Cytotoxic Activity of Origanum Vulgare L. on Hepatocellular Carcinoma cell Line HepG2 and Evaluation of its Biological Activity
Abstract
:1. Introduction
2. Results
2.1. Chemical Composition of Oregano EO
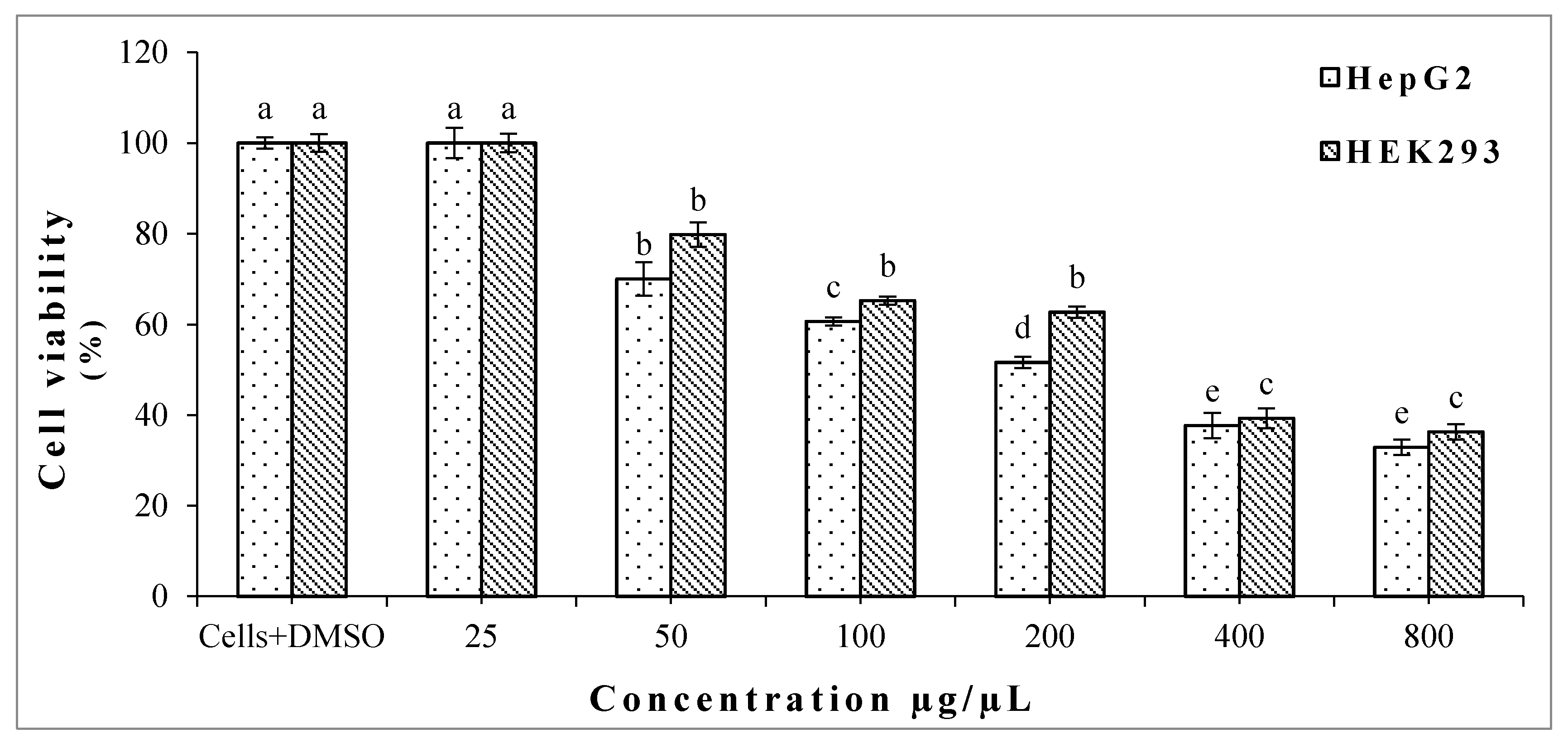
2.2. Cytotoxic Effect of Crude Oregano EO on HepG2 and HEK293 Cells
2.2.1. MTT Assay
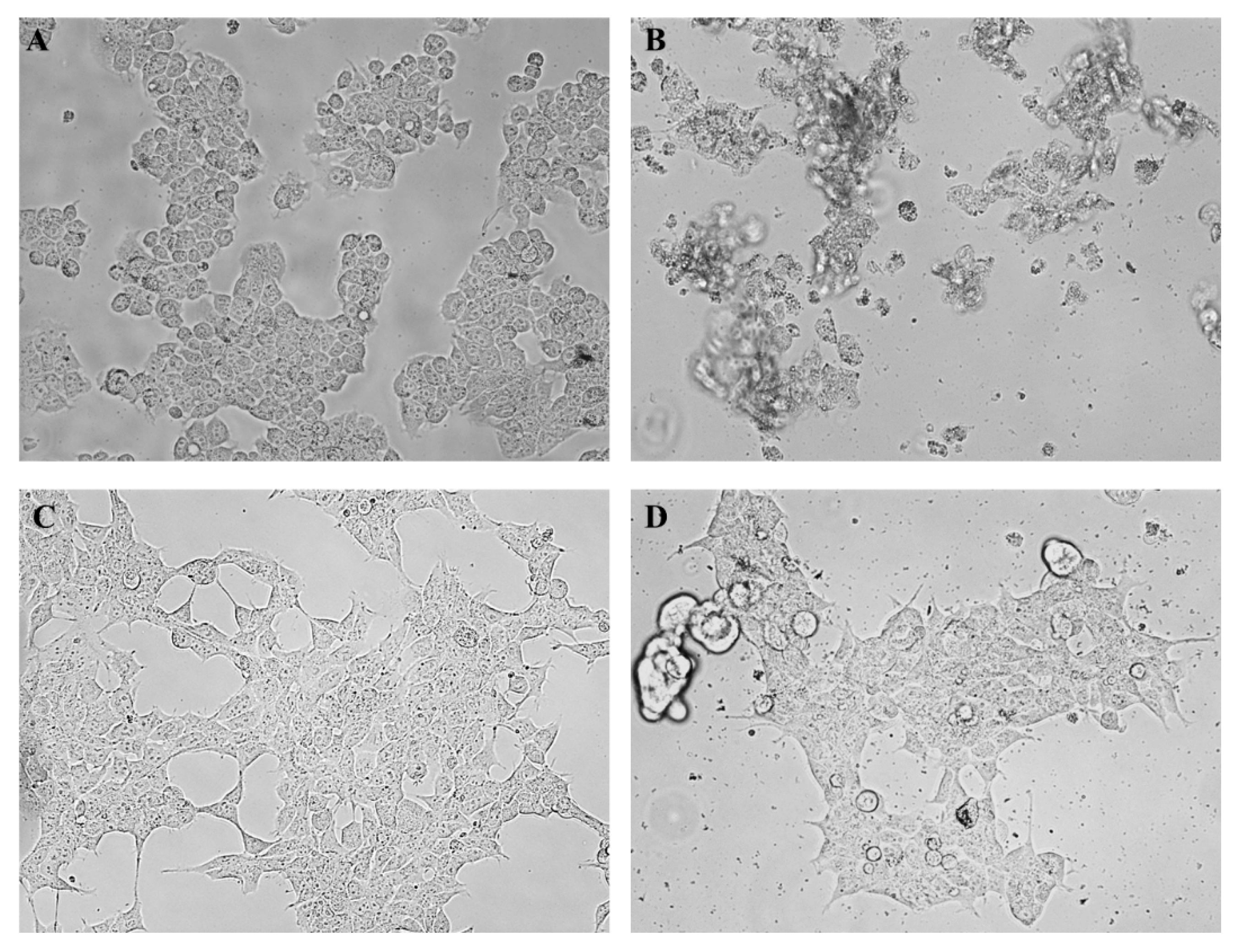
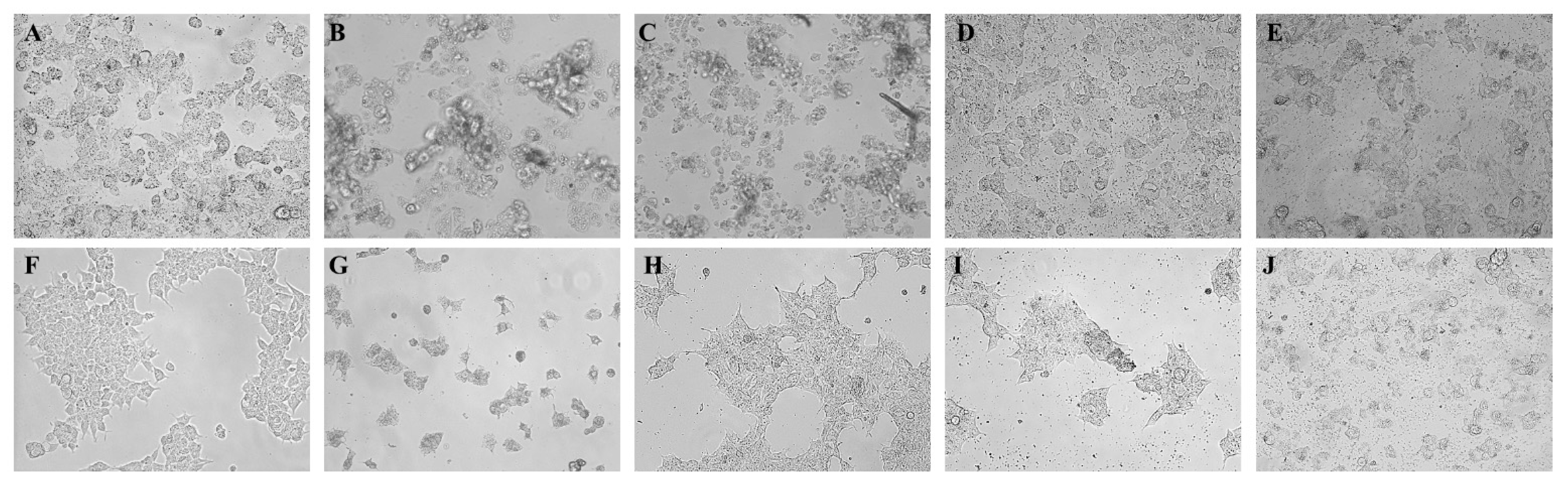
2.2.2. Observation of Morphological Changes
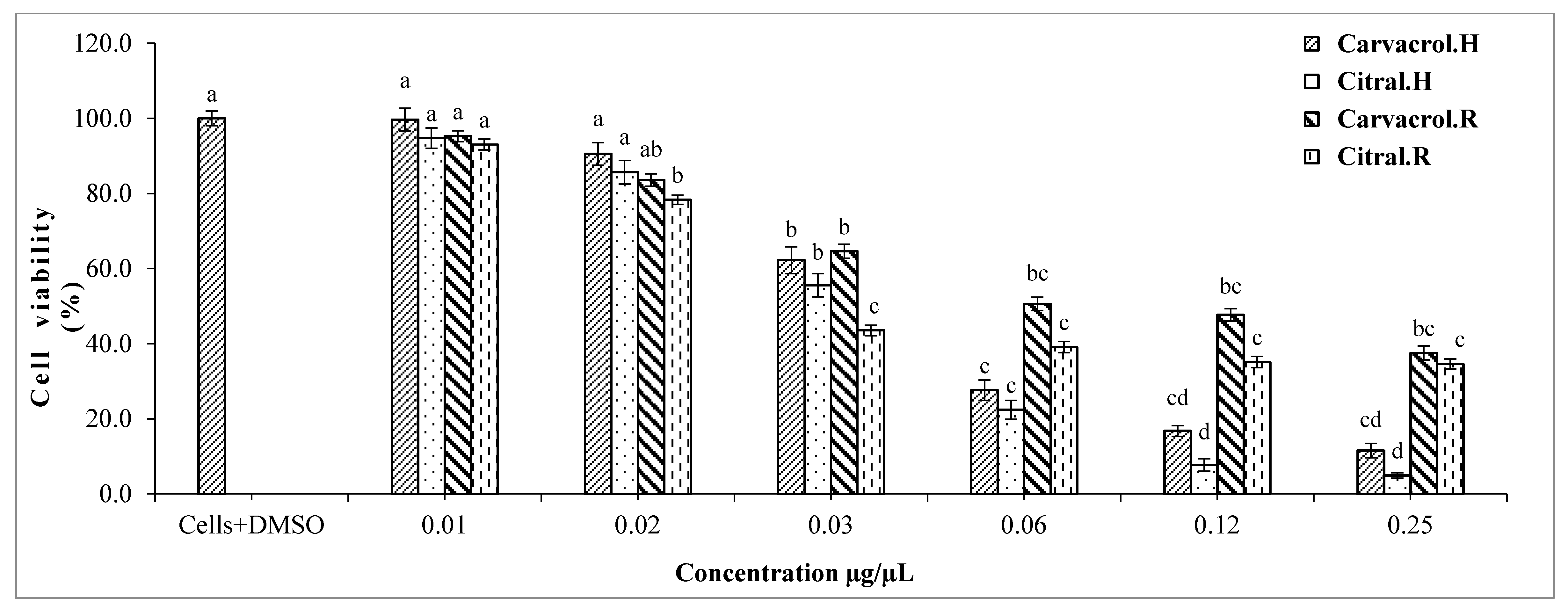
2.3. Cytotoxic Effect of Single Substances on HepG2 Cells
MTT Assay
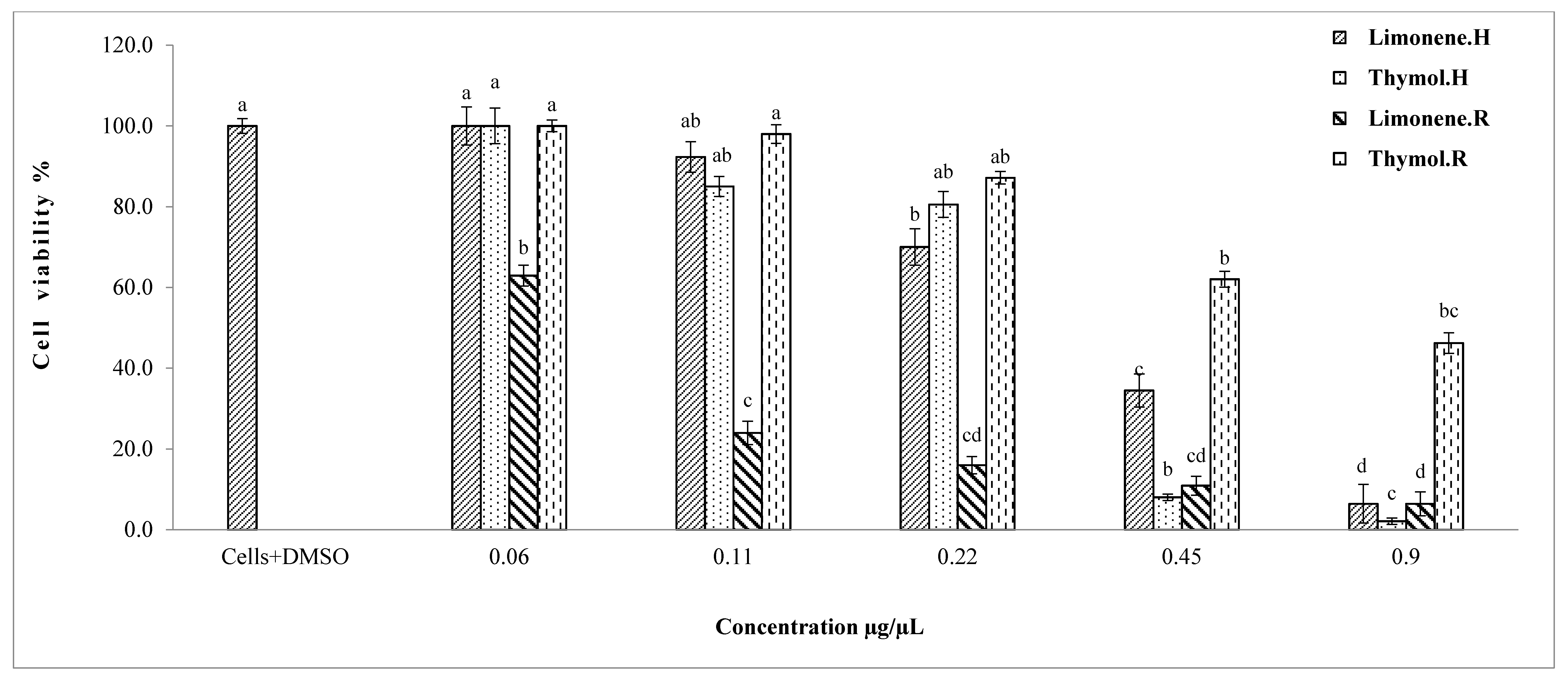
2.4. Cytotoxic Effect of Single Constituents on HEK293 Cells
MTT Assay
2.5. Observation of Morphological Changes
2.6. In Vitro Antibacterial Assay
2.7. Phytotoxic Activity Assay
3. Discussion
4. Materials and Methods
4.1. Chemicals
4.2. Plant Material
4.3. Isolation of the Volatile Oil
4.4. GC-FID Analysis
4.5. GC/MS Analysis
4.6. Identification of the Essential Oil Components
4.7. Cytotoxicity Test
4.7.1. Cell Cultures and Treatments
4.7.2. Cell Viability Assay (MTT Test)
4.7.3. Observation of Morphological Changes
4.8. Antibacterial Activity Test
4.8.1. Tested Bacterial Strains
4.8.2. Bactericidal Assay
4.9. Phytotoxic Activity Test
5. Conclusions
Author Contributions
Conflicts of Interest
References
- Elshafie, H.S.; Mancini, E.; Camele, I.; De Martino, L.; De Feo, V. In vivo antifungal activity of two essential oils from Mediterranean plants against postharvest brown rot disease of peach fruit. Ind. Crops Prod. 2015, 66, 11–15. [Google Scholar] [CrossRef]
- Orafidiya, L.O.; Agbani, E.O.; Oyedele, A.O.; Babalola, O.O.; Onayemi, O. Preliminary clinical tests on topical preparations of Ocimum gratissimum Linn. Leaf essential oil for the treatment of Acne vulgaris. Clin. Drug Investig. 2002, 22, 313–319. [Google Scholar] [CrossRef]
- Elshafie, H.S.; Sakr, S.; Mang, S.M.; De Feo, V.; Camele, I. Antimicrobial activity and chemical composition of three essential oils extracted from Mediterranean aromatic plants. J. Med. Food 2016, 19, 1096–1103. [Google Scholar] [CrossRef] [PubMed]
- Elshafie, H.S.; Ghanney, N.; Mang, S.M.; Ferchichi, A.; Camele, I. An in vitro attempt for controlling severe phyto and human pathogens using essential oils from Mediterranean plants of genus Schinus. J. Med. Food 2016, 19, 266–273. [Google Scholar] [CrossRef] [PubMed]
- Edris, A.E. Pharmaceutical and therapeutic potentials of essential oils and their individual volatile constituents: A review. Phytother. Res. 2007, 21, 308–323. [Google Scholar] [CrossRef] [PubMed]
