A Novel Synthesis of the Efficient Anti-Coccidial Drug Halofuginone Hydrobromide
Abstract
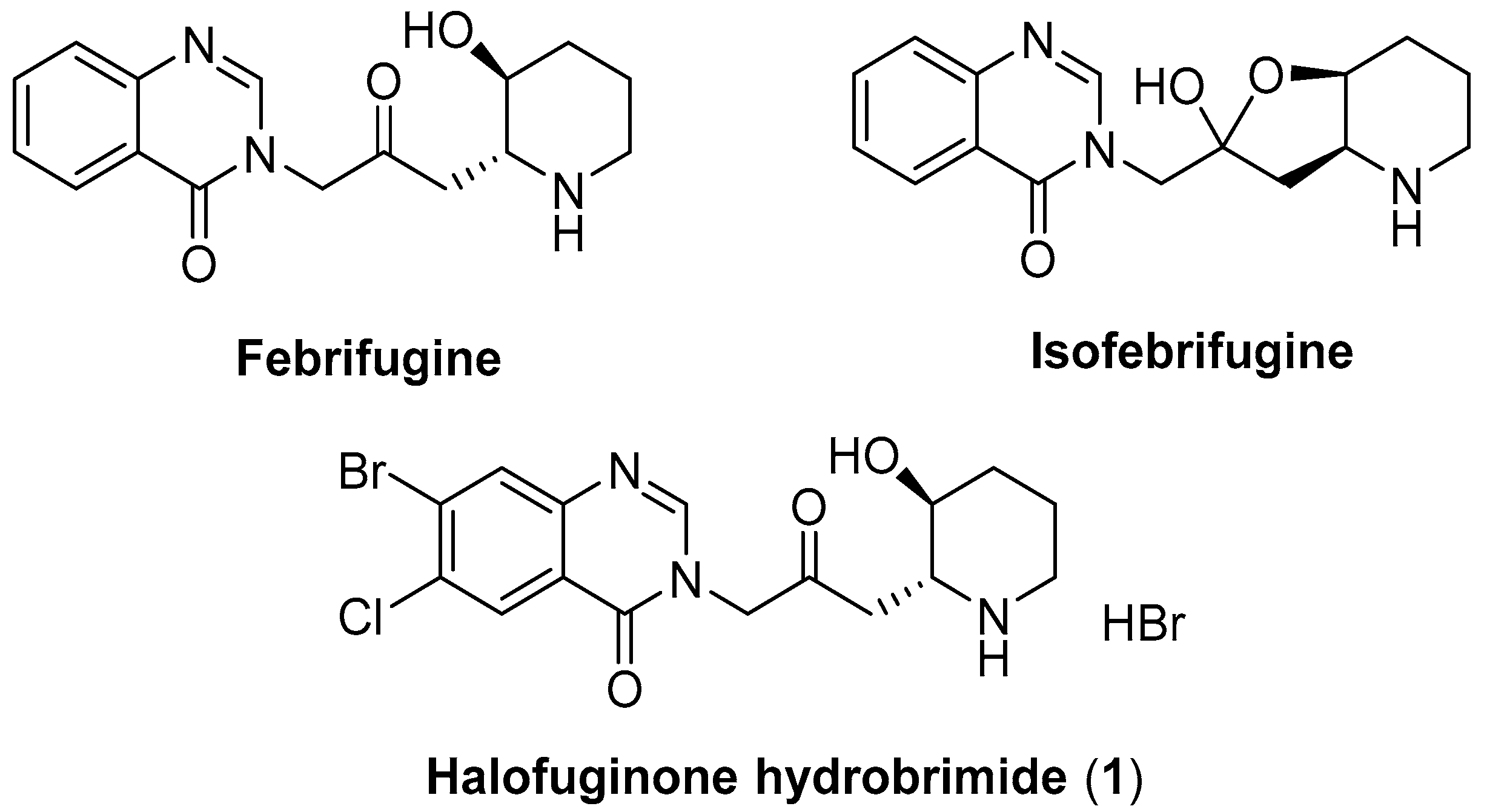
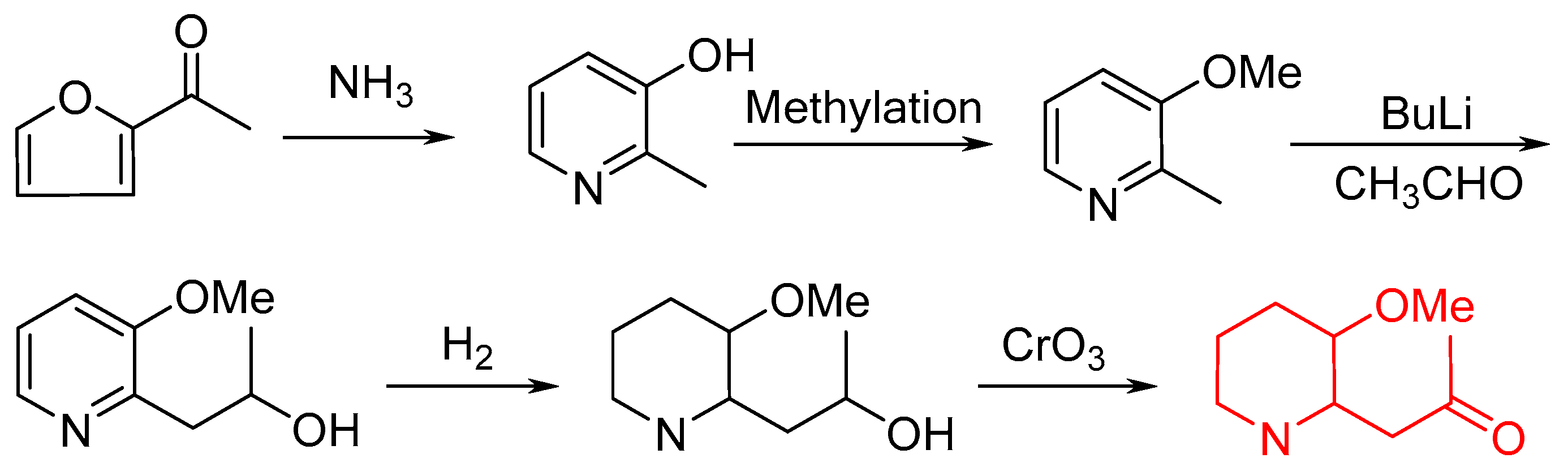
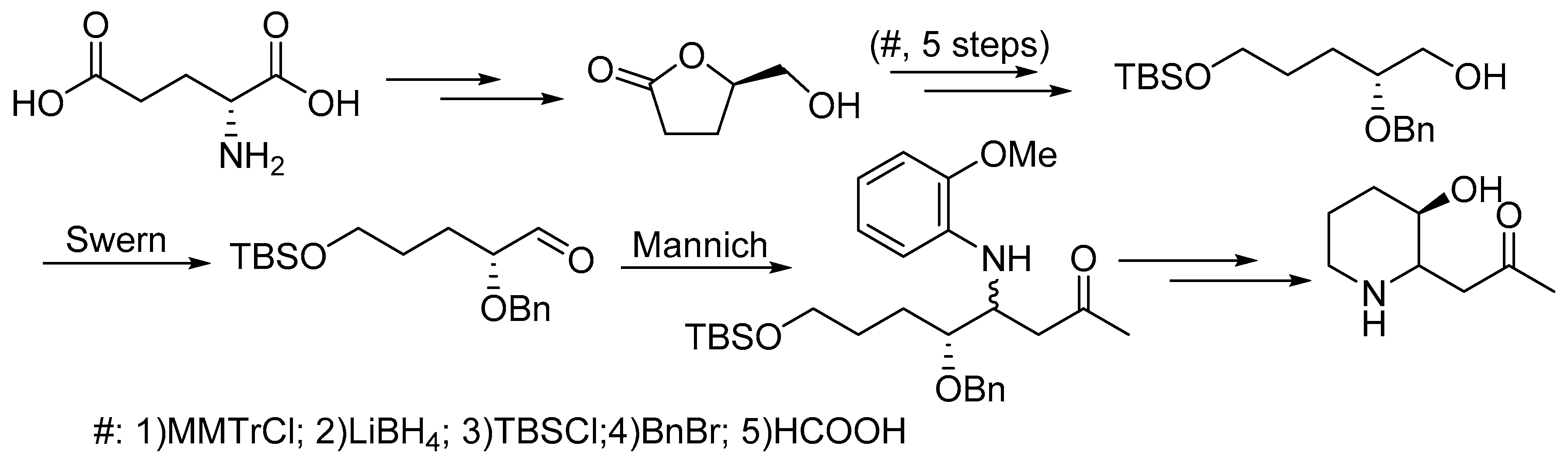
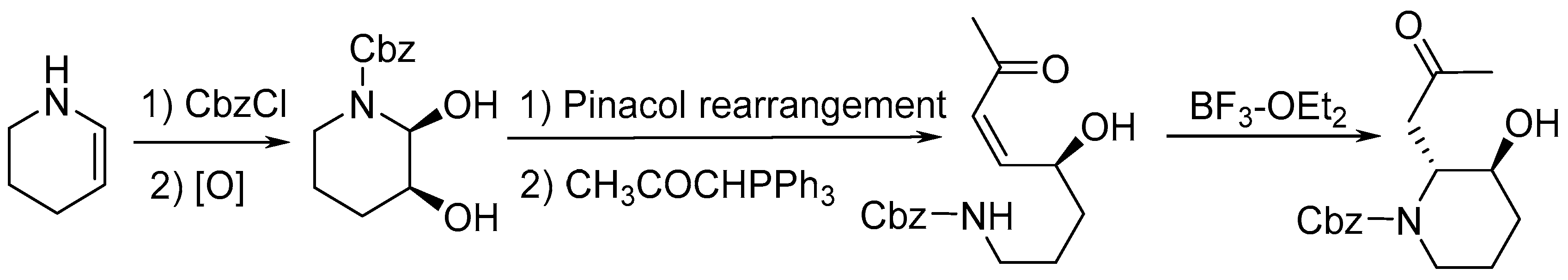
:1. Introduction
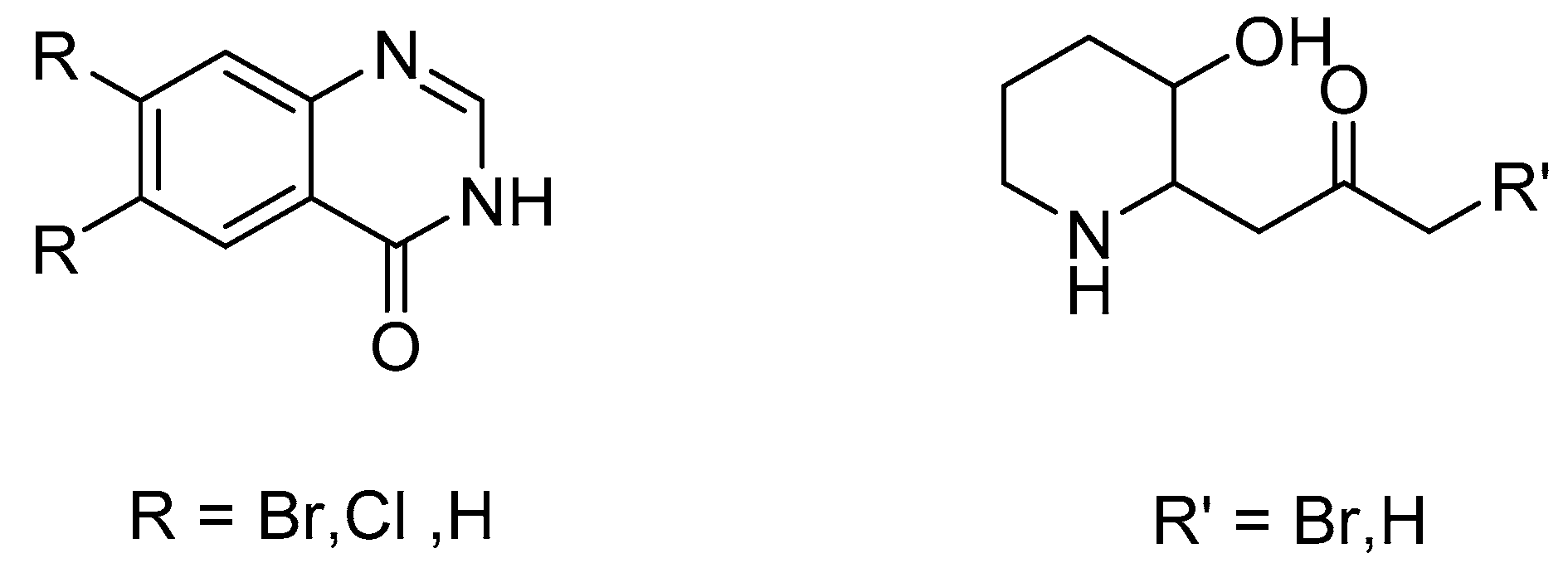
2. Results and Discussion
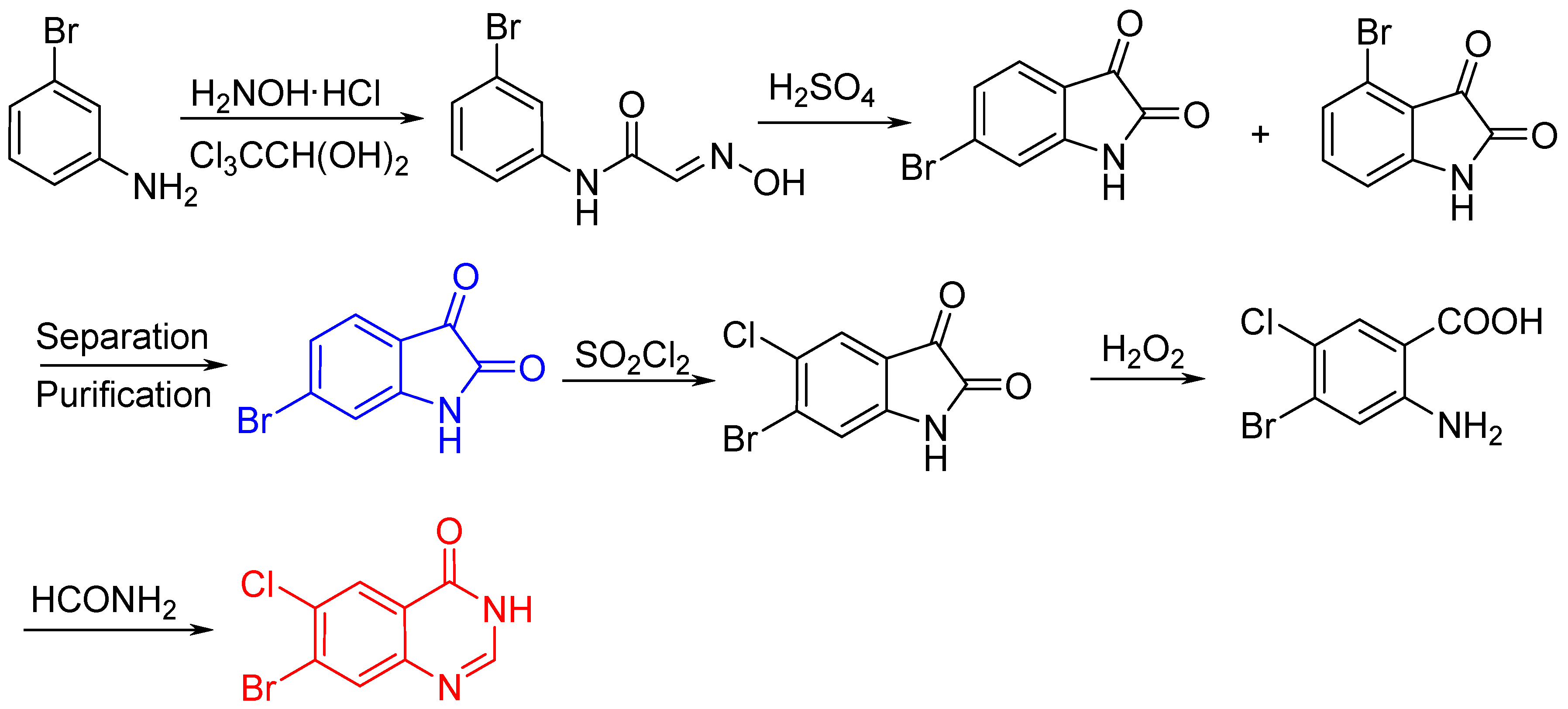
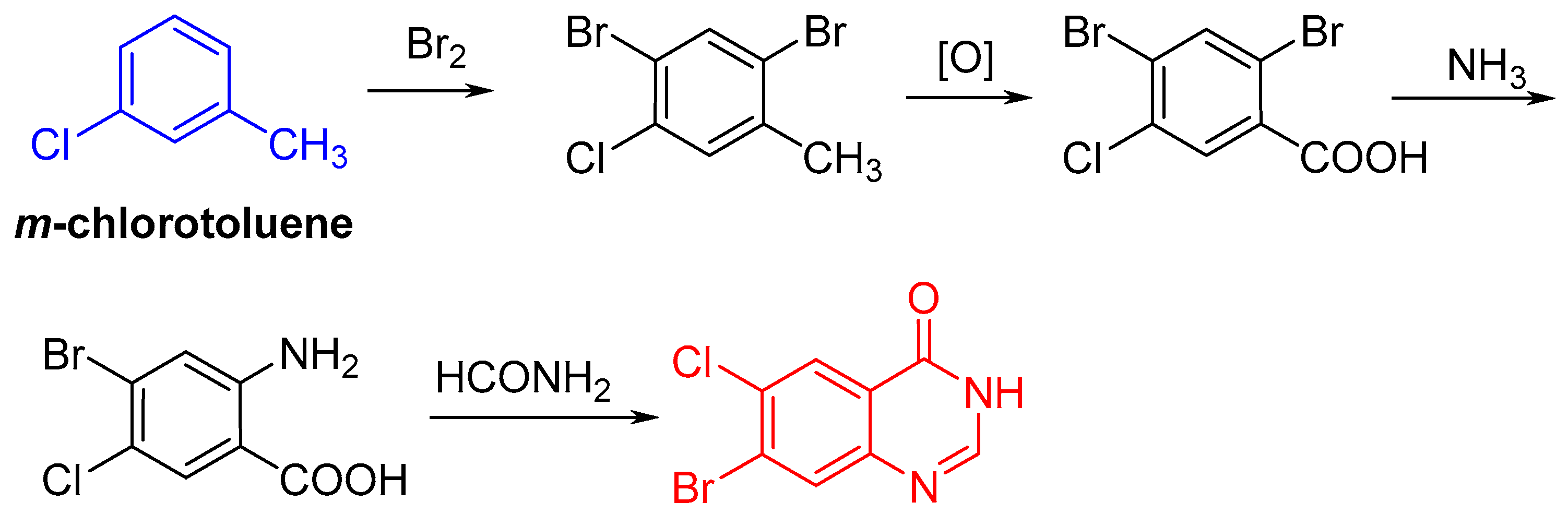
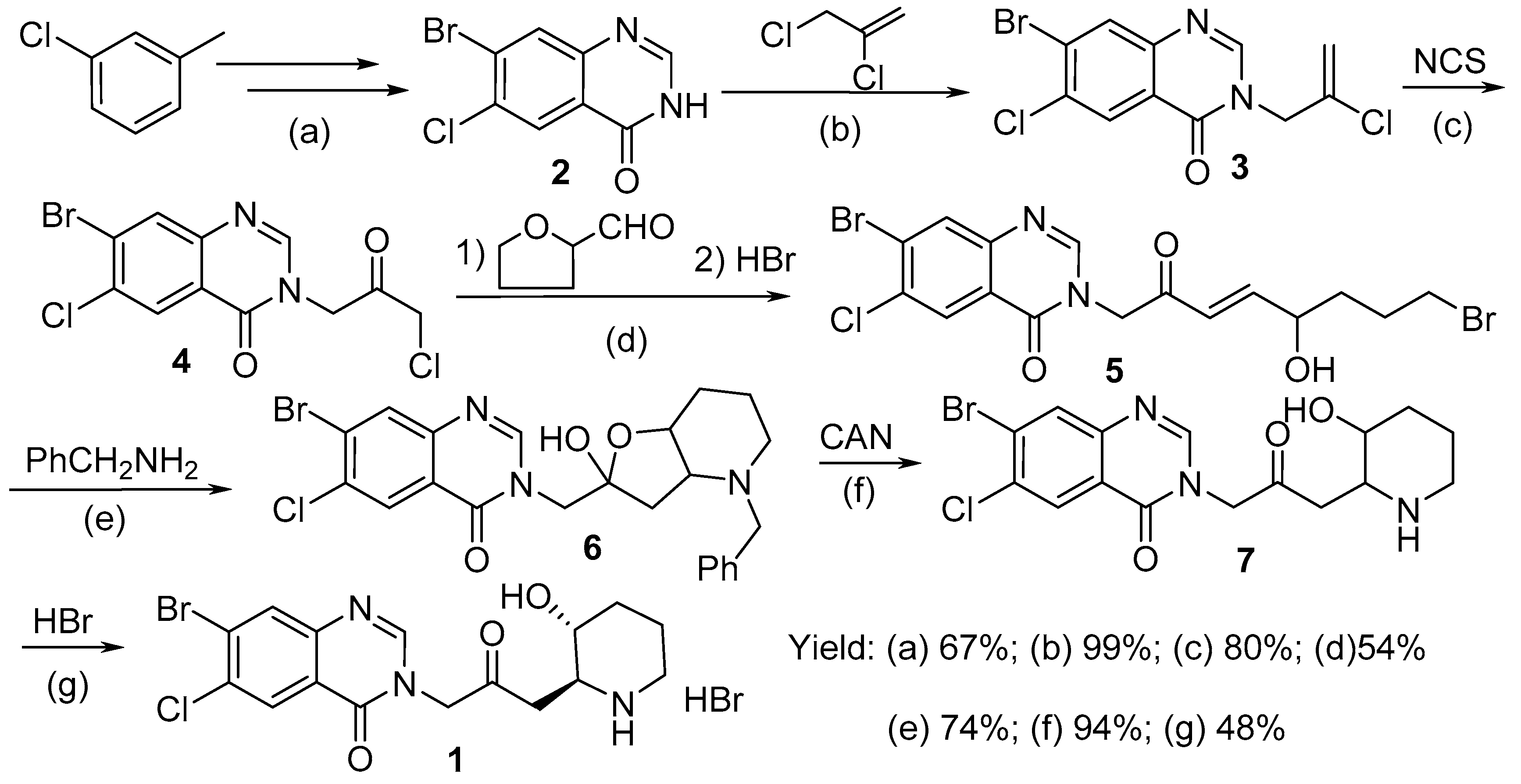
2.1. Synthesis of Halofuginone Hydrobromide
2.2. Evaluation on Antioccidial Activity of Halofuginone Hydrobromide (Hh) in Chickens
3. Experimental Section
3.1. General Information
3.2. Synthesis
3.2.1. Synthesis of 7-bromo-6-choroquinazolin-4(3H)-one (2)
3.2.2. 7-Bromo-6-chloro-3-(2-chloropropenyl)quinazolin-4(3H)-one (3)
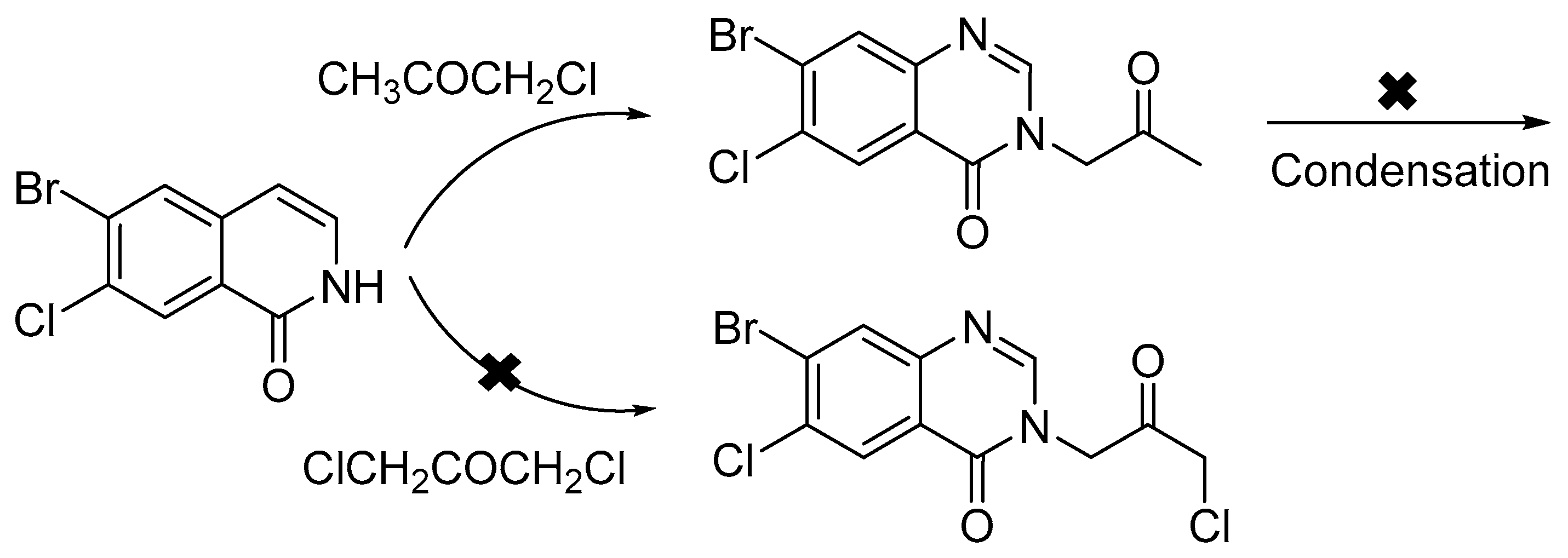
3.2.3. 7-Bromo-6-chloro-3-(3-chloroacetonyl)quinazalin-4(3H)-one (4)
3.2.4. 7-Bromo-6-chloro-3-(8-bromo-5-hydroxyl-2-oxo-3-octenyl) quinazolin-4(3H)-one (5)
3.2.5. 7-Bromo-6-chloro-3-[(4-benzyl-2-hydroxyl-octahydrofuran[3,2-b]pyridine-2)methyl]quinazol-in-4(3H)-one (6)
3.2.6. 7-Bromo-6-chloro-3-[3-(3-hydroxyl-2-piperidyl)-2-oxopropyl]quinazolin-4(3H)-one (7)
3.2.7. (±)-trans-7-Bromo-6-chloro-3-[3-(3-hydroxy-2-piperidyl)acetonyl]quinazolin-4(3H)-one hydro-bromide (halofuginone hydrobromide, 1)
