Inhibitory Effects of Ginsenoside Rb1 on Early Atherosclerosis in ApoE-/- Mice via Inhibition of Apoptosis and Enhancing Autophagy
Abstract
:1. Introduction
2. Materials and Methods
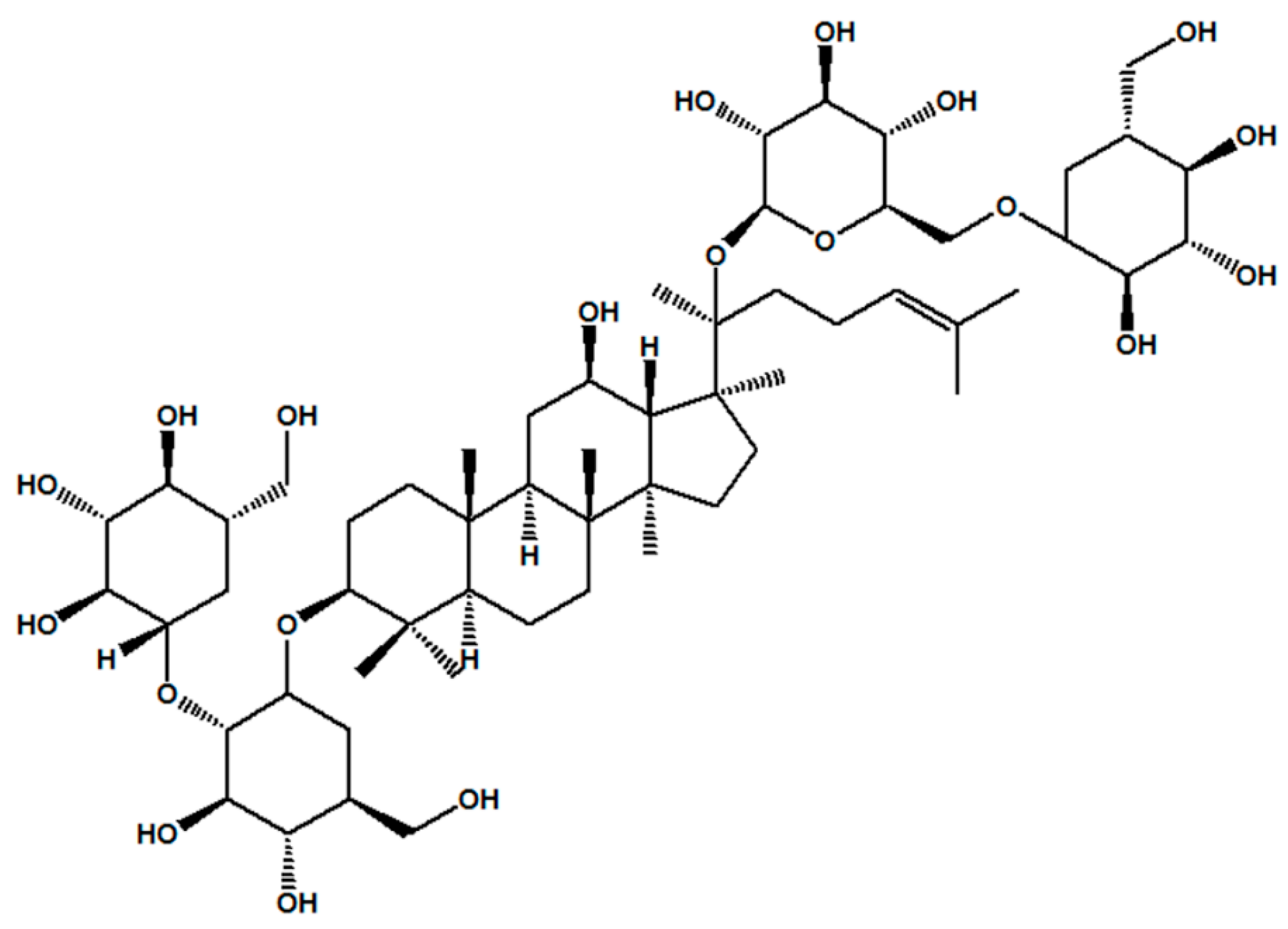
2.1. Reagents and Materials
2.2. Animals
2.3. Experimental Protocol
2.4. Serum Analysis of Lipids and Plasma Cytokines
2.5. Oil Red O Staining
2.6. Immunohistochemical Analysis
2.7. Detection of DNA Fragmentation
2.8. Transmission Electron Microscopy
2.9. Western Blot Analysis
2.10. Statistical Analysis
3. Results
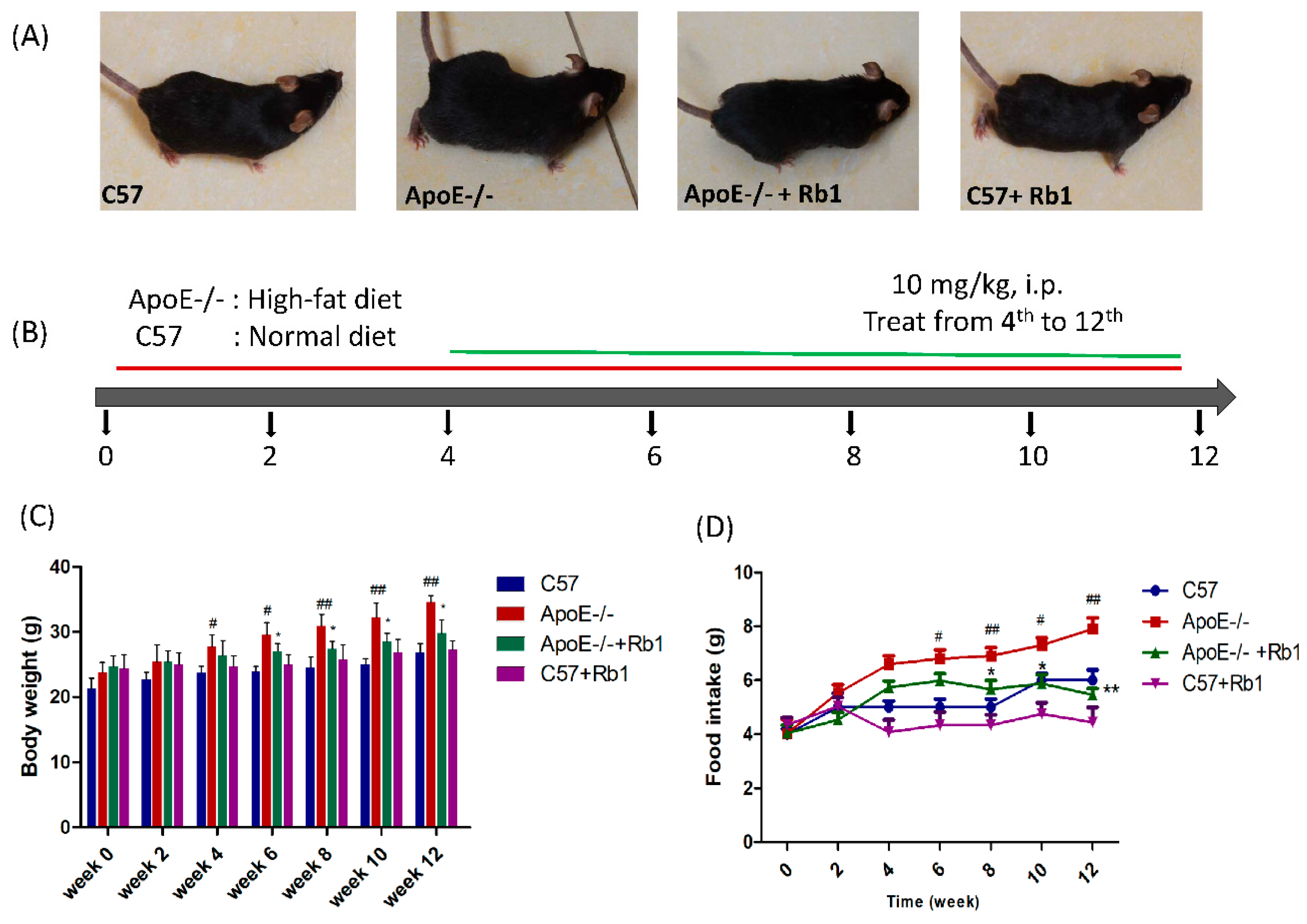
3.1. Ginsenoside Rb1 Decreased Body Weight and Food Intake in ApoE-/- Mice Fed a High-Fat Diet
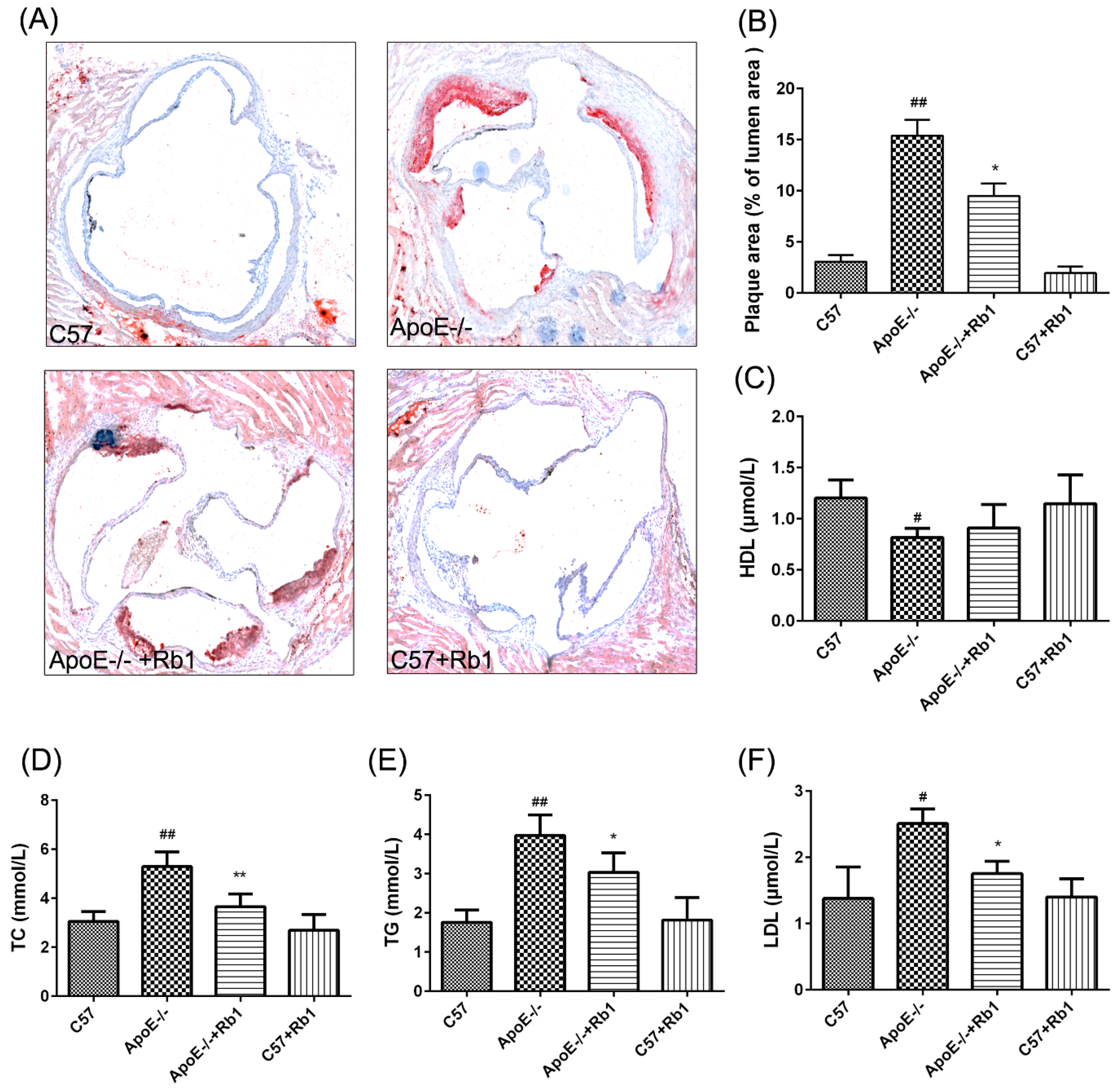
3.2. Ginsenoside Rb1 Reduced Atherosclerotic Development in ApoE-/- Mice
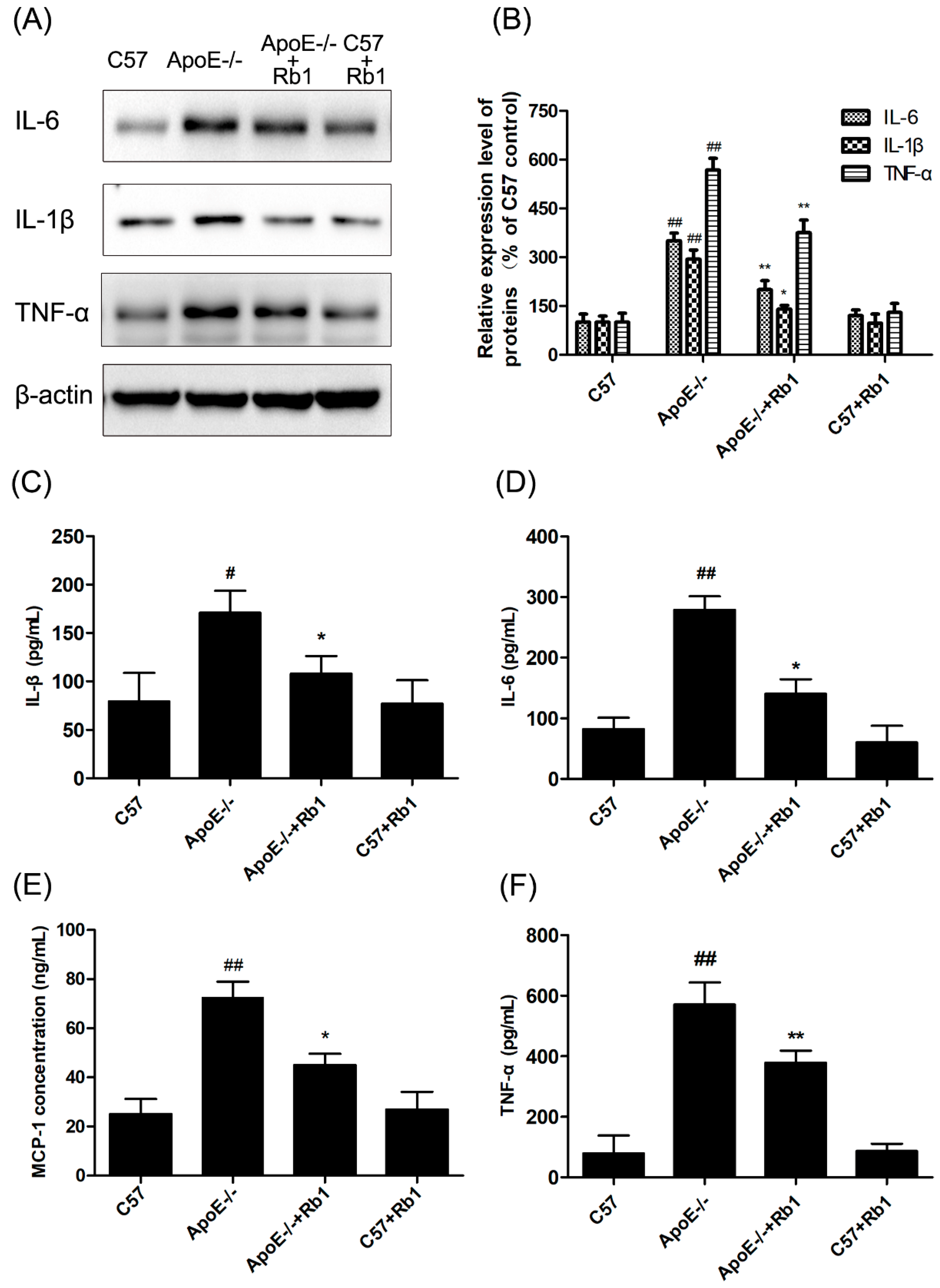
3.3. Ginsenoside Rb1 Suppressed Inflammatory Cytokines in the Serum of ApoE-/- Mice
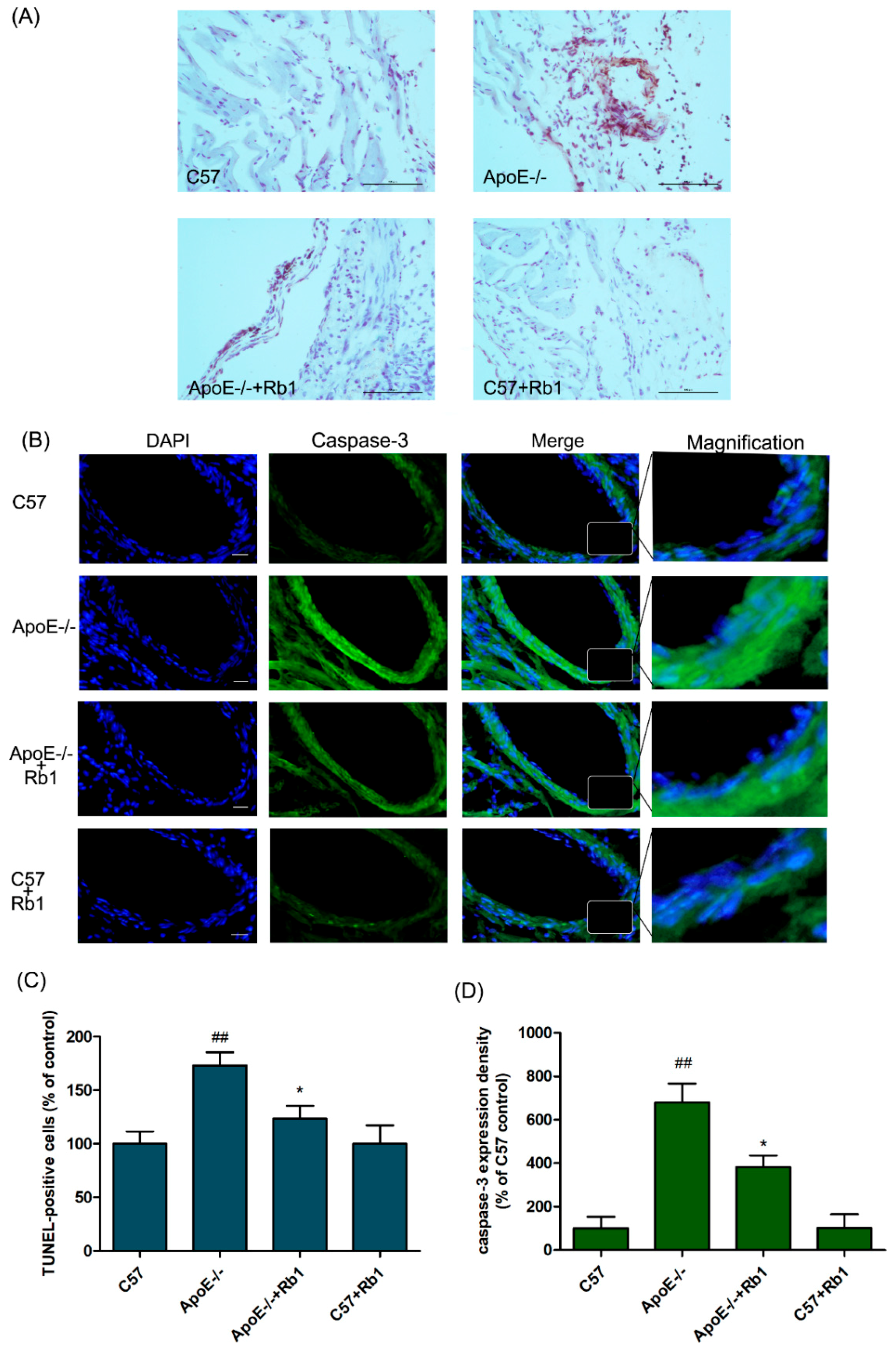
3.4. Ginsenoside Rb1 Attenuated the Percentage of Apoptotic Cells in the Aorta of ApoE-/- Mice
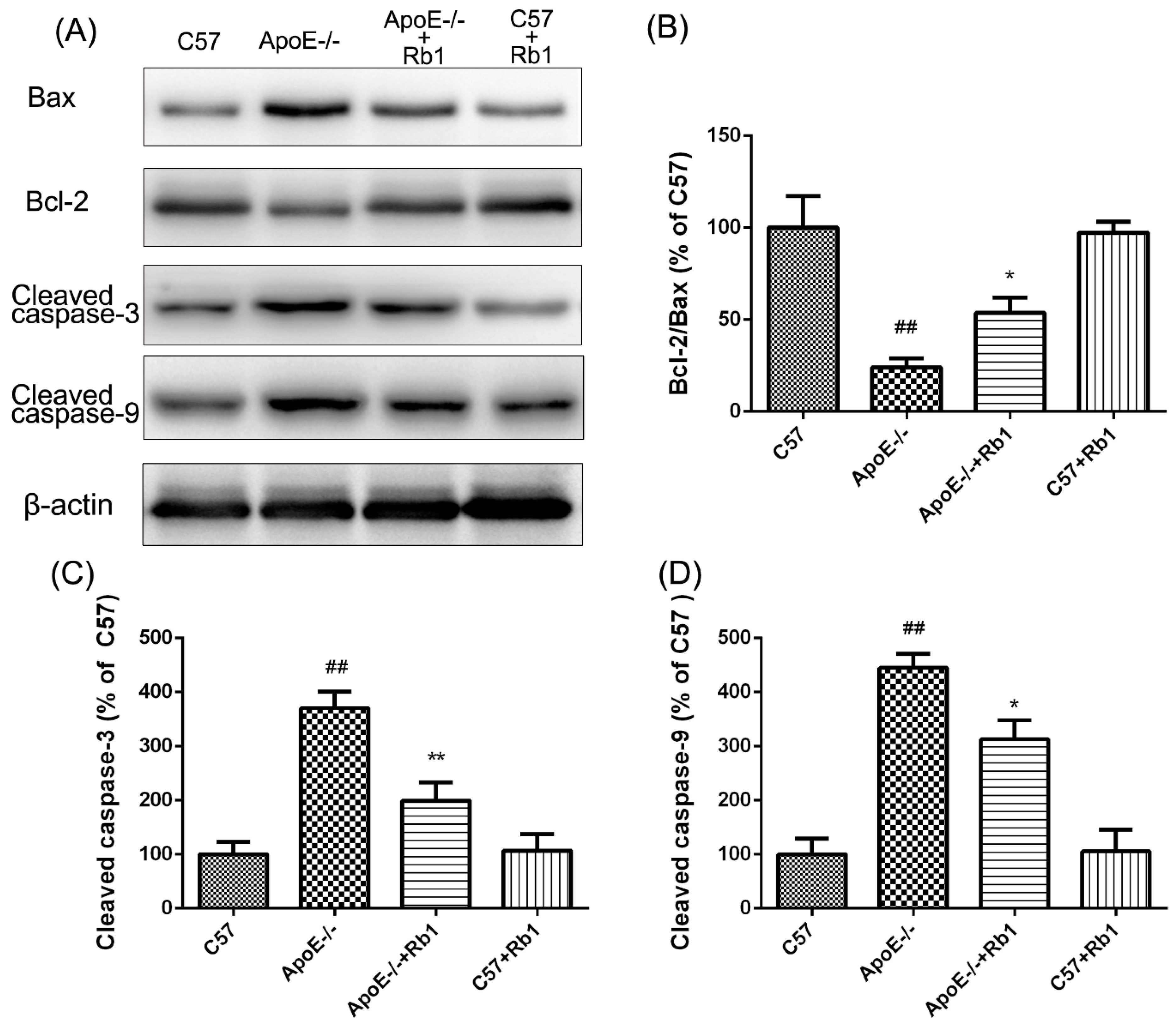
3.5. Ginsenoside Rb1 Regulated the Expression Levels of Apoptosis-Related Proteins in the Aorta of ApoE-/- Mice
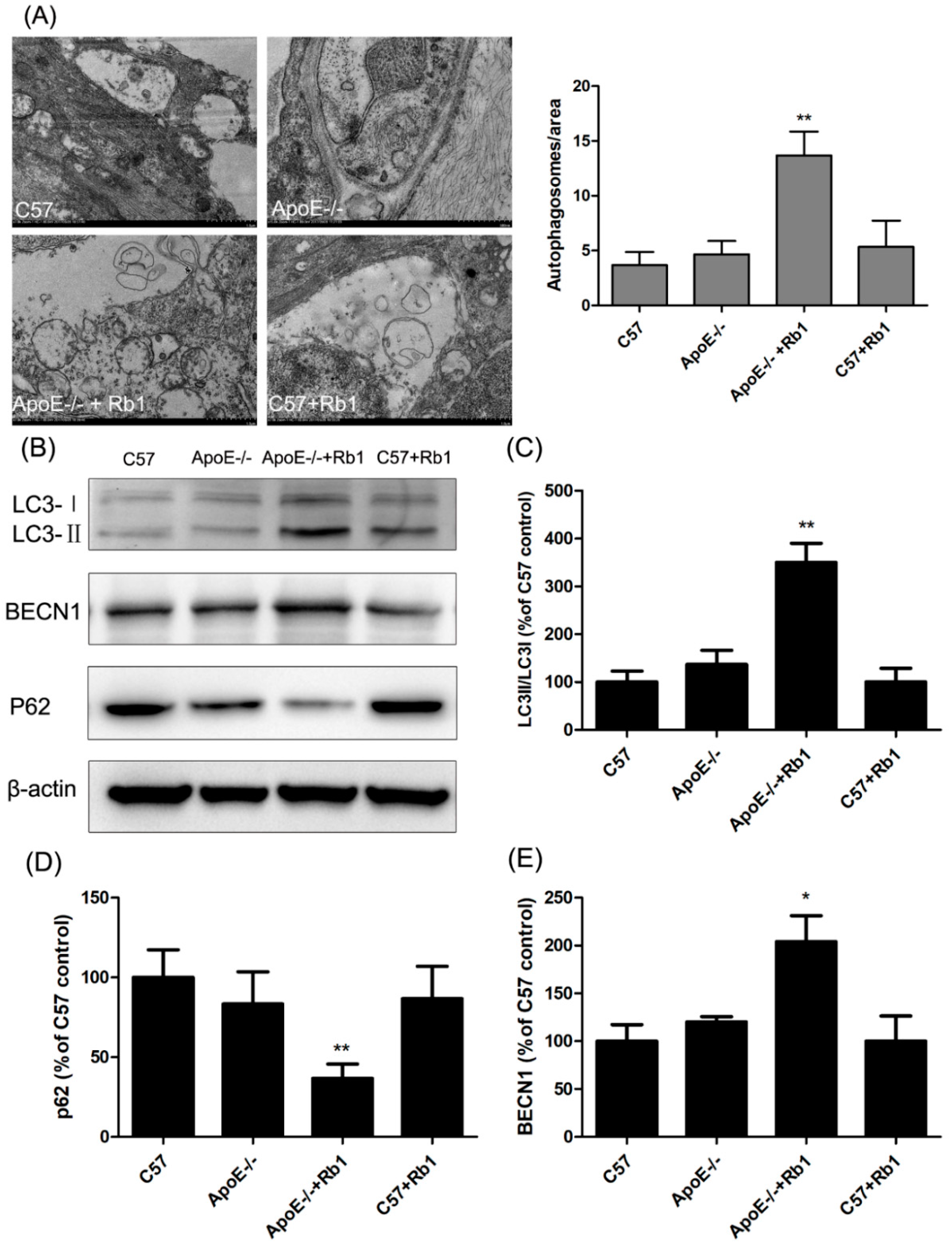
3.6. Ginsenoside Rb1 Administration Accelerated Autophagy in the Aorta of ApoE-/- Mice
4. Discussion
Author Contributions
Funding
Conflicts of Interest
References
- Lusis, A.J. Atherosclerosis. Nature 2000, 407, 233–241. [Google Scholar] [CrossRef] [PubMed]
