2. Results and Discussion
Structural Assessment of CH3XXH and CH3XX− (X = S, Se). We began by using the broadest variety of DFT methods (B3LYP, B3PW91, ωB97XD, M06-2X, M08-HX) and range of basis sets (6-31G(d) to 6-311++G(3df,3pd)) used in this study to obtain optimized structures for CH
3XXH and CH
3XX
− (X = S, Se). These are the smallest homoatomic persulf/selenides considered in this present study. Due to a paucity of corresponding experimental data, benchmark optimized structures were obtained at the QCISD/6-311+G(2df,p) level of theory. For simplicity, only the key C–X, X–X, and X–H (X = S, Se) distances are discussed herein and summarized in
Table 1; MP2 data are shown in
Table S1.
Method Sensitivity to Basis Set Changes: All five DFT methods showed similar overall sensitivities to changes in the basis set from 6-31G(d) to 6-311++G(3df,3pd). For example, the smallest variations were observed for the C–X and X–H (X = S, Se) bond distances; for any given method and chemical system (be it neutral or anionic) they varied by ≤ 0.020 Å. The only exceptions occurred for the C–Se bond in CH
3SeSe
− when using M06-2X method, which varied overall by 0.027 Å, and the Se–H bond using MP2, which varied by 0.025 Å (
Table S1). Notably, for any DFT method and chemical system the C–S bond distances showed greater variation than that of C–Se bonds. The only exceptions to this trend occurred for the C–X bonds in the CH
3XX
− anions when using the B3LYP or M06-2X methods. However, the reverse trend was observed for the X–H bonds; optimized Se–H bond lengths are more sensitive to basis set choice than S–H bonds (
Table 1 and
Table S1).
For all chemical systems (both neutral and anionic), regardless of the choice of DFT method, the largest variations upon changing basis set were observed in their X–X bonds. For CH
3SSH and CH
3SS
− it was found that the S–S bond distances varied by 0.032–0.043 and 0.044–0.051 Å, respectively. Meanwhile, in the corresponding CH
3SeSeH and CH
3SeSe
− species the Se–Se bond distances varied by 0.024–0.029 and 0.023–0.029 Å, respectively, except for using MP2 where the variation exceeded the trend for DFT data (
Table S1). That is, except for Se–H bonds, those bonds involving sulfur (i.e., C–X and X–X) in CH
3XXH and CH
3XX
− (X = S, Se) are most sensitive to the choice of basis set.
Effects of Increasing Basis Set Size: As can be seen in
Table 1, for all DFT methods considered and for both neutral CH
3XXH and anionic CH
3XX
− (X = S, Se), similar trends were generally observed upon increasing the basis set from double- to triple-zeta, and then subsequently by inclusion of diffuse and polarization functions.
For instance, increasing the basis set from 6-31G(d) to 6-311++G(3df,3pd) generally caused a systematic shortening in the X–H and C–X bonds. Notably, one does not need to increase the basis set significantly in order to get reasonable agreement with the corresponding values obtained at the QCISD/6-311+G(2df,p) level of theory. In fact, upon changing the basis set from 6-31G(d) to 6-311G(d) (i.e., double- to triple-zeta) resulted in their lengths differing from the benchmark values by ≤ 0.015 Å. Further increases in basis set size by inclusion of diffuse (i.e., 6-311G(d,p) to 6-311+G(d,p)) or f- and/or d-polarization functions on heavy atoms or hydrogen resulted in only minor individual decreases.
In contrast, modifying the basis set showed quite different trends for the X–X (X = S, Se) bonds. For instance, for all methods assessed, improving the basis set from 6-31G(d) to 6-311G(d) resulted in an increase in their optimized length for all chemical systems of up to 0.025 Å. The only exception occurred for the M06-2X method applied to CH3SeSe− for which the Se–Se bond shortened slightly by 0.008 Å. Notably, in CH3SSH and CH3SS− the observed ranges of bond lengthening were 0.009–0.015 and 0.014–0.027 Å, respectively, with the largest increases observed when using the B3LYP method. That is to say, the anionic persulfide is more sensitive to basis set changes than the neutral hydropersulfide. In contrast, for CH3SeSeH and CH3SeSe− the observed increases were quite similar lying in the range of 0.017–0.025 Å (except where noted above). The further inclusion of p-functions on hydrogen (i.e., 6-311G(d) to 6-311G(d,p)) or diffuse functions on heavy atoms (i.e., 6-311G(d,p) to 6-311+G(d,p)) had negligible effect on the X–X bond lengths in both the neutral CH3XXH and anionic CH3XX− (X = S, Se) systems.
