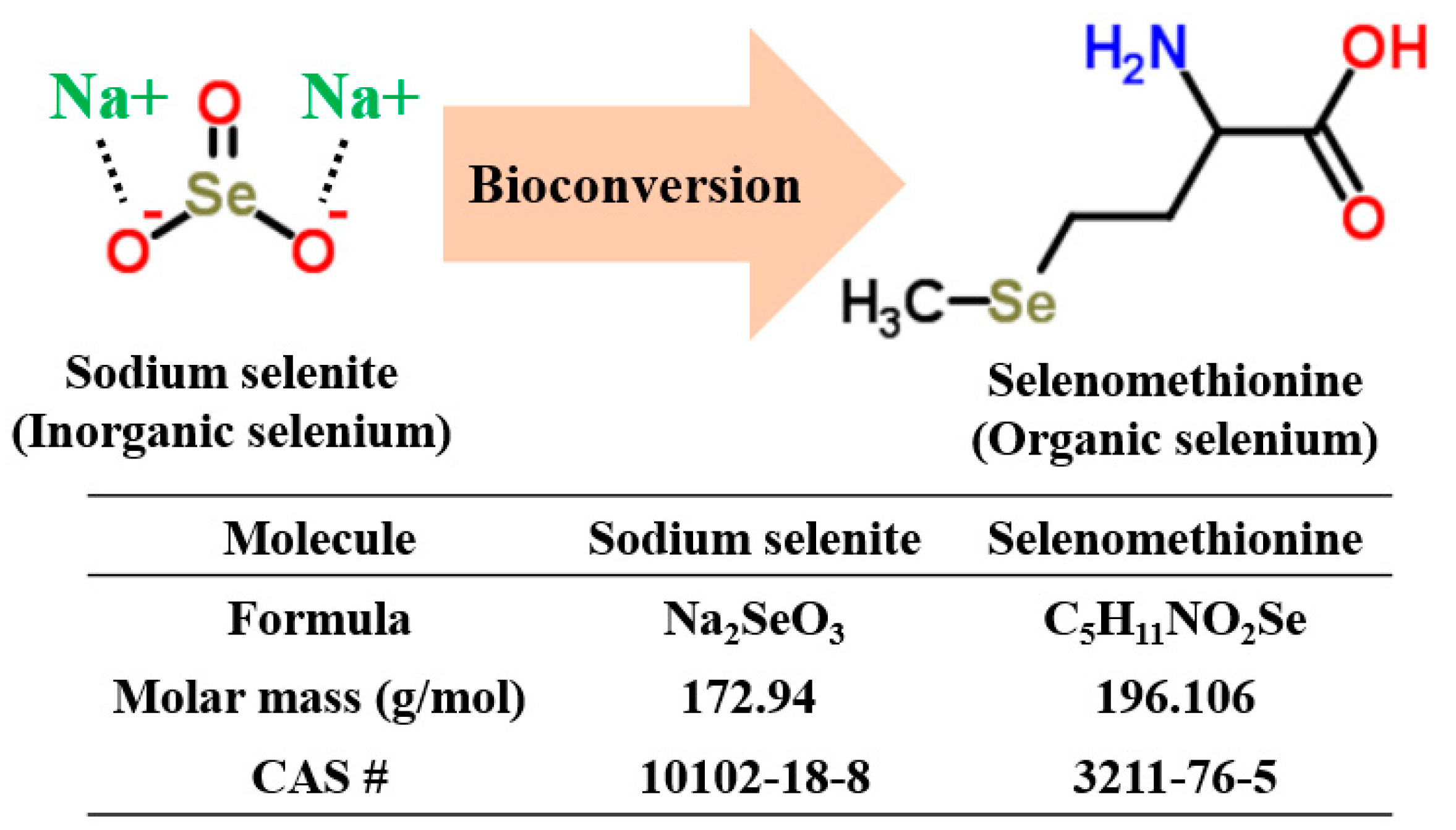
Production of Selenomethionine-Enriched Bifidobacterium bifidum BGN4 via Sodium Selenite Biocatalysis
Abstract
1. Introduction
2. Results and Discussions
2.1. Subsection Cell Screening and the Effect of the Cultivation Conditions on the Bacterial Cell Mass
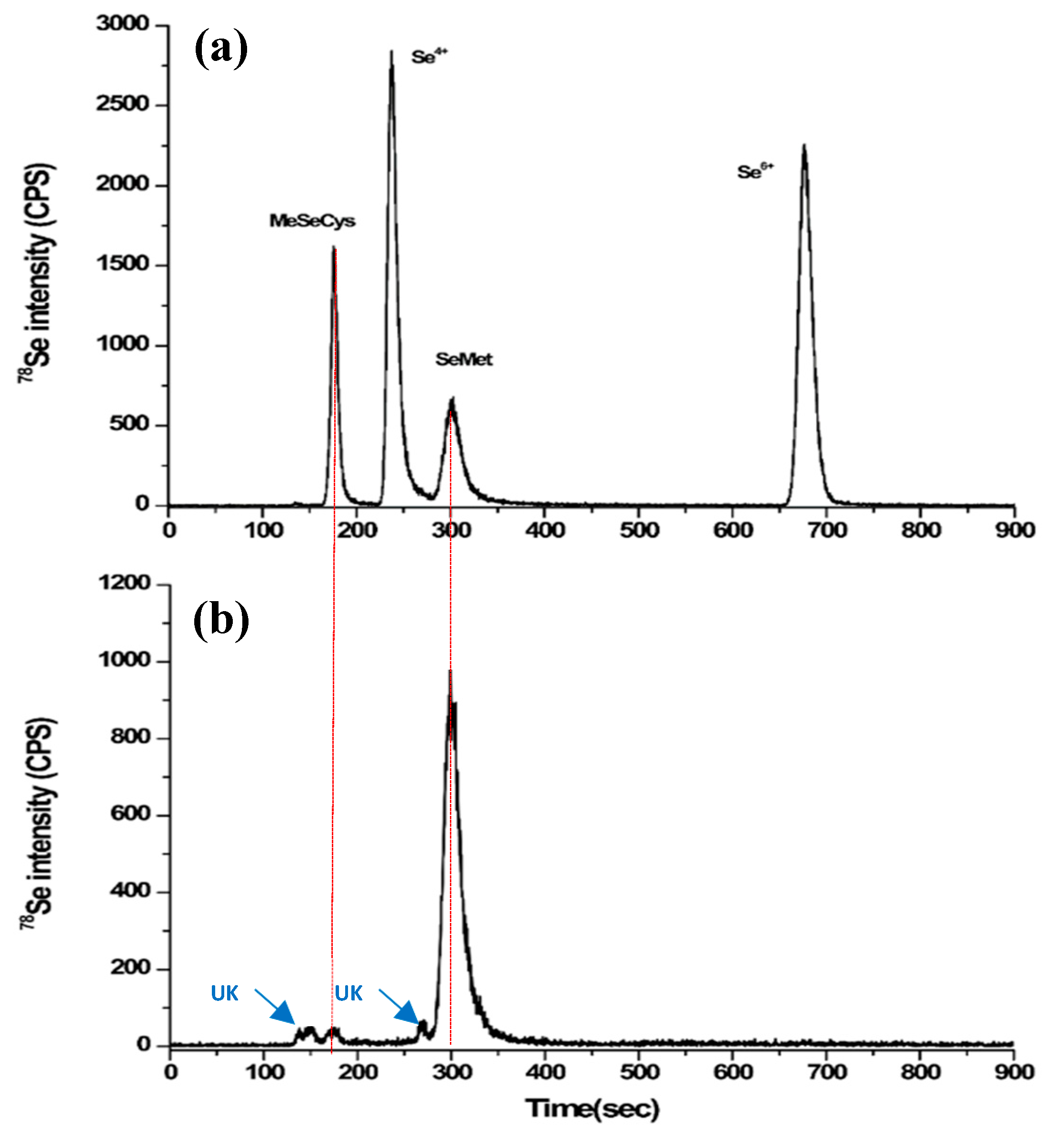
2.2. Quantification and Qualification of Organic Selenium in Microorganisms
3. Materials and Methods
3.1. Microbial Culture Condition and Initial Cell Screening
3.2. Selection of Selenium-Enriched Probiotics
3.3. Quantification of Total Selenium, Organic Selenium, and Selenium Species
3.4. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Stranges, S.; Marshall, J.R.; Trevisan, M.; Natarajan, R.; Donahue, R.; Combs, G.F.; Farinaro, E.; Clark, L.C.; Reid, M.E. Effects of Selenium Supplementation on Cardiovascular Disease Incidence and Mortality: Secondary Analyses in A Randomized Clinical Trial. Am. J. Epidemiol. 2006, 163, 694–699. [Google Scholar] [CrossRef] [PubMed]
