HPLC-PDA Combined with Chemometrics for Quantitation of Active Components and Quality Assessment of Raw and Processed Fruits of Xanthium strumarium L.
Abstract
:1. Introduction
2. Results and Discussion
2.1. Optimization of the Extraction Method
2.2. Optimization of Chromatographic Conditions
2.3. Method Validation
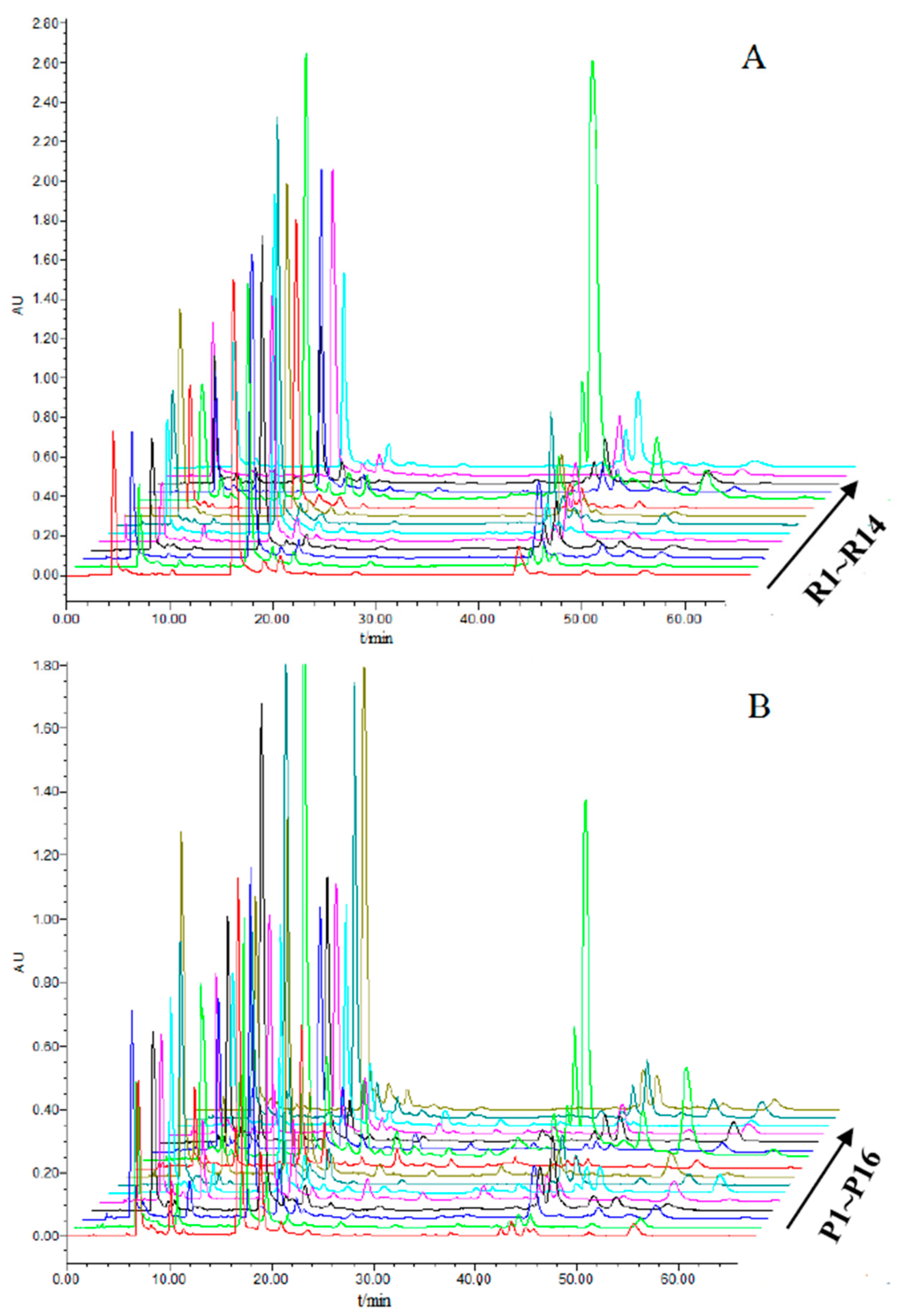
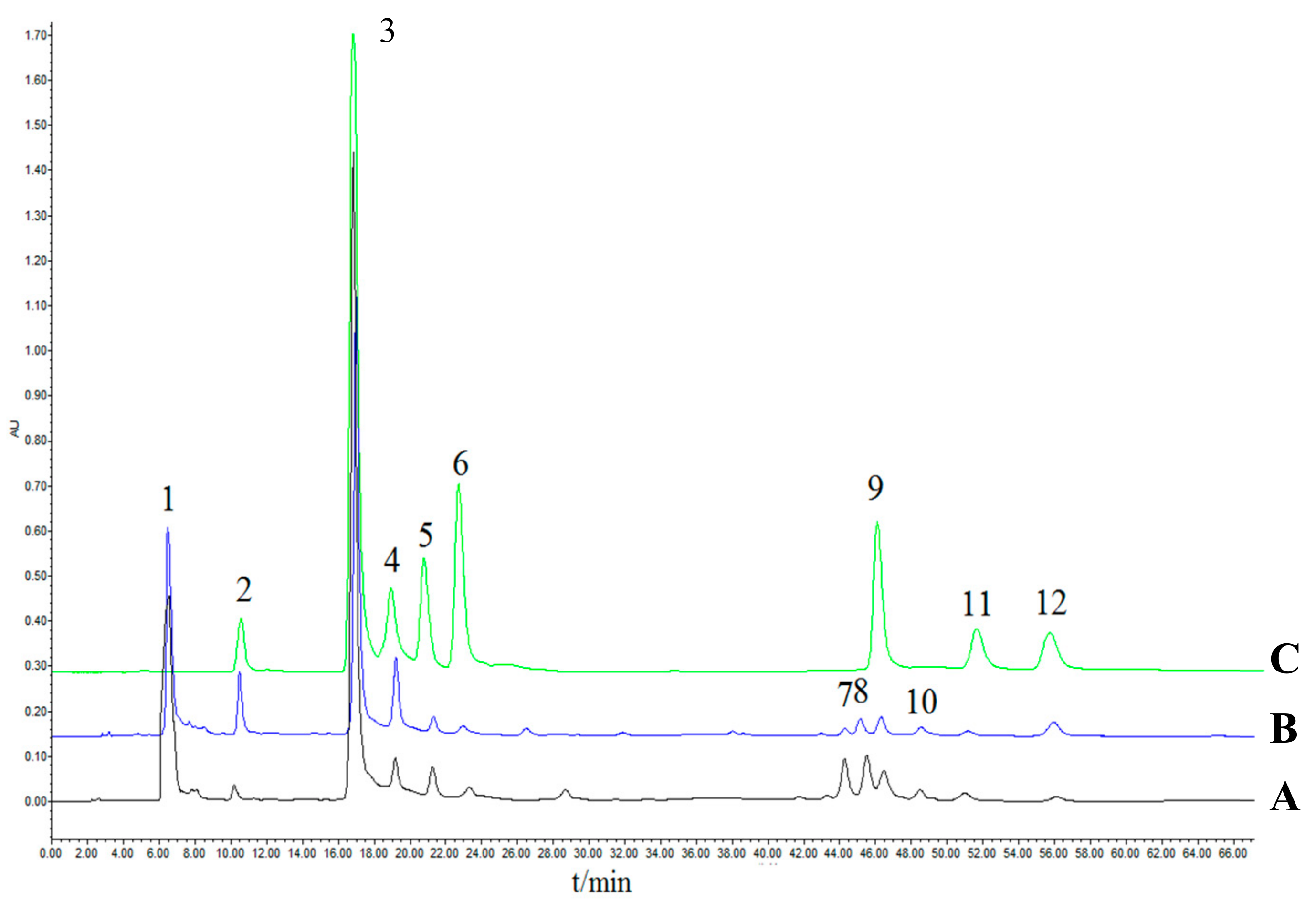
2.4. High-Performance Liquid Chromatography (HPLC) Fingerprints and Quantification of the Eight Active Components
2.4.1. Establishment of HPLC Fingerprints of Raw Xanthii Fructus (RXF) and Processed XF (PXF)
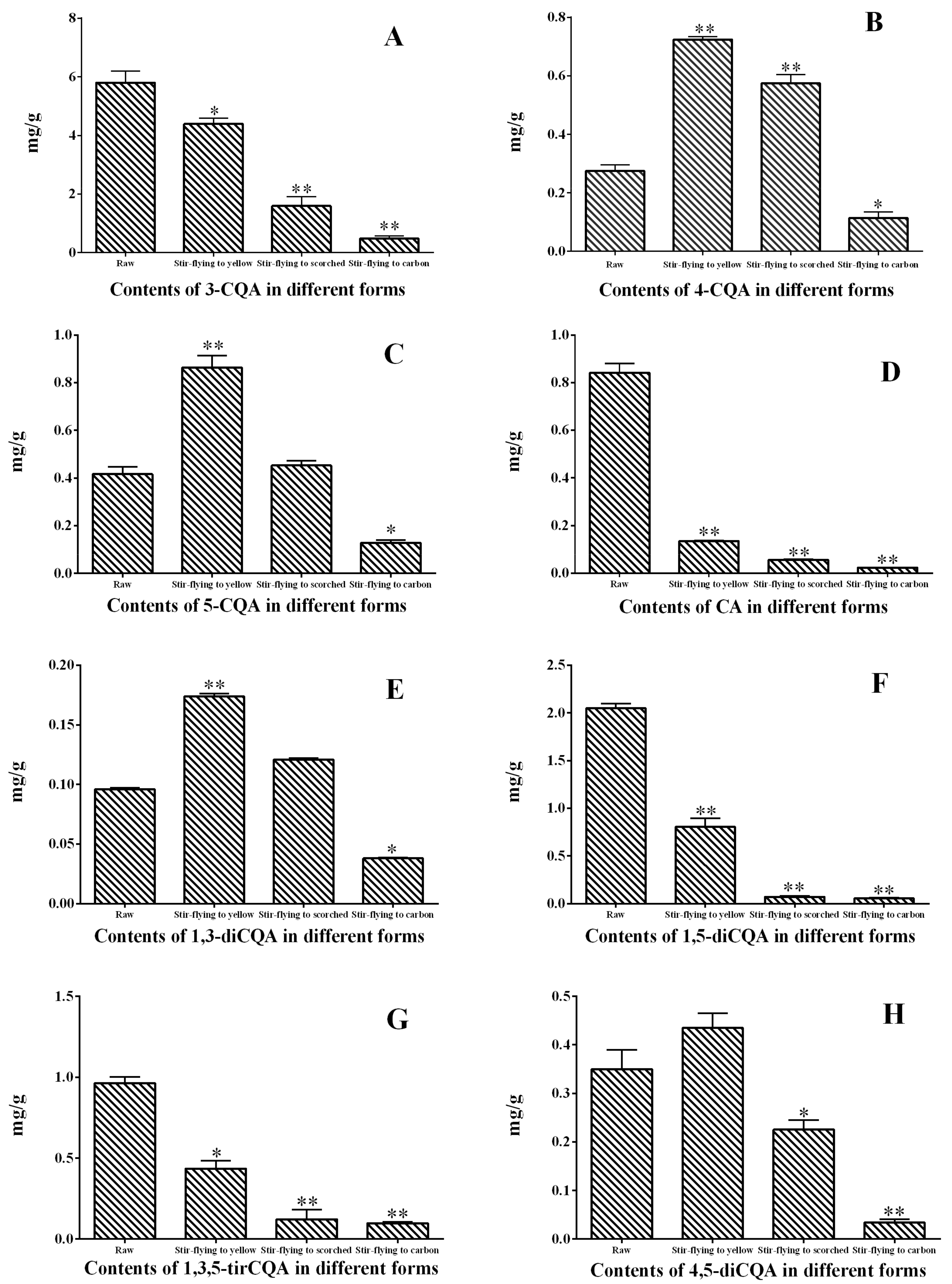
2.4.2. Quantification of Eight Caffeoylquinic Acids in Different Samples
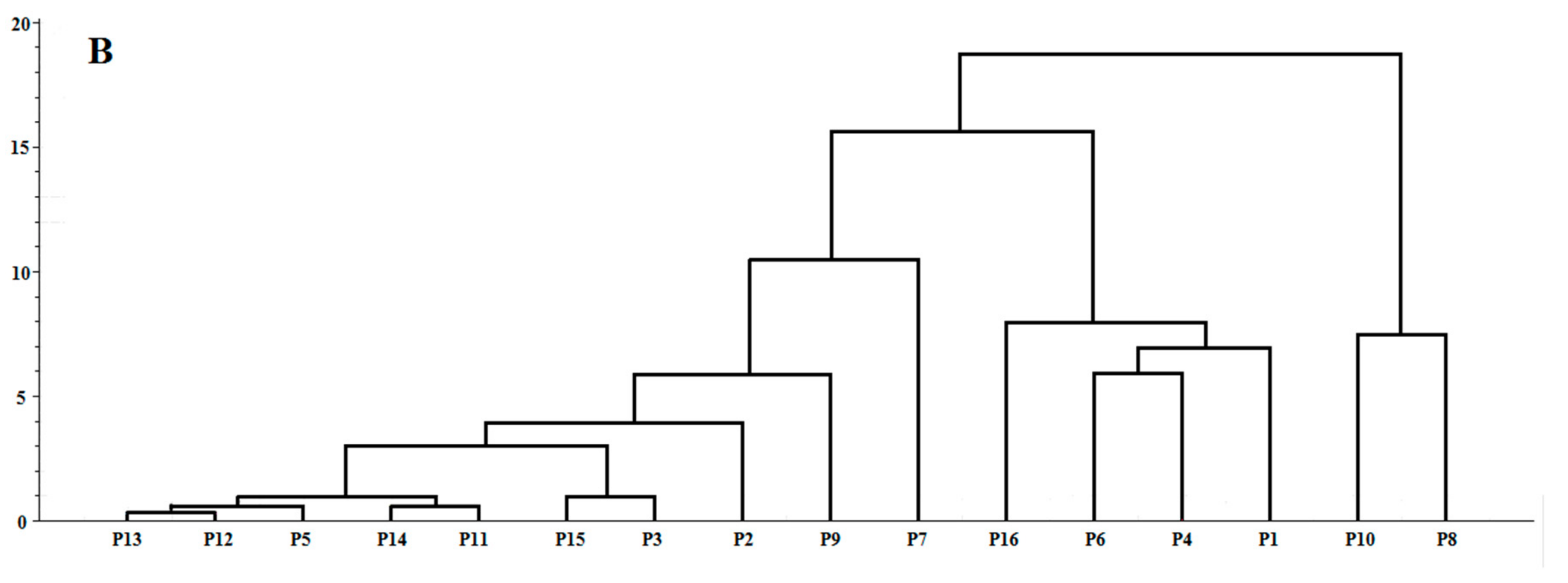
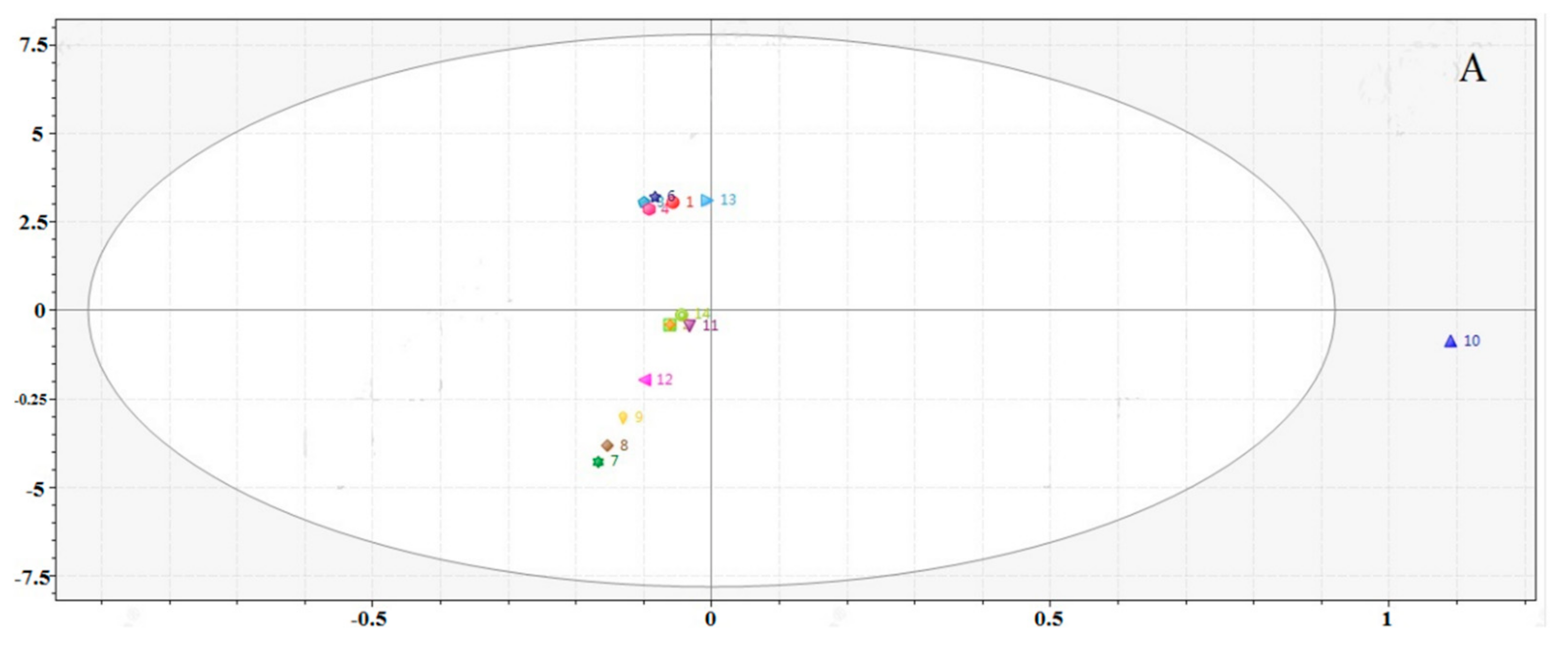
2.5. Chemometric Analysis
2.5.1. Quality Evaluation by Hierarchical Cluster Analysis (HCA)
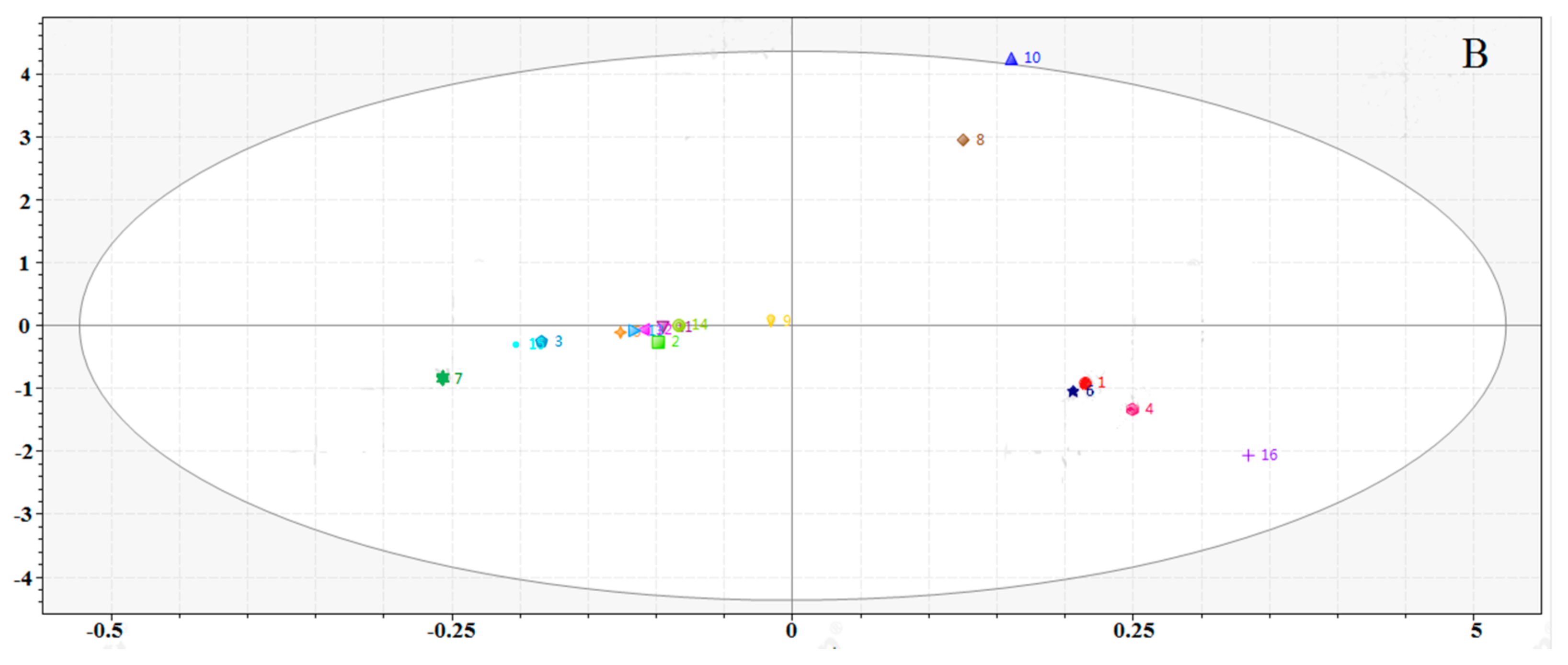
2.5.2. Quality Evaluation by Principal Component Analysis (PCA)
2.6. Comparison between RXF and PXF
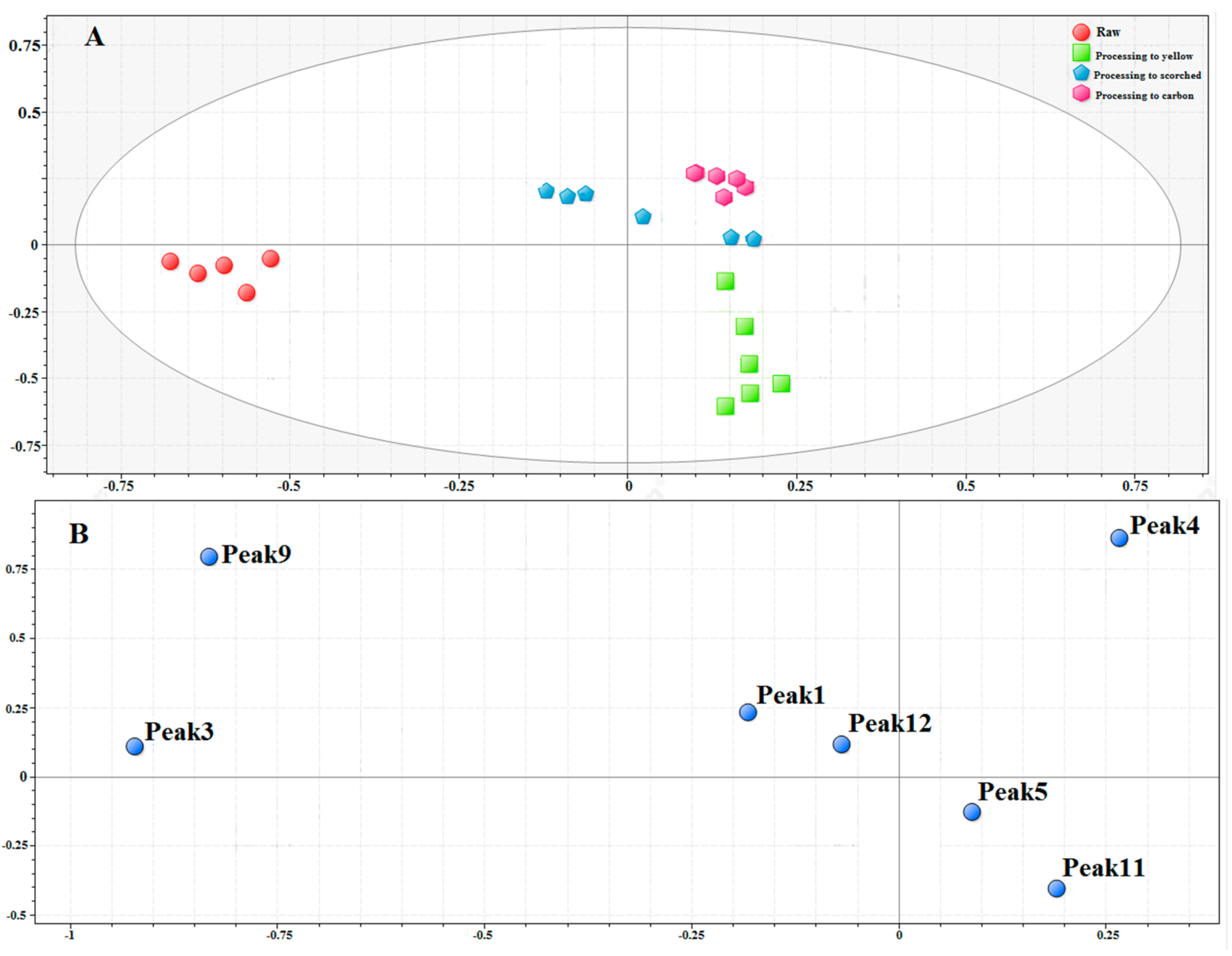
2.6.1. Comparison of Raw and Different Processed Products by Chemometric Analysis
2.6.2. Quality Assessment of RXF and PXF
3. Materials and Methods
3.1. Chemicals and Reagents
3.2. Plant Materials and Sample Preparation
3.2.1. Plant Materials
3.2.2. Sample Preparation
3.3. Standard Solution Preparation
3.4. Chromatographic Conditions
3.5. Method Validation
3.6. Comparison between RXF and Different Processed XF Samples
3.7. Data Analysis
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Xie, P.S.; Leung, A.Y. Understanding the traditional aspect of Chinese medicine in order to achieve meaningful quality control of Chinese materia medica. J. Chromatogr. A 2009, 1216, 1933–1940. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Z.; Liang, Z.; Chan, K.; Lu, G.; Lee, E.L.M.; Chen, H.; Li, L. A unique issue in the standardization of Chinese materia medica: Processing. Planta Med. 2010, 76, 1975–1986. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Liao, P.Y.; Zhu, H.T.; Chen, K.K.; Xu, M.; Zhang, Y.J.; Yang, C.R. The processing of Panax notoginseng and the transformation of its saponin components. Food Chem. 2012, 132, 1808–1813. [Google Scholar] [CrossRef]
