Leptodactylus latrans Amphibian Skin Secretions as a Novel Source for the Isolation of Antibacterial Peptides
Abstract
1. Introduction
2. Results
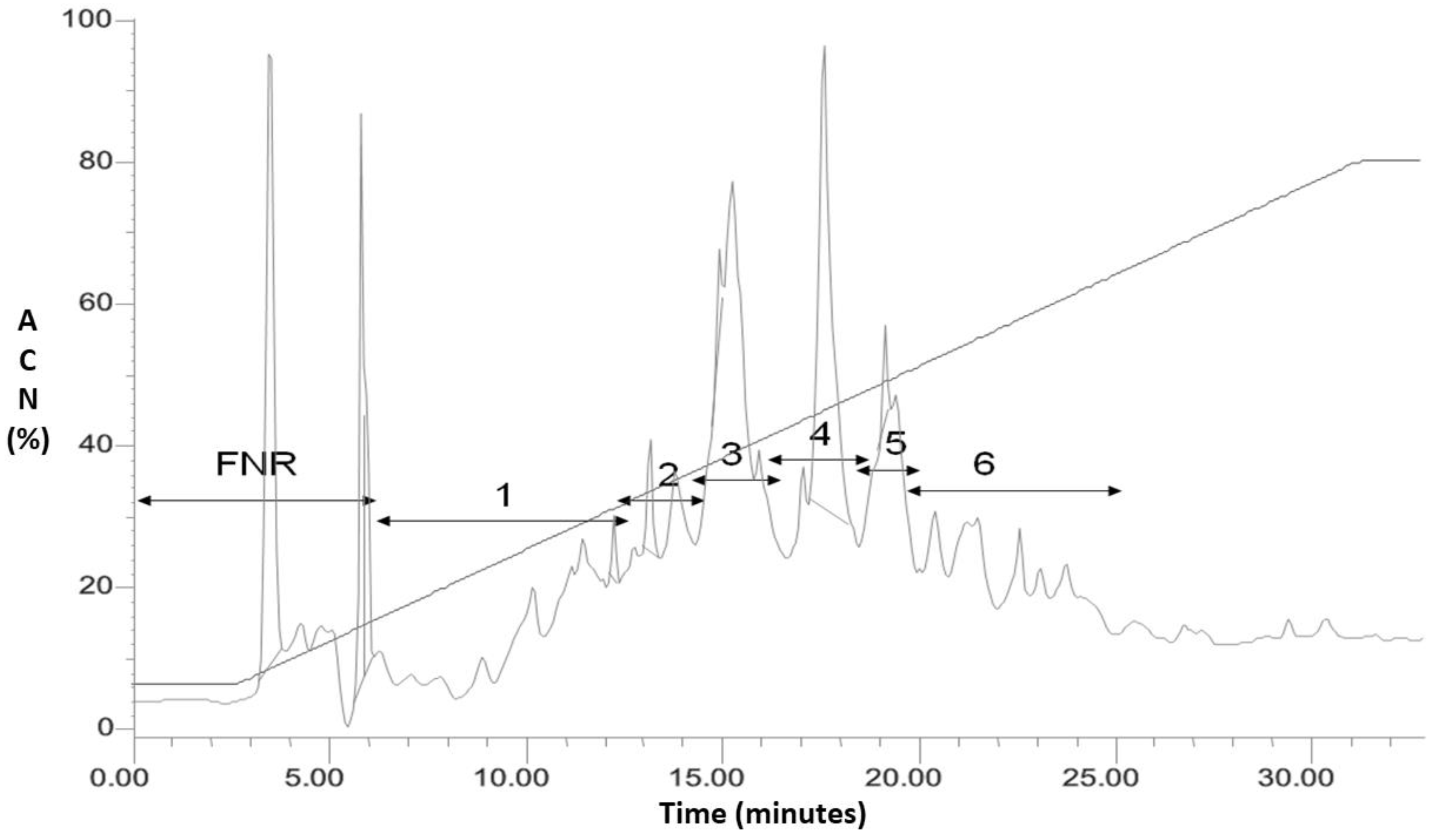
2.1. Analysis and Purification of the Complete Extract of L. latrans
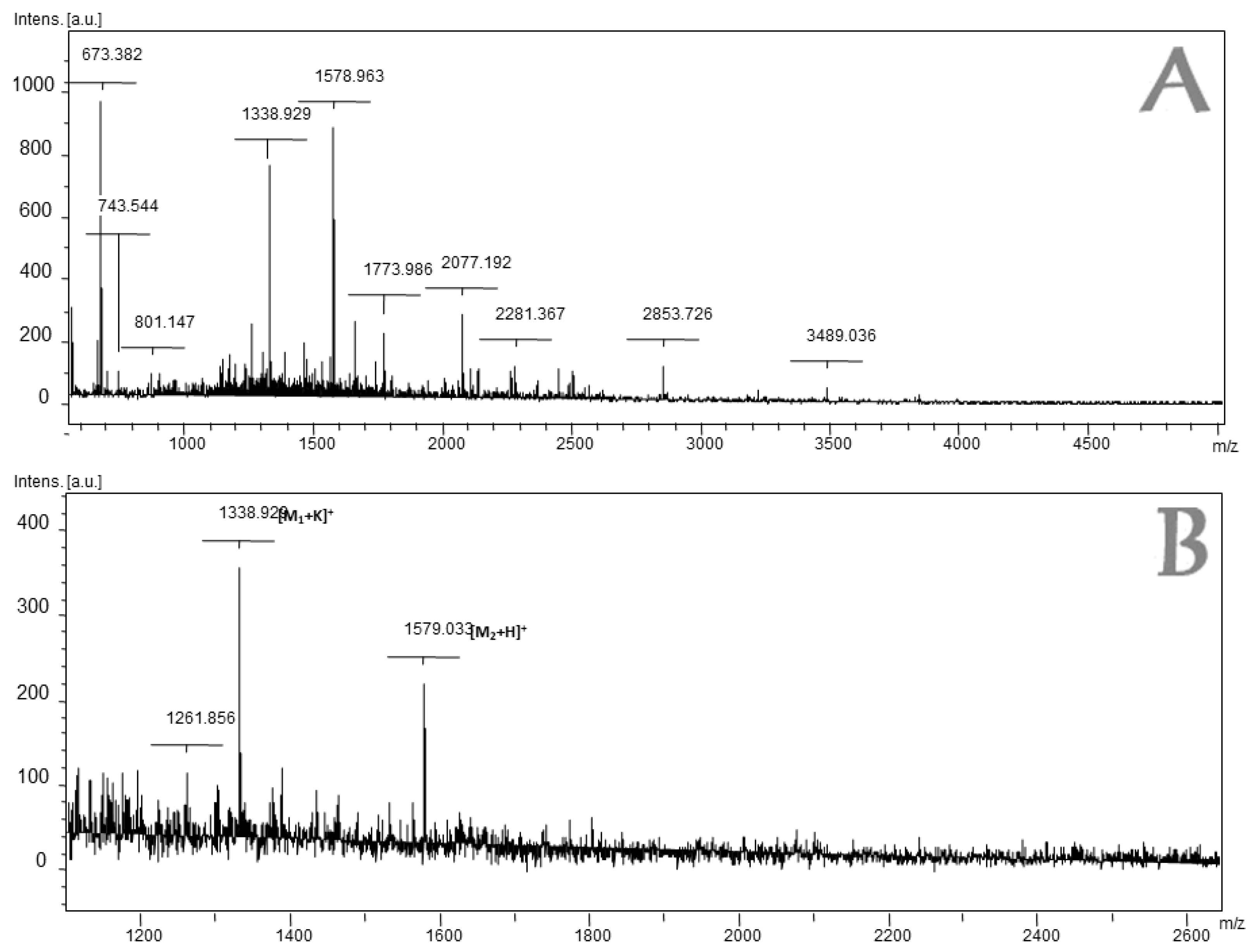
2.2. Mass Spectrometry Analysis of Fraction 4 of L. latrans
2.3. Solid-Phase Peptide Synthesis
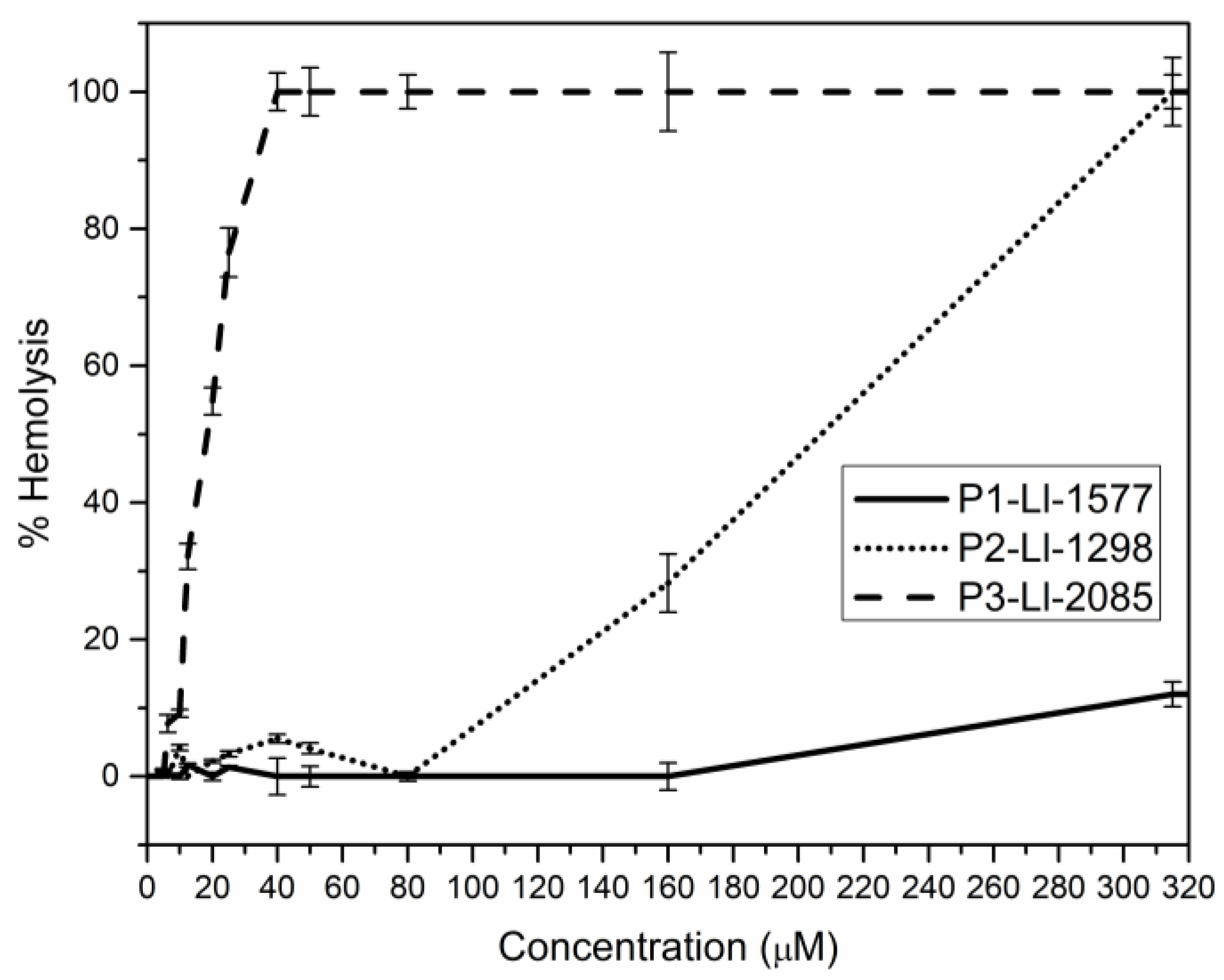
2.4. Antimicrobial and Hemolytic Activity of the Synthetic Peptides
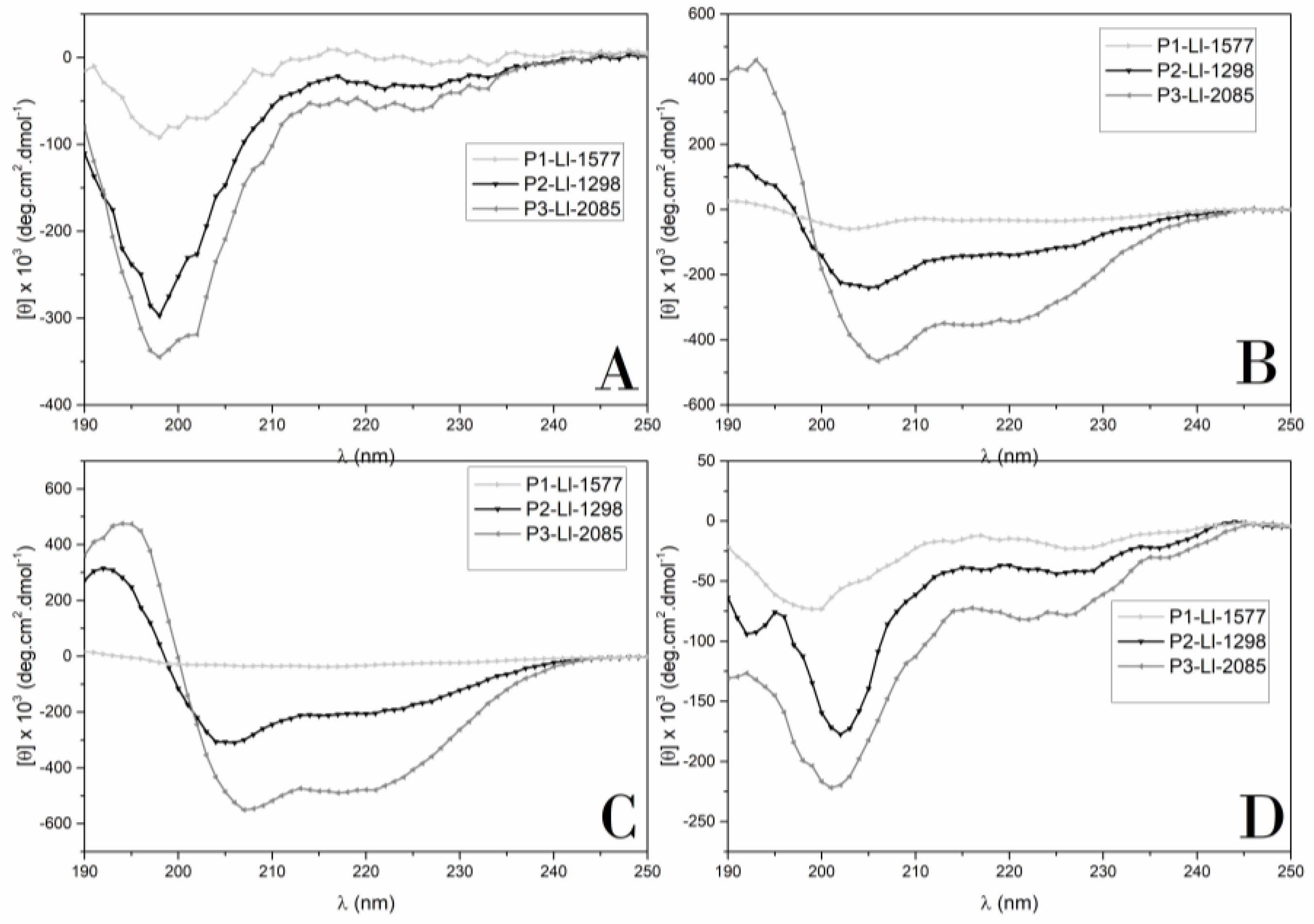
2.5. Secondary Structure Determination by Circular Dichroism (CD)
3. Discussion
4. Materials and Methods
4.1. Collection of Amphibian Specimens
4.2. Method for Biological Sampling. Transcutaneous Amphibian Stimulation (TAS)
4.3. Liquid Chromatography coupled to Mass Spectrometry (LC-MS-MS)
4.4. Structure Analysis
4.5. Circular Dichroism (CD) Analyses
4.6. Peptide Synthesis
4.7. Minimal Inhibitory Concentration (MIC) Determination
4.8. Hemolysis Assays
4.9. Calculation of Therapeutic Index
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Yeung, A.T.; Gellatly, S.L.; Hancock, R.E. Multifunctional cationic host defence peptides and their clinical applications. Cell. Mol. Life Sci. 2011, 68, 2161–2176. [Google Scholar] [CrossRef] [PubMed]
- Silver, L.L. Challenges of antibacterial discovery. Clin. Microbiol. Rev. 2011, 24, 71–109. [Google Scholar] [CrossRef] [PubMed]
- Silver, L.L. A persistent problem. J. Med. Microbiol. 2011, 60, 267–268. [Google Scholar] [CrossRef] [PubMed]
- Newman, D.J.; Cragg, G.M. Natural products as sources of new drugs over the last 25 years. J. Nat. Prod. 2007, 70, 461–477. [Google Scholar] [CrossRef] [PubMed]
- Newman, D.J.; Cragg, G.M. Natural products as sources of new drugs over the 30 years from 1981 to 2010. J. Nat. Prod. 2012, 75, 311–335. [Google Scholar] [CrossRef] [PubMed]
- Butler, M.S.; Buss, A.D. Natural products--the future scaffolds for novel antibiotics? Biochem. Pharmacol. 2006, 71, 919–929. [Google Scholar] [CrossRef] [PubMed]
- Saleem, M.; Nazir, M.; Ali, M.S.; Hussain, H.; Lee, Y.S.; Riaz, N.; Jabbar, A. Antimicrobial natural products: An update on future antibiotic drug candidates. Nat. Prod. Rep. 2010, 27, 238–254. [Google Scholar] [CrossRef] [PubMed]
- Albericio, F.; Kruger, H.G. Therapeutic peptides. Future Med. Chem. 2012, 4, 1527–1531. [Google Scholar] [CrossRef] [PubMed]
- Kaspar, A.A.; Reichert, J.M. Future directions for peptide therapeutics development. Drug Discov. Today 2013, 18, 807–817. [Google Scholar] [CrossRef] [PubMed]
- De la Torre, B.G.; Albericio, F. The Pharmaceutical Industry in 2017. An Analysis of FDA Drug Approvals from the Perspective of Molecules. Molecules 2018, 23, 533. [Google Scholar] [CrossRef] [PubMed]
