Identification of Maize Kernel Vigor under Different Accelerated Aging Times Using Hyperspectral Imaging
Abstract
1. Introduction
2. Results and Discussion
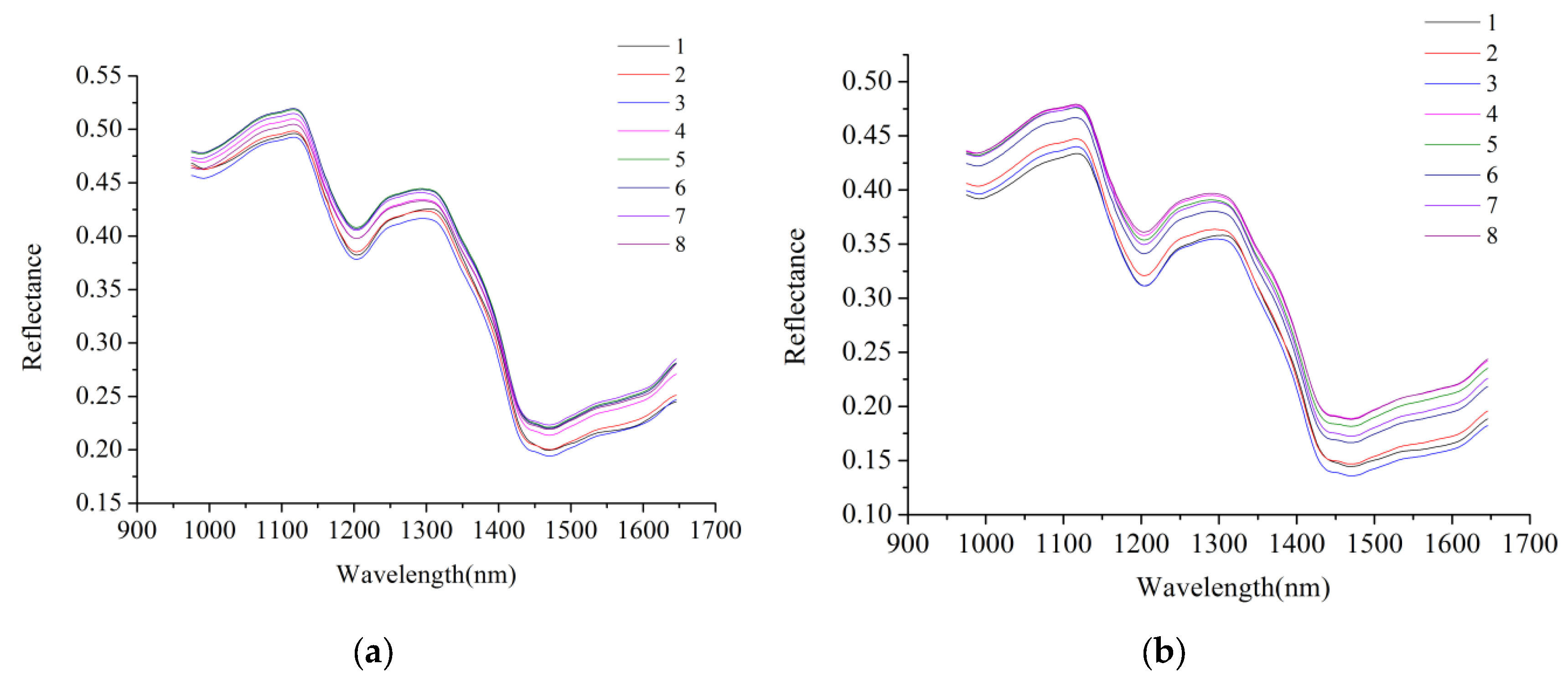
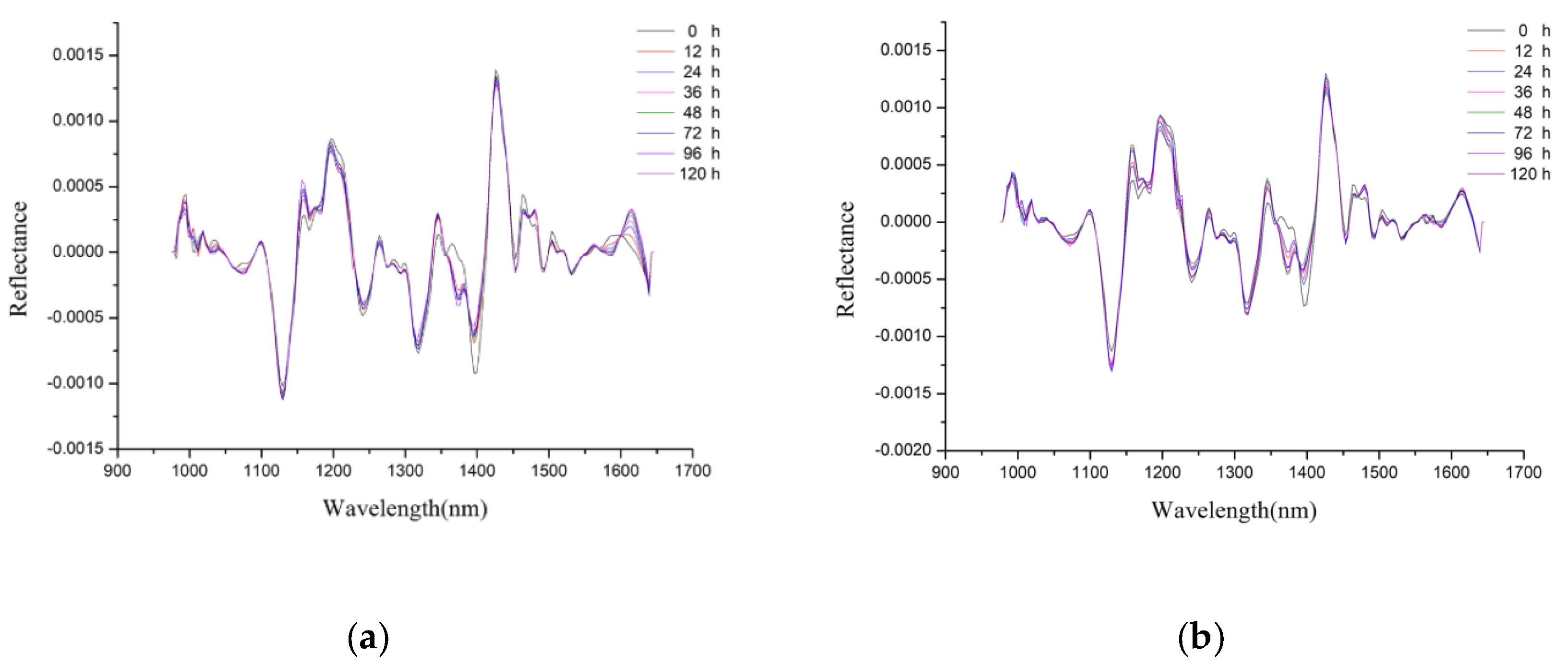
2.1. Spectral Profile
2.2. PCA Analysis
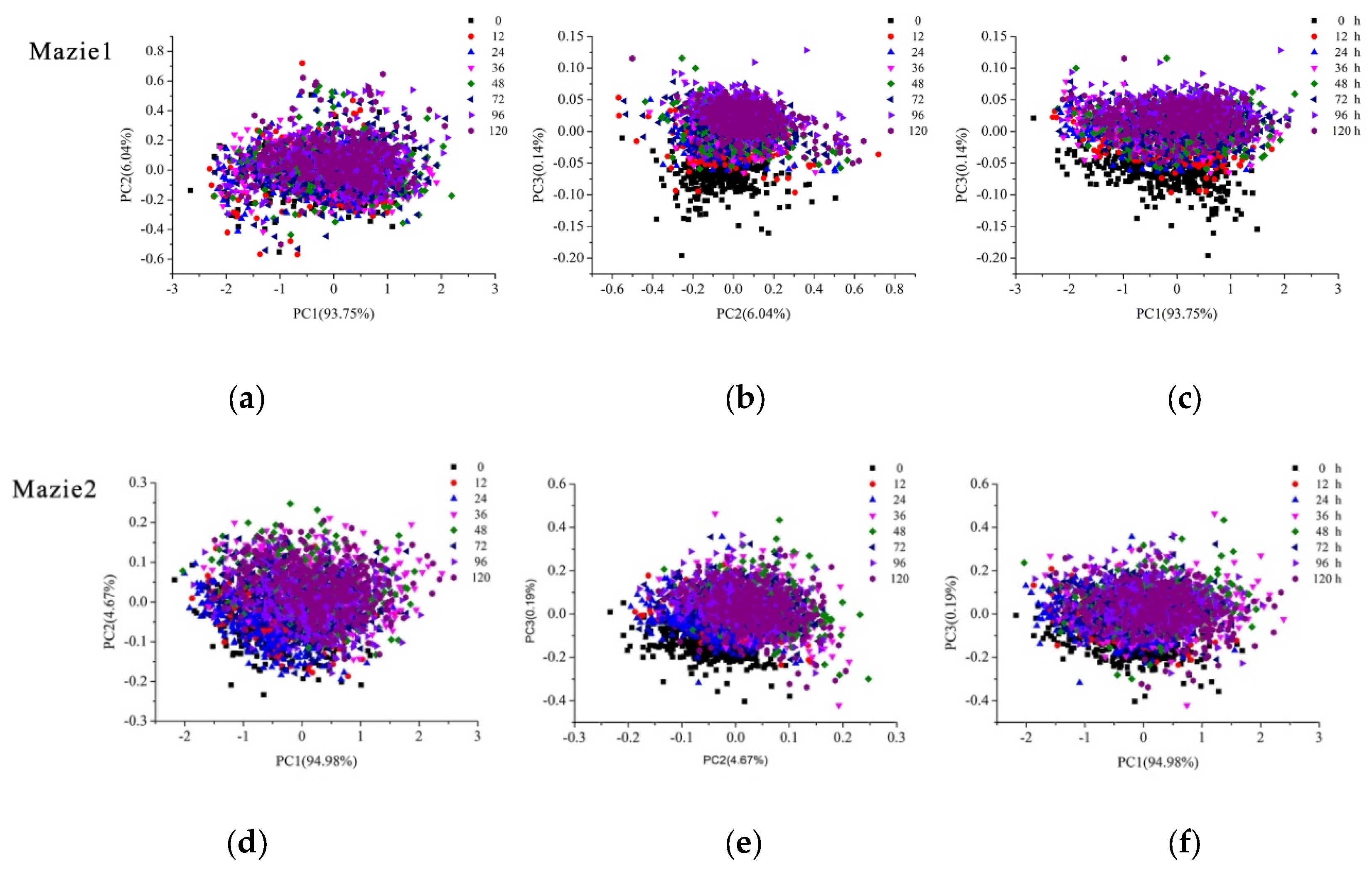
2.2.1. Pixel-Wise PCA Scores Visualization
2.2.2. Object-Wise PCA Scores Scatter Plots Analysis
2.3. Classification Models Based on Full Spectra
2.4. Optimal Wavelengths Selection
2.5. Classification Models on the Optimal Wavelengths
2.6. Germination Tests Analysis
3. Materials and Methods
3.1. Sample Preparation
3.2. Hyperspectral Imaging System
3.3. Hyperspectral Image Acquisition and Calibration
3.4. Spectral Reflectance Extraction and Preprocessing
3.5. Standard Germination Tests
3.6. Data analysis Methods
3.6.1. Principal Component Analysis
3.6.2. Optimal Wavelength Selection
3.6.3. Discriminant Model
3.6.4. Significance Test
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Xin, X.; Lin, X.H.; Zhou, Y.C.; Chen, X.L.; Liu, X.; Lu, X.X. Proteome analysis of maize seeds: The effect of artificial ageing. Physiol. Plant. 2011, 143, 126–138. [Google Scholar] [CrossRef] [PubMed]
- Woltz, J.M.; Tekrony, D.M. Accelerated aging test for corn seed. Seed Technol. 2001, 23, 21–34. [Google Scholar]
- Williams, P.; Manley, M.; Fox, G.; Geladi, P. Indirect detection of Fusarium verticillioides in maize (Zea maize L.) kernels by NIR hyperspectral imaging. J. Near Infrared Spectrosc. 2010, 18, 49–58. [Google Scholar] [CrossRef]
- Bittencourt, S.R.M.D.; Grzybowski, C.R.D.S.; Panobianco, M.; Vieira, R.D. Alternative methodology for the accelerated aging test for corn seeds. Ciênc. Rural 2012, 42, 1360–1365. [Google Scholar] [CrossRef]
- Han, Z.; Ku, L.; Zhang, Z.; Zhang, J.; Guo, S.; Liu, H.; Zhao, R.; Ren, Z.; Zhang, L.; Su, H. QTLs for seed vigor-related traits identified in maize seeds germinated under artificial aging conditions. PLoS ONE 2014, 9, 92535. [Google Scholar] [CrossRef] [PubMed]
- Gelmond, H.; Luria, I.; Woodstock, L.W.; Perl, M. The effect of accelerated aging of sorghum seeds on seedling vigour. J. Exp. Bot. 1978, 29, 489–495. [Google Scholar] [CrossRef]