- Lesgards, J.F.; Baldovini, N.; Vidal, N.; Pietri, S. Anticancer activities of essential oils constituents and synergy with conventional therapies: A review. Phytother. Res. 2014, 28, 1423–1446. [Google Scholar] [CrossRef] [PubMed]
- Vokou, S.; Kokkini, S.; Bessiere, J.M. Geographic variation of Greek oregano (Origanum vulgare ssp. hirtum) essential oils. Biochem. Syst. Ecol. 1993, 21, 287–295. [Google Scholar] [CrossRef]
- Elshafie, H.S.; Camele, I. Chapter 5: Investigating the effects of plant essential oils on post- harvest fruit decay. In Fungal Pathogenicity; Intech: Rijeka, Croatia, 2016; ISBN 978-953-51-4624-7. [Google Scholar]
- Kintzios, S. The Genera Origanum and Lippia, 1st ed.; Taylor & Francis: London, UK, 2004. [Google Scholar]
- Camele, I.; De Feo, V.; Altieri, L.; Mancini, E.; De Martino, L.; Rana, G.L. An attempt of postharvest orange fruit rot control using essential oils from Mediterranean plants. J. Med. Food 2010, 13, 1515–1523. [Google Scholar] [CrossRef] [PubMed]
- Orhan, I.E.; Ozcelik, B.; Kartal, M.; Kan, Y. Antimicrobial and antiviral effects of essential oils from selected Umbelliferae and Labiatae plants and individual essential oil components. Turk. J. Biol. 2012, 36, 239–246. [Google Scholar]
- Kazemi, M.; Mousavi, E.; Bandrez, N. Chemical compositions and antibacterial activity of the essential oils of Thymus vulgaris and Tanacetum parthenium. Res. J. Soil Biol. 2012, 4, 21–31. [Google Scholar] [CrossRef]
- Elshafie, H.S.; Mancini, E.; Sakr, S.; De Martino, L.; Mattia, C.A.; De Feo, V.; Camele, I. Antifungal activity of some constituents of Origanum vulgare L. essential oil against postharvest disease of peach fruit. J. Med. Food 2015, 18, 929–934. [Google Scholar] [CrossRef] [PubMed]
- Mancini, E.; Camele, I.; Elshafie, H.S.; De Martino, L.; Pellegrino, C.; Grulova, D.; De Feo, V. Chemical composition and biological activity of the essential oil of Origanum vulgare ssp. hirtum from different areas in the southern Apennines (Italy). Chem. Biodiver. 2014, 11, 639–651. [Google Scholar] [CrossRef] [PubMed]
- Camele, I.; Altieri, L.; De Martino, L.; De Feo, V.; Mancini, E.; Rana, G.L. In vitro control of post-harvest fruit rot fungi by some plant essential oil components. Int. J. Mol. Sci. 2012, 13, 2290–2300. [Google Scholar] [CrossRef] [PubMed]
- Adebayo, O.; Dang, T.; Bélanger, A.; Khanizadeh, S. Antifungal studies of selected essential oils and a commercial formulation against Botrytis cinerea. J. Food Res. 2013, 2, 217–226. [Google Scholar] [CrossRef]
