Chemical Components and Biological Activities of the Genus Phyllanthus: A Review of the Recent Literature
Abstract
:1. Introduction
2. Chemical Components
2.1. Phenylpropanoids
2.2. Terpenoids
2.3. Phenolic Compounds
2.4. Flavonoids
2.5. Alkaloids
2.6. Other Compounds
3. Biological Activities
3.1. Parasitology
3.2. Cardiovascular Protection
3.3. Antioxidant Activity
3.4. Anticancer Activity
3.5. Anti-Aging and Skin Protection Properties
3.6. Antidiabetic Activity
3.7. Organ Protective Effects
3.8. Diuretic Effects
3.9. Brain Functions
3.10. Analgesic and Anti-Inflammatory Activity
3.11. Immunomodulatory Effects
3.12. Antibacterial Activities
3.13. Antiviral Activity
3.14. Miscellaneous
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Haque, T.; Muhsin, M.D.A.; Akhter, T.; Haq, M.E.; Begum, R.; Chowdhury, S.F.U.A. Antimicrobial and analgesic activity of leaf extracts of Phyllanthus reticulatus Poir. (Family-Euphorbiaceae). Jahangirnagar Univ. J. Biol. Sci. 2016, 5, 81–85. [Google Scholar] [CrossRef]
- Petrovska, B.B. Historical review of medicinal plants’ usage. Pharmacogn. Rev. 2012, 6, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Aboelsoud, N.H. Herbal medicine in ancient Egypt. J. Med. Plants Res. 2010, 4, 82–86. [Google Scholar]
- Gismondi, A.; D’Agostino, A.; Canuti, L.; Di Marco, G.; Martínez-Labarga, C.; Angle, M.; Rickard, O.; Canini, A. Dental calculus reveals diet habits and medicinal plant use in the Early Medieval Italian population of Colonna. J. Archaeol. Sci. Rep. 2018, 20, 556–564. [Google Scholar] [CrossRef]
- Manjula, V.; Norman, T.S.J. Phyllanthus reticulatus for oral health. J. Med. Plants 2017, 5, 117–119. [Google Scholar]
- De Oliveira, C.N.F.; Frezza, T.F.; Garcia, V.L.; Figueira, G.M.; Mendes, T.M.F.; Allegretti, S.M. Schistosoma mansoni: In vivo evaluation of Phyllanthus amarus hexanic and ethanolic extracts. Exp. Parasitol. 2017, 183, 56–63. [Google Scholar] [CrossRef] [PubMed]
- Mao, X.; Wu, L.F.; Guo, H.L.; Chen, W.J.; Cui, Y.P.; Qi, Q.; Li, S.; Liang, W.Y.; Yang, G.H.; Shao, Y.Y. The genus Phyllanthus: An ethnopharmacological, phytochemical, and pharmacological review. Evid. Based Complement. Altern. 2016, 2016, 1–36. [Google Scholar] [CrossRef]
- Devi, S.; Kumar, D.; Kumar, M. In-Vitro antioxidant activities of methanolic extract of whole plant of Phyllanthus amarus (Euphorbiaceae). Int. J. Bot. Stud. 2016, 1, 30–32. [Google Scholar]
- Devi, S.; Kumar, M. In vitro Antioxidant potential of methanolic extract of whole plant of Phyllanthus amarus Schum (Euphorbiaceae). Int. J. Bot. Stud. 2017, 2, 100–102. [Google Scholar]
- Kaur, B.; Kaur, N.; Gautam, V. Evaluation of anti-helicobacter pylori (DSMZ 10242) activity and qualitative analysis of quercetin by HPLC in Phyllanthus niruri linn. World J. Pharm. Pharm. Sci. 2016, 5, 1691–1706. [Google Scholar]
- Nguyen, V.T.; Pham, H.N.T.; Bowyer, M.C.; van Altena, I.A.; Scarlett, C.J. Influence of solvents and novel extraction methods on bioactive compounds and antioxidant capacity of Phyllanthus amarus. Chem. Pap. 2016, 70, 556–566. [Google Scholar] [CrossRef]
- Zhang, J.; Miao, D.; Zhu, W.F.; Xu, J.; Liu, W.Y.; Kitdamrongtham, W.; Manosroi, J.; Abe, M.; Akihisa, T.; Feng, F. Biological activities of phenolics from the fruits of Phyllanthus emblica L. (Euphorbiaceae). Chem. Biodivers. 2017, 14, e1700404. [Google Scholar] [CrossRef] [PubMed]
- Tung, Y.-T.; Huang, C.-Z.; Lin, J.-H.; Yen, G.-C. Effect of Phyllanthus emblica L. fruit on methionine and choline-deficiency diet-induced nonalcoholic steatohepatitis. J. Food Drug Anal. 2018, 26, 1245–1252. [Google Scholar] [CrossRef] [PubMed]
- Boakye-Gyasi, E.; Kasange, E.A.; Biney, R.P.; Boadu-Mensah, K.; Agyare, C.; Woode, E. Anti-nociceptive effects of geraniin and an aqueous extract of the aerial parts of Phyllanthus muellerianus (Kuntze) Exell. in murine models of chemical nociception. Iran. J. Pharm. Sci. 2016, 12, 17–30. [Google Scholar]
- Mostofa, R.; Ahmed, S.; Begum, M.M.; Sohanur Rahman, M.; Begum, T.; Ahmed, S.U.; Tuhin, R.H.; Das, M.; Hossain, A.; Sharma, M.; et al. Evaluation of anti-inflammatory and gastric anti-ulcer activity of Phyllanthus niruri L. (Euphorbiaceae) leaves in experimental rats. BMC Complement. Altern. Med. 2017, 17, 267. [Google Scholar] [CrossRef] [PubMed]
- Menéndez-Perdomo, I.M.; Wong-Guerra, M.; Fuentes-León, F.; Carrazana, E.; Casadelvalle, I.; Vidal, A.; Sánchez-Lamar, Á. Antioxidant, photoprotective and antimutagenic properties of Phyllanthus spp. from Cuban flora. J. Pharm. Pharmacogn. Res. 2017, 5, 251–261. [Google Scholar]
- Guo, X.; Wang, X. Phyllanthus emblica Fruit Extract Activates Spindle Assembly Checkpoint, Prevents Mitotic Aberrations and Genomic Instability in Human Colon Epithelial NCM460 Cells. Int. J. Mol. Sci. 2016, 17, 1437. [Google Scholar] [CrossRef] [PubMed]