- De Jesus Souza, F.I.F.; Devilla, I.A.; de Souza, R.T.G.; Teixeira, I.R.; Spehar, C.R. Physiological quality of quinoa seeds submitted to different storage conditions. Afr. J. Agric. Res. 2016, 11, 1299–1308. [Google Scholar] [CrossRef]
- Mcdonough, C.M.; Floyd, C.D.; Waniska, R.D.; Rooney, L.W. Effect of accelerated aging on maize, sorghum, and sorghum meal. J. Cereal Sci. 2004, 39, 351–361. [Google Scholar] [CrossRef]
- Ambrose, A.; Kandpal, L.M.; Kim, M.S.; Lee, W.H.; Cho, B.K. High speed measurement of corn seed viability using hyperspectral imaging. Infrared Phys. Technol. 2016, 75, 173–179. [Google Scholar] [CrossRef]
- Williams, P.; Geladi, P.; Fox, G.; Manley, M. Maize kernel hardness classification by near infrared (NIR) hyperspectral imaging and multivariate data analysis. Anal. Chim. Acta 2009, 653, 121–130. [Google Scholar] [CrossRef] [PubMed]
- Mcgoverin, C.M.; Engelbrecht, P.; Geladi, P.; Manley, M. Characterisation of non-viable whole barley, wheat and sorghum grains using near-infrared hyperspectral data and chemometrics. Anal. Bioanalyt. Chem. 2011, 401, 2283–2289. [Google Scholar] [CrossRef] [PubMed]
- Nansen, C.; Zhao, G.; Dakin, N.; Zhao, C.; Turner, S.R. Using hyperspectral imaging to determine germination of native Australian plant seeds. J. Photochem. Photobiol. B 2015, 145, 19–24. [Google Scholar] [CrossRef] [PubMed]
- Kandpal, L.M.; Lohumi, S.; Kim, M.S.; Kang, J.S.; Cho, B.K. Near-infrared hyperspectral imaging system coupled with multivariate methods to predict viability and vigor in muskmelon seeds. Sens. Actuators B Chem. 2016, 229, 534–544. [Google Scholar] [CrossRef]
- Liu, C.; Wei, L.; Lu, X.; Wei, C.; Yang, J.; Lei, Z. Nondestructive determination of transgenic Bacillus thuringiensis rice seeds (Oryza sativa L.) using multispectral imaging and chemometric methods. Food Chem. 2014, 153, 87–93. [Google Scholar] [CrossRef] [PubMed]
- Williams, P.J.; Kucheryavskiy, S. Classification of maize kernels using NIR hyperspectral imaging. Food Chem. 2016, 209, 131–138. [Google Scholar] [CrossRef] [PubMed]
- Fiore, A.D.; Reverberi, M.; Ricelli, A.; Pinzari, F.; Serranti, S.; Fabbri, A.A.; Bonifazi, G.; Fanelli, C. Early detection of toxigenic fungi on maize by hyperspectral imaging analysis. Int. J. Food Microbiol. 2010, 144, 64–71. [Google Scholar] [CrossRef] [PubMed]
