Study of the Direct Red 81 Dye/Copper(II)-Phenanthroline System
Abstract
:1. Introduction
2. Results and Discussion
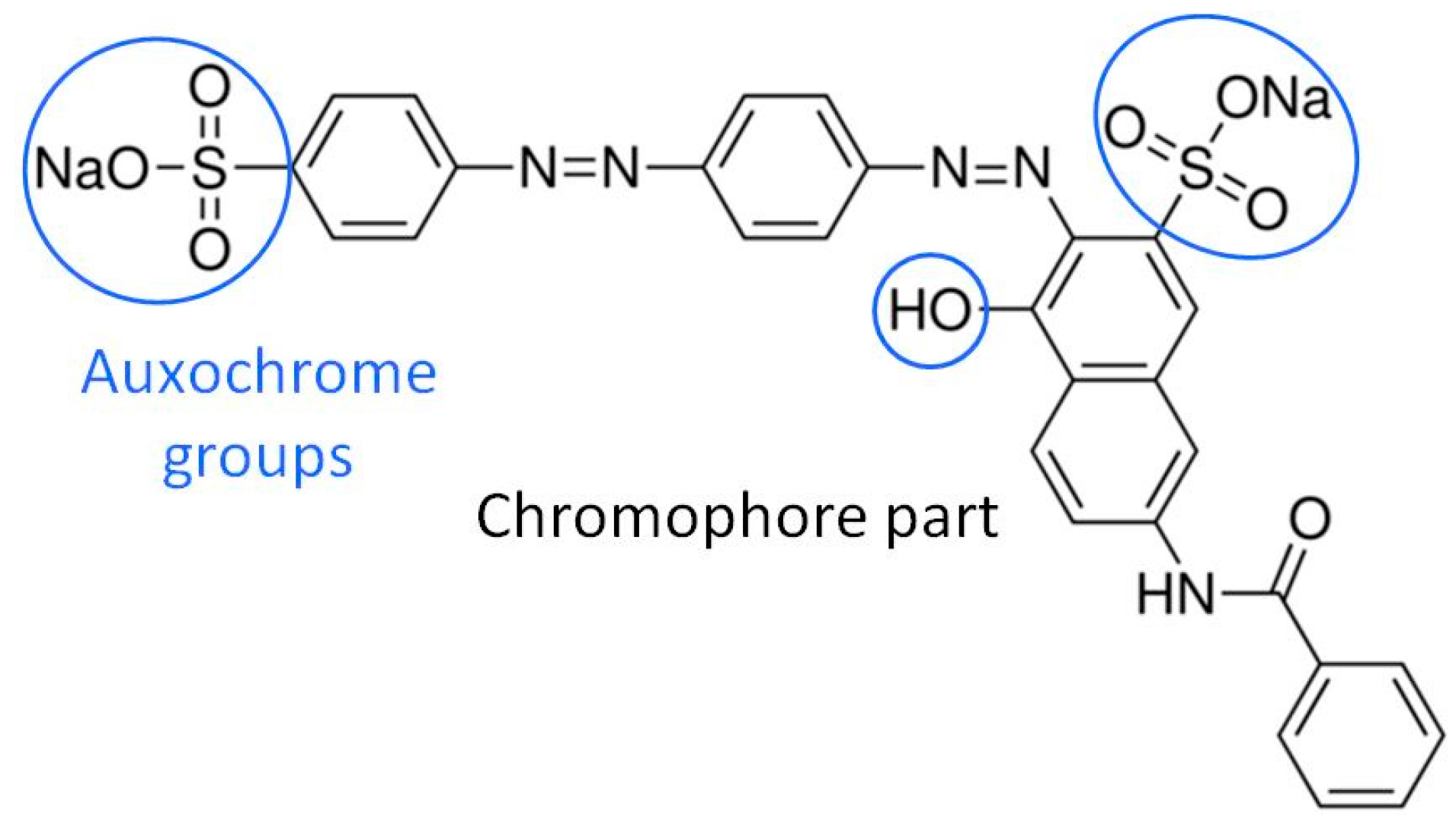
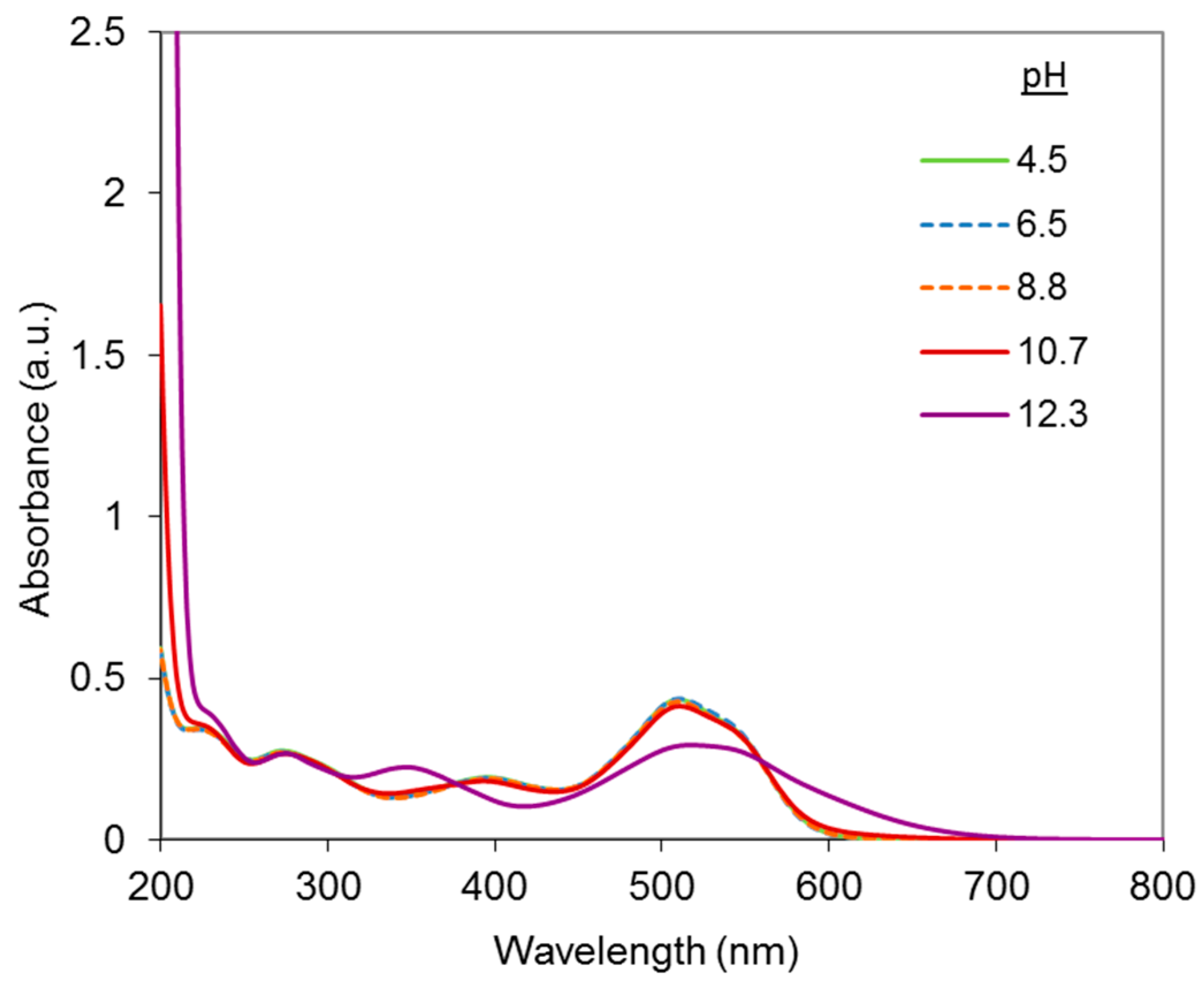
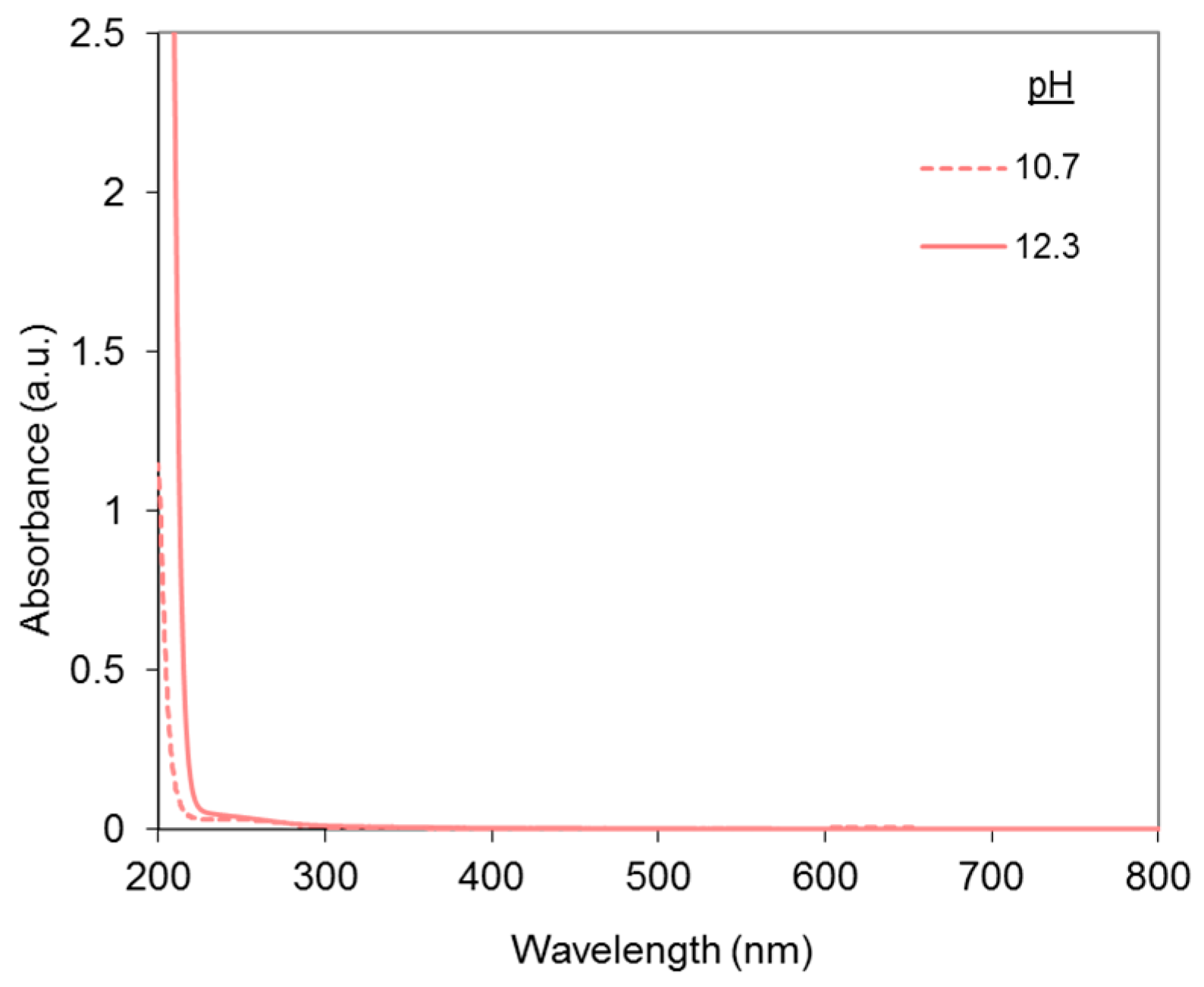
2.1. UV-Visible Analysis of the Dye
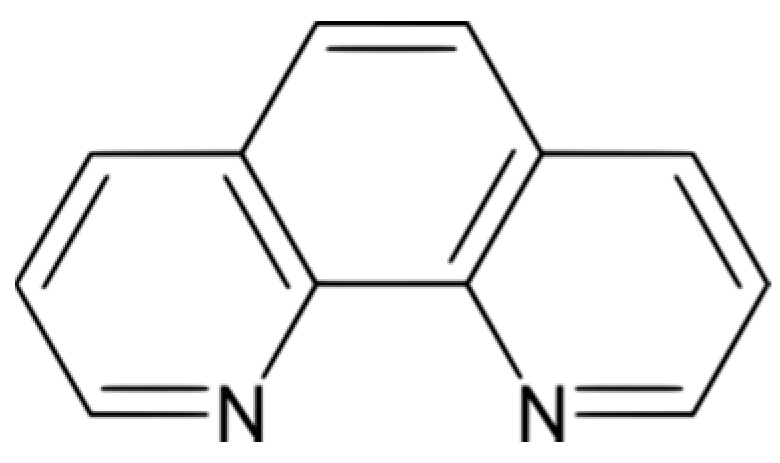
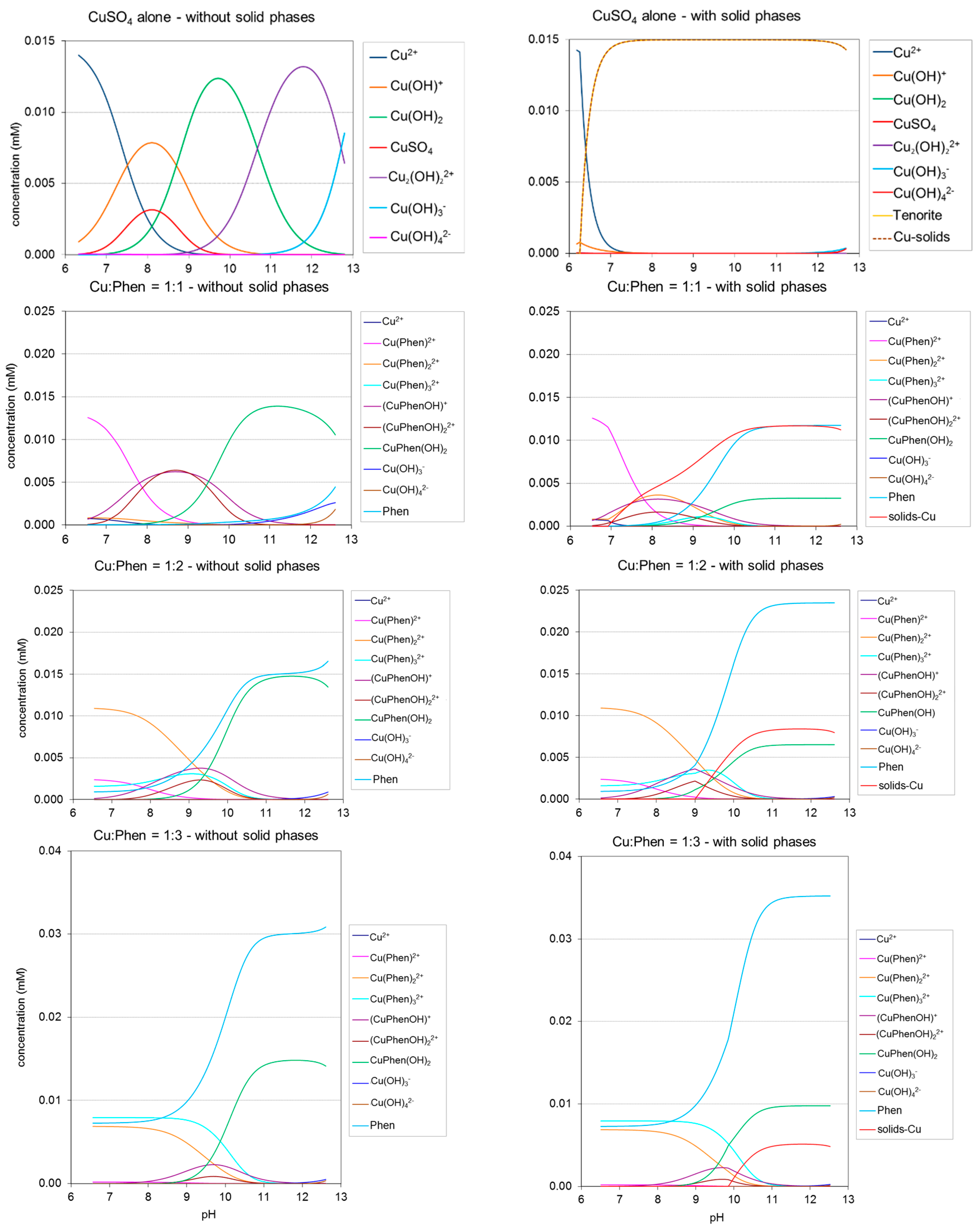
2.2. Copper-Phenanthroline Speciation
2.3. Analysis of the Cu-Phen Complex by UV-Vis Spectroscopy
2.3.1. CuSO4
2.3.2. Phenanthroline
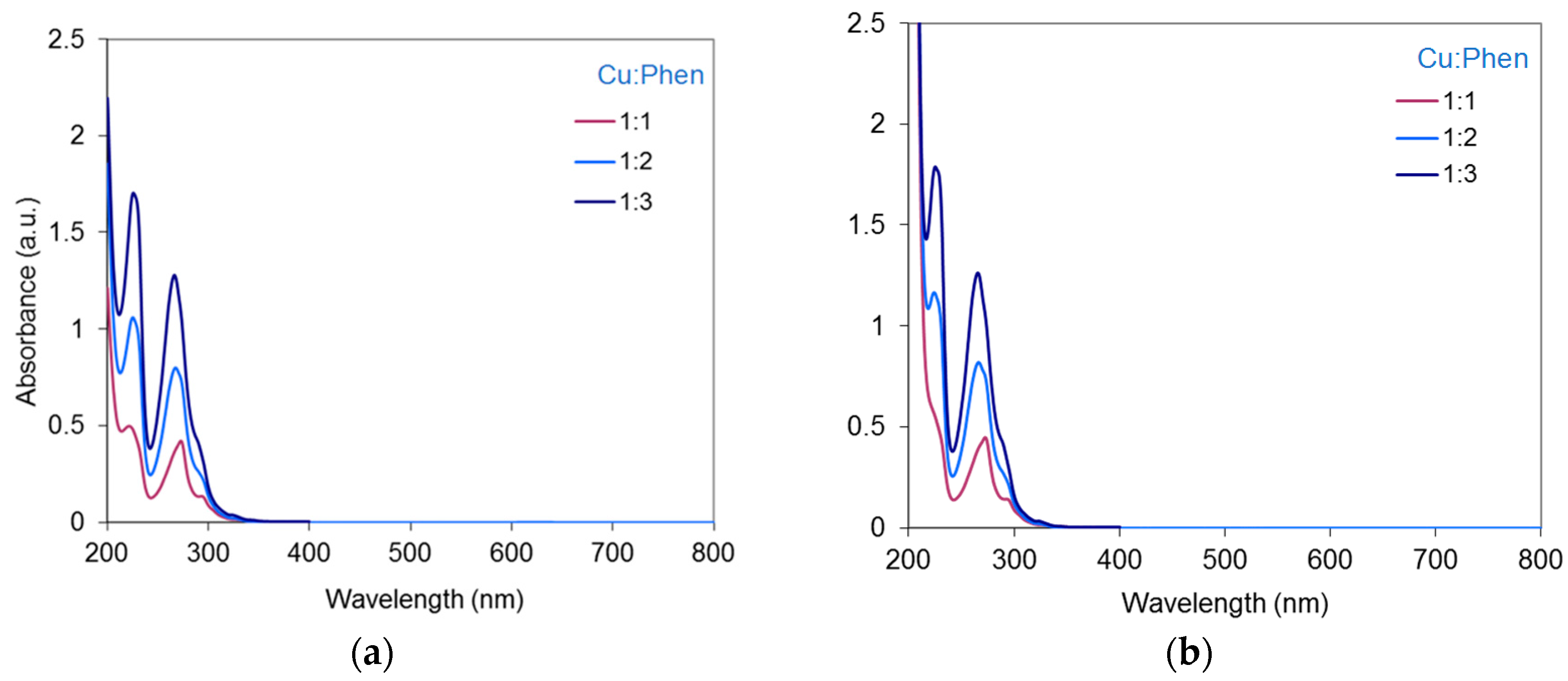
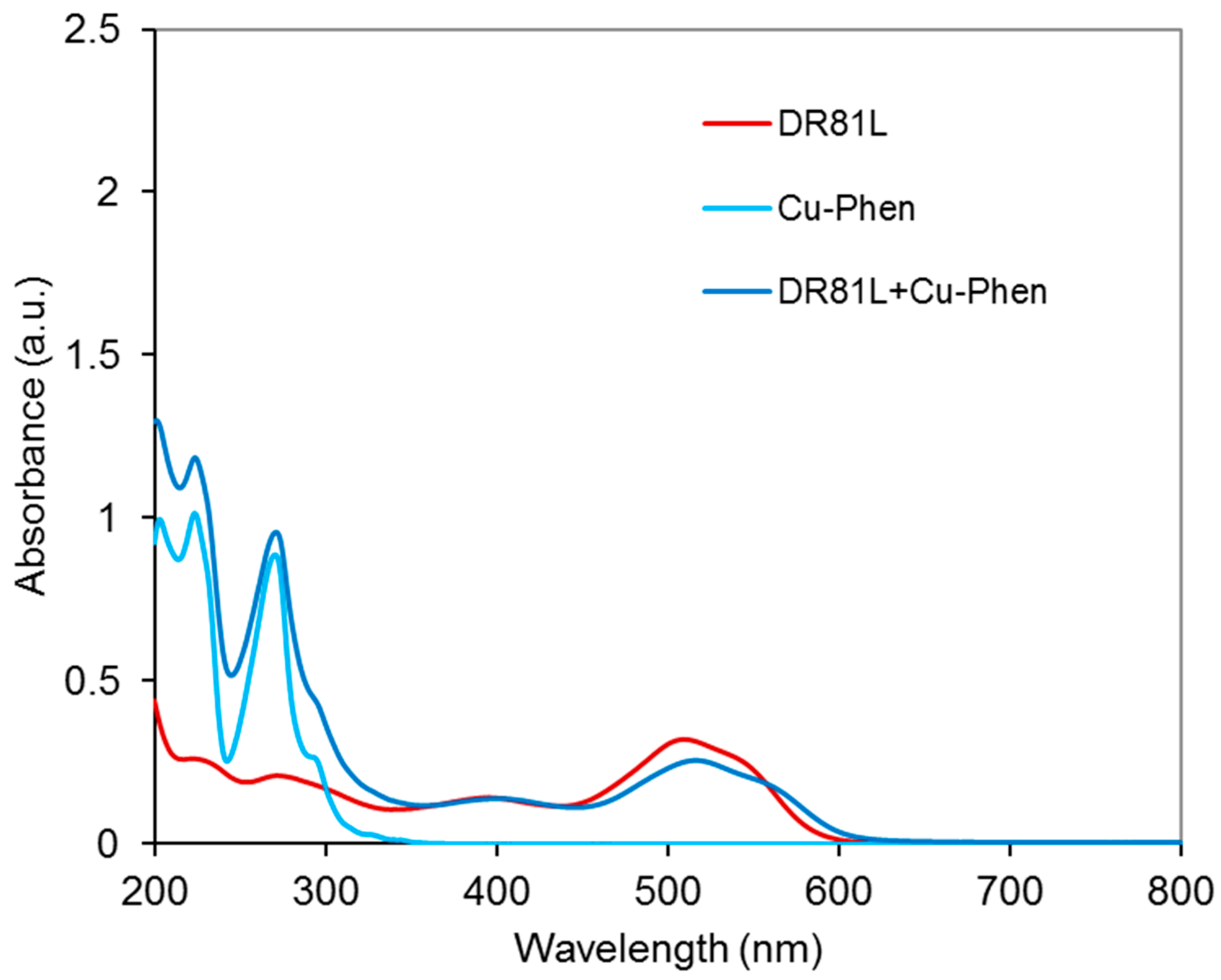
2.3.3. Cu-Phen
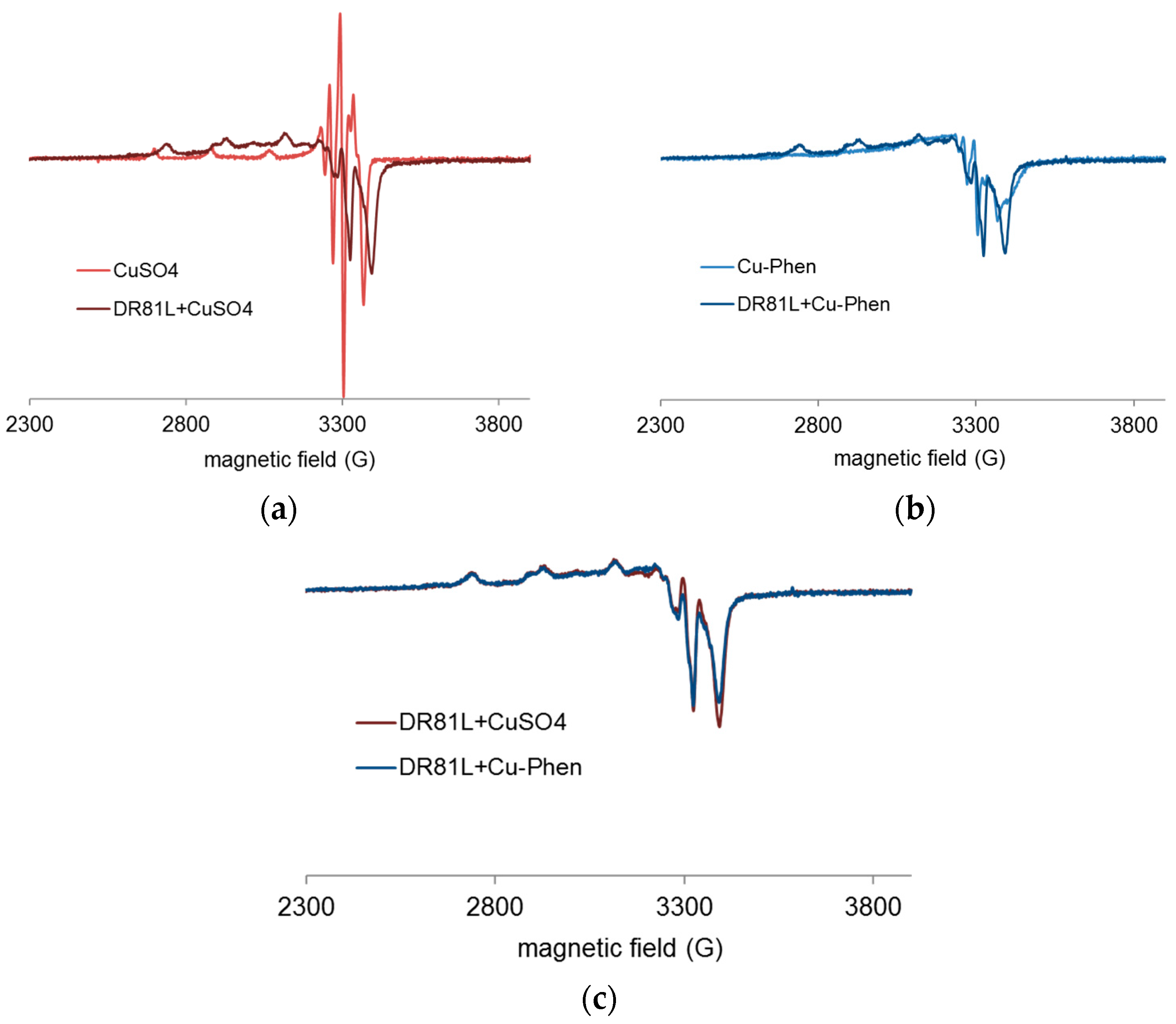
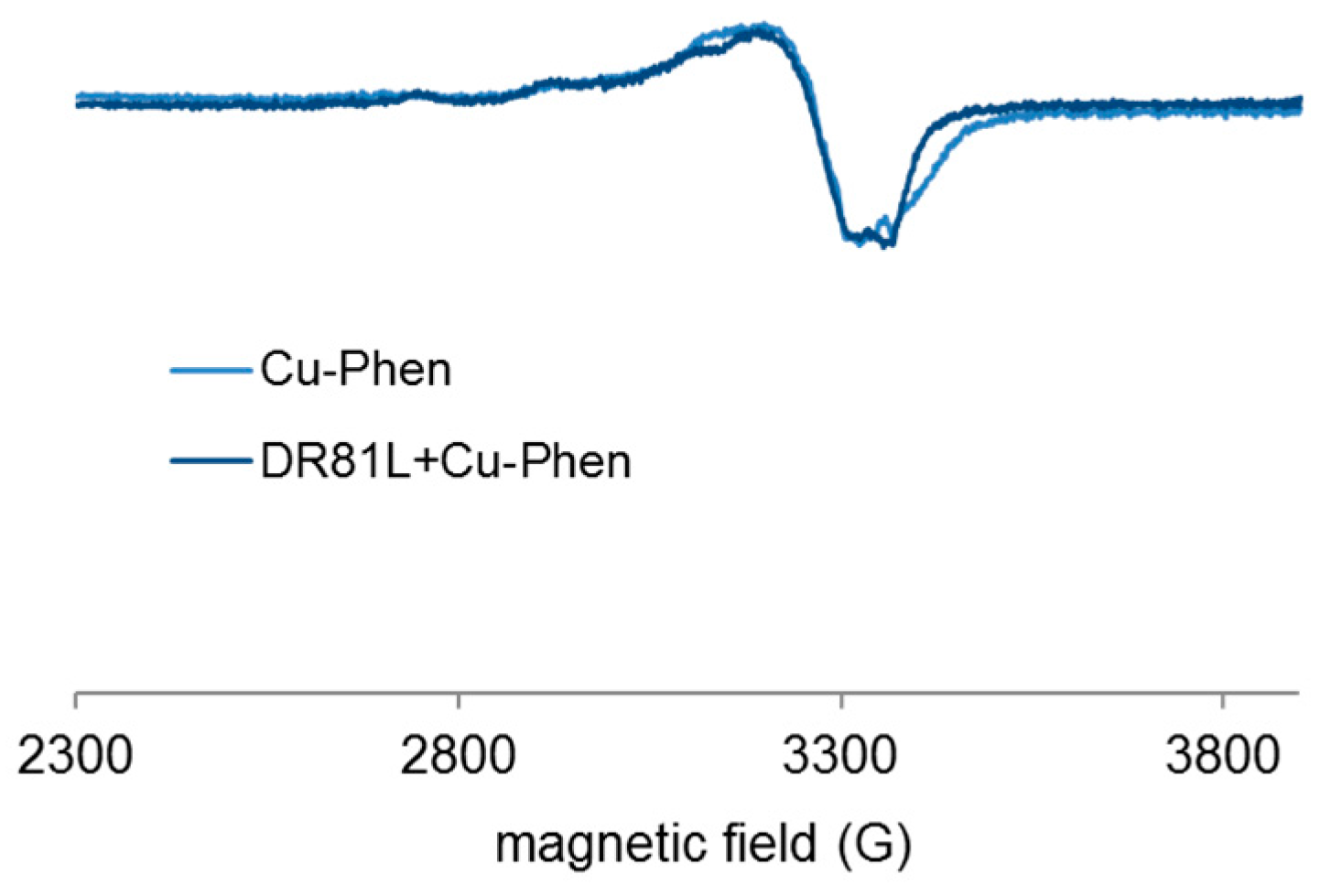
2.3.4. Evidence of Cu-Phen Coordination by EPR
2.4. Analysis of the DR81 Dye/Cu-Phen System
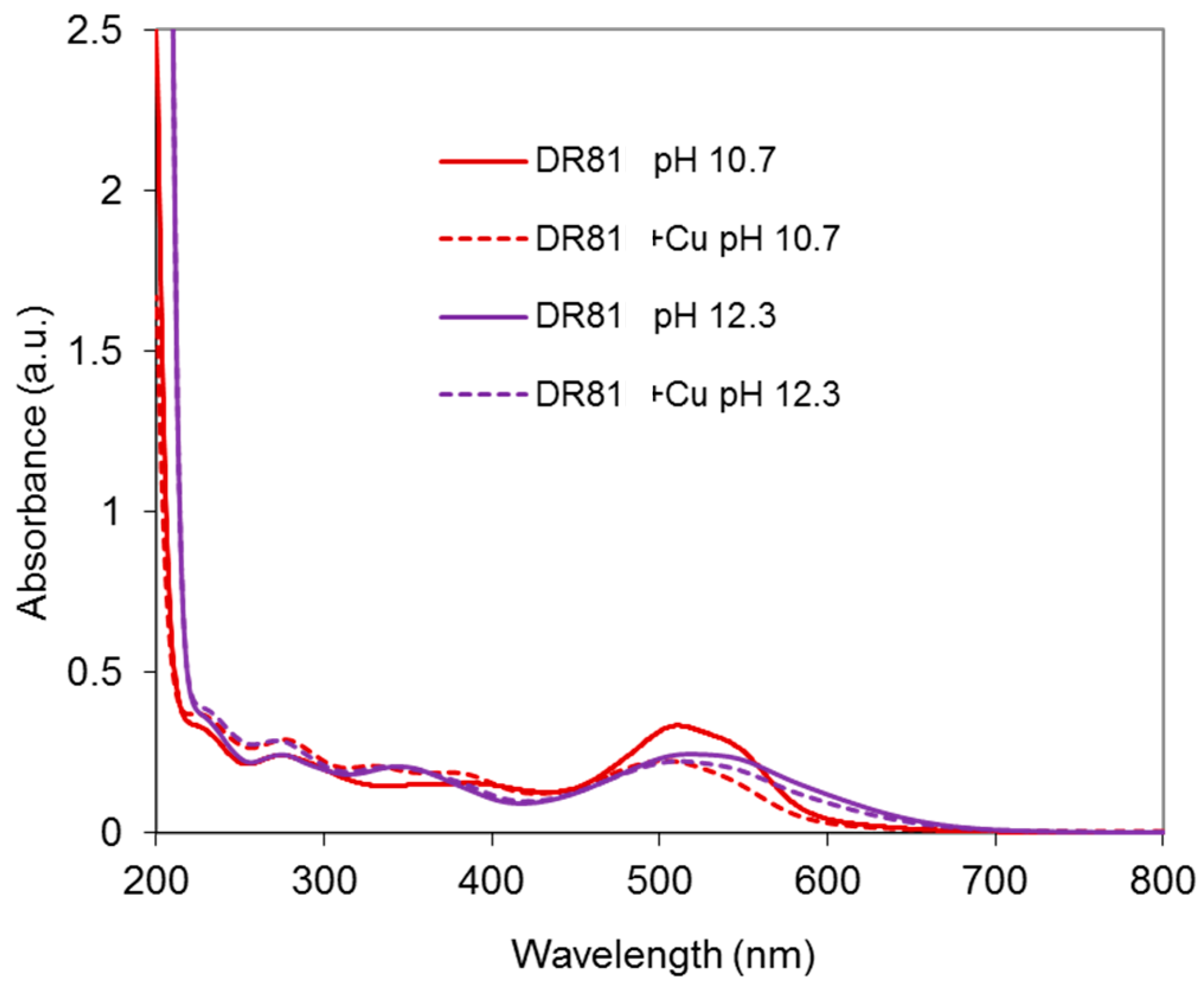
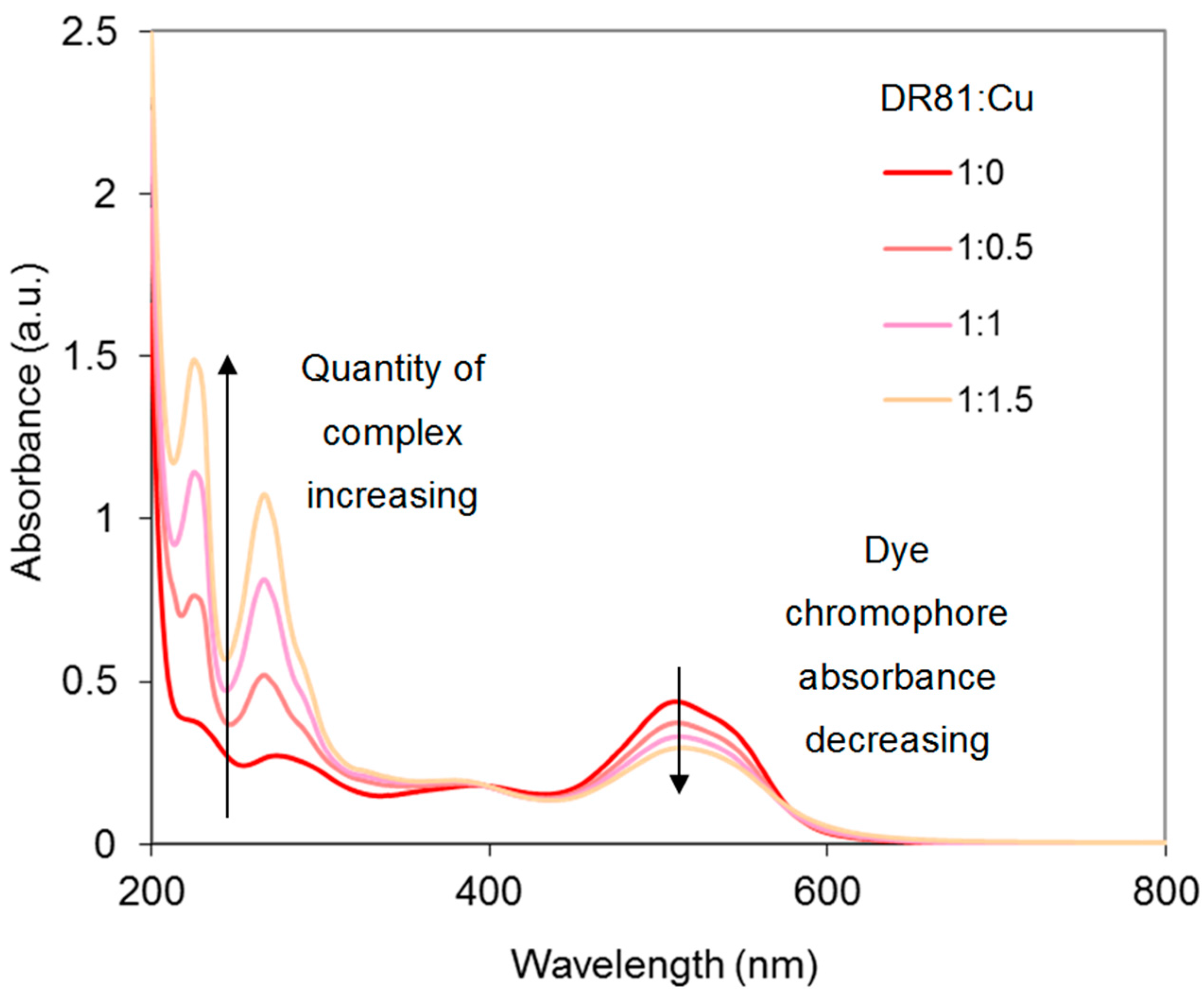
2.4.1. Dye/CuSO4 System
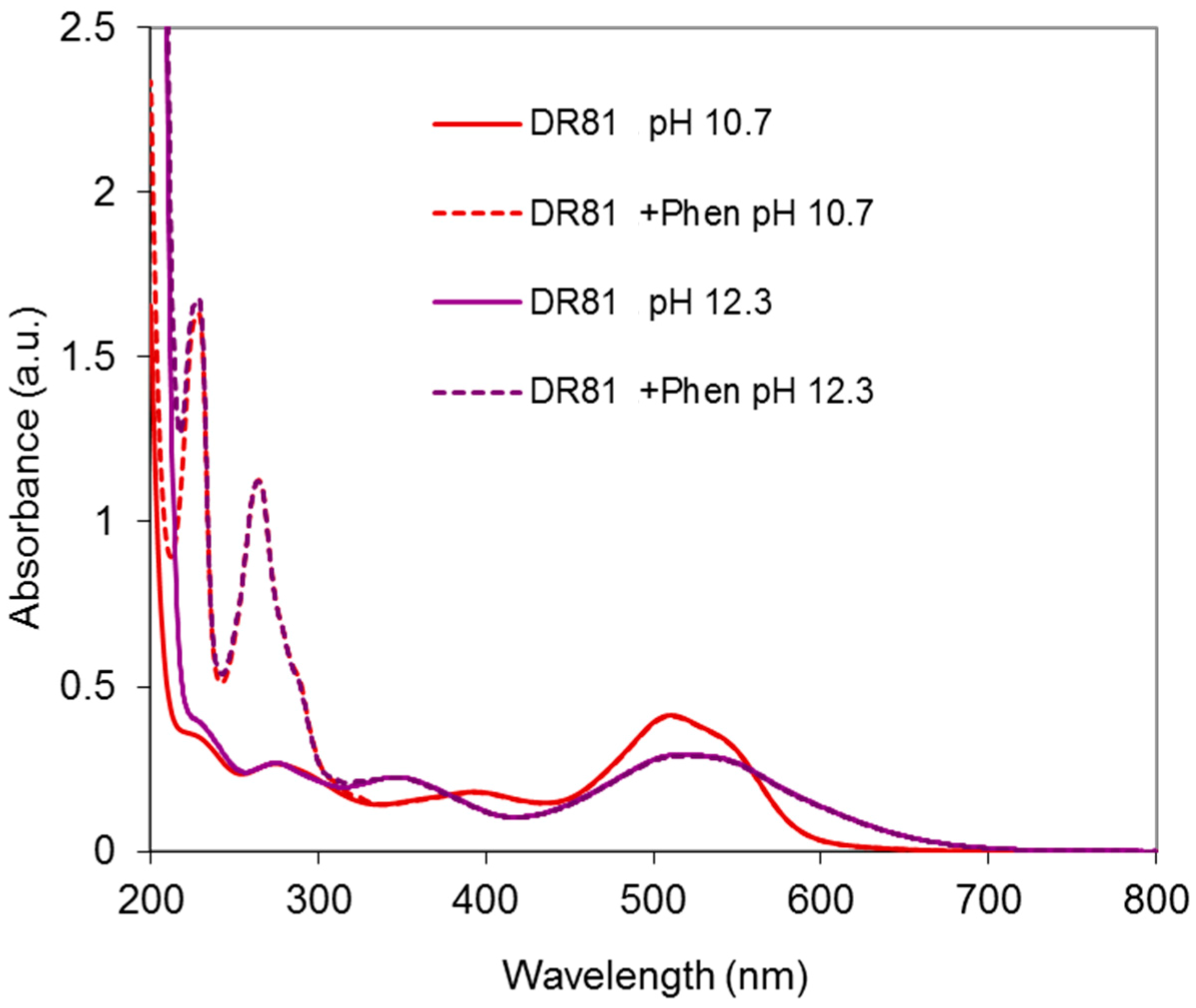
2.4.2. Dye/Phenanthroline System
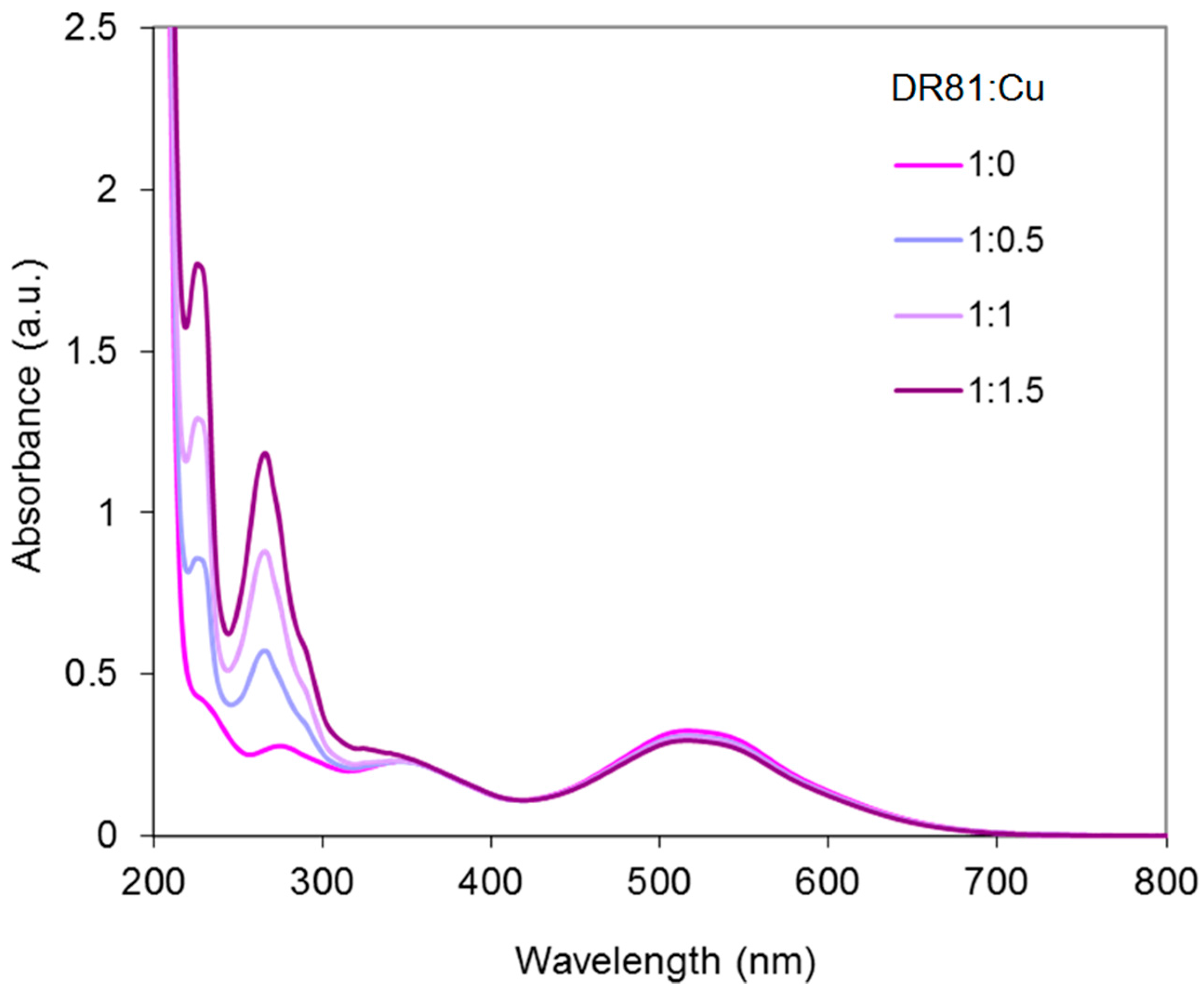
2.4.3. Dye/Cu-Phen System
Results at Alkaline pH
Results in Neutral Conditions
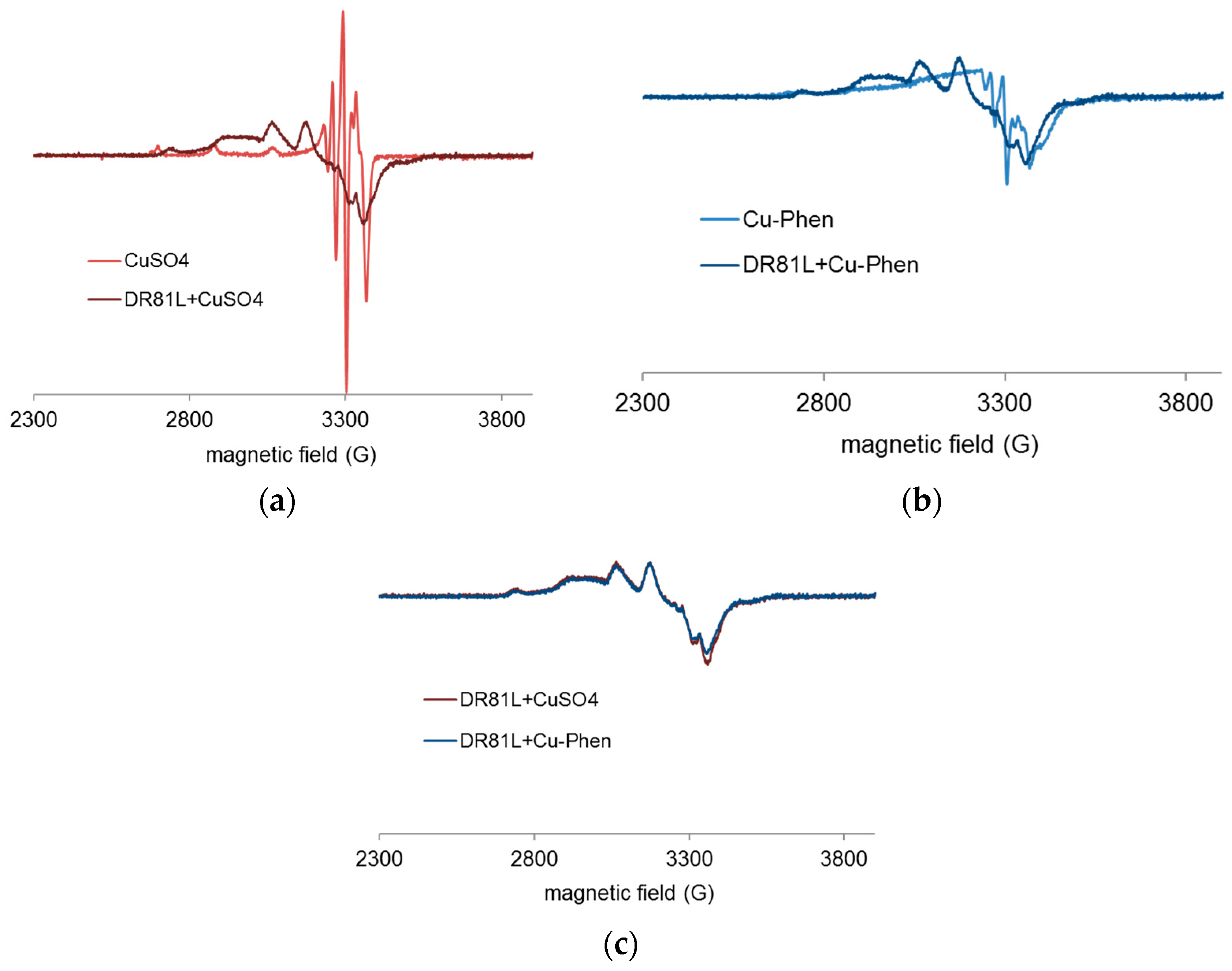
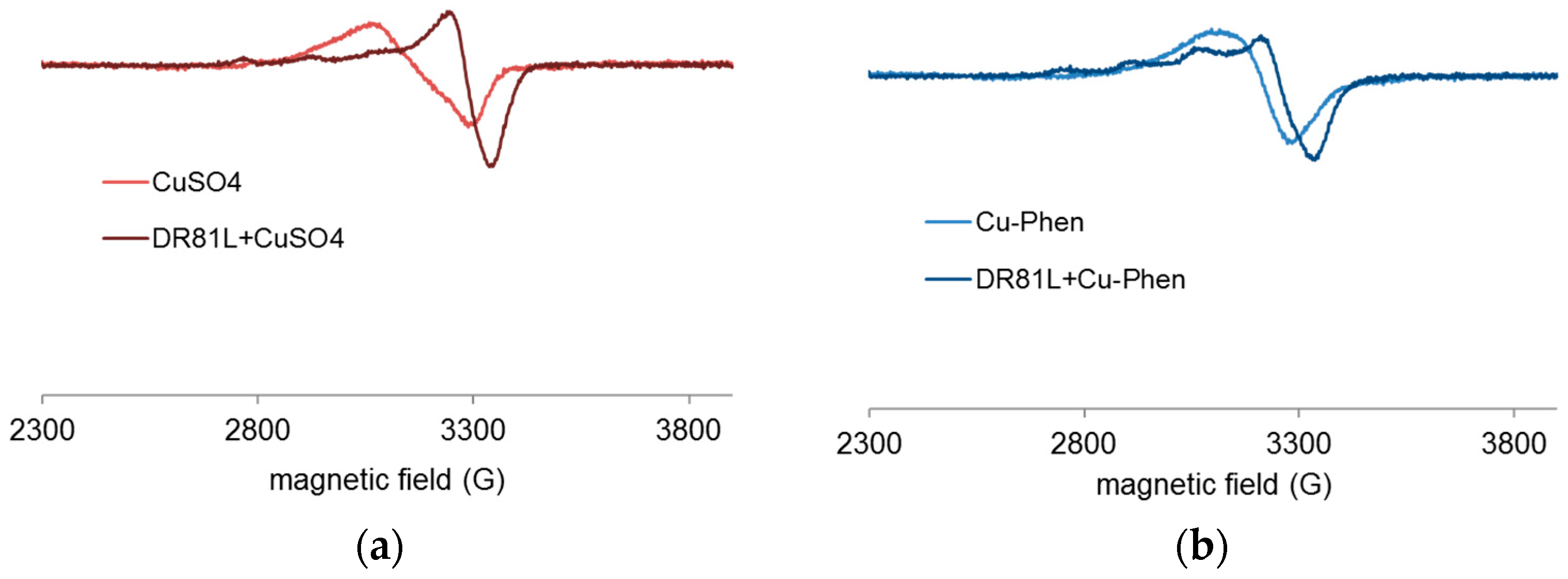
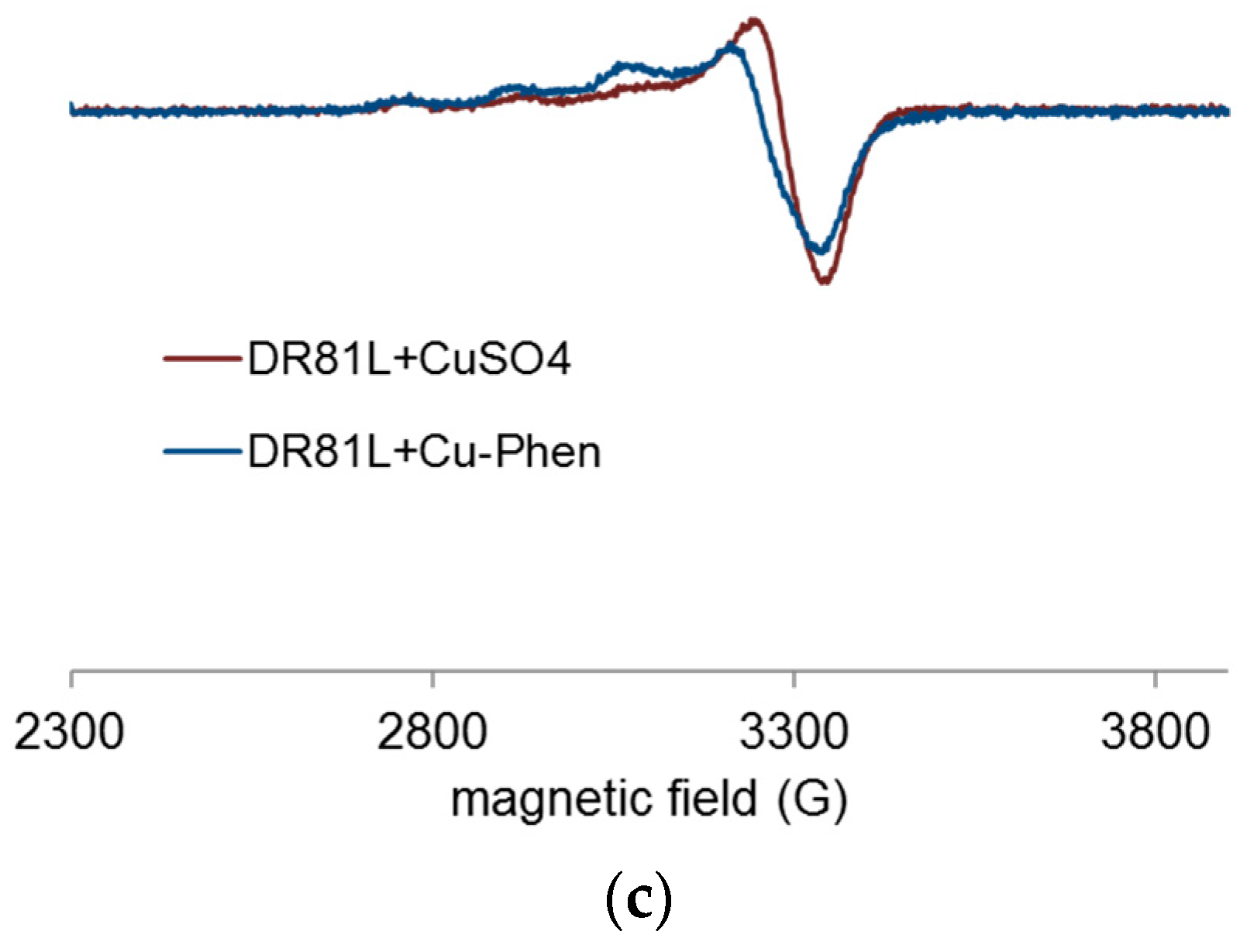
2.4.4. Proof of Dye-Cu-Phen Coordination
- (1)
- In strong alkaline conditions (ionized dye at pH 12.3) and at near neutral pH (unionized dye, pH 6.5), with or without phenanthroline, the chromophore’s electronic structure was hardly affected by copper complexation.
- (2)
- In medium alkaline conditions, pH 10.7 (unionized dye), with or without phenanthroline, the chromophore structure was partly modified.
