Effects of Red Ginseng Extract on the Pharmacokinetics and Elimination of Methotrexate via Mrp2 Regulation
Abstract
:1. Introduction
2. Results
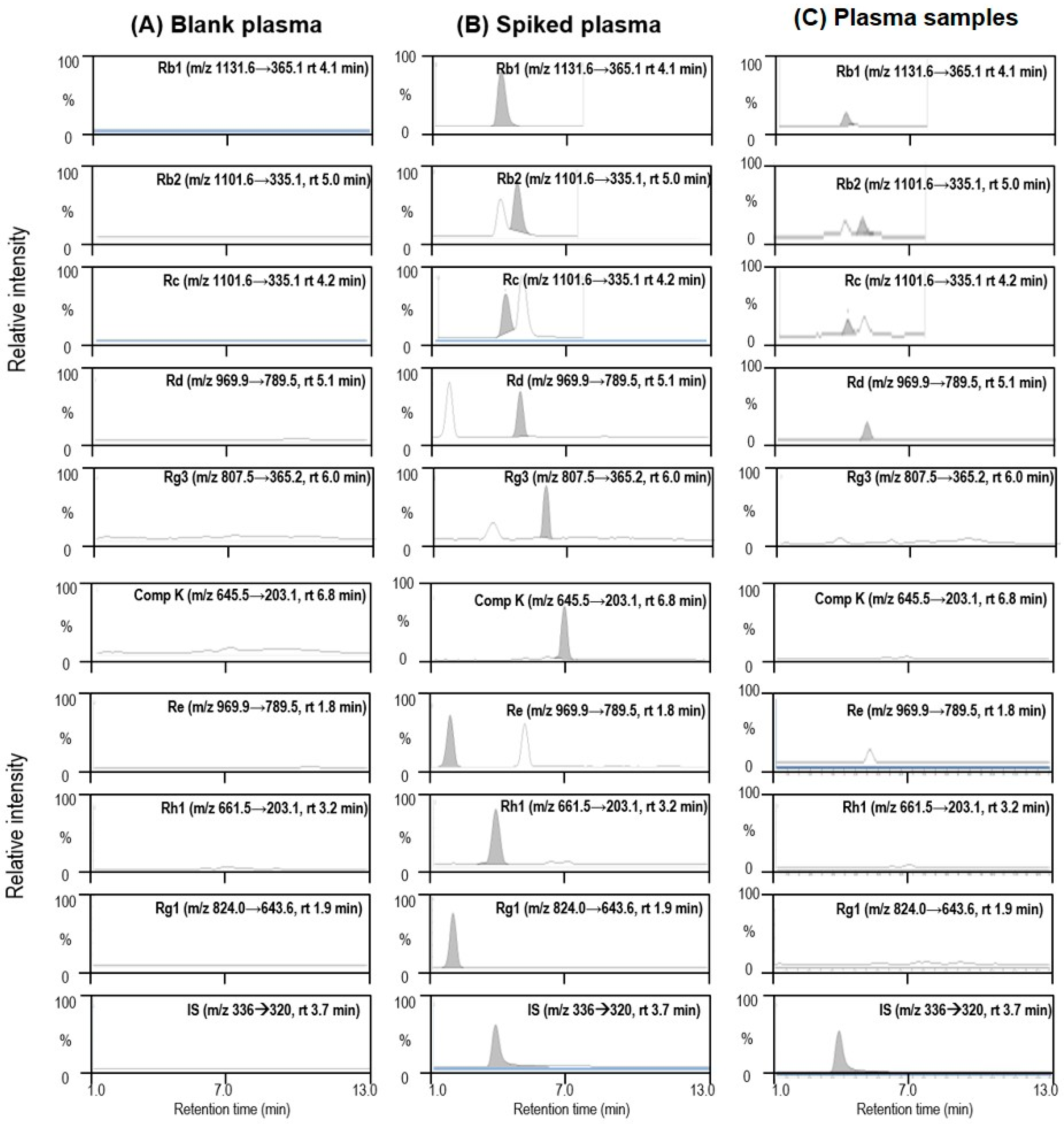
2.1. Concentration of Ginsenosides in Red Ginseng Extracts and Rat Plasma
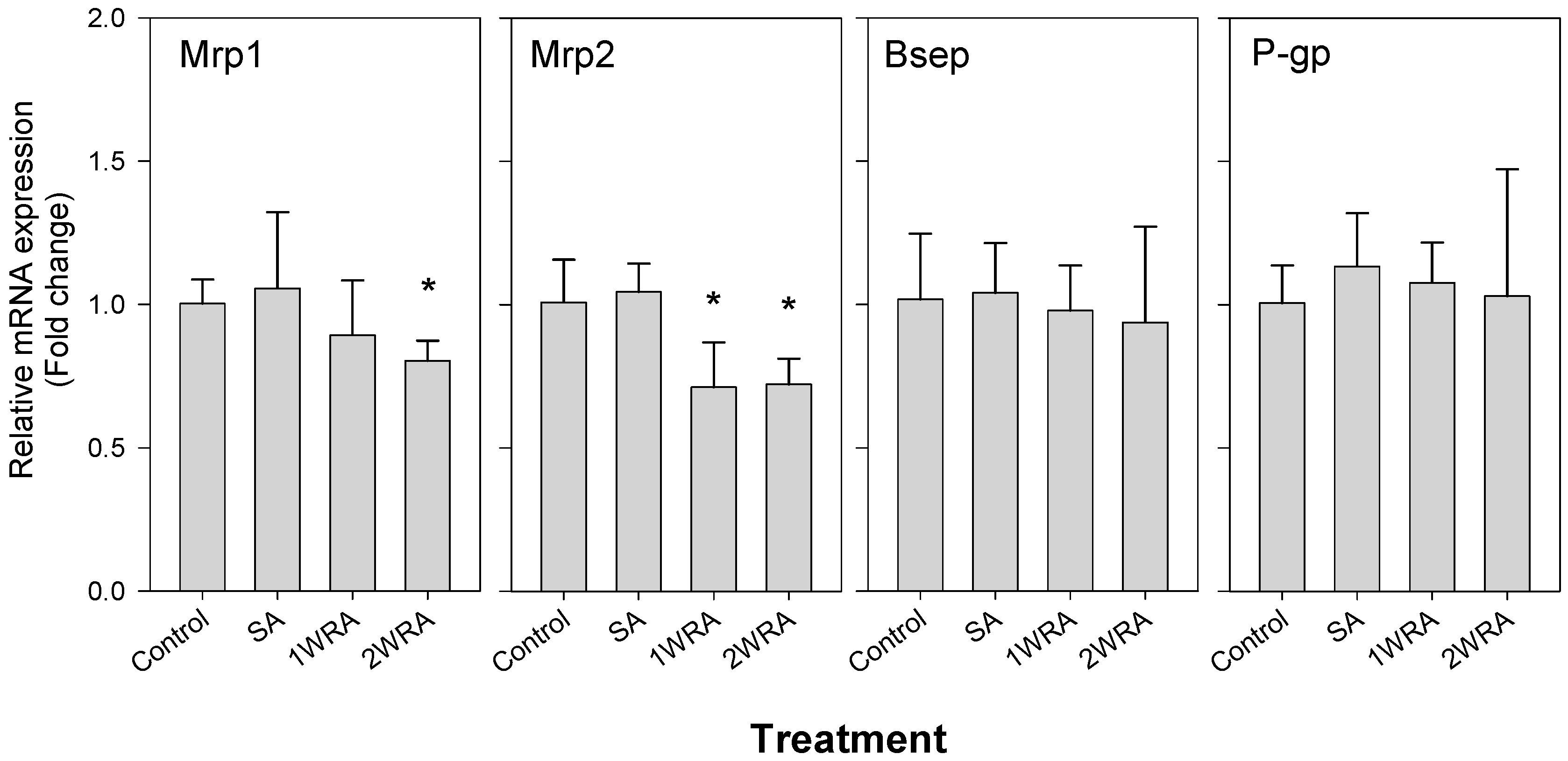
2.2. Analysis of the mRNA Expression of Efflux Transporters
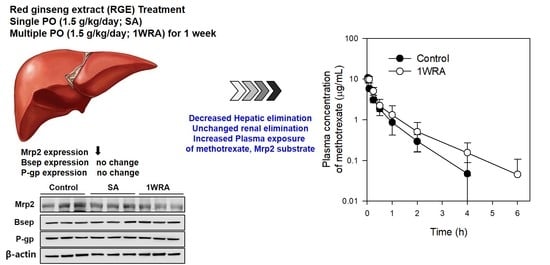
2.3. Western Blot Analysis of Mrp2
2.4. Effect of RGE Treatment on Biochemical Parameters for Liver and Renal Function
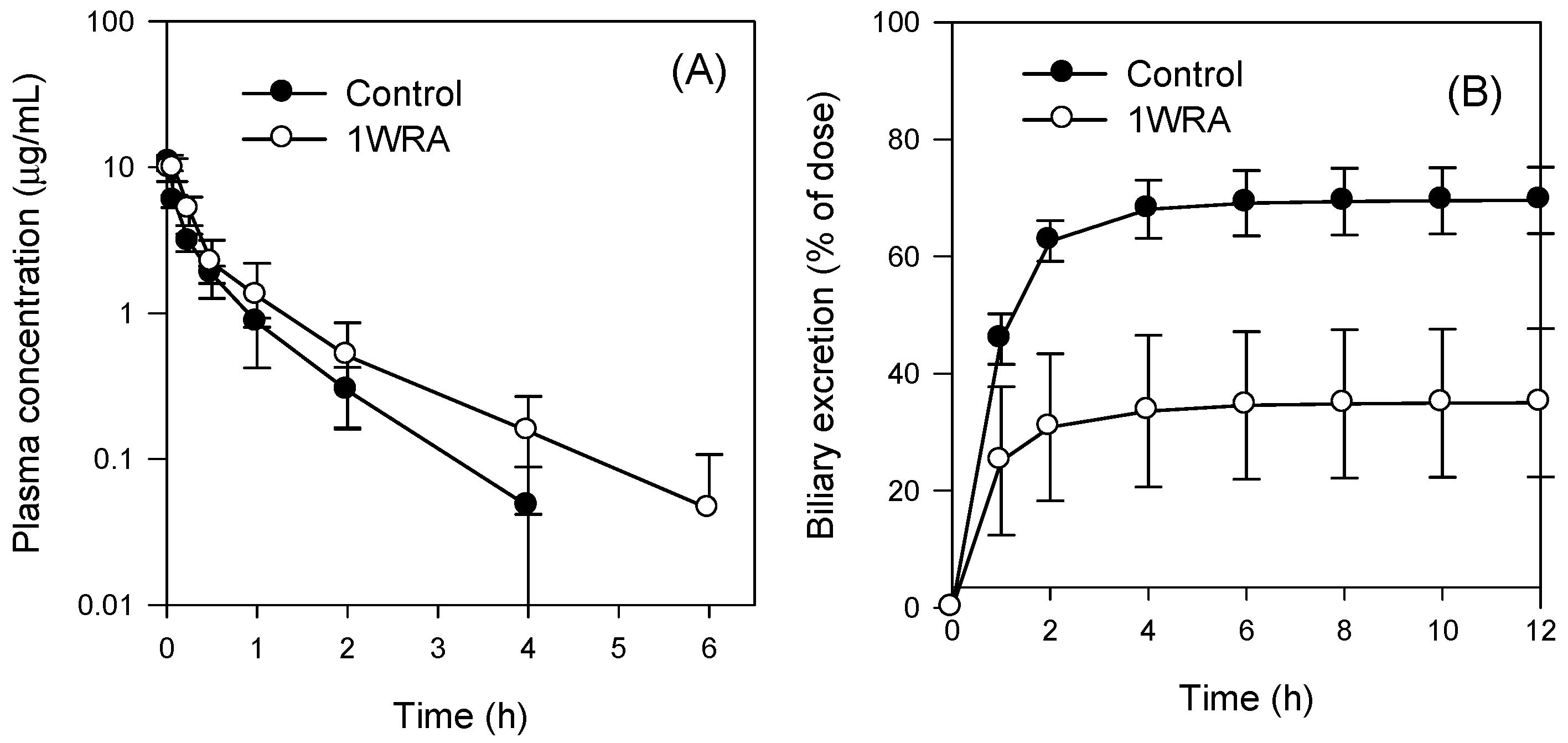
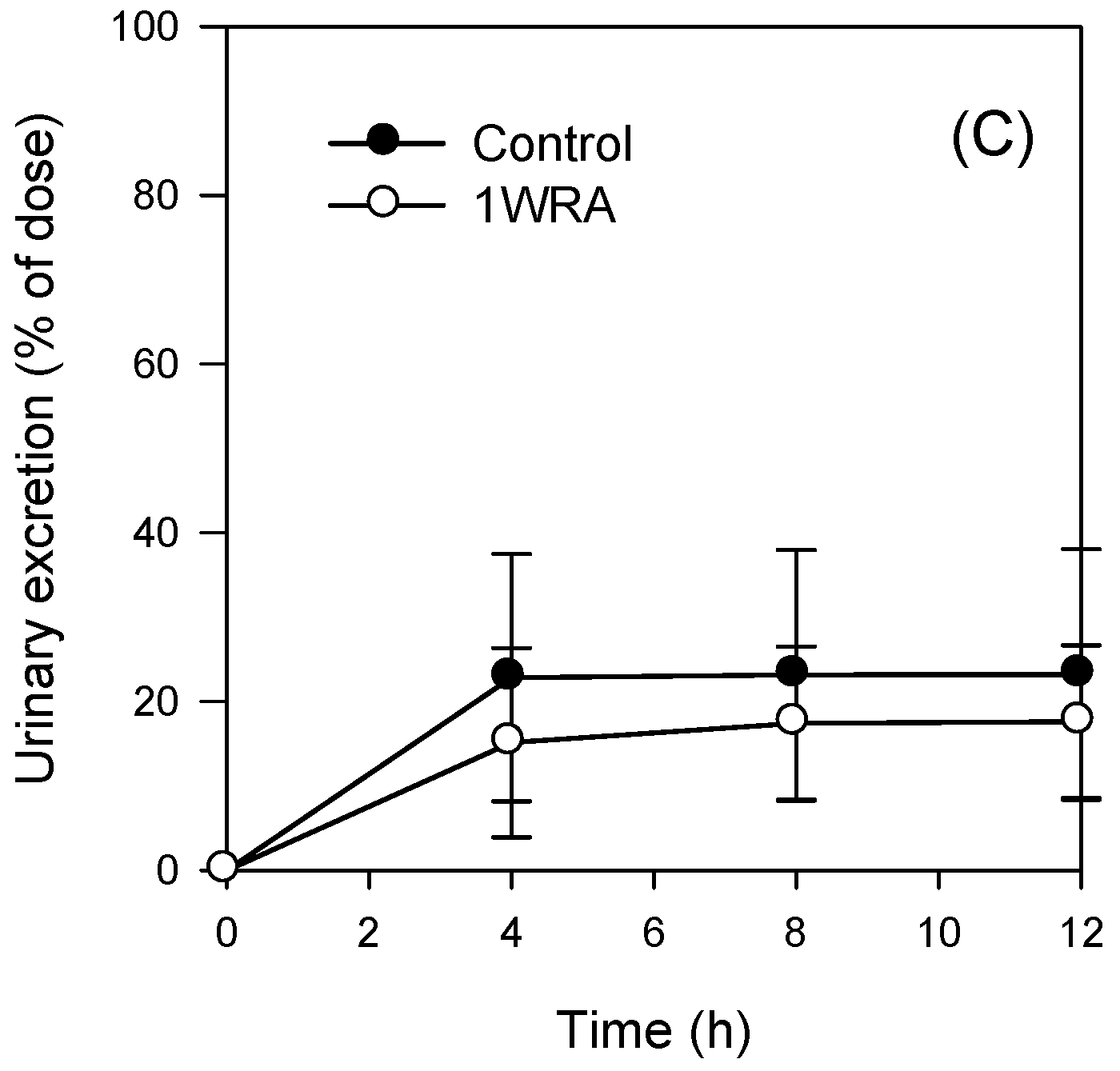
2.5. Pharmacokinetics of Methotrexate
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Animals
4.3. RGE Administration
4.4. Bioanalysis of 14 Ginsenosides
4.5. Real-Time Reverse-Transcription Polymerase Chain Reaction (RT-PCR) Analysis
4.6. Western Blot Analysis
4.7. Pharmacokinetics of Methotrexate
4.8. Data Analysis
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Ru, W.; Wang, D.; Xu, Y.; He, X.; Sun, Y.E.; Qian, L.; Zhou, X.; Qin, Y. Chemical constituents and bioactivities of panax ginseng (c. A. Mey.). Drug Discov. Ther. 2015, 9, 23–32. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.H.; Kim, J.H. A review on the medicinal potentials of ginseng and ginsenosides on cardiovascular diseases. J. Ginseng Res. 2014, 38, 161–166. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.M.; Bae, B.S.; Park, H.W.; Ahn, N.G.; Cho, B.G.; Cho, Y.L.; Kwak, Y.S. Characterization of korean red ginseng (panax ginseng meyer): History, preparation method, and chemical composition. J. Ginseng Res. 2015, 39, 384–391. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Yi, Y.S.; Kim, M.Y.; Cho, J.Y. Role of ginsenosides, the main active components of panax ginseng, in inflammatory responses and diseases. J. Ginseng Res. 2017, 41, 435–443. [Google Scholar] [CrossRef] [PubMed]
