Assessing Nutritional Traits and Phytochemical Composition of Artisan Jams Produced in Comoros Islands: Using Indigenous Fruits with High Health-Impact as an Example of Biodiversity Integration and Food Security in Rural Development
Abstract
:1. Introduction
2. Results and Discussion
2.1. Nutraceutical Properties
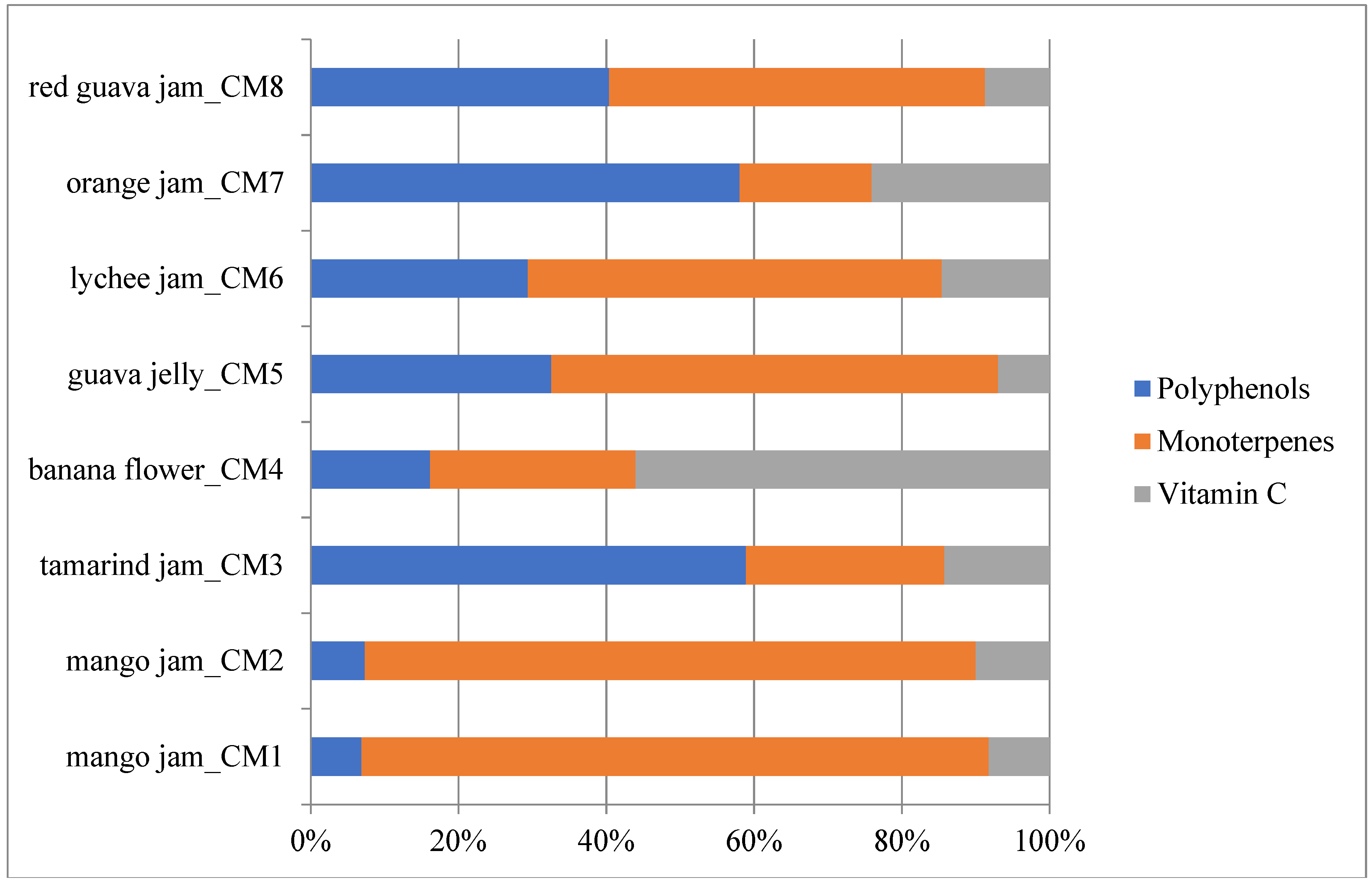
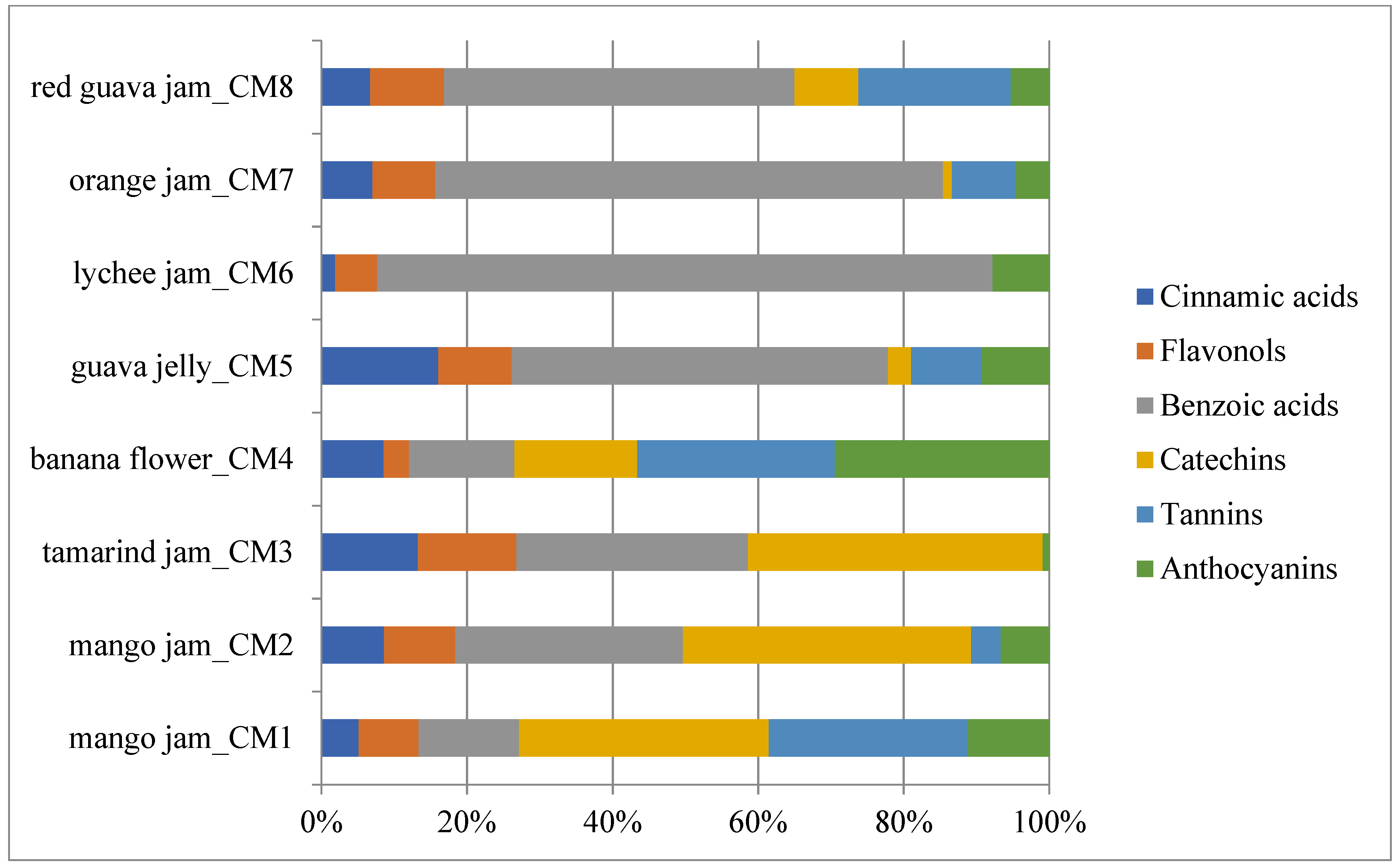
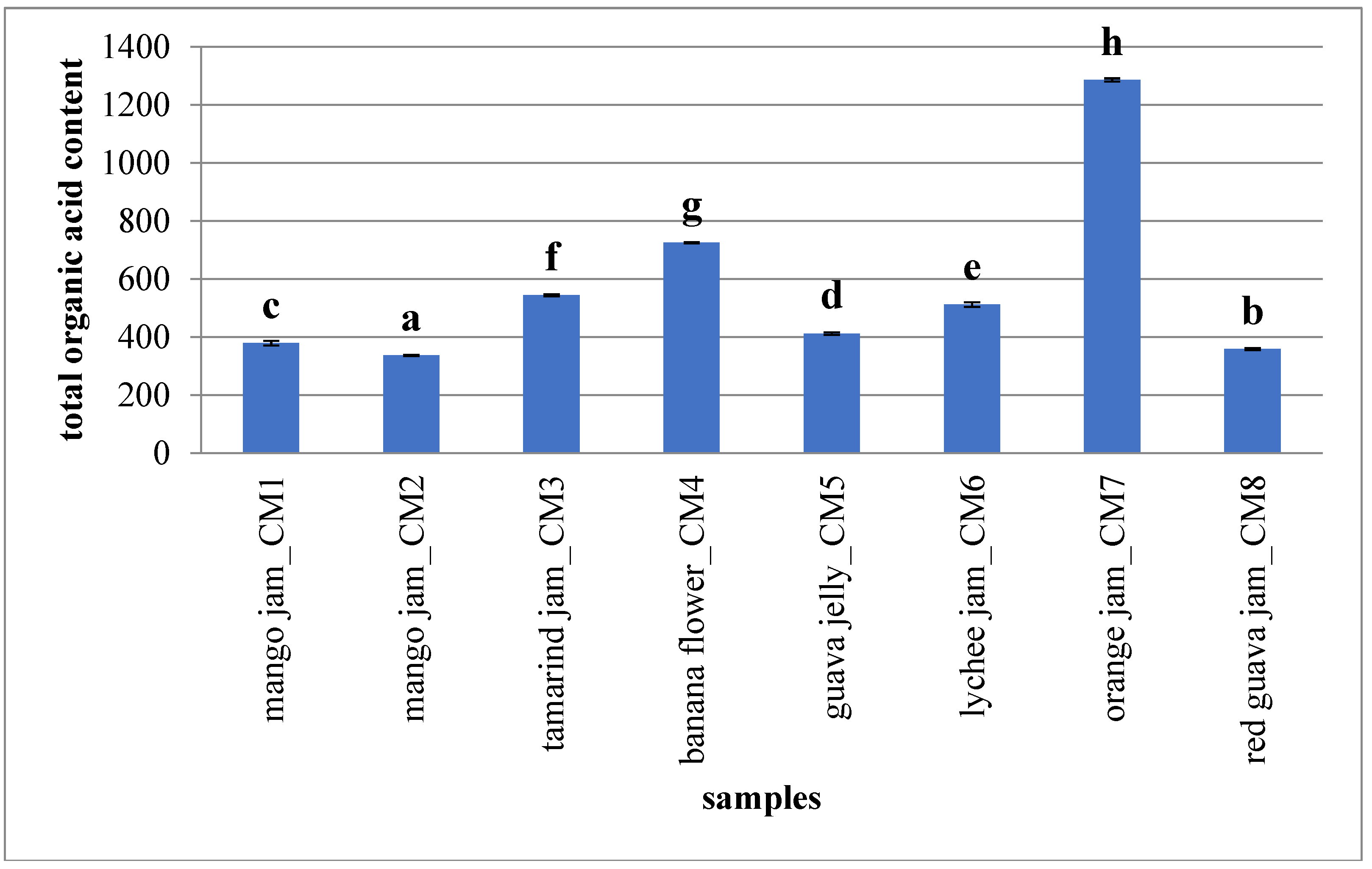
2.2. Phytochemical Composition
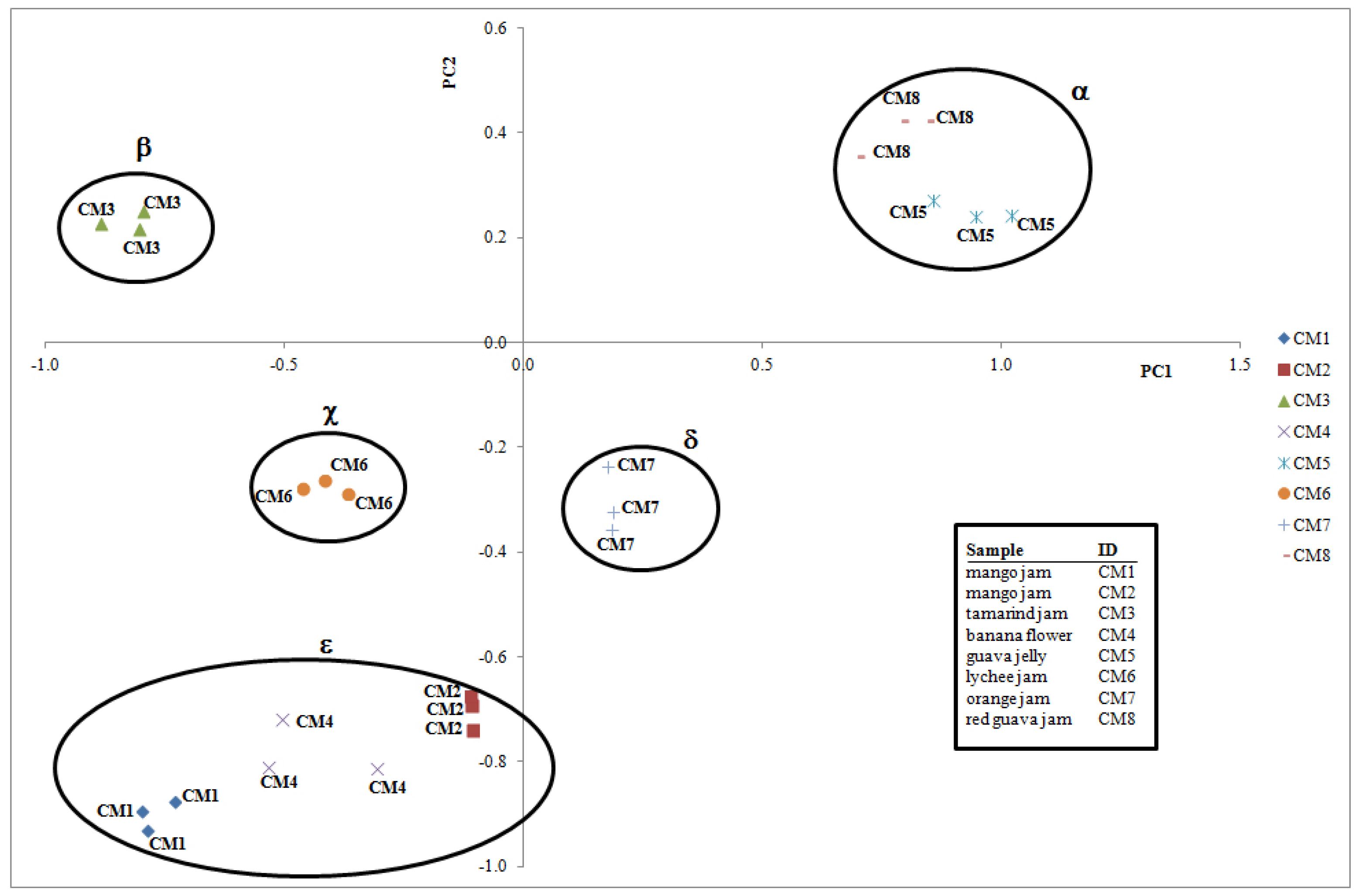
2.3. Multivariate Analysis
3. Materials and Methods
3.1. Plant. Material
3.2. Preparation of Fruit-Derived Products
3.3. Spectrophotometric Analysis
3.4. Chromatographic Analysis
3.5. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Sewall, B.J.; Granek, E.F.; Trewhella, W.J. The endemic Comoros Islands fruit bat Rousettus obliviosus: Ecology, conservation, and red list status. Oryx 2003, 37, 344–352. [Google Scholar] [CrossRef]
