Kinetics, Mechanism and Theoretical Studies of Norbornene-Ethylene Alternating Copolymerization Catalyzed by Organopalladium(II) Complexes Bearing Hemilabile α-Amino–pyridine
Abstract
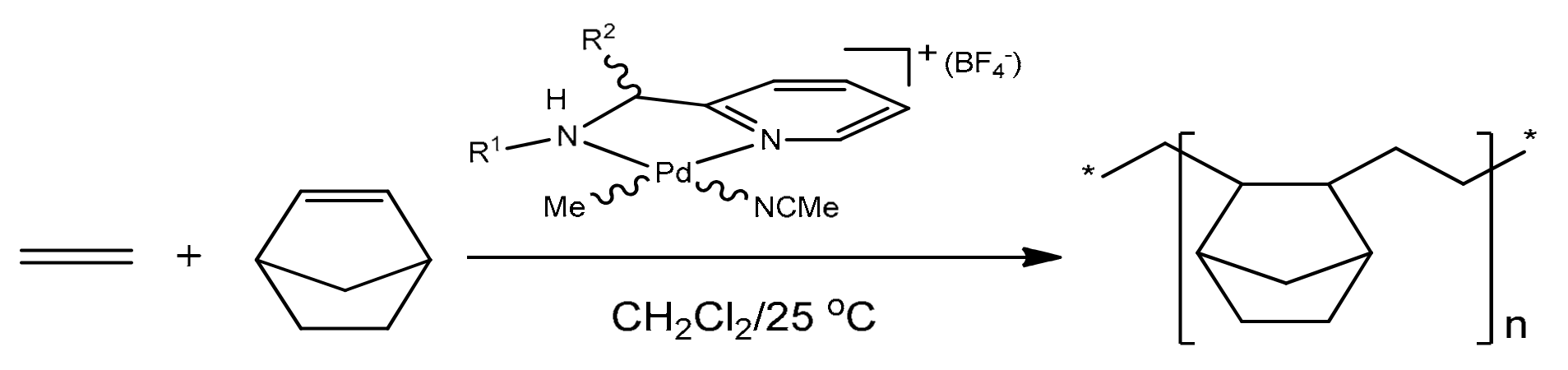
:1. Introduction
2. Results and Discussion
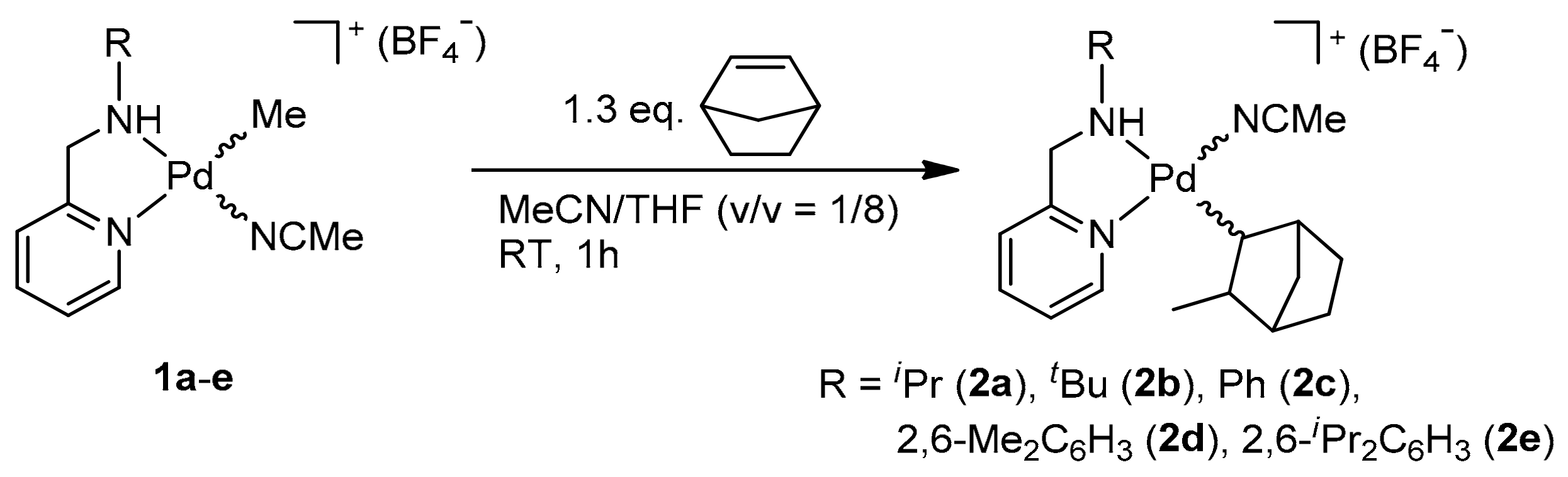
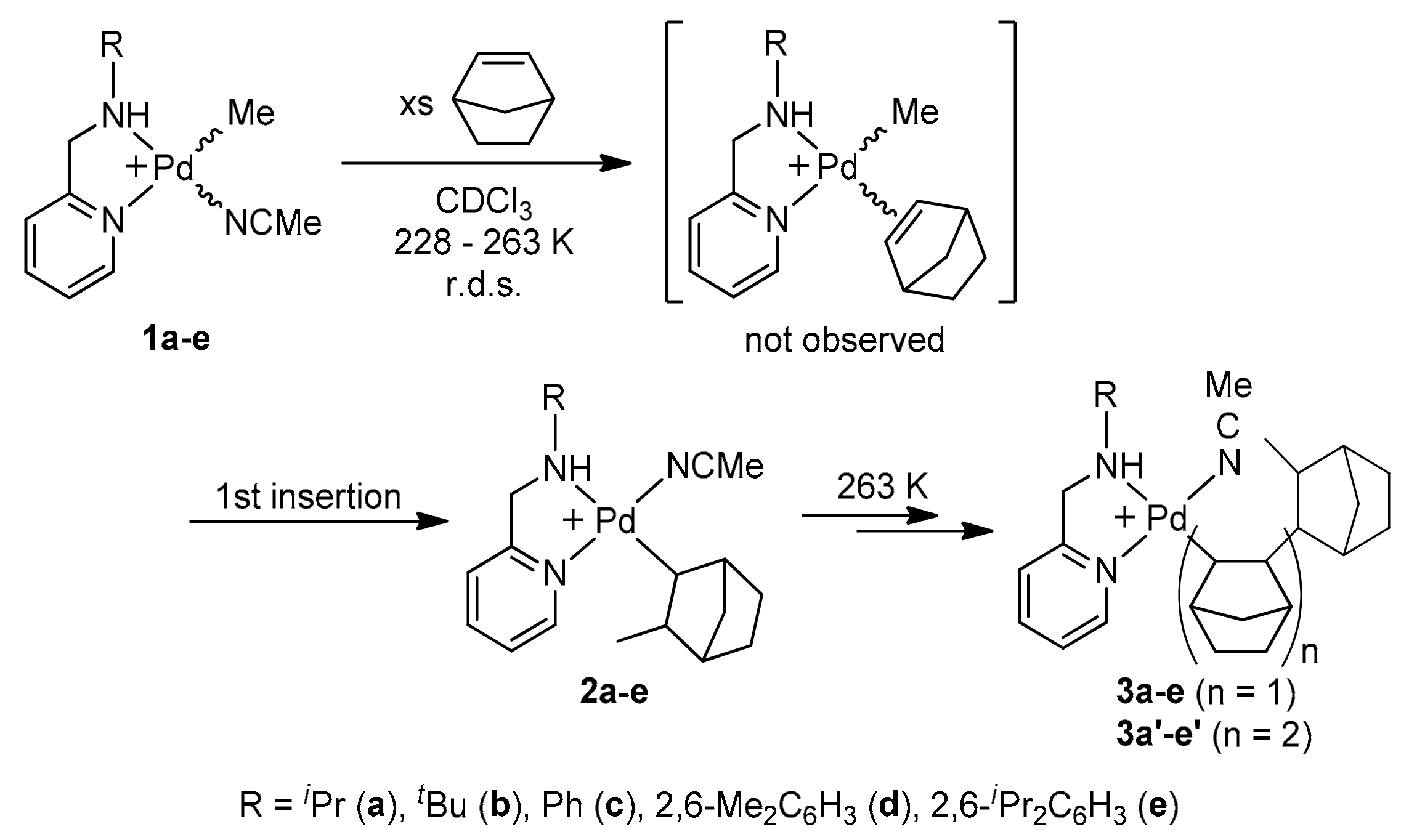
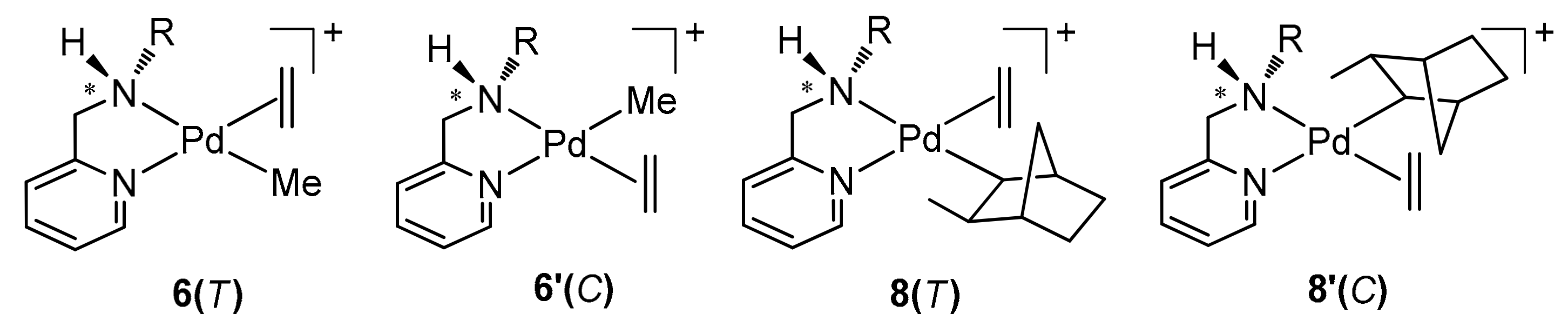
2.1. Synthesis and Characterization of Norbornyl Palladium Complexes
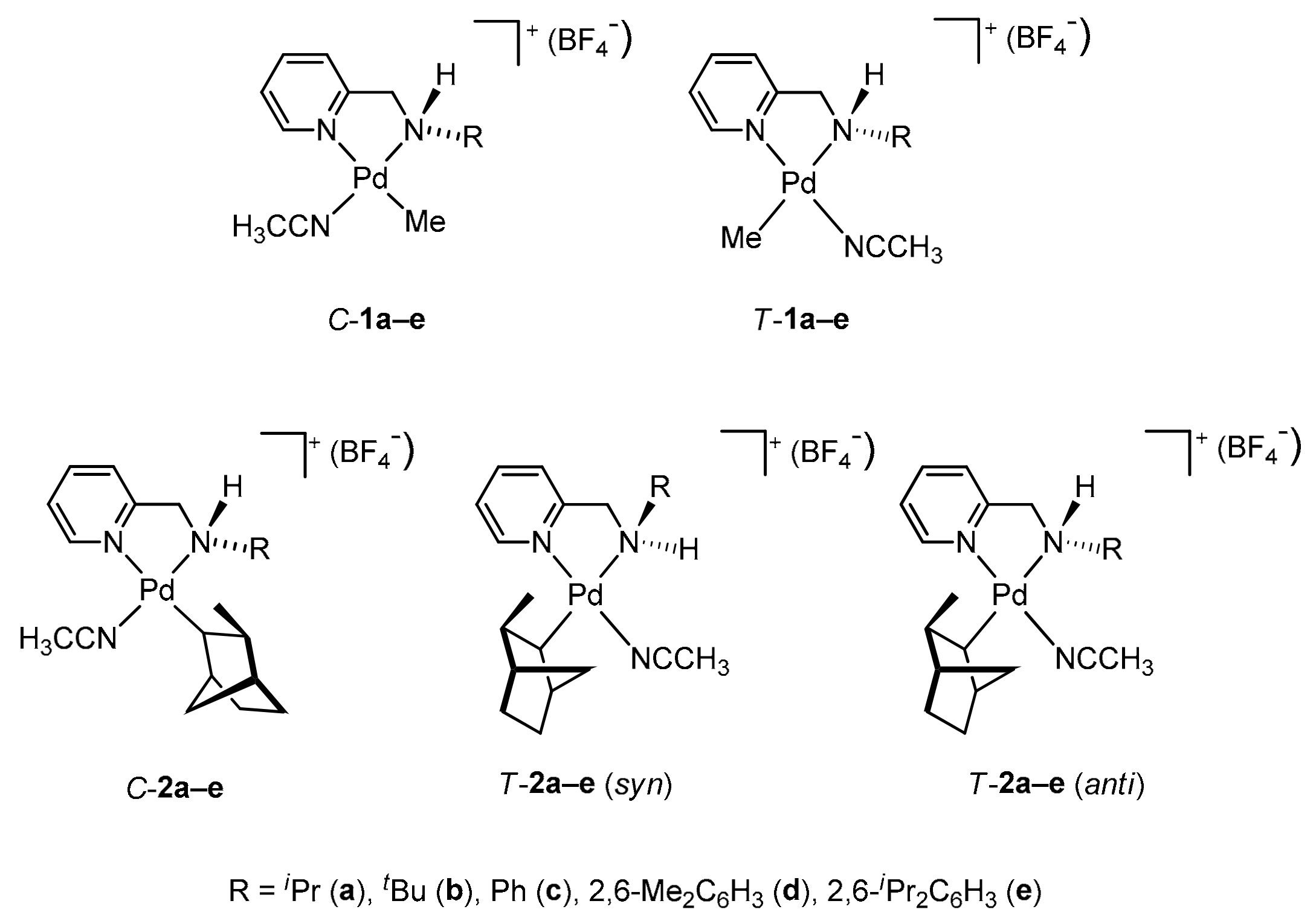
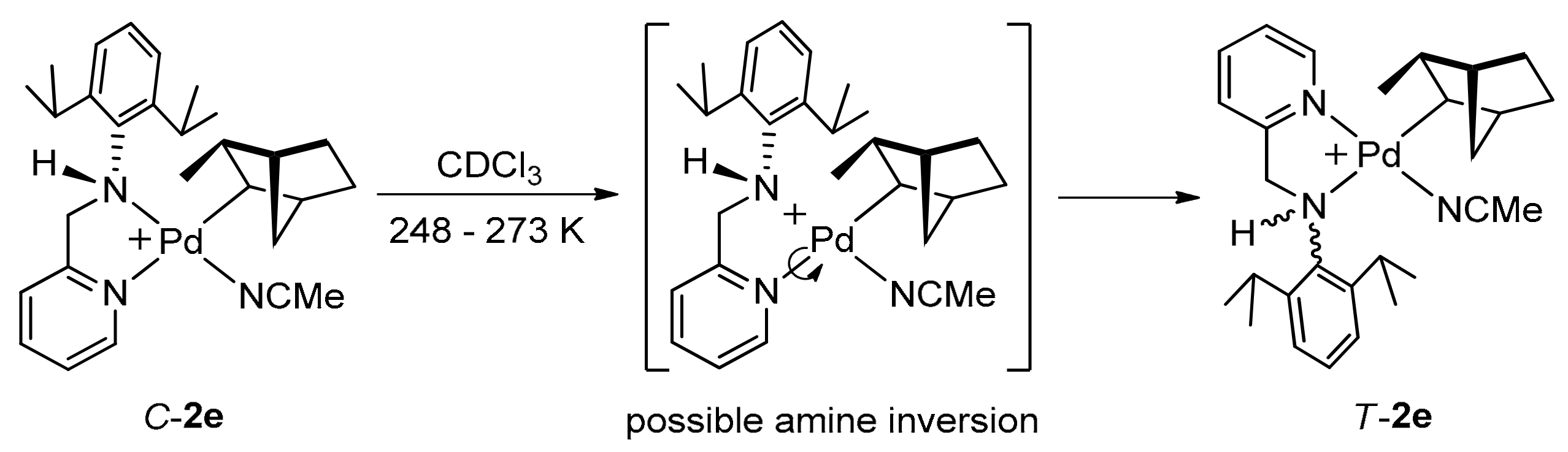
2.2. Geometrical Isomerisation of Norbornylpalladium Complex
2.3. Kinetics of Norbornene Insertion Reactions in Organopalladium(II) Complexes
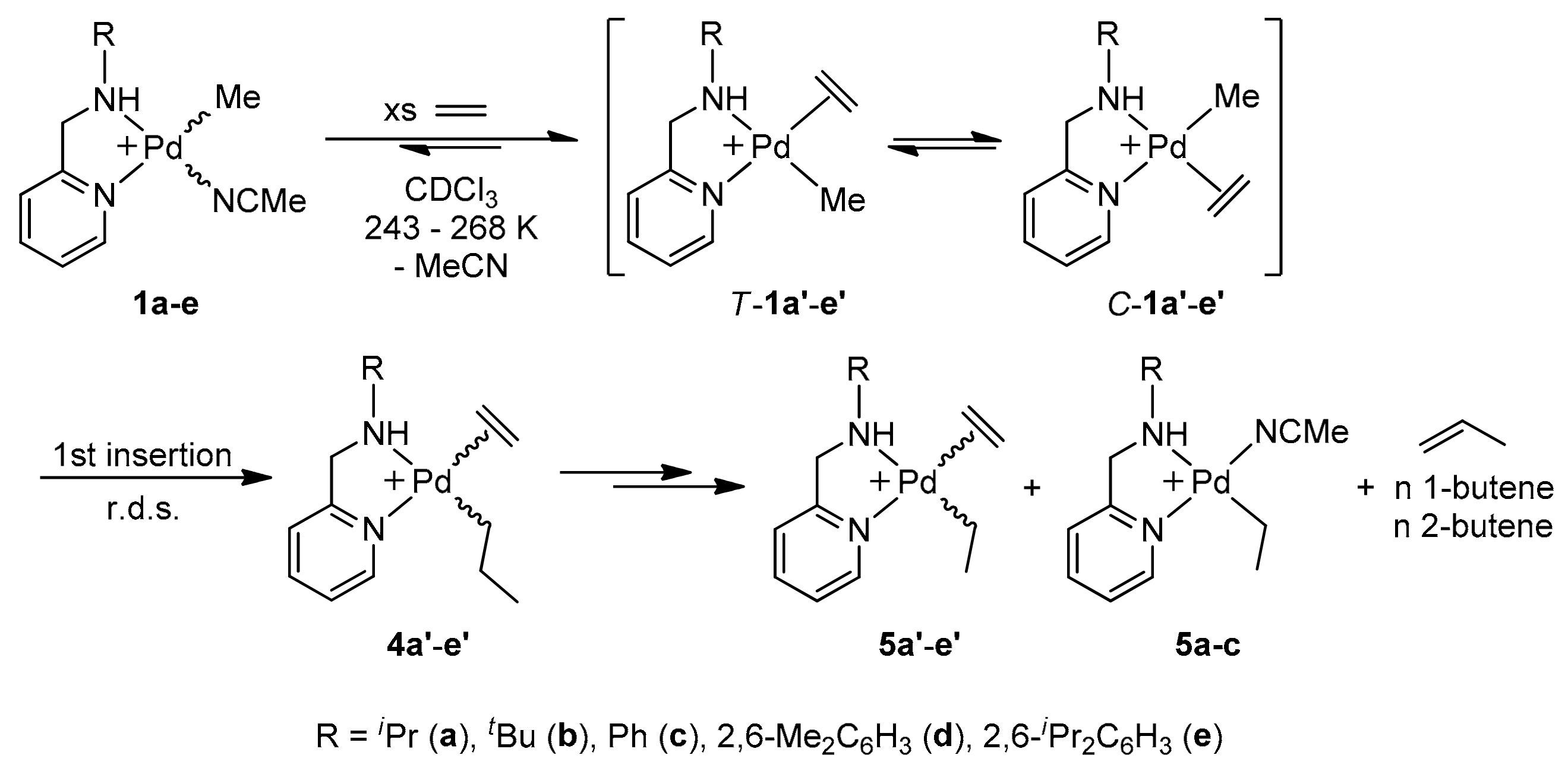
2.4. Kinetics of Ethylene Insertion Reactions in Organopalladium(II) Complexes
2.5. DFT Approach for E–N Copolymerization
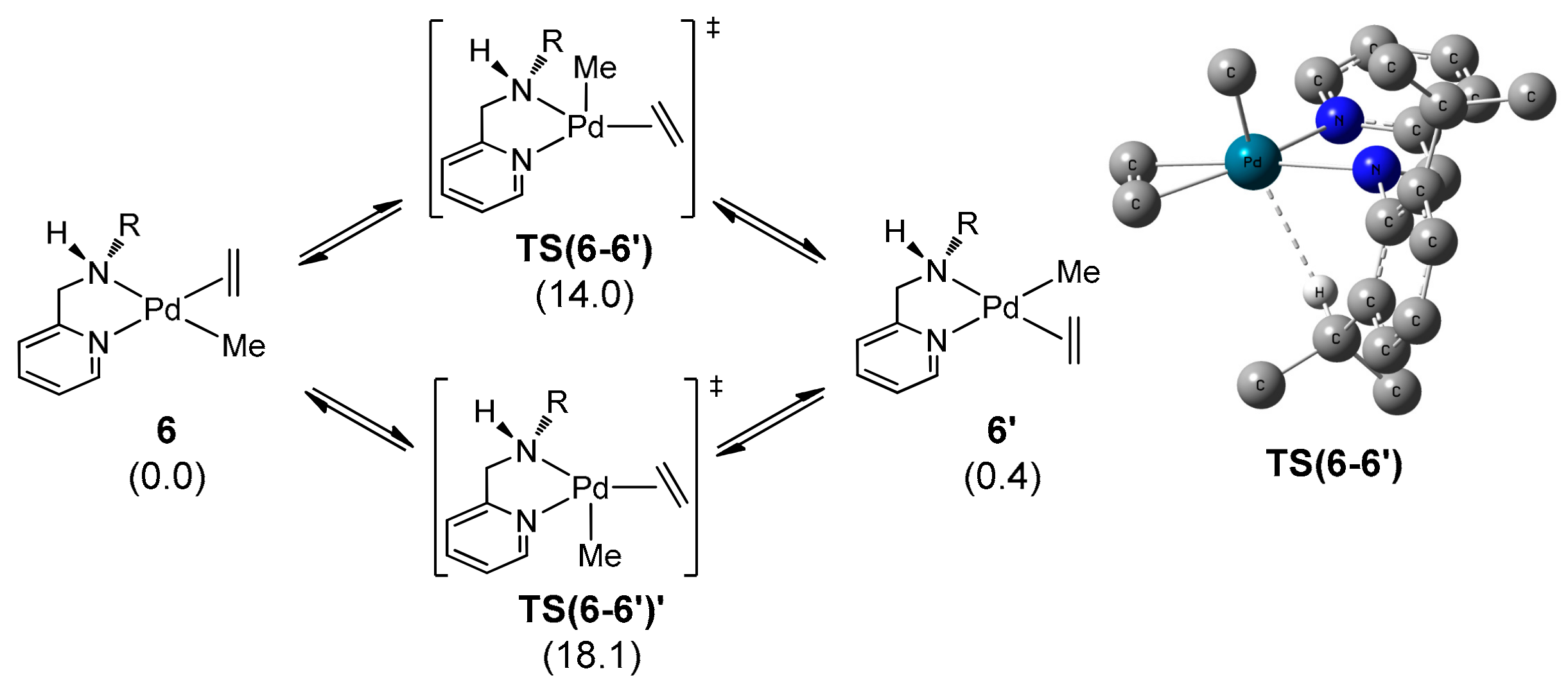
2.5.1. C–T Isomerization
2.5.2. Ethylene Insertion into (C2H4)Pd-Me
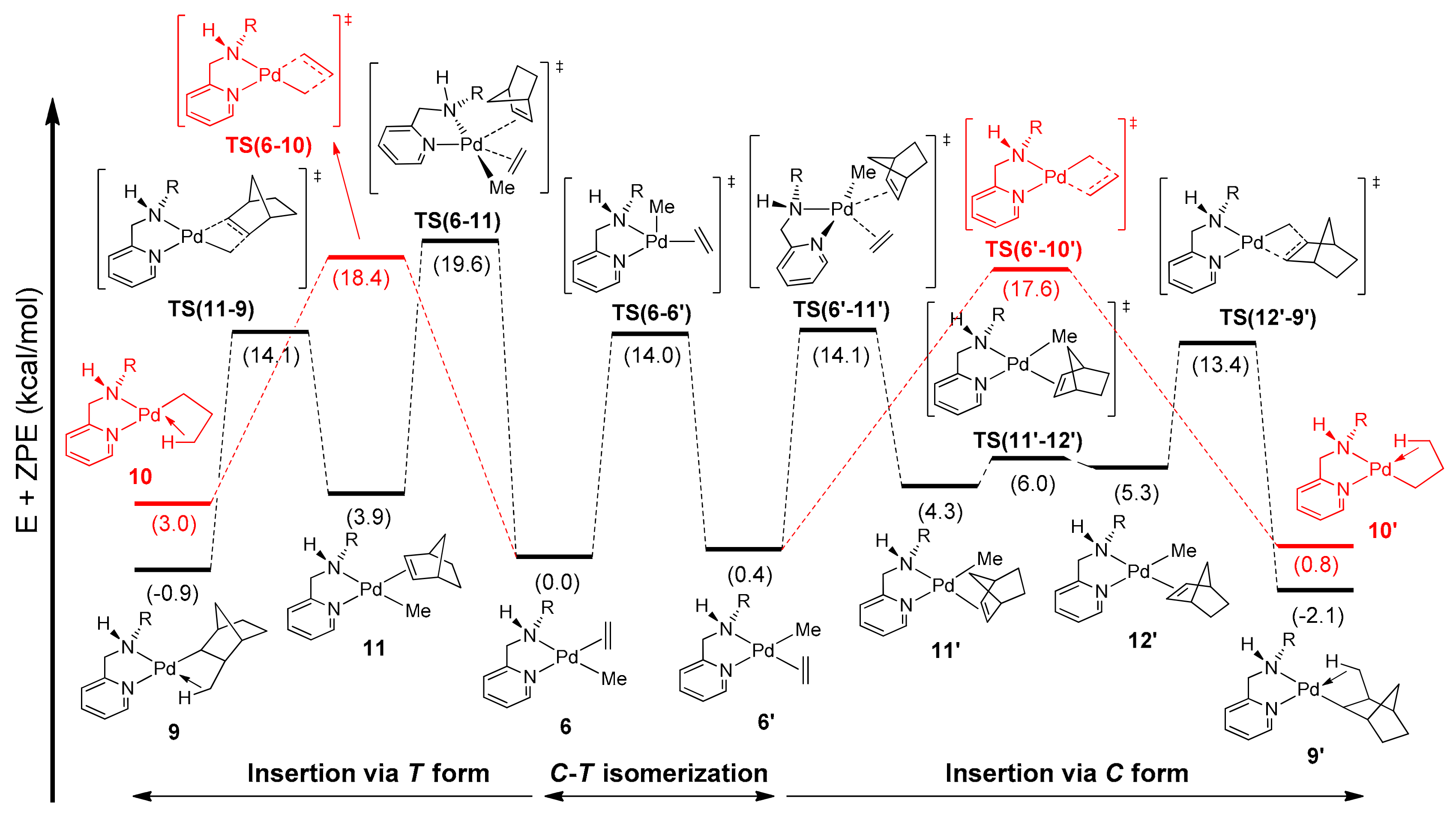
2.5.3. Norbornene Insertion into (C2H4)Pd-Me.
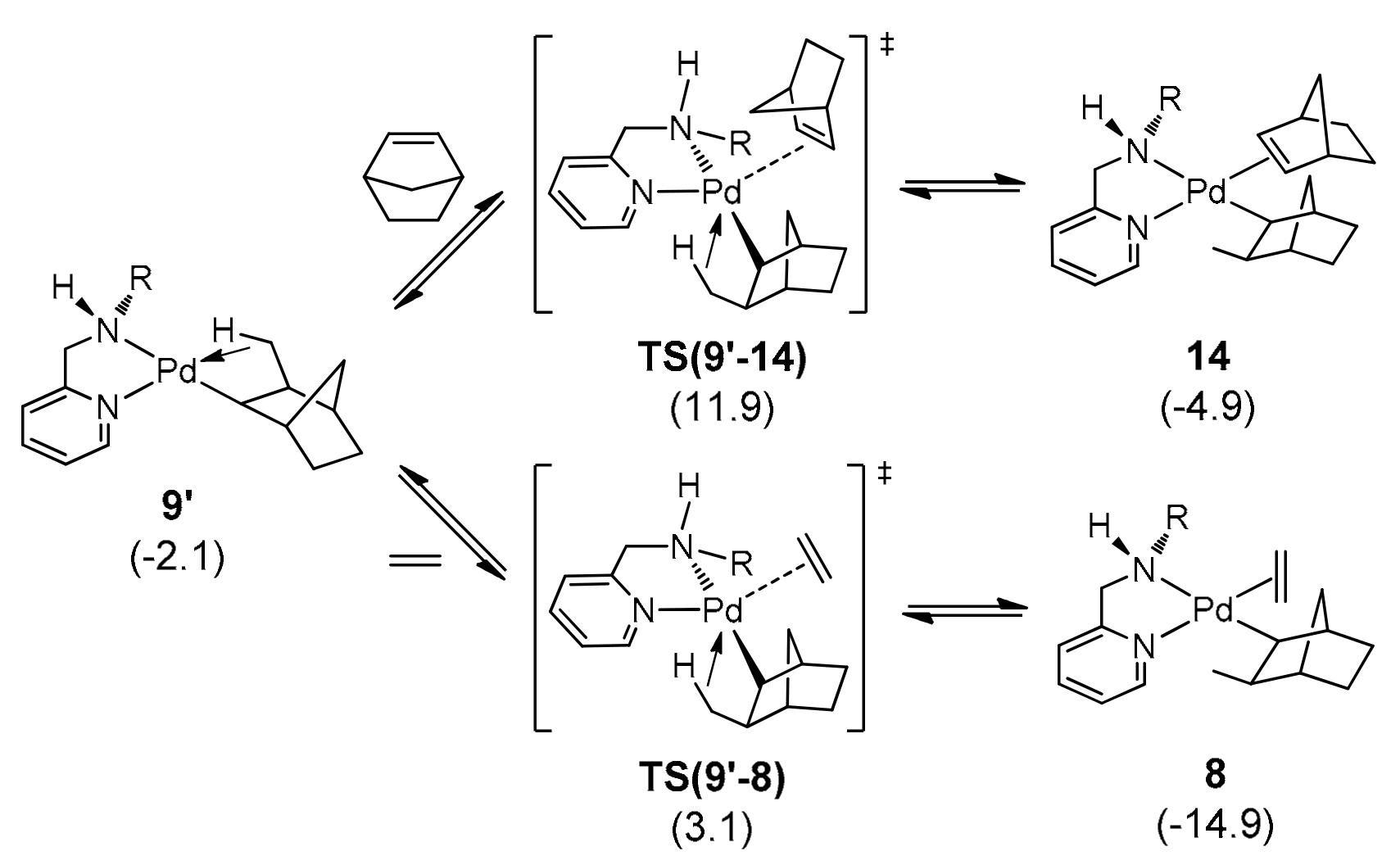
2.5.4. Ethylene and Norbornene Insertion into (C2H4)Pd-C7H10Me
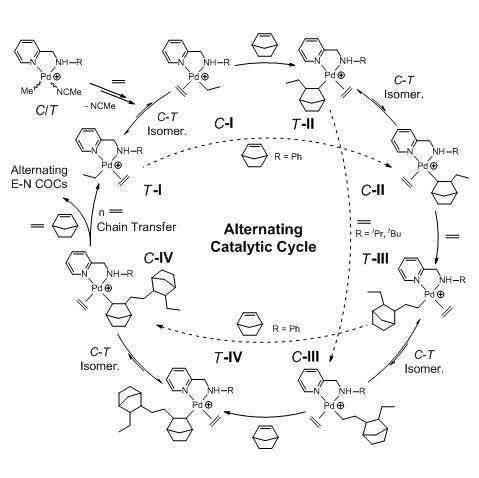
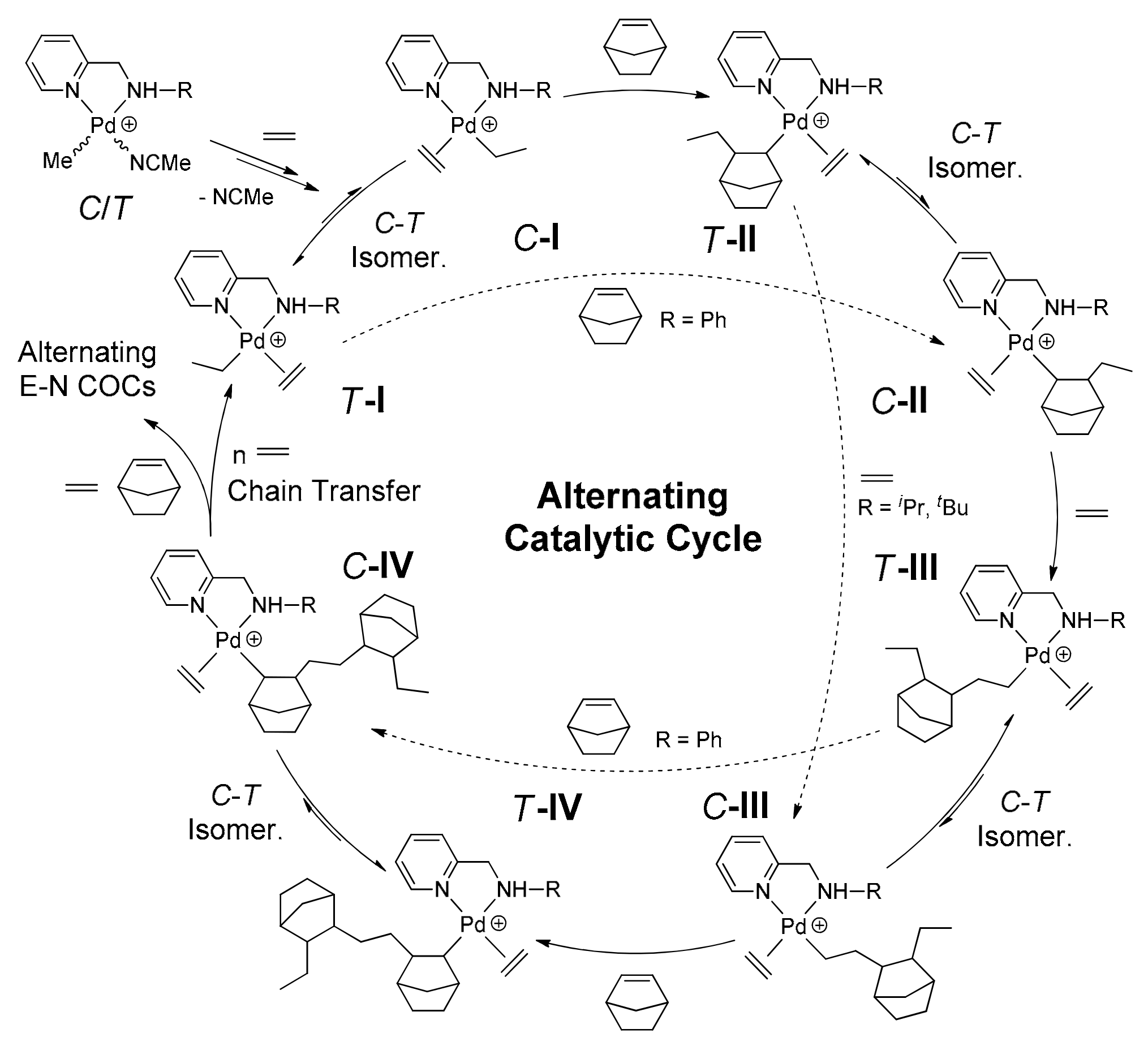
2.5.5. Mechanism of Alternating E–N Copolymerization
3. Materials and Methods
3.1. Materials
3.2. Measurements
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Kaminsky, W.; Bark, A.; Arndt-Rosenau, M. New polymers by homogenous zirconocene/aluminoxane catalysts. Chem. Macromol. Symp. 1991, 47, 83–93. [Google Scholar] [CrossRef]
- Cherdron, H.; Brekner, M.-J.; Osan, F. Cycloolefin-copolymere: Eine neue klasse transparenter thermoplaste. Angew. Makromol. Chem. 1994, 223, 121–133. [Google Scholar] [CrossRef]
- Lamonte, R.R.; McNally, D. Cyclic olefin copolymers. Adv. Mater. Process 2001, 3, 1–4. [Google Scholar]
- Kaminsky, W. Olefin polymerization catalyzed by metallocenes. Adv. Catal. 2001, 46, 89–159. [Google Scholar]
- Nunes, P.S.; Ohlsson, P.D.; Ordeig, O.; Kutter, J.P. Cyclic olefin polymers: Emerging materials for lab-on-a-chip applications. Microfluid Nanofluid 2010, 9, 145–161. [Google Scholar] [CrossRef]
- Boggioni, L.; Tritto, I. State of the art of cyclic olefin polymers. MRS Bull. 2013, 38, 245–251. [Google Scholar] [CrossRef]
- Hasan, T.; Ikeda, T.; Shiono, T.J. Homo- and Copolymerization of Norbornene Derivatives with Ethene by ansa-Fluorenylamidodimethyltitanium Activated with Methylaluminoxane. Polym. Sci. Part A Polym. Chem. 2007, 45, 4581–4587. [Google Scholar] [CrossRef]
- Nishizawa, O.; Misaka, H.; Sakai, R.; Kakuchi, T.; Satoh, T. Copolymerization of Ethylene and Norbornene Using Cyclopentadienylzirconium Trichloride Activated by Isobutyl-Modified Methylaluminoxane. J. Polym. Sci. Part A Polym. Chem. 2008, 46, 7411–7418. [Google Scholar] [CrossRef]
- Sudhakar, P.J. Appreciable Norbornene Incorporation in the Copolymerization of Ethylene/Norbornene Using Titanium Catalysts Containing Trianionic N[CH2CH(Ph)O]33− Ligands. Polym. Sci. Part A Polym. Chem. 2008, 46, 444–452. [Google Scholar] [CrossRef]
- Hu, H.; Gao, H.; Song, K.; Liu, F.; Long, J.; Zhang, L.; Zhu, F.; Wu, Q. Novel bis(benzoin) titanium catalyst for homo- and copolymerization of norbornene with ethylene: Synthesis, characterization and catalytic properties. Polymer 2008, 49, 4552–4558. [Google Scholar] [CrossRef]
- Long, Y.-Y.; Ye, W.-P.; Shi, X.-C.; Li, Y.-S.J. Living Copolymerization of Ethylene with Norbornene Mediated by Heteroligated (Salicylaldiminato)(β-enaminoketonato)Titanium Catalysts. Polym. Sci. Part A Polym. Chem. 2009, 47, 6072–6082. [Google Scholar] [CrossRef]
- Gao, H.-Y.; Hu, H.; Wu, Q. High norbornene incorporation in ethylene–norbornene copolymerization with a bis(α-alkyloxoimine) titanium-MAO catalyst. Sci. China Chem. 2010, 53, 1634–1640. [Google Scholar] [CrossRef]
- Padmanabhan, S.; Vijayakrishna, K.; Mani, R. Copolymerization of ethylene and norbornene by zirconium complexes containing symmetrically tuned trianionic ligands. Polym. Bull. 2010, 65, 13–23. [Google Scholar] [CrossRef]
- Cabrera, A.; Schneider, Y.; Valderrama, M.; Galland, G.B.; Rojas, R.S. Synthesis and Characterization of Materials Prepared via the Copolymerization of Ethylene with 1-Octadecene and Norbornene Using a [(π-Cyano-nacnac)Cp] Zirconium Complex. J. Polym. Sci. A Polym. Chem. 2011, 49, 1535–1542. [Google Scholar] [CrossRef]
