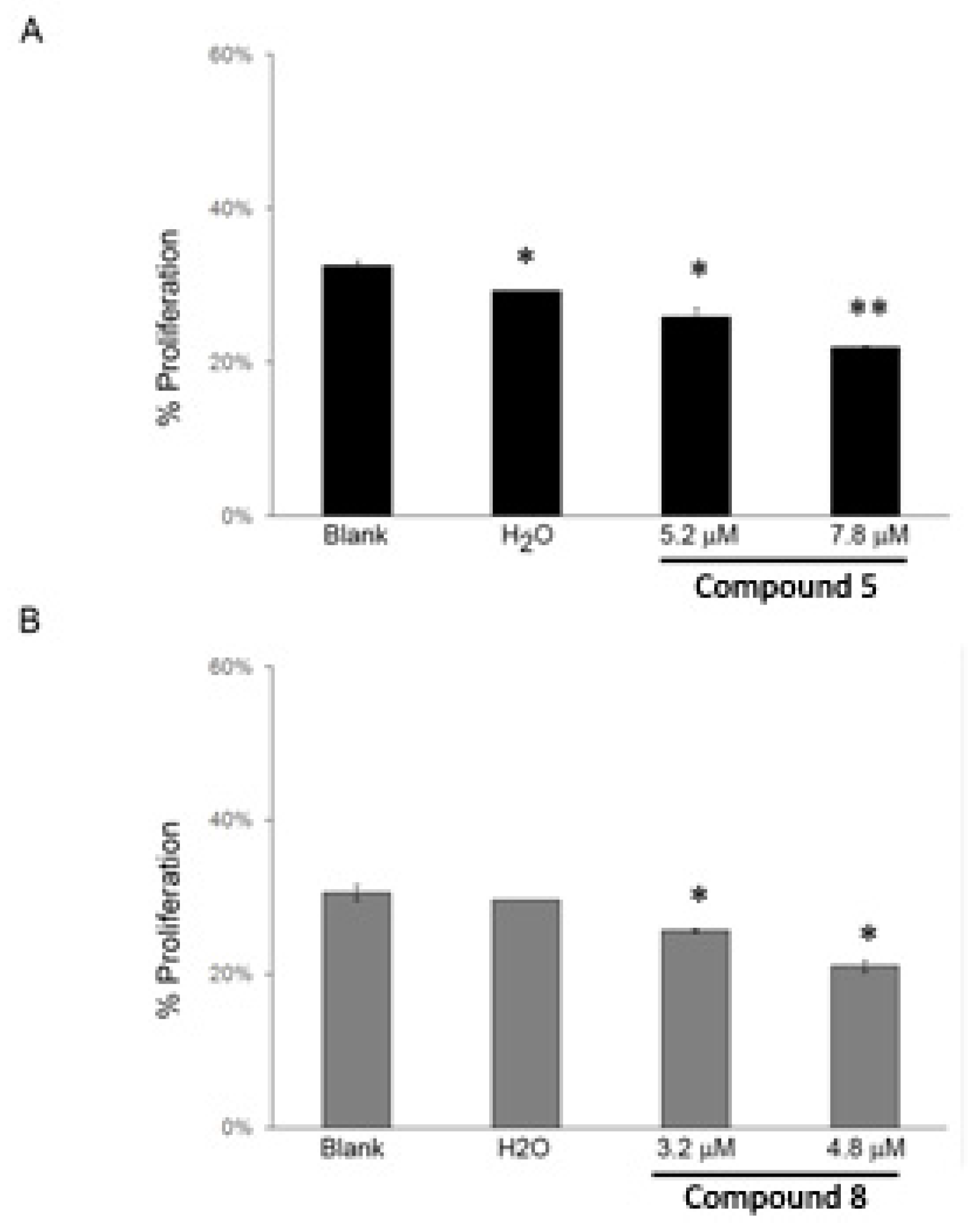
2.5. Effect of the Compounds 5 and 8 on NCI-H460 Cell Cycle Profile and Cellular Proliferation
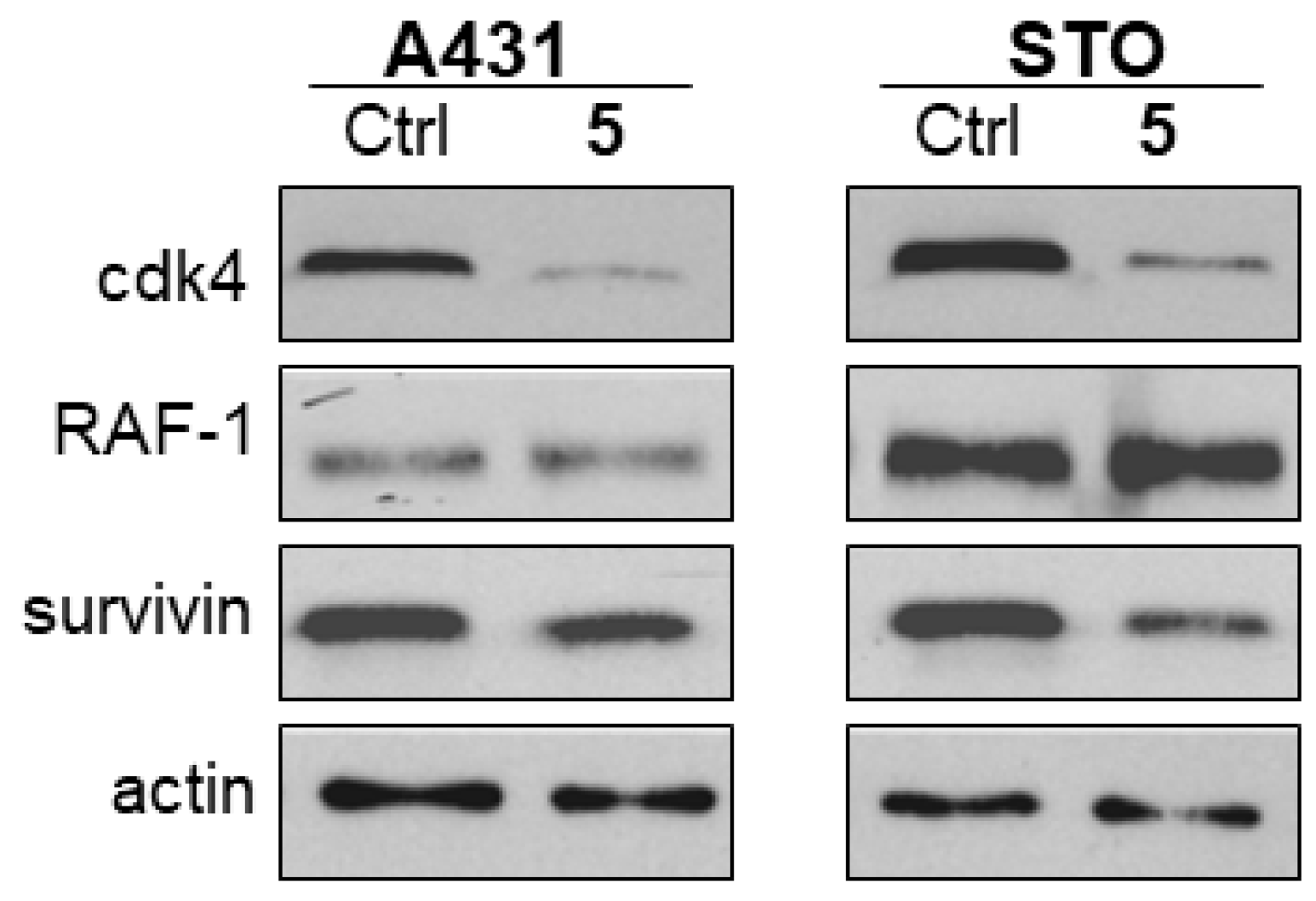
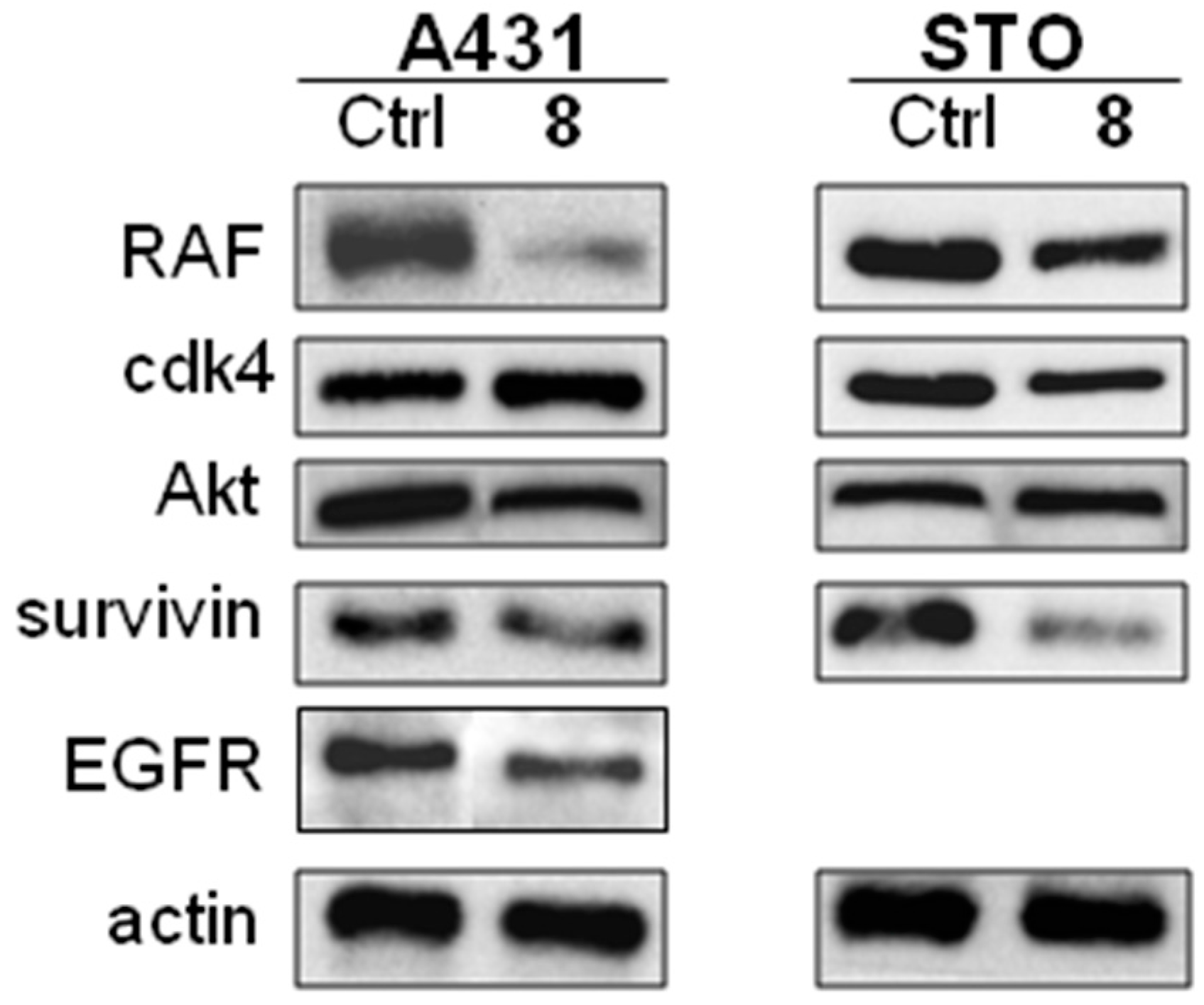
2.6. Effect of Compounds 5 and 8 on HSP90 Client Proteins
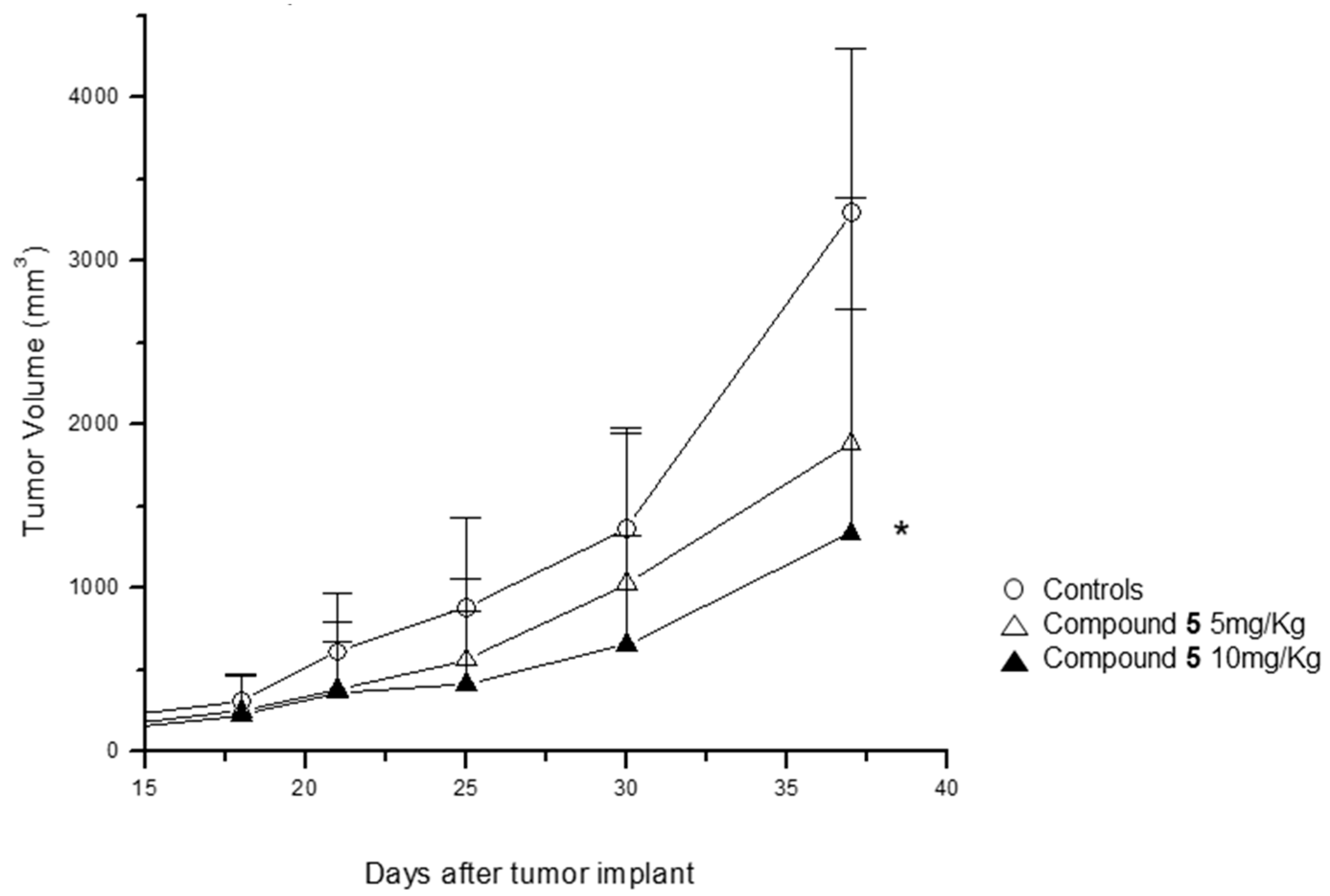
2.7. Effect of Compound 5 in the Growth of a Tumor Xenograft Mouse Model
3. Material and Methods
3.1. Chemistry
3.2. Cell Lines and Culture Conditions
3.3. Cell-Counting Assay and Determination of IC50 Concentrations
3.4. HSP90 Binding Activity
3.5. Analysis of Cell Growth Inhibition with the Sulforhodamine B Assay and Determination of GI50 Concentrations
3.6. Effect of Compounds 5 and 8 on Cell Cycle Profile, Cellular Proliferation and Apoptosis in NCI-H460 Cells
3.6.1. Drug Treatments
3.6.2. Determination of % Viable Cells
3.6.3. Analysis of Cell Cycle Profile
3.6.4. Analysis of Cellular Proliferation
3.6.5. Analysis of Apoptosis
3.7. Effect of Compounds 5 and 8 on Protein Expression Analyzed by Western Blot
3.8. Analysis of the Effect of Compound 5 on the Tumor Growth of Xenografted Mouse Models
3.9. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Morrow, G.; Hightower, L.E.; Tanguay, R.M. Small heat shock proteins: Big folding machines. Cell Stress Chaperones 2015, 20, 207–212. [Google Scholar] [CrossRef] [PubMed]