In general, more significant changes in the X–X lengths were observed upon inclusion of either a second set of d- or a set of f-functions on heavy atoms (i.e., 6-311G(d,p) to 6-311G(2d,p) or 6-311G(df,p)). Specifically, for both these basis set changes the S–S bonds in CH3SSH and CH3SS− shortened by 0.010 to 0.017 Å, except for MP2 where the distance increases for the 6-311G(2d,p) basis set. In contrast, for the corresponding selenium containing systems, the inclusion of a second set of d-functions on heavy atoms marginally lengthened Se–Se bonds by ≤ 0.004 Å, while the inclusion of a set of f-functions on heavy atoms shortened the Se–Se bonds by 0.010–0.017 Å. Combining or adding further diffuse and polarization functions by use of the 6-311+G(2df,p) or 6-311++G(3df,3pd) basis sets respectively, resulted in all DFT methods except B3LYP giving X–X (X = S, Se) bond lengths that were markedly shorter than their corresponding benchmark values. In the case of B3LYP (which overestimates the length of these bonds) increasing the basis set to 6-311+G(2df,p) or 6-311++G(3df,3pd) was in fact required in order to get good agreement with the benchmark values.
Importantly, for all DFT methods that were considered (with the exception of B3LYP), the 6-311G(2d,p) and 6-311G(df,p) basis sets gave optimized C–X, X–X, and X–H distances for CH
3XXH and CH
3XX
− (X = S) that were in closest general agreement with their corresponding benchmark values. Meanwhile, for the corresponding Se analogues, the best performing basis sets were generally 6-311G(d), 6-311G(d,p), 6-311+G(d,p), and 6-311G(2d,p). It is also noted that the M08-HX method does not offer much if any improvement over M06-2X. In fact, when Se is in the system, it slightly underestimates the bond lengths. As such, subsequent tables showing optimized parameters will only include results obtained using the B3PW91, ωB97XD and M06-2X methods in combination with the identified preferred basis sets. It should be noted that, for completeness, the corresponding values for the other methods are included in the
Supplementary Materials. Since MP2 trends were mostly similar to DFT, we do not include results obtained with this method.
Effect of Conjugation: CH2CHXXH and CH2CHXX− (X = S, Se). These model systems were examined to gain insights into the influence of conjugation on the neutral and anionic persulfide and perselenide groups. Based on the results obtained for the CH
3XX
−/H systems, optimized structures were obtained using only the B3PW91, ωB97XD, and M06-2X methods in combination with the 6-311+G(d,p), 6-311G(2d,p), and 6-311G(df,p) basis sets. The most significant changes were observed in their C–X and X–X bonds, hence only these optimized values are shown in
Table 2.
Table S2 includes the full set of optimized parameters with all DFT functionals and the X–H bonds.
In general, upon changing basis sets and methods similar trends were observed as for the CH3XX−/H systems, though with some key differences. For instance, for X = S, the closest agreement of the optimized bond lengths with the benchmark values was obtained for the 6-311G(df,p) and 6-311G(2d,p) basis sets. In contrast, for X = Se the X–X bond is more sensitive to the choice of basis set. In particular, for CH2CHSeSe− the best agreement with the benchmark values are obtained using the 6-311+G(d,p) or 6-311G(2d,p) basis sets; inclusion of f-functions results in too short an Se–Se bond. Meanwhile, for neutral CH2CHSeSeH it is essential to include f-functions in order to obtain good agreement with the corresponding benchmark values.
Comparison of the optimized bond lengths in CH
3XX
−/H and CH
2CHXX
−/H (X = S, Se) shows that for all species, at all levels of theory, the optimized C–X distance in CH
2CHXX
−/H is 0.04–0.05 Å shorter than in the corresponding CH
3XX
−/H analogue (cf.
Table 1). This is also observed when structures are optimized at the M06-2X/aug-cc-pVTZ level of theory (data not shown). In contrast, the X–X bond lengths in CH
2CHXX
−/H (X = S, Se) are all within ±0.01 Å of their optimized values in the corresponding CH
3XX
−/H analogue. When X = S the X–X bond in the anion is 0.017 Å longer in the conjugated species compared to 0.023 Å in the alkyl terminated species. It is noted that this again indicates that deprotonation of RXXH (X = S, Se) causes the X–X bond to lengthen, although conjugation lessens the bond lengthening upon going to the anion.
