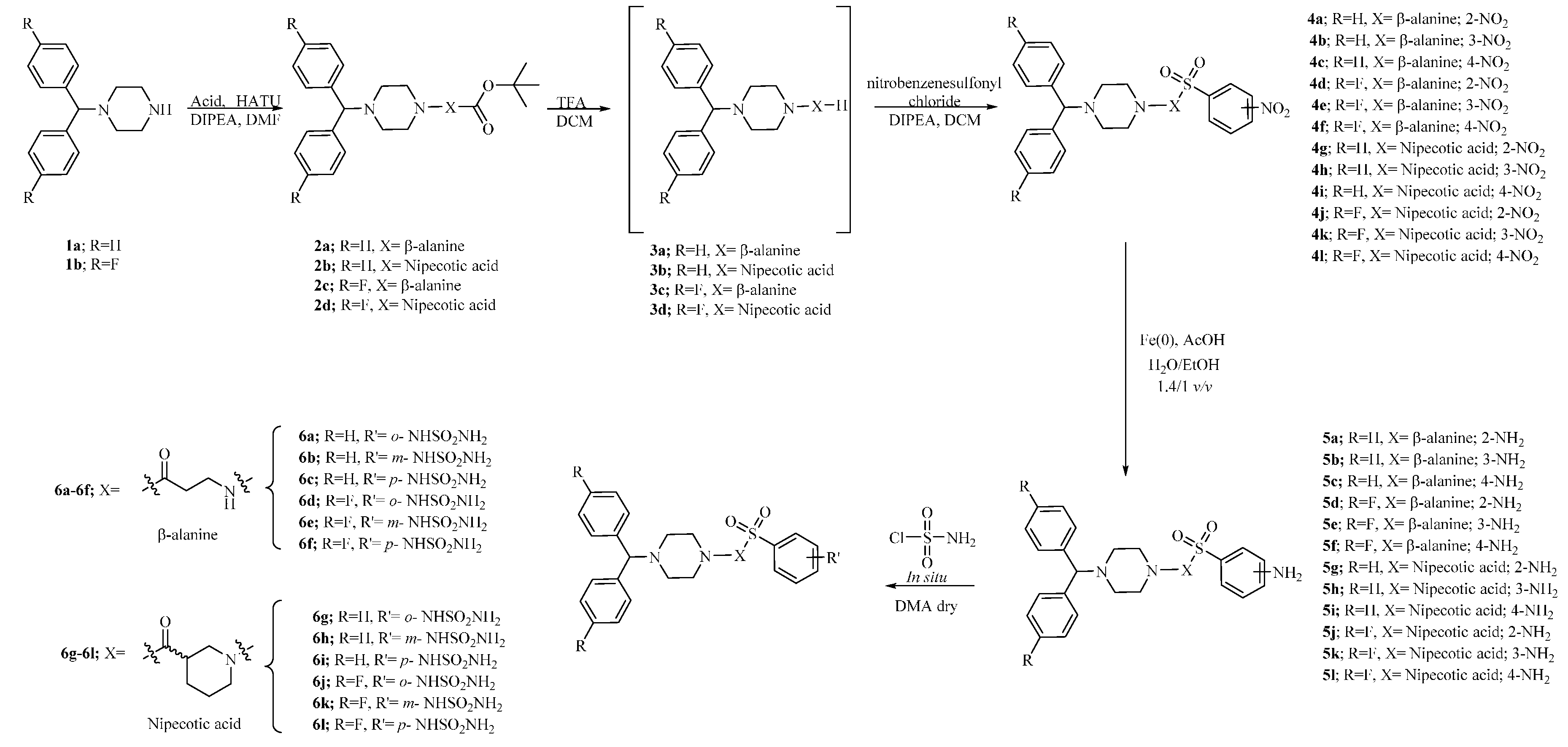
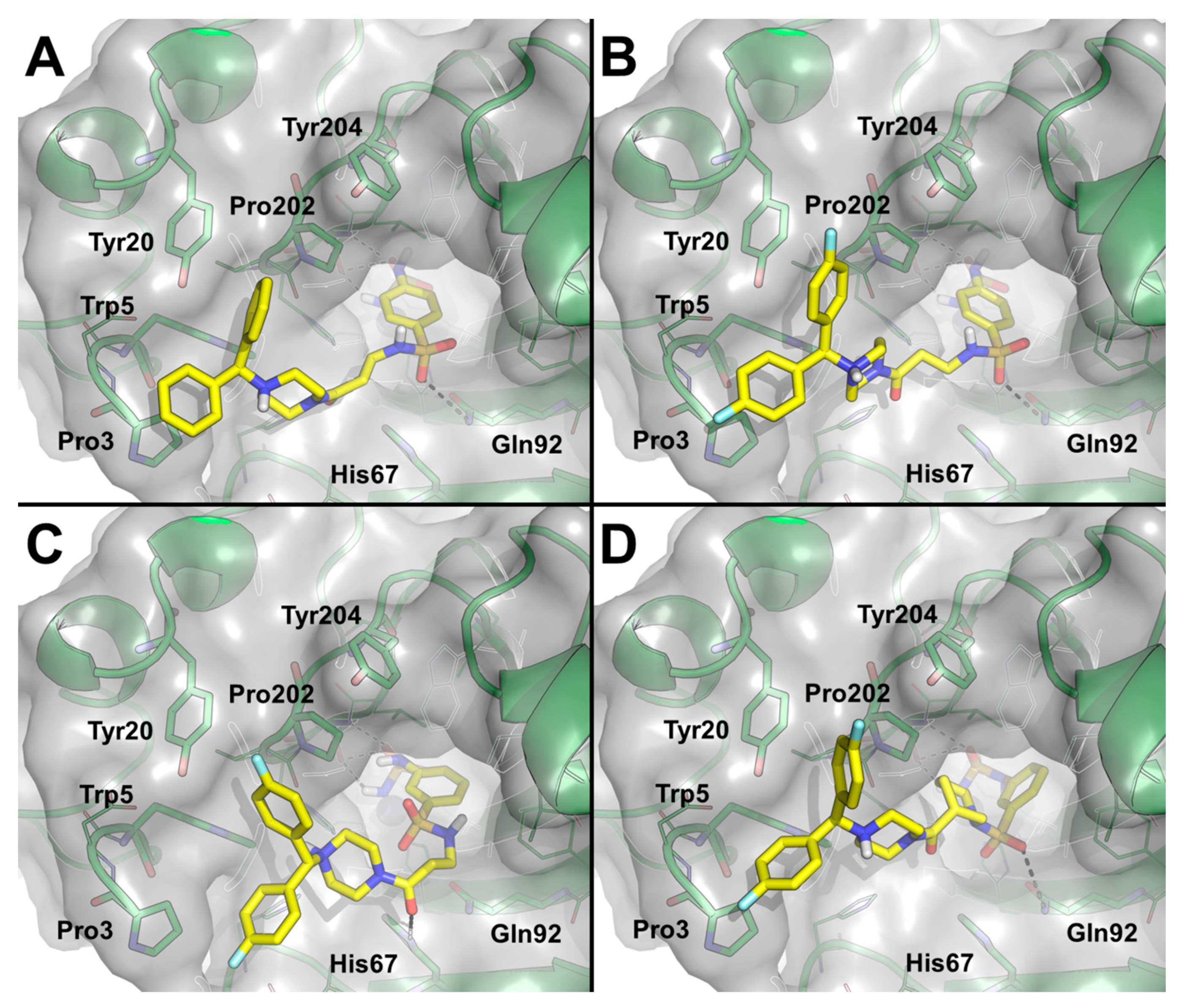
3.1. Chemistry
Anhydrous solvents and all reagents were purchased from Sigma-Aldrich (Milan, Italy), Alfa Aesar (Milan, Italy) and TCI (Milan, Italy). All reactions involving air- or moisture-sensitive compounds were performed under a nitrogen atmosphere using dried glassware and syringes techniques to transfer solutions. Nuclear magnetic resonance spectra (1H-NMR: 400 MHz; 13C-NMR: 100 MHz; 19F-NMR: 376 MHz) were recorded in DMSO-d6 using an Avance III 400 MHz spectrometer (Bruker, Milan, Italy). Chemical shifts are reported in parts per million (ppm) and the coupling constants (J) are expressed in Hertz (Hz). Splitting patterns are designated as follows: s, singlet; d, doublet; t, triplet; q, quadruplet; m, multiplet; brs, broad singlet; dd, double of doublets. The assignment of exchangeable protons (OH and NH) was confirmed by the addition of D2O. Analytical thin-layer chromatography (TLC) was carried out on silica gel F-254 plates (Merck, Milan, Italy). Melting points (m.p.) were carried out in open capillary tubes and are uncorrected. The solvents used in MS measures were acetone, acetonitrile (Chromasolv grade), purchased from Sigma–Aldrich and mQ water 18 MX, obtained from Millipore’s Simplicity system (Milan, Italy). The mass spectra were obtained using a 1200 L triple quadrupole system (Varian, Palo Alto, CA, USA) equipped by Electrospray Source (ESI) operating in both positive and negative ions. Stock solutions of analytes were prepared in acetone at 1.0 mg mL−1 and stored at 4 °C. Working solutions of each analyte were freshly prepared by diluting stock solutions in a mixture of mQ H2O/ACN 1:1 (v/v) up to a concentration of 1.0 μg mL−1. The mass spectra of each analyte were acquired by introducing, via syringe pump at 10 μL min−1, of the its working solution. Raw-data were collected and processed by Varian Workstation Vers. 6.8 software.