- Vidyasagar, A.; Wilson, N.A.; Djamali, A. Heat shock protein 27 (HSP27): Biomarker of disease and therapeutic target. Fibrogenesis Tissue Repair 2012, 5, 7. [Google Scholar] [CrossRef] [PubMed]
- Lianos, G.D.; Alexiou, G.A.; Mangano, A.; Mangano, A.; Rausei, S.; Boni, L.; Dionigi, G.; Roukos, D.H. The role of heat shock proteins in cancer. Cancer Lett. 2015, 360, 114–118. [Google Scholar] [CrossRef] [PubMed]
- Jego, G.; Hazoume, A.; Seigneuric, R.; Garrido, C. Targeting heat shock proteins in cancer. Cancer Lett. 2013, 332, 275–285. [Google Scholar] [CrossRef] [PubMed]
- Khalil, A.A.; Kabapy, N.F.; Deraz, S.F.; Smith, C. Heat shock proteins in oncology: Diagnostic biomarkers or therapeutic targets? Biochim. Biophys. Acta 2011, 1816, 89–104. [Google Scholar] [CrossRef] [PubMed]
- Neckers, L. Hsp90 inhibitors as novel cancer chemotherapeutic agents. Trends Mol. Med. 2002, 8 (Suppl. 4), S55–S61. [Google Scholar] [CrossRef]
- Shrestha, L.; Bolaender, A.; Patel, H.J.; Taldone, T. Heat Shock Protein (HSP) Drug Discovery and Development: Targeting Heat Shock Proteins in Disease. Curr. Top Med. Chem. 2016, 16, 16–2753. [Google Scholar] [CrossRef]
- Garcia-Carbonero, R.; Carnero, A.; Paz-Ares, L. Inhibition of HSP90 molecular chaperones: Moving into the clinic. Lancet Oncol. 2013, 14, e358–e369. [Google Scholar] [CrossRef]
