Acetylation of Microcrystalline Cellulose by Transesterification in AmimCl/DMSO Cosolvent System
Abstract
1. Introduction
2. Results and Discussion
2.1. Transesterification of Microcrystalline Cellulose (MCC)
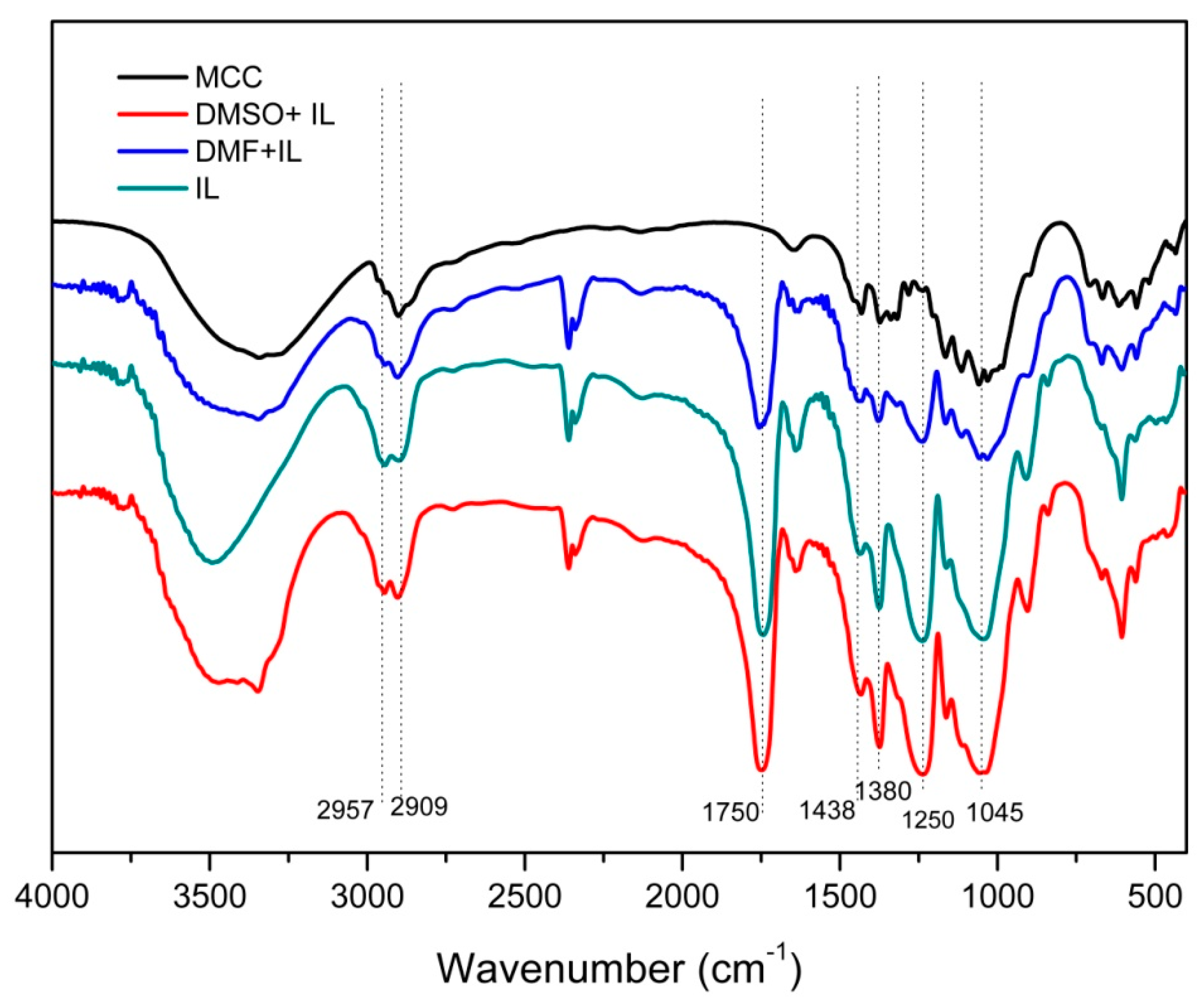
2.2. FT-IR Spectra
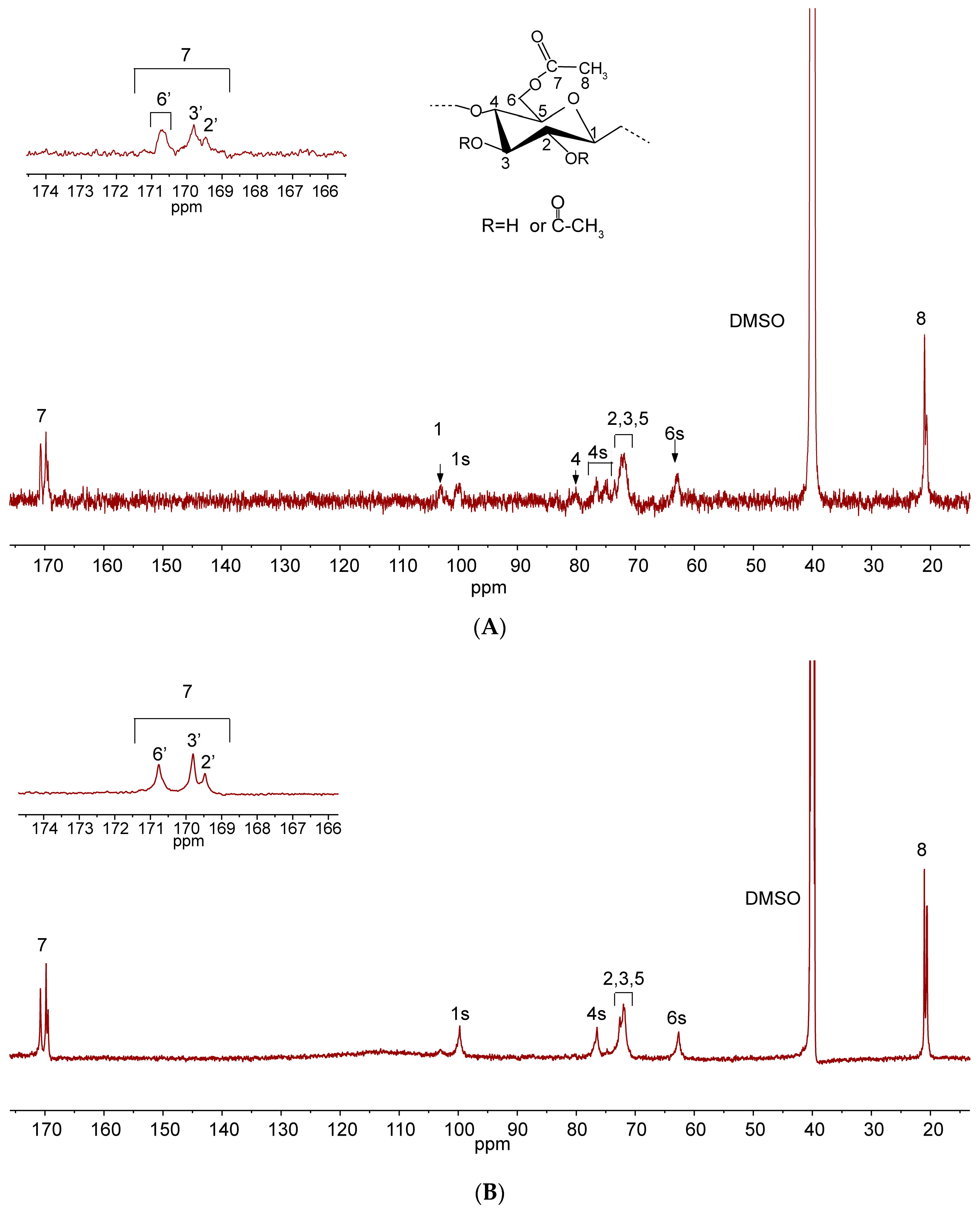
2.3. NMR Analysis
2.4. TGA/DTG Analysis
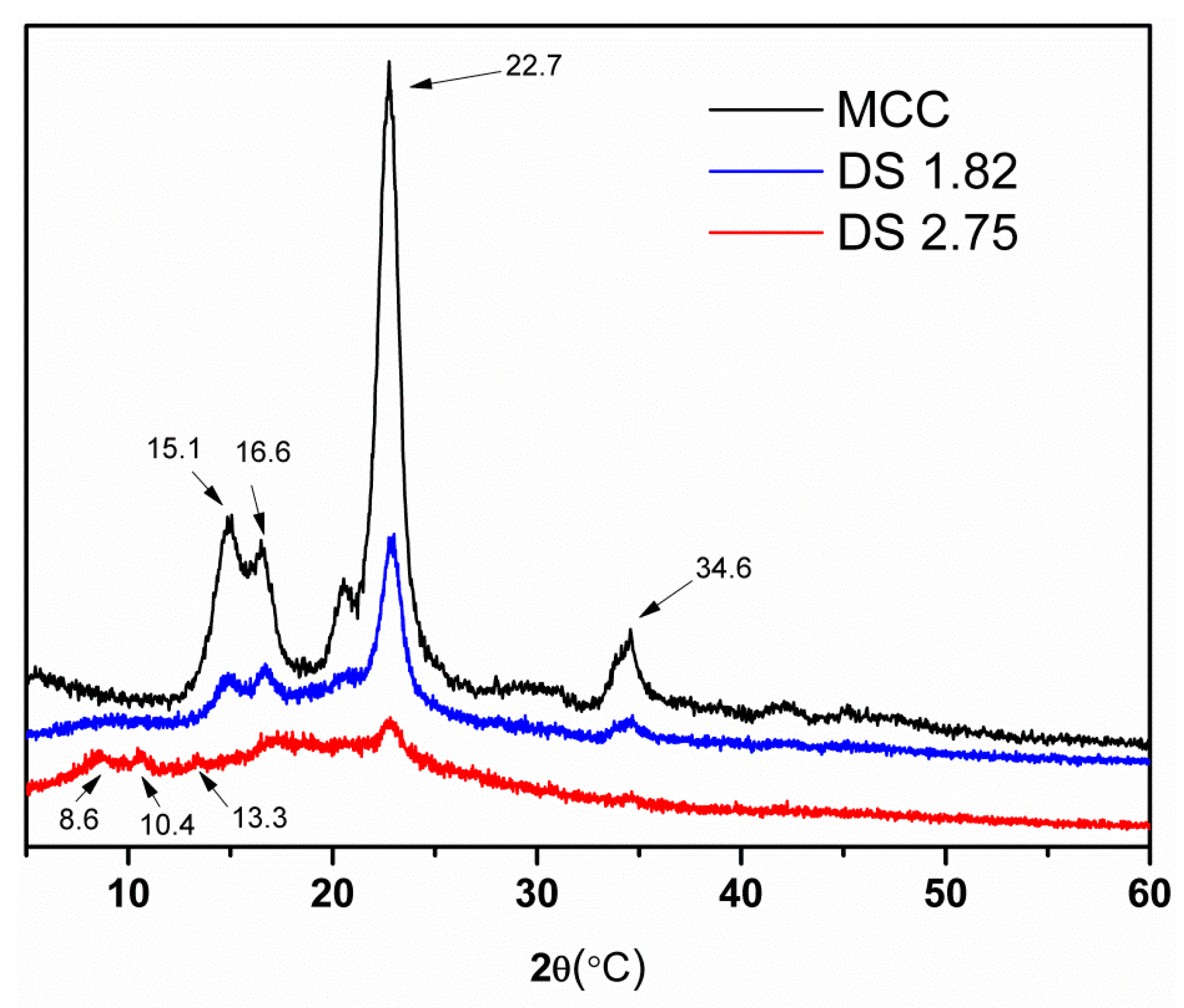
2.5. XRD
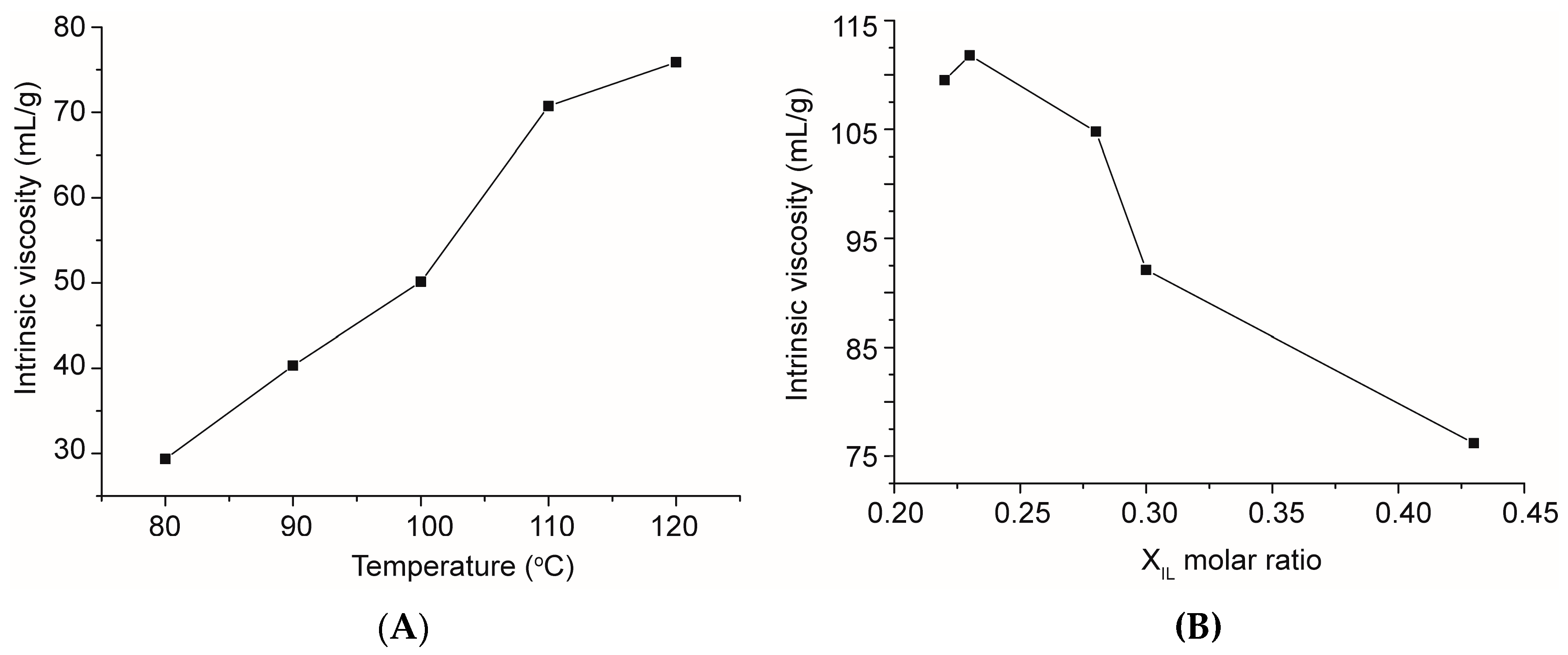
2.6. Intrinsic Viscosity Analysis
3. Materials and Methods
3.1. Materials
3.2. Synthesis of CA via Transesterification
3.3. Characterization
3.3.1. FT-IR
3.3.2. XRD
3.3.3. TGA/DTG
3.3.4. NMR Analysis
3.3.5. Determination of DS
3.3.6. Intrinsic Viscosity Analysis
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Heinze, T.; Liebert, T. 4.2 Chemical characteristics of cellulose acetate. Macromol. Symp. 2004, 208, 167–238. [Google Scholar] [CrossRef]
- Bilal, M.; Asgher, M.; Iqbal, H.M.; Hu, H.; Zhang, X. Biotransformation of lignocellulosic materials into value-added products—A review. Int. J. Biol. Macromol. 2017, 98, 447–458. [Google Scholar] [CrossRef] [PubMed]
- Tang, Z.; Fu, Y.; Ma, Z. Bovine serum albumin as an effective sensitivity enhancer for peptide-based amperometric biosensor for ultrasensitive detection of prostate specific antigen. Biosens. Bioelectron. 2017, 94, 394–399. [Google Scholar] [CrossRef] [PubMed]