- Noorudheen, N.; Chandrasekharan, D.K. Effect of ethanolic extract of Phyllanthus emblica on captan induced oxidative stress in vivo. South Indian J. Biol. Sci. 2016, 2, 95–102. [Google Scholar] [CrossRef]
- Guo, X.H.; Ni, J.; Xue, J.L.; Wang, X. Phyllanthus emblica Linn. fruit extract potentiates the anticancer efficacy of mitomycin C and cisplatin and reduces their genotoxicity to normal cells in vitro. J. Zhejiang Univ. Sci. B 2017, 18, 1031–1045. [Google Scholar] [CrossRef] [PubMed]
- Jagtap, S.; Khare, P.; Mangal, P.; Kondepudi, K.K.; Bishnoi, M.; Bhutani, K.K. Protective effects of phyllanthin, a lignan from Phyllanthus amarus, against progression of high fat diet induced metabolic disturbances in mice. RSC Adv. 2016, 6, 58343–58353. [Google Scholar] [CrossRef]
- Lee, N.Y.; Khoo, W.K.; Adnan, M.A.; Mahalingam, T.P.; Fernandez, A.R.; Jeevaratnam, K. The pharmacological potential of Phyllanthus niruri. J. Pharm. Pharmacol. 2016, 68, 953–969. [Google Scholar] [CrossRef] [PubMed]
- Yang, B.; Kortesniemi, M.; Liu, P.; Karonen, M.; Salminen, J.P. Analysis of hydrolyzable tannins and other phenolic compounds in emblic leafflower (Phyllanthus emblica L.) fruits by high performance liquid chromatography–electrospray ionization mass spectrometry. J. Agric. Food Chem. 2012, 60, 8672–8683. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.; Lai, Y.; Zhou, L.; Wu, Y.; Zhu, H.; Hu, Z.; Yang, J.; Zhang, J.; Wang, J.; Luo, Z.; et al. Enantiomeric lignans and neolignans from Phyllanthus glaucus: Enantioseparation and their absolute configurations. Sci. Rep. 2016, 6, 24809. [Google Scholar] [CrossRef] [PubMed]
- Muthusamy, A.; Prasad, H.N.N.; Sanjay, E.R.; Rao, M.R.; Satyamoorthy, K. Impact of precursors and plant growth regulators on in vitro growth, bioactive lignans, and antioxidant content of Phyllanthus species. In Vitro Cell. Dev. Plant 2016, 52, 598–607. [Google Scholar] [CrossRef]
- Pereira, R.G.; Garcia, V.L.; Rodrigues, M.V.N.; Martínez, J. Extraction of lignans from Phyllanthus amarus Schum. & Thonn using pressurized liquids and low pressure methods. Sep. Purif. Technol. 2016, 158, 204–211. [Google Scholar]
- Pereira, R.G.; Nakamura, R.N.; Rodrigues, M.V.N.; Osorio-Tobón, J.F.; Garcia, V.L.; Martinez, J. Supercritical fluid extraction of phyllanthin and niranthin from Phyllanthus amarus Schum. & Thonn. J. Supercrit. Fluids 2017, 127, 23–32. [Google Scholar]
- Borges, L.D.C.; Negrão-Neto, R.; Pamplona, S.; Fernandes, L.; Barros, M.; Fontes-Júnior, E.; Maia, C.; Silva, C.Y.Y.; Silva, M.N.D. Anti-Inflammatory and Antinociceptive Studies of Hydroalcoholic Extract from the Leaves of Phyllanthus brasiliensis (Aubl.) Poir. and Isolation of 5-O-β-d-Glucopyranosyljusticidin B and Six Other Lignans. Molecules 2018, 23, 941. [Google Scholar] [CrossRef] [PubMed]
- Fan, Y.Y.; Gan, L.S.; Liu, H.C.; Li, H.; Xu, C.H.; Zuo, J.P.; Ding, J.; Yue, J.M.; Phainanolide, A. Highly modified and oxygenated triterpenoid from Phyllanthus hainanensis. Org. Lett. 2017, 19, 4580–4583. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Xie, S.S.; Hu, Z.X.; Wu, Z.D.; Guo, Y.; Zhang, J.W.; Wang, J.P.; Xue, Y.B. Triterpenoids from Whole Plants of Phyllanthus urinaria. Chin. Herb. Med. 2017, 9, 193–196. [Google Scholar] [CrossRef]
- Chung, C.Y.; Liu, C.H.; Burnouf, T.; Wang, G.H.; Chang, S.P.; Jassey, A.; Tai, C.J.; Tai, C.J.; Huang, C.J.; Richardson, C.D.; et al. Activity-based and fraction-guided analysis of Phyllanthus urinaria identifies loliolide as a potent inhibitor of hepatitis C. virus entry. Antivir. Res. 2016, 130, 58–68. [Google Scholar] [CrossRef] [PubMed]
- Duong, T.H.; Bui, X.H.; Pogam, P.L.; Nguyen, H.H.; Tran, T.T.; Nguyen, T.A.T.; Chavasiri, W.; Boustie, J.; Nguyen, K.P.P. Two novel diterpenes from the roots of Phyllanthus acidus (L.) Skeel. Tetrahedron 2017, 73, 5634–5638. [Google Scholar] [CrossRef]
- Zheng, X.H.; Yang, J.; Lv, J.J.; Zhu, H.T.; Wang, D.; Xu, M.; Yang, C.R.; Zhang, Y.J. Phyllaciduloids A-D: Four new cleistanthane diterpenoids from Phyllanthus acidus (L.) Skeels. Fitoterapia 2018, 125, 89–93. [Google Scholar] [CrossRef] [PubMed]
- Yan, H.; Han, L.R.; Zhang, X.; Feng, J.T. Two new Anti-TMV active chalconoid analogues from the root of Phyllanthus emblica. Nat. Prod. Res. 2017, 31, 2143–2148. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Liu, T.T.; Chen, G. An effective β-cyclodextrin polyurethane spherical adsorbent for the chromatographic enrichment of corilagin from Phyllanthus niruri L. extract. React. Funct. Polym. 2016, 102, 119–129. [Google Scholar] [CrossRef]
- Tram, N.C.T.; Son, N.T.; Thao, D.T.; Cuong, N.M. Kaempferol and kaempferol glycosides from Phyllanthus acidus leaves. Vietnam J. Chem. 2016, 54, 790–793. [Google Scholar]