- Ravikanth, L.; Singh, C.B.; Jayas, D.S.; White, N.D.G. Classification of contaminants from wheat using near-infrared hyperspectral imaging. Biosyst. Eng. 2015, 135, 73–86. [Google Scholar] [CrossRef]
- Weinstock, B.A.; Janni, J.; Hagen, L.; Wright, S. Prediction of oil and oleic acid concentrations in individual corn (Zea mays L.) kernels using near-infrared reflectance hyperspectral imaging and multivariate analysis. Appl. Spectrosc. 2006, 60, 9. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.H.; Lu, F.M.; Chang, Y.C. Development of a near-infrared imaging system for determination of rice moisture. Cereal Chem. 2006, 83, 498–504. [Google Scholar] [CrossRef]
- Caporaso, N.; Whitworth, M.B.; Fisk, I.D. Protein content prediction in single wheat kernels using hyperspectral imaging. Food Chem. 2017, 240, 32–42. [Google Scholar] [CrossRef] [PubMed]
- Wei, W.; Heitschmidt, G.W.; Windham, W.R.; Peggy, F.; Xinzhi, N.; Xuan, C. Feasibility of detecting aflatoxin B1 on inoculated maize kernels surface using Vis/NIR hyperspectral imaging. J. Food Sci. 2014, 80. [Google Scholar] [CrossRef]
- Wang, L.; Pu, H.; Sun, D.W.; Liu, D.; Wang, Q.; Xiong, Z. Application of hyperspectral imaging for prediction of textural properties of maize seeds with different storage periods. Food Anal. Methods 2015, 8, 1535–1545. [Google Scholar] [CrossRef]
- Kafle, G.K.; Khot, L.R.; Jarolmasjed, S.; Si, Y.; Lewis, K. Robustness of near infrared spectroscopy based spectral features for non-destructive bitter pit detection in honeycrisp apples. Postharvest Biol. Technol. 2016, 120, 188–192. [Google Scholar] [CrossRef]
- Kamruzzaman, M.; Elmasry, G.; Sun, D.W.; Allen, P. Application of NIR hyperspectral imaging for discrimination of lamb muscles. J. Food Eng. 2011, 104, 332–340. [Google Scholar] [CrossRef]
- Qin, J.; Burks, T.F.; Kim, M.S.; Chao, K.; Ritenour, M.A. Detecting citrus canker by hyperspectral reflectance imaging and PCA-based image classification method. In Proceedings of the SPIE—The International Society for Optical Engineering, Orlando, FL, USA, 15 April 2008. [Google Scholar]
- Liu, D.; Ma, J.; Sun, D.-W.; Pu, H.; Gao, W.; Qu, J.; Zeng, X.-A. Prediction of color and pH of salted porcine meats using visible and near-infrared hyperspectral imaging. Food Bioprocess Technol. 2014, 7, 3100–3108. [Google Scholar] [CrossRef]
