Proteinaceous Regulators and Inhibitors of Protein Tyrosine Phosphatases
Abstract
1. Introduction
2. Inhibiting and Augmenting PTP Activities
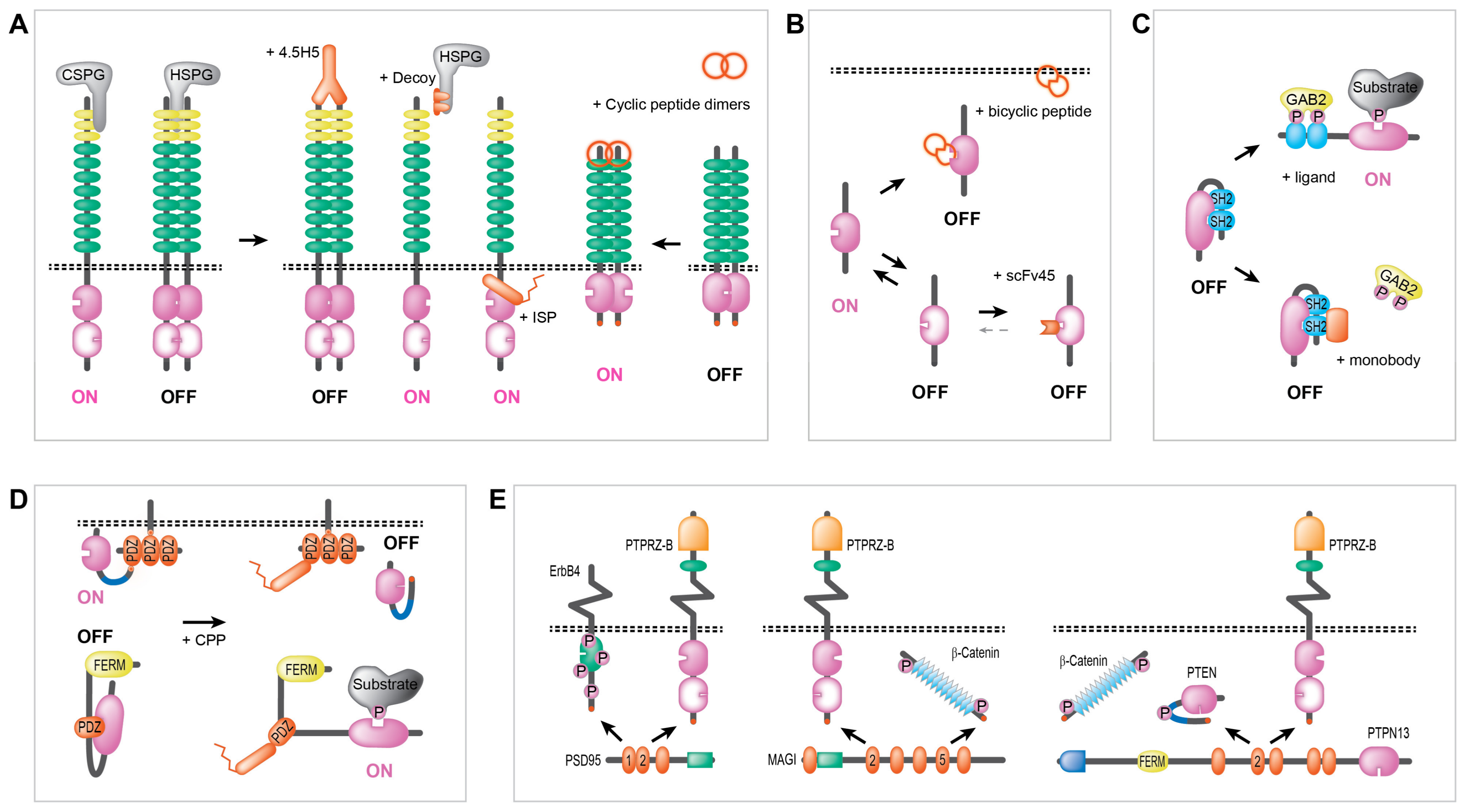
2.1. Intra-Molecular (Allosteric) Regulation of PTP Activity
2.2. Inter-Molecular Regulation of PTP Activity
2.3. Regulating Anchoring and Scaffolding Activity of PTPs
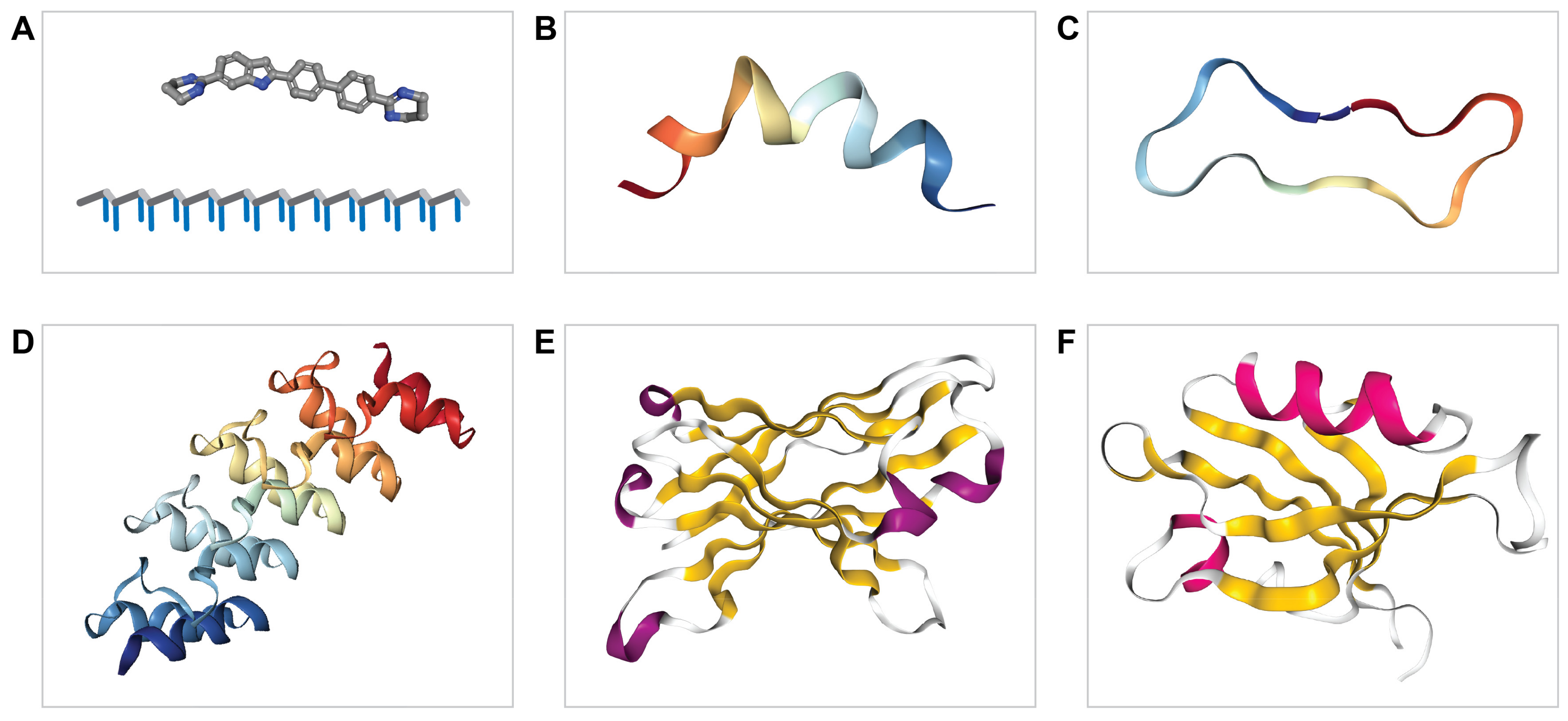
3. Expanding the Druggable Genome Using Proteinaceous Drugs
4. Towards Proteinaceous Drugs that Regulate Protein Tyrosine Phosphatases
5. Untying Gordian Knots in Glioblastomas
6. Conclusions and Perspectives
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Cohen, P. Protein kinases—The major drug targets of the twenty-first century? Nat. Rev. Drug Discov. 2002, 1, 309–315. [Google Scholar] [CrossRef] [PubMed]
- Hendriks, W.J.; Pulido, R. Protein tyrosine phosphatase variants in human hereditary disorders and disease susceptibilities. Biochim. Biophys. Acta 2013, 1832, 1673–1696. [Google Scholar] [CrossRef] [PubMed]
- Hendriks, W.J.; Elson, A.; Harroch, S.; Pulido, R.; Stoker, A.; den Hertog, J. Protein tyrosine phosphatases in health and disease. FEBS J. 2013, 280, 708–730. [Google Scholar] [CrossRef] [PubMed]
- Heinrich, R.; Neel, B.G.; Rapoport, T.A. Mathematical models of protein kinase signal transduction. Mol. Cell 2002, 9, 957–970. [Google Scholar] [CrossRef]
- Hornberg, J.J.; Bruggeman, F.J.; Binder, B.; Geest, C.R.; de Vaate, A.J.; Lankelma, J.; Heinrich, R.; Westerhoff, H.V. Principles behind the multifarious control of signal transduction. Erk phosphorylation and kinase/phosphatase control. FEBS J. 2005, 272, 244–258. [Google Scholar] [CrossRef] [PubMed]
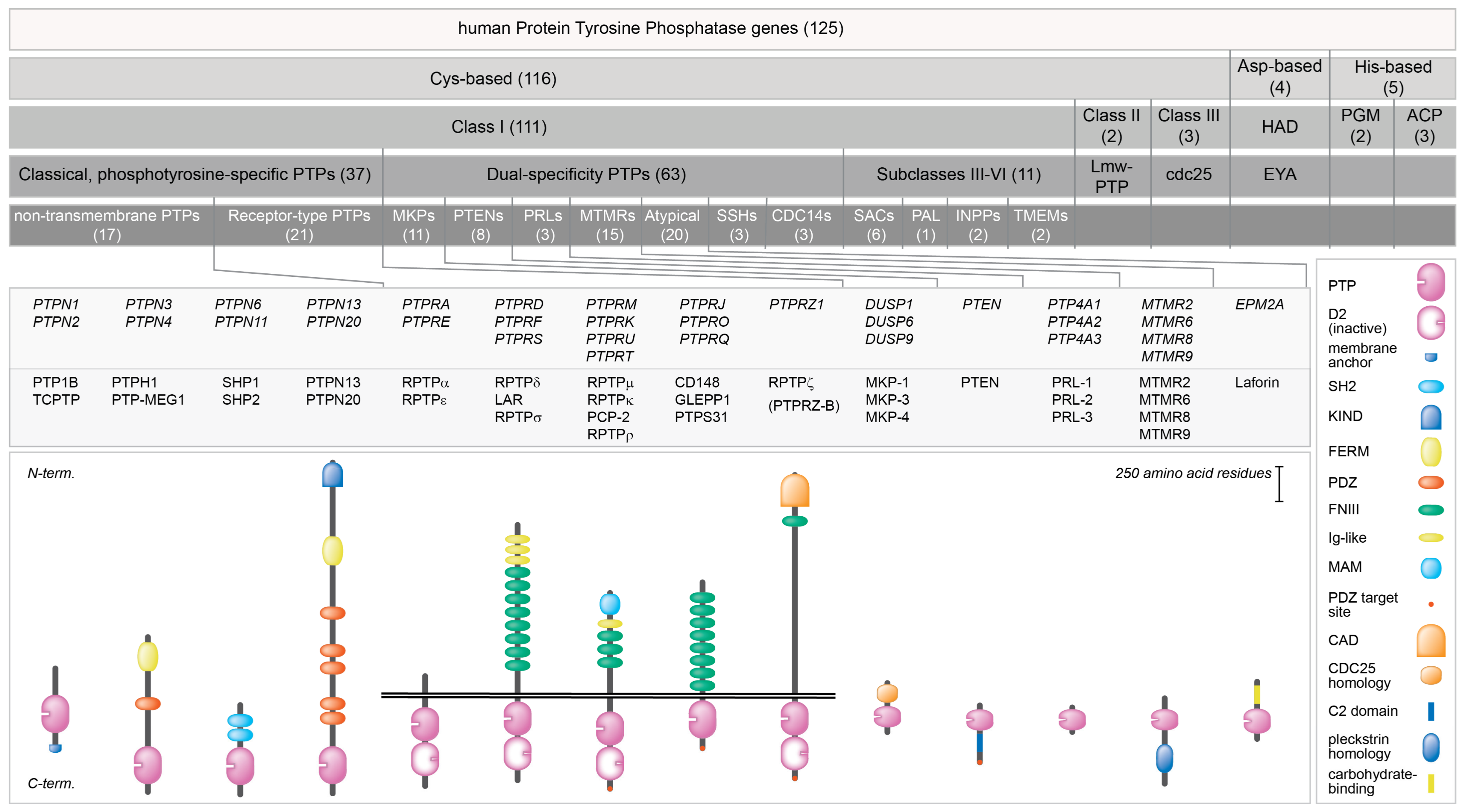
- Alonso, A.; Pulido, R. The extended human ptpome: A growing tyrosine phosphatase family. FEBS J. 2016, 283, 1404–1429. [Google Scholar] [CrossRef] [PubMed]
- Barford, D.; Jia, Z.; Tonks, N.K. Protein tyrosine phosphatases take off. Nat. Struct. Biol. 1995, 2, 1043–1053. [Google Scholar] [CrossRef] [PubMed]
- Andersen, J.N.; Mortensen, O.H.; Peters, G.H.; Drake, P.G.; Iversen, L.F.; Olsen, O.H.; Jansen, P.G.; Andersen, H.S.; Tonks, N.K.; Moller, N.P. Structural and evolutionary relationships among protein tyrosine phosphatase domains. Mol. Cell. Biol. 2001, 21, 7117–7136. [Google Scholar] [CrossRef] [PubMed]
- He, R.J.; Yu, Z.H.; Zhang, R.Y.; Zhang, Z.Y. Protein tyrosine phosphatases as potential therapeutic targets. Acta Pharmacol. Sin. 2014, 35, 1227–1246. [Google Scholar] [CrossRef] [PubMed]
- Peti, W.; Page, R. Strategies to make protein serine/threonine (pp1, calcineurin) and tyrosine phosphatases (ptp1b) druggable: Achieving specificity by targeting substrate and regulatory protein interaction sites. Bioorg. Med. Chem. 2015, 23, 2781–2785. [Google Scholar] [CrossRef] [PubMed]
- Hopkins, A.L.; Groom, C.R. The druggable genome. Nat. Rev. Drug Discov. 2002, 1, 727–730. [Google Scholar] [CrossRef] [PubMed]
- Stanford, S.M.; Bottini, N. Targeting tyrosine phosphatases: Time to end the stigma. Trends Pharmacol. Sci. 2017, 38, 524–540. [Google Scholar] [CrossRef] [PubMed]
- Elchebly, M.; Payette, P.; Michaliszyn, E.; Cromlish, W.; Collins, S.; Loy, A.L.; Normandin, D.; Cheng, A.; Himms-Hagen, J.; Chan, C.C.; et al. Increased insulin sensitivity and obesity resistance in mice lacking the protein tyrosine phosphatase-1b gene. Science 1999, 283, 1544–1548. [Google Scholar] [CrossRef] [PubMed]
- Klaman, L.D.; Boss, O.; Peroni, O.D.; Kim, J.K.; Martino, J.L.; Zabolotny, J.M.; Moghal, N.; Lubkin, M.; Kim, Y.B.; Sharpe, A.H.; et al. Increased energy expenditure, decreased adiposity, and tissue-specific insulin sensitivity in protein-tyrosine phosphatase 1b-deficient mice. Mol. Cell. Biol. 2000, 20, 5479–5489. [Google Scholar] [CrossRef] [PubMed]
- Tartaglia, M.; Niemeyer, C.M.; Fragale, A.; Song, X.; Buechner, J.; Jung, A.; Hahlen, K.; Hasle, H.; Licht, J.D.; Gelb, B.D. Somatic mutations in ptpn11 in juvenile myelomonocytic leukemia, myelodysplastic syndromes and acute myeloid leukemia. Nat. Genet. 2003, 34, 148–150. [Google Scholar] [CrossRef] [PubMed]
- Zinker, B.A.; Rondinone, C.M.; Trevillyan, J.M.; Gum, R.J.; Clampit, J.E.; Waring, J.F.; Xie, N.; Wilcox, D.; Jacobson, P.; Frost, L.; et al. Ptp1b antisense oligonucleotide lowers ptp1b protein, normalizes blood glucose, and improves insulin sensitivity in diabetic mice. Proc. Natl. Acad. Sci. USA 2002, 99, 11357–11362. [Google Scholar] [CrossRef] [PubMed]
- Pandey, S.K.; Yu, X.X.; Watts, L.M.; Michael, M.D.; Sloop, K.W.; Rivard, A.R.; Leedom, T.A.; Manchem, V.P.; Samadzadeh, L.; McKay, R.A.; et al. Reduction of low molecular weight protein-tyrosine phosphatase expression improves hyperglycemia and insulin sensitivity in obese mice. J. Biol. Chem. 2007, 282, 14291–14299. [Google Scholar] [CrossRef] [PubMed]
- Bollu, L.R.; Mazumdar, A.; Savage, M.I.; Brown, P.H. Molecular pathways: Targeting protein tyrosine phosphatases in cancer. Clin. Cancer Res.: Off. J. Am. Assoc. Cancer Res. 2017, 23, 2136–2142. [Google Scholar] [CrossRef] [PubMed]
- Ostman, A.; Frijhoff, J.; Sandin, A.; Bohmer, F.D. Regulation of protein tyrosine phosphatases by reversible oxidation. J. Biochem. 2011, 150, 345–356. [Google Scholar] [CrossRef] [PubMed]