- Soifoini, T.; Donno, D.; Jeannoda, V.; Rakotoniaina, E.; Hamidou, S.; Achmet, S.M.; Solo, N.R.; Afraitane, K.; Giacoma, C.; Beccaro, G.L. Bioactive compounds, nutritional traits, and antioxidant properties of Artocarpus altilis (Parkinson) fruits: Exploiting a potential functional food for food security on the Comoros Islands. J. Food Qual. 2018, in press. [Google Scholar] [CrossRef]
- Clevidence, B. Tropical and subtropical fruits: Phytonutrients and anticipated health benefits. In Proceedings of the III International Symposium on Tropical and Subtropical Fruits, Fortaleza, Ceara, Brazil, 12–17 September 2004; pp. 485–498. [Google Scholar]
- Lim, Y.Y.; Lim, T.T.; Tee, J.J. Antioxidant properties of several tropical fruits: A comparative study. Food Chem. 2007, 103, 1003–1008. [Google Scholar] [CrossRef]
- Sajise, P.; Ahmad, I. Conservation and sustainable use of tropical and sub-tropical fruits: Current status and prospects. In Proceedings of the International Workshop on Tropical and Subtropical Fruits, Chiang Mai, Thailand, April 2007; pp. 97–112. [Google Scholar]
- Chivandi, E.; Mukonowenzou, N.; Nyakudya, T.; Erlwanger, K.H. Potential of indigenous fruit-bearing trees to curb malnutrition, improve household food security, income and community health in sub-saharan africa: A review. Food Res. Int. 2015, 76, 980–985. [Google Scholar] [CrossRef]
- Reardon, T.; Barrett, C.B.; Berdegué, J.A.; Swinnen, J.F.M. Agrifood industry transformation and small farmers in developing countries. World Dev. 2009, 37, 1717–1727. [Google Scholar] [CrossRef]
- Singh, S.; Jain, S.; Singh, S.; Singh, D. Quality changes in fruit jams from combinations of different fruit pulps. J. Food Process. Preserv. 2009, 33, 41–57. [Google Scholar] [CrossRef]
- Codex_Alimentarius. Codex Standard for Jams, Jellies and Marmalades (Codex Stan 296-2009). Available online: http://www.codexalimentarius.org (accessed on 27 March 2018).
