Derivatization of Methylglyoxal for LC-ESI-MS Analysis—Stability and Relative Sensitivity of Different Derivatives
Abstract
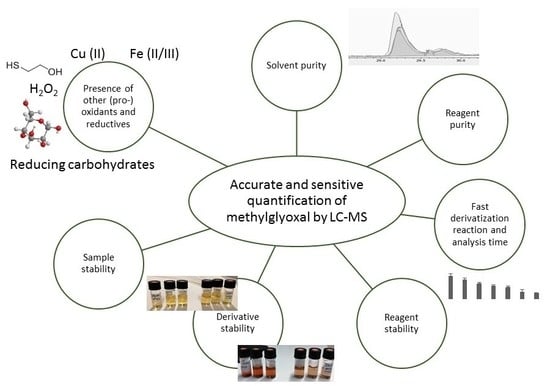
1. Introduction
2. Results and Discussion
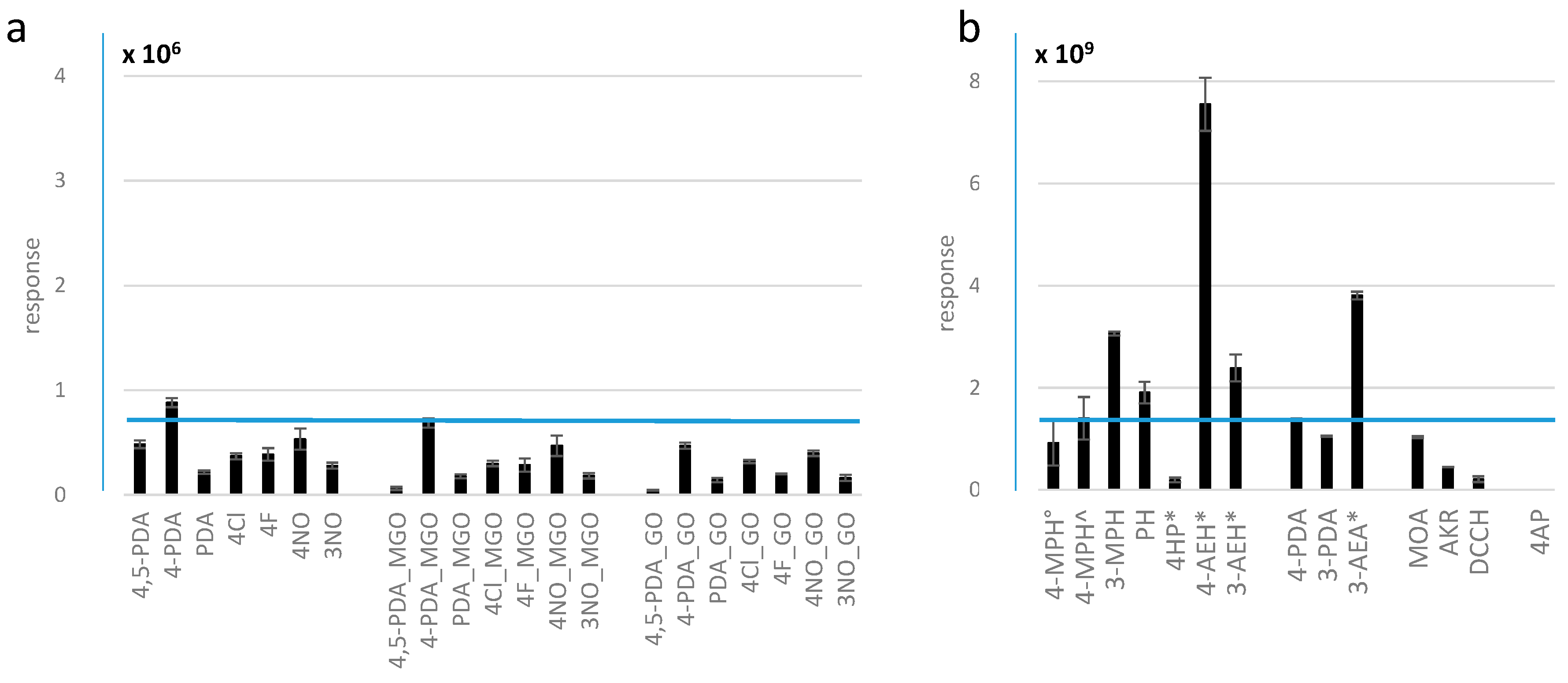
2.1. Relative Response of Methylglyoxal Derivatives in LC-MS Analysis
2.2. Extent of Degradation, Solubility, and Practical Considerations
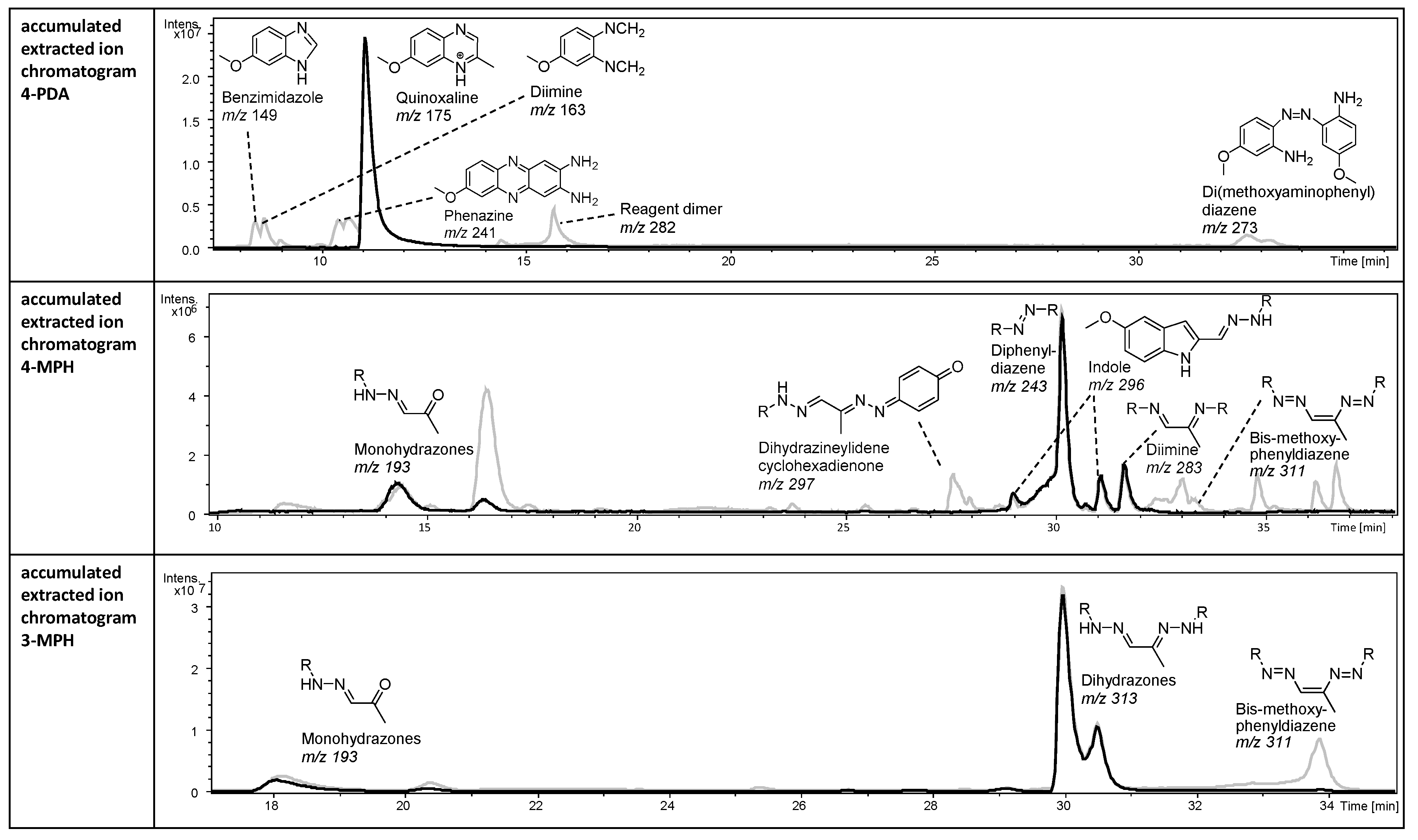
2.2.1. Multiple Reaction Products in the Chromatograms of 4-MPH and 4-PDA Reaction Mixtures
2.2.2. 3-MPH, Anilines, and Hydroxylamines Exhibit Higher Stability Compared to 4-MPH
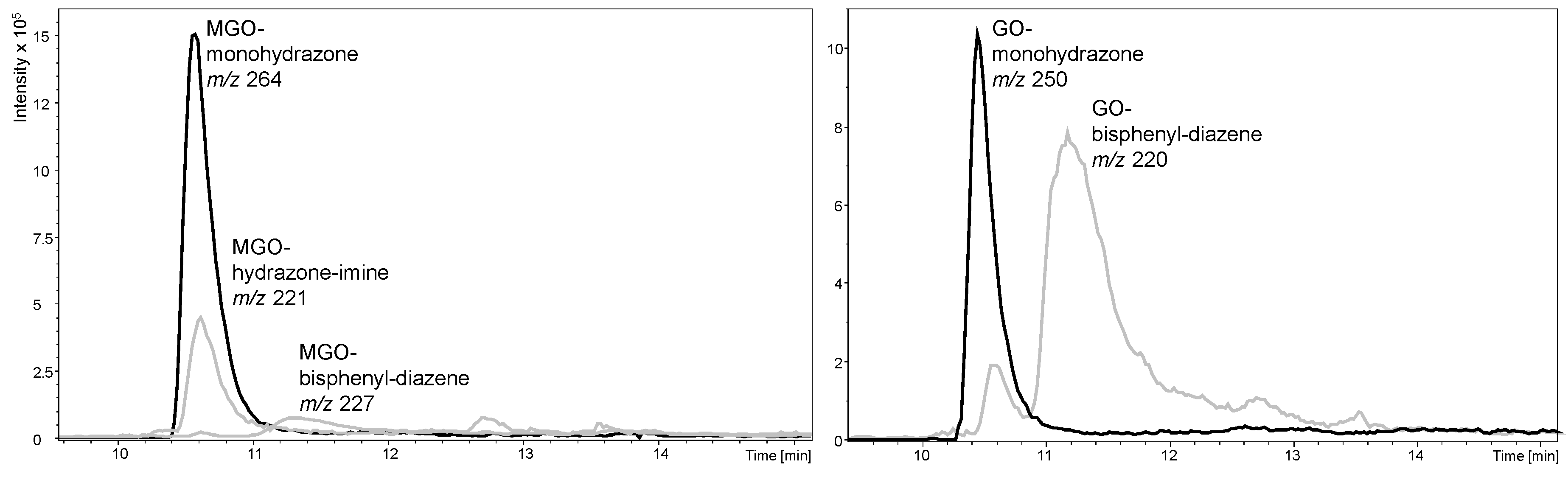
2.2.3. Reaction Products of Phenylhydrazines Featuring a Permanent Charge Provide Excellent Solubility but Insufficient Stability
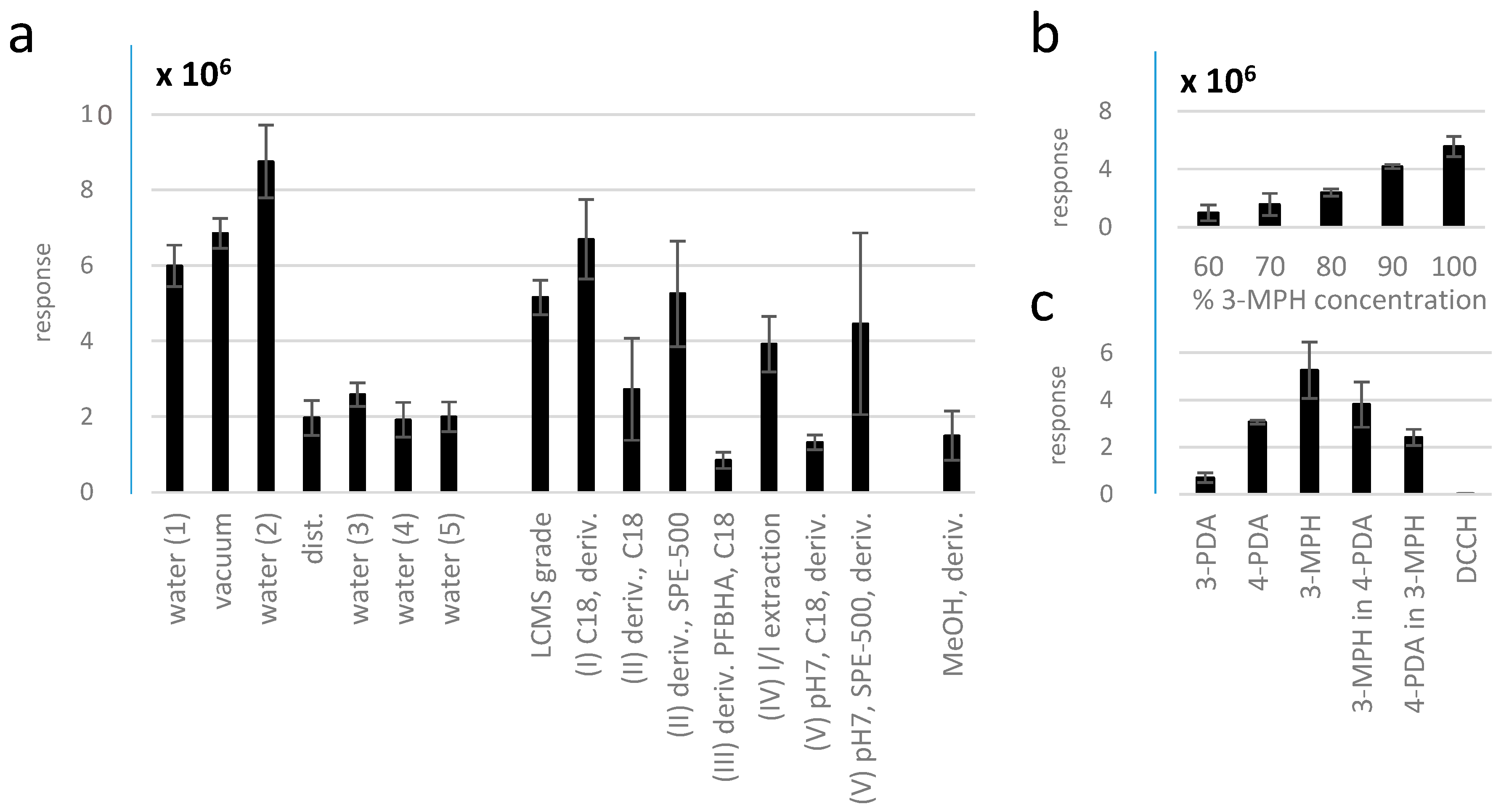
2.3. Application of 3-MPH as Derivatizing Agent to Explore the Contamination of Laboratory Water with Methylglyoxal
2.4. Analysis of Carbonyl Reaction Products of Linoleic and Linolenic Acids Oxidation by Cu(II) and Hydrogen Peroxide After Derivatization with 3-MPH
3. Materials and Methods
3.1 Materials and Chemicals
3.2. Response and Stability of Methylglyoxal Derivatives
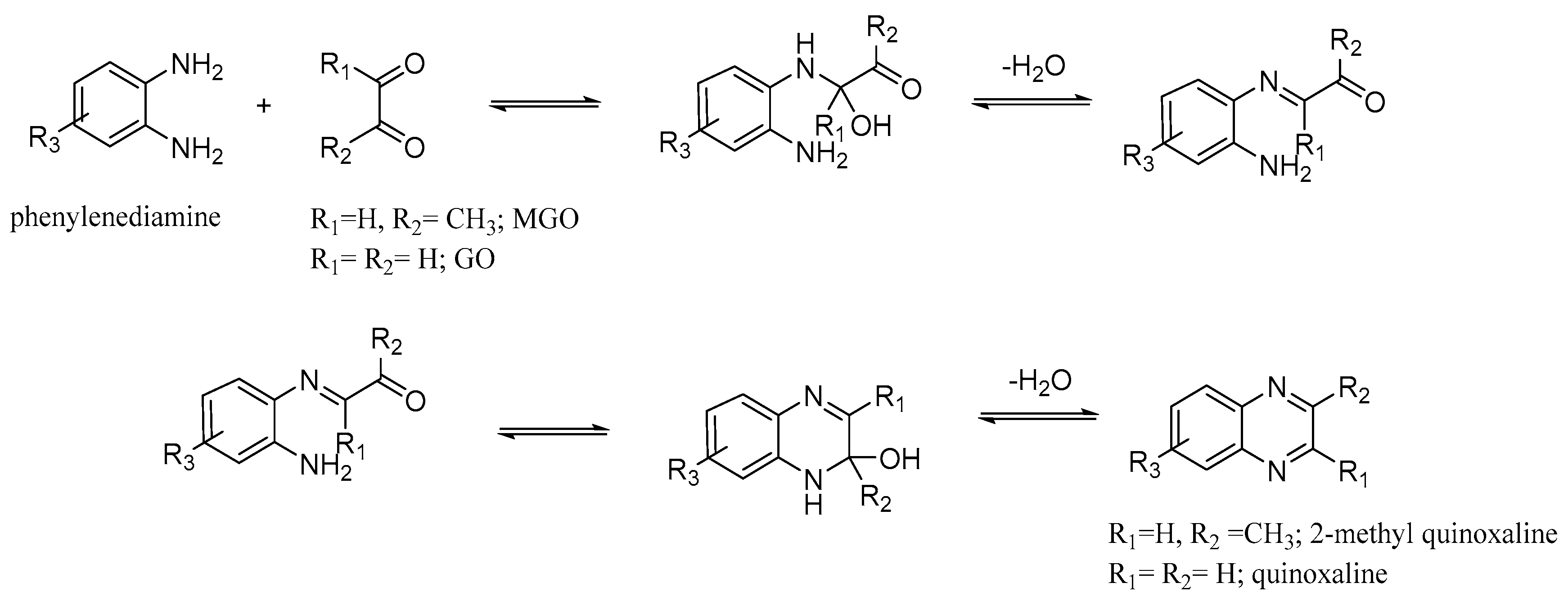
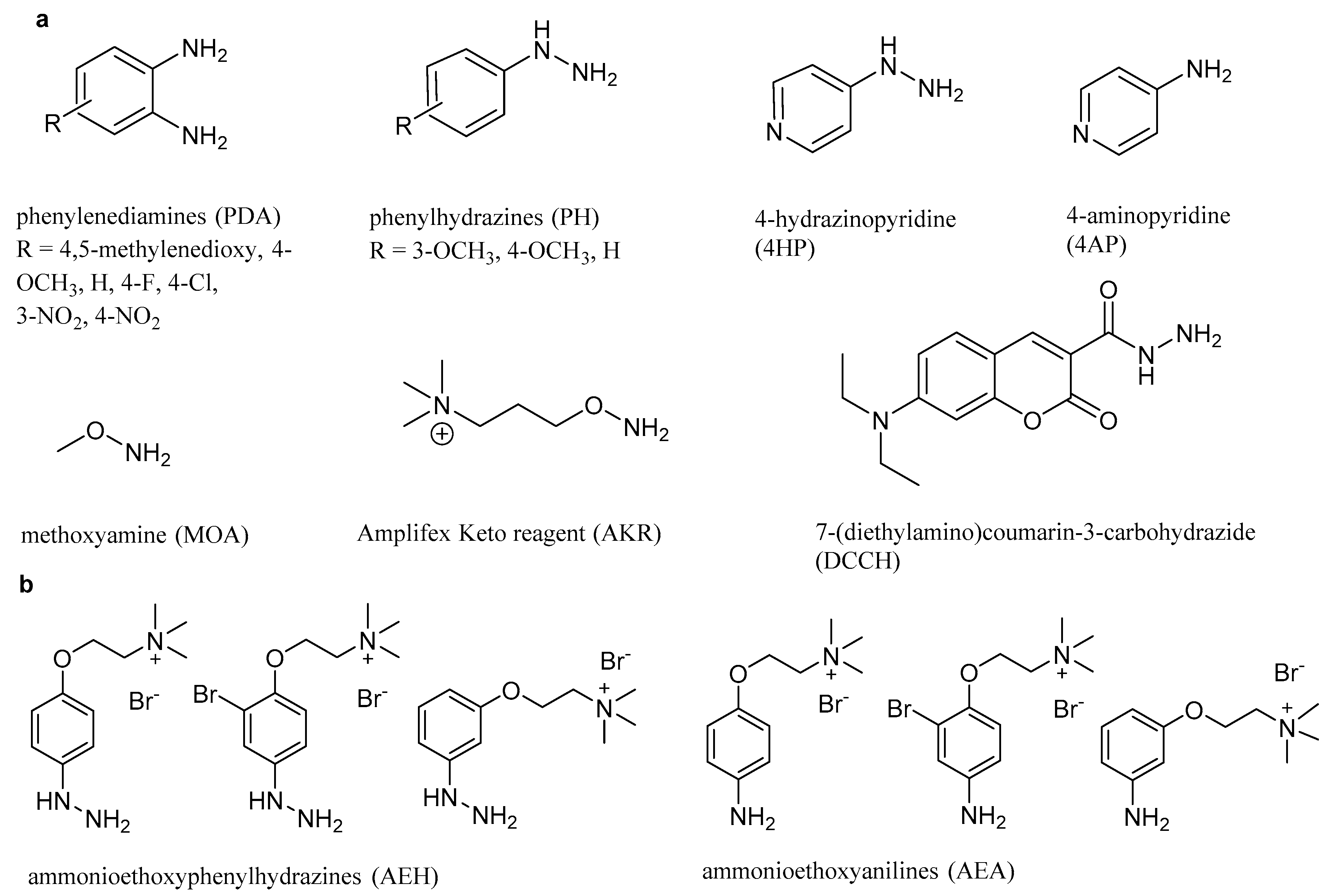
3.2.1. Reaction with Anilines
3.2.2. Comparison of Phenylenediamines (PDA)
3.2.3. Reaction with Phenylhydrazines, 4-Hydrazinopyridine, and Methoxyphenylenediamines
3.2.4. Reaction with the Amplifex™ Keto Reagent, Methoxyamine, and 7-(Diethylamino)coumarin-3-carbohydrazide
3.2.5. Relative Response for Different Types of Reagents
3.2.6. Optimized Derivatization Protocol with 3-MPH
3.3. Application of 3-MPH as Derivatization Agent to Explore the Contamination of Laboratory Water with Methylglyoxal
3.4. Analysis of Carbonyl Reaction Products of Linoleic and Linolenic Acids Oxidation by Hydrogen Peroxide After Derivatization with 3-MPH