- Russo, M.; Galletti, G.C.; Bocchini, P.; Carnacini, A. Essential oil chemical composition of wild populations of Italian oregano spice (Origanum vulgare ssp. hirtum (Link) Ietswaart): A preliminary evaluation of their use in chemotaxonomy by cluster analysis. 1. Inflorescences. J. Agric. Food Chem. 1998, 46, 3741–3746. [Google Scholar] [CrossRef]
- Mockute, D.; Bernotiene, G.; Judzentiene, A. The essential oil of Origanum vulgare L. ssp. vulgare growing wild in Vilnius district (Lithuania). Phytochemistry 2001, 57, 65–69. [Google Scholar] [CrossRef]
- D’Antuono, F.L.; Galletti, G.C.; Bocchini, P. Variability of essential oil content and composition of Origanum vulgare L. populations from a north Mediterranean area (Liguria Region, Northern Italy). Ann. Bot. 2000, 86, 471–478. [Google Scholar] [CrossRef]
- Pérez, R.A.; Navarro, T.; de Lorenzo, C. HS–SPME analysis of the volatile compounds from spices as a source of flavour in ‘Campo Real’ table olive preparations. Flavour Frag. J. 2007, 22, 265–273. [Google Scholar] [CrossRef]
- El Babili, F.; Bouajila, J.; Souchard, J.P.; Bertrand, C.; Bellvert, F.; Fouraste, I.; Moulis, C.; Valentin, A. Oregano: Chemical analysis and evaluation of its antimalarial, antioxidant, and cytotoxic activities. J. Food Sci. 2011, 76, 512–518. [Google Scholar] [CrossRef] [PubMed]
- Bakkali, F.; Averbeck, S.; Averbeck, D.; Zhiri, A.; Baudoux, D.; Idaomar, M. Antigenotoxic effects of three essential oils in diploid yeast Saccharomyces cerevisiae after treatments with UVC radiation, 8-MOP plus UVA and MMS. Mut. Res. 2006, 606, 27–28. [Google Scholar] [CrossRef] [PubMed]
- Mezzoug, N.; Elhadri, A.; Dallouh, A.; Amkiss, S.; Skali, S.; Abrini, J.; Zhiri, A.; Baudoux, D.; Diallo, B.; El Jaziri, M.; et al. Investigation of the mutagenic and antimutagenic effects of Origanum compactum essential oil and some of its constituents. Mutat. Res. Genet. Toxicol. Environ. 2007, 629, 100–110. [Google Scholar] [CrossRef] [PubMed]
- Kokkini, S.; Karousou, R.; Dardioti, A.; Krigas, S.; Lanarasm, T. Autumn essential oils of Greek oregano. Phytochemistry 1997, 44, 883–886. [Google Scholar] [CrossRef]
- Russo, R.; Corasaniti, M.T.; Bagetta, G.; Morrone, L.A. Exploitation of Cytotoxicity of Some Essential Oils for Translation in Cancer Therapy. Evid. Based Complement. Alternat. Med. 2015, 2015, 397821. [Google Scholar] [CrossRef] [PubMed]
- Lam, L.K.T.; Zheng, B. Effects of essential oils of glutathione S-transferase activity in mice. J. Agric. Food Chem. 1991, 39, 660–662. [Google Scholar] [CrossRef]
- Coccimiglio, J.; Alipour, M.; Jiang, Z.H.; Gottardo, C.; Suntres, Z. Antioxidant, Antibacterial, and Cytotoxic Activities of the Ethanolic Origanum vulgare Extract and Its Major Constituents. Oxid. Med. Cell. Longev. 2016, 2016, 8. [Google Scholar] [CrossRef] [PubMed]