3. Materials and Methods
3.1. Raw Materials
3.2. Stock Solution Preparation
3.3. Reactions
3.4. Centrifugation
3.5. UV-Vis Spectroscopy
3.6. EPR Spectroscopy
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Alén, R. Papermaking additives. In Papermaking Science and Technology: Vol. 4. Papermaking Chemistry; Papermaking science and technology; Fapet Oy: Helsinki, Finland, 2007; ISBN 978-952-5216-24-0. [Google Scholar]
- Göttsching, L.; Pakarinen, H. Papermaking Science and Technology: Vol. 7. Recycled Fiber and Deinking; Papermaking Science and Technology; Fapet Oy: Helsinki, Finland, 2000; Volume 7, ISBN 978-95-2-521607-1. [Google Scholar]
- Blau, F. Über neue organische Metallverbindungen. Monatschefte Chem. Verwandte Teile Anderer Wiss. 1898, 19, 647–689. [Google Scholar] [CrossRef]
- Sigman, D.S.; Graham, D.R.; D’Aurora, V.; Stern, A.M. Oxygen-dependent cleavage of DNA by the 1,10-phenanthroline cuprous complex. Inhibition of Escherichia coli DNA polymerase I. J. Biol. Chem. 1979, 254, 12269–12272. [Google Scholar] [PubMed]
- Sigman, D.S. Nuclease activity of 1,10-phenanthroline-copper ion. Acc. Chem. Res. 1986, 19, 180–186. [Google Scholar] [CrossRef]
- Thederahn, T.B.; Kuwabara, M.D.; Larsen, T.A.; Sigman, D.S. Nuclease activity of 1,10-phenanthroline-copper: Kinetic mechanism. J. Am. Chem. Soc. 1989, 111, 4941–4946. [Google Scholar] [CrossRef]
- Lu, L.-P.; Zhu, M.-L.; Yang, P. Crystal structure and nuclease activity of mono(1,10-phenanthroline) copper complex. J. Inorg. Biochem. 2003, 95, 31–36. [Google Scholar] [CrossRef]