- Yun, T.K.; Choi, S.Y.; Yun, H.Y. Epidemiological study on cancer prevention by ginseng: Are all kinds of cancers preventable by ginseng? J. Korean Med. Sci. 2001, 16, 192S–197S. [Google Scholar] [CrossRef] [PubMed]
- Gui, Q.F.; Xu, Z.R.; Xu, K.Y.; Yang, Y.M. The efficacy of ginseng-related therapies in type 2 diabetes mellitus: An updated systematic review and meta-analysis. Medicine 2016, 95, e2584. [Google Scholar] [CrossRef] [PubMed]
- Park, T.Y.; Hong, M.; Sung, H.; Kim, S.; Suk, K.T. Effect of korean red ginseng in chronic liver disease. J. Ginseng Res. 2017, 41, 450–455. [Google Scholar] [CrossRef] [PubMed]
- Ji, H.Y.; Liu, K.H.; Kong, T.Y.; Jeong, H.U.; Choi, S.Z.; Son, M.; Cho, Y.Y.; Lee, H.S. Evaluation of da-9801, a new herbal drug for diabetic neuropathy, on metabolism-mediated interaction. Arch. Pharm. Res. 2013, 36, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Gurley, B.J.; Gardner, S.F.; Hubbard, M.A.; Williams, D.K.; Gentry, W.B.; Cui, Y.; Ang, C.Y. Clinical assessment of effects of botanical supplementation on cytochrome p450 phenotypes in the elderly: St john′s wort, garlic oil, panax ginseng and ginkgo biloba. Drugs Aging 2005, 22, 525–539. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.S.; Kim, Y.; Jeon, J.Y.; Kim, M.G. Effect of red ginseng on cytochrome p450 and p-glycoprotein activities in healthy volunteers. J. Ginseng Res. 2016, 40, 375–381. [Google Scholar] [CrossRef] [PubMed]