3.1.1. General Procedure for the Synthesis of Compounds 2a–d
Compounds
1a,
b (1.0 eq) and the appropriate
N-Boc-protected carboxylic acid (1.1 eq) in DMF (10.0 mL) were treated with DIPEA (2.0 eq), and HATU (1.5 eq) at r.t. for 30 min. When the reaction was complete (TLC monitoring), it was quenched with slush and extracted with ethyl acetate (3 × 15 mL). The combined organic layers were washed with H
2O (3 × 15 mL), dried over Na
2SO
4, filtered-off and concentrated under reduced pressure to afford the title compounds
2a–
d [
17] as white solids.
tert-Butyl (3-(4-benzhydrylpiperazin-1-yl)-3-oxopropyl)carbamate (2a). Using 1a and N-Boc-β-alanine as starting materials and the general procedure described above compound 2a was obtained in 95% yield. 1H-NMR: δ 1.12 (9H, s, 3 × CH3), 2.26 (4H, m, 2 × piperazine-CH2), 2.47 (2H, t, J = 7.2, COCH2), 3.08 (2H, m, CH2NH), 3.38 (4H, m, 2 × piperazine-CH2), 4.30 (1H, s, CH), 7.17 (2H, appt, J = 7.4, Ar-H), 7.28 (4H, appt, J = 7.4, Ar-H), 7.41 (4H, d, J = 7.4, Ar-H), 7.6 (1H, brs, NH).
tert-Butyl 3-(4-benzhydrylpiperazine-1-carbonyl)piperidine-1-carboxylate (2b). Using 1a and N-Boc-nipecotic acid as starting materials compound 2b was obtained in 80% yield. 1H-NMR: δ 1.13 (9H, s, 3 × CH3), 1.30 (1H, m, piperidine-CH), 1.50 (1H, m, piperidine-CH), 1.73 (2H, m, piperidine-CH2), 2.30 (4H, m, 2 × piperazine-CH2), 2.75 (3H, m, piperidine-CH2, COCH), 3.49 (4H, m, 2 × piperazine-CH2), 3.71 (2H, m, piperidine-CH2), 4.30 (1H, s, CH) 7.18 (2H, appt, J = 7.2, Ar-H), 7.29 (4H, appt, J = 7.2, Ar-H), 7.42 (4H, d, J = 7.2, Ar-H).
tert-Butyl (3-(4-(bis(4-fluorophenyl)methyl)piperazin-1-yl)-3-oxopropyl)carbamate (2c). Using 1b and N-Boc-β-alanine as starting materials compound 2c was obtained in 99% yield. 1H-NMR: δ 1.12 (9H, s, 3 × CH3), 2.26(4H, m, 2 × piperazine-CH2), 2.47 (2H, t, J = 7.2, COCH2), 3.08 (2H, m, CH2NH), 3.38 (4H, m, 2 × piperazine-CH2), 4.30 (1H, s, CH), 7.12 (4H, m, Ar-H), 7.42 (4H, m, Ar-H), 7.6 (1H, brs, NH).