- Longsheng, Z.; Wei, F.; Tianxi, L. Flexible hierarchical membranes of WS 2 nanosheets grown on graphene-wrapped electrospun carbon nanofibers as advanced anodes for highly reversible lithium storage. Nanoscale 2016, 8, 16387–16394. [Google Scholar]
- Cai, J.; Zhang, L. Rapid dissolution of cellulose in LiOH/UREA and NaOH/UREA aqueous solutions. Macromol. Biosci. 2005, 5, 539–548. [Google Scholar] [CrossRef] [PubMed]
- Vitz, J.; Erdmenger, T.; Haensch, C.; Schubert, U.S. Extended dissolution studies of cellulose in imidazolium based ionic liquids. Green Chem. 2009, 11, 417–424. [Google Scholar] [CrossRef]
- Schlufter, K.; Schmauder, H.-P.; Dorn, S.; Heinze, T. Efficient homogeneous chemical modification of bacterial cellulose in the ionic liquid 1-N-butyl-3-methylimidazolium chloride. Macromol. Rapid Commun. 2006, 27, 1670–1676. [Google Scholar] [CrossRef]
- Wu, J.; Zhang, J.; Zhang, H.; He, J.S.; Ren, Q.; Guo, M. Homogeneous acetylation of cellulose in a new ionic liquid. Biomacromolecules 2004, 5, 266–268. [Google Scholar] [CrossRef] [PubMed]
- Pinkert, A.; Marsh, K.N.; Pang, S.; Staiger, M.P. Ionic liquids and their interaction with cellulose. Chem. Rev. 2009, 109, 6712–6728. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Chen, X.; Su, M.; Ye, J.; Hong, J.; Yang, Z. Direct production of all-wood plastics by kneading in ionic liquids/DMSO. Chem. Eng. J. 2015, 279, 136–142. [Google Scholar] [CrossRef]
- Gu, S.; Wang, J.; Wei, X.; Cui, H.; Wu, X.; Wu, F. Enhancement of lipase-catalyzed synthesis of caffeic acid phenethyl ester in ionic liquid with dmso co-solvent. Chin. J. Chem. Eng. 2014, 22, 1314–1321. [Google Scholar] [CrossRef]