- Fasinu, P.S.; Bouic, P.J.; Rosenkranz, B. An overview of the evidence and mechanisms of herb-drug interactions. Front. Pharmacol. 2012, 3, 69. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.W.; Serag, E.S.; Sneed, K.B.; Liang, J.; Chew, H.; Pan, S.Y.; Zhou, S.F. Clinical herbal interactions with conventional drugs: From molecules to maladies. Curr. Med. Chem. 2011, 18, 4836–4850. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.W.; Sneed, K.B.; Pan, S.Y.; Cao, C.; Kanwar, J.R.; Chew, H.; Zhou, S.F. Herb-drug interactions and mechanistic and clinical considerations. Curr. Drug Metab. 2012, 13, 640–651. [Google Scholar] [CrossRef] [PubMed]
- Leslie, E.M.; Deeley, R.G.; Cole, S.P. Multidrug resistance proteins: Role of p-glycoprotein, mrp1, mrp2, and bcrp (abcg2) in tissue defense. Toxicol. Appl. Pharmacol. 2005, 204, 216–237. [Google Scholar] [CrossRef] [PubMed]
- Rappa, G.; Finch, R.A.; Sartorelli, A.C.; Lorico, A. New insights into the biology and pharmacology of the multidrug resistance protein (mrp) from gene knockout models. Biochem. Pharmacol. 1999, 58, 557–562. [Google Scholar] [PubMed]
- Lorico, A.; Rappa, G.; Finch, R.A.; Yang, D.; Flavell, R.A.; Sartorelli, A.C. Disruption of the murine mrp (multidrug resistance protein) gene leads to increased sensitivity to etoposide (vp-16) and increased levels of glutathione. Cancer Res. 1997, 57, 5238–5242. [Google Scholar] [PubMed]
- Dietrich, C.G.; Geier, A.; Oude Elferink, R.P. Abc of oral bioavailability: Transporters as gatekeepers in the gut. Gut 2003, 52, 1788–1795. [Google Scholar] [CrossRef] [PubMed]
- Oude Elferink, R.P.; Meijer, D.K.; Kuipers, F.; Jansen, P.L.; Groen, A.K.; Groothuis, G.M. Hepatobiliary secretion of organic compounds; molecular mechanisms of membrane transport. Biochim. Biophys. Acta 1995, 1241, 215–268. [Google Scholar] [CrossRef]