- Wang, L.; Zhou, G.B.; Liu, P.; Song, J.H.; Liang, Y.; Yan, X.J.; Chen, S.J. Dissection of mechanisms of Chinese medicinal formula Realgar-Indigo naturalis as an effective treatment for promyelocytic leukemia. Proc. Natl. Acad. Sci. USA 2008, 105, 4826–4831. [Google Scholar] [CrossRef] [PubMed]
- Yan, S.K.; Xin, W.F.; Luo, G.A.; Wang, Y.M.; Cheng, Y.Y. An approach to develop two-dimensional fingerprint for the quality control of Qingkailing injection by high-performance liquid chromatography with diode array detection. J. Chromatogr. A 2005, 1090, 90–97. [Google Scholar] [CrossRef] [PubMed]
- Qin, K.; Wang, B.; Li, W.; Cai, H.; Chen, D.; Liu, X.; Cai, B. Quality assessment of raw and processed Arctium lappa L. through multicomponent quantification, chromatographic fingerprint, and related chemometric analysis. J. Sep. Sci. 2015, 38, 1491–1498. [Google Scholar] [CrossRef] [PubMed]
- Tao, Y.; Du, Y.; Su, D.; Li, W.; Cai, B. UHPLC-MS/MS quantification combined with chemometrics for the comparative analysis of different batches of raw and wine-processed Dipsacus asper. J. Sep. Sci. 2017, 40, 1686–1693. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Li, L.; Xiao, Y.Q.; Yao, J.Q.; Li, P.Y.; Yu, D.R.; Ma, Y.L. Global metabolite profiling and diagnostic ion filtering strategy by LC–QTOF MS for rapid identification of raw and processed pieces of Rheum palmatum L. Food Chem. 2016, 192, 531–540. [Google Scholar] [CrossRef] [PubMed]
- Chinese Pharmacopoeia Commission; Pharmacopoeia of Peoploe’s Republic of China Chemical Industry Press: Beijing, China, 2015.
- Dai, Y.H.; Cui, Z.; Li, J.L. Research progress in the chemical constituents and pharmacological activities of Xanthium L. J. Shenyang Pharm. Univ. 2007, 12, 016. [Google Scholar]
- Yang, M.; Zhang, D.K.; Zhong, L.Y.; Wang, F. Study on culture and philosophy of processing of traditional Chinese medicines. Chin. Med. Mater. Med. 2013, 38, 2223–2226. [Google Scholar]
- Han, T.; Li, H.L.; Zhang, Q.Y.; Han, P.; Zheng, H.C.; Rahman, K.; Qin, L.P. Bioactivity-guided fractionation for anti-inflammatory and analgesic properties and constituents of Xanthium strumarium L. Phytomedicine 2007, 14, 825–829. [Google Scholar] [CrossRef] [PubMed]
- Jiang, H.; Yang, L.; Xing, X.D.; Zhang, Y.Y.; Yan, M.L.; Yang, B.Y.; Wang, Q.H.; Kuang, H.X. Chemical constituents from fruits of Xanthium sibiricum. Chin. Tradit. Herb. Drugs 2017, 48, 47–51. [Google Scholar]