- Ravasio, A.; Boggioni, L.; Scalcione, G.; Bertini, F.; Piovani, D.; Tritto, I. Living Copolymerization of Ethylene with Norbornene by Fuorinated Enolato-Imine Titanium Catalyst. J. Polym. Sci. Part A Polym. Chem. 2012, 50, 3867–3874. [Google Scholar] [CrossRef]
- Yang, G.; Hong, M.; Li, Y.; Yu, S. Synthesis of Novel Bis(β-enaminoketonato)titanium Catalyst with High Activity and Excellent Ability to Copolymerize Olefins. Macromol. Chem. Phys. 2012, 213, 2311–2318. [Google Scholar] [CrossRef]
- Tritto, I.; Boggioni, L.; Ferro, D.R. Metallocene catalyzed ethene- and propene co-norbornene polymerization: Mechanisms from a detailed microstructural analysis. Coord. Chem. Rev. 2006, 250, 212–241. [Google Scholar] [CrossRef]
- Ruchatz, D.; Fink, G. Ethene−Norbornene Copolymerization Using Homogenous Metallocene and Half-Sandwich Catalysts: Kinetics and Relationships between Catalyst Structure and Polymer Structure. 2. Comparative Study of Different Metallocene- and Half-Sandwich/Methylaluminoxane Catalysts and Analysis of the Copolymers by 13C Nuclear Magnetic Resonance Spectroscopy. Macromolecules 1998, 31, 4674–4680. [Google Scholar] [PubMed]
- Ruchatz, D.; Fink, G. Ethene−Norbornene Copolymerization with Homogeneous Metallocene and Half-Sandwich Catalysts: Kinetics and Relationships between Catalyst Structure and Polymer Structure. 4. Development of Molecular Weights. Macromolecules 1998, 31, 4684–4686. [Google Scholar] [CrossRef] [PubMed]
- Tritto, I.; Marestin, C.; Boggioni, L.; Sacchi, M.C.; Brintzinger, H.-H.; Ferro, D.R. Stereoregular and Stereoirregular Alternating Ethylene−Norbornene Copolymers. Macromolecules 2001, 34, 5770–5777. [Google Scholar] [CrossRef]
- Nomura, K.; Tsubota, M.; Fujiki, M. Efficient Ethylene/Norbornene Copolymerization by (Aryloxo)(indenyl)titanium(IV) Complexes−MAO Catalyst System. Macromolecules 2003, 36, 3797–3799. [Google Scholar] [CrossRef]
- Yoshida, Y.; Mohri, J.-I.; Ishii, S.-I.; Mitani, M.; Saito, J.; Matsui, S.; Makio, H.; Nakano, T.; Tanaka, H.; Onda, M.; et al. Living Copolymerization of Ethylene with Norbornene Catalyzed by Bis(Pyrrolide−Imine) Titanium Complexes with MAO. J. Am. Chem. Soc. 2004, 126, 12023–12032. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, Y.; Matsui, S.; Fujita, T. Bis(pyrrolide–imine) Ti complexes with MAO: A new family of high performance catalysts for olefin polymerization. J. Organomet. Chem. 2005, 690, 4382–4397. [Google Scholar] [CrossRef]
- Nomura, K.; Wang, W.; Fujiki, M.; Liu, J. Notable norbornene (NBE) incorporation in ethylene–NBE copolymerization catalysed by nonbridged half-titanocenes: Better correlation between NBE incorporation and coordination energy. Chem. Commun. 2006, 25, 2659–2661. [Google Scholar] [CrossRef] [PubMed]
- Nomura, K.; Yamada, J.; Wang, W.; Liu, J. Effect of ketimide ligand for ethylene polymerization and ethylene/norbornene copolymerization catalyzed by (cyclopentadienyl)(ketimide)titanium complexes–MAO catalyst systems: Structural analysis for Cp∗TiCl2(N=CPh2). J. Organomet. Chem. 2007, 692, 4675–4682. [Google Scholar] [CrossRef]
- Zuo, W.; Sun, W.-H.; Zhang, S.; Hao, P.; Shiga, A. Highly Active Ethylene Polymerization and Copolymerization with Norbornene Using Bis(imino-indolide) Titanium Dichloride–MAO system. J. Polym. Sci. Part A Polym. Chem. 2007, 45, 3415–3430. [Google Scholar] [CrossRef]
- Gao, M.; Wang, C.; Sun, X.; Qian, C.; Ma, Z.; Bu, S.; Tang, Y.; Xie, Z. Ethylene–Norbornene Copolymerization by New Titanium Complexes Bearing Tridentate Ligands. Sidearm Effects on Catalytic Activity. Macromol. Rapid Commun. 2007, 28, 1511–1516. [Google Scholar] [CrossRef]
- Li, Y.; Gao, H.; Wu, Q. Homo-and Copolymerization of Ethylene and Norbornene with Bis(β-diketiminato) Titanium Complexes Activated with Methylaluminoxane. J. Polym. Sci. Part A Polym. Chem. 2008, 46, 93–101. [Google Scholar] [CrossRef]
- Yao, Z.; Lv, F.; Liu, S.-J.; Cao, K. Synthesis of Ethylene and Norbornene Copolymer with Metallocene Catalysts and Characteristic Analysis. J. Appl. Polym. Sci. 2008, 107, 286–291. [Google Scholar] [CrossRef]