- Pulido, R. Pten: A yin-yang master regulator protein in health and disease. Methods 2015, 77–78, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Hou, S.Q.; Ouyang, M.; Brandmaier, A.; Hao, H.; Shen, W.H. Pten in the maintenance of genome integrity: From DNA replication to chromosome segregation. BioEssays: News Rev. Mol. Cell. Dev. Biol. 2017, 39. [Google Scholar] [CrossRef] [PubMed]
- Barford, D.; Neel, B.G. Revealing mechanisms for sh2 domain mediated regulation of the protein tyrosine phosphatase shp-2. Structure 1998, 6, 249–254. [Google Scholar] [CrossRef]
- Chan, G.; Kalaitzidis, D.; Neel, B.G. The tyrosine phosphatase shp2 (ptpn11) in cancer. Cancer Metastasis Rev. 2008, 27, 179–192. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.N.; LaMarche, M.J.; Chan, H.M.; Fekkes, P.; Garcia-Fortanet, J.; Acker, M.G.; Antonakos, B.; Chen, C.H.; Chen, Z.; Cooke, V.G.; et al. Allosteric inhibition of shp2 phosphatase inhibits cancers driven by receptor tyrosine kinases. Nature 2016, 535, 148–152. [Google Scholar] [CrossRef] [PubMed]
- Garcia Fortanet, J.; Chen, C.H.; Chen, Y.N.; Chen, Z.; Deng, Z.; Firestone, B.; Fekkes, P.; Fodor, M.; Fortin, P.D.; Fridrich, C.; et al. Allosteric inhibition of shp2: Identification of a potent, selective, and orally efficacious phosphatase inhibitor. J. Med. Chem. 2016, 59, 7773–7782. [Google Scholar] [CrossRef] [PubMed]
- Xie, J.; Si, X.; Gu, S.; Wang, M.; Shen, J.; Li, H.; Shen, J.; Li, D.; Fang, Y.; Liu, C.; et al. Allosteric inhibitors of shp2 with therapeutic potential for cancer treatment. J. Med. Chem. 2017. [CrossRef] [PubMed]
- Nunes-Xavier, C.; Roma-Mateo, C.; Rios, P.; Tarrega, C.; Cejudo-Marin, R.; Tabernero, L.; Pulido, R. Dual-specificity map kinase phosphatases as targets of cancer treatment. Anti-Cancer Agents Med. Chem. 2011, 11, 109–132. [Google Scholar] [CrossRef]
- Camps, M.; Nichols, A.; Gillieron, C.; Antonsson, B.; Muda, M.; Chabert, C.; Boschert, U.; Arkinstall, S. Catalytic activation of the phosphatase mkp-3 by erk2 mitogen-activated protein kinase. Science 1998, 280, 1262–1265. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Sun, J.P.; Zhou, B.; Zhang, Z.Y. Structural basis of docking interactions between erk2 and map kinase phosphatase 3. Proc. Natl. Acad. Sci. USA 2006, 103, 5326–5331. [Google Scholar] [CrossRef] [PubMed]
- Zhou, B.; Zhang, J.; Liu, S.; Reddy, S.; Wang, F.; Zhang, Z.Y. Mapping erk2-mkp3 binding interfaces by hydrogen/deuterium exchange mass spectrometry. J. Biol. Chem. 2006, 281, 38834–38844. [Google Scholar] [CrossRef] [PubMed]
- Muda, M.; Theodosiou, A.; Rodrigues, N.; Boschert, U.; Camps, M.; Gillieron, C.; Davies, K.; Ashworth, A.; Arkinstall, S. The dual specificity phosphatases m3/6 and mkp-3 are highly selective for inactivation of distinct mitogen-activated protein kinases. J. Biol. Chem. 1996, 271, 27205–27208. [Google Scholar] [CrossRef] [PubMed]
- Kaltenmeier, C.T.; Vollmer, L.L.; Vernetti, L.A.; Caprio, L.; Davis, K.; Korotchenko, V.N.; Day, B.W.; Tsang, M.; Hulkower, K.I.; Lotze, M.T.; et al. A tumor cell-selective inhibitor of mitogen-activated protein kinase phosphatases sensitizes breast cancer cells to lymphokine-activated killer cell activity. J. Pharmacol. Exp. Therap. 2017, 361, 39–50. [Google Scholar] [CrossRef] [PubMed]
- Korotchenko, V.N.; Saydmohammed, M.; Vollmer, L.L.; Bakan, A.; Sheetz, K.; Debiec, K.T.; Greene, K.A.; Agliori, C.S.; Bahar, I.; Day, B.W.; et al. In vivo structure-activity relationship studies support allosteric targeting of a dual specificity phosphatase. ChemBioChem: Eur. J. Chem. Biol. 2014, 15, 1436–1445. [Google Scholar] [CrossRef] [PubMed]
- Stoker, A.W. Rptps in axons, synapses and neurology. Semin. Cell Dev. Biol. 2015, 37, 90–97. [Google Scholar] [CrossRef] [PubMed]
- Madan, L.L.; Veeranna, S.; Shameer, K.; Reddy, C.C.; Sowdhamini, R.; Gopal, B. Modulation of catalytic activity in multi-domain protein tyrosine phosphatases. PLoS ONE 2011, 6, e24766. [Google Scholar] [CrossRef] [PubMed]
- Streuli, M.; Krueger, N.X.; Thai, T.; Tang, M.; Saito, H. Distinct functional roles of the two intracellular phosphatase like domains of the receptor-linked protein tyrosine phosphatases lca and lar. EMBO J. 1990, 9, 2399–2407. [Google Scholar] [PubMed]
- Xu, Z.; Weiss, A. Negative regulation of cd45 by differential homodimerization of the alternatively spliced isoforms. Nat. Immunol. 2002, 3, 764–771. [Google Scholar] [CrossRef] [PubMed]
- Jiang, G.; den Hertog, J.; Hunter, T. Receptor-like protein tyrosine phosphatase alpha homodimerizes on the cell surface. Mol. Cell. Biol. 2000, 20, 5917–5929. [Google Scholar] [CrossRef] [PubMed]
- Gross, S.; Blanchetot, C.; Schepens, J.; Albet, S.; Lammers, R.; den Hertog, J.; Hendriks, W. Multimerization of the protein-tyrosine phosphatase (ptp)-like insulin-dependent diabetes mellitus autoantigens ia-2 and ia-2beta with receptor ptps (rptps). Inhibition of rptpalpha enzymatic activity. J. Biol. Chem. 2002, 277, 48139–48145. [Google Scholar] [CrossRef] [PubMed]