- Jiang, H.; Ma, G.X.; Yang, L.; Xing, X.D.; Yan, M.L.; Zhang, Y.Y.; Xu, X.D. Rearranged ent-kauranoid glycosides from the fruits of Xanthium strumarium and their antiproliferative activity. Phytochem. Lett. 2016, 18, 192–196. [Google Scholar] [CrossRef]
- Jiang, H.; Yang, L.; Liu, C.; Hou, H.; Wang, Q.H.; Wang, Z.; Kuang, H.X. Four new glycosides from the fruit of Xanthium sibiricum Patr. Molecules 2013, 18, 12464–12473. [Google Scholar] [CrossRef] [PubMed]
- Jiang, H.; Yang, L.; Ma, G.X.; Xing, X.D.; Yan, M.L.; Zhang, Y.Y.; Xu, X.D. New phenylpropanoid derivatives from the fruits of Xanthium sibiricum and their anti-inflammatory activity. Fitoterapia 2017, 117, 11–15. [Google Scholar] [CrossRef] [PubMed]
- Jiang, H.; Zhang, Y.Y.; Zhang, Y.; Yang, L.; Wang, Q.H.; Kuang, H.X. Isolation and identification of chemical constituents from the fruit of Patr. Inf. Tradit. Chin. Med. 2016, 33, 8–10. [Google Scholar]
- Rad, J.S.; Alfatemi, S.M.H.; Rad, M.S.; Iriti, M. In-vitro antioxidant and antibacterial activities of Xanthium strumarium L. extracts on methicillin-susceptible and methicillin-resistant Staphylococcus aureus. Anc. Sci. Life 2013, 33, 109–113. [Google Scholar] [PubMed]
- Sroka, Z.; Cisowski, W. Hydrogen peroxide scavenging, antioxidant and anti-radical activity of some phenolic acids. Food Chem. Toxicol. 2003, 41, 753–758. [Google Scholar] [CrossRef]
- Fernández, M.A.; Garcia, M.D.; Saenz, M.T. Antibacterial activity of the phenolic acids fractions of Scrophularia frutescens and Scrophularia sambucifolia. J. Ethnopharmacol. 1996, 53, 11–14. [Google Scholar] [CrossRef]
- Gomes, C.A.; Girão da Cruz, T.; Andrade, J.L.; Milhazes, N.; Borges, F.; Marques, M.P.M. Anticancer activity of phenolic acids of natural or synthetic origin: A structure-activity study. J. Med. Chem. 2003, 46, 5395–5401. [Google Scholar] [CrossRef] [PubMed]
- Breinholt, V. Desirable vs. harmful levels of intake of flavonoids and phenolic acid. Publ. R. Soc. Chem. 1999, 240, 93–105. [Google Scholar]
- Hwang, S.J.; Kim, Y.W.; Park, Y.; Lee, H.J.; Kim, K.W. Anti-inflammatory effects of chlorogenic acid in lipopolysaccharide-stimulated RAW 264.7 cells. Inflamm. Res. 2014, 63, 81–90. [Google Scholar] [CrossRef] [PubMed]
- Chao, C.Y.; Mong, M.C.; Chan, K.C.; Yin, M.C. Anti-glycative and anti-inflammatory effects of caffeic acid and ellagic acid in kidney of diabetic mice. Mol. Nutr. Food Res. 2010, 54, 388–395. [Google Scholar] [CrossRef] [PubMed]