- Coucharriere, C. Mise au Point et Etude d’un Système D’activation du Peroxyde D’hydrogène en Délignification et Blanchiment des Pâtes Chimiques. Ph.D. Thesis, University of Grenoble, Grenoble, France, 2000. [Google Scholar]
- Marlin, N.; Coucharriere, C.; Mortha, G.; Lachenal, D.; Larnicol, P.; Hostachy, J.C. Use of o-phenanthroline as a catalyst in hydrogen peroxide stages. In Proceedings of the 13th International Symposium on Wood Fibre and Pulping Chemistry, Auckland, New Zealand, 16–19 May 2005; APPITA: Carlton, Australia, 2005; pp. 29–34. [Google Scholar]
- Vladut, N.I. Système Chimique Délignifiant à Base de Peroxyde D’hydrogène. Ph.D. Thesis, University of Grenoble, Grenoble, France, 2012. [Google Scholar]
- Cabanne, L.; Larnicol, P.; Coucharriere, C. Hydrogen Peroxide Activation in Delignification and Bleaching of Wood Pulp Using a Phenanthroline-Copper Complex. WO03080925 (A1), 2 October 2003. [Google Scholar]
- Blanc, J.; Calais, C.; Dubois, J.L. Process for Delignification and Bleaching of Paper Pulp Using Activated Hydrogen Peroxide. WO02012028800 (A1), 28 June 2012. [Google Scholar]
- Korpi, H.; Lahtinen, P.; Sippola, V.; Krause, O.; Leskelä, M.; Repo, T. An efficient method to investigate metal-ligand combinations for oxygen bleaching. Appl. Catal. A Gen. 2004, 268, 199–206. [Google Scholar] [CrossRef]
- Korpi, H.; Figiel, P.J.; Lankinen, E.; Ryan, P.; Leskelä, M.; Repo, T. On in situ prepared Cu–Phenanthroline complexes in aqueous alkaline solutions and their use in the catalytic oxidation of veratryl alcohol. Eur. J. Inorg. Chem. 2007, 2007, 2465–2471. [Google Scholar] [CrossRef]