3.5. Instrumental Parameters of LC-ESI-MS
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Dakin, H.D.; Dudley, H.W. An enzyme concerned with the formation of hydroxy acids from ketonic aldehydes. J. Biol. Chem. 1913, 14, 155–157. [Google Scholar]
- Dakin, H.D.; Dudley, H.W. The interconversion of alpha-amino acids, alpha-hydroxy acids and alpha-ketonic aldehydes. Part II. J. Biol. Chem. 1913, 15, 127–143. [Google Scholar]
- Neuberg, C. Über die Zerstörung von Milchsäurealdehyd und Methylglyoxal durch tierische Organe. Biochem. Z. 1913, 49, 502–506. [Google Scholar]
- Beisswenger, P.J. Methylglyoxal in diabetes: Link to treatment, glycemic control and biomarkers of complications. Biochem. Soc. Trans. 2014, 42, 450–456. [Google Scholar] [CrossRef] [PubMed]
- Brownlee, M. Biochemistry and molecular cell biology of diabetic complications. Nature 2001, 414, 813–820. [Google Scholar] [CrossRef] [PubMed]
- Szent-Györgyi, A. The living state and cancer. Proc. Natl. Acad. Sci. USA 1977, 74, 2844–2847. [Google Scholar] [CrossRef] [PubMed]
- Baynes, J.W.; Thorpe, S.R. Glycoxidation and lipoxidation in atherogenesis. Free Radic. Biol. Med. 2000, 28, 1708–1716. [Google Scholar] [CrossRef]
- Thornalley, P.J. Protein and nucleotide damage by glyoxal and methylglyoxal in physiological systems - role in ageing and disease. Drug Metabol. Drug Interact. 2008, 23, 125–150. [Google Scholar] [CrossRef] [PubMed]
- Hovatta, I.; Tennant, R.S.; Helton, R.; Marr, R.A.; Singer, O.; Redwine, J.M.; Ellison, J.A.; Schadt, E.E.; Verma, I.M.; Lockhart, D.J.; et al. Glyoxalase 1 and glutathione reductase 1 regulate anxiety in mice. Nature 2005, 438, 662–666. [Google Scholar] [CrossRef] [PubMed]
- Benton, C.S.; Miller, B.H.; Skwerer, S.; Suzuki, O.; Schultz, L.E.; Cameron, M.D.; Marron, J.S.; Pletcher, M.T.; Wiltshire, T. Evaluating genetic markers and neurobiochemical analytes for fluoxetine response using a panel of mouse inbred strains. Psychopharmacology 2012, 221, 297–315. [Google Scholar] [CrossRef] [PubMed]