- Finley, J.; Ip, C.; Lisk, D.; Davis, C.; Hintze, K.; Whanger, P. Cancer-Protective Properties of High-Selenium Broccoli. J. Agric. Food Chem. 2001, 49, 2679–2683. [Google Scholar] [CrossRef] [PubMed]
- Rayman, M. The Importance of Selenium to Human Health. Lancet 2000, 356, 233–241. [Google Scholar] [CrossRef]
- Chen, X. Selenium and Cardiomyopathy (Keshan Disease). Acta Pharmacol. Toxicol. 2009, 59, 325–330. [Google Scholar] [CrossRef]
- Moreno-Reyes, R.; Suetens, C.; Mathieu, F.; Begaux, F.; Zhu, D.; Rivera, M.; Boelaert, M.; Nève, J.; Perlmutter, N.; Vanderpas, J. Kashin-Beck Osteoarthropathy In Rural Tibet In Relation to Selenium and Iodine Status. N. Engl. J. Med. 1998, 339, 1112–1120. [Google Scholar] [CrossRef] [PubMed]
- Wei, J.; Zeng, C.; Gong, Q.; Yang, H.; Li, X.; Lei, G.; Yang, T. The Association Between Dietary Selenium Intake and Diabetes: A Cross-Sectional Study Among Middle-Aged and Older Adults. Nutr. J. 2015, 14, 18. [Google Scholar] [CrossRef] [PubMed]
- Monsen, E.R. Dietary Reference Intakes for Vitamin C, Vitamin E, Selenium, and Carotenoids. J. Am. Diet. Assoc. 2000, 100, 637–640. [Google Scholar] [CrossRef]