- Terao, H.; Iwashita, A.; Ishii, S.; Tanaka, H.; Yoshida, Y.; Mitani, M.; Fujita, T. Ethylene/Norbornene Copolymerization Behavior of Bis(phenoxy–imine) Ti Complexes Combined with MAO. Macromolecules 2009, 42, 4359–4361. [Google Scholar] [CrossRef]
- Hong, M.; Wang, Y.-X.; Mu, H.-L.; Li, Y.-S. Efficient Synthesis of Hydroxylated Polyethylene via Copolymerization of Ethylene with 5-Norbornene-2-methanol Using Bis(β-enaminoketonato)titanium Catalysts. Organometallics 2011, 30, 4678–4686. [Google Scholar] [CrossRef]
- Zhang, W.; Wang, Y.; Redshaw, C.; Hao, X.; Sun, W.-H. 2-Aldiminophenoxytitanium chloride complexes: Synthesis, characterization, and ethylene (co-)polymerization behavior. J. Organomet. Chem. 2012, 715, 119–128. [Google Scholar] [CrossRef]
- Tao, P.; Mu, H.-L.; Liu, J.-Y.; Li, Y.-S. New Half-Sandwich Chromium(III) Complexes Bearing Phenoxy-Phosphine (Oxide) [O,P(=O)] Ligands: Synthesis, Structures, and Catalytic Properties for Ethylene (Co)Polymerization. Organometallics 2013, 32, 4805–4812. [Google Scholar] [CrossRef]
- Nomura, K.; Bahuleyan, B.K.; Zhang, S.; Sharma, P.M.V.; Katao, S.; Igarashi, A.; Inagaki, A.; Tamm, M. Synthesis and Structural Analysis of (Imido)vanadium(V) Dichloride Complexes Containing Imidazolin-2-iminato- and Imidazolidin-2-iminato Ligands, and their Use as Catalyst Precursors for Ethylene (Co)polymerization. Inorg. Chem. 2014, 53, 607–623. [Google Scholar] [CrossRef] [PubMed]
- Diteepeng, N.; Tang, X.; Hou, X.; Li, Y.-S.; Phomphrai, K.; Nomura, K. Ethylene polymerisation and ethylene/norbornene copolymerisation by using aryloxo-modified vanadium(V) complexes containing 2,6-difluoro-, dichloro-phenylimido complexes. Dalton Trans. 2015, 44, 12273–12281. [Google Scholar] [CrossRef] [PubMed]
- Pei, L.; Tang, Y.; Gao, H. Homo- and Copolymerization of Ethylene and Norbornene with Anilido–Imine Chromium Catalysts. Polymers 2016, 8, 69. [Google Scholar] [CrossRef]
- Yoon, K.-B.; Choi, Y.S.; Noh, S.K.; Lee, D.-H. Ethylene/Norbornene Copolymerization with iPr(Cp)(Flu)ZrCl2 Catalyst: Effect of MAO Cocatalyst and 3rd Monomer. Macromol. Symp. 2007, 260, 27–33. [Google Scholar] [CrossRef]
- Bredeau, S.; Boggioni, L.; Bertini, F.; Tritto, I.; Monteverde, F.; Alexandre, M.; Dubois, P. Ethylene–Norbornene Copolymerization by Carbon Nanotube-Supported Metallocene Catalysis: Generation of High-Performance Polyolefinic Nanocomposites. Macromol. Rapid Commun. 2007, 28, 822–827. [Google Scholar] [CrossRef]
- Gentil, S.; Pirio, N.; Meunier, P.; Gallou, F.; Paquette, L.A. Ethene/Norbornene Copolymerization with (Isodicyclopentadienyl)titanium Complex–MAO Catalyst. Collect. Czech. Chem. Commun. 2007, 72, 697–702. [Google Scholar]
- Tada, T.; Cai, Z.; Nakayama, Y.; Shiono, T. Efficient Molecular Weight Control with Trialkylaluminum in Ethylene/Norbornene Copolymerization by [Ph2C(Flu)(3-MeCp)]ZrCl2/Methylaluminoxane Catalyst. Macromol. Chem. Phys. 2010, 211, 2132–2137. [Google Scholar] [CrossRef]
- Huang, W.; Li, B.; Wang, Y.; Zhang, W.; Wang, L.; Li, Y.; Sun, W.-H.; Redshaw, C. Synthesis, characterization and ethylene (co-)polymerization behavior of half-titanocene 2-(1-(arylimino)ethyl)quinolin-8-olate chlorides. Catal. Sci. Technol. 2011, 1, 1208–1215. [Google Scholar] [CrossRef]
- Zou, Y.; Wang, D.; Wurst, K.; Kühnel, C.; Reinhardt, I.; Decker, U.; Gurram, V.; Camadanli, S.; Buchmeiser, M.R. Group 4 Dimethylsilylenebisamido Complexes Bearing the 6-[2-(Diethylboryl)phenyl]pyrid-2-yl Motif: Synthesis and Use in Tandem Ring Opening Metathesis/Vinyl-Insertion Copolymerization of Cyclic Olefins with Ethylene. Chem. Eur. J. 2011, 17, 13832–13846. [Google Scholar] [CrossRef] [PubMed]
- Carvalhoa, M.F.N.N.; Galvão, A.M.; Kredatusová, J.; Merna, J.; Pinheiro, P.F.; Salema, M.M. Synthesis and catalytic activity of camphor titanium complexes. Inorg. Chim. Acta 2012, 383, 244–249. [Google Scholar] [CrossRef]
- Hong, M.; Cui, L.; Liu, S.; Li, Y. Synthesis of Novel Cyclic Olefin Copolymer (COC) with High Performance via Effective Copolymerization of Ethylene with Bulky Cyclic Olefin. Macromolecules 2012, 45, 5397–5402. [Google Scholar] [CrossRef]
- Tang, X.-Y.; Wang, Y.-X.; Li, B.-X.; Liu, J.-Y.; Li, Y.-S. Highly Efficient Ethylene/Norbornene Copolymerization by o-Di(phenyl)phosphanylphenolate-Based Half-Titanocene Complexes. J. Polym. Sci. Part A Polym. Chem. 2013, 51, 1585–1594. [Google Scholar] [CrossRef]
