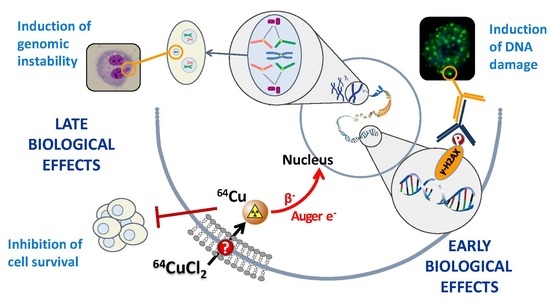
Radiobiological Characterization of 64CuCl2 as a Simple Tool for Prostate Cancer Theranostics
Abstract
1. Introduction
2. Results
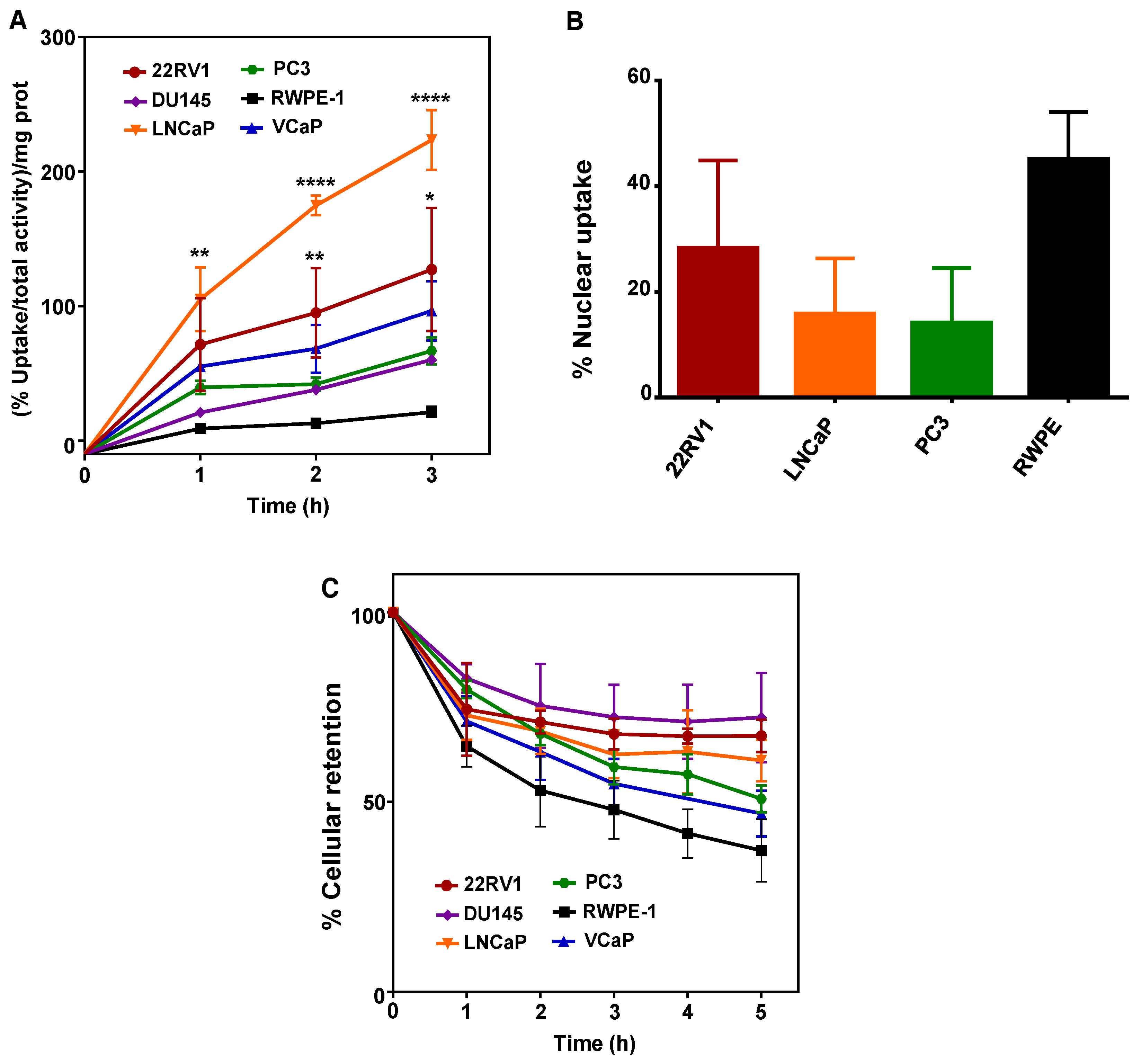
2.1. 64CuCl2 Exhibits Increased Uptake in PCa Cell Lines
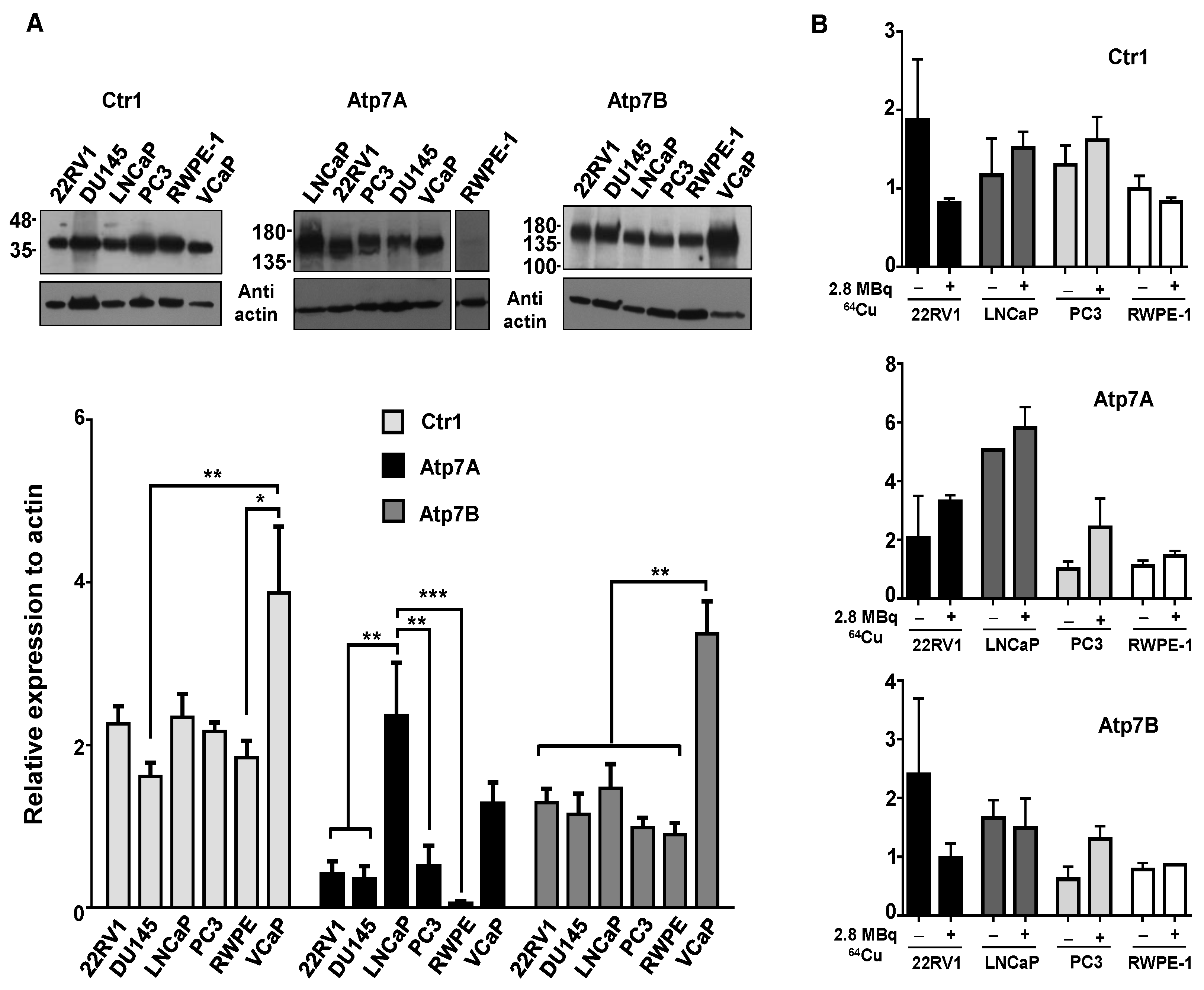
2.2. Increased 64CuCl2 Uptake Does Not Reflect Differential Expression of Copper Transporters
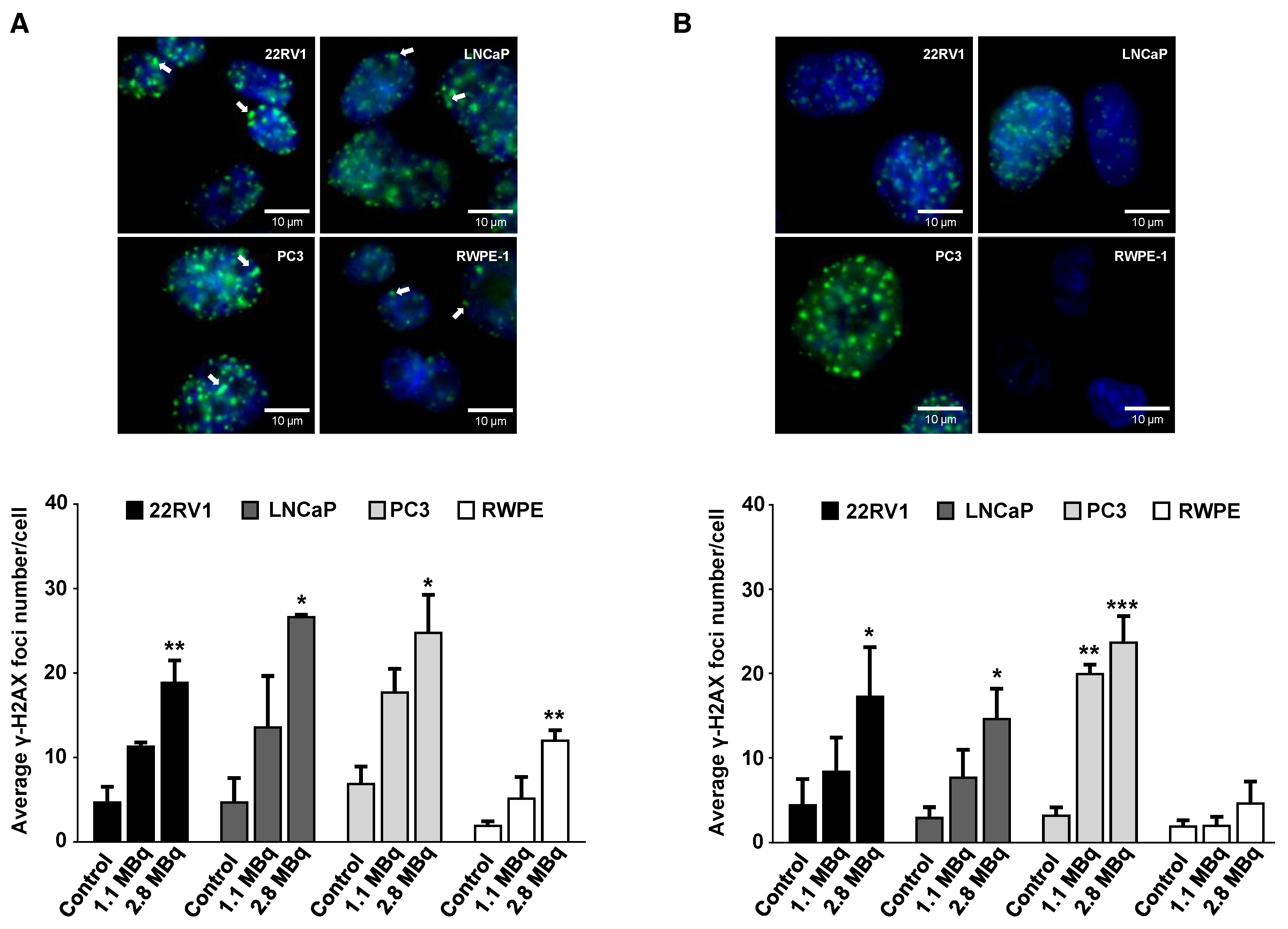
2.3. 64CuCl2 Induces DNA Damage in PCa Cells
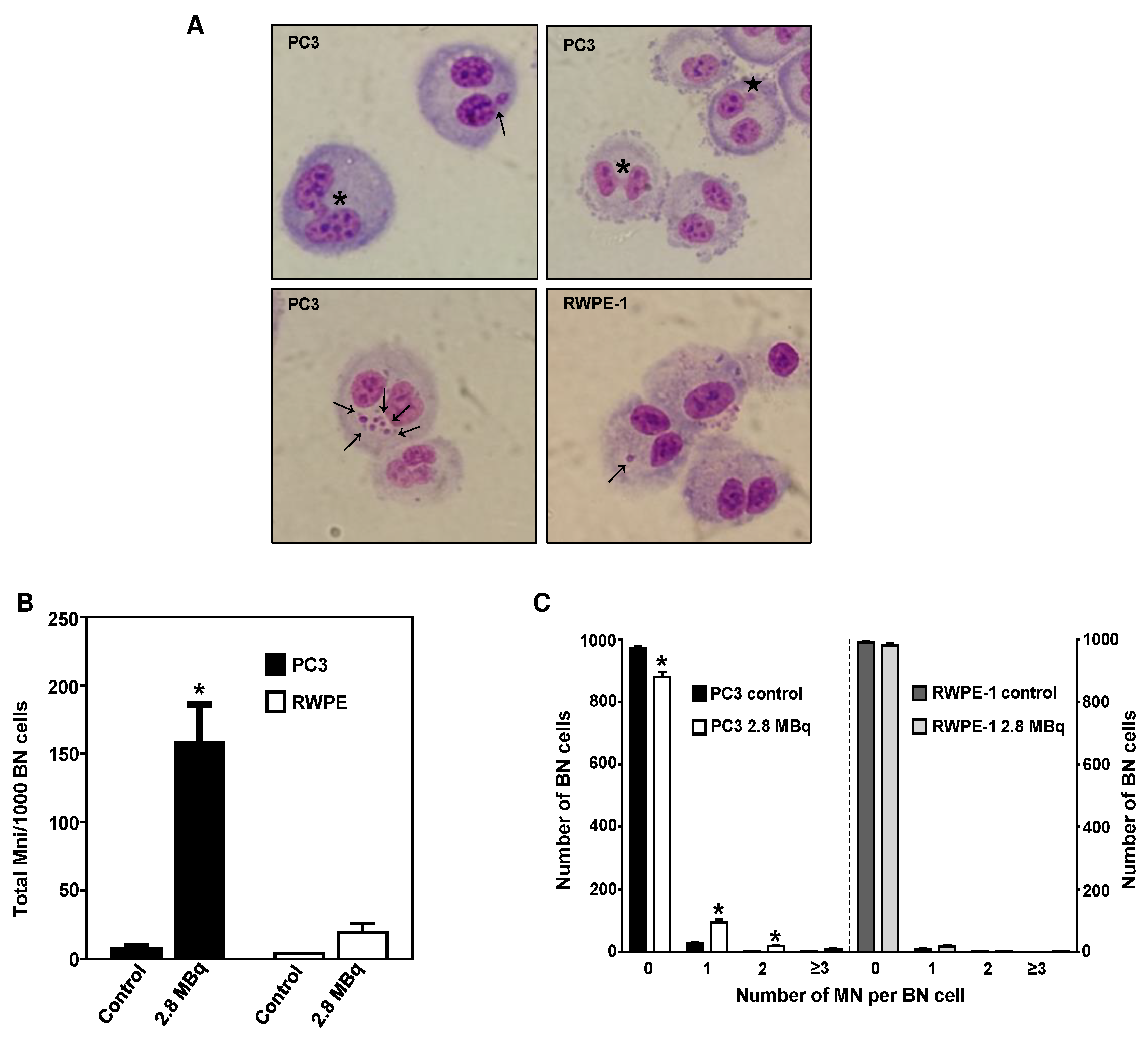
2.4. 64CuCl2 Induces Genomic Instability in PCa Cells
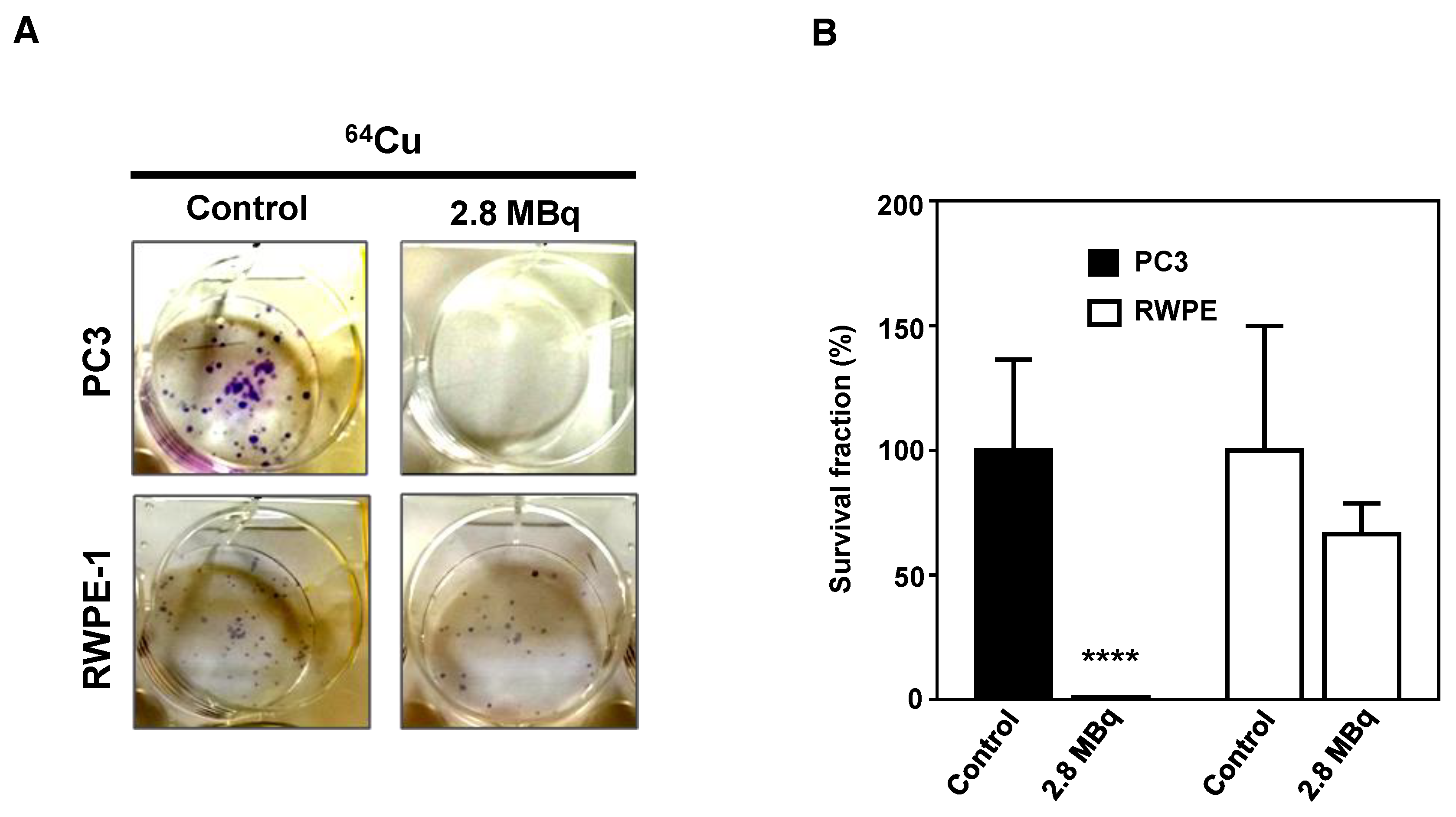
2.5. 64CuCl2 Exhibits Potent Anti-Proliferative Activity PCa Cells
3. Discussion
4. Materials and Methods
4.1. Cell Culture
4.2. Production of 64CuCl2
4.3. Cellular and Nuclear Uptake Assays
4.4. Efflux Assays
4.5. Protein Extraction
4.6. Western Blotting
4.7. γ-H2AX Assay and Foci Analysis
4.8. Cytokinesis-Blocked Micronucleus (CBMN) Assay
4.9. Cell Proliferation and Colony Formation Assay
4.10. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| ATSM | diacetyl-bis N4-methylthiosemicarbazone |
| BN | binucleated |
| CT | computed tomography |
| CBMN | cytokinesis-block micronucleus |
| DSBs | double-strand breaks |
| LET | linear energy transfer |
| MBq | megabecquerel |
| MN | micronucleus |
| MNi | micronuclei |
| PBS | phosphate-buffered saline |
| PCa | prostate cancer |
| PET | positron emission tomography |
| PET/CT | positron emission tomography/computed tomography |
References
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2018. CA Cancer J. Clin. 2018, 68, 7–30. [Google Scholar] [CrossRef] [PubMed]