tert-Butyl 3-(4-(bis(4-fluorophenyl)methyl)piperazine-1-carbonyl)piperidine-1-carboxylate (2d). Using 1b and N-Boc-nipecotic acid are as starting materials compound 2d was obtained in 95% yield. 1H-NMR: δ 1.13 (9H, s, 3 × CH3), 1.30 (1H, m, piperidine-CH), 1.50 (1H, m, piperidine-CH), 1.73 (2H, m, piperidine-CH2), 2.30 (4H, m, 2 × piperazine-CH2), 2.75 (3H, m, piperidine-CH2, COCH), 3.49 (4H, m, 2 × piperazine-CH2), 3.71 (2H, m, piperidine-CH2), 4.30 (1H, s, CH), 7.12 (4H, m, Ar-H), 7.42 (4H, m, Ar-H).
3.1.2. General Procedure for the Synthesis of Compounds 4a–l
A stirred solution of compounds 2a–d (1.0 eq) in DCM (10.0 mL) was treated with TFA (3.0 eq) and stirred at r.t. for 2h. The reaction mixture was concentrated to dry and co-distilled twice with DCM to afford the corresponding alkyl amines 3a–d as TFA salts (not isolated), which were readily dissolved in DCM (10.0 mL) and treated with DIPEA (5.0 eq) and the appropriate sulfonyl chloride (1.2 eq). The reaction solutions were stirred at r.t. for 1 h, then concentrated to dry and the residue obtained was purified by silica gel column chromatography using ethyl acetate in n-hexane (20–40% v/v) as eluents to afford the titled compounds 4a–l as white solids.
N-(3-(4-Benzhydrylpiperazin-1-yl)-3-oxopropyl)-2-nitrobenzenesulfonamide (4a). Using 3a and 2-nitro- benzenesulfonyl chloride as starting materials compound 4a was obtained in 29% yield according to the general procedure described above; TLC: Rf = 0.39 (ethyl acetate/n-hexane 70% v/v); 1H-NMR: δ 2.22 (4H, m, 2 × piperazine-CH2), 2.45 (2H, t, J = 7.2, COCH2), 3.08 (2H, m, CH2NH), 3.38 (4H, m, 2 × piperazine-CH2), 4.28 (1H, s, CH), 7.17 (2H, appt, J = 7.4, Ar-H), 7.28 (4H, appt, J = 7.4, Ar-H), 7.41 (4H, d, J = 7.4, Ar-H), 7.91 (5H, m, overlapping signals; exchangeable with D2O, SO2NHCH2, 4 × Ar-H); 13C-NMR: δ 32.4, 41.0, 44.7, 51.1, 74.6, 124.4, 126.9, 127.5, 128.5, 129.5, 132.5, 132.7, 134.0, 142.4, 147.7, 168.2; MS (ESI) m/z = 508.9 [M + 1]+.
N-(3-(4-Benzhydrylpiperazin-1-yl)-3-oxopropyl)-3-nitrobenzenesulfonamide (4b). Using 3a and 3-nitro-benzenesulfonyl chloride as starting materials compound 4b was obtained in 56% yield; TLC: Rf = 0.28 (ethyl acetate/n-hexane 50% v/v); 1H-NMR: δ 2.23 (4H, m, 2 × piperazine-CH2), 2.42 (2H, t, J = 6.8, COCH2), 2.98 (2H, t, J = 6.8, CH2NH), 3.37 (4H, m, 2 × piperazine-CH2), 4.27 (1H, s, CH), 7.18 (2H, appt, J = 7.4, Ar-H), 7.28 (4H, appt, J = 7.4, Ar-H), 7.40 (4H, d, J = 7.4, Ar-H), 7.88 (1H, t, J = 7.6, Ar-H), 7.96 (1H, s, exchangeable with D2O, CH2NHSO2), 8.19 (1H, d, J = 7.6, Ar-H), 8.46 (1H, d, J = 7.6, Ar-H), 8.51 (1H, s, Ar-H); 13C-NMR: δ 32.4, 38.8, 44.7, 51.0, 74.7, 121.3, 126.8, 126.9, 127.5, 128.5, 131.2, 132.5, 142.1, 142.4, 147.9, 168.7; MS (ESI) m/z = 508.9 [M + 1]+.