- Aquilina, G.; Azimonti, G.; Bampidis, V.; de Lourdes Bastos, M.; Bories, G.; Chesson, A.; Cocconcelli, P.S.; Flachowsky, G.; Gropp, J.; Kolar, B.; et al. Safety and Efficacy of Selenium Compounds (E8) As Feed Additives for All Animal Species: Sodium Selenite, Based on A Dossier Submitted by Retorte Gmbh Selenium Chemicals and Metals. EFSA J. 2016, 14, 4398. [Google Scholar]
- Brown, K.; Pickard, K.; Nicol, F.; Beckett, G.; Duthie, G.; Arthur, J. Effects of Organic and Inorganic Selenium Supplementation on Selenoenzyme Activity in Blood Lymphoctyes, Granulocytes, Platelets and Erythrocytes. Clin. Sci. 2000, 98, 593–599. [Google Scholar] [CrossRef] [PubMed]
- Mahima, A.; Kumar, A.; Amit, K.; Anu, R.; Vinod, K.; Debashis, R. Inorganic Versus Organic Selenium Supplementation: A Review. Pak. J. Biol. Sci. 2012, 15, 418–425. [Google Scholar] [PubMed]
- Probiotics Market Size, Share & Trends Analysis Report by Application (Food & Beverages, Dietary Supplements, Animal Feed), By End-Use, By Region, And Segment Forecast, 2018–2024. Available online: http://www.grandviewresearch.com/industry-analysis/probiotics-market (accessed on 14 September 2018).