3.3. Evaluation on the Antioccidial Activity of Halofuginone Hydrobromide in Chickens
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Koepfli, J.B.; Mead, J.F.; Brockman, J.A., Jr. Alkaloids of Dichroa febrifuga; isolation and degradative studies. J. Am. Chem. Soc. 1949, 71, 1048–1054. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, S.; Ueno, M.; Suzuki, R.; Ishitani, H.; Kim, H.S.; Wataya, Y. Catalytic asymmetric synthesis of antimalarial alkaloids febrifugine and isofebrifugine and their biological activity. J. Org. Chem. 1999, 64, 6833–6841. [Google Scholar] [CrossRef] [PubMed]
- Emanuel, W.; Gerald, B.; Sidney, K. Method for Treating Coccidiosis with Quinazolinones. U.S. Patent US3,320,124, 16 May 1967. [Google Scholar]
- Anderson, A.; Goodall, E.; Bliss, G.W.; Woodhouse, R.N. Analysis of the anti-coccidial drug, halofuginone, in chicken tissue and chicken feed using high-performance liquid chromatography. J. Chromatogr. 1981, 212, 347–355. [Google Scholar] [CrossRef]
- Zhang, D.F.; Sun, B.B.; Yue, Y.Y.; Yu, H.J.; Zhang, H.L.; Zhou, Q.J.; Du, A.F. Anticoccidial effect of halofuginone hydrobromide against Eimeria tenella with associated histology. Parasitol. Res. 2012, 111, 695–701. [Google Scholar] [CrossRef] [PubMed]