- Eurostat. Cancer Statistics–Specific Cancers. 2017. Available online: https://ec.europa.eu/eurostat/statistics-explained/index.php/Cancer_statistics_-_specific_cancers (accessed on 11 November 2018).
- Sumanasuriya, S.; De Bono, J. Treatment of advanced prostate cancer—A review of current therapies and future promise. Cold Spring Harb. Perspect. Med. 2018, 8. [Google Scholar] [CrossRef] [PubMed]
- Kitajima, K.; Yamamoto, S.; Fukushima, K.; Minamimoto, R.; Kamai, T.; Jadvar, H. Update on advances in molecular PET in urological oncology. Jpn J. Radiol. 2016, 34, 470–485. [Google Scholar] [CrossRef] [PubMed]
- Marko, J.; Gould, C.F.; Bonavia, G.H.; Wolfman, D.J. State of the art imaging of prostate cancer. Urol. Oncol. 2016, 34, 134–146. [Google Scholar] [CrossRef] [PubMed]
- Kelloff, G.J.; Choyke, P.; Coffey, D.S. Challenges in clinical prostate cancer: Role of imaging. AJR Am. J. Roentgenol. 2009, 192, 1455–1470. [Google Scholar] [CrossRef] [PubMed]
- Weissleder, R.; Schwaiger, M.C.; Gambhir, S.S.; Hricak, H. Imaging approaches to optimize molecular therapies. Sci. Transl. Med. 2016, 8, 355. [Google Scholar] [CrossRef] [PubMed]
- Niccoli Asabella, A.; Cascini, G.L.; Altini, C.; Paparella, D.; Notaristefano, A.; Rubini, G. The copper radioisotopes: A systematic review with special interest to 64Cu. Biomed. Res. Int. 2014, 2014. [Google Scholar] [CrossRef] [PubMed]
- Ramogida, C.F.; Orvig, C. Tumour targeting with radiometals for diagnosis and therapy. Chem. Commun. (Camb) 2013, 49, 4720–4739. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Pena, M.M.O.; Nose, Y.; Thiele, D.J. Biochemical characterization of the human copper transporter Ctr1. J. Biol. Chem. 2002, 277, 4380–4387. [Google Scholar] [CrossRef] [PubMed]