- Lahtinen, P.; Korpi, H.; Haavisto, E.; Leskelä, M.; Repo, T. Parallel screening of homogeneous copper catalysts for the oxidation of benzylic alcohols with molecular oxygen in aqueous solutions. J. Comb. Chem. 2004, 6, 967–973. [Google Scholar] [CrossRef] [PubMed]
- Korpi, H. Copper di-Imine Complexes: Structures and Catalytic Activity in the Oxidation of Alcohols by Dioxygen. Ph.D. Thesis, University of Helsinki, Helsinki, Finland, 2005. [Google Scholar]
- Sippola, V.O.; Krause, A.O.I. Bis(o-phenanthroline)copper-catalysed oxidation of lignin model compounds for oxygen bleaching of pulp. Catal. Today 2005, 100, 237–242. [Google Scholar] [CrossRef]
- Alderighi, L.; Gans, P.; Ienco, A.; Peters, D.; Sabatini, A.; Vacca, A. Hyperquad simulation and speciation (HySS): A utility program for the investigation of equilibria involving soluble and partially soluble species. Coord. Chem. Rev. 1999, 184, 311–318. [Google Scholar] [CrossRef]
- Marlin, N.; Fernandes, J.; Benattar, N. New ways to improve color-stripping of deinked pulps and dyed effluents. In Proceedings of the 14th International Symposium on Wood Fibre and Pulping Chemistry, Durban, South Africa, 25–28 June 2007; TAPPSA: Kloof, South Africa, 2007. [Google Scholar]
- Walger, E.; Rivollier, C.; Marlin, N.; Mortha, G. Activated H2O2 discoloration of a model azoic dye-colored pulp. In Proceedings of the 13th European Workshop on Lignocellulosics and Pulp, Seville, Spain, 24–27 June 2014; De Gruyter: Seville, Spain, 2014; pp. 863–866. [Google Scholar]
