Trichosanthis Pericarpium Aqueous Extract Protects H9c2 Cardiomyocytes from Hypoxia/Reoxygenation Injury by Regulating PI3K/Akt/NO Pathway
Abstract
1. Introduction
2. Results
2.1. TPAE Inhibited Cell Death Induced by H/R
2.2. TPAE Prevented Apoptosis in H9c2 Cells after H/R
2.3. TPAE Activated PI3K/Akt Signaling Pathway
2.4. Inhibition of PI3K Alleviated the Cardioprotection of TPAE
2.5. TPAE Mitigated H/R-Induced Decrease of NO Production via PI3K/Akt/NOS Pathway
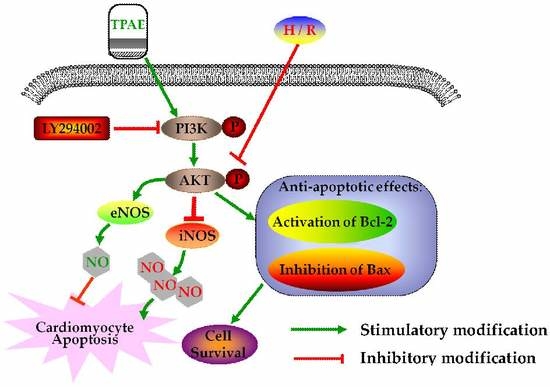
3. Discussion
4. Materials and Methods
4.1. Materials and Chemicals
4.2. Extract Preparation
4.3. Cell Culture and Hypoxia/Reoxygenation Model (H/R)
4.4. Experimental Protocols
4.5. Flow Cytometric Detection of Apoptosis
4.6. Enzyme-Linked Immunosorbent Assay
4.7. RNA Isolation and Purification
4.8. Quantitative Real-Time PCR to Confirm Markers
4.9. Western Blotting Analysis
4.10. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Huang, X.; Zuo, L.; Lv, Y.; Chen, C.; Yang, Y.; Xin, H.; Li, Y.; Qian, Y. Asiatic Acid Attenuates Myocardial Ischemia/Reperfusion Injury via Akt/GSK-3beta/HIF-1alpha Signaling in Rat H9c2 Cardiomyocytes. Molecules 2016, 21, 1248. [Google Scholar] [CrossRef] [PubMed]
- Yusuf, S.; Reddy, S.; Ounpuu, S.; Anand, S. Global burden of cardiovascular diseases: Part I: General considerations, the epidemiologic transition, risk factors, and impact of urbanization. Circulation 2001, 104, 2746–2753. [Google Scholar] [CrossRef] [PubMed]
- Buja, L.M. Myocardial ischemia and reperfusion injury. Cardiovasc. Pathol. 2005, 14, 170–175. [Google Scholar] [CrossRef] [PubMed]
- Gao, L.N.; Zhou, X.; Zhang, Y.; Cui, Y.L.; Yu, C.Q.; Gao, S. The anti-inflammatory activities of ethanol extract from Dan-Lou prescription in vivo and in vitro. BMC Complement. Altern. Med. 2015, 15, 317. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Xia, W.; Liu, W.W.; Xu, C.B.; Gu, N. Gualoupi (Pericarpium Trichosanthis) injection in combination with convention therapy for the treatment of angina pectoris: A Meta-analysis. J. Tradit. Chin. Med. 2017, 37, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Liao, P.; Sun, G.; Zhang, C.; Wang, M.; Sun, Y.; Zhou, Y.; Sun, X.; Jian, J. Bauhinia championii Flavone Attenuates Hypoxia-Reoxygenation Induced Apoptosis in H9c2 Cardiomyocytes by Improving Mitochondrial Dysfunction. Molecules 2016, 21, 1469. [Google Scholar] [CrossRef] [PubMed]
- Hausenloy, D.J.; Yellon, D.M. Myocardial ischemia-reperfusion injury: A neglected therapeutic target. J. Clin. Investig. 2013, 123, 92–100. [Google Scholar] [CrossRef] [PubMed]
- Qi, J.Y.; Wang, L.; Gu, D.S.; Guo, L.H.; Zhu, W.; Zhang, M.Z. Protect effects of Danlou Tablet against murine myocardial ischemia and reperfusion injury in vivo. Chin. J. Integr. Med. 2016. [Google Scholar] [CrossRef]
- Ding, Y.F.; Peng, Y.R.; Shen, H.; Shu, L.; Wei, Y.J. Gualou Xiebai decoction inhibits cardiac dysfunction and inflammation in cardiac fibrosis rats. BMC Complement. Altern. Med. 2016, 16, 49. [Google Scholar] [CrossRef] [PubMed]