- Hansson, G.K. Inflammation, atherosclerosis, and coronary artery disease. N. Engl. J. Med. 2005, 352, 1685–1695. [Google Scholar] [CrossRef] [PubMed]
- Haddy, N.; Sass, C.; Droesch, S.; Zaiou, M.; Siest, G.; Ponthieux, A.; Lambert, D.; Visvikis, S. IL-6, TNF-alpha and atherosclerosis risk indicators in a healthy family population: The STANISLAS cohort. Atherosclerosis 2003, 170, 277–283. [Google Scholar] [CrossRef]
- Kablakziembicka, A.; Przewlocki, T.; Sokołowski, A.; Tracz, W.; Podolec, P. Carotid intima-media thickness, hs-CRP and TNF-α are independently associated with cardiovascular event risk in patients with atherosclerotic occlusive disease. Atherosclerosis 2011, 214, 185–190. [Google Scholar] [CrossRef] [PubMed]
- Jia, Z.; Nallasamy, P.; Liu, D.; Shah, H.; Li, J.Z.; Chitrakar, R.; Si, H.; McCormick, J.; Zhu, H.; Zhen, W.; et al. Luteolin protects against vascular inflammation in mice and tnf-alpha-induced monocyte adhesion to endothelial cells via suppressing iκbα/nf-κb signaling pathway. J. Nutr. Biochem. 2015, 26, 293–302. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Tian, F.; Wang, J.; Jing, J.; Zhou, S.S.; Chen, Y.D. Atherosclerosis-Associated Endothelial Cell. Apoptosis by MiR-429-Mediated Down Regulation of Bcl-2. Cell. Physiol. Biochem. 2015, 37, 1421–1430. [Google Scholar] [PubMed]
- Wu, G.; Cai, J.; Han, Y.; Chen, J.; Huang, Z.P.; Chen, C.; Cai, Y.; Huang, H.; Yang, Y.; Liu, Y.; et al. LincRNA-p21 regulates neointima formation, vascular smooth muscle cell proliferation, apoptosis, and atherosclerosis by enhancing p53 activity. Circulation 2014, 130, 1452–1465. [Google Scholar] [CrossRef] [PubMed]