- Putakala, M.; Gujjala, S.; Nukala, S.; Bongu, S.B.R.; Chintakunta, N.; Desireddy, S. Cardioprotective effect of Phyllanthus amarus against high fructose diet induced myocardial and aortic stress in rat model. Biomed. Pharmacother. 2017, 95, 1359–1368. [Google Scholar] [CrossRef] [PubMed]
- Komlaga, G.; Genta-Jouve, G.; Cojean, S.; Dickson, R.A.; Mensah, M.L.; Loiseau, P.M.; Champy, P.; Beniddir, M.A. Antiplasmodial Securinega alkaloids from Phyllanthus fraternus: Discovery of natural (+)-allonorsecurinine. Tetrahedron Lett. 2017, 58, 3754–3756. [Google Scholar] [CrossRef]
- Zeng, Z.; Lv, W.; Jing, Y.; Chen, Z.; Song, L.; Liu, T.; Yu, R. Structural characterization and biological activities of a novel polysaccharide from Phyllanthus emblica. Drug Discov. Ther. 2017, 11, 54–63. [Google Scholar] [CrossRef] [PubMed]
- Navarro, M.; Moreira, I.; Arnaez, E.; Quesada, S.; Azofeifa, G.; Alvarado, D.; Monagas, M.J. Proanthocyanidin Characterization, Antioxidant and Cytotoxic Activities of Three Plants Commonly Used in Traditional Medicine in Costa Rica: Petiveria alliaceae L., Phyllanthus niruri L. and Senna reticulata Willd. Plants 2017, 6, 50. [Google Scholar] [CrossRef] [PubMed]
- Navarro, M.; Moreira, I.; Arnaez, E.; Quesada, S.; Azofeifa, G.; Vargas, F.; Alvarado, D.; Chen, P. Flavonoids and Ellagitannins Characterization, Antioxidant and Cytotoxic Activities of Phyllanthus acuminatus Vahl. Plants 2017, 6, 62. [Google Scholar] [CrossRef] [PubMed]
- Laulloo, S.J.; Bhowon, M.; Chua, L.; Gaungoo, H. Phytochemical Screening and Antioxidant Properties of Phyllanthus emblica from Mauritius. Chem. Nat. Compd. 2018, 54, 50–55. [Google Scholar] [CrossRef]
- Kumar, S.; Singh, A.; Bajpai, V.; Singh, B.; Kumar, B. Development of a UHPLC–MS/MS method for the quantitation of bioactive compounds in Phyllanthus species and its herbal formulations. J. Sep. Sci. 2017, 40, 3422–3429. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Singh, A.; Kumar, B. Identification and characterization of phenolics and terpenoids from ethanolic extracts of Phyllanthus species by HPLC-ESI-QTOF-MS/MS. J. Pharm. Anal. 2017, 7, 214–222. [Google Scholar] [CrossRef] [PubMed]
- Ghafar, S.Z.A.; Mediani, A.; Ramli, N.S.; Abas, F. Antioxidant, α-glucosidase, and nitric oxide inhibitory activities of Phyllanthus acidus and LC–MS/MS profile of the active extract. Food Biosci. 2018, 25, 134–140. [Google Scholar] [CrossRef]
- Ojezele, M.O.; Moke, E.G.; Onyesom, I. Impact of generic antimalarial or Phyllanthus amarus and vitamin co-administration on antioxidant status of experimental mice infested with Plasmodium berghei. Beni-Suef Univ. J. Basic Appl. Sci. 2017, 6, 260–265. [Google Scholar] [CrossRef]
- Peter, S.; Dey, S.; Veerakyathappa, B.; Kumar, S.R.; Paulad, C. Therapeutic activity of Partially purified fractions of Emblica officinalis (Syn. Phyllanthus emblica) dried fruits against Trypanosoma evansi. J. Pharm. Pharmacol. 2016, 4, 546–558. [Google Scholar]
- Chansriniyom, C.; Bunwatcharaphansakun, P.; Eaknai, W.; Nalinratana, N.; Ratanawong, A.; Khongkow, M.; Luechapudiporn, R. A synergistic combination of Phyllanthus emblica and Alpinia galanga against H2O2 -induced oxidative stress and lipid peroxidation in human ECV304 cells. J. Funct. Foods 2018, 43, 44–54. [Google Scholar] [CrossRef]
- Akporowhe, S.; Onyesom, I. Phyllanthus amarus augments the serum antioxidant capacity and invigorates the blood in experimental mice. Biosci. Biotechnol. Res. Commun. 2016, 9, 15–18. [Google Scholar]
- Rusmana, D.; Wahyudianingsih, R.; Elisabeth, M.; Balqis, B.; Maesaroh, M.; Widowati, W. Antioxidant Activity of Phyllanthus niruri Extract, Rutin and Quercetin. Indones. Biomed. J. 2017, 9, 84–90. [Google Scholar] [CrossRef]
- Singh, R.P.; Pal, A.; Pal, K. Antioxidant activity of ethanolic and aqueous extract of Phyllanthus niruri—In vitro. World J. Pharm. Pharm. Sci. 2016, 5, 1994–2000. [Google Scholar]
- Angamuthu, J.; Ganapathy, M.; Evanjelene, V.K. Evaluation of antioxidant activity of Phyllanthus acidus. World J. Pharm. Pharm. Sci. 2016, 5, 1011–1016. [Google Scholar]
- Nguyen, V.T.; Sakoff, J.A.; Scarlett, C.J. Physicochemical Properties, Antioxidant and Cytotoxic Activities of Crude Extracts and Fractions from Phyllanthus amarus. Medicines 2017, 4, 42. [Google Scholar] [CrossRef] [PubMed]
- Ramandeep, K.; Nahid, A.; Neelabh, C.; Navneet, K. Phytochemical Screening of Phyllanthus niruri collected from Kerala Region and its Antioxidant and Antimicrobial Potentials. J. Pharm. Sci. Res. 2017, 9, 1312–1316. [Google Scholar]
- Dai, M.; Wahyuni, A.; Dk, I.; Azizah, T.; Suhendi, A.; Saifudin, A. Antioxidant activity of Phyllanthus niruri L. herbs: In vitro and in vivo models and isolation of active compound. Nat. J. Physiol. Pharm. Pharmacol. 2016, 6, 32–37. [Google Scholar] [CrossRef]