- Hu, F.; Koon, C.M.; Chan, J.Y.; Lau, K.M.; Fung, K.P. The cardioprotective effect of danshen and gegen decoction on rat hearts and cardiomyocytes with post-ischemia reperfusion injury. BMC Complement. Altern. Med. 2012, 12, 249. [Google Scholar] [CrossRef] [PubMed]
- Mao, S.; Wang, L.; Ouyang, W.; Zhou, Y.; Qi, J.; Guo, L.; Zhang, M.; Hinek, A. Traditional Chinese medicine, Danlou tablets alleviate adverse left ventricular remodeling after myocardial infarction: Results of a double-blind, randomized, placebo-controlled, pilot study. BMC Complement. Altern. Med. 2016, 16, 447. [Google Scholar] [CrossRef]
- Chinese Pharmacopoeia Commission. Pharmacopoeia of the People’s Republic of China, 10th ed.; China Medical Science and Technology Press: Beijing, China, 2015; p. 114. ISBN 978–7-5067–7337–9. [Google Scholar]
- Li, C.; Fan, G.W.; Zhang, H.; Zhu, M.J. Protective effects of Trichosanthes on cardiovascular system. Int. J. Clin. Exp. Med. 2016, 9, 17106–17112. [Google Scholar] [CrossRef]
- Fu, N.; Li, H.; Sun, J.; Xun, L.; Gao, D.; Zhao, Q. Trichosanthes pericarpium Aqueous Extract Enhances the Mobilization of Endothelial Progenitor Cells and Up-regulates the Expression of VEGF, eNOS, NO, and MMP-9 in Acute Myocardial Ischemic Rats. Front. Physiol. 2017, 8, 1132. [Google Scholar] [CrossRef]
- Zhang, H.J.; Chen, R.C.; Sun, G.B.; Yang, L.P.; Zhu, Y.D.; Xu, X.D.; Sun, X.B. Protective effects of total flavonoids from Clinopodium chinense (Benth.) O. Ktze on myocardial injury in vivo and in vitro via regulation of Akt/Nrf2/HO-1 pathway. Phytomedicine 2018, 40, 88–97. [Google Scholar] [CrossRef]
- Chun, J.N.; Cho, M.; So, I.; Jeon, J.H. The protective effects of Schisandra chinensis fruit extract and its lignans against cardiovascular disease: A review of the molecular mechanisms. Fitoterapia 2014, 97, 224–233. [Google Scholar] [CrossRef]
- Mullonkal, C.J.; Toledo-Pereyra, L.H. Akt in ischemia and reperfusion. J. Investig. Surg. 2007, 20, 195–203. [Google Scholar] [CrossRef]
- Hausenloy, D.J.; Tsang, A.; Yellon, D.M. The reperfusion injury salvage kinase pathway: A common target for both ischemic preconditioning and postconditioning. Trends Cardiovas. Med. 2005, 15, 69–75. [Google Scholar] [CrossRef]
- Wang, J.; Ji, S.Y.; Liu, S.Z.; Jing, R.; Lou, W.J. Cardioprotective effect of breviscapine: Inhibition of apoptosis in H9c2 cardiomyocytes via the PI3K/Akt/eNOS pathway following simulated ischemia/reperfusion injury. Pharmazie 2015, 70, 593–597. [Google Scholar] [CrossRef]
- Chern, C.M.; Liou, K.T.; Wang, Y.H.; Liao, J.F.; Yen, J.C.; Shen, Y.C. Andrographolide inhibits PI3K/AKT-dependent NOX2 and iNOS expression protecting mice against hypoxia/ischemia-induced oxidative brain injury. Planta Med. 2011, 77, 1669–1679. [Google Scholar] [CrossRef]
- Brix, B.; Mesters, J.R.; Pellerin, L.; Johren, O. Endothelial cell-derived nitric oxide enhances aerobic glycolysis in astrocytes via HIF-1alpha-mediated target gene activation. J. Neurosci. 2012, 32, 9727–9735. [Google Scholar] [CrossRef]