- Jogunola, O.; Eta, V.; Hedenstrom, M.; Sundman, O.; Salmi, T.; Mikkola, J.P. Ionic liquid mediated technology for synthesis of cellulose acetates using different co-solvents. Carbohydr. Polym. 2016, 135, 341–348. [Google Scholar] [CrossRef] [PubMed]
- Rinaldi, R. Instantaneous dissolution of cellulose in organic electrolyte solutions. Chem. Commun. (Camb.) 2011, 47, 511–513. [Google Scholar] [CrossRef] [PubMed]
- Gale, E.; Wirawan, R.H.; Silveira, R.L.; Pereira, C.S.; Johns, M.A.; Skaf, M.S.; Scott, J.L. Directed discovery of greener cosolvents: New cosolvents for use in ionic liquid based organic electrolyte solutions for cellulose dissolution. ACS Sustain. Chem. Eng. 2016, 4, 6200–6207. [Google Scholar] [CrossRef]
- Kakuchi, R.; Ito, R.; Nomura, S.; Abroshan, H.; Ninomiya, K.; Ikai, T.; Maeda, K.; Kim, H.J.; Takahashi, K. A mechanistic insight into the organocatalytic properties of imidazolium-based ionic liquids and a positive co-solvent effect on cellulose modification reactions in an ionic liquid. RSC Adv. 2017, 7, 9423–9430. [Google Scholar] [CrossRef]
- Cao, Y.; Wu, J.; Zhang, J.; Li, H.; Zhang, Y.; He, J. Room temperature ionic liquids (RTILS): A new and versatile platform for cellulose processing and derivatization. Chem. Eng. J. 2009, 147, 13–21. [Google Scholar] [CrossRef]