- Global Probiotics Market 2017–2021. Available online: https://www.researchandmarkets.com/reports/4231709/global-probiotics-market-2017-2021?gclid=Cj0KCQjw6NjNBRDKARIsAFn3NMrYxxMExFwUMz0FGGYeE31PNIu8sAgcXKxML_4qJqcNm94I9a4Gae4aApILEALw_wcB (accessed on 14 September 2018).
- Ku, S.; Park, M.S.; Ji, G.E.; You, H.J. Review on Bifidobacterium bifidum BGN4: Functionality and Nutraceutical Applications as a Probiotic Microorganism. Int. J. Mol. Sci. 2016, 17, 1544. [Google Scholar] [CrossRef] [PubMed]
- Park, M.S.; Kwon, B.; Ku, S.; Ji, G.E. The Efficacy of Bifidobacterium longum BORI and Lactobacillus acidophilus AD031 Probiotic Treatment in Infants with Rotavirus Infection. Nutrients 2017, 9, 887. [Google Scholar] [CrossRef] [PubMed]
- Ku, S.; You, H.J.; Ji, G.E. Enhancement of Anti-Tumorigenic Polysaccharide Production, Adhesion, and Branch Formation of Bifidobacterium bifidum BGN4 by Phytic Acid. Food Sci. Biotechnol. 2009, 18, 749–754. [Google Scholar]
- Pavli, F.; Tassou, C.; Nychas, G.; Chorianopoulos, N. Probiotic Incorporation in Edible Films and Coatings: Bioactive Solution for Functional Foods. Int. J. Mol. Sci. 2018, 19, 150. [Google Scholar] [CrossRef] [PubMed]
- Hariharan, H.; Al-Dhabi, N.A.; Karuppiah, P.; Rajaram, S. Microbial synthesis of selinium nanocomposite using Saccharomyces cerevisiae and its antimicrobial activity against pathogens causing nosocomial infection. Chalcogenide Lett. 2012, 9, 509–515. [Google Scholar]
- Zhang, L.; Li, D.; Gao, P. Expulsion of Selenium/Protein Nanoparticles Through Vesicle-Like Structures by Saccharomyces Cerevisiae Under Microaerophilic Environment. World J. Microbiol. Biotechnol. 2012, 28, 3381–3386. [Google Scholar] [CrossRef] [PubMed]
- Nunes, R.; Luz, J.; Freitas, R.; Higuchi, A.; Kasuya, M.; Vanetti, M. Selenium Bioaccumulation in Shiitake Mushrooms: A Nutritional Alternative Source of This Element. J. Food Sci. 2012, 77, C983–C986. [Google Scholar] [CrossRef] [PubMed]
- Deng, Y.; Man, C.; Fan, Y.; Wang, Z.; Li, L.; Ren, H.; Cheng, W.; Jiang, Y. Preparation of Elemental Selenium-Enriched Fermented Milk by Newly Isolated Lactobacillus Brevis from Kefir Grains. Int. Dairy J. 2015, 44, 31–36. [Google Scholar] [CrossRef]
- Xia, S.; Chen, L.; Liang, J. Enriched Selenium and Its Effects on Growth and Biochemical Composition In lactobacillus Bulgaricus. J. Agric. Food Chem. 2007, 55, 2413–2417. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Zhou, K.; Zhang, J.; Chen, Q.; Liu, G.; Shang, N.; Qin, W.; Li, P.; Lin, F. Accumulation and Species Distribution of Selenium in Se-Enriched Bacterial Cells of The Bifidobacterium Animalis 01. Food Chem. 2009, 115, 727–734. [Google Scholar] [CrossRef]
- Ku, S.; You, H.J.; Park, M.S.; Ji, G.E. Whole-cell biocatalysis for producing ginsenoside Rd from Rb1 using Lactobacillus rhamnosus GG. J. Microbiol. Biotechnol. 2016, 26, 1206–1215. [Google Scholar] [CrossRef] [PubMed]