- Hamada, M.; Nakamura, M.; Tran, M.T.; Moriguchi, T.; Hong, C.; Ohsumi, T.; Dinh, T.T.; Kusakabe, M.; Hattori, M.; Katsumata, T.; et al. MafB promotes atherosclerosis by inhibiting foam-cell apoptosis. Nat. Commun. 2014, 5, 3147. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pedro, J.M.B.-S.; Wei, Y.; Sica, V.; Maiuri, M.C.; Zou, Z.; Kroemer, G.; Levine, B. BAX and BAK1 are dispensable for ABT-737-induced dissociation of the BCL2-BECN1 complex and autophagy. Autophagy 2015, 11, 452–459. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huber, T.B.; Edelstein, C.L.; Hartleben, B.; Inoki, K.; Jiang, M.; Koya, D.; Kume, S.; Lieberthal, W.; Pallet, N.; Quiroga, A.; et al. Emerging role of autophagy in kidney function, diseases and aging. Autophagy 2012, 8, 1009–1031. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xiong, Y.; Yepuri, G.; Forbiteh, M.; Yu, Y.; Montani, J.-P.; Yang, Z.; Ming, X.-F. ARG2 impairs endothelial autophagy through regulation of MTOR and PRKAA/AMPK signaling in advanced atherosclerosis. Autophagy 2014, 10, 2223–2238. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guo, F.-X.; Hu, Y.-W.; Zheng, L.; Wang, Q. Shear Stress in Autophagy and Its Possible Mechanisms in the Process of Atherosclerosis. DNA Cell Biol. 2017, 36, 335–346. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Evans, T.D.; Jeong, S.J.; Zhang, X.; Sergin, I.; Razani, B. TFEB and trehalose drive the macrophage autophagy-lysosome system to protect against atherosclerosis. Autophagy 2018, 14, 724–726. [Google Scholar] [CrossRef] [PubMed]