- Kim, D.O.; Padilla-Zakour, O. Jam processing effect on phenolics and antioxidant capacity in anthocyanin-Rich fruits: Cherry, plum, and raspberry. J. Food Sci. 2004, 69, S395–S400. [Google Scholar] [CrossRef]
- Rosa, A.; Atzeri, A.; Deiana, M.; Scano, P.; Incani, A.; Piras, C.; Cesare Marincola, F. Comparative antioxidant activity and 1H NMR profiling of Mediterranean fruit products. Food Res. Int. 2015, 69, 322–330. [Google Scholar] [CrossRef]
- Puiggròs, F.; Solà, R.; Bladé, C.; Salvadó, M.-J.; Arola, L. Nutritional biomarkers and foodomic methodologies for qualitative and quantitative analysis of bioactive ingredients in dietary intervention studies. J. Chromatogr. A 2011, 1218, 7399–7414. [Google Scholar] [CrossRef] [PubMed]
- Donno, D.; Cavanna, M.; Beccaro, G.L.; Mellano, M.G.; Torello-Marinoni, D.; Cerutti, A.K.; Bounous, G. Currants and strawberries as bioactive compound sources: Determination of antioxidant profiles with HPLC-DAD/MS. J. App. Bot. Food Qual. 2013, 86, 1–10. [Google Scholar]
- Gonzalez-Molina, E.; Moreno, D.A.; Garcia-Viguera, C. Genotype and harvest time influence the phytochemical quality of fino lemon juice (Citrus limon (L.) Burm. F.) for industrial use. J. Agric. Food Chem. 2008, 56, 1669–1675. [Google Scholar] [CrossRef] [PubMed]
- Tibaldi, G.; Fontana, E.; Nicola, S. Growing conditions and postharvest management can affect the essential oil of Origanum vulgare L. spp. hirtum (Link) ietswaart. Ind. Crop. Prod. 2011, 34, 1516–1522. [Google Scholar] [CrossRef]
- Bengoechea, M.L.; Sancho, A.I.; Bartolomé, B.; Estrella, I.; Gómez-Cordovés, C.; Hernández, M.T. Phenolic composition of industrially manufactured purees and concentrates from peach and apple fruits. J. Agric. Food Chem. 1997, 45, 4071–4075. [Google Scholar] [CrossRef]
- Dragovic-Uzelac, V.; Pospišil, J.; Levaj, B.; Delonga, K. The study of phenolic profiles of raw apricots and apples and their purees by HPLC for the evaluation of apricot nectars and jams authenticity. Food Chem. 2005, 91, 373–383. [Google Scholar] [CrossRef]
- Clifford, M.N. Anthocyanins—Nature, occurrence and dietary burden. J. Agric. Food Chem. 2000, 80, 1063–1072. [Google Scholar] [CrossRef]
- Banaś, A.; Korus, A.; Tabaszewska, M. Quality assessment of low-sugar jams enriched with plant raw materials exhibiting health-promoting properties. J. Food Sci. Technol. 2018, 55, 408–417. [Google Scholar] [CrossRef] [PubMed]
- Poiana, M.-A.; Moigradean, D.; Dogaru, D.; Mateescu, C.; Raba, D.; Gergen, I. Processing and storage impact on the antioxidant properties and color quality of some low sugar fruit jams. Rom. Biotechnol. Lett. 2011, 16, 6504–6512. [Google Scholar]
- Rababah, T.M.; Al-Mahasneh, M.A.; Kilani, I.; Yang, W.; Alhamad, M.N.; Ereifej, K.; Al-u’datt, M. Effect of jam processing and storage on total phenolics, antioxidant activity, and anthocyanins of different fruits. J. Sci. Food Agric. 2011, 91, 1096–1102. [Google Scholar] [CrossRef] [PubMed]
- Heimler, D.; Romani, A.; Ieri, F. Plant polyphenol content, soil fertilization and agricultural management: A review. Eur. Food Res. Technol. 2017, 243, 1107–1115. [Google Scholar] [CrossRef]
- Donno, D.; Randriamampionona, D.; Andriamaniraka, H.; Torti, V.; Mellano, M.G.; Giacoma, C.; Beccaro, G.L. Biodiversity and traditional medicinal plants from Madagascar: Phytochemical evaluation of Brachylaena ramiflora (DC.) Humbert decoctions and infusions. J. Appl. Bot. Food Qual. 2017, 90, 205–2013. [Google Scholar]
- Šavikin, K.; Zdunić, G.; Janković, T.; Tasić, S.; Menković, N.; Stević, T.; Đorđević, B. Phenolic content and radical scavenging capacity of berries and related jams from certificated area in Serbia. Plant. Food Hum. Nutr. 2009, 64, 212–217. [Google Scholar] [CrossRef] [PubMed]
- Da Silva Pinto, M.; Lajolo, F.M.; Genovese, M.I. Bioactive compounds and antioxidant capacity of strawberry jams. Plant. Food Hum. Nutr. 2007, 62, 127–131. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Cao, G.; Prior, R.L. Oxygen radical absorbing capacity of anthocyanins. J. Agric. Food Chem. 1997, 45, 304–309. [Google Scholar] [CrossRef]
- Veberic, R.; Slatnar, A.; Bizjak, J.; Stampar, F.; Mikulic-Petkovsek, M. Anthocyanin composition of different wild and cultivated berry species. LWT-Food Sci. Technol. 2015, 60, 509–517. [Google Scholar] [CrossRef]
- Jia, N.; Xiong, Y.L.; Kong, B.; Liu, Q.; Xia, X. Radical scavenging activity of black currant (Ribes nigrum L.) extract and its inhibitory effect on gastric cancer cell proliferation via induction of apoptosis. J. Funct. Foods 2012, 4, 382–390. [Google Scholar] [CrossRef]
- Donno, D.; Beccaro, G.L.; Mellano, M.G.; Cerutti, A.K.; Bounous, G. Goji berry fruit (Lycium spp.): Antioxidant compound fingerprint and bioactivity evaluation. J. Funct. Foods 2015, 18, 1070–1085. [Google Scholar] [CrossRef]
- Silva, B.M.; Andrade, P.B.; Gonçalves, A.C.; Seabra, R.M.; Oliveira, M.B.; Ferreira, M.A. Influence of jam processing upon the contents of phenolics, organic acids and free amino acids in quince fruit (Cydonia oblonga Miller). Eur. Food Res. Technol. 2004, 218, 385–389. [Google Scholar] [CrossRef]