- Kilpeläinen, I.; Xie, H.; King, A.; Granstrom, M.; Heikkinen, S.; Argyropoulos, D.S. Dissolution of wood in ionic liquids. J. Agric. Food Chem. 2007, 55, 9142–9148. [Google Scholar] [CrossRef] [PubMed]
- Schenzel, A.; Hufendiek, A.; Barner-Kowollik, C.; Meier, M.A.R. Catalytic transesterification of cellulose in ionic liquids: Sustainable access to cellulose esters. Green Chem. 2014, 16, 3266. [Google Scholar] [CrossRef]
- Sung Jun, L.; Hae Sung, L.; Sang Won, J.; Hyun-Chul, K.; Se Geun, L.; Tae Hwan, O. Effect of dimethyl sulfoxide on synthesis of thermoplastic cellulose-graft-poly(l-lactide) copolymer using ionic liquid as reaction media. J. Appl. Polym. Sci. 2015, 132, 41331–41337. [Google Scholar]
- Casarano, R.; El Seoud, O.A. Successful application of an ionic liquid carrying the fluoride counter-ion in biomacromolecular chemistry: Microwave-assisted acylation of cellulose in the presence of 1-allyl-3-methylimidazolium fluoride/DMSO mixtures. Macromol. Biosci. 2013, 13, 191–202. [Google Scholar] [CrossRef] [PubMed]
- Cao, X.; Sun, S.; Peng, X.; Zhong, L.; Sun, R.; Jiang, D. Rapid synthesis of cellulose esters by transesterification of cellulose with vinyl esters under the catalysis of NaOH or KOH in DMSO. J. Agric. Food Chem. 2013, 61, 2489–2495. [Google Scholar] [CrossRef] [PubMed]
- Achtel, C.; Heinze, T. Homogeneous acetylation of cellulose in the new solvent triethyloctylammonium chloride in combination with organic liquids. Macromol. Chem. Phys. 2016, 217, 2041–2048. [Google Scholar] [CrossRef]