- Sergin, I.; Evans, T.D.; Zhang, X.; Bhattacharya, S.; Stokes, C.J.; Song, E.; Ali, S.; Dehestani, B.; Holloway, K.B.; Micevych, P.S.; et al. Exploiting macrophage autophagy-lysosomal biogenesis as a therapy for atherosclerosis. Nat. Commun. 2017, 8, 15750. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, Y.; Li, Y.; Yang, W.; Gao, S.; Lin, J.; Wang, T.; Zhou, K.; Hu, H. Ginsenoside Rb1 inhibit apoptosis in rat model of Alzheimer’s disease induced by Abeta1-40. Am. J. Transl. Res. 2018, 10, 796–805. [Google Scholar]
- Zhao, D.; Zhang, M.; Yuan, H.; Meng, C.; Zhang, B.; Wu, H. Ginsenoside Rb1 protects against spinal cord ischemia-reperfusion injury in rats by downregulating the Bax/Bcl-2 ratio and caspase-3 and p-Ask-1 levels. Exp. Mol. Pathol. 2018, 105, 229–235. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Liu, W.; Lu, Y.; Tian, H.; Duan, C.; Lu, L.; Gao, G.; Wu, X.; Wang, X.; Yang, H. Piperlongumine restores the balance of autophagy and apoptosis by increasing BCL2 phosphorylation in rotenone-induced Parkinson disease models. Autophagy 2018, 14, 845–861. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Y.; Shen, L.; Zhang, J. Anti-amnestic and anti-aging effects of ginsenoside Rg1 and Rb1 and its mechanism of action. Acta Pharmacol. Sin. 2005, 26, 143–149. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ai, Q.; Sun, G.; Luo, Y.; Dong, X.; Hu, R.; Meng, X.; Sun, X. Ginsenoside Rb1 prevents hypoxia-reoxygenation-induced apoptosis in H9c2 cardiomyocytes via an estrogen receptor-dependent crosstalk among the Akt, JNK, and ERK 1/2 pathways using a label-free quantitative proteomics analysis. RSC Adv. 2015, 5, 26346–26363. [Google Scholar] [CrossRef]