- Ramadan, A.; Abo el-Sooud, K.; el-Bahy, M.M. Anticoccidial efficacy of toltrazuril and halofuginone against Eimeria tenella infection in broiler chickens in Egypt. Res. Vet. Sci. 1997, 62, 175–178. [Google Scholar] [CrossRef]
- Granot, I.; Halevy, O.; Hurwitz, S.; Pines, M. Halofuginone: An inhibitor of collagen type I synthesis. Biochim. Biophys. 1993, 1156, 107–112. [Google Scholar] [CrossRef]
- Pines, M. Halofuginone for fibrosis, regeneration and cancer in the gastrointestinal tract. World J. Gastroenterol. 2014, 20, 14778–14786. [Google Scholar] [CrossRef] [PubMed]
- Yavas, G.; Calik, M.; Calik, G.; Yavas, C.; Ata, O.; Esme, H. The effect of halofuginone in the amelioration of radiation induced-lung fibrosis. Med. Hypotheses 2013, 80, 357–359. [Google Scholar] [CrossRef] [PubMed]
- Lamora, A.; Mullard, M.; Amiaud, J.; Brion, R.; Heymann, D.; Redini, F.; Verrecchia, F. Anticancer activity of halofuginone in a preclinical model of osteosarcoma: Inhibition of tumor growth and lung metastases. Oncotarget 2015, 6, 14413–14427. [Google Scholar] [CrossRef] [PubMed]
- Cook, J.A.; Choudhuri, R.; Degraff, W.; Gamson, J.; Mitchell, J.B. Halofuginone enhances the radiation sensitivity of human tumor cell lines. Cancer Lett. 2010, 289, 119–126. [Google Scholar] [CrossRef] [PubMed]
