Pentafluorosulfanyl-containing Triclocarban Analogs with Potent Antimicrobial Activity
Abstract
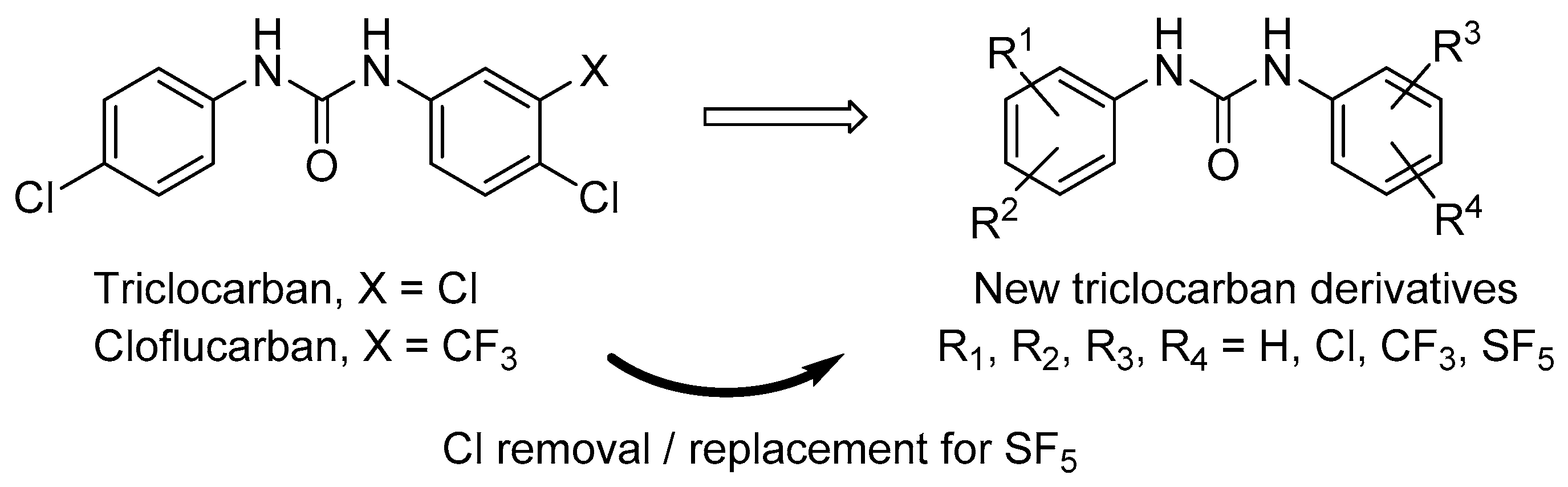
1. Introduction
2. Results and Discussion
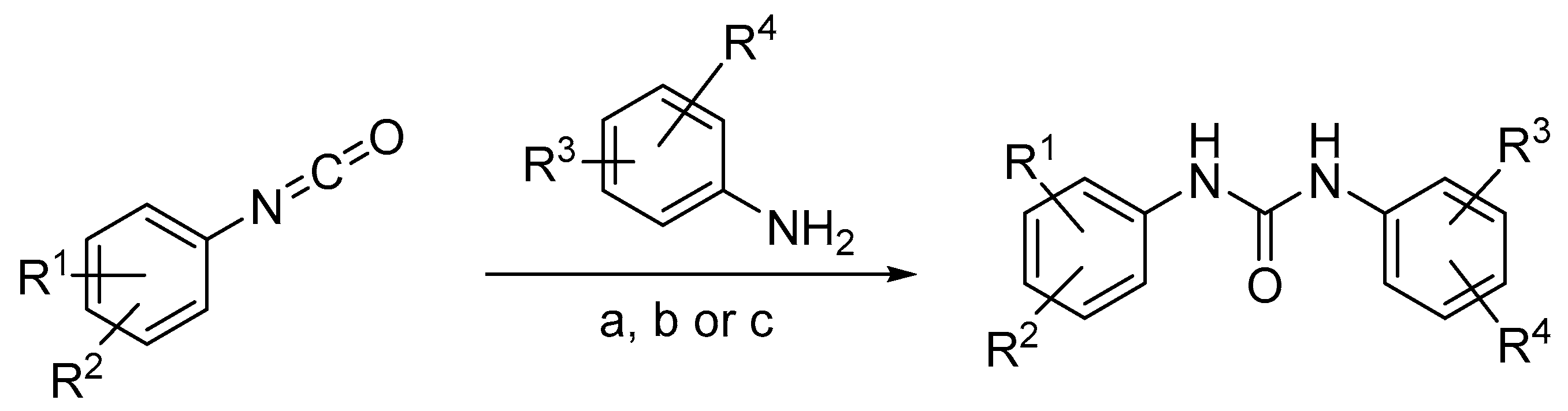
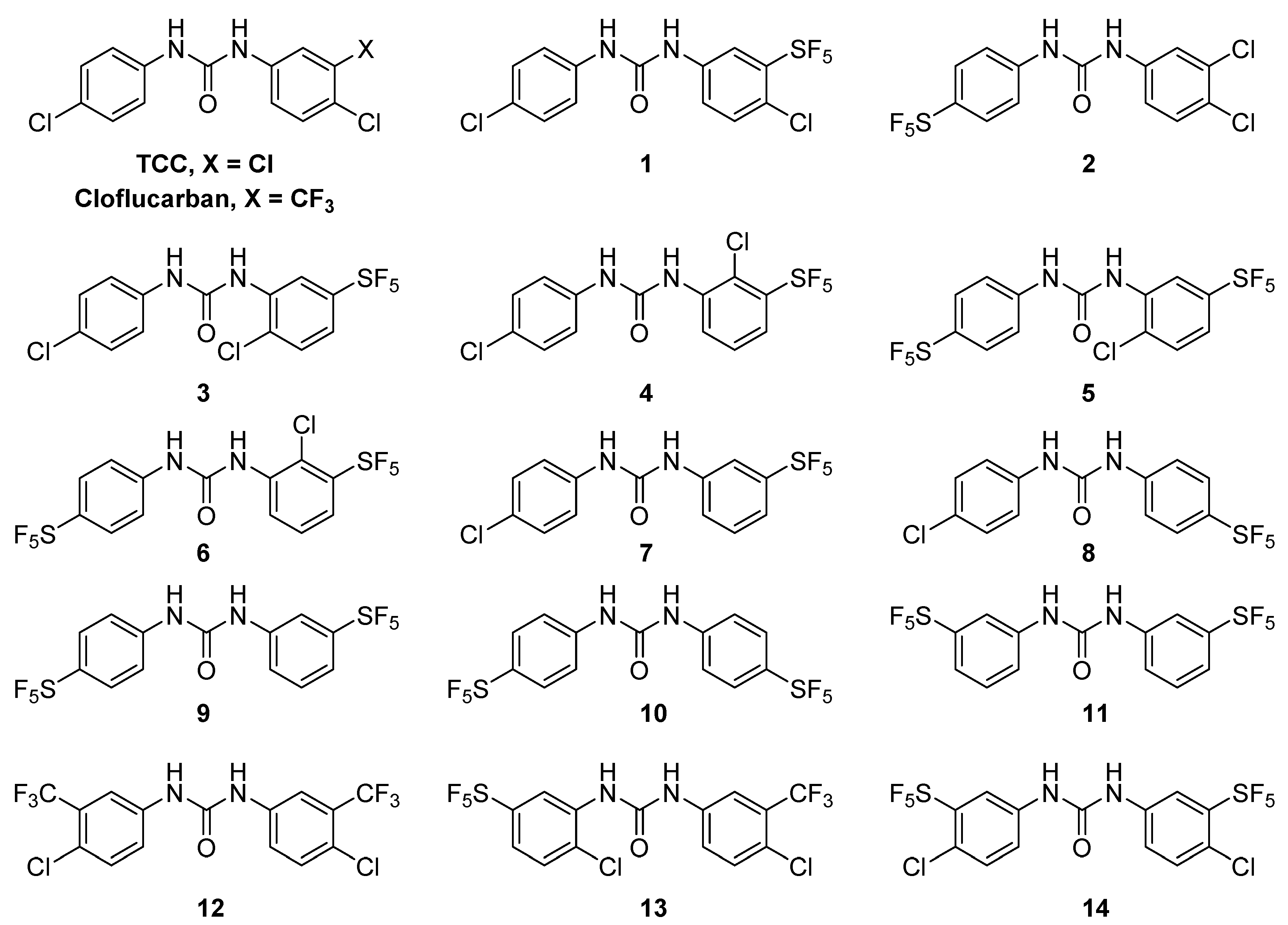
2.1. Chemistry
2.2. Antibacterial Activity, Selectivity Index and Structure-Activity Relationships
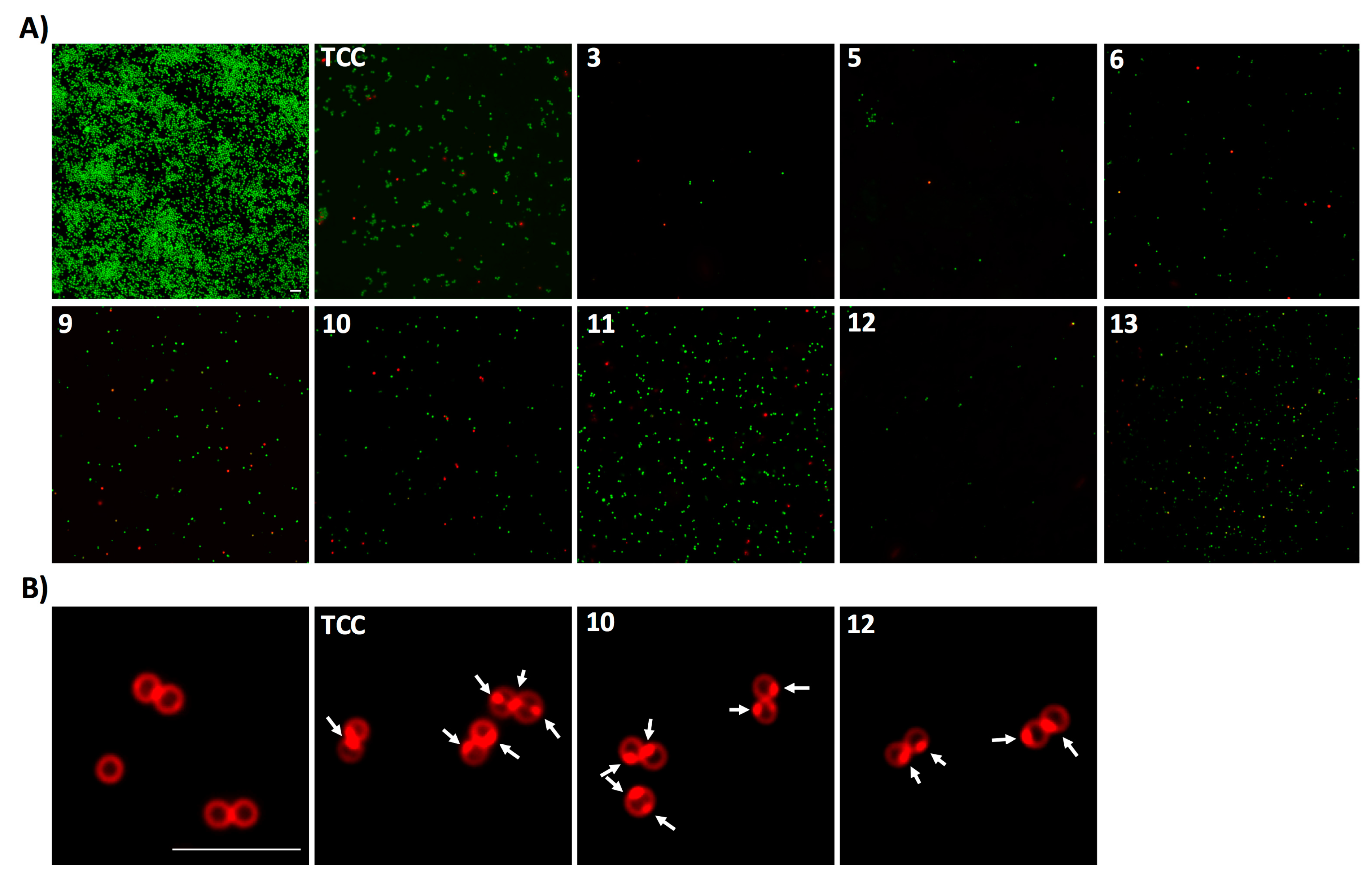
2.3. Diarylureas Show a Bacteriolytic Mode of Action
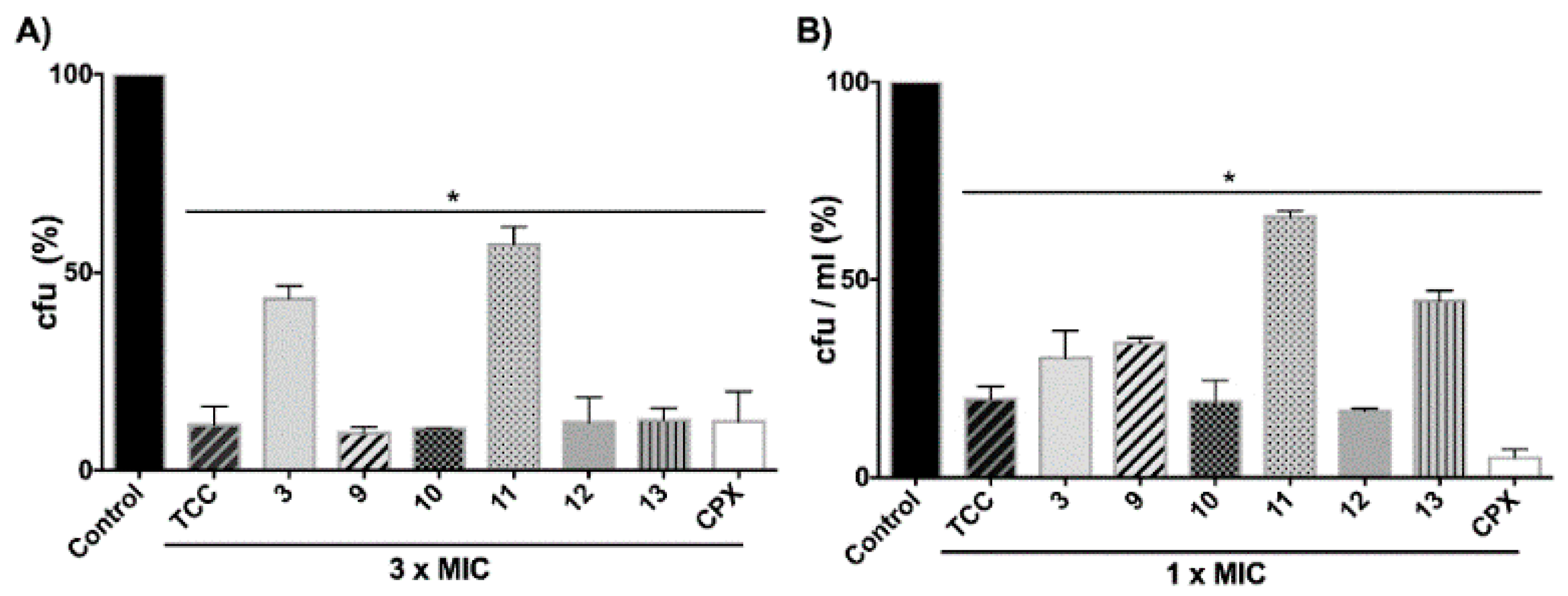
2.4. Antimicrobial Activity of the New Diarylureas on Removal of Biofilms in Catheters and on Disinfection on Contaminated Surfaces
2.5. New Compounds Show Less Spontaneous Mutation Rates Compared to TCC
3. Materials and Methods
3.1. Chemical Synthesis
3.1.1. General Methods
3.1.2. General Procedures for the Synthesis of Aryl Isocyanates
3.1.3. General Procedure A for the Synthesis of Ureas 2–6
3.1.4. General Procedure B for the Synthesis of Ureas 1, 7 and 8
3.1.5. General Procedure C for the Synthesis of Ureas 9–14
3.2. Bacterial Strains and Growth Conditions
3.3. Antibacterial Susceptibility Testing
3.4. Antibacterial Effect of Compounds on Cleaning a Surface
3.5. Antibacterial Effect of Compounds on Biofilms Growing on Catheters
3.6. Fluorescent Microscopy Viability Test Analysis
3.7. Spontaneous Mutation Frequency to Resistance
3.8. Mammalian Cytotoxicity Determination
4. Patents
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Schetty, G.; Stammbach, W.; Zinkernagel, R. Insecticidal Derivatives of Diphenyl Urea. U.S. Patent 2745874, 15 May 1956. [Google Scholar]
- Zhang, Y.; Anderson, M.; Weisman, J.L.; Lu, M.; Choy, C.J.; Boyd, V.A.; Price, J.; Siga, M.; Clark, J.; Connelly, M.; et al. Evaluation of diarylureas for activity against Plasmodium falciparum. ACS Med. Chem. Lett. 2010, 1, 460–465. [Google Scholar] [CrossRef] [PubMed]