- Bulet, P.; Stocklin, R.; Menin, L. Anti-microbial peptides: From invertebrates to vertebrates. Immunol. Rev. 2004, 198, 169–184. [Google Scholar] [CrossRef] [PubMed]
- Lehrer, R.I.; Ganz, T. Antimicrobial peptides in mammalian and insect host defence. Curr. Opin. Immunol. 1999, 11, 23–27. [Google Scholar] [CrossRef]
- Berkowitz, B.A.; Bevins, C.L.; Zasloff, M.A. Magainins: A new family of membrane-active host defense peptides. Biochem. Pharmacol. 1990, 39, 625–629. [Google Scholar] [CrossRef]
- Bevins, C.L.; Zasloff, M. Peptides from frog skin. Annu. Rev. Biochem. 1990, 59, 395–414. [Google Scholar] [CrossRef] [PubMed]
- Lehrer, R.I.; Ganz, T.; Selsted, M.E. Defensins: Endogenous antibiotic peptides of animal cells. Cell 1991, 64, 229–230. [Google Scholar] [CrossRef]
- Zasloff, M. Antimicrobial peptides of multicellular organisms. Nature 2002, 415, 389–395. [Google Scholar] [CrossRef] [PubMed]
- Nicolas, P. Multifunctional host defense peptides: Intracellular-targeting antimicrobial peptides. FEBS J. 2009, 276, 6483–6496. [Google Scholar] [CrossRef] [PubMed]
- Yeaman, M.R.; Yount, N.Y. Mechanisms of antimicrobial peptide action and resistance. Pharmacol. Rev. 2003, 55, 27–55. [Google Scholar] [CrossRef] [PubMed]
- Park, C.B.; Kim, H.S.; Kim, S.C. Mechanism of action of the antimicrobial peptide buforin II: Buforin II kills microorganisms by penetrating the cell membrane and inhibiting cellular functions. Biochem. Biophys. Res. Commun. 1998, 244, 253–257. [Google Scholar] [CrossRef] [PubMed]
- Conlon, J.M.; Kolodziejek, J.; Nowotny, N. Antimicrobial peptides from ranid frogs: Taxonomic and phylogenetic markers and a potential source of new therapeutic agents. Biochim. Biophys. Acta 2004, 1696, 1–14. [Google Scholar] [CrossRef] [PubMed]
- You, D.; Hong, J.; Rong, M.; Yu, H.; Liang, S.; Ma, Y.; Yang, H.; Wu, J.; Lin, D.; Lai, R. The first gene-encoded amphibian neurotoxin. J. Biol. Chem. 2009, 284, 22079–22086. [Google Scholar] [CrossRef] [PubMed]
- Daly, J.W.; Spande, T.F.; Garraffo, H.M. Alkaloids from amphibian skin: A tabulation of over eight-hundred compounds. J. Nat. Prod. 2005, 68, 1556–1575. [Google Scholar] [CrossRef] [PubMed]
- Giovannini, M.G.; Poulter, L.; Gibson, B.W.; Williams, D.H. Biosynthesis and degradation of peptides derived from Xenopus laevis prohormones. Biochem. J. 1987, 243, 113–120. [Google Scholar] [CrossRef] [PubMed]
- Dockray, G.J.; Hopkins, C.R. Caerulein secretion by dermal glands in Xenopus laevis. J. Cell Biol. 1975, 64, 724–733. [Google Scholar] [CrossRef] [PubMed]
- Mills, J.W.; Prum, B.E. Morphology of the exocrine glands of the frog skin. Am. J. Anat. 1984, 171, 91–106. [Google Scholar] [CrossRef] [PubMed]
- Siano, A.; Gatti, P.I.; Imaz, M.S.; Zerbini, E.; Simonetta, A.; Lajmanovich, R.; Tonarelli, G. A Comparative Study of the Biological Activity of Skin and Granular Gland Secretions of Leptodactylus latrans and Hypsiboas pulchellus from Argentina. Rec. Nat. Prod. 2014, 8, 8. [Google Scholar]