- Grbović, F.; Stanković, M.S.; Ćurčić, M.; Đorđević, N.; Šeklić, D.; Topuzović, M.; Marković, S. In Vitro Cytotoxic Activity of Origanum vulgare L. on HCT-116 and MDA-MB-231 Cell Lines. Plants 2013, 2, 371–378. [Google Scholar] [CrossRef] [PubMed]
- Sivropoulou, A.; Papanikolaou, E.; Nikolaou, C.; Kokkini, S.; Lanaras, T.; Arsenakis, M. Antimicrobial and cytotoxic activities of Origanum essential oils. J. Agric. Food Chem. 1996, 44, 1202–1205. [Google Scholar] [CrossRef]
- Armentano, M.F.; Bisaccia, F.; Miglionico, R.; Russo, D.; Nolfi, N.; Carmosino, M.; Andrade, P.B.; Valentão, P.; Diop, M.S.; Milella, L. Antioxidant and proapoptotic activities of Sclerocarya birrea [(A. Rich.) Hochst.] methanolic root extract on the hepatocellular carcinoma cell line HepG2. BioMed Res. Int. 2015, 2015, 11. [Google Scholar] [CrossRef] [PubMed]
- Tai, C.J.; Wang, C.K.; Chang, Y.J.; Lin, C.S.; Tai, C.J. Aqueous Extract of Solanum nigrum Leaves Induces Autophagy and Enhances Cytotoxicity of Cisplatin, Doxorubicin, Docetaxel, and 5-Fluorouracil in Human Colorectal Carcinoma Cells. Evid Based Complement Alternat Med. 2013, 2013, 10. [Google Scholar]
- Venkatesan, T.; Choi, Y.W.; Kim, Y.K. The cytotoxic nature of Acanthopanax sessiliflorus stem bark extracts in human breast cancer cells. Saudi J. Biol. Sci. 2015, 22, 752–759. [Google Scholar]
- Tuttolomondo, T.; La Bella, S.; Licata, M.; Virga, G.; Leto, C.; Saija, A.; Trombetta, D.; Tomaino, A.; Speciale, A.; Napoli, E.M.; et al. Biomolecular characterization of wild sicilian oregano: Phytochemical screening of essential oils and extracts, and evaluation of their antioxidant activities. Chem. Biodiver. 2013, 10, 411–433. [Google Scholar] [CrossRef] [PubMed]
- De Martino, L.; D’Arena, G.; Minervini, M.M.; Deaglio, S.; Fusco, B.; Cascavilla, N.; De Feo, V. Verbena officinalis essential oil and its component citral as apoptotic-inducing agent in chronic lympocytic leukemia. Int. J. Immunopath. Pharmacol. 2009, 22, 1097–1104. [Google Scholar] [CrossRef] [PubMed]
- De Martino, L.; D’Arena, G.; Minervini, M.M.; Deaglio, S.; Sinisi, N.P.; Cascavilla, P.; De Feo, V. Active caspase-3 detection to evaluate apoptosis induced by Verbena officinalis essential oil and citral in chronic lymphocytic leukaemia cells. Braz. J. Pharmacogn. 2011, 21, 869–873. [Google Scholar] [CrossRef]
- De Martino, L.; Iorio, M.; Coppola, G.; Campana, A.; Savastano, C.; Fusco, B.M.; De Feo, V. Verbena officinalis essential oil and citral as apoptotic inductors in leukocytes of healthy subjects and chronic myeloid leukemic patients. Pharmacologyonline 2008, 170–175. [Google Scholar]