- Bilwes, A.M.; den Hertog, J.; Hunter, T.; Noel, J.P. Structural basis for inhibition of receptor protein-tyrosine phosphatase-alpha by dimerization. Nature 1996, 382, 555–559. [Google Scholar] [CrossRef] [PubMed]
- Jiang, G.; den Hertog, J.; Su, J.; Noel, J.; Sap, J.; Hunter, T. Dimerization inhibits the activity of receptor-like protein-tyrosine phosphatase-alpha. Nature 1999, 401, 606–610. [Google Scholar] [CrossRef] [PubMed]
- Majeti, R.; Bilwes, A.M.; Noel, J.P.; Hunter, T.; Weiss, A. Dimerization-induced inhibition of receptor protein tyrosine phosphatase function through an inhibitory wedge. Science 1998, 279, 88–91. [Google Scholar] [CrossRef] [PubMed]
- Noordman, Y.E.; Augustus, E.D.; Schepens, J.T.; Chirivi, R.G.; Rios, P.; Pulido, R.; Hendriks, W.J. Multimerisation of receptor-type protein tyrosine phosphatases ptpbr7 and ptp-sl attenuates enzymatic activity. Biochim. Biophys. Acta 2008, 1783, 275–286. [Google Scholar] [CrossRef] [PubMed]
- Stoker, A.W. Detection and identification of ligands for mammalian rptp extracellular domains. Methods Mol. Biol. 2016, 1447, 267–281. [Google Scholar] [PubMed]
- Meng, K.; Rodriguez-Pena, A.; Dimitrov, T.; Chen, W.; Yamin, M.; Noda, M.; Deuel, T.F. Pleiotrophin signals increased tyrosine phosphorylation of beta beta-catenin through inactivation of the intrinsic catalytic activity of the receptor-type protein tyrosine phosphatase beta/zeta. Proc. Natl. Acad. Sci. USA 2000, 97, 2603–2608. [Google Scholar] [CrossRef] [PubMed]
- Fukada, M.; Fujikawa, A.; Chow, J.P.; Ikematsu, S.; Sakuma, S.; Noda, M. Protein tyrosine phosphatase receptor type z is inactivated by ligand-induced oligomerization. FEBS Lett. 2006, 580, 4051–4056. [Google Scholar] [CrossRef] [PubMed]
- Walchli, S.; Espanel, X.; Hooft van Huijsduijnen, R. Sap-1/ptprh activity is regulated by reversible dimerization. Biochem. Biophys. Res. Commun. 2005, 331, 497–502. [Google Scholar] [CrossRef] [PubMed]
- Toledano-Katchalski, H.; Tiran, Z.; Sines, T.; Shani, G.; Granot-Attas, S.; den Hertog, J.; Elson, A. Dimerization in vivo and inhibition of the nonreceptor form of protein tyrosine phosphatase epsilon. Mol. Cell. Biol. 2003, 23, 5460–5471. [Google Scholar] [CrossRef] [PubMed]
- Pavic, K.; Rios, P.; Dzeyk, K.; Koehler, C.; Lemke, E.A.; Kohn, M. Unnatural amino acid mutagenesis reveals dimerization as a negative regulatory mechanism of vhr's phosphatase activity. ACS Chem. Biol. 2014, 9, 1451–1459. [Google Scholar] [CrossRef] [PubMed]
- Raththagala, M.; Brewer, M.K.; Parker, M.W.; Sherwood, A.R.; Wong, B.K.; Hsu, S.; Bridges, T.M.; Paasch, B.C.; Hellman, L.M.; Husodo, S.; et al. Structural mechanism of laforin function in glycogen dephosphorylation and lafora disease. Mol. Cell 2015, 57, 261–272. [Google Scholar] [CrossRef] [PubMed]
- Heinrich, F.; Chakravarthy, S.; Nanda, H.; Papa, A.; Pandolfi, P.P.; Ross, A.H.; Harishchandra, R.K.; Gericke, A.; Losche, M. The pten tumor suppressor forms homodimers in solution. Structure 2015, 23, 1952–1957. [Google Scholar] [CrossRef] [PubMed]
- Papa, A.; Wan, L.; Bonora, M.; Salmena, L.; Song, M.S.; Hobbs, R.M.; Lunardi, A.; Webster, K.; Ng, C.; Newton, R.H.; et al. Cancer-associated pten mutants act in a dominant-negative manner to suppress pten protein function. Cell 2014, 157, 595–610. [Google Scholar] [CrossRef] [PubMed]
- Berger, P.; Schaffitzel, C.; Berger, I.; Ban, N.; Suter, U. Membrane association of myotubularin-related protein 2 is mediated by a pleckstrin homology-gram domain and a coiled-coil dimerization module. Proc. Natl. Acad. Sci. USA 2003, 100, 12177–12182. [Google Scholar] [CrossRef] [PubMed]
- Zou, J.; Chang, S.C.; Marjanovic, J.; Majerus, P.W. Mtmr9 increases mtmr6 enzyme activity, stability, and role in apoptosis. J. Biol. Chem. 2009, 284, 2064–2071. [Google Scholar] [CrossRef] [PubMed]
- Zou, J.; Zhang, C.; Marjanovic, J.; Kisseleva, M.V.; Majerus, P.W.; Wilson, M.P. Myotubularin-related protein (mtmr) 9 determines the enzymatic activity, substrate specificity, and role in autophagy of mtmr8. Proc. Natl. Acad. Sci. USA 2012, 109, 9539–9544. [Google Scholar] [CrossRef] [PubMed]
- Rios, P.; Li, X.; Kohn, M. Molecular mechanisms of the prl phosphatases. FEBS J. 2013, 280, 505–524. [Google Scholar] [CrossRef] [PubMed]
- Bai, Y.; Yu, Z.H.; Liu, S.; Zhang, L.; Zhang, R.Y.; Zeng, L.F.; Zhang, S.; Zhang, Z.Y. Novel anticancer agents based on targeting the trimer interface of the prl phosphatase. Cancer Res. 2016, 76, 4805–4815. [Google Scholar] [CrossRef] [PubMed]
- Pulido, R.; Stoker, A.W.; Hendriks, W.J. Ptps emerge as pips: Protein tyrosine phosphatases with lipid-phosphatase activities in human disease. Hum. Mol. Genet. 2013, 22, R66–76. [Google Scholar] [CrossRef] [PubMed]
- Khan, Z.; Lafon, M. Pdz domain-mediated protein interactions: Therapeutic targets in neurological disorders. Curr. Med. Chem. 2014, 21, 2632–2641. [Google Scholar] [CrossRef] [PubMed]
- Jeong, J.; VanHouten, J.N.; Kim, W.; Dann, P.; Sullivan, C.; Choi, J.; Sneddon, W.B.; Friedman, P.A.; Wysolmerski, J.J. The scaffolding protein nherf1 regulates the stability and activity of the tyrosine kinase her2. J. Biol. Chem. 2017, 292, 6555–6568. [Google Scholar] [CrossRef] [PubMed]