- Allaman, I.; Bélanger, M.; Magistretti, P.J. Methylglyoxal, the dark side of glycolysis. Front. Neurosci. 2015, 9, 23. [Google Scholar] [CrossRef] [PubMed]
- Semchyshyn, H.M. Reactive carbonyl species in vivo: Generation and dual biological effects. Sci. World J. 2014, 2014. [Google Scholar] [CrossRef] [PubMed]
- Fu, T.-M.; Jacob, D.J.; Wittrock, F.; Burrows, J.P.; Vrekoussis, M.; Henze, D.K. Global budgets of atmospheric glyoxal and methylglyoxal, and implications for formation of secondary organic aerosols. J. Geophys. Res. 2008, 113. [Google Scholar] [CrossRef]
- Vogel, M.; Büldt, A.; Karst, U. Hydrazine reagents as derivatizing agents in environmental analysis—A critical review. Fresenius J. Anal. Chem. 2000, 366, 781–791. [Google Scholar] [CrossRef] [PubMed]
- Chalmers, R.A.; Watts, R.W.E. Derivatives for the identification and quantitative determination of some keto- and aldo-carboxylic acids by gas-liquid chromatography. Analyst 1972, 97, 951–957. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, K.; Tanaka, M.; Kawai, S. Gas chromatographic determination of low-molecular-weight carbonyl compounds in aqueous solution as their O-(2,3,4,5,6-pentafluorobenzyl) oximes. J. Chromatogr. A 1980, 187, 413–417. [Google Scholar] [CrossRef]
- McLellan, A.C.; Phillips, S.A.; Thornalley, P.J. The assay of methylglyoxal in biological systems by derivatization with 1,2-diamino-4,5-dimethoxybenzene. Anal. Biochem. 1992, 206, 17–23. [Google Scholar] [CrossRef]
- Rotondo, A.; Bruno, G.; Brancatelli, G.; Nicolò, F.; Armentano, D. A phenyl-salicyliden-imine as a suitable ligand to build functional materials. Inorganica Chim. Acta 2009, 362, 247–252. [Google Scholar] [CrossRef]