Effects of Mixed Sulfur/Selenium: CH3XYH (X = S, Se; Y = Se, S). We then considered mixed sulfur/selenide species for which selected optimized parameters are given in
Table 3. Full optimized parameters for all DFT methods and basis sets are shown in
Table S3.
For all four mixed species the optimized lengths of the C–X bond (X = S, Se) are all within 0.01 Å of those obtained for their analogous persulfide or perselenide (i.e., CH
3XXH (X = S, Se)) at the same level of theory (cf.
Table 1). Thus, a similar method and basis set trends were also observed and do not require further detailed discussion. However, it should be noted that, while the X–Y bond lengths in the neutral species CH
3SSeH and CH
3SeSH are close to each other, the bond is consistently predicted to be marginally shorter (<0.01 Å) in CH
3SSeH. The changes observed in the CH
3X–Y bond length upon deprotonation (
i.e., CH
3XYH to CH
3XY
−), depends on whether S or Se is the terminal atom. When S is the terminal atom, upon deprotonation of the thiol group the Se–S bond lengthens marginally by <0.01 Å. In contrast, when Se is the terminal atom deprotonation causes the S–Se bond to lengthen by ≥0.04 Å. The larger impact of deprotonating a selenol versus thiol group is also seen in the species shown in
Table 1 and
Table 2, though to a smaller extent. We found that when the sulfur atom is in the middle of the chain, it has a larger Mulliken charge compared to selenium (−0.23 vs. −0.13) in the anionic species. By comparison, the charge of the terminal chalcogen is the same in both species. Thus, repulsive interactions cause the bond length to increase when sulfur is in the center of the chain.
Extending a Sulfide: CH3SSSH and CH2CHSSSH. Polysulfides, but to-date not polyselenides, have been suggested to be potentially biochemically important. Thus, for completeness, we considered the effect of extending the persulfide chain. Specifically, the simplest alkyl- and conjugate-containing trisulfides, CH
3SSSH and CH
2CHSSSH, were examined. The C–S
1 bond in all species had very similar optimized lengths (see
Table S4 in the
Supplementary Materials), for all methods and basis sets used, to those obtained for the analogous alkyl persulfides (cf.
Table 1); the observed shortened C–X bond in the conjugated neutral and anionic persulfides (
Table 2) was not observed in CH
2CHSSSH, further evidence that it is due to delocalization across the persulf/selenide and conjugated R-group (see
Supplementary Materials). Thus, in
Table 4 only the optimized lengths of the S–S bonds are given.
As can be seen in
Table 4, the B3PW91 method gives the worst agreement with the benchmark values, especially for the deprotonated (anionic) species with errors of up to 0.07 Å. Furthermore, for any DFT method, the largest errors in the optimized S
i–S
j bond lengths are observed upon use of the 6-311+G(d,p) basis set. The best agreement is instead obtained using the M06-2X and ωB97XD methods, the former performing slightly better in conjunction with the 6-311G(2d,p) basis set. Indeed, their errors lie in the ranges of 0.000–0.011 Å and −0.003–0.009 Å, respectively.
Notably, in CH
3SSSH and CH
2CHSSSH, the CS
1–S
2 bond is predicted to be shorter than the S
2–S
3H bond by >0.02 and <0.01 Å, respectively. However, upon deprotonation of the terminal thiol group in each, i.e., formation of CH
3SSS
− and CH
2CHSSS
−, the CS
1–S
2 bond lengthens significantly by ~0.05 Å, from ~2.06 and 2.07 Å in CH
3SSSH and CH
2CHSSSH, respectively, to approximately 2.1 and 2.12 Å. In contrast, the S
2–S
3 bond in CH
3SSS
− and CH
2CHSSS
− has shortened by 0.01–0.02 Å. Mulliken charges on S
1 were found to decrease more than they do on S
2 upon deprotonation in the benchmark calculation, for both alkyl and conjugated polysulfur species. However, the difference between the two was small compared to the large charge on S
3; it becomes much more negative upon deprotonation. This indicates that there is some degree of charge delocalization along the sulfur chain (
Table S5).