- Brewer, G.J. Anticopper therapy against cancer and diseases of inflammation and fibrosis. Drug. Discov. Today 2005, 10, 1103–1109. [Google Scholar] [CrossRef]
- Ishida, S.; Andreux, P.; Poitry-Yamate, C.; Auwerx, J.; Hanahan, D. Bioavailable copper modulates oxidative phosphorylation and growth of tumors. Proc. Natl. Acad. Sc.i USA 2013, 110, 19507–19512. [Google Scholar] [CrossRef] [PubMed]
- Evangelista, L.; Luigi, M.; Cascini, G.L. New issues for copper-64: From precursor to innovative PET tracers in clinical oncology. Curr. Radiopharm. 2013, 6, 117–123. [Google Scholar] [CrossRef] [PubMed]
- McMillan, D.D.; Maeda, J.; Bell, J.J.; Genet, M.D.; Phoonswadi, G.; Mann, K.A.; Kraft, S.L.; Kitamura, H.; Fujimori, A.; Yoshii, Y.; et al. Validation of 64Cu-ATSM damaging DNA via high-LET Auger electron emission. J. Radiat. Res. 2015, 56, 784–791. [Google Scholar] [CrossRef] [PubMed]
- Weeks, A.J.; Paul, R.L.; Marsden, P.K.; Blower, P.J.; Lloyd, D.R. Radiobiological effects of hypoxia-dependent uptake of 64Cu-ATSM: Enhanced DNA damage and cytotoxicity in hypoxic cells. Eur. J. Nucl. Med. Mol. Imaging 2010, 37, 330–338. [Google Scholar] [CrossRef] [PubMed]
- Obata, A.; Kasamatsu, S.; Lewis, J.S.; Furukawa, T.; Takamatsu, S.; Toyohara, J.; Asai, T.; Welch, M.J.; Adams, S.G.; Saji, H.; et al. Basic characterization of 64Cu-ATSM as a radiotherapy agent. Nucl. Med. Biol. 2005, 32, 21–28. [Google Scholar] [CrossRef] [PubMed]
- Qin, C.; Liu, H.; Chen, K.; Hu, X.; Ma, X.; Lan, X.; Zhang, Y.; Cheng, Z. Theranostics of malignant melanoma with 64CuCl2. J. Nucl. Med. 2014, 55, 812–817. [Google Scholar] [CrossRef] [PubMed]