- Han, T.; Zhang, Q.Y.; Zhang, H.; Wen, J.; Wang, Y.; Huang, B.K.; Qin, L.P. Authentication and quantitative analysis on the chemical profile of Xanthium fruit (Cang-Er-Zi) by high-performance liquid chromatography-diode-array detection tandem mass spectrometry method. Anal. Chim. Acta 2009, 634, 272–278. [Google Scholar] [CrossRef] [PubMed]
- Xie, B.; Gong, T.; Tang, M.; Mi, D.; Zhang, X.; Liu, J.; Zhang, Z. An approach based on HPLC-fingerprint and chemometrics to quality consistency evaluation of Liuwei Dihuang Pills produced by different manufacturers. J. Pharm. Biomed. Anal. 2008, 48, 1261–1266. [Google Scholar] [CrossRef] [PubMed]
- García-Salas, P.; Gómez-Caravaca, A.M.; Morales-Soto, A.; Segura-Carretero, A.; Fernández-Gutiérrez, A. Identification and quantification of phenolic compounds in diverse cultivars of eggplant grown in different seasons by high-performance liquid chromatography coupled to diode array detector and electrospray-quadrupole-time of flight-mass spectrometry. Food Res. Int. 2014, 57, 114–122. [Google Scholar] [CrossRef]
- Sahu, P.K.; Rao, R.N.; Cecchi, T.; Swain, S.; Patro, C.S.; Panda, J. An overview of experimental designs in hplc method development and validation. J. Pharm. Biomed. Anal. 2018, 147, 590–611. [Google Scholar] [CrossRef] [PubMed]
- Xie, D.M.; Qin, M.J.; Huang, L.Q. Herbal textural research on species of Xanthii Fructus. Chin. J. Mat. Med. 2015, 40, 1842–1844. [Google Scholar]
- Bridges, C.C., Jr. Hierarchical cluster analysis. Psychol. Rep. 1966, 18, 851–854. [Google Scholar] [CrossRef]
- Chen, X.J.; Ji, H.; Zhang, Q.W.; Tu, P.F.; Wang, Y.T.; Guo, B.L.; Li, S.P. A rapid method for simultaneous determination of 15 flavonoids in Epimedium using pressurized liquid extraction and ultra-performance liquid chromatography. J. Pharm. Biomed. Anal. 2008, 46, 226–235. [Google Scholar] [CrossRef] [PubMed]
- Caliński, T.; Harabasz, J. A dendrite method for cluster analysis. Commun. Stat-Theory Methods 1974, 3, 1–27. [Google Scholar] [CrossRef]
- Bro, R.; Smilde, A.K. Principal component analysis. Anal. Methods 2014, 6, 2812–2831. [Google Scholar] [CrossRef]
- Huang, Y.; Wu, Z.; Su, R.; Ruan, G.; Du, F.; Li, G. Current application of chemometrics in traditional Chinese herbal medicine research. J. Chromatogr. B 2016, 1026, 27–35. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples are available from the authors. |