- Tang, X.-Y.; Liu, J.-Y.; Li, Y.-S. Phosphine-Thiophenolate Half-Titanocene Chlorides: Synthesis, Structure, and Their Application in Ethylene (Co-)Polymerization. Catalysts 2013, 3, 261–275. [Google Scholar] [CrossRef]
- Tritto, I.; Boggioni, L.; Scalcione, G.; Sidari, D.; Galotto, N.G. Novel norbornene copolymers with transition metal catalysts. J. Organomet. Chem. 2015, 798, 367–374. [Google Scholar] [CrossRef]
- Zhao, W.; Yan, Q.; Tsutsumi, K.; Nomura, K. Efficient Norbornene (NBE) Incorporation in Ethylene/NBE Copolymerization by Half-Titanocene Catalysts Containing Chlorinated Aryloxo Ligands. Organometallics 2016, 35, 1895–1905. [Google Scholar] [CrossRef]
- Zhao, W.; Nomura, K. Design of Efficient Molecular Catalysts for Synthesis of Cyclic Olefin Copolymers (COC) by Copolymerization of Ethylene and α-Olefins with Norbornene or Tetracyclododecene. Catalysts 2016, 6, 175. [Google Scholar] [CrossRef]
- Li, Y.; Yang, J.; Wang, B.; Li, Y. Efficient copolymerization of ethylene with norbornene or its derivatives using half-metallocene zirconium (IV) catalysts. RSC Adv. 2016, 6, 59590–59599. [Google Scholar] [CrossRef]
- Ruchatz, D.; Fink, G. Ethene−Norbornene Copolymerization Using Homogenous Metallocene and Half-Sandwich Catalysts: Kinetics and Relationships between Catalyst Structure and Polymer Structure. 1. Kinetics of the Ethene−Norbornene Copolymerization Using the [(Isopropylidene)(η5-inden-1-ylidene-η5-cyclopentadienyl)]zirconium Dichloride/Methylaluminoxane Catalyst. Macromolecules 1998, 31, 4669–4673. [Google Scholar]
- Ruchatz, D.; Fink, G. Ethene−Norbornene Copolymerization with Homogeneous Metallocene and Half-Sandwich Catalysts: Kinetics and Relationships between Catalyst Structure and Polymer Structure. 3. Copolymerization Parameters and Copolymerization Diagrams. Macromolecules 1998, 31, 4681–4683. [Google Scholar] [CrossRef] [PubMed]
- McKnight, A.L.; Waymouth, R.M. Ethylene/Norbornene Copolymerizations with Titanium CpA Catalysts. Macromolecules 1999, 32, 2816–2825. [Google Scholar] [CrossRef]
- Kaminsky, W.; Beulich, I.; Arndt-Rosenau, M. Copolymerization of Ethene with Cyclic and Other Sterically Hindered Olefins. Macromol. Symp. 2001, 173, 211–225. [Google Scholar] [CrossRef]
- Tritto, I.; Boggioni, L.; Jansen, J.C.; Thorshaug, K.; Sacchi, M.C.; Ferro, D.R. Ethylene–Norbornene Copolymers from Metallocene-Based Catalysts: Microstructure at Tetrad Level and Reactivity Ratios. Macromolecules 2002, 35, 616–623. [Google Scholar] [CrossRef]
- Park, S.Y.; Choi, K.Y.; Song, K.H.; Jeong, B.G. Kinetic Modeling of Ethylene−Norbornene Copolymerization Using Homogeneous Metallocene Catalysts. Macromolecules 2003, 36, 4216–4225. [Google Scholar] [CrossRef]
- Hasan, T.; Ikeda, T.; Shiono, T. Ethene−Norbornene Copolymer with High Norbornene Content Produced by ansa-Fluorenylamidodimethyltitanium Complex Using a Suitable Activator. Macromolecules 2004, 37, 8503–8509. [Google Scholar] [CrossRef]
- Tritto, I.; Boggioni, L.; Ferro, D.R. Alternating Isotactic Ethylene−Norbornene Copolymers by C1-Symmetric Metallocenes: Determination of the Copolymerization Parameters and Mechanistic Considerations on the Basis of Pentad Analysis. Macromolecules 2004, 37, 9681–9693. [Google Scholar] [CrossRef]
- Vijayakrishna, K.; Sundararajan, G. Non-Cp type titanium precatalysts for ethylene/norbornene copolymerization. Polymer 2006, 47, 8289–8296. [Google Scholar] [CrossRef]
- Park, S.Y.; Lee, J.; Choi, K.Y. A Reduced Third Order Markov Model for Ethylene–Norbornene Copolymerization Kinetics with Homogeneous Metallocene Catalysts. Macromol. React. Eng. 2007, 1, 68–77. [Google Scholar] [CrossRef]
- Ni, J.; Lü, C.; Zhang, Y.; Liu, Z.; Mu, Y. Norbornene polymerization and ethylene/norbornene copolymerization catalyzed by constrained geometry cyclopentadienyl-phenoxytitanium catalysts. Polymer 2008, 49, 211–216. [Google Scholar] [CrossRef]
- Li, H.; Li, J.; Zhang, Y.; Mu, Y. Homo- and copolymerization of 5-ethylidene-2-norbornene with ethylene by [2-C5Me4-4,6-tBu2C6H2O]TiCl2/AliBu3/Ph3CB(C6F5)4 catalyst system and epoxidation of the resulting copolymer Polymer 2008, 49, 2839–2844. Polymer 2008, 49, 2839–2844. [Google Scholar] [CrossRef]
- Ravasio, A.; Boggioni, L.; Tritto, I. Copolymerization of Ethylene with Norbornene by Neutral Aryl Phosphine Sulfonate Palladium Catalyst. Macromolecules 2011, 44, 4180–4186. [Google Scholar] [CrossRef]