- Evers, R.; de Haas, M.; Sparidans, R.; Beijnen, J.; Wielinga, P.R.; Lankelma, J.; Borst, P. Vinblastine and sulfinpyrazone export by the multidrug resistance protein mrp2 is associated with glutathione export. Br. J. Cancer 2000, 83, 375–383. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, T.; Nishio, K.; Tanabe, S. The mrp family and anticancer drug metabolism. Curr. Drug Metab. 2001, 2, 367–377. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.J.; Kang, Y.J.; Kwon, M.; Choi, Y.A.; Choi, M.K.; Chi, H.Y.; Yoo, H.H.; Shim, C.K.; Song, I.S. Ursodeoxycholate restores biliary excretion of methotrexate in rats with ethinyl estradiol induced-cholestasis by restoring canalicular mrp2 expression. Int. J. Mol. Sci. 2018, 19, 1120. [Google Scholar] [CrossRef] [PubMed]
- Park, S.H.; Jang, J.H.; Chen, C.Y.; Na, H.K.; Surh, Y.J. A formulated red ginseng extract rescues pc12 cells from pcb-induced oxidative cell death through nrf2-mediated upregulation of heme oxygenase-1 and glutamate cysteine ligase. Toxicology 2010, 278, 131–139. [Google Scholar] [CrossRef] [PubMed]
- Seong, S.J.; Kang, W.Y.; Heo, J.K.; Jo, J.; Choi, W.G.; Liu, K.H.; Lee, S.; Choi, M.K.; Han, Y.H.; Lee, H.S.; et al. A comprehensive in vivo and in vitro assessment of the drug interaction potential of red ginseng. Clin. Ther. 2018, 40, 1322–1337. [Google Scholar] [CrossRef] [PubMed]
- Endale, M.; Im, E.J.; Lee, J.Y.; Kim, S.D.; Yayeh, T.; Song, Y.B.; Kwak, Y.S.; Kim, C.; Kim, S.H.; Roh, S.S.; et al. Korean red ginseng saponin fraction rich in ginsenoside-rb1, rc and rb2 attenuates the severity of mouse collagen-induced arthritis. Mediat. Inflamm. 2014, 2014, 748964. [Google Scholar] [CrossRef] [PubMed]