- Jiang, H.; Chen, Q. Development of electronic nose and near infrared spectroscopy analysis techniques to monitor the critical time in SSF process of feed protein. Sensors 2014, 14, 19441–19456. [Google Scholar] [CrossRef] [PubMed]
- Kamruzzaman, M.; Barbin, D.; Elmasry, G.; Sun, D.W.; Allen, P. Potential of hyperspectral imaging and pattern recognition for categorization and authentication of red meat. Innov. Food Sci. Emerg. Technol. 2012, 16, 316–325. [Google Scholar] [CrossRef]
- Zhang, C.; Feng, X.; Wang, J.; Liu, F.; He, Y.; Zhou, W. Mid-infrared spectroscopy combined with chemometrics to detect Sclerotinia stem rot on oilseed rape (Brassica napus L.) leaves. Plant Methods 2017, 13, 39. [Google Scholar] [CrossRef] [PubMed]
- Devos, O.; Ruckebusch, C.; Durand, A.; Duponchel, L.; Huvenne, J.-P. Support vector machines (SVM) in near infrared (NIR) spectroscopy: Focus on parameters optimization and model interpretation. Chemom. Intell. Lab. Syst. 2009, 96, 27–33. [Google Scholar] [CrossRef]
- Zhang, L.D.; Su, S.G.; Wang, L.S.; Li, J.H.; Yang, L.M. Study on application of fourier transformation near-infrared spectroscopy analysis with support vector machine (SVM). Spectrosc. Spect. Anal. 2005, 25, 33–35. [Google Scholar] [CrossRef]
- Campsvalls, G.; Gómezchova, L.; Calpemaravilla, J.; Soriaolivas, E.; Martínguerrero, J.D.; Moreno, J. Support vector machines for crop classification using hyperspectral data. Lecture Notes Comput. Sci. 2003, 2652, 134–141. [Google Scholar]
- Dai, Q.; Cheng, J.H.; Sun, D.W.; Pu, H.; Zeng, X.A.; Xiong, Z. Potential of visible/near-infrared hyperspectral imaging for rapid detection of freshness in unfrozen and frozen prawns. J. Food Eng. 2015, 149, 97–104. [Google Scholar] [CrossRef]
Sample Availability: Not available. |
| Sample Variety | C 1 | G 2 | Cal. 3 (%) | Pre. 4 (%) | Cv. 5 |
|---|---|---|---|---|---|
| Maize 1 | 256.00 | 1.74 | 81.53 | 68.15 | 58.13 |
| Maize 2 | 256.00 | 3.03 | 78.47 | 60.16 | 63.84 |
| Maize Mixed | 256.00 | 5.28 | 73.43 | 59.90 | 57.23 |
| Sample Variety | Sample Number | Pre. | Accuracy (%) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | ||||