| Peak | Compound | Linear Regression | Precision Relative Standard Deviation (RSD) (%) | Recovery (%) | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Regressive Equation 1 | Linear Range (μg/mL) | R2 | LOD (μg/mL) | LOQ (μg/mL) | Intra-Day (n = 6) | Inter-Day (n = 3) | Raw XF (RXF) | Processed XF (PXF) | ||
| 2 | 5-CQA | Y = 23991X + 3544.6 | 1.31~131 | 0.9996 | 0.03 | 0.07 | 0.23 | 1.98 | 95.45~104.34 | 96.34~101.72 |
| 3 | 3-CQA | Y = 31325X − 106752 | 10.80~1080 | 0.9995 | 0.20 | 0.68 | 0.12 | 1.80 | 97.45~108.45 | 94.23~101.34 |
| 4 | 4-CQA | Y = 322269X − 54947 | 10.30~215 | 0.9995 | 0.17 | 0.58 | 1.62 | 1.97 | 96.22~103.67 | 95.67~105.21 |
| 5 | CA | Y = 30259X − 54947 | 1.89~224 | 0.9998 | 0.13 | 0.45 | 1.03 | 1.53 | 94.78~102.77 | 95.69~107.23 |
| 6 | 1,3-diCQA | Y = 40113X − 83400 | 1.05~720 | 0.9991 | 0.06 | 0.21 | 1.57 | 1.88 | 99.76~105.38 | 98.23~104.09 |
| 9 | 1,5-diCQA | Y = 23487X − 310575 | 4.99~543 | 0.9995 | 0.02 | 0.07 | 1.75 | 2.07 | 95.46~100.98 | 94.67~100.45 |
| 11 | 1,3,5-tirCQA | Y = 19547X − 1918.3 | 2.34~642 | 0.9994 | 0.01 | 0.04 | 1.63 | 2.11 | 96.24~106.77 | 97.86~107.33 |
| 12 | 4,5-diCQA | Y = 33218X − 60842 | 1.55~234 | 0.9997 | 0.02 | 0.07 | 1.51 | 1.97 | 94.66~103.22 | 95.78~105.44 |
| No. | Region and Location (Latitude, Longitude) | Time of Collection (Specimen No.) | Content of Investigated Components (n = 3, mg/g ± SD) | Total Content (n = 3, mg/g ± SD) | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| CA (5) 1 | 3-CQA (3) | 4-CQA (4) | 5-CQA (2) | 1,3-diCQA (6) | 1,5-diCQA (9) | 1,3,5-tirCQA (11) | 4,5-diCQA (12) | ||||
| R1 | Binxian, Heilongjiang (45°45 N 127°28 E) | September, 2016 (RBH201609-01) | 0.304 ± 0.010 | 7.080 ± 0.021 | 0.522 ± 0.009 | 0.094 ± 0.004 | 0.035 ± 0.003 | 1.250 ± 0.012 | 0.202 ± 0.009 | 0.158 ± 0 .003 | 9.645 ± 0.011 |
| R2 | Tieling, Lioaning (42°13 N 123°50 E) | September, 2016 (RTL201609-02) | 0.168 ± 0.012 | 6.218 ± 0.022 | 0.502 ± 0.012 | 0.070 ± 0.011 | 0.089 ± 0.002 | 0.567 ± 0.009 | 0.346 ± 0.010 | 0.110 ± 0.004 | 8.070 ± 0.110 |
| R3 | Baotou, Inner Mongolia (40°39 N 109°50 E) | September, 2016 (RBI201609-03) | 0.278 ± 0.013 | 6.800 ± 0.020 | 0.340 ± 0.013 | 0.088 ± 0.008 | 0.383 ± 0.004 | 3.512 ± 0.010 | 0.880 ± 0.008 | 0.274 ± 0.005 | 12.555 ± 0.123 * |