Obtaining Reliable and Accurate Thermochemistry for CH3SSH and CH2CHSSH. Two of the most common goals when applying computational methods to the study of chemical problems are obtaining reliable and accurate optimized structures and thermochemical data. For biochemical or related problems, common reactions often require knowledge of proton affinities (PAs), gas phase basicities (GPBs), hydrogen affinities (HAs) and bond dissociation enthalpies (BDEs). Furthermore, given the size of the systems often encountered there is simultaneously considerable interest in identifying a DFT-based approach for calculating such properties. Thus, having assessed the use of DFT methods for the accurate optimization of structures, we also assessed the ability of the DFT methods B3LYP, B3PW91, ωB97XD, M06-2X, M08-HX, in combination with a range of basis sets to provide reliable and accurate biochemically-relevant thermochemical data.
Given the poor performance of B3LYP and minimal improvement of M08-HX over M06-2X in obtaining reliable structures, vide supra, here we only report the performance of B3PW91, ωB97XD, and M06-2X, unless otherwise noted. Furthermore, we have focused on reporting basis sets that for such systems have been previously used (e.g., 6-31G(d)), shown herein to be most consistently reliable (i.e., 6-311G(2d,p)), or often used for calculating accurate thermochemistry (i.e., 6-311+G(2df,p) and 6-311++G(3df,3pd)). The results obtained are summarized in
Table 5, although the data for all functionals and basis sets that were studied are shown in
Tables S6–S11.
The benchmark values were again obtained using the QCISD/6-311+G(2df,p) level of theory. The thermochemical values obtained for the conjugated persulfides compared to the alkyl persulfides reflects the trends observed structurally. For instance, the BDE(S–S) for CH2CHSSH is slightly higher by 5.7 kJ mol−1 than that of CH3SSH, while the PA and GPB of CH2CHSS− are 23.6 and 24.4 kJ mol−1 lower, respectively, than those of CH3SS−. This is due to delocalization across the persulfide and CH2CH group in CH2CHSSH/−. Meanwhile, the hydrogen affinity of CH2CHSS• is predicted to be only marginally higher than that of CH3SS• by 0.8 kJ mol−1. Notably, we did see spin contamination in the QCISD calculations with a spin of 0.88. This may be the reason for lower energies calculated by QCISD compared to DFT methods where spin contamination was much lower (0.77).
A DFT-based model was determined to be accurate if it gave values within 10 kJ mol
−1, generally held to be experimental accuracy, of the benchmark values. From
Table 5 it can be seen that not all methods or basis set choices were reliable, nor did all thermochemical properties exhibit the same method/basis set requirement. For instance, for accurate determination of the BDE(S–S) of both CH
3SSH and CH
2CHSSH, the best performance was observed for B3PW91 and ωB97XD in combination with the 6-31G(d) basis set. The former method slightly underestimating compared to the benchmark value while the latter overestimated slightly. The M06-2X method only gave an accurate BDE(S–S) for CH
2CHSSH and again when using the 6-31G(d) basis set. In general, the use of basis sets larger than 6-31G(d) gave BDE(S–S) values that are markedly higher (14–36 kJ mol
−1) than those obtained at the QCISD/6-311+G(2df,p) level of theory.
For the PA and GPB of the CH
3SS
− and CH
2CHSS
− anions the M06-2X method again only gives accurate values when used in combination with the 6-31G(d) basis set. In contrast, the B3PW91 and ωB97XD give accurate values for all the basis sets considered. However, the triple-zeta basis sets (6-311G(2d,p), 6-311+G(2df,p), and 6-311++G(3df,3pd)) gave best agreement with calculated values within 5 kJ mol
−1 of their corresponding benchmark value (see
Table 5).
The calculated values of the hydrogen affinity of CH
3SS
• and CH
2CHSS
• follow almost the same method and basis set trends and accuracy as that observed for the PA and GPB of CH
3SS
− and CH
2CHSS
−. Namely, the B3PW91 and ωB97XD methods in conjunction with any of the basis sets considered give calculated values within ±10 kJ mol
−1. The only exception occurs when at the B3PW91/6-31G(d) level of theory for CH
2CHSS
•, which gives a HA value 10.7 kJ mol
−1 lower than the corresponding QCISD/6-311+G(2df,p) benchmark value (see
Table 5). Meanwhile, the M06-2X method is inconsistent; for CH
3SS
• only the 6-31G(d) and 6-311+G(2df,p) basis sets give values within 10 kJ mol
−1 of the benchmark values while for CH
2CHSS
• it gives good agreement for all basis sets considered herein.