- Pal, R.; Kim, K.-H. Experimental choices for the determination of carbonyl compounds in air. J. Sep. Sci. 2007, 30, 2708–2718. [Google Scholar] [CrossRef] [PubMed]
- Brady, O.L.; Elsmie, G.V. The use of 2,4-dinitrophenylhydrazine as a reagent for aldehydes and ketones. Analyst 1926, 51, 77–78. [Google Scholar] [CrossRef]
- Fischer, E. Über einige Osazone und Hydrazone der Zuckergruppe. Ber. Dtsch. Chem. Ges. 1894, 27, 2486–2492. [Google Scholar] [CrossRef]
- Oppermann, H.; Ding, Y.; Sharma, J.; Berndt Paetz, M.; Meixensberger, J.; Gaunitz, F.; Birkemeyer, C. Metabolic response of glioblastoma cells associated with glucose withdrawal and pyruvate substitution as revealed by GC-MS. Nutr. Metab. 2016, 13. [Google Scholar] [CrossRef] [PubMed]
- Bilova, T.; Lukasheva, E.; Brauch, D.; Greifenhagen, U.; Paudel, G.; Tarakhovskaya, E.; Frolova, N.; Mittasch, J.; Balcke, G.U.; Tissier, A.; et al. A snapshot of the plant glycated proteome-structural, functional, and mechanistic aspects. J. Biol. Chem. 2016, 291, 7621–7636. [Google Scholar] [CrossRef] [PubMed]
- Eggink, M.; Wijtmans, M.; Ekkebus, R.; Lingeman, H.; de Esch, I.J.P.; Kool, J.; Niessen, W.M.A.; Irth, H. Development of a selective ESI-MS derivatization reagent: Synthesis and optimization for the analysis of aldehydes in biological mixtures. Anal. Chem. 2008, 80, 9042–9051. [Google Scholar] [CrossRef] [PubMed]
- Milkovska-Stamenova, S.; Schmidt, R.; Frolov, A.; Birkemeyer, C. GC-MS Method for the Quantitation of Carbohydrate Intermediates in Glycation Systems. J. Agric. Food Chem. 2015, 63, 5911–5919. [Google Scholar] [CrossRef] [PubMed]
- Ojeda, A.G.; Wrobel, K.; Escobosa, A.R.C.; Garay-Sevilla, M.E.; Wrobel, K. High-performance liquid chromatography determination of glyoxal. methylglyoxal, and diacetyl in urine using 4-methoxy-o-phenylenediamine as derivatizing reagent. Anal. Biochem. 2014, 449, 52–58. [Google Scholar] [CrossRef] [PubMed]