- Conlon, J.M. Structural diversity and species distribution of host-defense peptides in frog skin secretions. Cell. Mol. Life Sci. 2011, 68, 2303–2315. [Google Scholar] [CrossRef] [PubMed]
- Conlon, J.M.; Kolodziejek, J.; Nowotny, N. Antimicrobial peptides from the skins of North American frogs. Biochim. Biophys. Acta 2009, 1788, 1556–1563. [Google Scholar] [CrossRef] [PubMed]
- Thomas, P.; Vineeth Kumar, T.V.; Reshmy, V.; Kumar, K.S.; George, S. A mini review on the antimicrobial peptides isolated from the genus Hylarana (Amphibia: Anura) with a proposed nomenclature for amphibian skin peptides. Mol. Biol. Rep. 2012, 39, 6943–6947. [Google Scholar] [CrossRef] [PubMed]
- Xi, X.; Li, B.; Chen, T.; Kwok, H.F. A review on bradykinin-related peptides isolated from amphibian skin secretion. Toxins 2015, 7, 951–970. [Google Scholar] [CrossRef] [PubMed]
- Nascimento, A. Cytolytic Peptides and Proteases from the Skin Secretion of the Frog Leptodactylus Ocellatus. Ph.D. Thesis, University of Brasilia, Brasilia, Brazil, 2007. [Google Scholar]
- Bottari, E.; Festa, M.R.; Jasionowska, R. Zinc(II) Complexes with Aspartate and Glutamate. J. Coord. Chem. 1990, 21, 215–224. [Google Scholar] [CrossRef]
- Eisenberg, D.; Weiss, R.M.; Terwilliger, T.C. The helical hydrophobic moment: A measure of the amphiphilicity of a helix. Nature 1982, 299, 371–374. [Google Scholar] [CrossRef] [PubMed]
- Conlon, J.M.; Al-Ghaferi, N.; Abraham, B.; Leprince, J. Strategies for transformation of naturally-occurring amphibian antimicrobial peptides into therapeutically valuable anti-infective agents. Methods 2007, 42, 349–357. [Google Scholar] [CrossRef] [PubMed]
- Zhu, W.L.; Nan, Y.H.; Hahm, K.S.; Shin, S.Y. Cell selectivity of an antimicrobial peptide melittin diastereomer with D-amino acid in the leucine zipper sequence. J. Biochem. Mol. Biol. 2007, 40, 1090–1094. [Google Scholar] [CrossRef] [PubMed]
- Cruciani, R.A.; Barker, J.L.; Durell, S.R.; Raghunathan, G.; Guy, H.R.; Zasloff, M.; Stanley, E.F. Magainin 2, a natural antibiotic from frog skin, forms ion channels in lipid bilayer membranes. Eur. J. Pharmacol. 1992, 226, 287–296. [Google Scholar] [CrossRef]
- Nascimento, A.; Zanotta, L.C.; Kyaw, C.M.; Schwartz, E.N.F.; Schwartz, C.A.; Sebben, A.; Sousa, M.V.; Fontes, W.; Castro, M.S. Ocellatins: New antimicrobial peptides from the skin secretion of the South American frog Leptodactylus ocellatus (Anura: Leptodactylidae). Protein J. 2004, 23, 501–508. [Google Scholar] [CrossRef] [PubMed]
- King, J.D.; Al-Ghaferi, N.; Abraham, B.; Sonnevend, A.; Leprince, J.; Nielsen, P.F.; Conlon, J.M. Pentadactylin: An antimicrobial peptide from the skin secretions of the South American bullfrog Leptodactylus pentadactylus. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2005, 141, 393–397. [Google Scholar] [CrossRef] [PubMed]