Several overall trends are suggested in this examination of the performance of the DFT methods B3PW91, ωB97XD, and M06-2X, in conjunction with a range of basis sets, for the noted important thermochemical properties. In particular, the M06-2X functional is the least consistent and usually gives values that differ from the benchmark values by more than 10 kJ mol
−1. Furthermore, for all DFT functionals the values obtained using the 6-311+G(2df,p) basis set are within 4 kJ mol
−1 of the corresponding values obtained using the much larger and more expensive 6-311++G(3df,3pd) basis set. Hence, for the remainder of this report, for simplicity, only thermochemical values obtained using the B3PW91 and ωB97XD functionals in combination with basis sets no larger than 6-311+G(2df,p) are discussed, unless otherwise noted (see
Tables S6–S11 for the complete datasets).
Thermochemistry of Selenium-Containing Species. As noted above in the structural assessment, for selenium containing species considered herein; that is CH
3SeSe
•/
−/H, CH
2CHSeSe
•/
−/H, CH
3SSe
•/
−/H, and CH
3SeS
•/
−/H, the smallest consistently reliable basis set was 6-311+G(d,p), though with exceptions as noted. Hence, for these species we have limited our discussion herein to results obtained using B3PW91 and ωB97XD in conjunction with the 6-311+G(d,p) and 6-311+G(2df,p) basis sets. The results are shown in
Table 6.
As for the analogous purely sulfur-containing species (cf.
Table 5), the calculated PAs and GPBs of all species obtained using B3PW91 or ωB97XD with either basis set choice gives values within 10 kJ mol
−1 of the corresponding benchmark values. A similar consistency is observed for the calculated HAs, though with some exceptions. In particular, at the ωB97XD/6-311+G(d,p) level of theory the calculated HAs of CH
2CHSeSe
• and CH
3SSe
• are 11.2 and 12.4 kJ mol
−1, respectively, higher than their corresponding QCISD/6-311+G(2df,p) values. Again, as seen in
Table 5, the calculated RX—YH BDEs of all species are generally markedly overestimated by 12.7–26.3 kJ mol
−1 using either DFT method and basis set. Only three values fall within the desired 10 kJ mol
−1 error margin and all occur for the mixed chalcogen species; all using the 6-311+G(d,p) basis set.
The thermochemical values provided in
Table 5 and
Table 6 show several key differences between persulfides and perselenides and the mixed chalcogens. These are most clearly and simply illustrated by examination of the calculated benchmark values. In particular, increasing the number of Se atoms in an RX–YH bond reduces its BDE as shown by comparing those of CH
3SSH (236.3 kJ mol
−1), CH
3SSeH (218.7 kJ mol
−1), CH
3SeSH (223.2 kJ mol
−1), and CH
3SeSeH (202.7 kJ mol
−1). In addition, a conjugated group adjacent to the RX–XH group increases its BDE slightly by 3–6 kJ mol
−1. Meanwhile, the HAs are reasonably consistent and depend primarily on whether the formal radical terminal is a sulfur or selenium. For the former, all values lie within the range 282.7 kJ mol
−1 (CH
3SS
•) to 286.1 kJ mol
−1 (CH
3SeS
•), while the latter are notably lower, between 265.9 kJ mol
−1 (CH
2CHSeSe
•) and 267.3 kJ mol
−1 (CH
3SeSe
• and CH
3SSe
•).
Comparison of the PAs and GPBs of these perselenides and mixed per-sulf/selenides with their analogous persulfides (cf.
Table 5), shows several interesting trends. It is noted that, for simplicity, as the observed trends were the same for all the DFT methods, the values discussed here refer to those obtained at the benchmark level of theory. Firstly, systematically increasing the occurrence of Se in a per-chalcogenide group decreases their PA and GPB values. For instance, the PAs of CH
3SS
−, CH
3SeS
−, CH
3SSe
−, and CH
3SeSe
− are 1445.4 kJ mol
−1, 1438.9 kJ mol
−1, 1411.3 kJ mol
−1, and 1404.3 kJ mol
−1. Simultaneously, their GPB values decrease from 1415.1 kJ mol
−1, to 1408.6 kJ mol
−1 and 1381.1 kJ mol
−1, to 1374.0 kJ mol
−1. In addition, replacing CH
3- with CH
2CH- decreases the PA and GPB values of the pure persulfides and perselenides by 24–25 and 17–18 kJ mol
−1, respectively. This perhaps reflects a larger delocalization when a conjugated group is adjacent
and that the effect is less for the selenides.