- Sharpe, P.E.; Lowe, R.W. The bleaching of colored recycled fibers. In Proceedings of the 1993 Pulping Conference, Boston, MA, USA, 15–17 June 1993; TAPPI Press: Boston, MA, USA, 1993; Volume 3, pp. 1205–1217. [Google Scholar]
- Walger, E. Study of the Activation of Hydrogen Peroxide by the Copper(II)-Phenanthroline Complex for the Color-Stripping of Recovered Cellulosic Fibers; Université Grenoble Alpes: Grenoble, France, 2016. [Google Scholar]
- Moeller, T. Inorganic Chemistry : An Advanced Textbook; Wiley: New York, NY, USA, 1952. [Google Scholar]
- Ringbom, A. Les Complexes en Chimie Analytique : Comment Choisir Rationnellement les Meilleures Méthodes D’analyse Complexométrique; Dunod: Paris, France, 1967. [Google Scholar]
- Schilt, A.A. Analytical Applications of 1,10-Phenanthroline and Related Compounds, 1st ed.; International Series of Monographs in Analytical Chemistry; Pergamon Press: Oxford, UK; London, UK; Edinburgh, UK, 1969; Volume 32. [Google Scholar]
- Smith, R.M.; Martell, A.E. Critical Stability Constants; Plenum Press: New York, NY, USA; London, UK, 1975; ISBN 0-306-35212-5. [Google Scholar]
- Mcbryde, W.A.E. A Critical Review of Equilibrium Data for Proton and Metal Complexes of 1,10-Phenanthroline, 2,2′-Bipyridyl and Related Compounds; Pergamon Press: Oxford, UK; New York, NY, USA; Toronto, ON, Canada; Rushcutters Bay, Australia; Paris, France; Kronberg-Taunus, Germany, 1978; ISBN 0-08-022344-3. [Google Scholar]