- Rollins-Smith, L.A.; King, J.D.; Nielsen, P.F.; Sonnevend, A.; Conlon, J.M. An antimicrobial peptide from the skin secretions of the mountain chicken frog Leptodactylus fallax (Anura:Leptodactylidae). Regul. Pept. 2005, 124, 173–178. [Google Scholar] [CrossRef] [PubMed]
- Nascimento, A.; Chapeaurouge, A.; Perales, J.; Sebben, A.; Sousa, M.V.; Fontes, W.; Castro, M.S. Purification, characterization and homology analysis of ocellatin 4, a cytolytic peptide from the skin secretion of the frog Leptodactylus ocellatus. Toxicon 2007, 50, 1095–1104. [Google Scholar] [CrossRef] [PubMed]
- Conlon, J.M.; Abdel-Wahab, Y.H.; Flatt, P.R.; Leprince, J.; Vaudry, H.; Jouenne, T.; Condamine, E. A glycine-leucine-rich peptide structurally related to the plasticins from skin secretions of the frog Leptodactylus laticeps (Leptodactylidae). Peptides 2009, 30, 888–892. [Google Scholar] [CrossRef] [PubMed]
- Sousa, J.C.; Berto, R.F.; Gois, E.A.; Fontenele-Cardi, N.C.; Honorio, J.E. Jr.; Konno, K.; Richardson, M.; Rocha, M.F.; Camargo, A.A.; Pimenta, D.C.; et al. Leptoglycin: A new Glycine/Leucine-rich antimicrobial peptide isolated from the skin secretion of the South American frog Leptodactylus pentadactylus (Leptodactylidae). Toxicon 2009, 54, 23–32. [Google Scholar] [CrossRef] [PubMed]
- Castro, M.S.; Ferreira, T.C.; Cilli, E.M.; Crusca, E. Jr.; Mendes-Giannini, M.J.; Sebben, A.; Ricart, C.A.; Sousa, M.V.; Fontes, W. Hylin a1, the first cytolytic peptide isolated from the arboreal South American frog Hypsiboas albopunctatus (“spotted treefrog”). Peptides 2009, 30, 291–296. [Google Scholar] [CrossRef] [PubMed]
- Castro, M.S.; Matsushita, R.H.; Sebben, A.; Sousa, M.V.; Fontes, W. Hylins: Bombinins H structurally related peptides from the skin secretion of the Brazilian tree-frog Hyla biobeba. Protein Pept. Lett. 2005, 12, 89–93. [Google Scholar] [CrossRef] [PubMed]
- Mangoni, M.L.; Saugar, J.M.; Dellisanti, M.; Barra, D.; Simmaco, M.; Rivas, L. Temporins, small antimicrobial peptides with leishmanicidal activity. J. Biol. Chem. 2005, 280, 984–990. [Google Scholar] [CrossRef] [PubMed]
- Michael Conlon, J. The temporins. In Handbook of Biologically Active Peptides; Abba, J.K., Ed.; Academic Press: Burlington, NJ, USA, 2006; pp. 305–309. [Google Scholar]
- Dathe, M.; Wieprecht, T.; Nikolenko, H.; Handel, L.; Maloy, W.L.; MacDonald, D.L.; Beyermann, M.; Bienert, M. Hydrophobicity, hydrophobic moment and angle subtended by charged residues modulate antibacterial and haemolytic activity of amphipathic helical peptides. FEBS Lett. 1997, 403, 208–212. [Google Scholar] [CrossRef]