| Maize 1 | Cal. | 1 (400) | 400 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 100.00 |
| 2 (400) | 0 | 384 | 16 | 0 | 0 | 0 | 0 | 0 | 96.00 | ||
| 3 (400) | 0 | 14 | 356 | 26 | 3 | 0 | 1 | 0 | 89.00 | ||
| 4 (400) | 0 | 3 | 34 | 306 | 40 | 11 | 5 | 1 | 76.50 | ||
| 5 (400) | 0 | 0 | 16 | 74 | 228 | 68 | 2 | 12 | 57.00 | ||
| 6 (400) | 0 | 0 | 4 | 25 | 76 | 261 | 8 | 26 | 65.30 | ||
| 7 (400) | 0 | 0 | 0 | 1 | 5 | 18 | 327 | 49 | 81.80 | ||
| 8 (400) | 0 | 0 | 0 | 5 | 9 | 20 | 19 | 347 | 86.80 | ||
| Pre. | 1 (200) | 199 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 99.50 | |
| 2 (200) | 1 | 150 | 47 | 0 | 0 | 2 | 0 | 0 | 75.00 | ||
| 3 (200) | 0 | 19 | 158 | 17 | 6 | 0 | 0 | 0 | 79.00 | ||
| 4 (200) | 0 | 4 | 26 | 114 | 39 | 16 | 0 | 1 | 57.00 | ||
| 5 (200) | 0 | 0 | 2 | 11 | 92 | 87 | 0 | 8 | 46.00 | ||
| 6 (199) | 0 | 1 | 2 | 22 | 66 | 100 | 3 | 5 | 50.30 | ||
| 7 (200) | 0 | 0 | 0 | 1 | 10 | 12 | 117 | 60 | 58.50 | ||
| 8 (199) | 0 | 0 | 0 | 1 | 6 | 17 | 16 | 159 | 79.90 | ||
| Maize 2 | Cal. | 1 (400) | 400 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 100.00 |
| 2 (400) | 0 | 374 | 24 | 0 | 0 | 0 | 2 | 0 | 93.50 | ||
| 3 (400) | 0 | 16 | 384 | 0 | 0 | 0 | 0 | 0 | 96.00 | ||
| 4 (400) | 0 | 0 | 0 | 279 | 37 | 27 | 10 | 47 | 69.80 | ||
| 5 (400) | 0 | 0 | 0 | 38 | 322 | 6 | 0 | 34 | 80.50 | ||
| 6 (400) | 0 | 1 | 0 | 30 | 2 | 256 | 95 | 16 | 64.00 | ||
| 7 (400) | 0 | 1 | 0 | 17 | 1 | 105 | 259 | 17 | 64.80 | ||
| 8 (400) | 0 | 1 | 0 | 79 | 53 | 22 | 8 | 237 | 59.30 | ||
| Pre. | 1 (200) | 196 | 0 | 3 | 0 | 0 | 0 | 0 | 1 | 98.00 | |
| 2 (200) | 1 | 156 | 36 | 0 | 0 | 1 | 6 | 0 | 78.00 | ||
| 3 (199) | 1 | 21 | 177 | 0 | 0 | 0 | 0 | 0 | 88.90 | ||
| 4 (200) | 0 | 2 | 0 | 90 | 36 | 23 | 10 | 39 | 45.00 | ||
| 5 (200) | 1 | 0 | 0 | 47 | 109 | 4 | 2 | 37 | 54.50 | ||
| 6 (200) | 0 | 3 | 0 | 12 | 3 | 80 | 92 | 10 | 40.00 | ||
| 7 (200) | 0 | 6 | 1 | 19 | 0 | 71 | 93 | 10 | 46.50 | ||
| 8 (200) | 0 | 1 | 0 | 71 | 41 | 16 | 10 | 61 | 30.50 | ||
| Maize mixed | Cal. | 1 (800) | 800 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 100.00 |
| 2 (800) | 0 | 654 | 134 | 0 | 0 | 8 | 0 | 4 | 81.80 | ||
| 3 (800) | 0 | 123 | 657 | 17 | 1 | 2 | 0 | 0 | 82.10 | ||
| 4 (800) | 0 | 5 | 30 | 499 | 126 | 35 | 40 | 65 | 62.40 | ||
| 5 (800) | 0 | 4 | 19 | 156 | 422 | 129 | 22 | 48 | 52.80 | ||