- Neckers, L.; Workman, P. Hsp90 molecular chaperone inhibitors: Are we there yet? Clin. Cancer Res. 2012, 18, 64–76. [Google Scholar] [CrossRef] [PubMed]
- Hadden, M.K.; Hill, S.A.; Davenport, J.; Matts, R.L.; Blagg, B.S. Synthesis and evaluation of Hsp90 inhibitors that contain the 1,4-naphthoquinone scaffold. Bioorg. Med. Chem. 2009, 17, 634–640. [Google Scholar] [CrossRef] [PubMed]
- Rios, D.; Benites, J.; Valderrama, J.A.; Farias, M.; Pedrosa, R.C.; Verrax, J.; Buc Calderon, P. Biological evaluation of 3-acyl-2-arylamino-1,4-naphthoquinones as inhibitors of Hsp90 chaperoning function. Curr. Top. Med. Chem. 2012, 12, 2094–2102. [Google Scholar] [CrossRef] [PubMed]
- Davenport, J.; Balch, M.; Galam, L.; Girgis, A.; Hall, J.; Blagg, B.S.; Matts, R.L. High-throughput screen of natural product libraries for hsp90 inhibitors. Biology 2014, 3, 101–138. [Google Scholar] [CrossRef] [PubMed]
- Bargiotti, A.; Musso, L.; Dallavalle, S.; Merlini, L.; Gallo, G.; Ciacci, A.; Giannini, G.; Cabri, W.; Penco, S.; Vesci, L.; et al. Isoxazolo(aza)naphthoquinones: A new class of cytotoxic Hsp90 inhibitors. Eur. J. Med. Chem. 2012, 53, 64–75. [Google Scholar] [CrossRef] [PubMed]
- Uetrecht, J. Screening for the potential of a drug candidate to cause idiosyncratic drug reactions. Drug Discov. Today 2003, 8, 832–837. [Google Scholar] [CrossRef]