- El, A.M.; Sanchez-Puelles, J.M.; Royo, I.; Lopez-Hernandez, E.; Sanchez-Ferrer, A.; Acena, J.L.; Rodriguez-Manas, L.; Angulo, J. FM19G11 reverses endothelial dysfunction in rat and human arteries through stimulation of the PI3K/Akt/eNOS pathway, independently of mTOR/HIF-1alpha activation. Brit. J. Pharmacol. 2015, 172, 1277–1291. [Google Scholar] [CrossRef]
- Kuriyama, S.; Morio, Y.; Toba, M.; Nagaoka, T.; Takahashi, F.; Iwakami, S.; Seyama, K.; Takahashi, K. Genistein attenuates hypoxic pulmonary hypertension via enhanced nitric oxide signaling and the erythropoietin system. Am. J. Physiol. Lung. C. 2014, 306, L996–L1005. [Google Scholar] [CrossRef] [PubMed]
- He, T.; Ai, M.; Zhao, X.; Xing, Y. Inducible Nitric Oxide Synthase Mediates Hypoxia-Induced Hypoxia-Inducible Factor-1α Activation and Vascular Endothelial Growth Factor Expression in Oxygen-Induced Retinopathy. Pathobiology 2007, 74, 336–343. [Google Scholar] [CrossRef] [PubMed]
- He, F.; Xu, B.L.; Chen, C.; Jia, H.J.; Wu, J.X.; Wang, X.C.; Sheng, J.L.; Huang, L.; Cheng, J. Methylophiopogonanone A suppresses ischemia/reperfusion-induced myocardial apoptosis in mice via activating PI3K/Akt/eNOS signaling pathway. Acta Pharmacol. Sin. 2016, 37, 763–771. [Google Scholar] [CrossRef] [PubMed]
- Venardos, K.M.; Rajapakse, N.W.; Williams, D.; Hoe, L.S.; Peart, J.N.; Kaye, D.M. Cardio-protective effects of combined l-arginine and insulin: Mechanism and therapeutic actions in myocardial ischemia-reperfusion injury. Eur. J. Pharmacol. 2015, 769, 64–70. [Google Scholar] [CrossRef] [PubMed]
- Kanno, S.; Lee, P.C.; Zhang, Y.; Ho, C.; Griffith, B.P.; Shears, L.N.; Billiar, T.R. Attenuation of myocardial ischemia/reperfusion injury by superinduction of inducible nitric oxide synthase. Circulation 2000, 101, 2742–2748. [Google Scholar] [CrossRef] [PubMed]
- Ferdinandy, P.; Danial, H.; Ambrus, I.; Rothery, R.A.; Schulz, R. Peroxynitrite is a major contributor to cytokine-induced myocardial contractile failure. Circ. Res. 2000, 87, 241–247. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Loukili, N.; Rosenblatt-Velin, N.; Pacher, P.; Feihl, F.; Waeber, B.; Liaudet, L. Peroxynitrite is a key mediator of the cardioprotection afforded by ischemic postconditioning in vivo. PLoS ONE 2013, 8, e70331. [Google Scholar] [CrossRef]
- Oyama, J.; Shimokawa, H.; Momii, H.; Cheng, X.; Fukuyama, N.; Arai, Y.; Egashira, K.; Nakazawa, H.; Takeshita, A. Role of nitric oxide and peroxynitrite in the cytokine-induced sustained myocardial dysfunction in dogs in vivo. J. Clin. Investig. 1998, 101, 2207–2214. [Google Scholar] [CrossRef] [PubMed]
- Nossuli, T.O.; Hayward, R.; Jensen, D.; Scalia, R.; Lefer, A.M. Mechanisms of cardioprotection by peroxynitrite in myocardial ischemia and reperfusion injury. Am. J. Physiol. 1998, 275, H509–H519. [Google Scholar] [CrossRef] [PubMed]
- Dhalla, N.S.; Elmoselhi, A.B.; Hata, T.; Makino, N. Status of myocardial antioxidants in ischemia-reperfusion injury. Cardiovasc. Res. 2000, 47, 446–456. [Google Scholar] [CrossRef]