- Peng, F.; Lu, X.; Janisse, J.; Muzik, O.; Shields, A.F. PET of human prostate cancer xenografts in mice with increased uptake of 64CuCl2. J. Nucl. Med. 2006, 47, 1649–1652. [Google Scholar] [PubMed]
- Ferrari, C.; Asabella, A.N.; Villano, C.; Giacobbi, B.; Coccetti, D.; Panichelli, P.; Rubini, G. Copper-64 Dichloride as Theranostic Agent for Glioblastoma Multiforme: A Preclinical Study. Biomed. Res. Int. 2015, 2015. [Google Scholar] [CrossRef] [PubMed]
- Catalogna, G.; Talarico, C.; Dattilo, V.; Gangemi, V.; Calabria, F.; D’Antona, L.; Schenone, S.; Musumeci, F.; Bianco, C.; Perrotti, N.; et al. The SGK1 Kinase Inhibitor SI113 Sensitizes Theranostic Effects of the 64CuCl2 in Human Glioblastoma Multiforme Cells. Cell. Physiol. Biochem. 2017, 43, 108–119. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.I.; Jang, S.J.; Park, J.H.; Lee, Y.J.; Lee, T.S.; Woo, K.S.; Park, H.; Choe, J.G.; An, G.I.; Kang, J.H. Detection of increased 64Cu uptake by human copper transporter 1 gene overexpression using PET with 64CuCl2 in human breast cancer xenograft model. J. Nucl. Med. 2014, 55, 1692–1698. [Google Scholar] [CrossRef] [PubMed]
- Piccardo, A.; Paparo, F.; Puntoni, M.; Righi, S.; Bottoni, G.; Bacigalupo, L.; Zanardi, S.; DeCensi, A.; Ferrarazzo, G.; Gambaro, M.; et al. 64CuCl2 PET/CT in Prostate Cancer Relapse. J. Nucl. Med. 2018, 59, 444–451. [Google Scholar] [CrossRef] [PubMed]
- Capasso, E.; Durzu, S.; Piras, S.; Zandieh, S.; Knoll, P.; Haug, A.; Hacker, M.; Meleddu, C.; Mirzaei, S. Role of 64CuCl2 PET/CT in staging of prostate cancer. Ann. Nucl. Med. 2015, 29, 482–488. [Google Scholar] [CrossRef] [PubMed]