- Cowan, N.; Dätwyler, P.; Ernst, B.; Wang, C.; Vennerstro, J.L.; Spangenberg, T.; Keiser, J. Activities of N,N′-diarylurea MMV665852 analogs against Schistosoma mansoni. Antimicrob. Agents Chemother. 2015, 59, 1935–1941. [Google Scholar] [CrossRef] [PubMed]
- Brown, J.R.; North, E.J.; Hurdle, J.G.; Morisseau, C.; Scarborough, J.S.; Sun, D.; Korduláková, J.; Scherman, M.S.; Jones, V.; Grzegorzewicz, A.; et al. The structure-activity relationship of urea derivatives as anti-tuberculosis agents. Bioorg. Med. Chem. 2011, 19, 5585–5595. [Google Scholar] [CrossRef] [PubMed]
- Sviripa, V.; Zhang, W.; Conroy, M.D.; Schmidt, E.S.; Liu, A.X.; Truong, J.; Chunming, L.; Watt, D.S. Fluorinated N,N′-diarylureas as AMPK activators. Bioorg. Med. Chem. Lett. 2013, 23, 1600–1603. [Google Scholar] [CrossRef] [PubMed]
- Jin, Q.; Nie, H.; McCleland, B.W.; Widdowson, K.L.; Palovich, M.R.; Elliott, J.D.; Goodman, R.M.; Burman, M.; Sarau, H.M.; Ward, K.W.; et al. Discovery of potent and orally bioavailable N,N′-diarylurea antagonists for the CXCR2 chemokine receptor. Bioorg. Med. Chem. Lett. 2004, 14, 4375–4378. [Google Scholar] [CrossRef] [PubMed]
- Denoyelle, S.; Chen, T.; Chen, L.; Wang, Y.; Klosi, E.; Halperin, J.A.; Aktas, B.H.; Chorev, M. In vitro inhibition of translation initiation by N,N′-diarylureas–potential anti-cancer agents. Bioorg. Med. Chem. Lett. 2012, 22, 402–409. [Google Scholar] [CrossRef] [PubMed]
- Roman, D.P.; Barnett, E.H.; Balske, R.J. Cutaneous antiseptic activity of 3,4,4′-trichlorocarbanilide. Proc. Sci. Sect. Toilet. Goods Assoc. 1957, 28, 1213–1214. [Google Scholar]