- Andrianto, D.; Widianti, W.; Bintang, M. Antioxidant and Cytotoxic Activity of Phyllanthus acidus Fruit Extracts. IOP Conf. Ser. Earth Environ. Sci. 2017, 58, 012022. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhao, L.; Guo, X.; Li, C.; Li, H.; Lou, H.; Ren, D. Chemical constituents from Phyllanthus emblica and the cytoprotective effects on H2O2-induced PC12 cell injuries. Arch. Pharm. Res. 2014, 39, 1202–1211. [Google Scholar] [CrossRef] [PubMed]
- Kalpana, S.; Ramakrushna, B.; Anitha, S. Evaluation of in vitro antioxidant and α-amylase inhibitory activity of Phyllanthus indofischeri Bennet. Int. J. Pharm. Pharm. Sci. 2016, 8, 131–136. [Google Scholar]
- Sabir, S.M.; Shah, R.H.; Shah, A.H. Total Phenolic and Ascorbic Acid Contents and Antioxidant Activities of Twelve Different Ecotypes of Phyllanthus emblica from Pakistan. Chiang Mai J. Sci. 2017, 44, 904–911. [Google Scholar]
- Tahir, I.; Khan, M.R.; Shah, N.A.; Aftab, M. Evaluation of phytochemicals, antioxidant activity and amelioration of pulmonary fibrosis with Phyllanthus emblica leaves. BMC Complement. Altern. Med. 2016, 16, 406. [Google Scholar] [CrossRef] [PubMed]
- Boakye, Y.D.; Agyare, C.; Dapaah, S.O. In vitro and in vivo antioxidant properties of Phyllanthus muellerianus and its major constituent, geraniin. Oxid. Antioxid. Med. Sci. 2016, 5, 70–78. [Google Scholar] [CrossRef]
- Huang, S.T.; Huang, C.C.; Sheen, J.M.; Lin, T.K.; Liao, P.L.; Huang, W.L.; Wang, P.W.; Liou, C.W.; Chuang, J.H. Phyllanthus urinaria’s Inhibition of Human Osteosarcoma Xenografts Growth in Mice is Associated with Modulation of Mitochondrial Fission/Fusion Machinery. Am. J. Chin. Med. 2016, 44, 1507–1523. [Google Scholar] [CrossRef] [PubMed]
- Perera, D.; Soysa, P.; Wijeratne, S. Polyphenols contribute to the antioxidant and antiproliferative activity of Phyllanthus debilis plant in-vitro. BMC Complement. Altern. Med. 2016, 16, 339. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Z.Z.; Chen, L.H.; Liu, S.S.; Deng, Y.; Zheng, G.H.; Gu, Y.; Ming, Y.L. Bioguided Fraction and Isolation of the Antitumor Components from Phyllanthus niruri L. Biomed. Res. Int. 2016, 2016, 9729275. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.R.; Chen, Q.Q.; Lam, C.W.; Wang, C.Y.; Xu, F.G.; Liu, B.M.; Zhang, W. Effect of Phyllanthus amarus Extract on 5-Fluorouracil-Induced Perturbations in Ribonucleotide and Deoxyribonucleotide Pools in HepG2 Cell Line. Molecules 2016, 21, 1254. [Google Scholar] [CrossRef] [PubMed]
- Sawitri, E. Apoptosis of Colorectal Cancer Cell on Sprague-Dawley Rats Induced with 1, 2 Dimethylhidrazine and Phyllanthus niruri Linn Extrac. Int. J. Sci. Eng. 2016, 10, 45–50. [Google Scholar]
- Wang, C.C.; Yuan, J.R.; Wang, C.F.; Yang, N.; Chen, J.; Liu, D.; Song, J.; Feng, L.; Tan, X.B.; Jia, X.B. Anti-inflammatory Effects of Phyllanthus emblica L. on Benzopyrene-Induced Precancerous Lung Lesion by Regulating the IL-1beta/miR-101/Lin28B Signaling Pathway. Integr. Cancer Ther. 2017, 16, 505–515. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Wang, H. Phyllanthus emblica L. extract activates Nrf2 signalling pathway in HepG2 cells. Biomed. Res. 2017, 28, 3383–3386. [Google Scholar]
- Desai, K.; Braganza, V. Cytotoxic activity and phytochemical investigation of Phyllanthus emblica L. leaves. Int. J. Pharm. Sci. Res. 2016, 7, 2015–2019. [Google Scholar]
- Stefanowicz-Hajduk, J.; Sparzak-Stefanowska, B.; Krauze-Baranowska, M.; Ochocka, J.R. Securinine from Phyllanthus glaucus Induces Cell Cycle Arrest and Apoptosis in Human Cervical Cancer HeLa Cells. PLoS ONE 2016, 11, e0165372. [Google Scholar] [CrossRef] [PubMed]
- Vernhes Tamayo, M.; Schuch, A.P.; Yagura, T.; Baly Gil, L.; Menck, C.F.M.; Sánchez-Lamar, A. Genoprotective effect of Phyllanthus orbicularis extract against UVA, UVB and solar radiation. Photochem. Photobiol. 2018, 94, 1026–1031. [Google Scholar] [CrossRef] [PubMed]
- Tansirikongkol, A. Comparative in vitro anti-aging activities of Phyllanthus emblica L. extract, Manilkara sapota L. extract and its combination. Thai J. Pharm. Sci. 2016, 40, 108–111. [Google Scholar]
- Srinivasan, P.; Vijayakumar, S.; Kothandaraman, S.; Palani, M. Anti-diabetic activity of quercetin extracted from Phyllanthus emblica L. fruit: In silico and in vivo approaches. J. Pharm. Anal. 2018, 8, 109–118. [Google Scholar] [CrossRef] [PubMed]
- Rao, N.K.; Bethala, K.; Sisinthy, S.P.; Manickam, S. Antihyperglycemic and In Vivo Antioxidant Activities of Phyllanthus watsonii A. Shaw Roots in Streptozotocin Induced Type 2 Diabetic Rats. Int. J. Pharmacogn. Phytochem. Res. 2016, 8, 335–340. [Google Scholar]
- Singh, S.; Chauhan, M.G.; Kaur, B.; Kumar, B.; Gulati, M.; Singh, S.K. Characterization, organoleptic evaluation and standardization of aqueous extracts of antidiabetic herbs Trigonella foenum, Allium sativum, Aloe vera, Phyllanthus niruri. J. Pharm. Res. 2017, 11, 1370–1375. [Google Scholar]