- Jayat, C.; Ratinaud, M.H. Cell cycle analysis by flow cytometry: Principles and applications. Biology of the cell/under the auspices of the. Eur. Cell Biol. Organ. 1993, 78, 15–25. [Google Scholar] [CrossRef]
- Nagashima, T.; DeArmond, S.J.; Murovic, J.; Hoshino, T. Immunocytochemical demonstration of S-phase cells by anti-bromodeoxyuridine monoclonal antibody in human brain tumor tissues. Acta Neuropathol. 1985, 67, 155–159. [Google Scholar] [CrossRef] [PubMed]
- Familoni, O.B.; Ionica, I.; Bower, J.F.; Snieckus, V. Intramolecular Anionic Friedel-Crafts Equivalents. A General Regiospecific Route to Substituted and Naturally Occurring Xanthen-9-ones. Synlett 1997, 1997, 1081–1083. [Google Scholar] [CrossRef]
- Kruger, S.; Mann, F.G. Xanthones and thioxanthones. Part V. The preparation and properties of 9-thia-2-aza-anthrone and 9-thia-4-aza-anthrone. J. Chem. Soc. 1954, 3905–3910. [Google Scholar] [CrossRef]
- Vaz, J.A.; Ferreira, I.C.; Tavares, C.; Almeida, G.M.; Martins, A.; Helena Vasconcelos, M. Suillus collinitus methanolic extract increases p53 expression and causes cell cycle arrest and apoptosis in a breast cancer cell line. Food Chem. 2012, 135, 596–602. [Google Scholar] [CrossRef] [PubMed]
- Reis, F.S.; Sousa, D.; Barros, L.; Martins, A.; Morales, P.; Ferreira, I.C.; Vasconcelos, M.H. Leccinum vulpinum Watling induces DNA damage, decreases cell proliferation and induces apoptosis on the human MCF-7 breast cancer cell line. Food Chem. Toxicol. 2016, 90, 45–54. [Google Scholar] [CrossRef] [PubMed]