- Diamanti, S.J.; Ghosh, P.; Shimizu, F.; Bazan, G.C. Ethylene Homopolymerization and Copolymerization with Functionalized 5-Norbornen-2-yl Monomers by a Novel Nickel Catalyst System. Macromolecules 2003, 36, 9731–9735. [Google Scholar] [CrossRef]
- Joe, S.S.D.J.; Na, S.J.; Park, Y.-W.; Choi, C.H.; Lee, B.Y. Ethylene/Polar Norbornene Copolymerizations by Bimetallic Salicylaldimine-Nickel Catalysts. Macromolecules 2005, 38, 10027–10033. [Google Scholar]
- Wang, L.; Li, Y.; Zhu, F.; Wu, Q. Copolymerization of Norbornene and Methyl Acrylate by β-ketoiminato Palladium Complexes/MAO. Polym. Bull. 2006, 57, 73–81. [Google Scholar] [CrossRef]
- Liu, S.; Borkar, S.; Newsham, D.; Yennawar, H.; Sen, A. Synthesis of Palladium Complexes with an Anionic P∼O Chelate and Their Use in Copolymerization of Ethene with Functionalized Norbornene Derivatives: Unusual Functionality Tolerance. Organometallics 2007, 26, 210–216. [Google Scholar] [CrossRef]
- Chen, Z.; Mesgar, M.; White, P.S.; Daugulis, O.; Brookhart, M. Synthesis of Branched Ultrahigh-Molecular-Weight Polyethylene Using Highly Active Neutral, Single-Component Ni(II) Catalysts. ACS Catal. 2015, 5, 631–636. [Google Scholar] [CrossRef]
- Chen, Z.; Allen, K.E.; White, P.S.; Daugulis, O.; Brookhart, M. Synthesis of Branched Polyethylene with “Half-Sandwich” Pyridine–Imine Nickel Complexes. Organometallics 2016, 35, 1756–1760. [Google Scholar] [CrossRef]
- Bezier, D.; Daugulis, O.; Brookhart, M. Oligomerization of Ethylene Using a Diphosphine Palladium Catalyst. Organometallics 2017, 36, 443–447. [Google Scholar] [CrossRef]
- Ji, P.; Guo, L.; Hu, X.; Li, W. Ethylene polymerization by salicylaldimine Nickel(II) complexes derived from arylnaphthylamine. J. Polym. Res. 2017, 24, 30–38. [Google Scholar] [CrossRef]
- Huang, Y.; He, J.; Liu, Z.; Cai, G.; Zhang, S.; Li, X. A highly active chiral (S,S)-bis(oxazoline) Pd(II) alkyl complex/activator catalytic system for vinyl polymerization of norbornene in air and water. Poly. Chem. 2017. [Google Scholar] [CrossRef]
- Brekner, M.-J.; Osan, F.; Rohrmann, J.; Antberg, M.U.S. Process for the Preparation of Chemically Homogeneous Cycloolefin Copolymers. U.S. Patent 5,324,801, 28 January 1994. [Google Scholar]
- Benedikt, G.M.; Elce, E.; Goodall, B.L.; Kalamarides, H.A.; McIntosh, L.H.; Rhodes, L.F.; Selvy, K.T. Copolymerization of Ethene with Norbornene Derivatives Using Neutral Nickel Catalysts. Macromolecules 2002, 35, 8978–8988. [Google Scholar] [CrossRef]
- Kiesewetter, J.; Kaminsky, W. Ethene/Norbornene Copolymerization with Palladium(II) α-Diimine Catalysts: From Ligand Screening to Discrete Catalyst Species. Chem. Eur. J. 2003, 9, 1750–1758. [Google Scholar] [CrossRef] [PubMed]
- Kiesewetter, J.; Arikan, B.; Kaminsky, W. Copolymerization of ethene with norbornene using palladium(II) α-diimine catalysts: Influence of feed composition, polymerization temperature, and ligand structure on copolymer properties and microstructure. Polymer 2006, 47, 3302–3314. [Google Scholar] [CrossRef]
- Rodriguez, B.A.; Delferro, M.; Marks, T.J. Neutral Bimetallic Nickel(II) Phenoxyiminato Catalysts for Highly Branched Polyethylenes and Ethylene−Norbornene Copolymerizations. Organometallics 2008, 27, 2166–2168. [Google Scholar] [CrossRef]
- Tritto, I.; Boggioni, L.; Ravasio, A.; Scalcione, G. Cycloolefin Copolymers by Early and Late Transition Metal Catalysts. Macromol. React. Eng. 2013, 7, 91–97. [Google Scholar] [CrossRef]
- Gao, H.Y.; Liu, Y.; Li, G.L.; Xiao, Z.F.; Liang, G.D.; Wu, Q. Catalytic synthesis of polyethylene-block-polynorbornene copolymers using a living polymerization nickel catalyst. Polym. Chem. 2014, 5, 6012–6018. [Google Scholar] [CrossRef]
- Lin, Y.-C.; Yu, K.-H.; Huang, S.-L.; Liu, Y.-H.; Wang, Y.; Liu, S.-T.; Chen, J.-T. Alternating ethylene–norbornene copolymerization catalyzed by cationic organopalladium complexes bearing hemilabile bidentate ligands of α-amino–pyridines. Dalton Trans. 2009, 41, 9058–9067. [Google Scholar] [CrossRef] [PubMed]
- Lim, N.K.; Arndtsen, B.A. Novel Coupling of Two Mechanistically Distinct Polymerizations on a Single Metal Center: Palladium-Mediated Synthesis of Poly(norbornene)–Poly(THF) Block Copolymers. Macromolecules 2000, 33, 2305–2307. [Google Scholar] [CrossRef]