- Pathak, N.; Bandyopadhyay, A.; Kumar, G.; Chaurasia, R.; Varma, K. Comparative study to evaluate the anti-diabetic activity of commercially available extract of Tinospora cordifolia and Phyllanthus emblica in streptozocin induced diabetic rat. Int. J. Basic Clin. Pharmacol. 2016, 5, 1641–1646. [Google Scholar] [CrossRef]
- Zhou, J.; Zhang, C.; Zheng, G.H.; Qiu, Z. Emblic Leaf flower (Phyllanthus emblica L.) Fruits Ameliorate Vascular Smooth Muscle Cell Dysfunction in Hyperglycemia: An Underlying Mechanism Involved in Ellagitannin Metabolite Urolithin A. Evid. Based Complement. Altern. Med. 2018, 2018, 8478943. [Google Scholar] [CrossRef] [PubMed]
- Nadro, M.S.; Elkanah, G. Hypoglycaemic effect of fractions and crude methanolic leaf extract of Phyllanthus fraternus in streptozotocin—Induced diabetic and normal rats. J. Med. Plants Res. 2017, 11, 58–65. [Google Scholar] [CrossRef]
- Beidokhti, M.N.; Andersen, M.V.; Eid, H.M.; Villavicencio, M.L.S.; Staerk, D.; Haddad, P.S.; Jäger, A.K. Investigation of antidiabetic potential of Phyllanthus niruri L. using assays for α-glucosidase, muscle glucose transport, liver glucose production, and adipogenesis. Biochem. Bioph. Res. Commun. 2017, 493, 869–874. [Google Scholar] [CrossRef] [PubMed]
- Mediani, A.; Abas, F.; Maulidiani, M.; Khatib, A.; Tan, C.P.; Ismail, I.S.; Shaari, K.; Ismail, A.; Lajis, N.H. Metabolic and biochemical changes in streptozotocin induced obese-diabetic rats treated with Phyllanthus niruri extract. J. Pharm. Biomed. Anal. 2016, 128, 302–312. [Google Scholar] [CrossRef] [PubMed]
- Hossain, M.N.; Rahmatullah, M. Oral glucose tolerance tests with a formulation containing Phyllanthus emblica fruits and Trigonella foenum-graecum seeds. World J. Pharm. Pharm. Sci. 2016, 5, 232–239. [Google Scholar]
- Bongu, S.B.R.; Sagree, S.; Gudapareddy, V.; Putakala, M.; Nukala, S.; Gujjala, S.; Bellamkonda, R.; Desireddy, S. Protective role of aqueous extract of Phyllanthus amarus on oxidative stress in pancreas of streptozotocin induced diabetic male Wistar rats. J. Exp. Appl. Anim. Sci. 2016, 2, 23–30. [Google Scholar] [CrossRef]
- Chaimum-aom, N.; Chomko, S.; Talubmook, C. Toxicology and Oral glucose Tolerance Test (OGTT) of Thai Medicinal Plant Used for Diabetes controls, Phyllanthus acidus L. (Euphorbiaceae). Pharmacogn. J. 2016, 9, 58–61. [Google Scholar] [CrossRef]
- Olubunmi, O.P.; Yinka, O.S.; Oladele, O.J.; John, O.A.; Boluwatife, B.D.; Oluseyi, F.S. Aberrations in Renal Function Parameters Following Oral Administration of Phyllanthus amarus in Cadmium-Induced Kidney Damage in Adult Wistar Rats. J. Dis. Med. Plants 2017, 3, 60–67. [Google Scholar]
- Abbas, N.; Naz, M.; Alyousef, L.; Ahmed, E.S.; Begum, A. Comparative study of hepatoprotective effect produced by Cuminum cyminum, fruits of Phyllanthus emblicus and silymarin against cisplatin-induced hepatotoxicity. Int. J. Pharm. Sci. Res. 2017, 8, 2026–2032. [Google Scholar]
- Chaphalkar, R.; Apte, K.G.; Talekar, Y.; Ojha, S.K.; Nandave, M. Antioxidants of Phyllanthus emblica L. bark extract provide hepatoprotection against ethanol-induced hepatic damage: A comparison with silymarin. Oxid. Med. Cell. Longev. 2017, 2017, 3876040. [Google Scholar] [CrossRef] [PubMed]
- Iqbal, Z.; Asif, M.; Aslam, N.; Akhtar, N.; Asmawi, M.Z.; Fei, Y.M.; Jabeen, Q. Clinical investigations on gastroprotective effects of ethanolic extract of Phyllanthus emblica L. fruits. J. Herb. Med. 2017, 7, 11–17. [Google Scholar] [CrossRef]
- Hassan, M.R.A.; Mustapha, N.R.N.; Jaya, F.; Arjunan, S.; Ooi, E.T.; Said, R.M.; Menon, J.; Tee, H.P.; Omar, H.; Aiman, S. Efficacy and Safety of Phyllanthus niruri in Non-alcoholic Steatohepatitis Treatment: Pilot Study from Malaysia. J. Pharm. Pract. Commun. Med. 2017, 3, 131–137. [Google Scholar] [CrossRef]
- Lu, C.C.; Yang, S.H.; Hsia, S.M.; Wu, C.H.; Yen, G.C. Inhibitory effects of Phyllanthus emblica L. on hepatic steatosis and liver fibrosis in vitro. J. Funct. Foods 2016, 20, 20–30. [Google Scholar] [CrossRef]
- Yao, N.; Kamagaté, M.; Amonkan, A.K.; Koffi, C.; Kpahe, F.; Kouamé, M.; Dié-Kacou, H. Comparative effects of aqueuos extract of Phyllanthus amarus and its fractions on urinary excretion in rat. J. Phytopharmacol. 2016, 5, 182–184. [Google Scholar]
- Yao, A.N.; Kamagate, M.; Amonkan, A.K.; Chabert, P.; Kpahe, F.; Koffi, C.; Kouame, M.N.; Auger, C.; Kati-Coulibaly, S.; Schini-Kerth, V.; et al. The acute diuretic effect of an ethanolic fraction of Phyllanthus amarus (Euphorbiaceae) in rats involves prostaglandins. BMC Complement. Altern. Med. 2018, 18, 94. [Google Scholar] [CrossRef] [PubMed]