- Ahn, C.K.; Zhao, B.; Chen, J.; Cherednichenko, G.; Sanmarti, E.; Denison, M.S.; Lasley, B.; Pessah, I.N.; Kültz, D.; Chang, D.P.Y.; et al. In vitro biologic activities of the antimicrobials triclocarban, its analogs, and triclosan in bioassay screens: Receptor-based bioassay screens. Environ. Health Perspect. 2008, 16, 1203–1210. [Google Scholar] [CrossRef] [PubMed]
- Schebb, N.H.; Inceoglu, B.; Ahn, K.C.; Morisseau, C.; Gee, S.J.; Hammock, B.D. Investigation of human exposure to triclocarban after showering and preliminary evaluation of its biological effects. Environ. Sci. Technol. 2011, 45, 3109–3115. [Google Scholar] [CrossRef] [PubMed]
- Venkatesan, A.K.; Pycke, B.F.G.; Barber, L.B.; Lee, K.E.; Halden, R.U. Occurrence of triclosan, triclocarban, and its lesser chlorinated congeners in Minnesota freshwater sediments collected near wastewater treatment plants. J. Hazard. Mater. 2012, 229–230, 29–35. [Google Scholar] [CrossRef] [PubMed]
- Scharpf, L.G., Jr.; Hill, I.D.; Maibach, H.I. Percutaneous penetration and disposition of triclocarban in man: Body showering. Arch. Environ. Health 1975, 30, 7–14. [Google Scholar] [CrossRef] [PubMed]
- Schebb, N.H.; Buchholz, B.A.; Hammock, B.D.; Rice, R.H.J. Metabolism of the antibacterial triclocarban by human epidermal keratinocytes to yield protein adducts. Biochem. Mol. Toxicol. 2012, 26, 230–234. [Google Scholar] [CrossRef] [PubMed]
- Xie, W.; Zhang, W.; Ren, J.; Li, W.; Zhou, L.; Cui, Y.; Chen, H.; Yu, W.; Zhuang, X.; Zhang, Z.; et al. Metabonomics indicates inhibition of fatty acid synthesis, β-oxidation, and tricarboxylic acid cycle in triclocarban-induced cardiac metabolic alterations in male mice. J. Agric. Food. Chem. 2018, 66, 1533–1542. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Qiu, H.; Morisseau, C.; Hwang, S.H.; Tsai, H.; Ulu, A.; Chiamvimonvat, N.; Hammock, B.D. Inhibition of soluble epoxide hydrolase contributes to the anti-inflammatory effect of antimicrobial triclocarban in a murine model. Toxicol. Appl. Pharmacol. 2011, 255, 200–206. [Google Scholar] [CrossRef] [PubMed]
- Chalew, T.E.; Halden, R.U. Environmental exposure of aquatic and terrestrial biota to triclosan and triclocarban. J. Am. Water Works Assoc. 2009, 45, 4–13. [Google Scholar] [CrossRef] [PubMed]
- Halden, R.U.; Paull, D.H. Analysis of triclocarban in aquatic samples by liquid chromatography electrospray ionization mass spectrometry. Environ. Sci. Technol. 2004, 38, 4849–4855. [Google Scholar] [CrossRef] [PubMed]
- Ying, G.; Yu, X.; Kookana, R.S. Biological degradation of triclocarban and triclosan in a soil under aerobic and anaerobic conditions and comparison with environmental fate modelling. Environ. Pollut. 2007, 150, 300–305. [Google Scholar] [CrossRef] [PubMed]
- Gledhill, W.E. Biodegradation of 3,4,4′-trichlorocarbanilide, TCC®, in sewage and activated sludge. Water Res. 1975, 9, 649–654. [Google Scholar] [CrossRef]
- FDA Issues Final Rule on Safety and Effectiveness of Antibacterial Soaps. Available online: https://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm517478.htm (accessed on 14 October 2018).
- Hiles, R.A. Metabolism and toxicity of halogenated carbanilides: Absorption, distribution and excretion of radioactivity from 3,4,4′-trichloro[14C]carbanilide (TCC) and 3-trifluoromethyl-4,4′-dichloro[14C]carbanilide (TFC) in rats. Food Cosmet. Toxicol. 1977, 15, 205–211. [Google Scholar] [CrossRef]