- Reddy, K.R.; Surekha, K.; Lee, G.-H.; Peng, S.-M.; Chen, J.-T.; Liu, S.-T. Study of Insertion of Olefins and/or Carbon Monoxide into Phosphine−Imine Palladium Methyl Complexes. Organometallics 2001, 20, 1292–1299. [Google Scholar] [CrossRef]
- Setsune, J.-I.; Yamauchi, T.; Tanikawa, S.; Hirose, Y.; Watanabe, J.-Y. Insertion of CO and Alkenes into the (methyl)Pd Complexes of the Bidentate Porphyrin Ligand. Organometallics 2004, 23, 6058–6065. [Google Scholar] [CrossRef]
- Agostinho, M.; Braunstein, P.; Welter, R. Phosphinito- and phosphonito-oxazoline Pd(II) complexes as CO/ethylene insertion intermediates: synthesis and structural characterization. Dalton Trans. 2007, 7, 759–770. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.-J.; Yang, F.-Z.; Lin, Y.-F.; Chang, Y.-C.; Yu, K.-H.; Chang, M.-C.; Lee, G.-H.; Liu, Y.-H.; Wang, Y.; Liu, S.-T.; et al. Unsymmetrical bidentate ligands of α-aminoaldimines leading to sterically controlled selectivity of geometrical isomerism in square planar coordination. Dalton Trans. 2008, 43, 5945–5956. [Google Scholar] [CrossRef] [PubMed]
- Amatore, C.; Catellani, M.; Deledda, S.; Jutand, A.; Motti, E. Rates of the Oxidative Addition of Benzyl Halides to a Metallacyclic Palladium(II) Complex and of the Reductive Elimination from a Benzyl-Palladium (IV) Complex. Organometallics 2008, 27, 4549–4554. [Google Scholar] [CrossRef]
- Walter, M.D.; White, P.S.; Brookhart, M. γ-Agostic interactions stabilize the propagating species in the vinyl addition polymerization of norbornene. Chem. Commun. 2009, 42, 6361–6363. [Google Scholar] [CrossRef] [PubMed]
- Walter, M.D.; Moorhouse, R.A.; Urbin, S.A.; White, P.S.; Brookhart, M. γ-Agostic Species as Key Intermediates in the Vinyl Addition Polymerization of Norbornene with Cationic (allyl)Pd Catalysts: Synthesis and Mechanistic Insights. J. Am. Chem. Soc. 2009, 131, 9055–9069. [Google Scholar] [CrossRef] [PubMed]
- Kang, M.; Sen, A. Reaction of Palladium 1,5-Cyclooctadiene Alkyl Chloride with Norbornene Derivatives: Relevance to Metal-Catalyzed Addition Polymerization of Functionalized Norbornenes. Organometallics 2004, 23, 5396–5398. [Google Scholar] [CrossRef]
- Vicente, J.; Arcas, A.; Fernández-Hernández, J.M.; Bautista, D. New Acetonyl Palladium(II) Complexes. Organometallics 2008, 27, 3978–3985. [Google Scholar] [CrossRef]
- Takamiya, I.; Yamashita, M.; Nozaki, K. Syntheses of Ester-Substituted Norbornyl Palladium Complexes Ligated with tBu3P: Studies on the Insertion of exo- and endo-Monomers in the Ester-Substituted Norbornene Polymerization. Organometallics 2008, 27, 5347–5352. [Google Scholar] [CrossRef]
- Dieck, H.; Svoboda, M. Metallierung einer nichtaktivierten Alkyl-Gruppe im Nickelkomplex. Chem. Ber. 1976, 109, 1657–1664. [Google Scholar] [CrossRef]
- Tempel, D.J.; Johnson, L.K.; Huff, R.L.; White, P.S.; Brookhart, M. Mechanistic Studies of Pd(II)–α-Diimine-Catalyzed Olefin Polymerizations. J. Am. Chem. Soc. 2000, 122, 6686–6700. [Google Scholar] [CrossRef]
- Diez, V.; Cuevas, J.V.; García-Herbosa, G.; Aullón, G.; Charmant, J.P.H.; Carbayo, A.; Muñoz, A. 1H NMR Direct Observation of Enantiomeric Exchange in Palladium(II) and Platinum(II) Complexes Containing N,N’ Bidentate Aryl-pyridin-2-ylmethyl-amine Ligands. Inorg. Chem. 2007, 46, 568–577. [Google Scholar] [CrossRef] [PubMed]
- Dell’Anna, M.M.; Giardina-Papa, D.; Ibáñez, S.; Martín, A.; Mastrorilli, P.; Nobile, C.F.; Peruzzini, M. Insertion of CO and Strained Olefins into Organometallic (Ferrocenylmethyl)phosphane Palladium Complexes. Eur. J. Inorg. Chem. 2009, 29, 4454–4463. [Google Scholar] [CrossRef]
- Espenson, J.H. Chemical Kinetics and Reaction Mechanisms, 2nd ed.; McGraw-Hill: New York, NY, USA, 2002; pp. 156–160. [Google Scholar]
- Gates, D.P.; Svejda, S.A.; Oñate, E.; Killian, C.M.; Johnson, L.K.; White, P.S.; Brookhart, M. Synthesis of Branched Polyethylene using (α-Diimine)nickel(II) Catalysts: Influence of Temperature, Ethylene Pressure, and Ligand Structure on Polymer Properties. Macromolecules 2000, 33, 2320–2334. [Google Scholar] [CrossRef]