- Di Mola, A.; Massa, A.; De Feo, V.; Basile, A.; Pascale, M.; Aquino, R.P.; de Caprariis, P. Antiproliferative effect of citral and citral-related compounds on pancreatic and human B-lymphoma cell lines. Med. Chem. Res. 2017, 26, 631–639. [Google Scholar] [CrossRef]
- Yilar, M.; Bayan, Y.; Aksit, H.; Onaran, A.; Kadioglu, I.; Yanar, Y. Bioherbicidal Effects of Essential Oils Isolated from Thymus fallax F., Mentha dumetorum Schult. and Origanum vulgare L. Asian J. Chem. 2013, 25, 4807. [Google Scholar]
- Angelini, L.G.; Carpanese, G.; Cioni, P.L.; Morelli, I.; Macchia, M.; Flamini, G. Essential Oils from Mediterranean Lamiaceae as Weed Germination Inhibitors. J. Agric. Food Chem. 2003, 51, 6158–6164. [Google Scholar] [CrossRef] [PubMed]
- Council of Europe. European Pharmacopoeia, 5th ed.; Council of Europe: Strasbourg CEDEX, France, 2004; Volume I, p. 217. [Google Scholar]
- Jennings, W.; Shibamoto, T. Qualitative Analysis of flavour and fragrance volatiles by glass capillary gas chromatography; Academic Press: New York, NY, USA, 1980. [Google Scholar]
- Davies, N.W. Gas chromatographic retention indices of monoterpenes and sesquiterpenes on methyl silicone and Carbowax 20M phases. J. Chromatogr. 1990, 503, 1–24. [Google Scholar] [CrossRef]
- Goodner, K.L. Practical retention index models of OV-101, DB-1, DB-5, and DB-Wax for flavor and fragrance compounds. Lwt-Food Sci. Technol. 2008, 41, 951–958. [Google Scholar] [CrossRef]
- Adams, R.P. Identification of Essential Oil Components by Gas Chromatography/Mass Spectroscopy, 4th ed.; Allured Publishing Corporation: Carol Stream, IL, USA, 2007. [Google Scholar]
- Wiley Registry of Mass Spectral Data, with NIST Spectral Data CD Rom, 7th ed.; John Wiley & Sons: New York, NY, USA, 1998.
- Gerbino, A.; Schena, G.; Milano, S.; Milella, L.; Barbosa, A.F.; Armentano, F.; Procino, G.; Svelto, M.; Carmosino, M. Spilanthol from Acmella Oleracea lowers the intracellular levels of cAMP impairing NKCC2 phosphorylation and water channel AQP2 membrane expression in mouse kidney. PLoS ONE 2016, 11, e0156021. [Google Scholar] [CrossRef] [PubMed]
- Bhunia, M.C.; Johnson, B. Ray Purificaction, characterization and antimicrobial spectrum of a bacteriocin produced by Pediococcus acidolactici. J. Appl. Bacteriol. 1988, 65, 261–268. [Google Scholar] [CrossRef] [PubMed]
- King, E.O.; Ward, M.K.; Raney, D.E. Two simple media for demonstration of pyocyanin and fluorescin. J. Lab. Clin. Med. 1954, 44, 301–307. [Google Scholar] [PubMed]
Sample Availability: Samples of the compounds are not available from the authors. |