Extending the persulfides to trisulfides. We also examined the effect of extending the persulfide group by an additional sulfur to a trisulfide; specifically, we considered key bio-relevant thermochemistry of CH
3SSS
•/
−/H and CH
2CHSSS
•/
−/H. Based on the trends observed for persulfide systems, thermochemical values were obtained using the B3PW91 and ωB97XD DFT methods in combination with the 6-311+G(2df,p) basis set, and again at the QCISD/6-311+G(2df,p) benchmark level of theory (all other data shown in
Table S9). As can be seen in
Table 7, both DFT methods give thermochemical results in generally good agreement with the benchmark values with B3PW91 slightly preferred, although the differences in average errors are relatively small.
Comparison of the calculated benchmark values of the trisulfides with those of the corresponding persulfides (cf.
Table 6) shows that the BDE of RS
1S
2—S
3H (i.e., the BDE of the terminal S
2–S
3 bond), where R = CH
3- and CH
2CH-, decreases significantly by 53.3 and 56.3 kJ mol
−1, respectively. Similarly, their calculated PAs and GPBs decrease markedly by 31–33 and 18–19 kJ mol
−1 for R = CH
3- and CH
2CH-, respectively. As a result of these changes the RSS–SH BDE, and PAs and GPBs of RSSS
− all lie within a narrower range (≤13 kJ mol
−1) of each other. This perhaps reflects in part a decrease in the influence of the R group on the increasingly removed S–SH bond. In contrast, the hydrogen affinities of CH
3SSS
• and CH
2CHSSS
• are 6.7 and 11.5 kJ mol
−1 higher than that of their corresponding persulfide analogues with values of 289.4 and 295.0 kJ mol
−1, respectively.
Cysteine-derived polysulfides: CysSSnH (n = 1–3). Within biological systems, as noted above, per- and polysulfide derivatives of cysteine play important roles. Hence, we also considered the structures and thermochemistry of such species using the ωB97XD/6-311G(2d,p) level of theory. This level was selected as it emerged in our earlier study as the most able to provide reliable structures and thermochemistry for related model systems (see above). Selected parameters of optimized structures obtained for
CysSS
nH (
n = 0–3) and
CysSS
n− (
n = 0–3) are provided in
Table 8 and representative optimized structures are shown in
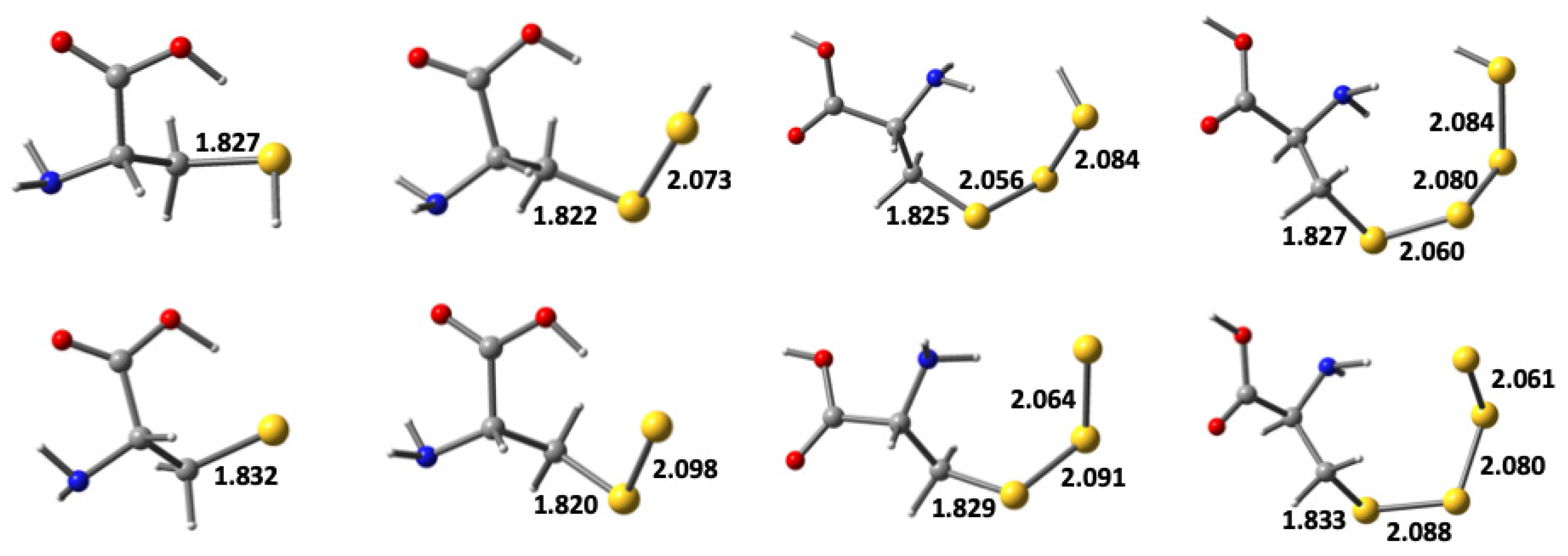
Figure 1, at this level of theory.