- Yang, F.-Z.; Wang, Y.-H.; Chang, M.-C.; Yu, K.-H.; Huang, S.-L.; Liu, Y.-H.; Wang, Y.; Liu, S.-T.; Chen, J.-T. Kinetic and Mechanistic Studies of Geometrical Isomerism in Neutral Square-Planar Methylpalladium Complexes Bearing Unsymmetrical Bidentate Ligands of α-Aminoaldimines. Inorg. Chem. 2009, 48, 7639–7644. [Google Scholar] [CrossRef] [PubMed]
- Johnson, L.K.; Killian, C.M.; Brookhart, M. New Pd(II)- and Ni(II)-Based Catalysts for Polymerization of Ethylene and α-Olefins. J. Am. Chem. Soc. 1995, 117, 6414–6415. [Google Scholar] [CrossRef]
- Johnson, L.K.; Mecking, S.; Brookhart, M. Copolymerization of Ethylene and Propylene with Functionalized Vinyl Monomers by Palladium(II) Catalysts. J. Am. Chem. Soc. 1996, 118, 267–268. [Google Scholar] [CrossRef]
- Killian, C.M.; Tempel, D.J.; Johnson, L.K.; Brookhart, M. Living Polymerization of α-Olefins Using NiII-α-Diimine Catalysts. Synthesis of New Block Polymers Based on α-Olefins. J. Am. Chem. Soc. 1996, 118, 11664–11665. [Google Scholar] [CrossRef]
- Mecking, S.; Johnson, L.K.; Wang, L.; Brookhart, M. Mechanistic Studies of the Palladium-Catalyzed Copolymerization of Ethylene and α-Olefins with Methyl Acrylate. J. Am. Chem. Soc. 1998, 120, 888–899. [Google Scholar] [CrossRef]
- Berry, R.S. Correlation of Rates of Intramolecular Tunneling Processes, with Application to Some Group V Compounds. J. Chem. Phys. 1960, 32, 933–938. [Google Scholar] [CrossRef]
- Ugi, I.; Marquarding, D.; Klusacek, H.; Gillespie, P.; Ramirez, F. Berry Pseudorotation and Turnstile Rotation. Acc. Chem. Res. 1971, 4, 288–296. [Google Scholar] [CrossRef]
- Casares, J.A.; Espinet, P. Dynamic Behavior of [Pd(C6F5)2(SPPynPh3-n)] Complexes: Evidence for a Turnstile Mechanism in Intramolecular Exchange. Inorg. Chem. 1997, 36, 5428–5431. [Google Scholar] [CrossRef]
- Anderson, G.K.; Cross, R.J. Isomerisation Mechanisms of Square-Planar Complexes. Chem. Soc. Rev. 1980, 9, 185–215. [Google Scholar] [CrossRef]
- Ozawa, F.; Ito, T.; Nakamura, Y.; Yamamoto, A. Mechanisms of Thermal Decomposition of trans- and cis-Dialkylbis-(tertiary phosphine)palladium(II). Reductive Elimination and trans to cis Isomerization. Bull. Chem. Soc. Jpn. 1981, 54, 1868–1880. [Google Scholar] [CrossRef]
- Pavonessa, R.S.; Trogler, W.C. Solvent-Dependent Reactions of Carbon Dioxide with a Platinum(II) Dihydride. Reversible Formation of a Platinum(II) Formatohydride and a Cationic Platinum(II) Dimer, [Pt2H3(PEt3)4][HCO2]. J. Am. Chem. Soc. 1982, 104, 3529–3530. [Google Scholar] [CrossRef]
- Low, J.J.; Goddard III, W.A. Theoretical Studies of Oxidative Addition and Reductive Elimination. 3. C–H and C–C Reductive Coupling from Palladium and Platinum Bis(phosphine) Complexes. J. Am. Chem. Soc. 1986, 108, 6115–6128. [Google Scholar] [CrossRef]
- Tatsumi, K.; Hoffmann, R.; Yamamoto, A.; Stille, J.K. Reductive Elimination of d8-Organotransition Metal Complexes. Bull. Chem. Soc. Jpn. 1981, 54, 1857–1867. [Google Scholar] [CrossRef]
- Deubel, D.V.; Ziegler, T. Challenge of the Copolymerization of Olefins with N-Containing Polar Monomers. Systematic Screening of Nickel(II) and Palladium(II) Catalysts with Brookhart and Grubbs Ligands. 2. Chain-Propagation Barriers, Intrinsic Regioselectivity, and Curtin–Hammett Reactivity. Organometallics 2002, 21, 4432–4441. [Google Scholar]
- Noda, S.; Nakamura, A.; Kochi, T.; Chung, L.W.; Morokuma, K.; Nozaki, K. Mechanistic Studies on the Formation of Linear Polyethylene Chain Catalyzed by Palladium Phosphine—Sulfonate Complexes: Experiment and Theoretical Studies. J. Am. Chem. Soc. 2009, 131, 14088–14100. [Google Scholar] [CrossRef] [PubMed]
- Zheng, F.; Hutton, A.T.; van Sittert, C.G.C.E.; Moss, J.R.; Mapolie, S.F. Synthesis, structural characterization and cis-trans isomerization of novel (salicylaldiminato)platinum(II) complexes. Dalton Trans. 2013, 42, 11163–11179. [Google Scholar] [CrossRef] [PubMed]
- Musaev, D.G.; Svensson, M.; Morokuma, K.; Strömberg, S.; Zetterberg, K.; Siegbahn, P.E.M. Density Functional Study of the Mechanism of the Palladium(II)-Catalyzed Ethylene Polymerization Reaction. Organometallics 1997, 16, 1933–1945. [Google Scholar] [CrossRef]
- Froese, R.D.J.; Musaev, D.G.; Morokuma, K. Theoretical Study of Substituent Effects in the Diimine−M(II) Catalyzed Ethylene Polymerization Reaction Using the IMOMM Method. J. Am. Chem. Soc. 1998, 120, 1581–1587. [Google Scholar] [CrossRef]