- Yee, K.O.; Connolly, C.M.; Pines, M.; Lawler, J. Halofuginone inhibits tumor growth in the polyoma middle T antigen mouse via a thrombospondin-1 independent mechanism. Cancer Biol. Ther. 2006, 5, 218–224. [Google Scholar] [CrossRef] [PubMed]
- Casey, N.H.; Crosley, R.I.; Smith, G.A. Influence of continuous dietary halofuginone on broiler skin tensile strength and growth performance. J. S. Afr. Vet. Assoc. 1992, 63, 16–19. [Google Scholar] [PubMed]
- Edgar, S.A.; Flanagan, C. Efficacy of stenorol (halofuginone). III. For the control of coccidiosis in turkeys. Poult. Sci. 1979, 58, 1483–1489. [Google Scholar] [CrossRef] [PubMed]
- Edgar, S.A.; Flanagan, C. Efficacy of stenorol (halofuginone). II. Plus roxarsone or bacitracin md against selected strains of chicken Eimeria. Poult. Sci. 1979, 58, 1476–1482. [Google Scholar] [CrossRef] [PubMed]
- Edgar, S.A.; Flanagan, C. Efficacy of stenorol (halofuginone). I. Against recent field isolates of six species of chicken coccidia. Poult. Sci. 1979, 58, 1469–1475. [Google Scholar] [CrossRef] [PubMed]