- Li, G.; Qian, W.; Zhao, C. Analyzing the anti-ischemia-reperfusion injury effects of ginsenoside Rb1 mediated through the inhibition of p38α MAPK. Can. J. Physiol. Pharmacol. 2016, 94, 97–103. [Google Scholar] [CrossRef] [PubMed]
- Xiong, Y.; Shen, L.K.; Tso, P.; Xiong, Y.; Wang, G.; Woods, S.C.; Liu, M. Antiobesity and antihyperglycemic effects of ginsenoside Rb1 in rats. Diabetes 2010, 59, 2505–2512. [Google Scholar] [CrossRef] [PubMed]
- Shang, W.; Yang, Y.; Jiang, B.; Jin, H.; Zhou, L.; Liu, S.; Chen, M. Ginsenoside Rb1 promotes adipogenesis in 3T3-L1 cells by enhancing PPARgamma2 and C/EBPalpha gene expression. Life Sci. 2007, 80, 618–625. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.J.; Jin, Y.R.; Lim, W.C.; Park, W.K.; Cho, J.Y.; Jang, S.; Lee, S.K. Ginsenoside-Rb1 acts as a weak phytoestrogen in MCF-7 human breast cancer cells. Arch. Pharm. Res. 2003, 26, 58–63. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.U.; Bae, E.A.; Han, M.J.; Kim, N.J.; Kim, D.H. Hepatoprotective effect of ginsenoside Rb1 and compound K on tert-butyl hydroperoxide-induced liver injury. Liver Int. Off. J. Int. Assoc. Study Liver 2005, 25, 1069–1073. [Google Scholar] [CrossRef] [PubMed]