- Dathe, M.; Wieprecht, T. Structural features of helical antimicrobial peptides: Their potential to modulate activity on model membranes and biological cells. Biochim. Biophys. Acta 1999, 1462, 71–87. [Google Scholar] [CrossRef]
- Mangoni, M.L.; Grovale, N.; Giorgi, A.; Mignogna, G.; Simmaco, M.; Barra, D. Structure-function relationships in bombinins H, antimicrobial peptides from Bombina skin secretions. Peptides 2000, 21, 1673–1679. [Google Scholar] [CrossRef]
- Mangoni, M.L.; Rinaldi, A.C.; Di Giulio, A.; Mignogna, G.; Bozzi, A.; Barra, D.; Simmaco, M. Structure-function relationships of temporins, small antimicrobial peptides from amphibian skin. Eur. J. Biochem. 2000, 267, 1447–1454. [Google Scholar] [CrossRef] [PubMed]
- Ehrenstein, G.; Lecar, H. Electrically gated ionic channels in lipid bilayers. Q. Rev. Biophys. 1977, 10, 1–34. [Google Scholar] [CrossRef] [PubMed]
- Shai, Y. Mechanism of the binding, insertion and destabilization of phospholipid bilayer membranes by alpha-helical antimicrobial and cell non-selective membrane-lytic peptides. Biochim. Biophys. Acta 1999, 1462, 55–70. [Google Scholar] [CrossRef]
- Bessin, Y.; Saint, N.; Marri, L.; Marchini, D.; Molle, G. Antibacterial activity and pore-forming properties of ceratotoxins: A mechanism of action based on the barrel stave model. Biochim. Biophys. Acta 2004, 1667, 148–156. [Google Scholar] [CrossRef] [PubMed]
- Cespedes, G.F.; Lorenzon, E.N.; Vicente, E.F.; Mendes-Giannini, M.J.; Fontes, W.; Castro, M.S.; Cilli, E.M. Mechanism of action and relationship between structure and biological activity of Ctx-Ha: A new ceratotoxin-like peptide from Hypsiboas albopunctatus. Protein Pept. Lett. 2012, 19, 596–603. [Google Scholar] [CrossRef] [PubMed]
- Lorenzon, E.N.; Cespedes, G.F.; Vicente, E.F.; Nogueira, L.G.; Bauab, T.M.; Castro, M.S.; Cilli, E.M. Effects of dimerization on the structure and biological activity of antimicrobial peptide Ctx-Ha. Antimicrob. Agents Chemother. 2012, 56, 3004–3010. [Google Scholar] [CrossRef] [PubMed]
- Shai, Y.; Oren, Z. From “carpet” mechanism to de-novo designed diastereomeric cell-selective antimicrobial peptides. Peptides 2001, 22, 1629–1641. [Google Scholar] [CrossRef]
- Muller, D.M.; Carrasco, M.S.; Simonetta, A.C.; Beltramini, L.M.; Tonarelli, G.G. A synthetic analog of plantaricin 149 inhibiting food-borne pathogenic bacteria:evidence for alpha-helical conformation involved in bacteria-membrane interaction. J. Pept. Sci. 2007, 13, 171–178. [Google Scholar] [CrossRef] [PubMed]
- Lopes, J.L.; Nobre, T.M.; Siano, A.; Humpola, V.; Bossolan, N.R.; Zaniquelli, M.E.; Tonarelli, G.; Beltramini, L.M. Disruption of Saccharomyces cerevisiae by Plantaricin 149 and investigation of its mechanism of action with biomembrane model systems. Biochim. Biophys. Acta 2009, 1788, 2252–2258. [Google Scholar] [CrossRef] [PubMed]