- Uddin, M.S.; Mamun, A.A.; Hossain, M.S.; Akter, F.; Iqbal, M.A.; Asaduzzaman, M. Exploring the Effect of Phyllanthus emblica L. on Cognitive Performance, Brain Antioxidant Markers and Acetylcholinesterase Activity in Rats: Promising Natural Gift for the Mitigation of Alzheimer’s Disease. Ann. Neurosci. 2016, 23, 218–229. [Google Scholar] [CrossRef] [PubMed]
- Uddin, M.S.; Mamun, A.A.; Hossain, M.S.; Ashaduzzaman, M.; Noor, M.A.A.; Hossain, M.S.; Uddin, M.J.; Sarker, J.; Asaduzzaman, M. Neuroprotective Effect of Phyllanthus acidus L. on Learning and Memory Impairment in Scopolamine-Induced Animal Model of Dementia and Oxidative Stress: Natural Wonder for Regulating the Development and Progression of Alzheimer’s Disease. Adv. Alzheimer’s Dis. 2016, 5, 53–72. [Google Scholar] [CrossRef]
- Jang, H.; Srichayet, P.; Park, W.J.; Heo, H.J.; Kim, D.-O.; Tongchitpakdee, S.; Kim, T.-J.; Jung, S.H.; Lee, C.Y. Phyllanthus emblica L. (Indian gooseberry) extracts protect against retinal degeneration in a mouse model of amyloid beta-induced Alzheimer’s disease. J. Funct. Foods 2017, 37, 330–338. [Google Scholar] [CrossRef]
- Wagle, N.; Nagarjuna, S.; Sharma, H.; Dangi, N.B.; Sapkota, H.P.; Naik, B.S.; Padhaya, R.R. Evaluation of antinociceptive and anti-inflammatory activity of phytosterol present in chloroform extract of Phyllanthus maderaspatensis. Indian J. Physiol. Pharmacol. 2016, 60, 90–95. [Google Scholar] [PubMed]
- Hossain, M.; Akter, S.; Begum, Y.; Bulbul, I. Analgesic and Anti-inflammatory Activities of Ethanolic Leaf Extract of Phyllanthus acidus L. on Swiss Albino Mice. Eur. J. Med. Plants 2016, 13, 1–10. [Google Scholar] [CrossRef]
- Yoon, W.H.; Lee, K.H. Anti-inflammatory, Anti-arthritic and Analgesic Effect of the Herbal Extract Made from Bacopa monnieriis, Cassia fistula and Phyllanthus polyphyllus. Nat. Prod. Sci. 2017, 23, 108–112. [Google Scholar] [CrossRef]
- Manikandan, R.; Beulaja, M.; Thiagarajan, R.; Palanisamy, S.; Goutham, G.; Koodalingam, A.; Prabhu, N.M.; Kannapiran, E.; Basu, M.J.; Arulvasu, C.; et al. Biosynthesis of silver nanoparticles using aqueous extract of Phyllanthus acidus L. fruits and characterization of its anti-inflammatory effect against H2O2 exposed rat peritoneal macrophages. Process Biochem. 2017, 55, 172–181. [Google Scholar] [CrossRef]
- Hossain, M.; Akter, S.; Das, A.; Sarwar, M. CNS Depressant, Antidiarrheal and Antipyretic Activities of Ethanolic Leaf Extract of Phyllanthus acidus L. on Swiss Albino Mice. Br. J. Pharm. Res. 2016, 10, 1–9. [Google Scholar] [CrossRef]
- Khan, A.; Ahmed, T.; Rizwan, M.; Khan, N. Comparative therapeutic efficacy of Phyllanthus emblica (Amla) fruit extract and procaine penicillin in the treatment of subclinical mastitis in dairy buffaloes. Microb. Pathog. 2018, 115, 8–11. [Google Scholar] [CrossRef] [PubMed]
- Afrin, F.; Banik, S.; Hossain, M.S. Pharmacological activities of methanol extract of Phyllanthus acidus pulp. J. Med. Plants Res. 2016, 10, 790–795. [Google Scholar]
- Tjandrawinata, R.R.; Susanto, L.W.; Nofiarny, D. The use of Phyllanthus niruri L. as an immunomodulator for the treatment of infectious diseases in clinical settings. Asian Pac. J. Trop. Dis. 2017, 7, 132–140. [Google Scholar] [CrossRef]
- Putri, D.U.; Rintiswati, N.; Soesatyo, M.H.; Haryana, S.M. Immune modulation properties of herbal plant leaves: Phyllanthus niruri aqueous extract on immune cells of tuberculosis patient—In vitro study. Nat. Prod. Res. 2018, 32, 463–467. [Google Scholar] [CrossRef] [PubMed]
- Muthulakshmi, M.; Subramani, P.; Michael, R. Immunostimulatory effect of the aqueous leaf extract of Phyllanthus niruri on the specific and nonspecific immune responses of Oreochromis mossambicus Peters. Iran. J. Vet. Res. 2016, 17, 200–202. [Google Scholar] [PubMed]
- Ilangkovan, M.; Jantan, I.; Bukhari, S.N. Phyllanthin from Phyllanthus amarus inhibits cellular and humoral immune responses in Balb/C mice. Phytomedicine 2016, 23, 1441–1450. [Google Scholar] [CrossRef] [PubMed]
- Ilangkovan, M.; Jantan, I.; Mesaik, M.A.; Bukhari, S.N. Inhibitory Effects of the Standardized Extract of Phyllanthus amarus on Cellular and Humoral Immune Responses in Balb/C Mice. Phytother. Res. 2016, 30, 1330–1338. [Google Scholar] [CrossRef] [PubMed]
- Sabdoningrum, E.K.; Hidanah, S.; Wahjuni, R.S.; Chusniati, S.; Arimbi, A. An in vitro antibacterial activity test of Meniran Herbs’ (Phyllanthus Niruri L.) ethanol extract against Mycoplasma gallisepticum causes Chronic Respiratory Disease (CRD) in Broiler Chickens. KnE Life Sci. 2017, 3, 48–61. [Google Scholar] [CrossRef]
- Senjobi, C.T.; Ettu, A.O.; Otujo, C.O. Antibacterial and antifungal activities of leaf extracts of Phyllanthus amaru Schum and Thonn. J. Pharmacogn. Phytother. 2017, 9, 6–10. [Google Scholar] [CrossRef]