- Celebioglu, A.; Demirci, S.; Uyar, T. Cyclodextrin-grafted electrospun cellulose acetate nanofibers via “click” reaction for removal of phenanthrene. Appl. Surf. Sci. 2014, 305, 581–588. [Google Scholar] [CrossRef]
- Kono, H.; Hashimoto, H.; Shimizu, Y. NMR characterization of cellulose acetate: Chemical shift assignments, substituent effects, and chemical shift additivity. Carbohydr. Polym. 2015, 118, 91–100. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Chen, Y.; Wei, Y.; Zhang, A.; Liu, C. Homogeneous esterification mechanism of bagasse modified with phthalic anhydride in ionic liquid, Part 2: Reactive behavior of hemicelluloses. Carbohydr. Polym. 2017, 157, 1365–1373. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Xu, J.; Wang, K.; Cao, X.; Sun, R. Cellulose acetate fibers prepared from different raw materials with rapid synthesis method. Carbohydr. Polym. 2016, 137, 685–692. [Google Scholar] [CrossRef] [PubMed]
- Barud, H.S.; de Araujo Junior, A.M.; Santos, D.B.; de Assuncao, R.M.N.; Meireles, C.S.; Cerqueira, D.A.; Rodrigues Filho, G.; Ribeiro, C.A.; Messaddeq, Y.; Ribeiro, S.J.L. Thermal behavior of cellulose acetate produced from homogeneous acetylation of bacterial cellulose. Thermochim. Acta 2008, 471, 61–69. [Google Scholar] [CrossRef]
- Tosh, B. Thermal analysis of cellulose esters prepared from different molecular weight fractions of high alpha-cellulose pulp. Indian J. Chem. Technol. 2011, 18, 451–457. [Google Scholar]