| No. | Identified Substances | Ki (a) | Ki (b) | % (c) | Identification (d) |
|---|---|---|---|---|---|
| 1 | α-Pinene | 921 | 1032 | 0.1 | 1,2,3 |
| 2 | Sabinene | 966 | 1132 | t | 1,2 |
| 3 | 1-Octen-3-ol | 974 | 1154 | 0.1 | 1,2 |
| 4 | Octan-3-one | 981 | 1253 | t | 1,2 |
| 5 | 3-Octanol | 990 | 1393 | t | 1,2 |
| 6 | α-Phellandrene | 997 | 1150 | t | 1,2,3 |
| 7 | α-Terpinene | 1010 | 1189 | t | 1,2,3 |
| 8 | p-Cymene | 1018 | 1269 | t | 1,2,3 |
| 9 | Limonene | 1022 | 1205 | 1.3 | 1,2,3 |
| 10 | 1,8-Cineole | 1024 | 1213 | 0.1 | 1,2,3 |
| 11 | (e)-β-Ocimene | 1044 | 1262 | t | 1,2 |
| 12 | γ-Terpinene | 1054 | 1256 | t | 1,2,3 |
| 13 | cis-Sabinene hydrate | 1062 | 1556 | 0.3 | 1,2 |
| 14 | Terpinolene | 1083 | 1265 | t | 1,2 |
| 15 | Methyl benzoate | 1089 | t | 1,2 | |
| 16 | trans-Sabinene hydrate | 1093 | 1474 | t | 1,2 |
| 17 | endo-Fencol | 1115 | 1120 | 0.1 | 1,2 |
| 18 | 3-Thujanol | 1155 | t | 1,2 | |
| 19 | Borneol | 1160 | 1719 | t | 1,2,3 |
| 20 | Terpinen-4-ol | 1173 | 1611 | 0.8 | 1,2,3 |
| 21 | γ-Terpineol | 1209 | 1718 | 0.7 | 1,2,3 |
| 22 | trans-Carveol | 1214 | 1845 | t | 1,2 |
| 23 | Thymol methyl ether | 1231 | 1607 | 0.9 | 1,2 |
| 24 | Carvacrol methyl ether | 1241 | 1975 | 1.4 | 1,2 |
| 25 | Linalyl acetate | 1253 | 1665 | t | 1,2 |
| 26 | (e)-Citral | 1270 | 1727 | 2.5 | 1,2 |
| 27 | trans-Carvone oxide | 1280 | t | 1,2 | |
| 28 | Thymol and Carvacrol | 1283 | 21982239 | 74.8 | 1,2,3 |
| 29 | δ-Elemene | 1337 | 1476 | 0.1 | 1,2 |
| 30 | α-Cubebene | 1349 | 1466 | 0.1 | 1,2 |
| 31 | Thymol acetate | 1354 | 1867 | 0.1 | 1,2 |
| 32 | Eugenol | 1358 | 2186 | t | 1,2 |
| 33 | Piperitenone oxide | 1365 | 1983 | t | 1,2 |
| 34 | Cyclosativene | 1370 | 1492 | 0.1 | 1,2 |
| 35 | α-Ylangene | 1375 | 1493 | 0.2 | 1,2 |
| 36 | β-Bourbonene | 1384 | 1535 | 0.1 | 1,2 |
| 37 | β-Elemene | 1390 | 1598 | 0.1 | 1,2 |
| 38 | (e)-β-Damascone | 1413 | 1830 | t | 1,2 |
| 39 | trans-Caryophyllene | 1419 | 1612 | 0.4 | 1,2,3 |
| 40 | β-Copaene | 1428 | 0.2 | 1,2 | |
| 41 | trans-α-Bergamotene | 1434 | 1568 | 0.1 | 1,2 |
| 42 | α-Guaiene | 1437 | 0.1 | 1,2 | |
| 43 | Aromadendrene | 1442 | 1628 | 0.4 | 1,2 |
| 44 | α-Humulene | 1453 | 1689 | 0.4 | 1,2,3 |
| 45 | allo-Aromadendrene | 1458 | 1661 | 0.2 | 1,2,3 |
| 46 | cis-Cadina-1(6),4-diene | 1462 | 0.1 | 1,2 | |
| 47 | γ-Gurjunene | 1474 | 1687 | 0.4 | 1,2 |
| 48 | γ-Muurolene | 1480 | 1704 | 0.4 | 1,2 |
| 49 | Valencene | 1493 | 1741 | 0.2 | 1,2 |
| 50 | α-Muurolene | 1498 | 1740 | 0.1 | 1,2 |
| 51 | Germacrene A | 1505 | 1499 | 1.1 | 1,2 |
| 52 | δ-Amorphene | 1511 | 0.2 | 1,2 | |