- Jeffcoat, A.R.; Handy, R.W.; Francis, M.T.; Willis, S.; Wall, M.E.; Birch, C.G.; Hiles, R.A. The metabolism and toxicity of halogenated carbanilides. Biliary metabolites of 3,4,4′-trichloro-carbanilide and 3-trifluoromethyl-4,4′-dichlorocarbanilide in the rat. Drug Metab. Dispos. 1977, 5, 157–166. [Google Scholar] [PubMed]
- Markgraf, J.H.; Quinn, H. Antibacterial composition. U.S. Patent 3485919, 23 December 1969. [Google Scholar]
- Savoie, P.R.; Welch, J.T. Preparation and utility of organic pentafluorosulfanyl-containing compounds. Chem. Rev. 2015, 115, 1130–1190. [Google Scholar] [CrossRef] [PubMed]
- Altomonte, S.; Zanda, M. Synthetic chemistry and biological activity of pentafluorosulphanyl (SF5) organic molecules. J. Fuorine Chem. 2012, 143, 57–93. [Google Scholar] [CrossRef]
- Bassetto, M.; Ferla, S.; Pertusati, F. Polyfluorinated groups in medicinal chemistry. Future Med. Chem. 2015, 7, 527–546. [Google Scholar] [CrossRef] [PubMed]
- Welch, J.T. Applications of pentafluorosulfanyl substitution in life sciences research. In Fluorine in Pharmaceutical and Medicinal Chemistry: From Biophysical Aspects to Clinical Applications; Gouverneur, V., Müller, K., Eds.; World Scientific: London, UK, 2012; pp. 175–207. [Google Scholar]
- Saethre, L.J.; Berrah, N.; Bozek, J.D.; Boerve, K.J.; Carroll, T.X.; Kukk, E.; Gard, G.L.; Winter, R.; Thomas, T.D. Chemical insights from high-resolution X-ray photoelectron spectroscopy and ab initio theory: Propyne, trifluoropropyne, and ethynylsulfur pentafluoride. J. Am. Chem. Soc. 2001, 123, 10729–10737. [Google Scholar] [CrossRef] [PubMed]
- Kanishchev, O.S.; Dolbier, W.R., Jr. Generation of ortho-SF5-benzyne and its diels−alder reactions with furans: Synthesis of 1-SF5-naphthalene, its derivatives, and 1,6(1,7)-bis-SF5-naphthalenes. J. Org. Chem. 2016, 81, 11305–11311. [Google Scholar] [CrossRef] [PubMed]