- Thornalley, P.J.; Yurek-George, A.; Argirov, O.K. Kinetics and mechanism of the reaction of aminoguanidine with the α-oxoaldehydes glyoxal, methylglyoxal, and 3-deoxyglucosone under physiological conditions. Biochem. Pharmacol. 2000, 60, 55–65. [Google Scholar] [CrossRef]
- Kiontke, A.; Oliveira-Birkmeier, A.; Opitz, A.; Birkemeyer, C. Electrospray ionization efficiency is dependent on different molecular descriptors with respect to solvent pH and instrumental configuration. PLoS ONE 2016, 11. [Google Scholar] [CrossRef] [PubMed]
- Hoyen, H.; Vogel, M.; Karst, U. Recent developments in the determination of formaldehyde in air samples using derivatizing agents. Air Qual. Control 2003, 63, 295–298. [Google Scholar]
- Zwiener, C.; Glauner, T.; Frimmel, F.H. Method optimization for the determination of carbonyl compounds in disinfected water by DNPH derivatization and LC-ESI-MS-MS. Anal. Bioanal. Chem. 2002, 372, 615–621. [Google Scholar] [CrossRef] [PubMed]
- Nemet, I.; Varga-Defterdarović, L.; Turk, Z. Preparation and quantification of methylglyoxal in human plasma using reverse-phase high-performance liquid chromatography. Clin. Biochem. 2004, 37, 875–881. [Google Scholar] [CrossRef] [PubMed]
- Milic, I.; Hoffmann, R.; Fedorova, M. Simultaneous detection of low and high molecular weight carbonylated compounds derived from lipid peroxidation by electrospray ionization-tandem mass spectrometry. Anal. Chem. 2013, 85, 156–162. [Google Scholar] [CrossRef] [PubMed]
- Star-Weinstock, M.; Williamson, B.L.; Dey, S.; Pillai, S.; Purkayastha, S. LC-ESI-MS/MS analysis of testosterone at sub-picogram levels using a novel derivatization reagent. Anal. Chem. 2012, 84, 9310–9317. [Google Scholar] [CrossRef] [PubMed]
- ChemAxon’s Solubility Predictor. Available online: https://docs.chemaxon.com/display/docs/Solubility+Predictor (accessed on 7 May 2018).