| R4 | Baoding, Hebei (38°52 N 115°27 E) | October, 2016 (RBH201610-04) | 0.288 ± 0.011 | 6.114 ± 0.022 | 0.364 ± 0.011 | 0.070 ± 0.013 | 0.048 ± 0.001 | 0.990 ± 0.006 | 0.677 ± 0.010 | 0.220 ± 0.003 | 8.771 ± 0.015 |
| R5 | Jining, Shandong (35°24 N 116°34 E) | September, 2016 (RJS201609-05) | 0.176 ± 0.010 | 5.804 ± 0.019 | 0.604 ± 0.012 | 0.202 ± 0.011 | 0.042 ± 0.003 | 3.214 ± 0.011 | 0.258 ± 0.009 | 0.101 ± 0.003 | 10.401 ± 0.009 |
| R6 | Hanzhong, Shanxi (33°04 N 107°01 E) | September, 2016 (RHS201609-06) | 0.288 ± 0.009 | 7.634 ± 0.020 | 0.676 ± 0.011 | 0.094 ± 0.012 | 0.128 ± 0.004 | 0.378 ± 0.007 | 0.196 ± 0.007 | 0.114 ± 0.004 | 9.508 ± 0.010 |
| R7 | Zhumadian, Henan (33°00 N 114°01 E) | October, 2016 (RZH201610-07) | 0.418 ± 0.008 | 9.396 ± 0.020 | 0.892 ± 0.010 | 0.121 ± 0.010 | 0.669 ± 0.002 | 1.059 ± 0.009 | 0.238 ± 0.006 | 0.474 ± 0.003 | 13.267 ± 0.008 * |
| R8 | Suqian, Jiangsu (33°57 N 118°16 E) | October, 2016 (RSJ201610-08) | 0.378 ± 0.009 | 7.570 ± 0.023 | 0.758 ± 0.010 | 0.050 ± 0.011 | 0.068 ± 0.005 | 0.186 ± 0.008 | 0.038 ± 0.006 | 0.225 ± 0.006 | 9.273 ± 0.010 |
| R9 | Liuan, Anhui (31°44 N 116°31 E) | September, 2016 (RLA201609-09) | 0.262 ± 0.010 | 6.658 ± 0.024 | 0.582 ± 0.012 | 0.536 ± 0.012 | 0.068 ± 0.003 | 0.735 ± 0.009 | 0.360 ± 0.004 | 0.109 ± 0.003 | 9.310 ± 0.009 |
| R10 | Dazhou, Sichuan (31°12 N 107°27 E) | October, 2016 (RDS201610-10) | 0.896 ± 0.015 | 10.65 ± 0.021 | 0.456 ± 0.016 | 0.590 ± 0.014 | 6.257 ± 0.014 | 3.098 ± 0.023 | 5.409 ± 0.019 | 1.702 ± 0.010 | 29.058 ± 0.018 ** |
| R11 | Huanggang, Hubei (30°27 N 114°52 E) | September, 2016 (RHH201609-11) | 0.290 ± 0.011 | 6.678 ± 0.021 | 0.380 ± 0.018 | 0.070 ± 0.010 | 0.018 ± 0.003 | 0.870 ± 0.009 | 0.279 ± 0.003 | 0.226 ± 0.006 | 8.811 ± 0.010* |
| R12 | Shaoyang, Hunan (27°14 N 111°27 E) | October, 2016 (RSH201610-12) | 0.670 ± 0.010 | 9.396 ± 0.013 | 0.596 ± 0.012 | 0.340 ± 0.019 | 0.049 ± 0.007 | 1.667 ± 0.012 | 0.148 ± 0.011 | 0.608 ± 0.013 | 13.474 ± 0.013 * |
| R13 | Guilin, Guangxi (25°16 N 110°17 E) | October, 2016 (RGG201610-13) | 0.374 ± 0.016 | 7.616 ± 0.017 | 0.532 ± 0.013 | 0.108 ± 0.004 | 0.139 ± 0.002 | 2.811 ± 0.011 | 0.537 ± 0.009 | 0.301 ± 0.005 | 12.418 ± 0.010 * |