- Wojdyło, A.; Oszmiański, J.; Bielicki, P. Polyphenolic composition, antioxidant activity, and polyphenol oxidase (PPO) activity of quince (Cydonia oblonga Miller) varieties. J. Agric. Food Chem. 2013, 61, 2762–2772. [Google Scholar] [CrossRef] [PubMed]
- Cabral, T.A.; de Morais Cardoso, L.; Pinheiro-Sant’Ana, H.M. Chemical composition, vitamins and minerals of a new cultivar of lychee (Litchi chinensis cv. Tailandes) grown in Brazil. Fruits 2014, 69, 425–434. [Google Scholar] [CrossRef]
- Singh, D.; Wangchu, L.; Moond, S.K. Processed Products of Tamarind. Nat. Prod. Radiance 2007, 6, 315–321. [Google Scholar]
- Jimoh, S.; Onabanjo, O. Potentials of Tamarindus indica (L.) in jam production. J. Agric. Soc. Res. 2012, 12, 29–43. [Google Scholar]
- Marquina, V.; Araujo, L.; Ruíz, J.; Rodríguez-Malaver, A.; Vit, P. Composition and antioxidant capacity of the guava (Psidium guajava L.) fruit, pulp and jam. Arch. Latinoam. Nutr. 2008, 58, 98–102. [Google Scholar] [PubMed]
- Sánchez-Salcedo, E.M.; Mena, P.; García-Viguera, C.; Martínez, J.J.; Hernández, F. Phytochemical evaluation of white (Morus alba L.) and black (Morus nigra L.) mulberry fruits, a starting point for the assessment of their beneficial properties. J. Funct. Foods 2015, 12, 399–408. [Google Scholar] [CrossRef]
- Garcia-Viguera, C.; Tomás-Barberán, F.A.; Ferreres, F.; Artés, F.; Tomás-Lorente, F. Determination of Citrus jams genuineness by flavonoid analysis. Eur. Food Res. Technol. 1993, 197, 255–259. [Google Scholar]
- Del Rio, D.; Rodriguez-Mateos, A.; Spencer, J.P.; Tognolini, M.; Borges, G.; Crozier, A. Dietary (poly) phenolics in human health: Structures, bioavailability, and evidence of protective effects against chronic diseases. Antioxid. Redox Signal. 2013, 18, 1818–1892. [Google Scholar] [CrossRef] [PubMed]
- Kansci, G.; Koubala, B.B.; Mbome, I.L. Biochemical and physicochemical properties of four mango varieties and some quality characteristics of their jams. J. Food Process. Preserv. 2008, 32, 644–655. [Google Scholar] [CrossRef]
- López, R.; Ramírez, A.; de Fariñas Graziani, L. Physicochemical and microbiological evaluation of 3 commercial guava jams (Psidium guajava L.). Arch. Latinoam. Nutr. 2000, 50, 291–295. [Google Scholar] [PubMed]
- Seeram, N.P. Berry fruits: Compositional elements, biochemical activities, and the impact of their intake on human health, performance, and disease. J. Agric. Food Chem. 2008, 56, 627–629. [Google Scholar] [CrossRef] [PubMed]
- Ammar, I.; Ennouri, M.; Bouaziz, M.; Ben Amira, A.; Attia, H. Phenolic profiles, phytchemicals and mineral content of decoction and infusion of Opuntia ficus-indica flowers. Plant. Food Hum. Nutr. 2015, 70, 388–394. [Google Scholar] [CrossRef] [PubMed]
- Landete, J.M. Ellagitannins, ellagic acid and their derived metabolites: A review about source, metabolism, functions and health. Food Res. Int. 2011, 44, 1150–1160. [Google Scholar] [CrossRef]
- Ştefănuţ, M.N.; Căta, A.; Pop, R.; Moşoarcă, C.; Zamfir, A.D. Anthocyanins HPLC-DAD and MS characterization, total phenolics, and antioxidant activity of some berries extracts. Anal. Lett. 2011, 44, 2843–2855. [Google Scholar] [CrossRef]
- Garzon, G.; Wrolstad, R. Comparison of the stability of pelargonidin-based anthocyanins in strawberry juice and concentrate. J. Food Sci. 2002, 67, 1288–1299. [Google Scholar] [CrossRef]
- Kammerer, D.; Carle, R.; Schieber, A. Detection of peonidin and pelargonidin glycosides in black carrots (Daucus carota ssp. Sativus var. Atrorubens alef.) by high-performance liquid chromatography/electrospray ionization mass spectrometry. Rapid Commun. Mass Spectrom. 2003, 17, 2407–2412. [Google Scholar] [CrossRef] [PubMed]
- Schwarz, M.; Wray, V.; Winterhalter, P. Isolation and identification of novel pyranoanthocyanins from black carrot (Daucus carota L.) juice. J. Agric. Food Chem. 2004, 52, 5095–5101. [Google Scholar] [CrossRef] [PubMed]
- Papaefthimiou, D.; Papanikolaou, A.; Falara, V.; Givanoudi, S.; Kostas, S.; Kanellis, A.K. Genus Cistus: A model for exploring labdane-type diterpenes’ biosynthesis and a natural source of high value products with biological, aromatic, and pharmacological properties. Front. Chem. 2014, 2, 35. [Google Scholar] [CrossRef] [PubMed]
- Trombetta, D.; Castelli, F.; Sarpietro, M.G.; Venuti, V.; Cristani, M.; Daniele, C.; Saija, A.; Mazzanti, G.; Bisignano, G. Mechanisms of antibacterial action of three monoterpenes. Antimicrob. Agents Chemother. 2005, 49, 2474–2478. [Google Scholar] [CrossRef] [PubMed]
- Crowell, P.L. Prevention and therapy of cancer by dietary monoterpenes. J. Nutr. 1999, 129, S775–S778. [Google Scholar] [CrossRef] [PubMed]
- Cazares-Franco, M.C.; Ramirez-Chimal, C.; Herrera-Hernandez, M.G.; Nunez-Colin, C.A.; Hernandez-Martinez, M.A.; Guzman-Maldonado, S.H. Physicochemical, nutritional and health-related component characterization of the underutilized mexican serviceberry fruit Malacomeles denticulata (Kunth) G. N. Jones. Fruits 2014, 69, 47–60. [Google Scholar] [CrossRef]
- Mattila, P.H.; Hellström, J.; McDougall, G.; Dobson, G.; Pihlava, J.-M.; Tiirikka, T.; Stewart, D.; Karjalainen, R. Polyphenol and vitamin c contents in European commercial blackcurrant juice products. Food Chem. 2011, 127, 1216–1223. [Google Scholar] [CrossRef] [PubMed]