As can be seen, for both the neutral and deprotonated derivatives, the
r(C−S) bond length decreases slightly upon forming a perthiol/sulfide group, but then gradually lengthens, essentially returning to its length in cysteine, as one goes to the corresponding trithiol/sulfide and tetrathiol/sulfide derivatives. It is also noted that for both
CysSSH and
CysSS
−, the C–S and S–S bond lengths are in close agreement with the corresponding values obtained at the same level of theory for CH
3SSH (1.816 and 2.067 Å) and CH
3SS
− (1.816 and 2.092 Å), respectively (cf.
Table 1). In addition, for the neutral polysulfides the S–S bond in the chain that is farthest removed from the alkyl group has the longest length, with the S–S bonds being sequentially shorter the closer they are to the alkyl group. In contrast, for the corresponding anionic deprotonated series (
CysSS
n−,
n = 0–3), the opposite trend is observed; the S–S bond in the chain farthest from the alkyl group is shortest, and they get longer the closer they are to the alkyl group. In the latter species this trend may reflect a diminishing effect of the negative charge on the terminal sulfur atom the further removed the bond.
Thermochemistry of cysteine-derived polysulfides. As for the other species considered above, we calculated the RS
n–SH homolytic BDEs for
CysSS
nH (
n = 1–3), the PAs and GPBs of the
CysSS
n− (
n = 0–3) series of derivatives, and the hydrogen affinities (HAs) of the
CysSS
n• (
n = 0–3) series of species. However, all values were again only obtained using the chosen ωB97XD/6-311G(2d,p) level of theory. The results obtained are shown in
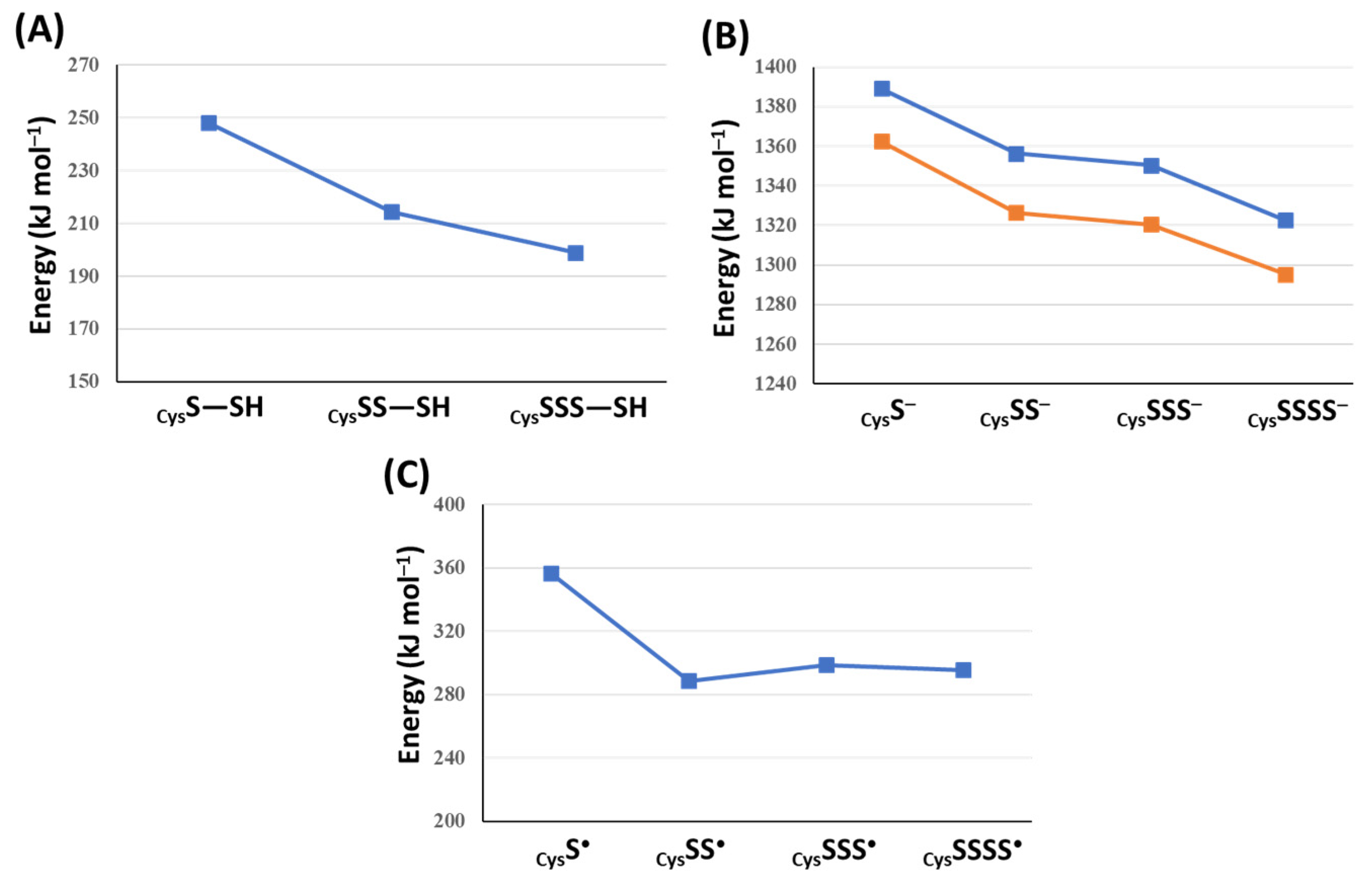
Figure 2A–C.
The calculated RS
n–SH (
n = 1–3) homolytic bond dissociation enthalpies, that is, the BDE of the terminal S–SH bond in the perthiol chain decreases by 33.7 kJ mol
−1 from 248.1 to 214.4, upon going from the perthiol (
n = 1) to trithiol (
n = 2). It then decreases a further 15.4 kJ mol
−1 to 199 kJ mol
−1 upon increasing to the chain further to the tetrathiol (
n = 3);