- Siano, A.; Humpola, M.V.; Rey, M.C.; Simonetta, A.; Tonarelli, G.G. Interaction of acylated and substituted antimicrobial peptide analogs with phospholipid-polydiacetylene vesicles. Correlation with their biological properties. Chem. Biol. Drug Des. 2011, 78, 85–93. [Google Scholar] [CrossRef] [PubMed]
- Lai, R.; Liu, H.; Hui Lee, W.; Zhang, Y. An anionic antimicrobial peptide from toad Bombina maxima. Biochem. Biophys. Res. Commun. 2002, 295, 796–799. [Google Scholar] [CrossRef]
- Brown, S.E.; Howard, A.; Kasprzak, A.B.; Gordon, K.H.; East, P.D. A peptidomics study reveals the impressive antimicrobial peptide arsenal of the wax moth Galleria mellonella. Insect Biochem. Mol. Biol. 2009, 39, 792–800. [Google Scholar] [CrossRef] [PubMed]
- Brogden, K.A.; De Lucca, A.J.; Bland, J.; Elliott, S. Isolation of an ovine pulmonary surfactant-associated anionic peptide bactericidal for Pasteurella haemolytica. Proc. Natl. Acad. Sci. USA 1996, 93, 412–416. [Google Scholar] [CrossRef] [PubMed]
- Heidari, M.; Hamir, A.; Cutlip, R.C.; Brogden, K.A. Antimicrobial anionic peptide binds in vivo to Mannheimia (Pasteurella) haemolytica attached to ovine alveolar epithelium. Int. J. Antimicrob. Agents 2002, 20, 69–72. [Google Scholar] [CrossRef]
- Schittek, B.; Hipfel, R.; Sauer, B.; Bauer, J.; Kalbacher, H.; Stevanovic, S.; Schirle, M.; Schroeder, K.; Blin, N.; Meier, F.; et al. Dermcidin: A novel human antibiotic peptide secreted by sweat glands. Nat. Immunol. 2001, 2, 1133–1137. [Google Scholar] [CrossRef] [PubMed]
- Rieg, S.; Garbe, C.; Sauer, B.; Kalbacher, H.; Schittek, B. Dermcidin is constitutively produced by eccrine sweat glands and is not induced in epidermal cells under inflammatory skin conditions. Br. J. Dermatol. 2004, 151, 534–539. [Google Scholar] [CrossRef] [PubMed]
- Lai, Y.P.; Peng, Y.F.; Zuo, Y.; Li, J.; Huang, J.; Wang, L.F.; Wu, Z.R. Functional and structural characterization of recombinant dermcidin-1L, a human antimicrobial peptide. Biochem. Biophys. Res. Commun. 2005, 328, 243–250. [Google Scholar] [CrossRef] [PubMed]
- Tyler, M.J.; Stone, D.J.; Bowie, J.H. A novel method for the release and collection of dermal, glandular secretions from the skin of frogs. J. Pharmacol. Toxicol. Methods 1992, 28, 199–200. [Google Scholar] [CrossRef]
- Kloczkowski, A.; Ting, K.L.; Jernigan, R.L.; Garnier, J. Combining the GOR V algorithm with evolutionary information for protein secondary structure prediction from amino acid sequence. Proteins 2002, 49, 154–166. [Google Scholar] [CrossRef] [PubMed]
- Camproux, A.C.; Tuffery, P.; Chevrolat, J.P.; Boisvieux, J.F.; Hazout, S. Hidden Markov model approach for identifying the modular framework of the protein backbone. Protein Engin. 1999, 12, 1063–1073. [Google Scholar] [CrossRef]