- Ass, B.A.; Ciacco, G.T.; Frollini, E. Cellulose acetates from linters and sisal: Correlation between synthesis conditions in DMAC/LiCL and product properties. Bioresour. Technol. 2006, 97, 1696–1702. [Google Scholar] [CrossRef] [PubMed]
- Elomaa, M. Determination of the degree of substitution of acetylated starch by hydrolysis, 1H NMR and TGA/IR. Carbohydr. Polym. 2004, 57, 261–267. [Google Scholar] [CrossRef]
- Sun, X.; Lu, C.; Zhang, W.; Tian, D.; Zhang, X. Acetone-soluble cellulose acetate extracted from waste blended fabrics via ionic liquid catalyzed acetylation. Carbohydr. Polym. 2013, 98, 405–411. [Google Scholar] [CrossRef] [PubMed]
- Pamies, R.; Hernandez Cifre, J.G.; Lopez Martinez, M.D.C.; Garcia de la Torre, J. Determination of intrinsic viscosities of macromolecules and nanoparticles, Comparison of single-point and dilution procedures. Colloid Polym. Sci. 2008, 286, 1223–1231. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are not avaliable from the authors. |

| Entry | T (°C) | Time (h) | Solvent a | DS | Solubility b | ||
|---|---|---|---|---|---|---|---|
| DMSO | DMF | Acetone | |||||
| CA-1 | 80 | 3 | IL0.6/DMSO0.4 | 1.82 | ++ | + | − |
| CA-2 | 90 | 3 | IL0.6/DMSO0.4 | 1.97 | ++ | + | − |
| CA-3 | 100 | 3 | IL0.6/DMSO0.4 | 2.16 | ++ | + | − |
| CA-4 | 110 | 3 | IL0.6/DMSO0.4 | 2.31 | ++ | + | ++ |
| CA-5 | 120 | 3 | IL0.6/DMSO0.4 | 2.38 | ++ | + | ++ |
| CA-6 | 100 | 3 | IL0.6/DMF0.4 | 1.82 | ++ | + | − |
| CA-7 | 110 | 3 | IL0.6/DMF0.4 | 2.01 | ++ | + | + |