- Pereira, E.; do Quental, L.; Palma, E.; Oliveira, M.C.; Mendes, F.; Raposinho, P.; Correia, I.; Lavrado, J.; Di Maria, S.; Belchior, A.; et al. Evaluation of Acridine Orange Derivatives as DNA-Targeted Radiopharmaceuticals for Auger Therapy: Influence of the Radionuclide and Distance to DNA. Sci. Rep. 2017, 7, 42544. [Google Scholar] [CrossRef] [PubMed]
- Cornelissen, B.; Vallis, K.A. Targeting the nucleus: An overview of Auger-electron radionuclide therapy. Curr. Drug Discov. Technol. 2010, 7, 263–279. [Google Scholar] [CrossRef] [PubMed]
- Cai, H.; Wu, J.S.; Muzik, O.; Hsieh, J.T.; Lee, R.J.; Peng, F. Reduced 64Cu uptake and tumor growth inhibition by knockdown of human copper transporter 1 in xenograft mouse model of prostate cancer. J. Nucl. Med. 2014, 55, 622–628. [Google Scholar] [CrossRef] [PubMed]
- Rogakou, E.P.; Pilch, D.R.; Orr, A.H.; Ivanova, V.S.; Bonner, W.M. DNA double-stranded breaks induce histone H2AX phosphorylation on serine 139. J. Biol. Chem. 1998, 273, 5858–5868. [Google Scholar] [CrossRef] [PubMed]
- Desai, N.; Davis, E.; O’Neill, P.; Durante, M.; Cucinotta, F.A.; Wu, H. Immunofluorescence detection of clustered gamma-H2AX foci induced by HZE-particle radiation. Rad. Res. 2005, 164, 518–522. [Google Scholar] [CrossRef]
- Fenech, M. Cytokinesis-block micronucleus cytome assay. Nat. Prot. 2007, 2, 1084–1104. [Google Scholar] [CrossRef] [PubMed]
- Franken, N.A.; Rodermond, H.M.; Stap, J.; Haveman, J.; van Bree, C. Clonogenic assay of cells in vitro. Nat. Prot. 2006, 1, 2315–2319. [Google Scholar] [CrossRef] [PubMed]
- Righi, S.; Ugolini, M.; Bottoni, G.; Puntoni, M.; Iacozzi, M.; Paparo, F.; Cabria, M.; Ceriani, L.; Gambaro, M.; Giovanella, L.; et al. Biokinetic and dosimetric aspects of 64CuCl2 in human prostate cancer: Possible theranostic implications. EJNMMI Res. 2018, 8, 18. [Google Scholar] [CrossRef] [PubMed]