- Abburi, R.; Kalkhof, S.; Oehme, R.; Kiontke, A.; Birkemeyer, C. Artifacts in amine analysis from anodic oxidation of organic solvents upon electrospray ionization for mass spectrometry. Eur. J. Mass Spectrom. 2012, 18, 301–312. [Google Scholar] [CrossRef] [PubMed]
- Hardie, R.L.; Thomson, R.H. 488. The oxidation of phenylhydrazine. J. Chem. Soc. 1957, 0, 2512–2518. [Google Scholar] [CrossRef]
- Huang, P.-K.C.; Kosower, E.M. Diazenes. III. Properties of phenyldiazene. J. Am. Chem. Soc. 1968, 90, 2367–2376. [Google Scholar] [CrossRef]
- Henning, C.; Liehr, K.; Girndt, M.; Ulrich, C.; Glomb, M.A. Extending the spectrum of α-dicarbonyl compounds in vivo. J. Biol. Chem. 2014, 289, 28676–28688. [Google Scholar] [CrossRef] [PubMed]
- Enders, E.; Kolbah, D.; Korunčev, D.; Müller, E. Hydrazines, Azines; Azo-, Azoxy-Compounds I; Diazenes I. In Methoden der Organischen Chemie, 4th ed.; Thieme: Stuttgart, Germany, 1967; ISBN 9783131959249. [Google Scholar]
- Milic, I.; Fedorova, M. Derivatization and detection of small aliphatic and lipid-bound carbonylated lipid peroxidation products by ESI-MS. Methods Mol. Biol. 2015, 1208, 3–20. [Google Scholar] [CrossRef] [PubMed]
- Nemet, I.; Varga-Defterdarović, L.; Turk, Z. Methylglyoxal in food and living organisms. Mol. Nutr. Food Res. 2006, 50, 1105–1117. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Cáceres, M.I.; Palomino-Vasco, M.; Mora-Diez, N.; Acedo-Valenzuela, M.I. Novel HPLC—fluorescence methodology for the determination of methylglyoxal and glyoxal. Application to the analysis of monovarietal wines “Ribera del Guadiana”. Food Chem. 2015, 187, 159–165. [Google Scholar] [CrossRef] [PubMed]
- Hamberg, M. Autoxidation of linoleic acid: Isolation and structure of four dihydroxyoctadecadienoic acids. Biochim. Biophys. Acta Lipids Lipid Metab. 1983, 752, 353–356. [Google Scholar] [CrossRef]