- Camproux, A.C.; Gautier, R.; Tuffery, P. A hidden markov model derived structural alphabet for proteins. J. Mol. Biol. 2004, 339, 591–605. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.Y.; Chen, X. Improving taxonomy-based protein fold recognition by using global and local features. Proteins 2011, 79, 2053–2064. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Liu, S.; Zhou, Y. SP5: Improving protein fold recognition by using torsion angle profiles and profile-based gap penalty model. PLoS ONE 2008, 3, e2325. [Google Scholar] [CrossRef] [PubMed]
- Sreerama, N.; Venyaminov, S.Y.; Woody, R.W. Analysis of protein circular dichroism spectra based on the tertiary structure classification. Anal. Biochem. 2001, 299, 271–274. [Google Scholar] [CrossRef] [PubMed]
- Sreerama, N.; Woody, R.W. Estimation of protein secondary structure from circular dichroism spectra: Comparison of CONTIN, SELCON, and CDSSTR methods with an expanded reference set. Anal. Biochem. 2000, 287, 252–260. [Google Scholar] [CrossRef] [PubMed]
- Sreerama, N.; Venyaminov, S.Y.; Woody, R.W. Estimation of protein secondary structure from circular dichroism spectra: Inclusion of denatured proteins with native proteins in the analysis. Anal. Biochem. 2000, 287, 243–251. [Google Scholar] [CrossRef] [PubMed]
- Sreerama, N.; Woody, R.W. A self-consistent method for the analysis of protein secondary structure from circular dichroism. Anal. Biochem. 1993, 209, 32–44. [Google Scholar] [CrossRef] [PubMed]
- Amsterdam, D. The laboratory diagnosis of tuberculosis in a period of resurgence: Challenge for the laboratory. Clin. Lab. Sci. 1996, 9, 207–212. [Google Scholar] [PubMed]
- Steinberg, D.A.; Hurst, M.A.; Fujii, C.A.; Kung, A.H.; Ho, J.F.; Cheng, F.C.; Loury, D.J.; Fiddes, J.C. Protegrin-1: A broad-spectrum, rapidly microbicidal peptide with in vivo activity. Antimicrob. Agents Chemother. 1997, 41, 1738–1742. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.; Hancock, R.E. Improved derivatives of bactenecin, a cyclic dodecameric antimicrobial cationic peptide. Antimicrob. Agents Chemother. 1999, 43, 1274–1276. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.; Hancock, R.E. Interaction of the cyclic antimicrobial cationic peptide bactenecin with the outer and cytoplasmic membrane. J. Biol. Chem. 1999, 274, 29–35. [Google Scholar] [CrossRef] [PubMed]
- Siano, A.; Humpola, M.V.; de Oliveira, E.; Albericio, F.; Simonetta, A.C.; Lajmanovich, R.; Tonarelli, G.G. Antimicrobial peptides from skin secretions of Hypsiboas pulchellus (Anura: Hylidae). J. Nat. Prod. 2014, 77, 831–841. [Google Scholar] [CrossRef] [PubMed]
- Siano, A.; Tonarelli, G.; Imaz, M.S.; Perin, J.C.; Ruggeri, N.; Lopez, M.; Santi, M.N.; Zerbini, E. Bactericidal and hemolytic activities of synthetic peptides derived from granulysin. Protein Pept. Lett. 2010, 17, 517–521. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds P1-Ll-1577, P2-Ll-1298, and P3-Ll-2085 are available from the authors. |