| CA-8 | 120 | 3 | IL0.6/DMF0.4 | 2.06 | ++ | + | + |
| CA-9 | 100 | 3 | IL | 1.84 | ++ | + | − |
| CA-10 | 110 | 3 | IL | 2.24 | ++ | + | ++ |
| CA-11 | 120 | 3 | IL | 2.35 | ++ | + | ++ |
| CA-12 | 120 | 6 | IL | 2.36 | ++ | + | ++ |
| CA-13 | 120 | 6 | IL0.43/DMSO0.57 | 2.38 | ++ | + | ++ |
| CA-14 | 120 | 6 | IL0.3/DMSO0.7 | 2.51 | ++ | + | ++ |
| CA-15 | 120 | 6 | IL0.28/DMSO0.72 | 2.59 | ++ | + | ++ |
| CA-16 | 120 | 6 | IL0.23/DMSO0.77 | 2.75 | ++ | + | ++ |
| CA-17 | 120 | 6 | IL0.22/DMSO0.78 | 2.71 | ++ | + | ++ |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, H.; Wen, X.; Zhang, X.; Liu, C. Acetylation of Microcrystalline Cellulose by Transesterification in AmimCl/DMSO Cosolvent System. Molecules 2017, 22, 1419. https://doi.org/10.3390/molecules22091419
Wang H, Wen X, Zhang X, Liu C. Acetylation of Microcrystalline Cellulose by Transesterification in AmimCl/DMSO Cosolvent System. Molecules. 2017; 22(9):1419. https://doi.org/10.3390/molecules22091419
Chicago/Turabian StyleWang, Huihui, Xiaoxiang Wen, Xueqin Zhang, and Chuanfu Liu. 2017. "Acetylation of Microcrystalline Cellulose by Transesterification in AmimCl/DMSO Cosolvent System" Molecules 22, no. 9: 1419. https://doi.org/10.3390/molecules22091419
APA StyleWang, H., Wen, X., Zhang, X., & Liu, C. (2017). Acetylation of Microcrystalline Cellulose by Transesterification in AmimCl/DMSO Cosolvent System. Molecules, 22(9), 1419. https://doi.org/10.3390/molecules22091419