- Knafo, S.; Sanchez-Puelles, C.; Palomer, E.; Delgado, I.; Draffin, J.E.; Mingo, J.; Wahle, T.; Kaleka, K.; Mou, L.; Pereda-Perez, I.; et al. Pten recruitment controls synaptic and cognitive function in alzheimer’s models. Nat. Neurosci. 2016, 19, 443–453. [Google Scholar] [CrossRef] [PubMed]
- Fujii, N.; Haresco, J.J.; Novak, K.A.; Stokoe, D.; Kuntz, I.D.; Guy, R.K. A selective irreversible inhibitor targeting a pdz protein interaction domain. J. Am. Chem. Soc. 2003, 125, 12074–12075. [Google Scholar] [CrossRef] [PubMed]
- Fujii, N.; Haresco, J.J.; Novak, K.A.; Gage, R.M.; Pedemonte, N.; Stokoe, D.; Kuntz, I.D.; Guy, R.K. Rational design of a nonpeptide general chemical scaffold for reversible inhibition of pdz domain interactions. Bioorg. Med. Chem. Lett. 2007, 17, 549–552. [Google Scholar] [CrossRef] [PubMed]
- Chung, J.S.; Cruz, P.D., Jr.; Ariizumi, K. Inhibition of t-cell activation by syndecan-4 is mediated by cd148 through protein tyrosine phosphatase activity. Eur. J. Immunol. 2011, 41, 1794–1799. [Google Scholar] [CrossRef] [PubMed]
- Iuliano, R.; Trapasso, F.; Sama, I.; Le Pera, I.; Martelli, M.L.; Lembo, F.; Santoro, M.; Viglietto, G.; Chiariotti, L.; Fusco, A. Rat protein tyrosine phosphatase eta physically interacts with the pdz domains of syntenin. FEBS Lett. 2001, 500, 41–44. [Google Scholar] [CrossRef]
- Babault, N.; Cordier, F.; Lafage, M.; Cockburn, J.; Haouz, A.; Prehaud, C.; Rey, F.A.; Delepierre, M.; Buc, H.; Lafon, M.; et al. Peptides targeting the pdz domain of ptpn4 are efficient inducers of glioblastoma cell death. Structure 2011, 19, 1518–1524. [Google Scholar] [CrossRef] [PubMed]
- Maisonneuve, P.; Caillet-Saguy, C.; Raynal, B.; Gilquin, B.; Chaffotte, A.; Perez, J.; Zinn-Justin, S.; Delepierre, M.; Buc, H.; Cordier, F.; et al. Regulation of the catalytic activity of the human phosphatase ptpn4 by its pdz domain. FEBS J. 2014, 281, 4852–4865. [Google Scholar] [CrossRef] [PubMed]
- Maisonneuve, P.; Caillet-Saguy, C.; Vaney, M.C.; Bibi-Zainab, E.; Sawyer, K.; Raynal, B.; Haouz, A.; Delepierre, M.; Lafon, M.; Cordier, F.; et al. Molecular basis of the interaction of the human protein tyrosine phosphatase non-receptor type 4 (ptpn4) with the mitogen-activated protein kinase p38 gamma. J. Biol. Chem. 2016, 291, 16699–16708. [Google Scholar] [CrossRef] [PubMed]
- Caillet-Saguy, C.; Toto, A.; Guerois, R.; Maisonneuve, P.; di Silvio, E.; Sawyer, K.; Gianni, S.; Wolff, N. Regulation of the human phosphatase ptpn4 by the inter-domain linker connecting the pdz and the phosphatase domains. Sci. Rep. 2017, 7, 7875. [Google Scholar] [CrossRef] [PubMed]
- Xiao, X.; He, Q.H.; Yu, L.Y.; Wang, S.Q.; Li, Y.; Yang, H.; Zhang, A.H.; Ma, X.H.; Peng, Y.J.; Chen, B. Structure-based optimization of salt-bridge network across the complex interface of ptpn4 pdz domain with its peptide ligands in neuroglioma. Comput. Biol. Chem. 2017, 66, 63–68. [Google Scholar] [CrossRef] [PubMed]
- Arkin, M.R.; Tang, Y.; Wells, J.A. Small-molecule inhibitors of protein-protein interactions: Progressing toward the reality. Chem. Biol. 2014, 21, 1102–1114. [Google Scholar] [CrossRef] [PubMed]
- London, N.; Raveh, B.; Schueler-Furman, O. Druggable protein-protein interactions--from hot spots to hot segments. Curr. Opin. Chem. Biol. 2013, 17, 952–959. [Google Scholar] [CrossRef] [PubMed]
- King, A.C.; Peterson, T.J.; Horvat, T.Z.; Rodriguez, M.; Tang, L.A. Venetoclax: A first-in-class oral bcl-2 inhibitor for the management of lymphoid malignancies. Ann. Pharmacother. 2017, 51, 410–416. [Google Scholar] [CrossRef] [PubMed]
- Frankson, R.; Yu, Z.H.; Bai, Y.; Li, Q.; Zhang, R.Y.; Zhang, Z.Y. Therapeutic targeting of oncogenic tyrosine phosphatases. Cancer Res. 2017, 77, 5701–5705. [Google Scholar] [CrossRef] [PubMed]
- Vargas, C.; Radziwill, G.; Krause, G.; Diehl, A.; Keller, S.; Kamdem, N.; Czekelius, C.; Kreuchwig, A.; Schmieder, P.; Doyle, D.; et al. Small-molecule inhibitors of af6 pdz-mediated protein-protein interactions. ChemMedChem 2014, 9, 1458–1462. [Google Scholar] [CrossRef] [PubMed]
- Bach, A.; Pedersen, S.W.; Dorr, L.A.; Vallon, G.; Ripoche, I.; Ducki, S.; Lian, L.Y. Biochemical investigations of the mechanism of action of small molecules zl006 and ic87201 as potential inhibitors of the nnos-pdz/psd-95-pdz interactions. Sci. Rep. 2015, 5, 12157. [Google Scholar] [CrossRef] [PubMed]
- Wynes, M.W.; Edelman, B.L.; Kostyk, A.G.; Edwards, M.G.; Coldren, C.; Groshong, S.D.; Cosgrove, G.P.; Redente, E.F.; Bamberg, A.; Brown, K.K.; et al. Increased cell surface fas expression is necessary and sufficient to sensitize lung fibroblasts to fas ligation-induced apoptosis: Implications for fibroblast accumulation in idiopathic pulmonary fibrosis. J. Immunol. 2011, 187, 527–537. [Google Scholar] [CrossRef] [PubMed]
- Laraia, L.; McKenzie, G.; Spring, D.R.; Venkitaraman, A.R.; Huggins, D.J. Overcoming chemical, biological, and computational challenges in the development of inhibitors targeting protein-protein interactions. Chem. Biol. 2015, 22, 689–703. [Google Scholar] [CrossRef] [PubMed]
- Sathish, J.G.; Sethu, S.; Bielsky, M.C.; de Haan, L.; French, N.S.; Govindappa, K.; Green, J.; Griffiths, C.E.; Holgate, S.; Jones, D.; et al. Challenges and approaches for the development of safer immunomodulatory biologics. Nat. Rev. Drug Discov. 2013, 12, 306–324. [Google Scholar] [CrossRef] [PubMed]