- Yamazaki, K.; Yokoi, H.; Sone, K. Spectrochemical studies on o-phenanthroline complex salts. J. Chem. Soc. Japan: Pure Chem. Sect. 1948, 69, 137–140. [Google Scholar] [CrossRef]
- Armaroli, N.; De Cola, L.; Balzani, V.; Sauvage, J.-P.; Dietrich-Buchecker, C.O.; Kern, J.-M. Absorption and luminescence properties of 1,10-phenanthroline, 2,9-diphenyl-1,10-phenanthroline, 2,9-dianisyl-1,10-phenanthroline and their protonated forms in dichloromethane solution. J. Chem. Soc. Faraday Trans. 1992, 88, 553–556. [Google Scholar] [CrossRef]
- Vallee, B.L.; Rupley, J.A.; Coombs, T.L.; Neurath, H. The Role of Zinc in Carboxypeptidase. J. Biol. Chem. 1960, 235, 64–69. [Google Scholar]
- Ni, Y.; Lin, D.; Kokot, S. Synchronous fluorescence, UV–visible spectrophotometric, and voltammetric studies of the competitive interaction of bis(1,10-phenanthroline)copper(II) complex and neutral red with DNA. Anal. Biochem. 2006, 352, 231–242. [Google Scholar] [CrossRef] [PubMed]
- Bovet, C.; Barron, A.R. Electron Paramagnetic Resonance Spectroscopy of Copper(II) Compounds—OpenStax CNX. Available online: http://cnx.org/contents/NB5XiHcn@2/Electron-Paramagnetic-Resonanc (accessed on 17 May 2016).
Sample Availability: Samples of the compound Direct Red 81 are available from the authors. |







| Cu:Phen | 1:0 | 1:1 | 1:2 | 1:3 | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| pH | 6.5 | 10.7 | 12.3 | 6.5 | 10.7 | 12.3 | 6.5 | 10.7 | 12.3 | 6.5 | 10.7 | 12.3 |
| Cu2+ | 90.2 | ε | ε | 5.1 | ε | ε | ε | ε | ε | ε | ε | ε |