- Georgé, S.; Brat, P.; Alter, P.; Amiot, M.J. Rapid determination of polyphenols and vitamin c in plant-derived products. J. Agric. Food Chem. 2005, 53, 1370–1373. [Google Scholar] [CrossRef] [PubMed]
- Donno, D.; Cerutti, A.K.; Prgomet, I.; Mellano, M.G.; Beccaro, G.L. Foodomics for mulberry fruit (Morus spp.): Analytical fingerprint as antioxidants’ and health properties’ determination tool. Food Res. Inter. 2015, 69, 179–188. [Google Scholar] [CrossRef]
- Fügel, R.; Carle, R.; Schieber, A. Quality and authenticity control of fruit purées, fruit preparations and jams—A review. Trends Food Sci. Technol. 2005, 16, 433–441. [Google Scholar] [CrossRef]
- Eyduran, S.P.; Ercisli, S.; Akin, M.; Beyhan, O.; Gecer, M.K.; Eyduran, E.; Erturk, Y.E. Organic acids, sugars, vitamin c, antioxidant capacity and phenolic compounds in fruits of white (Morus alba L.) and black (Morus nigra L.) mulberry genotypes. J. Appl. Bot. Food Qual. 2015, 88, 134–138. [Google Scholar]
- Soyer, Y.; Koca, N.; Karadeniz, F. Organic acid profile of turkish white grapes and grape juices. J. Food Compos. Anal. 2003, 16, 629–636. [Google Scholar] [CrossRef]
- Cejudo-Bastante, C.; Castro-Mejías, R.; Natera-Marín, R.; García-Barroso, C.; Durán-Guerrero, E. Chemical and sensory characteristics of orange based vinegar. J. Food Sci. Technol. 2016, 53, 3147–3156. [Google Scholar] [CrossRef] [PubMed]
- Flores, P.; Hellín, P.; Fenoll, J. Determination of organic acids in fruits and vegetables by liquid chromatography with tandem-mass spectrometry. Food Chem. 2012, 132, 1049–1054. [Google Scholar] [CrossRef]
- Chinnici, F.; Spinabelli, U.; Riponi, C.; Amati, A. Optimization of the determination of organic acids and sugars in fruit juices by ion-exclusion liquid chromatography. J. Food Compos. Anal. 2005, 18, 121–130. [Google Scholar] [CrossRef]
- Touati, N.; Tarazona-Díaz, M.P.; Aguayo, E.; Louaileche, H. Effect of storage time and temperature on the physicochemical and sensory characteristics of commercial apricot jam. Food Chem. 2014, 145, 23–27. [Google Scholar] [CrossRef] [PubMed]
- Tzouros, N.; Arvanitoyannis, I. Agricultural produces: Synopsis of employed quality control methods for the authentication of foods and application of chemometrics for the classification of foods according to their variety or geographical origin. Crit. Rev. Food Sci. Nutr. 2001, 41, 287–319. [Google Scholar] [CrossRef] [PubMed]
- Reid, L.M.; O’Donnell, C.P.; Downey, G. Potential of SPME-GC and chemometrics to detect adulteration of soft fruit purees. J. Agric. Food Chem. 2004, 52, 421–427. [Google Scholar] [CrossRef] [PubMed]
- Slinkard, K.; Singleton, V.L. Total phenol analysis: Automation and comparison with manual methods. Am. J. Enol. Vitic. 1977, 28, 49–55. [Google Scholar]
- Sánchez-Rangel, J.C.; Benavides, J.; Heredia, J.B.; Cisneros-Zevallos, L.; Jacobo-Velázquez, D.A. The Folin–Ciocalteu assay revisited: Improvement of its specificity for total phenolic content determination. Anal. Methods 2013, 5, 5990–5999. [Google Scholar] [CrossRef]
- Lee, J.; Durst, R.W.; Wrolstad, R.E. Determination of total monomeric anthocyanin pigment content of fruit juices, beverages, natural colorants, and wines by the pH differential method: Collaborative study. J. AOAC Int. 2005, 88, 1269–1278. [Google Scholar] [PubMed]
- Giusti, M.M.; Wrolstad, R.E. Characterization and measurement of anthocyanins by UV-visible spectroscopy. Curr. Protoc. Food Anal. Chem. 2001, 00, F1.2.1–F1.2.13. [Google Scholar] [CrossRef]
- Benzie, I.F.; Strain, J.J. Ferric reducing/antioxidant power assay: Direct measure of total antioxidant activity of biological fluids and modified version for simultaneous measurement of total antioxidant power and ascorbic acid concentration. Meth. Enzymol. 1999, 299, 15–27. [Google Scholar] [PubMed]
- Donno, D.; Mellano, M.G.; Prgomet, Z.; Beccaro, G.L. Advances in Ribes × nidigrolaria Rud. Bauer and A. Bauer fruits as potential source of natural molecules: A preliminary study on physico-chemical traits of an underutilized berry. Sci. Hortic. 2018, 237, 20–27. [Google Scholar] [CrossRef]
- Donno, D.; Mellano, M.G.; De Biaggi, M.; Riondato, I.; Rakotoniaina, E.N.; Beccaro, G.L. New findings in Prunus padus L. fruits as a source of natural compounds: Characterization of metabolite profiles and preliminary evaluation of antioxidant activity. Molecules 2018, 23, 725. [Google Scholar] [CrossRef] [PubMed]
- Mok, D.K.W.; Chau, F.T. Chemical information of chinese medicines: A challenge to chemist. Chemom. Intell. Lab. Syst. 2006, 82, 210–217. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are not available from the authors. |


| Total Polyphenolic Content | Antioxidant Activity | Total Anthocyanin Content | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| (mgGAE/100 gPr) | (mmol Fe2+/kgpr) | (mgC3G/100 gPr) | ||||||||
| Sample | ID | Mean Value | SD | Tukey Test | Mean Value | SD | Tukey Test | Mean Value | SD | Tukey Test |
| mango jam | CM1 | 11.11 | 1.34 | a | 4.71 | 2.07 | a | 1.48 | 0.26 | ab |
| mango jam | CM2 | 10.98 | 2.18 | a | 7.86 | 0.36 | b | 0.59 | 0.25 | a |
| tamarind jam | CM3 | 484.95 | 81.89 | d | 23.97 | 0.28 | d | 0.87 | 0.43 | ab |
| banana flower | CM4 | 13.06 | 1.85 | a | 19.23 | 1.29 | c | 4.49 | 1.62 | c |