- Bullock, B.N.; Jochim, A.L.; Arora, P.S. Assessing helical protein interfaces for inhibitor design. J. Am. Chem. Soc. 2011, 133, 14220–14223. [Google Scholar] [CrossRef] [PubMed]
- Liskamp, R.M.J.; Rijkers, D.T.S.; Bakker, S.E. Bioactive macrocyclic peptides and peptide mimics. In Modern Supramolecular Chemistry; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2008; pp. 1–27. [Google Scholar]
- Dohm, M.T.; Kapoor, R.; Barron, A.E. Peptoids: Bio-inspired polymers as potential pharmaceuticals. Curr. Pharm. Des. 2011, 17, 2732–2747. [Google Scholar] [CrossRef] [PubMed]
- Goodman, C.M.; Choi, S.; Shandler, S.; DeGrado, W.F. Foldamers as versatile frameworks for the design and evolution of function. Nat. Chem. Biol. 2007, 3, 252–262. [Google Scholar] [CrossRef] [PubMed]
- Accardo, A.; Aloj, L.; Aurilio, M.; Morelli, G.; Tesauro, D. Receptor binding peptides for target-selective delivery of nanoparticles encapsulated drugs. Int. J. Nanomed. 2014, 9, 1537–1557. [Google Scholar]
- Pelay-Gimeno, M.; Glas, A.; Koch, O.; Grossmann, T.N. Structure-based design of inhibitors of protein-protein interactions: Mimicking peptide binding epitopes. Angew. Chem. Int. Ed. 2015, 54, 8896–8927. [Google Scholar] [CrossRef] [PubMed]
- Kaspar, A.A.; Reichert, J.M. Future directions for peptide therapeutics development. Drug Discov. Today 2013, 18, 807–817. [Google Scholar] [CrossRef] [PubMed]
- Fosgerau, K.; Hoffmann, T. Peptide therapeutics: Current status and future directions. Drug Discov. Today 2015, 20, 122–128. [Google Scholar] [CrossRef] [PubMed]
- Felix, J.; Savvides, S.N. Mechanisms of immunomodulation by mammalian and viral decoy receptors: Insights from structures. Nat. Rev. Immunol. 2017, 17, 112–129. [Google Scholar] [CrossRef] [PubMed]
- Czajkowsky, D.M.; Hu, J.; Shao, Z.; Pleass, R.J. Fc-fusion proteins: New developments and future perspectives. EMBO Mol. Med. 2012, 4, 1015–1028. [Google Scholar] [CrossRef] [PubMed]
- Boldicke, T. Single domain antibodies for the knockdown of cytosolic and nuclear proteins. Protein Sci.: Publ. Protein Soc. 2017, 26, 925–945. [Google Scholar] [CrossRef] [PubMed]
- Strohl, W.R. Current progress in innovative engineered antibodies. Protein Cell 2017. [Google Scholar] [CrossRef] [PubMed]
- Sha, F.; Salzman, G.; Gupta, A.; Koide, S. Monobodies and other synthetic binding proteins for expanding protein science. Protein Sci.: Publ. Protein Soc. 2017, 26, 910–924. [Google Scholar] [CrossRef] [PubMed]
- Pluckthun, A. Designed ankyrin repeat proteins (darpins): Binding proteins for research, diagnostics, and therapy. In Annual Review of Pharmacology and Toxicology; Insel, P.A., Ed.; ANNUAL REVIEWS: Palo Alto, CA, USA, 2015; Volume 55, pp. 489–511. [Google Scholar]
- Wu, C.L.; Hardy, S.; Aubry, I.; Landry, M.; Haggarty, A.; Saragovi, H.U.; Tremblay, M.L. Identification of function-regulating antibodies targeting the receptor protein tyrosine phosphatase sigma ectodomain. PLoS ONE 2017, 12, e0178489. [Google Scholar] [CrossRef] [PubMed]
- Aricescu, A.R.; McKinnell, I.W.; Halfter, W.; Stoker, A.W. Heparan sulfate proteoglycans are ligands for receptor protein tyrosine phosphatase sigma. Mol. Cell. Biol. 2002, 22, 1881–1892. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Faux, C.; Nixon, J.; Alete, D.; Chilton, J.; Hawadle, M.; Stoker, A.W. Dimerization of protein tyrosine phosphatase sigma governs both ligand binding and isoform specificity. Mol. Cell. Biol. 2007, 27, 1795–1808. [Google Scholar] [CrossRef] [PubMed]
- Coles, C.H.; Mitakidis, N.; Zhang, P.; Elegheert, J.; Lu, W.; Stoker, A.W.; Nakagawa, T.; Craig, A.M.; Jones, E.Y.; Aricescu, A.R. Structural basis for extracellular cis and trans rptpsigma signal competition in synaptogenesis. Nat. Commun. 2014, 5, 5209. [Google Scholar] [CrossRef] [PubMed]
- Coles, C.H.; Shen, Y.; Tenney, A.P.; Siebold, C.; Sutton, G.C.; Lu, W.; Gallagher, J.T.; Jones, E.Y.; Flanagan, J.G.; Aricescu, A.R. Proteoglycan-specific molecular switch for rptpsigma clustering and neuronal extension. Science 2011, 332, 484–488. [Google Scholar] [CrossRef] [PubMed]
- Johnson, K.G.; Tenney, A.P.; Ghose, A.; Duckworth, A.M.; Higashi, M.E.; Parfitt, K.; Marcu, O.; Heslip, T.R.; Marsh, J.L.; Schwarz, T.L.; et al. The hspgs syndecan and dallylike bind the receptor phosphatase lar and exert distinct effects on synaptic development. Neuron 2006, 49, 517–531. [Google Scholar] [CrossRef] [PubMed]
- Thura, M.; Al-Aidaroos, A.Q.; Yong, W.P.; Kono, K.; Gupta, A.; Lin, Y.B.; Mimura, K.; Thiery, J.P.; Goh, B.C.; Tan, P.; et al. Prl3-zumab, a first-in-class humanized antibody for cancer therapy. JCI Insight 2016, 1, e87607. [Google Scholar] [CrossRef] [PubMed]
- Guo, K.; Tang, J.P.; Jie, L.; Al-Aidaroos, A.Q.; Hong, C.W.; Tan, C.P.; Park, J.E.; Varghese, L.; Feng, Z.; Zhou, J.; et al. Engineering the first chimeric antibody in targeting intracellular prl-3 oncoprotein for cancer therapy in mice. Oncotarget 2012, 3, 158–171. [Google Scholar] [CrossRef] [PubMed]