- Preto, A.; Goncalves, J.; Rebocho, A.P.; Figueiredo, J.; Meireles, A.M.; Rocha, A.S.; Vasconcelos, H.M.; Seca, H.; Seruca, R.; Soares, P.; et al. Proliferation and survival molecules implicated in the inhibition of BRAF pathway in thyroid cancer cells harbouring different genetic mutations. BMC Cancer 2009, 9. [Google Scholar] [CrossRef] [PubMed]
- Dos Santos, T.; Oliveira, M.; Sousa, D.; Lima, R.T.; Martins, A.; Ferreira, I.C.F.R.; Vasconcelos, M.H. Suillus luteus methanolic extract inhibits proliferation and increases expression of p-H2A.X in a non-small cell lung cancer cell line. J. Funct. Foods 2014, 6, 100–106. [Google Scholar] [CrossRef]
- Belaz, K.R.; Denadai, M.; Almeida, A.P.; Lima, R.T.; Vasconcelos, M.H.; Pinto, M.M.; Cass, Q.B.; Oliveira, R.V. Enantiomeric resolution of albendazole sulfoxide by semipreparative HPLC and in vitro study of growth inhibitory effects on human cancer cell lines. J. Pharm. Biomed. Anal. 2012, 66, 100–108. [Google Scholar] [CrossRef] [PubMed]
- Bizarro, A.; Ferreira, I.C.; Sokovic, M.; van Griensven, L.J.; Sousa, D.; Vasconcelos, M.H.; Lima, R.T. Cordyceps militaris (L.) Link Fruiting Body Reduces the Growth of a Non-Small Cell Lung Cancer Cell Line by Increasing Cellular Levels of p53 and p21. Molecules 2015, 20, 13927–13940. [Google Scholar] [CrossRef] [PubMed]