- De Jonge, M.J.; Dumez, H.; Verweij, J.; Yarkoni, S.; Snyder, D.; Lacombe, D.; Marreaud, S.; Yamaguchi, T.; Punt, C.J.; van Oosterom, A.; et al. Phase i and pharmacokinetic study of halofuginone, an oral quinazolinone derivative in patients with advanced solid tumours. Eur. J. Cancer 2006, 42, 1768–1774. [Google Scholar] [CrossRef] [PubMed]
- Baker, B.R.; Schaub, R.E.; Joseph, J.P.; McEvoy, F.J.; Williams, J.H. An antimalarial alkaloid from hydrangea. Xv. Synthesis of 5-, 6-, 7-, and 8-derivatives with two identical substituents. J. Org. Chem. 1952, 17, 149–156. [Google Scholar] [CrossRef]
- Schmand, H.; Kellermeier, B.; Bartels, G.; Schmidt, H.J. Verfahren zur Herstellung von Halogenanthranilsäuren. Ger. Patent DE59,206,660, 1 August 1996. [Google Scholar]
- Pavlidis, V.H.; Perry, P.J. The synthesis of a novel series of substituted 2-phenyl-4h-3,1-benzoxazin-4-ones. Synth. Commun. 1994, 24, 533–548. [Google Scholar] [CrossRef]
- Baur, K.G.; Disteldorf, W.; Hagen, H.; Kohler, R.; Markert, J. Process for the Preparation of Haloanthranilic Acids. Ger. Patent DE3,409,244, 19 September 1985. [Google Scholar]
- Baker, B.R.; Querry, M.V.; Kadish, A.F.; Williams, J.H. An antimalarial alkaloid from hydrangea. Iv. Functional derivatives of 3-alkyl-4-quinazolones. J. Org. Chem. 1952, 17, 35–51. [Google Scholar] [CrossRef]