- Guo, K.; Li, J.; Tang, J.P.; Tan, C.P.; Hong, C.W.; Al-Aidaroos, A.Q.; Varghese, L.; Huang, C.; Zeng, Q. Targeting intracellular oncoproteins with antibody therapy or vaccination. Sci. Transl. Med. 2011, 3, 99ra85. [Google Scholar] [CrossRef] [PubMed]
- Doody, K.M.; Stanford, S.M.; Sacchetti, C.; Svensson, M.N.; Coles, C.H.; Mitakidis, N.; Kiosses, W.B.; Bartok, B.; Fos, C.; Cory, E.; et al. Targeting phosphatase-dependent proteoglycan switch for rheumatoid arthritis therapy. Sci. Transl. Med. 2015, 7, 288ra276. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, T.; Takahashi, K.; Mernaugh, R.L.; Tsuboi, N.; Liu, H.; Daniel, T.O. A monoclonal antibody against cd148, a receptor-like tyrosine phosphatase, inhibits endothelial-cell growth and angiogenesis. Blood 2006, 108, 1234–1242. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ortuso, F.; Paduano, F.; Carotenuto, A.; Gomez-Monterrey, I.; Bilotta, A.; Gaudio, E.; Sala, M.; Artese, A.; Vernieri, E.; Dattilo, V.; et al. Discovery of ptprj agonist peptides that effectively inhibit in vitro cancer cell proliferation and tube formation. ACS Chem. Biol. 2013, 8, 1497–1506. [Google Scholar] [CrossRef] [PubMed]
- Van der Wijk, T.; Blanchetot, C.; Overvoorde, J.; den Hertog, J. Redox-regulated rotational coupling of receptor protein-tyrosine phosphatase alpha dimers. J. Biol. Chem. 2003, 278, 13968–13974. [Google Scholar] [CrossRef] [PubMed]
- Haque, A.; Andersen, J.N.; Salmeen, A.; Barford, D.; Tonks, N.K. Conformation-sensing antibodies stabilize the oxidized form of ptp1b and inhibit its phosphatase activity. Cell 2011, 147, 185–198. [Google Scholar] [CrossRef] [PubMed]
- Sha, F.; Gencer, E.B.; Georgeon, S.; Koide, A.; Yasui, N.; Koide, S.; Hantschel, O. Dissection of the bcr-abl signaling network using highly specific monobody inhibitors to the shp2 sh2 domains. Proc. Natl. Acad. Sci. USA 2013, 110, 14924–14929. [Google Scholar] [CrossRef] [PubMed]
- Tiganis, T. Ptp1b and tcptp—Nonredundant phosphatases in insulin signaling and glucose homeostasis. FEBS J. 2013, 280, 445–458. [Google Scholar] [CrossRef] [PubMed]
- Liao, H.; Pei, D. Cell-permeable bicyclic peptidyl inhibitors against t-cell protein tyrosine phosphatase from a combinatorial library. Org. Biomol. Chem. 2017, 15, 9595–9598. [Google Scholar] [CrossRef] [PubMed]
- Lian, W.; Jiang, B.; Qian, Z.; Pei, D. Cell-permeable bicyclic peptide inhibitors against intracellular proteins. J. Am. Chem. Soc. 2014, 136, 9830–9833. [Google Scholar] [CrossRef] [PubMed]
- Xie, Y.; Massa, S.M.; Ensslen-Craig, S.E.; Major, D.L.; Yang, T.; Tisi, M.A.; Derevyanny, V.D.; Runge, W.O.; Mehta, B.P.; Moore, L.A.; et al. Protein-tyrosine phosphatase (ptp) wedge domain peptides: A novel approach for inhibition of ptp function and augmentation of protein-tyrosine kinase function. J. Biol. Chem. 2006, 281, 16482–16492. [Google Scholar] [CrossRef] [PubMed]
- Lang, B.T.; Cregg, J.M.; DePaul, M.A.; Tran, A.P.; Xu, K.; Dyck, S.M.; Madalena, K.M.; Brown, B.P.; Weng, Y.L.; Li, S.; et al. Modulation of the proteoglycan receptor ptpsigma promotes recovery after spinal cord injury. Nature 2015, 518, 404–408. [Google Scholar] [CrossRef] [PubMed]
- Gardner, R.T.; Wang, L.; Lang, B.T.; Cregg, J.M.; Dunbar, C.L.; Woodward, W.R.; Silver, J.; Ripplinger, C.M.; Habecker, B.A. Targeting protein tyrosine phosphatase sigma after myocardial infarction restores cardiac sympathetic innervation and prevents arrhythmias. Nat. Commun. 2015, 6, 6235. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Wong, C.; Li, W.; Ruven, C.; He, L.; Wu, X.; Lang, B.T.; Silver, J.; Wu, W. Enhanced regeneration and functional recovery after spinal root avulsion by manipulation of the proteoglycan receptor ptpsigma. Sci. Rep. 2015, 5, 14923. [Google Scholar] [CrossRef] [PubMed]
- Barnea, M.; Olender, T.; Bedford, M.T.; Elson, A. Regulation of receptor-type protein tyrosine phosphatases by their c-terminal tail domains. Biochem. Soc. Trans. 2016, 44, 1295–1303. [Google Scholar] [CrossRef] [PubMed]
- Feldhammer, M.; Uetani, N.; Miranda-Saavedra, D.; Tremblay, M.L. Ptp1b: A simple enzyme for a complex world. Crit. Rev. Biochem. Mol. Biol. 2013, 48, 430–445. [Google Scholar] [CrossRef] [PubMed]
- Ye, F.; Zhang, M. Structures and target recognition modes of pdz domains: Recurring themes and emerging pictures. Biochem. J. 2013, 455, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Valiente, M.; Andres-Pons, A.; Gomar, B.; Torres, J.; Gil, A.; Tapparel, C.; Antonarakis, S.E.; Pulido, R. Binding of pten to specific pdz domains contributes to pten protein stability and phosphorylation by microtubule-associated serine/threonine kinases. J. Biol. Chem. 2005, 280, 28936–28943. [Google Scholar] [CrossRef] [PubMed]
- Stupp, R.; Mason, W.P.; van den Bent, M.J.; Weller, M.; Fisher, B.; Taphoorn, M.J.; Belanger, K.; Brandes, A.A.; Marosi, C.; Bogdahn, U.; et al. Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N. Engl. J. Med. 2005, 352, 987–996. [Google Scholar] [CrossRef] [PubMed]
- Navis, A.C.; van den Eijnden, M.; Schepens, J.T.; Hooft van Huijsduijnen, R.; Wesseling, P.; Hendriks, W.J. Protein tyrosine phosphatases in glioma biology. Acta Neuropathol. 2010, 119, 157–175. [Google Scholar] [CrossRef] [PubMed]