- Ku, S.; Zheng, H.; Park, M.S.; Ji, G.E. Optimization of β-glucuronidase activity from Lactobacillus delbrueckii Rh2 and and its use for biotransformation of baicalin and wogonoside. J. Korean Soc. Appl. Biol. Chem. 2011, 54, 275–280. [Google Scholar] [CrossRef]
- Li, Y.; Ku, S.; Park, M.S.; Li, Z.; Ji, G.E. Acceleration of Aglycone Isoflavone and -Aminobutyric Acid Production from Doenjang Using Whole-Cell Biocatalysis Accompanied by Protease Treatment. J. Microbiol. Biotechnol. 2017, 27, 1952–1960. [Google Scholar] [CrossRef] [PubMed]
- Ku, S.; You, H.J.; Park, M.S.; Ji, G.E. Effects of ascorbic acid on α-l-arabinofuranosidase and α-l-arabinopyranosidase activities from Bifidobacterium longum RD47 and its application to whole cell bioconversion of ginsenoside. J. Korean Soc. Appl. Biol. Chem. 2015, 58, 857–865. [Google Scholar] [CrossRef] [PubMed]
- Mrvčić, J.; Stanzer, D.; Šolić, E.; Stehlik-Tomas, V. Interaction of Lactic Acid Bacteria with Metal Ions: Opportunities for Improving Food Safety and Quality. World J. Microbiol. Biotechnol. 2012, 28, 2771–2782. [Google Scholar] [CrossRef] [PubMed]
- Mazo, V.; Gmoshinski, I.; Zorin, S. New Food Sources of Essential Trace Elements Produced by Biotechnology Facilities. Biotechnol. J. 2007, 2, 1297–1305. [Google Scholar] [CrossRef] [PubMed]
- Ito, T.; Kikuta, H.; Nagamori, E.; Honda, H.; Ogino, H.; Ishikawa, H.; Kobayashi, T. Lipase Production in Two-Step Fed-Batch Culture of Organic Solvent-Tolerant Pseudomonas Aeruginosa LST-03. J. Biosci. Bioeng. 2001, 91, 245–250. [Google Scholar] [CrossRef]
- Wang, T.; Tian, X.; Liu, T.; Wang, Z.; Guan, W.; Guo, M.; Chu, J.; Zhuang, Y. A Two-Stage Fed-Batch Heterotrophic Culture of Chlorella Protothecoides That Combined Nitrogen Depletion with Hyperosmotic Stress Strategy Enhanced Lipid Yield and Productivity. Process Biochem. 2017, 60, 74–83. [Google Scholar] [CrossRef]
- Li, X.; Liu, Y.; Wu, J.; Liang, H.; Qu, S. Microcalorimetric Study of Staphylococcus Aureus Growth Affected by Selenium Compounds. Thermochim. Acta 2002, 387, 57–61. [Google Scholar]
- Tran, P.A.; Webster, T.J. Selenium Nanoparticles Inhibit Staphylococcus Aureus Growth. Int. J. Nanomed. 2011, 6, 1553–1558. [Google Scholar]
- Diowksz, A.; Ambroziak, W.; Wladarezyk, M. Investigation of ability of selenium accumulation by lactic acid bacteria of Lactobacillus sp. and yeast S. cerevisiae. Pol. J. Food Nutr. Sci. 1999, 49, 17–21. [Google Scholar]
- Andreoni, V.; Luischi, M.M.; Cavalca, L.; Erba, D.; Ciappellano, S. Selenite tolerance and accumulation in the L. species. Ann. Microbiol. 2000, 50, 77–88. [Google Scholar]
- Siwek, M.; Galunsky, B.; Niemeyer, B. Isolation of selenium organic species from antarctic krill after enzymatic hydrolysis. Anal. Bioanal. Chem. 2005, 381, 737–741. [Google Scholar] [CrossRef] [PubMed]