- Baker, B.R.; Schaub, R.E.; McEvoy, F.J.; Williams, J.H. An antimalarial alkaloid from hydrangea. Xii. Synthesis of 3-[β-keto-γ-(3-hydroxy-2-piperidyl)propyl]-4-quinazolone, the alkaloid. J. Org. Chem. 1952, 17, 132–140. [Google Scholar] [CrossRef]
- Baker, B.R.; Schaub, R.E.; Querry, M.V.; Williams, J.H. An antimalarial alkaloid from hydrangea. Ix. Synthesis of 3-[β-keto-γ-(4-hydroxy-2-piperidyl)propyl]-4-quinazolone, an isomer. J. Org. Chem. 1952, 17, 97–108. [Google Scholar] [CrossRef]
- Baker, B.R.; Schaub, R.E.; Querry, M.V.; Williams, J.H. An antimalarial alkaloid from hydrangea. Viii. Attempted synthesis of 3-[β-keto-γ-(4-hydroxy-2-piperidyl)propyl]-4-quinazolone by the diketone approach. J. Org. Chem. 1952, 17, 77–96. [Google Scholar] [CrossRef]
- Baker, B.R.; Schaub, R.E.; Williams, J.H. An antimalarial alkaloid from hydrangea. Xi. Synthesis of 3-[β-keto-γ-(3- and 4-hydroxymethyl-2-pyrrolidyl)propyl]-4-quinazolones. J. Org. Chem. 1952, 17, 116–131. [Google Scholar] [CrossRef]
- Takeuchi, Y.; Azuma, K.; Takakura, K.; Abe, H.; Kim, H.-S.; Wataya, Y.; Harayama, T. Asymmetric synthesis of (+)-febrifugine and (+)-isofebrifugine using yeast reduction. Tetrahedron 2001, 57, 1213–1218. [Google Scholar] [CrossRef]