| CuOH+ | 8.8 | 0.5 | ε | 0.5 | ε | ε | ε | ε | ε | ε | ε | ε |
| Cu(OH)2 | 0.1 | 48.1 | 1.6 | ε | 1.7 | 0.3 | ε | 0.1 | 0.1 | ε | ε | ε |
| CuSO4 | 0.3 | ε | ε | ε | ε | ε | ε | ε | ε | ε | ε | ε |
| Cu2(OH)22+ | 0.6 | ε | ε | ε | ε | ε | ε | ε | ε | ε | ε | ε |
| Cu(OH)3− | ε a | 51.2 | 77.4 | ε | 1.8 | 14.5 | ε | 0.1 | 3.1 | ε | ε | 1.6 |
| Cu(OH)42− | ε | 0.2 | 21.0 | ε | ε | 3.9 | ε | ε | 0.8 | ε | ε | 0.4 |
| Cu(Phen)2+ | / | / | / | 84.3 | ε | ε | 15.9 | ε | ε | 1.3 | ε | ε |
| Cu(Phen)22+ | / | / | / | 5.5 | ε | ε | 72.8 | 0.6 | ε | 45.8 | 1.1 | ε |
| Cu(Phen)32+ | / | / | / | ε | ε | ε | 10.6 | 1.4 | ε | 52.8 | 4.9 | ε |
| (CuPhenOH)+ | / | / | / | 4.1 | 5.8 | 0.2 | 0.8 | 5.9 | 0.2 | 0.1 | 5.7 | 0.2 |
| (CuPhenOH)22+ | / | / | / | 0.4 | 0.9 | ε | ε | 0.9 | ε | ε | 0.8 | ε |
| CuPhen(OH)2 | / | / | / | ε | 89.7 | 81.2 | ε | 91.1 | 95.9 | ε | 87.4 | 97.7 |
| Phen | / | / | / | 0.1 | 3.5 | 18.7 | 6.1 | 96.8 | 104.0 | 48 | 189 | 202 |
| Cu:Phen | 1:0 | 1:1 | 1:2 | 1:3 | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| pH | 6.5 | 10.7 | 12.3 | 6.5 | 10.7 | 12.3 | 6.5 | 10.7 | 12.3 | 6.5 | 10.7 | 12.3 |
| Cu2+ | 29.9 | ε | ε | 5.1 | ε | ε | ε | ε | ε | ε | ε | ε |
| CuOH+ | 3.0 | ε | ε | 0.5 | ε | ε | ε | ε | ε | ε | ε | ε |
| Cu(OH)2 | ε a | ε | ε | ε | ε | ε | ε | ε | ε | ε | ε | ε |
| CuSO4 | 0.1 | ε | ε | ε | ε | ε | ε | ε | ε | ε | ε | ε |
| Cu2(OH)22+ | 0.1 | ε | ε | ε | ε | ε | ε | ε | ε | ε | ε | ε |
| Cu(OH)3− | ε | ε | 0.9 | ε | ε | 0.9 | ε | ε | 0.9 | ε | ε | 0.9 |
| Cu(OH)42− | ε | ε | 0.3 | ε | ε | 0.3 | ε | ε | 0.3 | ε | ε | 0.3 |