| R14 | Dali, Yunnan (25°36 N 100°15 E) | October, 2016 (RDY201610-14) | 0.602 ± 0.013 | 4.296 ± 0.019 | 0.264 ± 0.011 | 0.128 ± 0.006 | 0.092 ± 0.003 | 0.096 ± 0.003 | 0.203 ± 0.007 | 0.412 ± 0.009 | 6.093 ± 0.009 * |
| No. | Region or Pharmacy (Specimen No.) | Content of Investigated Components (n = 3, mg/g ± SD) | Total Content (n = 3, mg/g ± SD) | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| CA (5) 1 | 3-CQA (3) | 4-CQA (4) | 5-CQA (2) | 1,3-diCQA (6) | 1,5-diCQA (9) | 1,3,5-tirCQA (11) | 4,5-diCQA (12) | |||
| P1 | Binxian, Heilongjiang (PBH2016-01) | 0.223 ± 0.020 | 4.123 ± 0.032 | 1.051 ± 0.022 | 0.882 ± 0.010 | 0.056 ± 0.002 | 0.207 ± 0.004 | 0.114 ± 0.003 | 0.332 ± 0.005 | 6.988 ± 0.012 |
| P2 | Tieling, Lioaning (PTL2016-02) | 0.196 ± 0.013 | 3.670 ± 0.029 | 0.824 ± 0.018 | 0.531 ± 0.012 | 0.045 ±0.003 | 0.178 ± 0.007 | 0.098 ± 0.004 | 0.222 ± 0.009 | 5.764 ± 0.014 * |
| P3 | Baotou, Inner Mongolia (PBI2016-03) | 0.331 ± 0.010 | 5.306 ± 0.029 | 0.912 ± 0.024 | 0.586 ± 0.025 | 0.174 ± 0.009 | 1.492 ± 0.012 | 0.423 ± 0.010 | 0.389 ± 0.014 | 9.613 ± 0.013 |
| P4 | Baoding, Hebei (PBH2016-04) | 0.359 ± 0.012 | 5.024 ± 0.022 | 1.026 ± 0.021 | 0.891 ± 0.012 | 0.084 ± 0.010 | 0.273 ± 0.011 | 0.183 ± 0.012 | 0.330 ± 0.016 | 8.170 ± 0.012 |
| P5 | Jining, Shandong (PJS2016-05) | 0.181 ± 0.015 | 3.940 ± 0.024 | 1.191 ± 0.022 | 1.120 ± 0.024 | 0.501 ± 0.014 | 0.501 ± 0.015 | 0.190 ± 0.009 | 0.656 ± 0.021 | 8.280 ± 0.015 |
| P6 | QInling, Shanxi (PQS2016-06) | 0.121 ± 0.012 | 3.675 ± 0.022 | 0.630 ± 0.016 | 0.372 ± 0.011 | 0.097 ± 0.012 | 0.529 ± 0.014 | 0.079 ± 0.010 | 0.586 ± 0.014 | 6.089 ± 0.013* |
| P7 | Zhumadian, Henan (PZH2016-07) | 0.538 ± 0.014 | 7.238 ± 0.025 | 0.533 ± 0.012 | 0.140 ± 0.010 | 0.041 ± 0.011 | 1.068 ± 0.013 | 0.256 ± 0.012 | 0.317 ± 0.011 | 10.131 ± 0.014 * |
| P8 | Suqian, Jiangsu (PSJ2016-08) | 0.337 ± 0.010 | 5,142 ± 0.021 | 1.338 ± 0.011 | 1.145 ± 0.008 | 0.023 ± 0.009 | 0.017 ± 0.010 | 0.026 ± 0.011 | 0.714 ± 0.012 | 8.742 ± 0.013 |
| P9 | Liuan, Anhui (PLA2016-09) | 0.042 ± 0.009 | 1.565 ± 0.012 | 0.428 ± 0.010 | 0.533 ± 0.012 | 0.079 ± 0.014 | 0.070 ± 0.012 | 0.085 ± 0.011 | 0.189 ± 0.012 | 2.991 ± 0.011 ** |
| P10 | Dazhou, Sichuan (PDS2016-10) | 0.296 ± 0.015 | 7.567 ± 0.011 | 1.732 ± 0.012 | 1.221 ± 0.014 | 0.994 ± 0.011 | 9.688 ± 0.013 | 2.034 ± 0.16 | 2.677 ± 0.011 | 26.209 ± 0.012 ** |