- Yu, M.F.; Gorenne, I.; Su, X.; Moreland, R.S.; Kotlikoff, M.I. Sodium hydrosulfite contractions of smooth muscle are calcium and myosin phosphorylation independent. Am. J. Physiol. 1998, 275, L976–L982. [Google Scholar] [CrossRef] [PubMed]
- Lawson, W.J.; Holland, R.A.; Forster, R.E. Effect of temperature on deoxygenation rate of human red cells. J. Appl. Physiol. 1965, 20, 912–918. [Google Scholar] [CrossRef] [PubMed]
- Luo, L.; Lu, L.; Lu, Y.; Zhang, L.; Li, B.; Guo, K.; Chen, L.; Wang, Y.; Shao, Y.; Xu, J. Effects of hypoxia on progranulin expression in HT22 mouse hippocampal cells. Mol. Med. Rep. 2014, 9, 1675–1680. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Li, Y.; Ding, Y.; Wang, J.; Geng, J.; Yang, H.; Ren, J.; Tang, J.; Gao, J. Neuroprotective effects of gallic acid against hypoxia/reoxygenation-induced mitochondrial dysfunctions in vitro and cerebral ischemia/reperfusion injury in vivo. Brain Res. 2014, 1589, 126–139. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.Q.; Eyzaguirre, C. Effects of hypoxia induced by Na2S2O4 on intracellular calcium and resting potential of mouse glomus cells. Brain Res. 1999, 818, 118–126. [Google Scholar] [CrossRef]
- Konstantinidis, K.; Whelan, R.S.; Kitsis, R.N. Mechanisms of cell death in heart disease. Arterioscler. Throm. Vas. 2012, 32, 1552–1562. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Chen, Y.; Shen, Q.; Liu, G.; Ye, J.; Sun, G.; Sun, X. Myricitrin Attenuates High Glucose-Induced Apoptosis through Activating Akt-Nrf2 Signaling in H9c2 Cardiomyocytes. Molecules 2016, 21, 880. [Google Scholar] [CrossRef] [PubMed]
- Moncada, S.; Higgs, E.A. Endogenous nitric oxide: Physiology, pathology and clinical relevance. Eur. J. Clin. Invest. 1991, 21, 361–374. [Google Scholar] [CrossRef] [PubMed]
- Patruno, A.; Franceschelli, S.; Pesce, M.; Maccallini, C.; Fantacuzzi, M.; Speranza, L.; Ferrone, A.; De Lutiis, M.A.; Ricciotti, E.; Amoroso, R.; et al. Novel aminobenzyl-acetamidine derivative modulate the differential regulation of NOSs in LPS induced inflammatory response: Role of PI3K/Akt pathway. Biochim. Biophys. Acta. 2012, 1820, 2095–2104. [Google Scholar] [CrossRef] [PubMed]
- Povsic, T.J.; Najjar, S.S.; Prather, K.; Zhou, J.; Adams, S.D.; Zavodni, K.L.; Kelly, F.; Melton, L.G.; Hasselblad, V.; Heitner, J.F.; et al. EPC mobilization after erythropoietin treatment in acute ST-elevation myocardial infarction: The REVEAL EPC substudy. J. Thromb. Thrombolys. 2013, 36, 375–383. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Sun, G.B.; Sun, X.; Wang, H.W.; Meng, X.B.; Qin, M.; Sun, J.; Luo, Y.; Sun, X.B. Cardioprotective effect of salvianolic acid B against arsenic trioxide-induced injury in cardiac H9c2 cells via the PI3K/Akt signal pathway. Toxicol. Lett. 2013, 216, 100–107. [Google Scholar] [CrossRef] [PubMed]
- Oltvai, Z.N.; Milliman, C.L.; Korsmeyer, S.J. Bcl-2 heterodimerizes in vivo with a conserved homolog, Bax, that accelerates programmed cell death. Cell 1993, 74, 609–619. [Google Scholar] [CrossRef]
- Willis, S.; Day, C.L.; Hinds, M.G.; Huang, D.C. The Bcl-2-regulated apoptotic pathway. J. Cell Sci. 2003, 116, 4053–4056. [Google Scholar] [CrossRef] [PubMed]