| guava jelly | CM5 | 437.13 | 2.44 | d | 25.40 | 0.35 | d | 8.35 | 1.13 | d |
| lychee jam | CM6 | 266.96 | 7.95 | c | 23.01 | 0.07 | d | 3.15 | 0.66 | bc |
| orange jam | CM7 | 124.50 | 27.49 | b | 16.62 | 0.89 | c | 9.56 | 0.46 | d |
| red guava jam | CM8 | 625.34 | 67.86 | e | 25.50 | 0.28 | d | 4.99 | 0.67 | c |
| Mango Jam, CM1 | Mango Jam, CM2 | Tamarind Jam, CM3 | Banana Flower, CM4 | Guava Jelly, CM5 | Lychee Jam, CM6 | Orange Jam, CM7 | Red Guava Jam, CM8 | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mean Value | SD | Mean Value | SD | Mean Value | SD | Mean Value | SD | Mean Value | SD | Mean Value | SD | Mean Value | SD | Mean Value | SD | ||
| Cinnamic acids | caffeic acid | 0.678 | 0.170 | 0.761 | 0.101 | 0.498 | 0.134 | 0.749 | 0.154 | 0.687 | 0.034 | 0.764 | 0.116 | 0.571 | 0.110 | 0.744 | 0.204 |
| chlorogenic acid | n.d. | / | n.d. | / | 11.454 | 0.731 | n.d. | / | 13.766 | 0.546 | n.d. | / | 12.971 | 0.185 | n.d. | / | |
| coumaric acid | n.d. | / | n.d. | / | n.d. | / | 0.556 | 0.509 | n.d. | / | n.d. | / | n.d. | / | 5.583 | 0.797 | |
| ferulic acid | n.d. | / | n.d. | / | 1.377 | 0.217 | n.d. | / | n.d. | / | n.d. | / | 1.001 | 0.110 | n.d. | / | |
| Flavonols | hyperoside | n.d. | / | n.d. | / | n.d. | / | 0.356 | 0.033 | n.d. | / | 1.027 | 0.116 | n.d. | / | n.d. | / |
| isoquercitrin | n.d. | / | n.d. | / | 0.491 | 0.006 | n.d. | / | 0.525 | 0.034 | n.d. | / | 1.140 | 0.110 | 0.541 | 0.155 | |
| quercetin | n.d. | / | n.d. | / | 11.441 | 0.963 | n.d. | / | 7.562 | 0.034 | n.d. | / | 15.694 | 1.107 | 7.621 | 0.204 | |
| quercitrin | 0.161 | 0.030 | 0.007 | 0.003 | 1.640 | 0.257 | 0.173 | 0.019 | 1.001 | 0.034 | 1.334 | 0.116 | 1.152 | 0.110 | 1.477 | 0.256 | |
| rutin | 0.936 | 0.036 | 0.858 | 0.034 | n.d. | / | n.d. | / | n.d. | / | n.d. | / | n.d. | / | n.d. | / | |
| Benzoic acids | ellagic acid | 1.167 | 0.264 | 1.228 | 0.154 | 23.302 | 1.962 | 1.604 | 0.365 | 4.444 | 0.316 | 3.799 | 0.116 | 57.105 | 1.637 | 4.561 | 0.607 |
| gallic acid | 0.668 | 0.105 | 1.535 | 0.232 | 8.571 | 0.314 | 0.611 | 0.070 | 42.023 | 0.582 | 30.650 | 0.886 | 87.724 | 1.812 | 40.955 | 1.727 | |
| Catechins | catechin | 0.872 | 0.023 | 1.749 | 0.175 | 40.644 | 1.558 | 1.216 | 0.162 | 0.327 | 0.034 | n.d. | / | 0.436 | 0.110 | 4.287 | 0.204 |
| epicatechin | 3.683 | 0.328 | 1.747 | 0.400 | n.d. | / | 1.348 | 0.184 | 2.523 | 0.034 | n.d. | / | 2.015 | 0.110 | 4.031 | 0.157 | |
| Tannins | castalagin | n.d. | / | n.d. | / | n.d. | / | 2.595 | 0.425 | 4.384 | 0.034 | n.d. | / | 15.210 | 2.414 | 9.773 | 0.295 |
| vescalagin | 3.639 | 0.174 | 0.359 | 0.008 | n.d. | / | 1.554 | 0.371 | 4.339 | 0.205 | n.d. | / | 3.031 | 0.110 | 10.012 | 0.204 | |
| Mango Jam, CM1 | Mango Jam, CM2 | Tamarind Jam, CM3 | Banana Flower, CM4 | Guava Jelly, CM5 | Lychee Jam, CM6 | Orange Jam, CM7 | Red Guava Jam, CM8 | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mean Value | SD | Mean Value | SD | Mean Value | SD | Mean Value | SD | Mean Value | SD | Mean Value | SD | Mean Value | SD | Mean Value | SD | ||
| Monoterpenes | limonene | 103.529 | 3.116 | 31.201 | 0.588 | n.d. | / | n.d. | / | 102.750 | 0.582 | 33.602 | 0.687 | 18.818 | 0.472 | 51.319 | 3.204 |
| phellandrene | 27.843 | 2.380 | 7.809 | 0.540 | 7.497 | 0.219 | n.d. | / | 4.671 | 0.034 | 4.668 | 0.116 | n.d. | / | 4.946 | 0.204 | |
| sabinene | 8.140 | 0.045 | 12.187 | 1.146 | 11.273 | 1.180 | n.d. | / | 12.847 | 1.020 | 12.811 | 0.687 | n.d. | / | 12.332 | 0.797 | |
| γ-terpinene | 24.552 | 2.464 | 48.067 | 2.301 | n.d. | / | 17.949 | 2.097 | 39.002 | 0.034 | 19.069 | 0.492 | 37.084 | 0.505 | 43.005 | 0.155 | |
| terpinolene | n.d. | / | n.d. | / | 26.944 | 2.718 | 8.228 | 0.213 | 7.731 | 0.034 | 7.473 | 0.886 | 7.926 | 0.110 | 7.715 | 0.564 | |
| Vitamin C | ascorbic acid | 15.058 | 0.362 | 10.178 | 0.072 | 20.397 | 0.117 | 49.699 | 0.356 | 18.074 | 0.138 | 18.978 | 0.057 | 54.062 | 0.074 | 19.104 | 0.487 |
| dehydroascorbic acid | 0.813 | 0.130 | 1.815 | 0.163 | 3.767 | 0.283 | 3.123 | 0.213 | 1.138 | 0.192 | 1.196 | 0.225 | 32.003 | 0.401 | 1.279 | 0.234 | |
| Mango Jam, CM1 | Mango Jam, CM2 | Tamarind Jam, CM3 | Banana Flower, CM4 | Guava Jelly, CM5 | Lychee Jam, CM6 | Orange Jam, CM7 | Red Guava Jam, CM8 | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mean Value | SD | Mean Value | SD | Mean Value | SD | Mean Value | SD | Mean Value | SD | Mean Value | SD | Mean Value | SD | Mean Value | SD | ||
| Organic acids | citric acid | 16.217 | 4.058 | 29.696 | 0.469 | 522.583 | 2.566 | 694.580 | 3.627 | 326.950 | 3.030 | 268.422 | 2.629 | 287.911 | 4.614 | 200.408 | 2.204 |
| malic acid | 357.878 | 3.679 | 302.528 | 2.531 | n.d. | / | 21.926 | 1.696 | n.d. | / | n.d. | / | n.d. | / | n.d. | / | |
| oxalic acid | 4.741 | 0.198 | 4.573 | 0.416 | 21.444 | 0.530 | 8.441 | 0.102 | 8.194 | 0.117 | n.d. | / | 6.629 | 0.110 | 23.717 | 0.797 | |
| quinic acid | n.d. | / | n.d. | / | n.d. | / | n.d. | / | n.d. | / | 138.233 | 1.263 | 391.128 | 2.192 | n.d. | / | |
| succinic acid | n.d. | / | n.d. | / | n.d. | / | n.d. | / | n.d. | / | n.d. | / | 406.648 | 4.401 | n.d. | / | |
| tartaric acid | n.d. | / | n.d. | / | n.d. | / | n.d. | / | 76.724 | 1.513 | 105.358 | 4.157 | 193.497 | 3.661 | 134.637 | 1.729 | |