- Sheppard, W.A. Arylsulfur pentafluorides. J. Am. Chem. Soc. 1962, 84, 3072–3076. [Google Scholar] [CrossRef]
- Westphal, M.V.; Wolfstädter, B.T.; Plancher, J.; Gatfield, J.; Carreira, E.M. Evaluation of tert-butyl isosteres: Case studies of physicochemical and pharmacokinetic properties, efficacies, and activities. ChemMedChem. 2015, 10, 461–469. [Google Scholar] [CrossRef] [PubMed]
- Phillips, M.A.; Lotharius, J.; Marsh, K.; White, J.; Dayan, A.; White, K.L.; Njoroge, J.W.; El Mazouni, F.; Lao, Y.; Kokkonda, S.; et al. A long-duration dihydroorotate dehydrogenase inhibitor (DSM265) for prevention and treatment of malaria. Sci. Transl. Med. 2015, 7, 296ra111. [Google Scholar] [CrossRef] [PubMed]
- Murphy, C.D. Microbial degradation of fluorinated drugs: Biochemical pathways, impacts on the environment and potential applications. Appl. Microbiol. Biotechnol. 2016, 100, 2617–2627. [Google Scholar] [CrossRef] [PubMed]
- Jackson, D.A.; Mabury, S.A. Environmental properties of pentafluorosulfanyl compounds: Physical properties and photodegradation. Environ. Toxicol. Chem. 2009, 28, 1866–1879. [Google Scholar] [CrossRef] [PubMed]
- Zarei, M.; Vázquez-Carrera, M.; Vázquez, S.; Leiva, R.; Pujol, E. HRI Activators Useful for the Treatment of Cardiometabolic Diseases. Patent PCT WO2018/010856A1, 18 January 2018. [Google Scholar]
- Raasch, M.S. Ureido-Substituted Arylsulfur Pentafluorides. U.S. Patent 3073861, 15 January 1963. [Google Scholar]
- Karagiannidis, L.E.; Haynes, C.J.E.; Holder, K.J.; Kirby, I.L.; Moore, S.J.; Wells, N.J.; Gale, P.A. Highly effective yet simple transmembrane anion transporters based upon ortho-phenylenediamine bis-ureas. Chem. Commun. 2014, 50, 12050–12053. [Google Scholar] [CrossRef] [PubMed]