- Giommarelli, C.; Zuco, V.; Favini, E.; Pisano, C.; Dal Piaz, F.; De Tommasi, N.; Zunino, F. The enhancement of antiproliferative and proapoptotic activity of HDAC inhibitors by curcumin is mediated by Hsp90 inhibition. Cell. Mol. Life Sci. 2010, 67, 995–1004. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds 5 and 8 are available from the authors. |
| Binding Affinity (μM) | Cell Growth Inhibitory Activity (μM) | |||
|---|---|---|---|---|
| Compound | Binding HSP90 (FP) IC50 | NCI-H460 IC50 72 h | STO IC50 72 h | A431 IC50 72 h |
| 2a | n.a. | 0.7 ± 0.01 | n.d. | 0.2 ± 0.01 |
| 2b | n.a. | 3.2 ± 0.5 | n.d. | 3.1 ± 0.5 |
| 2c | n.a. | n.d. | n.d. | 0.46 ± 0.8 |
| 4 | >100 | >50 | >50 | >50 |
| 5 | n.a. | 4.8 ± 0.1 | 2.7 ± 0.8 | 1.36 ± 0.05 |
| 7 | 2.47 ± 0.09 | >50 | 46.9 | 46.9 |
| 8 | 3.7 ± 0.03 | n.d. | 0.26 ± 0.08 | 0.26 ± 0.40 |
| 10 | >100 | >50 | >50 | >50 |
| 17-AAG | 1.095 ±0.05 | 0.010 ± 0.002 | - | 0.069 ± 0.005 |
| Compound | GI50 (μM) | ||
|---|---|---|---|
| MCF7 | NCI-H460 | AGS | |
| 5 | 5.4 ± 0.6 | 5.2 ± 1.2 | 6.9 ± 1.2 |
| 8 | 3.2 ± 0.1 | 3.2 ± 0.1 | 3.1 ± 0.3 |
| (A) | Treatments | % Apoptosis | (B) | Treatments | % Apoptosis |
|---|---|---|---|---|---|
| Blank | 7.0 ± 0.6 | Blank | 5.2 ± 0.9 | ||
| H2O | 7.6 ± 1.2 | H2O | 6.4 ± 1.8 | ||
| Compound 5 | 5.2 μM | 7.2 ± 1.4 | Compound 8 | 3.2 μM | 6.7 ± 1.4 |