- Cunningham, D.; You, Z. In vitro and in vivo model systems used in prostate cancer research. J. Biol. Met. 2015, 2. [Google Scholar] [CrossRef] [PubMed]
- Brogsitter, C.; Zophel, K.; Kotzerke, J. 18F-Choline, 11C-choline and 11C-acetate PET/CT: Comparative analysis for imaging prostate cancer patients. EJNMMI 2013, 40 (Suppl. 1), S18–S27. [Google Scholar] [CrossRef] [PubMed]
- Jorgensen, J.T.; Persson, M.; Madsen, J.; Kjaer, A. High tumor uptake of 64Cu: Implications for molecular imaging of tumor characteristics with copper-based PET tracers. Nucl. Med. Biol. 2013, 40, 345–350. [Google Scholar] [CrossRef] [PubMed]
- Holzer, A.K.; Varki, N.M.; Le, Q.T.; Gibson, M.A.; Naredi, P.; Howell, S.B. Expression of the human copper influx transporter 1 in normal and malignant human tissues. J. Histochem. Cytochem. 2006, 54, 1041–1049. [Google Scholar] [CrossRef] [PubMed]
- Sage, E.; Shikazono, N. Radiation–induced clustered DNA lesions: Repair and mutagenesis. Free Radic. Biol. Med. 2017, 107, 125–135. [Google Scholar] [CrossRef] [PubMed]
- Cai, Z.; Chen, Z.; Bailey, K.E.; Scollard, D.A.; Reilly, R.M.; Vallis, K.A. Relationship between induction of phosphorylated H2AX and survival in breast cancer cells exposed to 111In-DTPA-hEGF. J. Nucl. Med. 2008, 49, 1353–1361. [Google Scholar] [CrossRef] [PubMed]
- Bookstein, R.; MacGrogan, D.; Hilsenbeck, S.G.; Sharkey, F.; Allred, D.C. P53 is mutated in a subset of advanced-stage prostate cancers. Cancer Res. 1993, 53, 3369–3373. [Google Scholar] [PubMed]
- Heidenberg, H.B.; Bauer, J.J.; McLeod, D.G.; Moul, J.W.; Srivastava, S. The role of the p53 tumor suppressor gene in prostate cancer: A possible biomarker? Urology 1996, 48, 971–979. [Google Scholar] [CrossRef]