| 53 | δ-Cadinene | 1523 | 1773 | 1.0 | 1,2 |
| 54 | Zonarene | 1530 | 1729 | 0.1 | 1,2 |
| 55 | α-Cadinene | 1535 | 1743 | 0.1 | 1,2 |
| 56 | α-Calacorene | 1540 | 1942 | 0.1 | 1,2 |
| 57 | β-Calacorene | 1561 | 1942 | 0.1 | 1,2 |
| 58 | Germacrene-d-4-ol | 1573 | 2069 | 0.1 | 1,2 |
| 59 | (−)-Spatulenol | 1574 | 2150 | 0.3 | 1,2,3 |
| 60 | Caryophyllene oxide | 1581 | 2008 | 0.3 | 1,2,3 |
| 61 | β-Atlantol | 1606 | 0.1 | 1,2 | |
| 62 | α-Muurolol | 1640 | 0.2 | 1,2 | |
| 63 | Cubenol | 1645 | 2080 | 0.1 | 1,2 |
| 64 | α-Cadinol | 1653 | 2255 | 0.4 | 1,2 |
| 65 | Oplopanone | 1734 | 2568 | t | 1,2 |
| 66 | Z-Lanceol | 1759 | 0.1 | 1,2 | |
| 67 | Khusinol acetate | 1815 | t | 1,2 | |
| 68 | Cedranediol-8S-13 | 1896 | 0.2 | 1,2 | |
| 69 | Columellarin | 1948 | t | 1,2 | |
| 70 | Ethyl hexadecanoate | 1989 | t | 1,2 | |
| 71 | Methyl linoleate | 2106 | t | 1,2 | |
| Total | 91.4 | ||||
| Monoterpene Hydrocarbons | 1.7 | ||||
| Oxygenated Monoterpenes | 4.2 | ||||
| Sesquiterpene Hydrocarbons | 5.8 | ||||
| Oxygenated Sesquiterpenes | 1.5 | ||||
| Phenolic compounds | 77.2 | ||||
| Other compounds | 0.1 |
| Single Substances | Trend-Line Equation | IC50 (mg/L) ± SDs |
|---|---|---|
| Limonene | Y = 24.51X − 12.89 | 294.3 ± 10.5 |
| Thymol | Y = 27.28X − 26.70 | 289.5 ± 6.6 |
| Carvacrol | Y = 19.90X − 18.25 | 48.3 ± 6.7 |
| Citral | Y = 20.47X − 26.49 | 35.5 ± 4.5 |
| Single Substances | Trend-Line Equation | IC50 (mg/L) ± SDs |
|---|---|---|
| Thymol | Y = 14.56X + 35 | 939.8 ± 29.6 |
| Limonene | Y = 12.61X − 13.80 | 120.3 ± 12.0 |
| Carvacrol | Y = 8.91X + 27.96 | 90.5 ± 9.9 |
| Citral | Y = 6.58X + 23.41 | 32.0 ± 5.1 |
| Diameter of Inhibition Zones (mm) | ||||
|---|---|---|---|---|
| Treatments | Conc (%) | G+ve | G−ve | |
| B. meg Act. | B. meg 100 | E. coli | ||
| Oregano EO | 50 | 40.0 ± 3.0a | 43.0 ± 3.2a | 22.0 ± 2.4a |
| 25 | 28.0 ± 1.5b | 31.0 ± 1.9b | 15.0 ± 1.3b | |
| Carvacrol | 50 | 45.0 ± 4.0a | 45.0 ± 2.8a | 20.0 ± 2.2a |
| 25 | 30.0 ± 2.2b | 35.0 ± 2.1b | 11.0 ± 1.7b | |
| Thymol | 50 | 30.0 ± 2.7b | 35.0 ± 3.2b | 20.0 ± 1.9a |
| 25 | 20.0 ± 1.4c | 0.0 ± 0.0e | 0.0 ± 0.0c | |
| Limonene | 50 | 22.0 ± 3.5c | 27.0 ± 1.7c | 15.0 ± 1.1b |
| 25 | 15.0 ± 2.7d | 17.0 ± 1.1d | 0.0 ± 0.0c | |
| Citral | 50 | 45.0 ± 25a | 45.0 ± 2.7a | 25.0 ± 2.7a |
| 25 | 40.0 ± 1.8a | 40.0 ± 1.8a | 15.0 ± 1.4b | |
| Doses | G.S. ± SD | R.E. ± SD | G.I. % | |
|---|---|---|---|---|
| Sinapis Arvensis | Control | 6.9 ± 0.7 | 1.4 ± 0.5 | 100.0 ± 0.0 |
| 0.062 µg/mL | 7.7 ± 0.6 | 1.3 ± 1.2 | 103.6 ± 4.6 | |
| 0.125 µg/mL | 6.3 ± 0.4 | 1.7 ± 1.0 | 110.9 ± 6.0 | |
| 0.25 µg/mL | 7.7 ± 0.7 | 1.9 ± 1.0 | 151.4 ± 8.0 | |