- Bourgonje, A.M.; Verrijp, K.; Schepens, J.T.; Navis, A.C.; Piepers, J.A.; Palmen, C.B.; van den Eijnden, M.; Hooft van Huijsduijnen, R.; Wesseling, P.; Leenders, W.P.; et al. Comprehensive protein tyrosine phosphatase mrna profiling identifies new regulators in the progression of glioma. Acta Neuropathol. Commun. 2016, 4, 96. [Google Scholar] [CrossRef] [PubMed]
- Sotelo, N.S.; Schepens, J.T.G.; Valiente, M.; Hendriks, W.; Pulido, R. Pten-pdz domain interactions: Binding of pten to pdz domains of ptpn13. Methods 2015, 77–78, 147–156. [Google Scholar] [CrossRef] [PubMed]
- Barnea, G.; Grumet, M.; Milev, P.; Silvennoinen, O.; Levy, J.B.; Sap, J.; Schlessinger, J. Receptor tyrosine phosphatase beta is expressed in the form of proteoglycan and binds to the extracellular matrix protein tenascin. J. Biol. Chem. 1994, 269, 14349–14352. [Google Scholar] [PubMed]
- Bourgonje, A.M.; Navis, A.C.; Schepens, J.T.; Verrijp, K.; Hovestad, L.; Hilhorst, R.; Harroch, S.; Wesseling, P.; Leenders, W.P.; Hendriks, W.J. Intracellular and extracellular domains of protein tyrosine phosphatase ptprz-b differentially regulate glioma cell growth and motility. Oncotarget 2014, 5, 8690–8702. [Google Scholar] [CrossRef] [PubMed]
- Foehr, E.D.; Lorente, G.; Kuo, J.; Ram, R.; Nikolich, K.; Urfer, R. Targeting of the receptor protein tyrosine phosphatase beta with a monoclonal antibody delays tumor growth in a glioblastoma model. Cancer Res. 2006, 66, 2271–2278. [Google Scholar] [CrossRef] [PubMed]
- Fujikawa, A.; Nagahira, A.; Sugawara, H.; Ishii, K.; Imajo, S.; Matsumoto, M.; Kuboyama, K.; Suzuki, R.; Tanga, N.; Noda, M.; et al. Small-molecule inhibition of ptprz reduces tumor growth in a rat model of glioblastoma. Sci. Rep. 2016, 6, 20473. [Google Scholar] [CrossRef] [PubMed]
- Rampling, R.; Peoples, S.; Mulholland, P.J.; James, A.; Al-Salihi, O.; Twelves, C.J.; McBain, C.; Jefferies, S.; Jackson, A.; Stewart, W.; et al. A cancer research uk first time in human phase i trial of ima950 (novel multipeptide therapeutic vaccine) in patients with newly diagnosed glioblastoma. Clin. Cancer Res.: Off. J. Am. Assoc. Cancer Res. 2016, 22, 4776–4785. [Google Scholar] [CrossRef] [PubMed]
- Kawachi, H.; Tamura, H.; Watakabe, I.; Shintani, T.; Maeda, N.; Noda, M. Protein tyrosine phosphatase zeta/rptpbeta interacts with psd-95/sap90 family. Brain Res. Mol. Brain Res. 1999, 72, 47–54. [Google Scholar] [CrossRef]
- Appert-Collin, A.; Hubert, P.; Cremel, G.; Bennasroune, A. Role of erbb receptors in cancer cell migration and invasion. Front. Pharmacol. 2015, 6, 283. [Google Scholar] [CrossRef] [PubMed]
- Fujikawa, A.; Chow, J.P.; Shimizu, H.; Fukada, M.; Suzuki, R.; Noda, M. Tyrosine phosphorylation of erbb4 is enhanced by psd95 and repressed by protein tyrosine phosphatase receptor type z. J. Biochem. 2007, 142, 343–350. [Google Scholar] [CrossRef] [PubMed]
- Iwakura, Y.; Nawa, H. Erbb1-4-dependent egf/neuregulin signals and their cross talk in the central nervous system: Pathological implications in schizophrenia and parkinson's disease. Front. Cell. Neurosci. 2013, 7, 4. [Google Scholar] [CrossRef] [PubMed]
- Bach, A.; Clausen, B.H.; Moller, M.; Vestergaard, B.; Chi, C.N.; Round, A.; Sorensen, P.L.; Nissen, K.B.; Kastrup, J.S.; Gajhede, M.; et al. A high-affinity, dimeric inhibitor of psd-95 bivalently interacts with pdz1–2 and protects against ischemic brain damage. Proc. Natl. Acad. Sci. USA 2012, 109, 3317–3322. [Google Scholar] [CrossRef] [PubMed]
- Buxbaum, J.D.; Georgieva, L.; Young, J.J.; Plescia, C.; Kajiwara, Y.; Jiang, Y.; Moskvina, V.; Norton, N.; Peirce, T.; Williams, H.; et al. Molecular dissection of nrg1-erbb4 signaling implicates ptprz1 as a potential schizophrenia susceptibility gene. Mol. Psychiatry 2008, 13, 162–172. [Google Scholar] [CrossRef] [PubMed]
- Fujikawa, A.; Fukada, M.; Makioka, Y.; Suzuki, R.; Chow, J.P.; Matsumoto, M.; Noda, M. Consensus substrate sequence for protein-tyrosine phosphatase receptor type z. J. Biol. Chem. 2011, 286, 37137–37146. [Google Scholar] [CrossRef] [PubMed]
- Adamsky, K.; Arnold, K.; Sabanay, H.; Peles, E. Junctional protein magi-3 interacts with receptor tyrosine phosphatase beta (rptp beta) and tyrosine-phosphorylated proteins. J. Cell Sci. 2003, 116, 1279–1289. [Google Scholar] [CrossRef] [PubMed]
- Dobrosotskaya, I.Y.; James, G.L. Magi-1 interacts with beta-catenin and is associated with cell-cell adhesion structures. Biochem. Biophys. Res. Commun. 2000, 270, 903–909. [Google Scholar] [CrossRef] [PubMed]
- MacDonald, B.T.; Tamai, K.; He, X. Wnt/beta-catenin signaling: Components, mechanisms, and diseases. Dev. Cell 2009, 17, 9–26. [Google Scholar] [CrossRef] [PubMed]
- Fukada, M.; Kawachi, H.; Fujikawa, A.; Noda, M. Yeast substrate-trapping system for isolating substrates of protein tyrosine phosphatases: Isolation of substrates for protein tyrosine phosphatase receptor type z. Methods 2005, 35, 54–63. [Google Scholar] [CrossRef] [PubMed]
- Freiss, G.; Chalbos, D. Ptpn13/ptpl1: An important regulator of tumor aggressiveness. Anti-Cancer Agents Med. Chem. 2011, 11, 78–88. [Google Scholar] [CrossRef]