| 7.8 μM | 7.3 ± 0.9 | 4.8 μM | 12.5 ± 1.7 ** |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bizarro, A.; Sousa, D.; Lima, R.T.; Musso, L.; Cincinelli, R.; Zuco, V.; De Cesare, M.; Dallavalle, S.; Vasconcelos, M.H. Synthesis and Evaluation of the Tumor Cell Growth Inhibitory Potential of New Putative HSP90 Inhibitors. Molecules 2018, 23, 407. https://doi.org/10.3390/molecules23020407
Bizarro A, Sousa D, Lima RT, Musso L, Cincinelli R, Zuco V, De Cesare M, Dallavalle S, Vasconcelos MH. Synthesis and Evaluation of the Tumor Cell Growth Inhibitory Potential of New Putative HSP90 Inhibitors. Molecules. 2018; 23(2):407. https://doi.org/10.3390/molecules23020407
Chicago/Turabian StyleBizarro, Ana, Diana Sousa, Raquel T. Lima, Loana Musso, Raffaella Cincinelli, Vantina Zuco, Michelandrea De Cesare, Sabrina Dallavalle, and M. Helena Vasconcelos. 2018. "Synthesis and Evaluation of the Tumor Cell Growth Inhibitory Potential of New Putative HSP90 Inhibitors" Molecules 23, no. 2: 407. https://doi.org/10.3390/molecules23020407
APA StyleBizarro, A., Sousa, D., Lima, R. T., Musso, L., Cincinelli, R., Zuco, V., De Cesare, M., Dallavalle, S., & Vasconcelos, M. H. (2018). Synthesis and Evaluation of the Tumor Cell Growth Inhibitory Potential of New Putative HSP90 Inhibitors. Molecules, 23(2), 407. https://doi.org/10.3390/molecules23020407