| 6 (800) | 0 | 7 | 4 | 52 | 70 | 480 | 110 | 77 | 60.00 | ||
| 7 (800) | 0 | 0 | 0 | 57 | 51 | 125 | 409 | 158 | 51.00 | ||
| 8 (800) | 0 | 2 | 1 | 40 | 64 | 75 | 87 | 531 | 66.40 | ||
| Pre. | 1 (400) | 394 | 2 | 3 | 0 | 0 | 1 | 0 | 0 | 98.50 | |
| 2 (400) | 4 | 246 | 130 | 0 | 2 | 11 | 3 | 4 | 61.50 | ||
| 3 (399) | 2 | 94 | 287 | 9 | 1 | 4 | 0 | 2 | 72.20 | ||
| 4 (400) | 0 | 9 | 16 | 205 | 90 | 26 | 25 | 29 | 51.30 | ||
| 5 (400) | 0 | 3 | 3 | 55 | 136 | 130 | 20 | 53 | 34.00 | ||
| 6 (399) | 0 | 3 | 5 | 36 | 64 | 187 | 74 | 30 | 46.90 | ||
| 7 (400) | 0 | 3 | 4 | 36 | 59 | 167 | 77 | 54 | 19.30 | ||
| 8 (399) | 0 | 3 | 0 | 40 | 59 | 51 | 58 | 188 | 47.10 | ||
| Sample Variety | Sample Number | Pre. | Accuracy (%) | |||
|---|---|---|---|---|---|---|
| Group 1 | Group 2 | Group 3 | ||||
| Maize 1 | Cal. | Group 1 (400) | 400 | 0 | 0 | 100.00 |
| Group 2 (800) | 0 | 770 | 30 | 96.25 | ||
| Group 3 (2000) | 0 | 57 | 1943 | 97.15 | ||
| Pre. | Group 1 (200) | 199 | 1 | 0 | 99.50 | |
| Group 2 (400) | 1 | 374 | 25 | 93.50 | ||
| Group 3 (998) | 0 | 35 | 963 | 96.49 | ||
| Maize 2 | Cal. | Group 1 (400) | 400 | 0 | 0 | 100.00 |
| Group 2 (800) | 0 | 798 | 2 | 99.75 | ||
| Group 3 (2000) | 0 | 3 | 1997 | 99.85 | ||
| Pre. | Group 1 (200) | 196 | 3 | 1 | 98.00 | |
| Group 2 (399) | 2 | 390 | 7 | 97.74 | ||
| Group 3 (1000) | 1 | 13 | 986 | 98.60 | ||
| Maize mixed | Cal. | Group 1 (800) | 800 | 0 | 0 | 100.00 |
| Group 2 (1600) | 0 | 1568 | 32 | 98.00 | ||
| Group 3 (4000) | 0 | 72 | 3928 | 98.20 | ||
| Pre. | Group 1 (400) | 394 | 5 | 1 | 98.50 | |
| Group 2 (799) | 6 | 757 | 36 | 94.74 | ||
| Group 3 (1998) | 0 | 49 | 1949 | 97.55 | ||
| Sample Variety | No. | Optimal Wavelengths (nm) |
|---|---|---|
| Maize 1 | 19 | 995, 1005, 1035, 1076, 1130, 1156, 1167, 1207, 1241, 1264, 1321, 1375, 1399, 1426, 1463, 1480, 1504, 1585, 1615 |
| Maize 2 | 18 | 1005, 1072, 1130, 1156, 1160, 1167, 1197, 1241, 1264, 1318, 1345, 1372, 1396, 1426, 1453, 1463, 1480, 1612 |
| Sample Variety | c | g | Cal. (%) | Pre. (%) | Cv. |
|---|---|---|---|---|---|
| Maize 1 | 256.00 | 27.86 | 70.47 | 57.45 | 71.31 |
| Maize 2 | 256.00 | 16.00 | 71.66 | 62.48 | 63.81 |
| Sample Variety | Sample Number | Prediction Value | Accuracy (%) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | ||||