| ID | Experimental MW | Amino Acid Sequence | Nr | Charge * | Secondary Structure Prediction | H | |
|---|---|---|---|---|---|---|---|
| PSIRED | GOR V | ||||||
| S1 | 1299.72 [M + H]+ | AAGKGLVSNLLEK | 13 | +1 | Helix (K4-E12) | Helix (L6-L10) | −0.05 |
| S2 | 1578.71 [M + H]+ | DEMKLDGFNMHLE | 13 | −3 | Coil | Coil | −0.18 |
| S3 | 673.382 [M + H]+ | GAMGKPL | 7 | +1 | - | - | 0.02 |
| S4 | 743.544 [M + H]+ | VVGDLLK | 7 | 0 | - | - | 0.06 |
| S5 | 801.147 [M + H]+ | DEEAKPI | 7 | −2 | - | - | −0.3 |
| ID | Amino Acid Sequence | Net Charge pH = 7 | Secondary Structure Prediction | Experimental MW (*) | Rel. Hydro AA/Total AA | H | |
|---|---|---|---|---|---|---|---|
| PSIPRED | GOR V | ||||||
| P1-Ll-1577 | DEMKLDGFNMHLE-NH2 | −2 | Coil | Coil | 1577.713 | 5/13 (38%) | −0.18 |
| P2-Ll-1298 | AAGKGLVSNLLEK-NH2 | +2 | Helix (K4-E12) | Helix (L6-L10) | 1298.75 | 6/13 (46%) | −0.05 |
| P3-Ll-2085 | GLLDFLKAAGKGLVSNLLEK-NH2 | +2 | Helix (L2-E19) | Helix (L3-A9, L13-L17) | 2085.205 | 10/20 (50%) | −0.01 |
| ID | MIC (µM) E. coli ATCC 25922 | MIC (µM) S. aureus ATCC 25923 |
|---|---|---|
| P1-Ll-1577 (*) | 20 | 40.5 |
| P2-Ll-1298 | 24.6 | 49 |
| P3-Ll-2085 | 15 | 15 |
| Analog Identification | MIC (μM) | MHC (μM) | Therapeutic Index (TI) | ||
|---|---|---|---|---|---|
| E. coli | S. aureus | E. coli | S. aureus | ||
| P1-Ll-1577 | 20 | 40.5 | 640 | 32 | 15.8 |
| P2-Ll-1298 | 24.6 | 49 | 320 | 13 | 6.5 |
| P3-Ll-2085 | 15 | 15 | 40 | 2.7 | 2.7 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Siano, A.; Humpola, M.V.; De Oliveira, E.; Albericio, F.; Simonetta, A.C.; Lajmanovich, R.; Tonarelli, G.G. Leptodactylus latrans Amphibian Skin Secretions as a Novel Source for the Isolation of Antibacterial Peptides. Molecules 2018, 23, 2943. https://doi.org/10.3390/molecules23112943
Siano A, Humpola MV, De Oliveira E, Albericio F, Simonetta AC, Lajmanovich R, Tonarelli GG. Leptodactylus latrans Amphibian Skin Secretions as a Novel Source for the Isolation of Antibacterial Peptides. Molecules. 2018; 23(11):2943. https://doi.org/10.3390/molecules23112943
Chicago/Turabian StyleSiano, Alvaro, Maria Veronica Humpola, Eliandre De Oliveira, Fernando Albericio, Arturo C. Simonetta, Rafael Lajmanovich, and Georgina G. Tonarelli. 2018. "Leptodactylus latrans Amphibian Skin Secretions as a Novel Source for the Isolation of Antibacterial Peptides" Molecules 23, no. 11: 2943. https://doi.org/10.3390/molecules23112943
APA StyleSiano, A., Humpola, M. V., De Oliveira, E., Albericio, F., Simonetta, A. C., Lajmanovich, R., & Tonarelli, G. G. (2018). Leptodactylus latrans Amphibian Skin Secretions as a Novel Source for the Isolation of Antibacterial Peptides. Molecules, 23(11), 2943. https://doi.org/10.3390/molecules23112943