- Scott, S.L.; Earle, J.D.; Gumerlock, P.H. Functional p53 increases prostate cancer cell survival after exposure to fractionated doses of ionizing radiation. Cancer Res. 2003, 63, 7190–7196. [Google Scholar] [PubMed]
- Van Oorschot, B.; Hovingh, S.E.; Rodermond, H.; Guclu, A.; Losekoot, N.; Geldof, A.A.; Barendsen, G.W.; Stalpers, L.J.; Franken, N.A. Decay of gamma-H2AX foci correlates with potentially lethal damage repair in prostate cancer cells. Oncol. Rep- 2013, 29, 2175–2180. [Google Scholar] [CrossRef] [PubMed]
- Robinson, D.; Van Allen, E.M.; Wu, Y.M.; Schultz, N.; Lonigro, R.J.; Mosquera, J.M.; Montgomery, B.; Taplin, M.E.; Pritchard, C.C.; Attard, G.; et al. Integrative clinical genomics of advanced prostate cancer. Cell 2015, 161, 1215–1228. [Google Scholar] [CrossRef] [PubMed]
- Dhawan, M.; Ryan, C.J.; Ashworth, A. DNA Repair Deficiency Is Common in Advanced Prostate Cancer: New Therapeutic Opportunities. Oncologist 2016, 21, 940–945. [Google Scholar] [CrossRef] [PubMed]
- Cheng, H.H. The resounding effect of DNA repair deficiency in prostate cancer. Urol. Oncol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Avila-Rodriguez, M.A.; Rios, C.; Carrasco-Hernandez, J.; Manrique-Arias, J.C.; Martinez-Hernandez, R.; Garcia-Perez, F.O.; Jalilian, A.R.; Martinez-Rodriguez, E.; Romero-Pina, M.E.; Diaz-Ruiz, A. Biodistribution and radiation dosimetry of [(64)Cu]copper dichloride: First in human study in healthy volunteers. EJNMMI Res. 2017, 7, 98. [Google Scholar] [CrossRef] [PubMed]
- Alves, F.; Alves, V.H.P.; Do Carmo, S.J.C.; Neves, A.C.B.; Silva, M.; Abrunhosa, A.J. Production of copper–64 and gallium–68 with a medical cyclotron using liquid targets. Mod. Phys. Lett. A 2017, 32, 1740013. [Google Scholar] [CrossRef]