- Reyes, L.H.; Guzman Mar, J.L.; Mizanur Rahman, G.M.; Seybert, B.; Fahrenholz, T.; Kingston, H.S. Simultaneous determination of arsenic and selenium species in fish tissues using microwave-assisted enzymatic extraction and ion chromatography–inductively coupled plasma mass spectrometry. Talanta 2009, 78, 983–990. [Google Scholar] [CrossRef] [PubMed]
- Yin, Y.; Wang, R.R.; Wang, Y.; Wang, J.J.; Xu, G.X. Preparation of Selenium-enriched Bifidobacterium Longum and its Effect on Tumor Growth and Immune Function of Tumor-Bearing Mice. Asian Pac. J. Cancer Prev. 2014, 15, 3681–3686. [Google Scholar] [CrossRef] [PubMed]
- Alzate, A.; Cañas, B.; Pérez-Munguía, S.; Hernández-Mendoza, H.; Pérez-Conde, C.; Gutiérrez, A.; Cámara, C. Evaluation of the Inorganic Selenium Biotransformation in Selenium-Enriched Yogurt by HPLC-ICP-MS. J. Agric. Food Chem. 2007, 55, 9776–9783. [Google Scholar] [CrossRef] [PubMed]
- Penas, E.; Martinez-Villaluenga, C.; Frias, J.; Sanchez-Martinez, M.J.; Perez-Corona, M.T.; Madrid, Y.; Camara, C.; Vidal-Valverde, C. Se improves indole glucosinolate hydrolysis products content, Se-methylselenocysteine content, antioxidant capacity and potential anti-inflammatory properties of sauerkraut. Food Chem. 2012, 132, 907–914. [Google Scholar] [CrossRef]
- Bryszewska, M.A.; Ambroziak, W.; Diowksz, A.; Baxter, M.J.; Langford, N.J.; Lewis, D.J. Changes in the chemical form of selenium observed during the manufacture of a selenium-enriched sourdough bread for use in a human nutrition study. Food. Addit. Contam. 2005, 22, 135–140. [Google Scholar] [CrossRef] [PubMed]
- Bryszewska, M.A.; Ambroziak, W.; Langford, N.J.; Baxter, M.J.; Colyer, A.; Lewis, D.J. The effect of consumption of selenium enriched rye/wheat sourdough bread on the body’s selenium status. Plant Foods Hum. Nutr. 2007, 62, 121–126. [Google Scholar] [CrossRef] [PubMed]
- Chinlu, W.; Richard, T.L. Organic selenium sources, selenomethionine and selenoyeast, have higher bioavailability than an inorganic selenium source, sodium selenite, in diets for channel catfish (Ictalurus punctatus). Aquaculture 1997, 152, 223–234. [Google Scholar]
- Claire, R.; Julie, A.S.; Antonio, T.B.; Jenny, L.B.; Larry, C.C.; Cindy, C.; Denise, R.; Claire, M.P.; Mark, A.N. Inhibitory effect of selenomethionine on the growth of three selected human tumor cell lines. Cancer Lett. 1998, 125, 103–110. [Google Scholar]
- Gerhard, N.S. Selenomethionine: A review of its nutritional significance, metabolism and toxicity. Am. Soc. Nutr. 2000, 130, 1653–1656. [Google Scholar]
- Toxnet Toxicology Data Network. Available online: https://toxnet.nlm.nih.gov/cgi-bin/sis/search/a?dbs+hsdb:@term+@DOCNO+3564 (accessed on 12 September 2018).