- Youle, R.J.; Strasser, A. The BCL-2 protein family: Opposing activities that mediate cell death. Nat. Rev. Mol. Cell Biol. 2008, 9, 47–59. [Google Scholar] [CrossRef] [PubMed]
- Del, P.G.; Venditti, A.; del, P.M.; Maurillo, L.; Buccisano, F.; Tamburini, A.; Cox, M.C.; Franchi, A.; Bruno, A.; Mazzone, C.; et al. Amount of spontaneous apoptosis detected by Bax/Bcl-2 ratio predicts outcome in acute myeloid leukemia (AML). Blood 2003, 101, 2125–2131. [Google Scholar] [CrossRef]
- Shaw, R.J.; Cantley, L.C. Ras, PI(3)K and mTOR signalling controls tumour cell growth. Nature 2006, 441, 424–430. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.; Chen, R.C.; Yang, Z.H.; Sun, G.B.; Wang, M.; Ma, X.J.; Yang, L.J.; Sun, X.B. Taxifolin prevents diabetic cardiomyopathy in vivo and in vitro by inhibition of oxidative stress and cell apoptosis. Food Chem. Toxicol. 2014, 63, 221–232. [Google Scholar] [CrossRef] [PubMed]
- Gu, S.; Zhang, W.; Chen, J.; Ma, R.; Xiao, X.; Ma, X.; Yao, Z.; Chen, Y. EPC-derived microvesicles protect cardiomyocytes from Ang II-induced hypertrophy and apoptosis. PLoS ONE 2014, 9, e85396. [Google Scholar] [CrossRef] [PubMed]
- Gao, M.X.; He, Q.; Han, W.Y.; Zhang, Z.T. Synthesis of 2-amino-4, 5-diarylpyrimidines and their protective effects against oxygen-glucose deprivation in PC12 cells. Acta Pharm. Sin. B 2010, 45, 1123–1127. [Google Scholar]
- Liu, M.H.; Li, G.H.; Peng, L.J.; Qu, S.L.; Zhang, Y.; Peng, J.; Luo, X.Y.; Hu, H.J.; Ren, Z.; Liu, Y.; et al. PI3K/Akt/FoxO3a signaling mediates cardioprotection of FGF-2 against hydrogen peroxide-induced apoptosis in H9c2 cells. Mol. Cell. Biochem. 2016, 414, 57–66. [Google Scholar] [CrossRef] [PubMed]
- Kanazawa, H.; Imoto, K.; Okada, M.; Yamawaki, H. Canstatin inhibits hypoxia-induced apoptosis through activation of integrin/focal adhesion kinase/Akt signaling pathway in H9c2 cardiomyoblasts. PLoS ONE 2017, 12, e173051. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.M.; Hsu, J.H.; Liou, S.F.; Chen, T.J.; Chen, L.Y.; Chiu, C.C.; Yeh, J.L. Baicalein, an active component of Scutellaria baicalensis Georgi, prevents lysophosphatidylcholine-induced cardiac injury by reducing reactive oxygen species production, calcium overload and apoptosis via MAPK pathways. BMC Complement. Altern. Med. 2014, 14, 233. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y.Q.; Chang, G.L.; Wang, Y.; Zhang, D.Y.; Cao, L.; Liu, J. Geniposide Prevents Hypoxia/Reoxygenation-Induced Apoptosis in H9c2 Cells: Improvement of Mitochondrial Dysfunction and Activation of GLP-1R and the PI3K/AKT Signaling Pathway. Cell. Physiol. Biochem. 2016, 39, 407–421. [Google Scholar] [CrossRef] [PubMed]
- Pooja, S.; Pushpanathan, M.; Gunasekaran, P.; Rajendhran, J. Endocytosis-Mediated Invasion and Pathogenicity of Streptococcus agalactiae in Rat Cardiomyocyte (H9C2). PLoS ONE 2015, 10, e139733. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.H.; Chen, Y.; Gao, C.Y.; Cui, Z.T.; Yao, J.M. Protective Effects of MicroRNA-126 on Human Cardiac Microvascular Endothelial Cells Against Hypoxia/Reoxygenation-Induced Injury and Inflammatory Response by Activating PI3K/Akt/eNOS Signaling Pathway. Cell. Physiol. Biochem. 2017, 42, 506–518. [Google Scholar] [CrossRef] [PubMed]