- Banni, S.; Contini, M.S.; Angioni, E.; Deiana, M.; Dessì, M.A.; Melis, M.P.; Carta, G.; Corongiu, F.P. A novel approach to study linoleic acid autoxidation: Importance of simultaneous detection of the substrate and its derivative oxidation products. Free Radic. Res. 1996, 25, 43–53. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Cui, P. Reactive carbonyl species derived from Omega-3 and Omega-6 fatty acids. J. Agric. Food Chem. 2015, 63, 6293–6296. [Google Scholar] [CrossRef] [PubMed]
- Shibamoto, T. Analytical methods for trace levels of reactive carbonyl compounds formed in lipid peroxidation systems. J. Pharmaceut. Biomed. Anal. 2006, 41, 12–25. [Google Scholar] [CrossRef] [PubMed]
- Qi, B.-L.; Liu, P.; Wang, Q.-Y.; Cai, W.-J.; Yuan, B.-F.; Feng, Y.-Q. Derivatization for liquid chromatography-mass spectrometry. Trends Anal. Chem. TrAC 2014, 59, 121–132. [Google Scholar] [CrossRef]
- El-Maghrabey, M.H.; Kishikawa, N.; Ohyama, K.; Imazato, T.; Ueki, Y.; Kuroda, N. Determination of human serum semicarbazide-sensitive amine oxidase activity via flow injection analysis with fluorescence detection after online derivatization of the enzymatically produced benzaldehyde with 1,2-diaminoanthraquinone. Anal. Chim. Acta 2015, 881, 139–147. [Google Scholar] [CrossRef] [PubMed]
- Grosjean, D.; Fung, K. Collection efficiencies of cartridges and microimpingers for sampling of aldehydes in air as 2,4-dinitrophenylhydrazones. Anal. Chem. 2002, 54, 1221–1224. [Google Scholar] [CrossRef]
Sample Availability: Samples of methylglyoxal, 4-PDA and 3-MPH and most of the synthesized compounds and intermediates are available from the authors at the date of publication. |
| LOD pmol/Injected | Literature Reference Value LOD pmol/Injected | |
|---|---|---|
| 4-PDA | 0.01 | 0.1 with HPLC-FD [26] |
| 3-MPH | 0.05 | n.a. |
| 3-AEH | 0.7 | n.a. |
| 3-AEA | 1 | n.a. a |
| DCCH | 1.5 | n.a. b |
| AKR | 2 | n.a. c |
| 4-MPH | >50 | n.a. |
| Tentative Identification | Ratio C18:2/C18:3 | p-Value t-Test |
|---|---|---|