- Hwang, Y.P.; Jeong, H.G. Ginsenoside Rb1 protects against 6-hydroxydopamine-induced oxidative stress by increasing heme oxygenase-1 expression through an estrogen receptor-related PI3K/Akt/Nrf2-dependent pathway in human dopaminergic cells. Toxicol. Appl. Pharmacol. 2010, 242, 18–28. [Google Scholar] [CrossRef] [PubMed]
- Zhou, P.; Lu, S.; Luo, Y.; Wang, S.; Yang, K.; Zhai, Y.; Sun, G.; Sun, X. Attenuation of TNF-α-Induced Inflammatory Injury in Endothelial Cells by Ginsenoside Rb1 via Inhibiting NF-κB, JNK and p38 Signaling Pathways. Front. Pharmacol. 2017, 8, 464. [Google Scholar] [CrossRef] [PubMed]
- Figg, N.L.; Bennett, M.R. Quantification of Apoptosis in Mouse Atherosclerotic Lesions. Methods Mol. Biol. 2015, 1339, 191–199. [Google Scholar] [PubMed]
- Zhao, J.; Ou, S.-L.; Wang, W.-Y.; Yan, C.; Chi, L.-X. MicroRNA-1907 enhances atherosclerosis-associated endothelial cell apoptosis by suppressing Bcl-2. AM. J. Transl. Res. 2017, 9, 3433–3442. [Google Scholar] [PubMed]
- Luo, Y.; Meng, X.; Zhou, P.; Lu, S.; Qin, M.; Xu, X.; Sun, G.; Sun, X. Elatoside C protects against ox-LDL-induced HUVECs injury by FoxO1-mediated autophagy induction. Biochim. Biophys. Acta 2017, 1863, 1654–1665. [Google Scholar] [CrossRef] [PubMed]
- Qiao, L.; Zhang, X.; Liu, M.; Liu, X.; Dong, M.; Cheng, J.; Zhang, X.; Zhai, C.; Song, Y.; Lu, H.; et al. Ginsenoside Rb1 Enhances Atherosclerotic Plaque Stability by Improving Autophagy and Lipid Metabolism in Macrophage Foam Cells. Front. Pharmacol. 2017, 8, 727. [Google Scholar] [CrossRef] [PubMed]
- Hu, S.Y. The Genus Panax (Ginseng) in Chinese Medicine. Econ. Bot. 1976, 30, 11–28. [Google Scholar] [CrossRef]