| 0.625 µg/mL | 7.0 ± 0.9 | 1.8 ± 0.8 | 130.4 ± 6.0 | |
| 1.25 µg/mL | 6.7 ± 0.4 | 1.5 ± 1.2 | 104.0 ± 4.0 | |
| 2.5 µg/mL | 6.7 ± 0.5 | 1.0 ± 0.8 | 69.4 ± 6.7 * | |
| Phalaris Canariensis | Control | 6.9 ± 0.7 | 1.4 ± 0.5 | 100.0 ± 0.0 |
| 0.062 µg/mL | 7.7 ± 0.6 | 1.3 ± 1.2 | 79.5 ± 5.4 ** | |
| 0.125 µg/mL | 6.3 ± 0.4 | 1.7 ± 1.0 | 107.6 ± 9.7 | |
| 0.25 µg/mL | 7.7 ± 0.7 | 1.9 ± 1.0 | 83.7 ± 6.4 | |
| 0.625 µg/mL | 7.0 ± 0.9 | 1.8 ± 0.8 | 95.3 ± 4.6 | |
| 1.25 µg/mL | 6.7 ± 0.4 | 1.5 ± 1.2 | 100.5 ± 5.4 | |
| 2.5 µg/mL | 6.7 ± 0.5 | 1.0 ± 0.8 | 83.7 ± 3.7 | |
| Lepidium Sativum | Control | 6.2 ± 1.0 | 0.8 ± 0.3 | 100.0 ± 8.0 |
| 0.062 µg/mL | 6.0 ± 1.0 | 0.8 ± 0.3 | 96.8 ± 6.0 | |
| 0.125 µg/mL | 4.3 ± 1.0 * | 0.4 ± 0.1 | 34.7 ± 4.2 * | |
| 0.25 µg/mL | 4.3 ± 0.5 * | 0.5 ± 0.2 | 43.34 ± 5.7 | |
| 0.625 µg/mL | 5.3 ± 0.9 | 0.6 ± 0.2 | 64.1 ± 8.0 | |
| 1.25 µg/mL | 5.3 ± 0.9 | 0.7 ± 0.1 | 74.8 ± 6.1 | |
| 2.5 µg/mL | 5.3 ± 0.7 | 0.7 ± 0.3 | 74.8 ± 4.9 | |
| Raphanus Sativus | Control | 6.9 ± 1.0 | 2.6 ± 0.8 | 100.0 ± 8.0 |
| 0.062 µg/mL | 3.3 ± 1.2 | 2.1 ± 1.7 | 67.3 ± 4.5 | |
| 0.125 µg/mL | 2.7 ± 0.8 * | 1.1 ± 0.3 * | 28.9 ± 6.2 * | |
| 0.25 µg/mL | 5.7 ± 0.4 | 1.5 ± 0.7 | 83.1 ± 8.4 | |
| 0.625 µg/mL | 2.3 ± 1.0 * | 1.5 ± 0.4 | 33.5 ± 5.4 * | |
| 1.25 µg/mL | 3.3 ± 0.8 | 1.6 ± 0.1 | 51.3 ± 4.5 | |
| 2.5 µg/mL | 3.3 ± 0.9 | 1.6 ± 0.4 | 51.3 ± 3.1 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Elshafie, H.S.; Armentano, M.F.; Carmosino, M.; Bufo, S.A.; De Feo, V.; Camele, I. Cytotoxic Activity of Origanum Vulgare L. on Hepatocellular Carcinoma cell Line HepG2 and Evaluation of its Biological Activity. Molecules 2017, 22, 1435. https://doi.org/10.3390/molecules22091435
Elshafie HS, Armentano MF, Carmosino M, Bufo SA, De Feo V, Camele I. Cytotoxic Activity of Origanum Vulgare L. on Hepatocellular Carcinoma cell Line HepG2 and Evaluation of its Biological Activity. Molecules. 2017; 22(9):1435. https://doi.org/10.3390/molecules22091435
Chicago/Turabian StyleElshafie, Hazem S., Maria F. Armentano, Monica Carmosino, Sabino A. Bufo, Vincenzo De Feo, and Ippolito Camele. 2017. "Cytotoxic Activity of Origanum Vulgare L. on Hepatocellular Carcinoma cell Line HepG2 and Evaluation of its Biological Activity" Molecules 22, no. 9: 1435. https://doi.org/10.3390/molecules22091435
APA StyleElshafie, H. S., Armentano, M. F., Carmosino, M., Bufo, S. A., De Feo, V., & Camele, I. (2017). Cytotoxic Activity of Origanum Vulgare L. on Hepatocellular Carcinoma cell Line HepG2 and Evaluation of its Biological Activity. Molecules, 22(9), 1435. https://doi.org/10.3390/molecules22091435