- Walsh, S.E.; Maillard, J.Y.; Russell, A.D.; Catrenich, C.E.; Charbonneau, D.L.; Bartolo, R.G. Activity and mechanisms of action of selected biocidal agents on Gram-positive and -negative bacteria. J. Appl. Microbiol. 2003, 94, 240–247. [Google Scholar] [CrossRef] [PubMed]
- Hughes, D.; Karlén, A. Discovery and preclinical development of new antibiotics. Upsala J. Med. Sci. 2014, 119, 162–169. [Google Scholar] [CrossRef] [PubMed]
- Kleemann, H.; Weck, R. Ortho-Substituted Pentafluorosulfanylbenzenes, Process for Their Preparation and Their Use as Valuable Synthetic Intermediates. U.S. Patent 7932416 B2, 26 April 2011. [Google Scholar]
- Barniol-Xicota, M.; Escandell, A.; Valverde, E.; Julián, E.; Torrents, E.; Vázquez, S. Antibacterial activity of novel benzopolycyclic amines. Bioorg. Med. Chem. 2015, 23, 290–296. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are available from the authors. |
| Compound | MIC50 (µg/mL) b,c | CC50 (µg/mL) d | ||||
|---|---|---|---|---|---|---|
| S. aureus | S. aureus MRSA | S. epidermis | S. mutants | E. faecalis | ||
| TCC | 0.5 (29) | 0.5 (29) | 0.5 (29) | NA e | NA | 14.5 |
| TFC | 0.5 (27.4) | 0.5 (27.4) | 0.5 (27.4) | NA | >20 | 13.7 |
| 1 | 1 (4.8) | 1 (4.8) | 1 (4.8) | 5 | 1 (4.8) | 4.8 |
| 2 | 1 (4.8) | 1 (4.8) | 1 (4.8) | 5 | 1 (4.8) | 4.8 |
| 3 | 0.5 (110) | 0.5 (110) | 1 (55) | NA | NA | 55 |
| 4 | 0.5 (56) | 0.5 (56) | 0.5 (56) | NA | 5 | 28 |
| 5 | 0.5 (10.2) | 0.5 (10.2) | 0.5 (10.2) | 0.5 (10.2) | 0.5 (10.2) | 5.6 |
| 6 | 0.5 (8.4) | 0.5 (8.4) | 0.5 (8.4) | 0.5 (8.4) | 0.5 (8.4) | 4.2 |
| 7 | 0.5 (26.2) | 0.5 (26.2) | 0.5 (26.2) | >20 | 5 | 13.1 |