- Ng, T.B. Pharmacological activity of sanchi ginseng (Panax notoginseng). J. Pharm. Pharmacol. 2010, 58, 1007–1019. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Zhao, Y.; Rayburn, E.R.; Hill, D.L.; Wang, H.; Zhang, R. In vitro anti-cancer activity and structure–activity relationships of natural products isolated from fruits of Panax ginseng. Cancer Chemother. Pharmacol. 2007, 59, 589–601. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.A.; Kim, S.; Chang, S.H.; Hwang, H.J.; Choi, Y.N. Anti-arthritic effect of ginsenoside Rb1 on collagen induced arthritis in mice. Int. Immunopharmacol. 2007, 7, 1286–1291. [Google Scholar] [CrossRef] [PubMed]
- Sang, H.L.; Jung, B.H.; Sun, Y.K.; Lee, E.H.; Chung, B.C. The antistress effect of ginseng total saponin and ginsenoside Rg3 and Rb1 evaluated by brain polyamine level under immobilization stress. Pharmacol. Res. 2006, 54, 46–49. [Google Scholar]
- Li, X.; Zhu, F.; Jiang, J.; Sun, C.; Zhong, Q.; Shen, M.; Wang, X.; Tian, R.; Shi, C.; Xu, M.; et al. Simultaneous inhibition of the ubiquitin-proteasome system and autophagy enhances apoptosis induced by ER stress aggravators in human pancreatic cancer cells. Autophagy 2016, 12, 1521–1537. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liao, X.; Sluimer, J.C.; Wang, Y.; Subramanian, M.; Brown, K.; Pattison, J.S.; Robbins, J.; Martinez, J.; Tabas, I. Macrophage Autophagy Plays a Protective Role in Advanced Atherosclerosis. Cell Metab. 2012, 15, 545–553. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, X.; Tang, Y.; Cui, Y.; Zhang, H.; Zhang, D. Autophagy is associated with cell fate in the process of macrophage-derived foam cells formation and progress. J. Biomed. Sci. 2016, 23, 57. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are not available from the authors. |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhou, P.; Xie, W.; Luo, Y.; Lu, S.; Dai, Z.; Wang, R.; Zhang, X.; Li, G.; Sun, G.; Sun, X. Inhibitory Effects of Ginsenoside Rb1 on Early Atherosclerosis in ApoE-/- Mice via Inhibition of Apoptosis and Enhancing Autophagy. Molecules 2018, 23, 2912. https://doi.org/10.3390/molecules23112912
Zhou P, Xie W, Luo Y, Lu S, Dai Z, Wang R, Zhang X, Li G, Sun G, Sun X. Inhibitory Effects of Ginsenoside Rb1 on Early Atherosclerosis in ApoE-/- Mice via Inhibition of Apoptosis and Enhancing Autophagy. Molecules. 2018; 23(11):2912. https://doi.org/10.3390/molecules23112912
Chicago/Turabian StyleZhou, Ping, Weijie Xie, Yun Luo, Shan Lu, Ziru Dai, Ruiying Wang, Xuelian Zhang, Guang Li, Guibo Sun, and Xiaobo Sun. 2018. "Inhibitory Effects of Ginsenoside Rb1 on Early Atherosclerosis in ApoE-/- Mice via Inhibition of Apoptosis and Enhancing Autophagy" Molecules 23, no. 11: 2912. https://doi.org/10.3390/molecules23112912
APA StyleZhou, P., Xie, W., Luo, Y., Lu, S., Dai, Z., Wang, R., Zhang, X., Li, G., Sun, G., & Sun, X. (2018). Inhibitory Effects of Ginsenoside Rb1 on Early Atherosclerosis in ApoE-/- Mice via Inhibition of Apoptosis and Enhancing Autophagy. Molecules, 23(11), 2912. https://doi.org/10.3390/molecules23112912