| oxobutanal * | 11 | 0.005 |
| oxopentanal * | 10 | 0.056 |
| oxononanoic acid * | 1.9 | 0.035 |
| methylglyoxal | 1.7 | 0.013 |
| glyoxal | 0.7 | 0.031 |
| acrolein * | 0.7 | 0.056 |
| pentenal * | 0.6 | 0.071 |
| malondialdehyde * | 0.4 | 0.015 |
| hexenedial * | 0.1 | 0.005 |
| Experiment | Gradient [min]/[%B] | Flow mL/min |
|---|---|---|
| 3- and 4-MPH, PDAs and MOA | 0/80, 5/80, 20/70, 30/30, 35/0, 42/0, 47/80, 54/80 | 0.6 |
| DCCH, 4-AEH | 0/90, 5/90, 15/0, 30/0, 40/90, 45/90 | 0.5 |
| Amplifex™ (AKR) | 0/0, 10/0, 30/50, 40/98, 45/98, 46/0, 56/0 | 0.3 |
| 3-AEH, 3-AEA | 0/98, 5/98, 15/0, 20/0, 20.1/98, 25/98 | 0.3 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fritzsche, S.; Billig, S.; Rynek, R.; Abburi, R.; Tarakhovskaya, E.; Leuner, O.; Frolov, A.; Birkemeyer, C. Derivatization of Methylglyoxal for LC-ESI-MS Analysis—Stability and Relative Sensitivity of Different Derivatives. Molecules 2018, 23, 2994. https://doi.org/10.3390/molecules23112994
Fritzsche S, Billig S, Rynek R, Abburi R, Tarakhovskaya E, Leuner O, Frolov A, Birkemeyer C. Derivatization of Methylglyoxal for LC-ESI-MS Analysis—Stability and Relative Sensitivity of Different Derivatives. Molecules. 2018; 23(11):2994. https://doi.org/10.3390/molecules23112994
Chicago/Turabian StyleFritzsche, Stefan, Susan Billig, Robby Rynek, Ramarao Abburi, Elena Tarakhovskaya, Olga Leuner, Andrej Frolov, and Claudia Birkemeyer. 2018. "Derivatization of Methylglyoxal for LC-ESI-MS Analysis—Stability and Relative Sensitivity of Different Derivatives" Molecules 23, no. 11: 2994. https://doi.org/10.3390/molecules23112994
APA StyleFritzsche, S., Billig, S., Rynek, R., Abburi, R., Tarakhovskaya, E., Leuner, O., Frolov, A., & Birkemeyer, C. (2018). Derivatization of Methylglyoxal for LC-ESI-MS Analysis—Stability and Relative Sensitivity of Different Derivatives. Molecules, 23(11), 2994. https://doi.org/10.3390/molecules23112994