- Sato, T.; Irie, S.; Kitada, S.; Reed, J.C. Fap-1: A protein tyrosine phosphatase that associates with fas. Science 1995, 268, 411–415. [Google Scholar] [CrossRef] [PubMed]
- Gump, J.M.; Staskiewicz, L.; Morgan, M.J.; Bamberg, A.; Riches, D.W.; Thorburn, A. Autophagy variation within a cell population determines cell fate through selective degradation of fap-1. Nat. Cell Biol. 2014, 16, 47–54. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.; Luan, C.H.; Hjort, E.E.; Bei, L.; Mishra, R.; Sakamoto, K.M.; Platanias, L.C.; Eklund, E.A. The role of fas-associated phosphatase 1 in leukemia stem cell persistence during tyrosine kinase inhibitor treatment of chronic myeloid leukemia. Leukemia 2016, 30, 1502–1509. [Google Scholar] [CrossRef] [PubMed]
- Revillion, F.; Puech, C.; Rabenoelina, F.; Chalbos, D.; Peyrat, J.P.; Freiss, G. Expression of the putative tumor suppressor gene ptpn13/ptpl1 is an independent prognostic marker for overall survival in breast cancer. Int. J. Cancer 2009, 124, 638–643. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Shen, D.; Parsons, D.W.; Bardelli, A.; Sager, J.; Szabo, S.; Ptak, J.; Silliman, N.; Peters, B.A.; van der Heijden, M.S.; et al. Mutational analysis of the tyrosine phosphatome in colorectal cancers. Science 2004, 304, 1164–1166. [Google Scholar] [CrossRef] [PubMed]
- Hoover, A.C.; Strand, G.L.; Nowicki, P.N.; Anderson, M.E.; Vermeer, P.D.; Klingelhutz, A.J.; Bossler, A.D.; Pottala, J.V.; Hendriks, W.J.; Lee, J.H. Impaired ptpn13 phosphatase activity in spontaneous or hpv-induced squamous cell carcinomas potentiates oncogene signaling through the map kinase pathway. Oncogene 2009, 28, 3960–3970. [Google Scholar] [CrossRef] [PubMed]
- Abaan, O.D.; Levenson, A.; Khan, O.; Furth, P.A.; Uren, A.; Toretsky, J.A. Ptpl1 is a direct transcriptional target of ews-fli1 and modulates ewing's sarcoma tumorigenesis. Oncogene 2005, 24, 2715–2722. [Google Scholar] [CrossRef] [PubMed]
- Herrmann, L.; Dittmar, T.; Erdmann, K.S. The protein tyrosine phosphatase ptp-bl associates with the midbody and is involved in the regulation of cytokinesis. Mol. Biol. Cell 2003, 14, 230–240. [Google Scholar] [CrossRef] [PubMed]
- Louis, D.N.; Perry, A.; Reifenberger, G.; von Deimling, A.; Figarella-Branger, D.; Cavenee, W.K.; Ohgaki, H.; Wiestler, O.D.; Kleihues, P.; Ellison, D.W. The 2016 world health organization classification of tumors of the central nervous system: A summary. Acta Neuropathol. 2016, 131, 803–820. [Google Scholar] [CrossRef] [PubMed]
- Palma, L.; Celli, P.; Maleci, A.; Di Lorenzo, N.; Cantore, G. Malignant monstrocellular brain tumours. A study of 42 surgically treated cases. Acta neurochir. 1989, 97, 17–25. [Google Scholar] [CrossRef] [PubMed]
- Srivas, R.; Shen, J.P.; Yang, C.C.; Sun, S.M.; Li, J.; Gross, A.M.; Jensen, J.; Licon, K.; Bojorquez-Gomez, A.; Klepper, K.; et al. A network of conserved synthetic lethal interactions for exploration of precision cancer therapy. Mol. Cell 2016, 63, 514–525. [Google Scholar] [CrossRef] [PubMed]
- Matsuda, S.; Kitagishi, Y. Magi scaffolding molecules involved in cancer cell signaling. J. Carcinog. Mutagen. 2013, S7, 005. [Google Scholar]
- Shi, Y.; Ping, Y.F.; Zhou, W.; He, Z.C.; Chen, C.; Bian, B.S.; Zhang, L.; Chen, L.; Lan, X.; Zhang, X.C.; et al. Tumour-associated macrophages secrete pleiotrophin to promote ptprz1 signalling in glioblastoma stem cells for tumour growth. Nat. Commun. 2017, 8, 15080. [Google Scholar] [CrossRef] [PubMed]
- Kegelman, T.P.; Wu, B.; Das, S.K.; Talukdar, S.; Beckta, J.M.; Hu, B.; Emdad, L.; Valerie, K.; Sarkar, D.; Furnari, F.B.; et al. Inhibition of radiation-induced glioblastoma invasion by genetic and pharmacological targeting of mda-9/syntenin. Proc. Natl. Acad. Sci. USA 2017, 114, 370–375. [Google Scholar] [CrossRef] [PubMed]
- Spanos, W.C.; Hoover, A.; Harris, G.F.; Wu, S.; Strand, G.L.; Anderson, M.E.; Klingelhutz, A.J.; Hendriks, W.; Bossler, A.D.; Lee, J.H. The pdz binding motif of human papillomavirus type 16 e6 induces ptpn13 loss, which allows anchorage-independent growth and synergizes with ras for invasive growth. J. Virol. 2008, 82, 2493–2500. [Google Scholar] [CrossRef] [PubMed]
- Trave, G. Pdz-peptide complexes: As exciting as ever. Structure 2011, 19, 1350–1351. [Google Scholar] [CrossRef] [PubMed]

© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hendriks, W.; Bourgonje, A.; Leenders, W.; Pulido, R. Proteinaceous Regulators and Inhibitors of Protein Tyrosine Phosphatases. Molecules 2018, 23, 395. https://doi.org/10.3390/molecules23020395
Hendriks W, Bourgonje A, Leenders W, Pulido R. Proteinaceous Regulators and Inhibitors of Protein Tyrosine Phosphatases. Molecules. 2018; 23(2):395. https://doi.org/10.3390/molecules23020395
Chicago/Turabian StyleHendriks, Wiljan, Annika Bourgonje, William Leenders, and Rafael Pulido. 2018. "Proteinaceous Regulators and Inhibitors of Protein Tyrosine Phosphatases" Molecules 23, no. 2: 395. https://doi.org/10.3390/molecules23020395
APA StyleHendriks, W., Bourgonje, A., Leenders, W., & Pulido, R. (2018). Proteinaceous Regulators and Inhibitors of Protein Tyrosine Phosphatases. Molecules, 23(2), 395. https://doi.org/10.3390/molecules23020395