- Palma, E.; Mendes, F.; Morais, G.R.; Rodrigues, I.; Santos, I.C.; Campello, M.P.; Raposinho, P.; Correia, I.; Gama, S.; Belo, D.; et al. Biophysical characterization and antineoplastic activity of new bis(thiosemicarbazonato) Cu(II) complexes. J. Inorg. Biochem. 2017, 167, 68–79. [Google Scholar] [CrossRef] [PubMed]
- Belchior, A.; Gil, O.M.; Almeida, P.; Vaz, P. Dose and Time Dependence of Targeted and Untargeted Effects after Very Low Doses of Alpha-Particle Irradiation of Human Lung Cancer Cells. Dose Response 2013, 11, 431–446. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guerreiro, J.F.; Alves, V.; Abrunhosa, A.J.; Paulo, A.; Gil, O.M.; Mendes, F. Radiobiological Characterization of 64CuCl2 as a Simple Tool for Prostate Cancer Theranostics. Molecules 2018, 23, 2944. https://doi.org/10.3390/molecules23112944
Guerreiro JF, Alves V, Abrunhosa AJ, Paulo A, Gil OM, Mendes F. Radiobiological Characterization of 64CuCl2 as a Simple Tool for Prostate Cancer Theranostics. Molecules. 2018; 23(11):2944. https://doi.org/10.3390/molecules23112944
Chicago/Turabian StyleGuerreiro, Joana Fernandes, Vítor Alves, Antero José Abrunhosa, António Paulo, Octávia Monteiro Gil, and Filipa Mendes. 2018. "Radiobiological Characterization of 64CuCl2 as a Simple Tool for Prostate Cancer Theranostics" Molecules 23, no. 11: 2944. https://doi.org/10.3390/molecules23112944
APA StyleGuerreiro, J. F., Alves, V., Abrunhosa, A. J., Paulo, A., Gil, O. M., & Mendes, F. (2018). Radiobiological Characterization of 64CuCl2 as a Simple Tool for Prostate Cancer Theranostics. Molecules, 23(11), 2944. https://doi.org/10.3390/molecules23112944