- Michalak, A.; Ziegler, T. DFT Studies on Substituent Effects in Palladium-Catalyzed Olefin Polymerization. Organometallics 2000, 19, 1850–1858. [Google Scholar] [CrossRef]

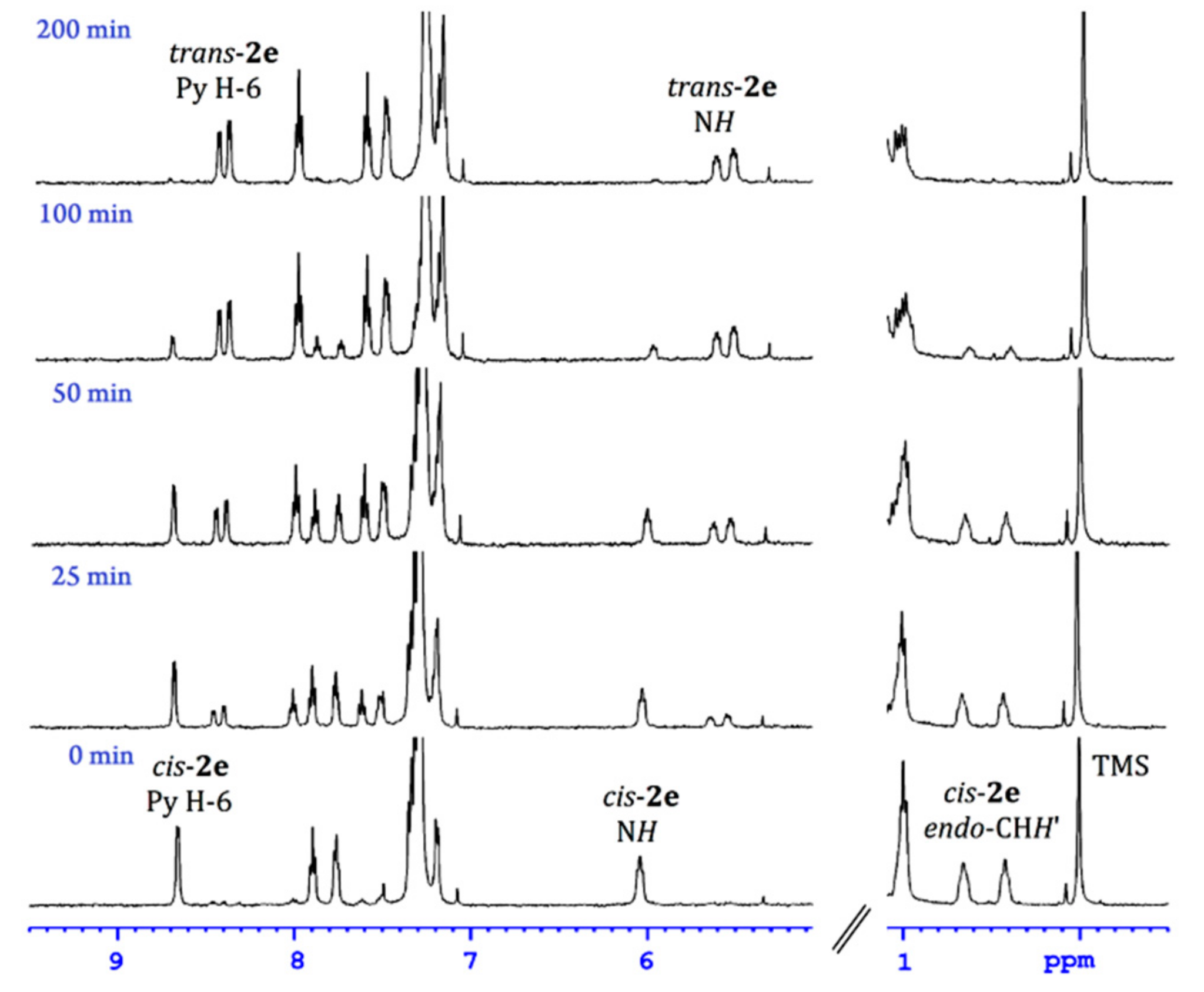

| RHNCH2(o-C6H4N) | Me-Pd [8] | MeC7H10-Pd | ||||
|---|---|---|---|---|---|---|
| R | T (%) | C (%) | T (%) (anti/syn) | C (%) | ||
| iPr | 1a | 82 | 18 | 2a | 100 (30/70) | 0 |
| tBu | 1b | 100 | 0 | 2b | 100 (27/73) | 0 |
| Ph | 1c | 93 | 7 | 2c | 96 (41/55) | 4 |
| 2,6-Me2C6H3 | 1d | 60 | 40 | 2d | 87 (47/40) | 13 |
| 2,6-iPr2C6H3 | 1e | 62 | 38 | 2e | 94 (44/50) | 6 |
| Entry | Cat | Temp. (K) | [Cat] (×10−3 M) | [N] a/[Cat] | kobs (10−4 s−1) | |
|---|---|---|---|---|---|---|
| C | T | |||||
| 1 | 1a | 263 | 5.3 | 28.7 | - b | 8.69 |
| 2 | 1b | 263 | 24.9 | 82.2 | - c | 2.40 |
| 3 | 1c | 263 | 2.0 | 23.2 | - b | 51.60 |
| 4 | 1d | 263 | 1.1 | 16.7 | 134.00 | 21.80 |
| 5 | 1e | 263 | 3.4 | 10.8 | - b | 5.47 |
| 6 | 1e | 263 | 3.4 | 24.2 | - b | 6.19 |
| 7 | 1e | 263 | 3.4 | 62.6 | - b | 10.10 |
| 8 | 1e | 263 | 3.4 | 134.5 | - b | 12.90 |
| 9 | 1e | 263 | 3.4 | 260.9 | - b | 23.80 |
| 10 | 1e | 263 | 1.7 | 21.0 | 110.00 | - |
| 11 | 1e | 258 | 3.3 | 31.1 | 109.00 | 2.35 |
| 12 | 1e | 253 | 3.4 | 26.3 | 41.30 | 1.29 |
| 13 | 1e | 243 | 3.3 | 28.0 | 16.80 | - |
| 14 | 1e | 238 | 3.4 | 27.2 | 9.68 | - |
| 15 | 1e | 233 | 3.3 | 20.1 | 7.11 | - |
| 16 | 1e | 233 | 3.3 | 33.7 | 10.80 | - |
| 17 | 1e | 233 | 3.3 | 40.5 | 12.90 | - |
| 18 | 1e | 228 | 3.4 | 31.3 | 2.50 | - |
| RHNCH2(o-C6H4N) | E-Coord. Complexes | |||||
|---|---|---|---|---|---|---|
| R | T (%) | C (%) | T (%) | C (%) | ||
| iPr | 1a’ | 58 | 42 | 5a’ | 59 | 41 |
| tBu | 1b’ | 63 | 37 | 5b’ | 58 | 42 |
| Ph | 1c’ | 100 | 0 | 5c’ | 100 | 0 |
| 2,6-Me2C6H3 | 1d’ | 59 | 41 | 5d’ | 64 | 36 |
| 2,6-iPr2C6H3 | 1e’ | 38 | 62 | 5e’ | 67 | 33 |
| Entry | Temp. (K) | Cat | [Cat] (10−3 M) | [E] a/[Cat] | kE (10−4 s−1) | ||
|---|---|---|---|---|---|---|---|
| C | T | Total b | |||||
| 1 | 263 | 1a | 5.3 | 18.3 | c | c | 6.44 |
| 2 | 263 | 1b | 3.6 | 21.3 | d | 8.05 | - |
| 3 | 263 | 1c | 4.4 | 25.9 | e | 1.93 | - |
| 4 | 263 | 1d | 3.2 | 17.7 | 4.74 | 4.72 | 4.28 |
| 5 | 268 | 1e | 3.4 | 21.7 | 13.10 | 14.30 | 13.60 |
| 6 | 263 | 1e | 3.3 | 14.3 | 4.36 | 3.80 | 4.14 |
| 7 | 263 | 1e | 3.3 | 25.0 | 4.46 | 4.19 | 4.37 |
| 8 | 263 | 1e | 3.3 | 51.9 | 4.91 | 4.27 | 4.71 |
| 9 | 258 | 1e | 3.4 | 49.4 | 3.40 | 3.20 | 3.37 |
| 10 | 253 | 1e | 3.4 | 32.5 | 1.16 | 1.17 | 1.16 |
| 11 | 243 | 1e | 3.3 | 27.2 | 0.41 | 0.41 | 0.41 |
| Cat | Act. b | NBcoc c (mol %) | Single c (mol %) | Diads c (mol %) | Triads c (mol %) | Alter. c (mol %) |
|---|---|---|---|---|---|---|
| 1a | 14 | 44.0 | 33.7 | 6.9 | 3.4 | 76.6 |
| 1b | 5 | 42.9 | 39.0 | 3.9 | 0 | 81.9 |
| 1c | 6 | 47.5 | 26.4 | 12.4 | 8.7 | 71.0 |
| 1d | 27 | 48.1 | 34.8 | 6.9 | 6.4 | 80.8 |
| 1e | 28 | 49.0 | 38.7 | 6.1 | 4.2 | 86.3 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yu, K.-H.; Huang, S.-L.; Liu, Y.-H.; Wang, Y.; Liu, S.-T.; Cheng, Y.-C.; Lin, Y.-F.; Chen, J.-T. Kinetics, Mechanism and Theoretical Studies of Norbornene-Ethylene Alternating Copolymerization Catalyzed by Organopalladium(II) Complexes Bearing Hemilabile α-Amino–pyridine. Molecules 2017, 22, 1095. https://doi.org/10.3390/molecules22071095
Yu K-H, Huang S-L, Liu Y-H, Wang Y, Liu S-T, Cheng Y-C, Lin Y-F, Chen J-T. Kinetics, Mechanism and Theoretical Studies of Norbornene-Ethylene Alternating Copolymerization Catalyzed by Organopalladium(II) Complexes Bearing Hemilabile α-Amino–pyridine. Molecules. 2017; 22(7):1095. https://doi.org/10.3390/molecules22071095
Chicago/Turabian StyleYu, Kuo-Hsuan, Shou-Ling Huang, Yi-Hung Liu, Yu Wang, Shiuh-Tzung Liu, Yuan-Chung Cheng, Ya-Fan Lin, and Jwu-Ting Chen. 2017. "Kinetics, Mechanism and Theoretical Studies of Norbornene-Ethylene Alternating Copolymerization Catalyzed by Organopalladium(II) Complexes Bearing Hemilabile α-Amino–pyridine" Molecules 22, no. 7: 1095. https://doi.org/10.3390/molecules22071095
APA StyleYu, K.-H., Huang, S.-L., Liu, Y.-H., Wang, Y., Liu, S.-T., Cheng, Y.-C., Lin, Y.-F., & Chen, J.-T. (2017). Kinetics, Mechanism and Theoretical Studies of Norbornene-Ethylene Alternating Copolymerization Catalyzed by Organopalladium(II) Complexes Bearing Hemilabile α-Amino–pyridine. Molecules, 22(7), 1095. https://doi.org/10.3390/molecules22071095