- Boakye, Y.D.; Agyare, C.; Hensel, A. Anti-infective properties and time-kill kinetics of Phyllanthus muellerianus and its major constituent, geraniin. Med. Chem. 2016, 6, 95–104. [Google Scholar] [CrossRef]
- Adesegun, A.; Samuel, F.; Adesina, O. Antibacterial Activity of the Volatile Oil of Phyllanthus muellerianus and Its Inhibition against the Extracellular Protease of Klebsiella granulomatis. Eur. J. Med. Plants 2016, 14, 1–10. [Google Scholar] [CrossRef]
- Pathmavathi, M.; Thamizhiniyan, P. Antimicrobial activity of various extracts of Plectranthus ambionicus and Phyllanthus amarus. J. Appl. Adv. Res. 2016, 1, 29–35. [Google Scholar] [CrossRef]
- Sathishkumar, M.; Saroja, M.; Venkatachalam, M.; Rajamanickam, A. Biosynthesis of zinc sulphide nanoparticles using Phyllanthus emblica and their antimicrobial activities. Elixir Elec. Eng. 2017, 102, 44411–44415. [Google Scholar]
- Oluboyo, B.O.; Oluboyo, A.O.; Kalu, S.O. Inhibitory effects of Phyllanthus amarus extracts on the growth of some pathogenic microorganisms. Afr. J. Clin. Exp. Microbiol. 2016, 17, 166–172. [Google Scholar] [CrossRef]
- Gunawan, I.; Bawa, I.; Putra, A.B. Isolation, characterization and antibacterial activity of triterpenoid compounds fraction chloroform bark Phyllanthus niruri L. World Pharm. Pharm. Sci. 2016, 5, 357–364. [Google Scholar]
- Haris, M.; Kumar, A.; Ahmad, A.; Abuzinadah, M.F.; Basheikh, M.; Khan, S.A.; Mujeeb, M. Microwave-assisted green synthesis and antimicrobial activity of silver nanoparticles derived from a supercritical carbon dioxide extract of the fresh aerial parts of Phyllanthus niruri L. Trop. J. Pharm. Res. 2017, 16, 2967–2976. [Google Scholar] [CrossRef]
- Uzor, B.; Umeh, L.; Manu, O. Phytochemical Composition and Antimicrobial Potential of Phyllanthus amarus Leaf Extract Against Some Clinical Isolates. Niger. J. Microbiol. 2016, 30, 3464–3467. [Google Scholar]
- Ahamath, J.M.; Vahith, R.A.; Manivel, V.; Elamparithi, R. Phytochemical Screening and Antimicrobial Activity of Phyllanthus niruri. J. Adv. Appl. Sci. Res. 2017, 1, 7. [Google Scholar]
- Gao, Q.; Li, X.; Huang, H.; Guan, Y.; Mi, Q.; Yao, J. The Efficacy of a Chewing Gum Containing Phyllanthus emblica Fruit Extract in Improving Oral Health. Curr. Microbiol. 2018, 75, 604–610. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Li, X.; Wang, J.-K.; Kuang, Y.; Qi, M.-X. Anti-hepatitis B viral activity of Phyllanthus niruri L. (Phyllanthaceae) in HepG2/C3A and SK-HEP-1 cells. Trop. J. Pharm. Res. 2017, 16, 1873–1879. [Google Scholar] [CrossRef]
- Chitra, R.; Vadivel, E.; Rajamani, K. Estimation of anti-hepatic viral compounds in Phyllanthus amarus in vitro cultures. J. Hortic. Sci. 2016, 3, 62–65. [Google Scholar]
- Sarma, K.; Borkakoty, B.; Parida, P.; Jakharia, A.; Dey, D.; Biswas, D.; Panda, D.; K Modi, M.; K Mohapatra, P.; Mahanta, J. In Silico Identification of Natural Lead Molecules from the Genus of Phyllanthus Against Hepatitis B Virus Reverse Transcriptase. Nat. Prod. J. 2016, 6, 292–304. [Google Scholar] [CrossRef]
- Faeji, C.O.; Oladunmoye, M.K.; Adebayo, I.A.; Adebolu, T.T. In-ovo biological activities of Phyllanthus amarus leaf extracts against Newcastle disease virus. J. Med. Plants Res. 2017, 11, 419–425. [Google Scholar] [CrossRef]
- Dinesh, S.; Sudharsana, S.; Mohanapriya, A.; Itami, T.; Sudhakaran, R. Molecular docking and simulation studies of Phyllanthus amarus phytocompounds against structural and nucleocapsid proteins of white spot syndrome virus. 3 Biotech 2017, 7, 353. [Google Scholar] [CrossRef] [PubMed]
- Sundaram, D.; Kesavan, K.; Kumaravel, H.; Mohammed, R.F.; Tohru, M.; Toshiaki, I.; Raja, S. Protective efficacy of active compounds from Phyllanthus amarus against white spot syndrome virus in freshwater crab (Paratelphusa hydrodomous). Aquac. Res. 2016, 47, 2061–2067. [Google Scholar] [CrossRef]
- Zhang, X.; Xia, Q.; Yang, G.; Zhu, D.; Shao, Y.; Zhang, J.; Cui, Y.; Wang, R.; Zhang, L. The anti-HIV-1 activity of polyphenols from Phyllanthus urinaria and the pharmacokinetics and tissue distribution of its marker compound, gallic acid. J. Tradit. Chin. Med. Sci. 2017, 4, 158–166. [Google Scholar] [CrossRef]
- Manjula, V.; Norman, T.S.J. Pharmacognostical study of Phyllanthus reticulatus: A tribal drug. Pharma Innov. J. 2017, 6, 107–109. [Google Scholar]
- Berezi, E.; Uwakwe, A.; Monago-Ighorodje, C.; Nwauche, K. Gastroprotective potentials of aqueous leaf extracts of Phyllanthus amarus on ibuprofen-induced ulcer in Wistar rats. Int. J. Adv. Res. Biol. Sci. 2017, 4, 138–146. [Google Scholar]
- Joshi, S.; Gajbhiye, S.V.; Thatte, U. Evaluation of gastric motility of Phyllanthus emblica and Asparagus racemosus in cold stress induced gastric damage. Int. J. Basic Clin. Pharmacol. 2016, 5, 1516–1519. [Google Scholar] [CrossRef]