- Mei, X.; Wang, H.X.; Li, J.S.; Liu, X.H.; Lu, X.F.; Li, Y.; Zhang, W.Y.; Tian, Y.G. Dusuqing granules (DSQ) suppress inflammation in Klebsiella pneumonia rat via NF-κB/MAPK signaling. BMC Complement. Altern. Med. 2017, 17, 216. [Google Scholar] [CrossRef] [PubMed]
- Sun, S.; Yang, S.; Dai, M.; Jia, X.; Wang, Q.; Zhang, Z.; Mao, Y. The effect of Astragalus polysaccharides on attenuation of diabetic cardiomyopathy through inhibiting the extrinsic and intrinsic apoptotic pathways in high glucose -stimulated H9C2 cells. BMC Complement. Altern. Med. 2017, 17, 310. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Ichikawa, T.; Villacorta, L.; Janicki, J.S.; Brower, G.L.; Yamamoto, M.; Cui, T. Nrf2 protects against maladaptive cardiac responses to hemodynamic stress. Arterioscler. Throm. Vas. 2009, 29, 1843–1850. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.; Fan, H.; Yang, L.; Shi, L.; Liu, Y. Tyrosol prevents ischemia/reperfusion-induced cardiac injury in H9c2 cells: Involvement of ROS, Hsp70, JNK and ERK, and apoptosis. Molecules 2015, 20, 3758–3775. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are available from the authors. |






| Gene Symbol | Reference Sequence | Primer | Sequence(5′-3′) | Product Size (bp) |
|---|---|---|---|---|
| Akt | NM_033230.2 | Forward | TCACCTCTGAGACCGACACC | 110 |
| Reverse | CCGTTCACTGTCCACACACTC | |||
| eNOS | NM_021838.2 | Forward | GGGATTCTGGCAAGACCGAT | 151 |
| Reverse | TTGTCCAAACACTCCACGCT | |||
| iNOS | NM_012611.3 | Forward | TTCCTCAGGCTTGGGTCTTGT | 213 |
| Reverse | ATCCTGTGTTGTTGGGCTGG | |||
| Bcl-2 | NM_016993.1 | Forward | GGATCCAGGATAACGGAGGC | 141 |
| Reverse | ATGCACCCAGAGTGATGCAG | |||
| Bax | NM_017059.2 | Forward | GGGCCTTTTTGCTACAGGGT | 106 |
| Reverse | TTCTTGGTGGATGCGTCCTG | |||
| GAPDH | NM_017008.3 | Forward | GGCACAGTCAAGGCTGAGAATG | 143 |
| Reverse | ATGGTGGTGAAGACGCCAGTA |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chu, D.; Zhang, Z. Trichosanthis Pericarpium Aqueous Extract Protects H9c2 Cardiomyocytes from Hypoxia/Reoxygenation Injury by Regulating PI3K/Akt/NO Pathway. Molecules 2018, 23, 2409. https://doi.org/10.3390/molecules23102409
Chu D, Zhang Z. Trichosanthis Pericarpium Aqueous Extract Protects H9c2 Cardiomyocytes from Hypoxia/Reoxygenation Injury by Regulating PI3K/Akt/NO Pathway. Molecules. 2018; 23(10):2409. https://doi.org/10.3390/molecules23102409
Chicago/Turabian StyleChu, Donghai, and Zhenqiu Zhang. 2018. "Trichosanthis Pericarpium Aqueous Extract Protects H9c2 Cardiomyocytes from Hypoxia/Reoxygenation Injury by Regulating PI3K/Akt/NO Pathway" Molecules 23, no. 10: 2409. https://doi.org/10.3390/molecules23102409
APA StyleChu, D., & Zhang, Z. (2018). Trichosanthis Pericarpium Aqueous Extract Protects H9c2 Cardiomyocytes from Hypoxia/Reoxygenation Injury by Regulating PI3K/Akt/NO Pathway. Molecules, 23(10), 2409. https://doi.org/10.3390/molecules23102409