| Sugars | fructose | 11.701 | 0.038 | 11.316 | 0.100 | 15.353 | 0.140 | 12.275 | 0.287 | 21.341 | 0.034 | 14.814 | 0.116 | 13.620 | 0.110 | 14.428 | 0.204 |
| glucose | 14.505 | 0.321 | 13.620 | 0.401 | 27.637 | 2.326 | 12.342 | 0.111 | 12.615 | 0.034 | 11.044 | 0.116 | 13.090 | 0.110 | 13.413 | 0.155 | |
| sucrose | 13.444 | 0.339 | 14.831 | 0.680 | 13.509 | 0.231 | 11.131 | 1.084 | 11.801 | 0.867 | 14.573 | 0.492 | 14.935 | 0.110 | 14.904 | 0.157 | |
| Method | Compounds of Interest | Stationary Phase | Mobile Phase | Flow (mL min−1) | Wavelength (nm) |
|---|---|---|---|---|---|
| A | cinnamic acids, flavanols | KINETEX-C18 column (4.6 × 150 mm, 5 μm) | A: 10 mM KH2PO4/H3PO4, pH = 2.8 | 1.5 | 330 |
| B: CH3CN | |||||
| B | benzoic acids, catechins, | KINETEX-C18 column (4.6 × 150 mm, 5 μm) | A: H2O/CH3OH/HCOOH (5:95:0.1 v/v/v), pH = 2.5 | 0.6 | 280 |
| tannins | B: CH3OH/HCOOH (100:0.1 v/v) | ||||
| C | monoterpenes | KINETEX-C18 column (4.6 × 150 mm, 5 μm) | A: H2O | 1.0 | 210, 220, |
| B: CH3CN | 235, 250 | ||||
| D | organic acids | KINETEX-C18 column (4.6 × 150 mm, 5 μm) | A: 10 mM KH2PO4/H3PO4, pH = 2.8 | 0.6 | 214 |
| B: CH3CN | |||||
| E | vitamins | KINETEX-C18 column (4.6 × 150 mm, 5 μm) | A: 5 mM C16H33N(CH3)3Br/50 mM KH2PO4, pH = 2.5 | 0.9 | 261, 348 |
| B: CH3OH | |||||
| F | sugars | SphereClone-NH2 column (4.6 × 250 mm, 5 μm) | A: H2O | 0.5 | 200, 267, |
| B: CH3CN | 286 |
| Class | Standard | Calibration Curve Equation | R2 | Calibration Curve Range (mg L−1) |
|---|---|---|---|---|
| Cinnamic acids | caffeic acid | y = 59.046x + 200.6 | 0.996 | 111–500 |
| chlorogenic acid | y = 13.583x + 760.05 | 0.984 | 111–500 | |
| coumaric acid | y = 8.9342x + 217.4 | 0.997 | 111–500 | |
| ferulic acid | y = 3.3963x − 4.9524 | 1.000 | 111–500 | |
| Flavonols | hyperoside | y = 7.1322x − 4.583 | 0.999 | 111–500 |
| isoquercitrin | y = 8.3078x + 26.621 | 0.999 | 111–500 | |
| quercetin | y = 3.4095x − 98.307 | 0.998 | 111–500 | |
| quercitrin | y = 2.7413x + 5.6367 | 0.998 | 111–500 | |
| rutin | y = 6.5808x + 30.831 | 0.999 | 111–500 | |
| Benzoic acids | ellagic acid | y = 29.954x + 184.52 | 0.998 | 62.5–250 |
| gallic acid | y = 44.996x + 261.86 | 0.999 | 62.5–250 | |
| Catechins | catechin | y = 8.9197x + 66.952 | 1.000 | 62.5–250 |
| epicatechin | y = 12.88x − 43.816 | 0.999 | 62.5–250 | |
| Tannins | castalagin | y = 4.236x − 8.535 | 1.000 | 62.5–250 |
| vescalagin | y = 4.939x − 1.232 | 1.000 | 62.5–250 | |
| Monoterpenes | limonene | y = 0.1894x − 5.420 | 0.999 | 125–1000 |
| phellandrene | y = 8.783x − 145.3 | 0.998 | 125–1000 | |
| sabinene | y = 18.14x − 1004 | 0.998 | 125–1000 | |
| γ-terpinene | y = 0.4886x − 23.02 | 0.999 | 125–1000 | |
| terpinolene | y = 26.52x + 876.8 | 0.999 | 125–1000 | |
| Organic acids | citric acid | y = 1.0603x − 22.092 | 1.000 | 167–1000 |
| malic acid | y = 1.415x − 80.254 | 0.996 | 167–1000 | |
| oxalic acid | y = 6.4502x + 6.1503 | 0.998 | 167–1000 | |
| quinic acid | y = 0.8087x − 38.021 | 0.998 | 167–1000 | |
| succinic acid | y = 0.9236x − 8.0823 | 0.995 | 167–1000 | |
| tartaric acid | y = 1.8427x + 15.796 | 1.000 | 167–1000 | |
| Vitamins | ascorbic acid | y = 42.71x + 27.969 | 0.999 | 100–1000 |
| dehydroascorbic acid | y = 4.1628x + 140.01 | 0.999 | 30–300 | |
| Sugar | fructose | y = 1.8548x + 1.2324 | 0.999 | 125–1000 |
| glucose | y = 0.1269x − 0.1107 | 0.998 | 125–1000 | |
| sucrose | y = 0.296x − 3.2202 | 1.000 | 125–1000 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Donno, D.; Mellano, M.G.; Hassani, S.; De Biaggi, M.; Riondato, I.; Gamba, G.; Giacoma, C.; Beccaro, G.L. Assessing Nutritional Traits and Phytochemical Composition of Artisan Jams Produced in Comoros Islands: Using Indigenous Fruits with High Health-Impact as an Example of Biodiversity Integration and Food Security in Rural Development. Molecules 2018, 23, 2707. https://doi.org/10.3390/molecules23102707
Donno D, Mellano MG, Hassani S, De Biaggi M, Riondato I, Gamba G, Giacoma C, Beccaro GL. Assessing Nutritional Traits and Phytochemical Composition of Artisan Jams Produced in Comoros Islands: Using Indigenous Fruits with High Health-Impact as an Example of Biodiversity Integration and Food Security in Rural Development. Molecules. 2018; 23(10):2707. https://doi.org/10.3390/molecules23102707
Chicago/Turabian StyleDonno, Dario, Maria Gabriella Mellano, Saandia Hassani, Marta De Biaggi, Isidoro Riondato, Giovanni Gamba, Cristina Giacoma, and Gabriele Loris Beccaro. 2018. "Assessing Nutritional Traits and Phytochemical Composition of Artisan Jams Produced in Comoros Islands: Using Indigenous Fruits with High Health-Impact as an Example of Biodiversity Integration and Food Security in Rural Development" Molecules 23, no. 10: 2707. https://doi.org/10.3390/molecules23102707
APA StyleDonno, D., Mellano, M. G., Hassani, S., De Biaggi, M., Riondato, I., Gamba, G., Giacoma, C., & Beccaro, G. L. (2018). Assessing Nutritional Traits and Phytochemical Composition of Artisan Jams Produced in Comoros Islands: Using Indigenous Fruits with High Health-Impact as an Example of Biodiversity Integration and Food Security in Rural Development. Molecules, 23(10), 2707. https://doi.org/10.3390/molecules23102707