- Ahmed, A.H. The Effect of Water Extracts of Phyllanthus emblica and Costus speciousus on Reducing Obesity in Albino Rats. Alex. Sci. Exch. 2017, 38, 463–472. [Google Scholar]
- Nambiar, S.S.; Venugopal, K.S.; Shetty, N.P.; Appaiah, K.A. Fermentation induced changes in bioactive properties of wine from Phyllanthus with respect to atherosclerosis. J. Food Sci. Technol. 2016, 53, 2361–2371. [Google Scholar] [CrossRef] [PubMed]
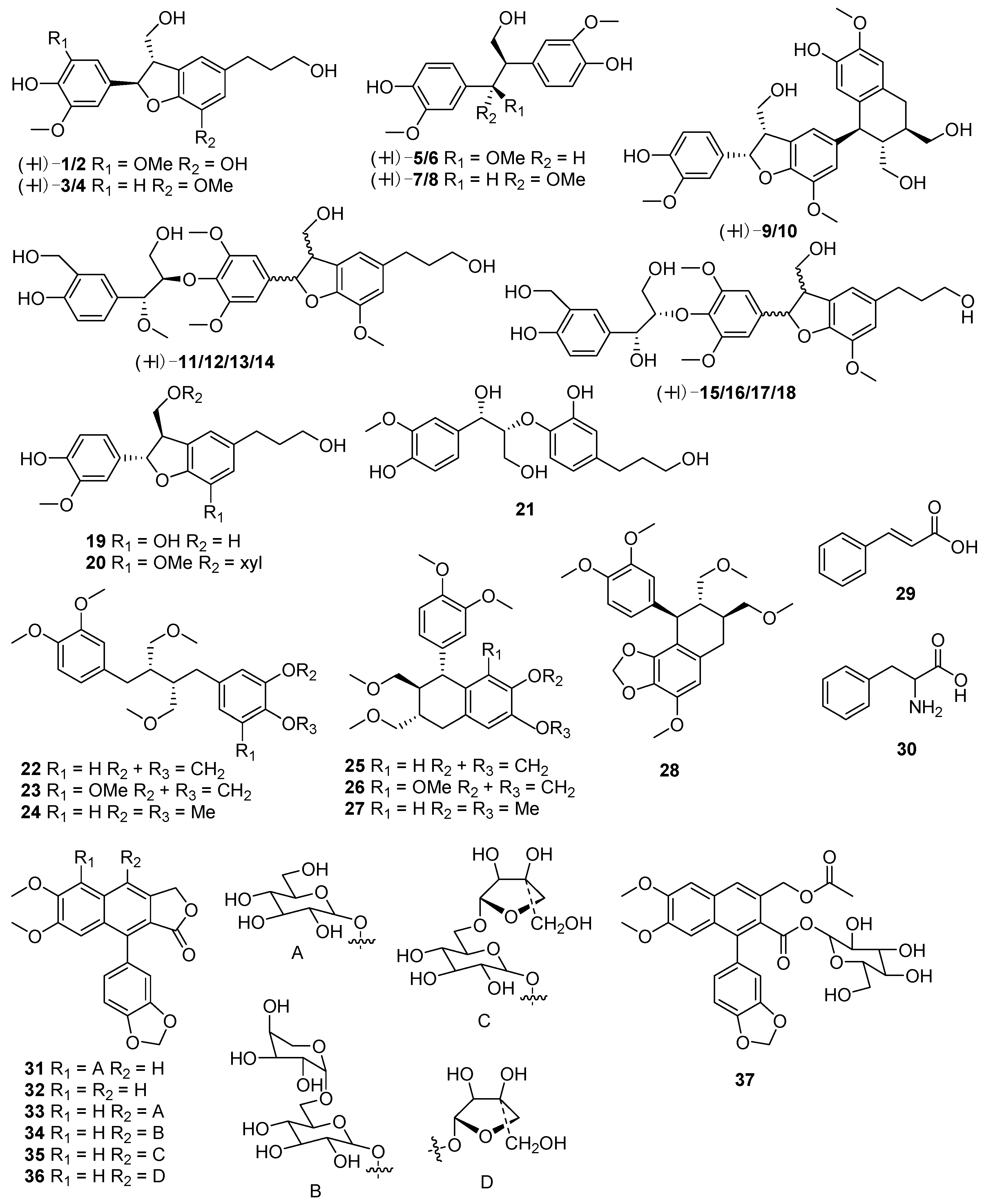

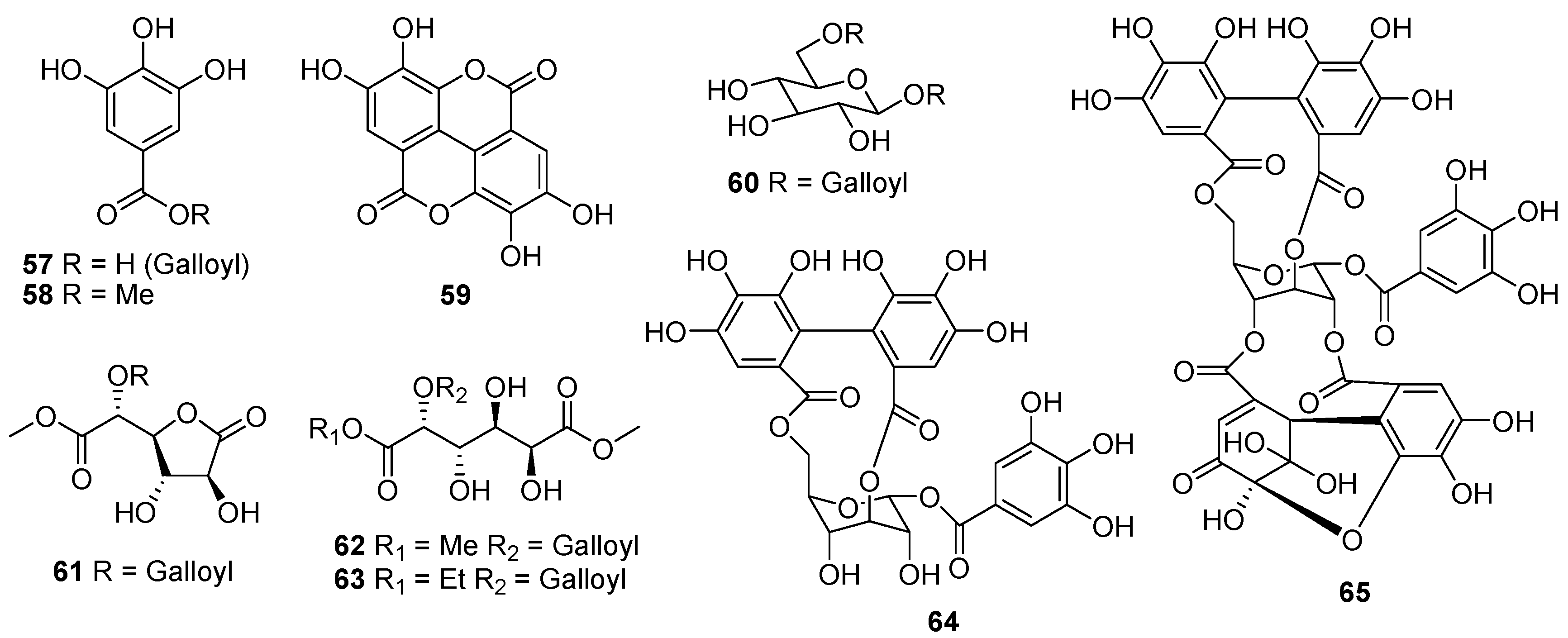
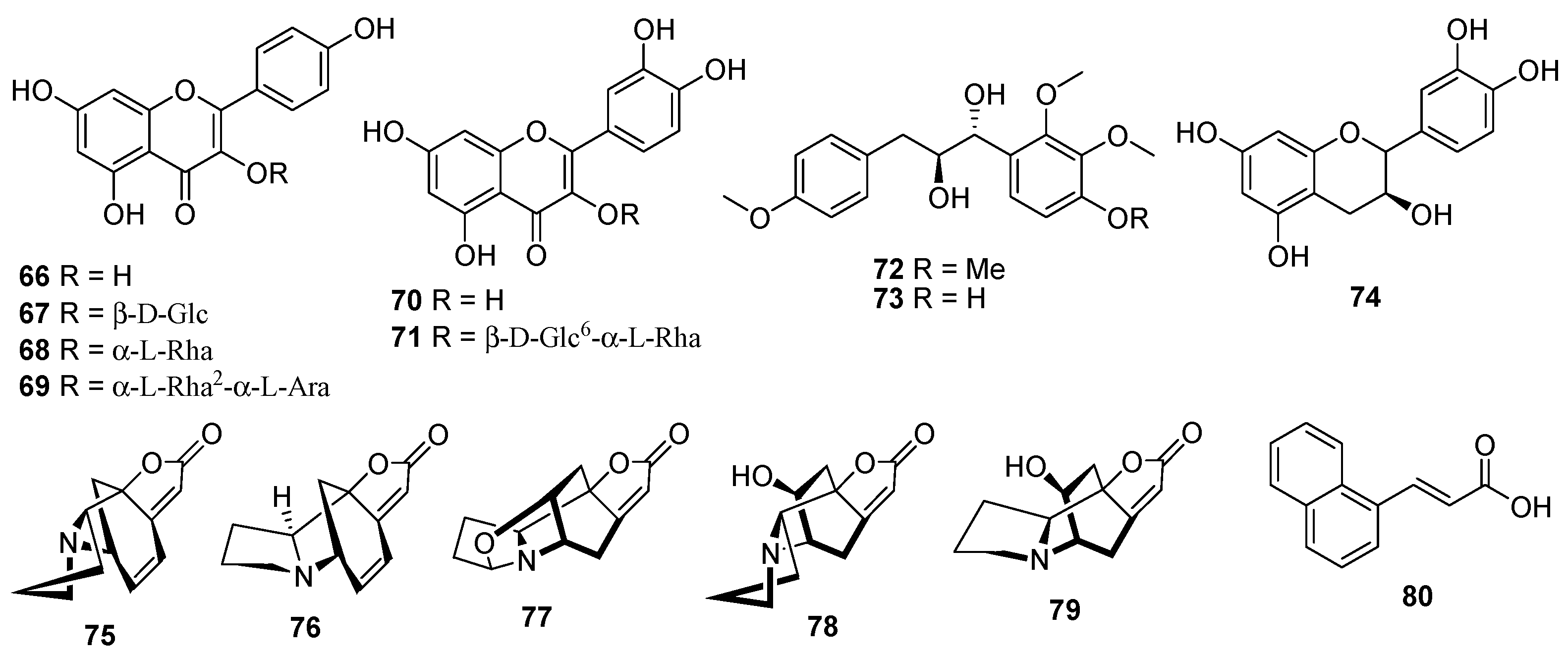
| No. | Compounds | Sources | Ref. |
|---|---|---|---|
| 1 | (+)-(7R,8S)-phyllanglaucin A | P. glaucus | [23] |
| 2 | (−)-(7S,8R)-phyllanglaucin A | P. glaucus | [23] |
| 3 | (7R,8S)-dihydrodehyd roconiferyl alcohol | P. glaucus | [23] |
| 4 | (7S,8R)-dihydrodehydrodiconiferyl alcohol | P. glaucus | [23] |
| 5 | (7R,8R)-4,4′-dihydroxy-3,7,3′-trimethoxy-8,1′-7′,8′,9′-trinor-neolignan-9-ol | P. glaucus | [23] |
| 6 | (7S,8S)-4,4′ -dihydroxy-3,7,3′-trimethoxy-8,1′-7′,8′,9′-trinor-neolignan-9-ol | P. glaucus | [23] |
| 7 | (7S,8R)-4,4′-dihydroxy-3,7,3′-trimethoxy-8,1′-7′,8′,9′-trinor-neolignan-9-ol | P. glaucus | [23] |
| 8 | (7R,8S)-4,4′-dihydroxy-3,7,3′-trimethoxy-8,1′-7′,8′,9′-trinor-neolignan-9-ol | P. glaucus | [23] |
| 9 | (+)-phyllanglaucin B | P. glaucus | [23] |
| 10 | (−)-phyllanglaucin B | P. glaucus | [23] |
| 11 | (+)-phyllanglaucin C | P. glaucus | [23] |
| 12 | (−)-phyllanglaucin C | P. glaucus | [23] |
| 13 | (+)-phyllanglaucin D | P. glaucus | [23] |
| 14 | (−)-phyllanglaucin D | P. glaucus | [23] |
| 15 | (7R,8S,7′R,8′R)-acernikol | P. glaucus | [23] |
| 16 | (7S,8R,7′S,8′S)-acernikol | P. glaucus | [23] |
| 17 | (7R,8S,7′S,8′S)-acernikol | P. glaucus | [23] |
| 18 | (7S,8R,7′R,8′R)-acernikol | P. glaucus | [23] |
| 19 | (7S,8R)-cedrusin | P. glaucus | [23] |
| 20 | (7S,8R)-dihydrodehydrodiconifenyl alcohol 9-O-β-d-xylopyranoside | P. glaucus | [23] |
| 21 | (7S,8R)-4,7,9,9′-tetrahydroxy-3,3′-dimethoxy-8-O-4′-neolignan | P. glaucus | [23] |
| 22 | 5-demethoxy-niranthin | P. amarus | [25] |
| 23 | niranthin | P. amarus | [25,26] |
| 24 | phyllanthin | P. amarus | [25,26] |
| 25 | filtetralin | P. amarus | [25] |
| 26 | 5-demethoxy-nirtetralin | P. amarus | [25] |
| 27 | nirtetralin | P. amarus | [25] |
| 28 | hipophyllanthin | P. amarus | [25] |
| 29 | cinnamic acid | P. amarus, P. urinaria | [24] |
| 30 | phenylalanine | P. amarus,P. urinaria | [24] |
| 31 | 5-O-β-d-glucopyranosyljusticidin B | P. brasiliensis | [27] |
| 32 | justicidin B | P. brasiliensis | [27] |
| 33 | cleistanthin B | P. brasiliensis | [27] |
| 34 | arabelline | P. brasiliensis | [27] |
| 35 | 4-O-β-d-apiofuranosyl-(1′″→6′′)-β-d-glucopyranosyldiphyllin | P. brasiliensis | [27] |
| 36 | tuberculatin | P. brasiliensis | [27] |
| 37 | phyllanthostatin A | P. brasiliensis | [27] |
| No. | Compounds | Sources | Ref. |
|---|---|---|---|
| 38 | phainanolide A | P. hainanensis | [28] |
| 39 | phainanoid G | P. hainanensis | [28] |
| 40 | phainanoid H | P. hainanensis | [28] |
| 41 | phainanoid I | P. hainanensis | [28] |
| 42 | 28-norlup-20(29)-ene-3,17β-diol | P. urinaria | [29] |
| 43 | betulin | P. urinaria | [29] |
| 44 | β-betulinic acid | P. urinaria | [29] |
| 45 | 3-oxofriedelan-28-oic acid | P. urinaria | [29] |
| 46 | oleanolic acid | P. urinaria | [29] |
| 47 | (E)-coumaroyltaraxerol | P. urinaria | [29] |
| 48 | (Z)-coumaroyltaraxerol | P. urinaria | [29] |
| 49 | phyllaciduloid A | P. acidus | [32] |
| 50 | spruceanol | P. acidus | [31] |
| 51 | phyllaciduloid B | P. acidus | [32] |
| 52 | phyllaciduloid C | P. acidus | [32] |
| 53 | phyllaciduloid D | P. acidus | [32] |
| 54 | phyllanes A | P. acidus | [31] |
| 55 | phyllanes B | P. acidus | [31] |
| 56 | (−)-loliolide | P. urinaria | [30] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nisar, M.F.; He, J.; Ahmed, A.; Yang, Y.; Li, M.; Wan, C. Chemical Components and Biological Activities of the Genus Phyllanthus: A Review of the Recent Literature. Molecules 2018, 23, 2567. https://doi.org/10.3390/molecules23102567
Nisar MF, He J, Ahmed A, Yang Y, Li M, Wan C. Chemical Components and Biological Activities of the Genus Phyllanthus: A Review of the Recent Literature. Molecules. 2018; 23(10):2567. https://doi.org/10.3390/molecules23102567
Chicago/Turabian StyleNisar, Muhammad Farrukh, Junwei He, Arsalan Ahmed, Youxin Yang, Mingxi Li, and Chunpeng Wan. 2018. "Chemical Components and Biological Activities of the Genus Phyllanthus: A Review of the Recent Literature" Molecules 23, no. 10: 2567. https://doi.org/10.3390/molecules23102567
APA StyleNisar, M. F., He, J., Ahmed, A., Yang, Y., Li, M., & Wan, C. (2018). Chemical Components and Biological Activities of the Genus Phyllanthus: A Review of the Recent Literature. Molecules, 23(10), 2567. https://doi.org/10.3390/molecules23102567