Figure 2A. This trend suggests that while the strength of the terminal S–SH bond does weaken as the chain is lengthened, it approaches a limiting value below 199.0 kJ mol
−1, although possibly not too much lower than that. In contrast, as seen in
Figure 2C, the hydrogen affinities of the
CysSS
n• (
n = 0–3) species decrease significantly by 67.8 kJ mol
−1 upon going from a cysteinyl thiyl radical (
n = 0; 356.5 kJ mol
−1) to the perthiyl radical (
n = 1; 288.7 kJ mol
−1). However, extending the chain further to
n = 2 or
n = 3 results in a slight increase in the HAs to 298.7 kJ mol
−1 and 295.6 kJ mol
−1, respectively. This suggests that at least for HAs of the polysulfur radicals, they are reasonably constant for the perthiyl and beyond.
Figure 2B shows that as the polysulfide chain is extended from the cysteine thiolate to
CysSSSS
− both the calculated PAs and GPBs decrease. However, while there are significant decreases of 33.0 (PA) and 35.9 (GPB) kJ mol
−1 going from the thiolate (
CysS
−) to perthiolate (
CysSS
−), this does not appear to continue upon extending the chain further. Indeed, extending the chain to
CysSSS
− results in only comparatively small further decreases of 5.8 and 5.9 kJ mol
−1 to 1350.5 and 1320.6 kJ mol
−1. However, upon extending the chain to
CysSSSS
−, larger decreases in both the PA and GPB are again observed; they decrease by 27.9 and 25.4 kJ mol
−1 to 1322.6 and 1295.2 kJ mol
−1. This may in part reflect that in the cysteinyl polysulfide derivatives, weak intramolecular hydrogen bonding was observed in some optimized structures between the terminal S
− center and the cysteine’s amino group. This interaction would also help decrease PA and GPB values. It should also be noted that comparison of the calculated PAs and GPBs of the cysteine-derived sulfides
CysSS
− and
CysSSS
− with that of their smaller corresponding analogs CH
3SS
− (cf.
Table 5) and CH
3SSS
− (cf.
Table 7) shows that that those of the former two species are markedly lower by 60-90 kJ mol
−1. Thus, overall, these results suggest that in biological systems, extending the polysulfide chain will increase its likelihood of being deprotonated and that hydrogen bonding may help to stabilize such anions.