- Cho, S.K.; Kim, D.; Yoo, D.; Jang, E.J.; Jun, J.B.; Sung, Y.K. Korean red ginseng exhibits no significant adverse effect on disease activity in patients with rheumatoid arthritis: A randomized, double-blind, crossover study. J. Ginseng Res. 2018, 42, 144–148. [Google Scholar] [CrossRef] [PubMed]
- Yu, T.; Rhee, M.H.; Lee, J.; Kim, S.H.; Yang, Y.; Kim, H.G.; Kim, Y.; Kim, C.; Kwak, Y.S.; Kim, J.H.; et al. Ginsenoside rc from korean red ginseng (panax ginseng c.A. Meyer) attenuates inflammatory symptoms of gastritis, hepatitis and arthritis. Am. J. Chin. Med. 2016, 44, 595–615. [Google Scholar] [CrossRef] [PubMed]
- Jhun, J.; Lee, J.; Byun, J.K.; Kim, E.K.; Woo, J.W.; Lee, J.H.; Kwok, S.K.; Ju, J.H.; Park, K.S.; Kim, H.Y.; et al. Red ginseng extract ameliorates autoimmune arthritis via regulation of stat3 pathway, th17/treg balance, and osteoclastogenesis in mice and human. Mediat. Inflamm. 2014, 2014, 351856. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.K. Pharmacokinetics of ginsenoside rb1 and its metabolite compound k after oral administration of korean red ginseng extract. J. Ginseng Res. 2013, 37, 451–456. [Google Scholar] [CrossRef] [PubMed]
- Xu, Q.F.; Fang, X.L.; Chen, D.F. Pharmacokinetics and bioavailability of ginsenoside rb1 and rg1 from panax notoginseng in rats. J. Ethnopharmacol. 2003, 84, 187–192. [Google Scholar] [CrossRef]