| Maize 1 | Cal. | 1 (400) | 400 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 100.00 |
| 2 (400) | 0 | 372 | 27 | 1 | 0 | 0 | 0 | 0 | 93.00 | ||
| 3 (400) | 0 | 33 | 314 | 14 | 23 | 12 | 1 | 3 | 78.50 | ||
| 4 (400) | 0 | 5 | 27 | 190 | 57 | 68 | 11 | 42 | 47.50 | ||
| 5 (400) | 0 | 3 | 43 | 54 | 196 | 66 | 3 | 35 | 49.00 | ||
| 6 (400) | 0 | 0 | 17 | 92 | 82 | 170 | 3 | 36 | 42.50 | ||
| 7 (400) | 0 | 0 | 0 | 19 | 7 | 13 | 319 | 42 | 79.80 | ||
| 8 (400) | 0 | 1 | 5 | 31 | 29 | 26 | 14 | 294 | 73.50 | ||
| Pre. | 1 (200) | 199 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 99.50 | |
| 2 (200) | 2 | 161 | 36 | 0 | 0 | 1 | 0 | 0 | 80.50 | ||
| 3 (200) | 0 | 36 | 131 | 6 | 14 | 6 | 0 | 7 | 65.50 | ||
| 4 (200) | 0 | 4 | 31 | 66 | 41 | 47 | 1 | 10 | 33.00 | ||
| 5 (200) | 0 | 2 | 25 | 37 | 77 | 37 | 2 | 20 | 38.50 | ||
| 6 (199) | 0 | 2 | 13 | 62 | 53 | 43 | 4 | 22 | 21.60 | ||
| 7 (200) | 0 | 0 | 0 | 11 | 10 | 10 | 121 | 48 | 60.50 | ||
| 8 (199) | 0 | 0 | 0 | 21 | 22 | 16 | 20 | 120 | 60.30 | ||
| Maize 2 | Cal. | 1 (400) | 400 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 100.00 |
| 2 (400) | 0 | 365 | 33 | 0 | 0 | 2 | 0 | 0 | 91.30 | ||
| 3 (400) | 0 | 25 | 375 | 0 | 0 | 0 | 0 | 0 | 93.80 | ||
| 4 (400) | 0 | 0 | 0 | 246 | 54 | 29 | 10 | 61 | 61.50 | ||
| 5 (400) | 0 | 0 | 0 | 57 | 295 | 6 | 0 | 42 | 73.80 | ||
| 6 (400) | 0 | 5 | 0 | 38 | 1 | 230 | 113 | 13 | 57.50 | ||
| 7 (400) | 0 | 2 | 0 | 21 | 1 | 165 | 196 | 15 | 49.00 | ||
| 8 (400) | 0 | 0 | 0 | 117 | 63 | 27 | 7 | 186 | 46.50 | ||
| Pre. | 1 (200) | 196 | 0 | 3 | 0 | 0 | 0 | 0 | 1 | 98.00 | |
| 2 (200) | 1 | 164 | 30 | 0 | 0 | 2 | 3 | 0 | 82.00 | ||
| 3 (199) | 2 | 15 | 182 | 0 | 0 | 0 | 0 | 0 | 91.50 | ||
| 4 (200) | 0 | 0 | 0 | 86 | 35 | 11 | 11 | 57 | 43.00 | ||
| 5 (200) | 1 | 0 | 0 | 36 | 126 | 3 | 2 | 32 | 63.00 | ||
| 6 (200) | 0 | 2 | 0 | 16 | 2 | 93 | 84 | 3 | 46.50 | ||
| 7 (200) | 0 | 2 | 0 | 20 | 0 | 75 | 94 | 9 | 47.00 | ||
| 8 (200) | 0 | 0 | 0 | 78 | 46 | 12 | 6 | 58 | 29.00 | ||
| Sample Variety | Sample Number | Pre. | Accuracy (%) | |||
|---|---|---|---|---|---|---|
| Group 1 | Group 2 | Group 3 | ||||
| Maize 1 | Cal. | Group 1 (400) | 400 | 0 | 0 | 100.00 |