| Cu(Phen)2+ | / | / | / | 84.3 | ε | ε | 15.9 | ε | ε | 1.3 | ε | ε |
| Cu(Phen)22+ | / | / | / | 5.5 | 0.1 | ε | 72.8 | 0.4 | ε | 45.8 | 0.9 | ε |
| Cu(Phen)32+ | / | / | / | ε | 0.2 | ε | 10.6 | 1.5 | ε | 52.8 | 4.6 | ε |
| (CuPhenOH)+ | / | / | / | 4.1 | 1.4 | ε | 0.8 | 2.7 | 0.1 | 0.1 | 4.0 | 0.1 |
| (CuPhenOH)22+ | / | / | / | 0.4 | ε | ε | ε | 0.2 | ε | ε | 0.4 | ε |
| CuPhen(OH)2 | / | / | / | ε | 21.3 | 21.7 | ε | 41.7 | 43.5 | ε | 60.9 | 65.2 |
| Phen | / | / | / | ε | 76.5 | 78.2 | 6.1 | 150.1 | 156.5 | 48.5 | 219.1 | 234.7 |
| Tenorite | 66.9 | 100.0 | 98.8 | ε | 77.0 | 77.0 | ε | 53.4 | 55.3 | ε | 29.2 | 33.5 |
| Solids-Cu | 66.9 | 100.0 | 98.8 | ε | 77.0 | 77.0 | ε | 53.4 | 55.3 | ε | 29.2 | 33.5 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Walger, E.; Marlin, N.; Molton, F.; Mortha, G. Study of the Direct Red 81 Dye/Copper(II)-Phenanthroline System. Molecules 2018, 23, 242. https://doi.org/10.3390/molecules23020242
Walger E, Marlin N, Molton F, Mortha G. Study of the Direct Red 81 Dye/Copper(II)-Phenanthroline System. Molecules. 2018; 23(2):242. https://doi.org/10.3390/molecules23020242
Chicago/Turabian StyleWalger, Elsa, Nathalie Marlin, Florian Molton, and Gérard Mortha. 2018. "Study of the Direct Red 81 Dye/Copper(II)-Phenanthroline System" Molecules 23, no. 2: 242. https://doi.org/10.3390/molecules23020242
APA StyleWalger, E., Marlin, N., Molton, F., & Mortha, G. (2018). Study of the Direct Red 81 Dye/Copper(II)-Phenanthroline System. Molecules, 23(2), 242. https://doi.org/10.3390/molecules23020242