- Leung, K.W.; Wong, A.S. Pharmacology of ginsenosides: A literature review. Chin. Med. 2010, 5, 20. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Masuda, M.; I’Izuka, Y.; Yamazaki, M.; Nishigaki, R.; Kato, Y.; Ni’inuma, K.; Suzuki, H.; Sugiyama, Y. Methotrexate is excreted into the bile by canalicular multispecific organic anion transporter in rats. Cancer Res. 1997, 57, 3506–3510. [Google Scholar] [PubMed]
- Liu, H.; Yang, J.; Du, F.; Gao, X.; Ma, X.; Huang, Y.; Xu, F.; Niu, W.; Wang, F.; Mao, Y.; et al. Absorption and disposition of ginsenosides after oral administration of panax notoginseng extract to rats. Drug Metab. Dispos. 2009, 37, 2290–2298. [Google Scholar] [CrossRef] [PubMed]
- Zheng, M.M.; Xu, F.X.; Li, Y.J.; Xi, X.Z.; Cui, X.W.; Han, C.C.; Zhang, X.L. Study on transformation of ginsenosides in different methods. Biomed. Res. Int. 2017, 2017, 8601027. [Google Scholar] [CrossRef] [PubMed]
- Miyamoto, E.; Odashima, S.; Kitagawa, I.; Tsuji, A. Stability kinetics of ginsenosides in aqueous solution. J. Pharm. Sci. 1984, 73, 409–410. [Google Scholar] [CrossRef] [PubMed]
- Ott, M.; Fricker, G.; Bauer, B. Pregnane x receptor (pxr) regulates p-glycoprotein at the blood-brain barrier: Functional similarities between pig and human pxr. J. Pharmacol. Exp. Ther. 2009, 329, 141–149. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.R.; Chung, T.Y.; Shin, H.; Son, S.H.; Park, K.K.; Choi, J.H.; Chung, W.Y. Red ginseng saponin extract attenuates murine collagen-induced arthritis by reducing pro-inflammatory responses and matrix metalloproteinase-3 expression. Biol. Pharm. Bull. 2010, 33, 604–610. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Bionaz, M.; Sloboda, D.M.; Ehrlich, L.; Li, S.; Newnham, J.P.; Dudenhausen, J.W.; Henrich, W.; Plagemann, A.; Challis, J.R.; et al. The dilution effect and the importance of selecting the right internal control genes for rt-qpcr: A paradigmatic approach in fetal sheep. BMC Res. Notes 2015, 8, 58. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Red ginseng extract are available from the authors. |