| P11 | Huanggang, Hubei (PHH2016-11) | 0.283 ± 0.013 | 3.306 ± 0.011 | 1.053 ± 0.012 | 0.871 ± 0.014 | 0.085 ± 0.011 | 0.143 ± 0.015 | 0.042 ± 0.013 | 0.246 ± 0.011 | 6.029 ± 0.011 |
| P12 | Shaoyang, Hunan (PSH2016-12) | 0.161 ± 0.010 | 3.624 ± 0.012 | 0.768 ± 0.011 | 0.542 ± 0.013 | 0.084 ± 0.011 | 0.546 ± 0.014 | 0.129 ± 0.012 | 0.563 ± 0.013 | 6.417 ± 0.012 |
| P13 | Guilin, Guangxi (PGG2016-13) | 0.209 ± 0.012 | 3.716 ± 0.011 | 0.901 ± 0.011 | 0.688 ± 0.023 | 0.067 ± 0.012 | 0.813 ± 0.021 | 0.137 ± 0.015 | 0.334 ± 0.020 | 6.865 ± 0.015 |
| P14 | Dali, Yunnan (PDY2016-14) | 0.195 ± 0.015 | 2.983 ± 0.021 | 0.921 ± 0.016 | 0.853 ± 0.024 | 0.030 ± 0.009 | 0.976 ± 0.016 | 0.045 ± 0.008 | 0.166 ± 0.010 | 6.169 ± 0.014 * |
| P15 | Hangzhou, Zhejiang (PHZ2016-15) | 0.237 ± 0.021 | 5.607 ± 0.017 | 0.359 ± 0.012 | 0.338 ± 0.018 | 0.020 ± 0.004 | 0.670 ± 0.012 | 0.791 ± 0.021 | 0.469 ± 0.023 | 8.491 ± 0.016 |
| P16 | Tongrentang, Beijing (PTB2016-16) | 0.406 ± 0.021 | 7.225 ± 0.016 | 0.697 ± 0.021 | 0.089 ± 0.010 | 0.103 ± 0.005 | 0.853 ± 0.011 | 0.232 ± 0.019 | 0.302 ± 0.021 | 9.907 ± 0.015 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jiang, H.; Yang, L.; Xing, X.; Yan, M.; Guo, X.; Yang, B.; Wang, Q.; Kuang, H. HPLC-PDA Combined with Chemometrics for Quantitation of Active Components and Quality Assessment of Raw and Processed Fruits of Xanthium strumarium L. Molecules 2018, 23, 243. https://doi.org/10.3390/molecules23020243
Jiang H, Yang L, Xing X, Yan M, Guo X, Yang B, Wang Q, Kuang H. HPLC-PDA Combined with Chemometrics for Quantitation of Active Components and Quality Assessment of Raw and Processed Fruits of Xanthium strumarium L. Molecules. 2018; 23(2):243. https://doi.org/10.3390/molecules23020243
Chicago/Turabian StyleJiang, Hai, Liu Yang, Xudong Xing, Meiling Yan, Xinyue Guo, Bingyou Yang, Qiuhong Wang, and Haixue Kuang. 2018. "HPLC-PDA Combined with Chemometrics for Quantitation of Active Components and Quality Assessment of Raw and Processed Fruits of Xanthium strumarium L." Molecules 23, no. 2: 243. https://doi.org/10.3390/molecules23020243
APA StyleJiang, H., Yang, L., Xing, X., Yan, M., Guo, X., Yang, B., Wang, Q., & Kuang, H. (2018). HPLC-PDA Combined with Chemometrics for Quantitation of Active Components and Quality Assessment of Raw and Processed Fruits of Xanthium strumarium L. Molecules, 23(2), 243. https://doi.org/10.3390/molecules23020243