- Cummins, L.; Kimura, E. Safety Evaluation of Selenium Sulfide Antidandruff Shampoos. Toxicol. Appl. Pharmacol. 1971, 20, 89–96. [Google Scholar] [CrossRef]
- Zhang, J.S. Evaluation of nanotoxicity of foods and drugs: Biological properties of red elemental selenium at nano size (nano-Se) in vitro and in vivo. In Nanotoxicity: From In Vivo and In Vitro Models to Health Risks; Saura, C.S., Daniel, A.C., Eds.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2009; pp. 97–113. [Google Scholar]
- Thomson, C. Assessment of requirements for selenium and adequacy of selenium status: A review. Eur. J. Clin. Nutr. 2004, 58, 391–402. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Lee, S.Y.; Ji, G.E. Timing of Bifidobacterium administration influences the development of allergy to ovalbumin in mice. Biotechnol. Lett. 2005, 27, 1361–1367. [Google Scholar] [CrossRef] [PubMed]
- Kim, N.; Kunisawa, J.; Kweon, M.N.; Ji, G.E.; Kiyono, H. Oral feeding of Bifidobacterium bifidum (BGN4) prevents CD4+ CD45RB high T cell-mediated inflammatory bowel disease by inhibition of disordered T cell activation. Clin. Immunol. 2007, 123, 30–39. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.; Ku, S.; Kim, S.; Lee, H.; Jin, H.; Kang, S.; Li, R.; Johnston, T.; Park, M.; Ji, G. Safety Evaluations of Bifidobacterium bifidum BGN4 and Bifidobacterium longum BORI. Int. J. Mol. Sci. 2018, 19, 1422. [Google Scholar] [CrossRef] [PubMed]
- Vonderheide, A.P.; Wrobel, K.; Kannamkumarath, S.S.; B′Hymer, C.; Montes-Bayón, M.; Ponce de León, C.; Caruso, J.A. Characterization of selenium species in Brazil nuts by HPLC-ICP-MS and ES-MS. J. Agric. Food Chem. 2002, 50, 5722–5728. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Selenomethionine-enriched Bifidobacterium bifidum BGN4 cells are available from the authors. |
| Cell Strains | Dry Weight of Selenium-Enriched LAB (mg) | ||
|---|---|---|---|
| SS 1 Added at 0 h | SS Added at 12 h | Control (No SS Added) | |
| L. bulgaricus KCTC 3188 | 15 ± 0.62 ** | 24.3 ± 0.55 | 25 ± 0.38 |
| L. acidophilus KCTC 3142 | 16.3 ± 0.40 ** | 22.8 ± 0.42 | 22.3 ± 0.26 |
| L. casei KFRI 704 | 15 ± 0.49 ** | 25.6 ± 0.31 | 25.8 ± 0.56 |
| L. brevis 353 | 21 ± 0.49 ** | 31.1 ± 0.55 | 30.5 ± 0.81 |
| B. bifidum BGN4 | 5.3 ± 0.50 ** | 20.9 ± 0.53 | 22 ± 0.35 |
| Cell Strains | Total Organic Selenium Content of LAB (µg/g) |
|---|---|
| L. bulgaricus KCTC 3188 | 111.7 ± 0.16 a |
| L. acidophilus KCTC 3142 | 134.4 ± 1.18 b |
| L. casei KFRI 704 | 35 ± 0.20 c |
| L. brevis 353 | 111.8 ± 0.86 a |
| B. bifidum BGN4 | 207.5 ± 1.25 d |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jin, W.; Yoon, C.; Johnston, T.V.; Ku, S.; Ji, G.E. Production of Selenomethionine-Enriched Bifidobacterium bifidum BGN4 via Sodium Selenite Biocatalysis. Molecules 2018, 23, 2860. https://doi.org/10.3390/molecules23112860
Jin W, Yoon C, Johnston TV, Ku S, Ji GE. Production of Selenomethionine-Enriched Bifidobacterium bifidum BGN4 via Sodium Selenite Biocatalysis. Molecules. 2018; 23(11):2860. https://doi.org/10.3390/molecules23112860
Chicago/Turabian StyleJin, Weihong, Cheolho Yoon, Tony V. Johnston, Seockmo Ku, and Geun Eog Ji. 2018. "Production of Selenomethionine-Enriched Bifidobacterium bifidum BGN4 via Sodium Selenite Biocatalysis" Molecules 23, no. 11: 2860. https://doi.org/10.3390/molecules23112860
APA StyleJin, W., Yoon, C., Johnston, T. V., Ku, S., & Ji, G. E. (2018). Production of Selenomethionine-Enriched Bifidobacterium bifidum BGN4 via Sodium Selenite Biocatalysis. Molecules, 23(11), 2860. https://doi.org/10.3390/molecules23112860