| 8 | 0.5 (26) | 0.5 (26) | 0.5 (26) | NA | 0.5 (26) | 13 |
| 9 | 0.5 (25) | 0.5 (25) | 0.5 (25) | 0.5 (25) | 0.5 (25) | 12.5 |
| 10 | 0.05 (412) | 0.05 (412) | 0.5 (41.2) | 0.5 (41.2) | 0.5 (41.2) | 20.6 |
| 11 | 0.5 (67) | 0.5 (67) | 0.5 (67) | 0.5 (67) | >10 | 33.5 |
| 12 | 0.5 (67.6) | 0.3 (112.7) | 0.5 (67.6) | 2 (16.9) | 2 (16.9) | 33.8 |
| 13 | 0.5 (49.2) | 0.5 (49.2) | 0.5 (49.2) | 0.5 (49.2) | 0.5 (49.2) | 24.6 |
| 14 | 0.5 (20) | 0.5 (20) | 0.5 (20) | 0.5 (20) | 0.5 (20) | 10 |
| Compound (10 µg/ mL) | Frequency of Mutation |
|---|---|
| TCC | 4 × 10−9 |
| 3 | 0 |
| 9 | 0 |
| 10 | 0 |
| 11 | 5 × 10−1 |
| 12 | 5 × 10−10 |
| 13 | 0 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pujol, E.; Blanco-Cabra, N.; Julián, E.; Leiva, R.; Torrents, E.; Vázquez, S. Pentafluorosulfanyl-containing Triclocarban Analogs with Potent Antimicrobial Activity. Molecules 2018, 23, 2853. https://doi.org/10.3390/molecules23112853
Pujol E, Blanco-Cabra N, Julián E, Leiva R, Torrents E, Vázquez S. Pentafluorosulfanyl-containing Triclocarban Analogs with Potent Antimicrobial Activity. Molecules. 2018; 23(11):2853. https://doi.org/10.3390/molecules23112853
Chicago/Turabian StylePujol, Eugènia, Núria Blanco-Cabra, Esther Julián, Rosana Leiva, Eduard Torrents, and Santiago Vázquez. 2018. "Pentafluorosulfanyl-containing Triclocarban Analogs with Potent Antimicrobial Activity" Molecules 23, no. 11: 2853. https://doi.org/10.3390/molecules23112853
APA StylePujol, E., Blanco-Cabra, N., Julián, E., Leiva, R., Torrents, E., & Vázquez, S. (2018). Pentafluorosulfanyl-containing Triclocarban Analogs with Potent Antimicrobial Activity. Molecules, 23(11), 2853. https://doi.org/10.3390/molecules23112853