| Ginsenoside | (μg Ginsenoside/g RGE) | |
|---|---|---|
| 20(s)-protopanaxadiol | Rb1 | 1925 ± 38.3 |
| Rb2 | 929.6 ± 32.0 | |
| Rc | 1015 ± 32.4 | |
| Rd | 541.6 ± 47.0 | |
| Rh2 | ND | |
| Rg3 | 1291 ± 47.5 | |
| F2 | ND | |
| Compound K | ND | |
| Protopanaxadiol | ND | |
| 20(s)-protopanaxatriol | Re | 532.1 ± 36.3 |
| Rh1 | 654.2 ± 31.2 | |
| Rg1 | 160.0 ± 0.4 | |
| F1 | ND | |
| Protopanaxatriol | ND | |
| Ginsenoside | Plasma Concentration (ng/mL) | |||
|---|---|---|---|---|
| SA (1.5 g/kg/day) | 1WRA (1.5 g/kg/day for 1 week) | 2WRA (1.5 g/kg/day for 2 weeks) | ||
| 20(s)-protopanaxadiol | Rb1 | 8.9 ± 2.3 | 33.6 ± 10.1 * | 32.1 ± 12.4 * |
| Rb2 | 7.2 ± 1.6 | 25.1 ± 6.4 * | 22.3 ± 7.8 * | |
| Rc | 6.1 ± 1.6 | 25.5 ± 7.9 * | 25.1 ± 8.5 * | |
| Rd | 1.8 ± 0.6 | 9.8 ± 2.5 * | 11.5 ± 4.5 * | |
| Rh2 | ND | ND | ND | |
| Rg3 | ND | ND | ND | |
| F2 | ND | ND | ND | |
| Compound K | ND | ND | ND | |
| Protopanaxadiol | ND | ND | ND | |
| 20(s)-protopanaxatriol | Re | ND | ND | ND |
| Rh1 | ND | ND | ND | |
| Rg1 | ND | ND | ND | |
| F1 | ND | ND | ND | |
| Protopanaxatriol | ND | ND | ND | |
| Biochemical Parameters | Control | 1WRA | |
|---|---|---|---|
| Alanine aminotransferase (ALT) | Unit/L | 72.7 ± 13.3 | 66.3 ± 28.0 |
| Aspartate aminotransferase (AST) | Unit/L | 27.3 ± 3.1 | 41.0 ± 29.8 |
| Triglyceride | mg/dL | 80.3 ± 57.0 | 73.7 ± 10.5 |
| Total cholesterol | mg/dL | 62.7 ± 13.3 | 53.7 ± 9.1 |
| HDL cholesterol | mg/dL | 54.7 ± 9.9 | 49.0 ± 1.7 |