| Group 2 (800) | 0 | 746 | 54 | 93.25 | ||
| Group 3 (2000) | 0 | 101 | 1899 | 94.95 | ||
| Pre. | Group 1 (200) | 199 | 1 | 0 | 99.50 | |
| Group 2 (400) | 2 | 364 | 34 | 91.00 | ||
| Group 3 (998) | 0 | 77 | 921 | 92.28 | ||
| Maize 2 | Cal. | Group 1 (400) | 400 | 0 | 0 | 100.00 |
| Group 2 (800) | 0 | 798 | 2 | 99.75 | ||
| Group 3 (2000) | 0 | 7 | 1993 | 99.65 | ||
| Pre. | Group 1 (200) | 196 | 3 | 1 | 98.00 | |
| Group 2 (399) | 3 | 391 | 5 | 97.99 | ||
| Group 3 (1000) | 1 | 4 | 995 | 99.50 | ||
| Sample Variety | Accelerating Aging Time (hrs) | Germination Rate (%) | Shoot Length (cm/seedling) | Root Length (cm/seedling) |
|---|---|---|---|---|
| Maize 1 | 0 | 92.00a | 11.30a | 23.15a |
| 12 | 90.67a | 12.26b | 21.42b | |
| 24 | 86.00a | 9.77c | 17.31cd | |
| 36 | 75.33b | 6.35d | 18.20c | |
| 48 | 73.67b | 8.95c | 16.68d | |
| 72 | 76.33b | 6.17d | 13.76e | |
| 96 | 59.00c | 5.60d | 12.80e | |
| 120 | 57.00c | 5.99d | 12.69e | |
| Maize 2 | 0 | 96.33a | 13.06a | 24.68a |
| 12 | 97.67a | 10.64b | 24.11a | |
| 24 | 93.00a | 10.09bc | 19.25b | |
| 36 | 82.67b | 8.78cd | 17.08c | |
| 48 | 79.00bc | 6.98e | 18.46b | |
| 72 | 75.33c | 7.27de | 12.80d | |
| 96 | 62.00d | 5.93e | 14.63e | |
| 120 | 63.67d | 6.73e | 12.13e |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Feng, L.; Zhu, S.; Zhang, C.; Bao, Y.; Feng, X.; He, Y. Identification of Maize Kernel Vigor under Different Accelerated Aging Times Using Hyperspectral Imaging. Molecules 2018, 23, 3078. https://doi.org/10.3390/molecules23123078
Feng L, Zhu S, Zhang C, Bao Y, Feng X, He Y. Identification of Maize Kernel Vigor under Different Accelerated Aging Times Using Hyperspectral Imaging. Molecules. 2018; 23(12):3078. https://doi.org/10.3390/molecules23123078
Chicago/Turabian StyleFeng, Lei, Susu Zhu, Chu Zhang, Yidan Bao, Xuping Feng, and Yong He. 2018. "Identification of Maize Kernel Vigor under Different Accelerated Aging Times Using Hyperspectral Imaging" Molecules 23, no. 12: 3078. https://doi.org/10.3390/molecules23123078
APA StyleFeng, L., Zhu, S., Zhang, C., Bao, Y., Feng, X., & He, Y. (2018). Identification of Maize Kernel Vigor under Different Accelerated Aging Times Using Hyperspectral Imaging. Molecules, 23(12), 3078. https://doi.org/10.3390/molecules23123078