- Sukemoto, S.; Oshige, M.; Sato, M.; Mimura, K.I.; Nishioka, H.; Abe, H.; Harayama, T.; Takeuchi, Y. Concise asymmetric synthesis of (+)febrifugine utilizing trans-selective intramolecular conjugate addition. Synthesis 2008, 2008, 3081–3087. [Google Scholar]
- Uesato, S.; Kuroda, Y.; Kato, M.; Fujiwara, Y.; Hase, Y.; Fujita, T. Conformational analysis of febrifugines and halofuginones in organic solvents. Chem. Pharm. Bull. 2008, 46, 1–5. [Google Scholar] [CrossRef]
- Johnson, J.; Reid, W.M. Anticoccidial drugs: Lesion scoring techniques in batteryand floor-pen experiments with chickens. Exp. Parasitol. 1970, 28, 30–36. [Google Scholar] [CrossRef]
- Du, H.R.; Zhang, Y.; Ma, D.L.; Jiang, C.L. Advances in the study of halofuginone as an anticoccidial drug. Heilongjiang Anim. Sci. Vet. Med. 2013, 2, 29–31. [Google Scholar]
Sample Availability: Samples of the compounds are available from the authors. |
| Catalyst [a] | Result | Solvent [b] | Result |
|---|---|---|---|
| Sodium methoxide | Incomplete reaction | Ethanol | Favorable reaction |
| Sodium ethoxide | Incomplete reaction | Isopropanol | Favorable reaction |
| Potassium tert-butoxide | Favorable reaction | tert-Butanol | Favorable reaction |
| Sodium hydride | More by-products | N,N-dimethylformamide | Favorable reaction, but complicated work-up |