| LDL cholesterol | mg/dL | 9.0 ± 1.0 | 7.7 ± 1.2 |
| Free Fatty acid | mg/dL | 161.3 ± 73.0 | 166.0 ± 12.2 |
| Blood urea nitrogen (BUN) | mg/dL | 34.9 ± 7.2 | 33.4 ± 12.1 |
| Creatinine | mg/dL | 0.5 ± 0.1 | 0.5 ± 0.1 |
| Parameters | Groups | ||
|---|---|---|---|
| Control (n = 4) | 1WRA (n = 6) | ||
| C0 | μg/mL | 15.6 ± 2.6 | 9.7 ± 1.7 * |
| AUC6h | μg min/mL | 3.8 ± 0.2 | 5.6 ± 2.2 * |
| AUC∞ | μg·min/mL | 3.8 ± 0.2 | 5.7 ± 2.3 * |
| t1/2 | h | 0.7 ± 0.2 | 0.9 ± 0.2 * |
| MRT | h | 0.7 ± 0.2 | 0.9 ± 0.3 |
| Vd | mL/kg | 518.1 ± 119.0 | 477.2 ± 109.3 |
| CLtotal | mL/min/kg | 13.1 ± 0.7 | 9.8 ± 3.1 * |
| CLbile | mL/min/kg | 9.2 ± 1.0 | 4.3 ± 2.4 * |
| CLurine | mL/min/kg | 2.3 ± 1.5 | 1.8 ± 1.1 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, S.; Kwon, M.; Choi, M.-K.; Song, I.-S. Effects of Red Ginseng Extract on the Pharmacokinetics and Elimination of Methotrexate via Mrp2 Regulation. Molecules 2018, 23, 2948. https://doi.org/10.3390/molecules23112948
Lee S, Kwon M, Choi M-K, Song I-S. Effects of Red Ginseng Extract on the Pharmacokinetics and Elimination of Methotrexate via Mrp2 Regulation. Molecules. 2018; 23(11):2948. https://doi.org/10.3390/molecules23112948
Chicago/Turabian StyleLee, Sowon, Mihwa Kwon, Min-Koo Choi, and Im-Sook Song. 2018. "Effects of Red Ginseng Extract on the Pharmacokinetics and Elimination of Methotrexate via Mrp2 Regulation" Molecules 23, no. 11: 2948. https://doi.org/10.3390/molecules23112948
APA StyleLee, S., Kwon, M., Choi, M.-K., & Song, I.-S. (2018). Effects of Red Ginseng Extract on the Pharmacokinetics and Elimination of Methotrexate via Mrp2 Regulation. Molecules, 23(11), 2948. https://doi.org/10.3390/molecules23112948