| N-butyl lithium | More by-products | Ether | Slow reaction |
| / | / | Tetrahydrofuran | More by-products |
| Group [#] | RBWGR [a] (%) | SR [b] (%) | ALS [c] | OS [d] | ACI [e] |
|---|---|---|---|---|---|
| I | 100 | 100 | 0 | 0 | 200.00 |
| II | 46.02 | 40 | 32 | 20 | 34.02 |
| III | 99.68 | 100 | 0 | 1 | 198.68 |
| IV | 106.54 | 100 | 2 | 1 | 203.54 |
| V | 93.02 | 100 | 11 | 5 | 177.02 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, J.; Yao, Q.; Liu, Z. A Novel Synthesis of the Efficient Anti-Coccidial Drug Halofuginone Hydrobromide. Molecules 2017, 22, 1086. https://doi.org/10.3390/molecules22071086
Zhang J, Yao Q, Liu Z. A Novel Synthesis of the Efficient Anti-Coccidial Drug Halofuginone Hydrobromide. Molecules. 2017; 22(7):1086. https://doi.org/10.3390/molecules22071086
Chicago/Turabian StyleZhang, Junren, Qizheng Yao, and Zuliang Liu. 2017. "A Novel Synthesis of the Efficient Anti-Coccidial Drug Halofuginone Hydrobromide" Molecules 22, no. 7: 1086. https://doi.org/10.3390/molecules22071086
APA StyleZhang, J., Yao, Q., & Liu, Z. (2017). A Novel Synthesis of the Efficient Anti-Coccidial Drug Halofuginone Hydrobromide. Molecules, 22(7), 1086. https://doi.org/10.3390/molecules22071086