N-(3-(4-Benzhydrylpiperazin-1-yl)-3-oxopropyl)-4-nitrobenzenesulfonamide (4c). Using 3a and 4-nitro-benzenesulfonyl chloride as starting materials compound 4c was obtained in 54% yield; TLC: Rf = 0.72 (ethyl acetate/n-hexane 70% v/v); 1H-NMR: δ 2.27 (4H, m, 2 × piperazine-CH2), 2.50 (2H, t, J = 6.8, COCH2), 3.05 (2H, t, J = 6.8, CH2NH), 3.37 (4H, m, 2 × piperazine-CH2), 4.32 (1H, s, CH), 7.24 (2H, appt, J = 7.4, Ar-H), 7.34 (4H, appt, J = 7.4, Ar-H), 7.46 (4H, d, J = 7.4, Ar-H), 8.03 (1H, s, exchangeable with D2O, CH2NHSO2), 8.08 (2H, d, J = 8.8, Ar-H), 8.46 (2H, d, J = 8.8, Ar-H); 13C-NMR: δ 32.4, 38.9, 44.6, 51.1, 74.7, 124.5, 126.9, 127.5, 128.0, 128.5, 142.4, 146.0, 149.4, 168.0; MS (ESI) m/z = 508.9 [M + 1]+.
N-(3-(4-(bis(4-Fluorophenyl)methyl)piperazin-1-yl)-3-oxopropyl)-2-nitrobenzenesulfonamide (4d). Using 3c and 2-nitrobenzenesulfonyl chloride as starting materials compound 4d was obtained in 53% yield; TLC: Rf = 0.30 (MeOH/DCM 10% v/v); 1H-NMR: δ 2.20 (4H, m, 2 × piperazine-CH2), 2.47 (2H, m, COCH2), 3.08 (2H, t, J = 6.8, CH2NH), 3.38 (4H, m, 2 × piperazine-CH2), 4.37 (1H, s, CH), 7.12 (4H, m, Ar-H), 7.42 (4H, m, Ar-H), 7.85–7.98 (5H, m, exchangeable with D2O, SO2NHCH2, 4 × Ar-H); 13C-NMR: δ 32.4, 39.1, 44.6, 50.8, 72.5, 116.2 (d, 2JC–F 21), 124.5, 129.3, 130.3 (d, 3JC–F 8), 132.7, 134.0, 138.3, 138.5, 147.7, 162.1 (d, 1JC–F 242), 168.2; δF (376 MHz, DMSO-d6) −115.6 (2F, s); MS (ESI) m/z = 543.43 [M − 1]+.
N-(3-(4-(bis(4-Fluorophenyl)methyl)piperazin-1-yl)-3-oxopropyl)-3-nitrobenzenesulfonamide (4e). Using 3c and 3-nitrobenzenesulfonyl chloride as starting materials compound 4e was obtained in 66%; TLC: Rf = 0.25 (MeOH/DCM 5% v/v); 1H-NMR: δ 2.19 (4H, m, 2 × piperazine-CH2), 2.43 (2H, m, COCH2), 2.98 (2H, t, J = 6.8, CH2NH), 3.34 (4H, m, 2 × piperazine-CH2), 4.36 (1H, s, CH), 7.11 (4H, t, J = 9.0, Ar-H), 7.42 (4H, mAr-H), 7.88 (1H, t, J = 7.6, Ar-H), 7.96 (1H, s, exchangeable with D2O, CH2NHSO2), 8.19 (1H, d, J = 7.6, Ar-H), 8.46 (1H, d, J = 7.6, Ar-H), 8.51 (1H, s, Ar-H); 13C-NMR: δ 32.4, 38.8, 44.7, 51.4, 72.6, 116.0 (d, 2JC–F 21), 121.4, 127.0, 130.3 (d, 3JC–F 8), 132.6, 138.2, 138.4, 142.1, 147.9, 162.1 (d, 1JC–F 242), 168.1; δF (376 MHz, DMSO-d6) −115.6 (2F, s); MS (ESI) m/z = 545.11 [M + 1]+.
N-(3-(4-(bis(4-Fluorophenyl)methyl)piperazin-1-yl)-3-oxopropyl)-4-nitrobenzenesulfonamide (4f). Using 3c and 4-nitrobenzenesulfonyl chloride as starting materials compound 4f was obtained in 48% yield; TLC: Rf = 0.48 (MeOH/DCM 5% v/v); 1H-NMR: δ 2.19 (4H, m, 2 × piperazine-CH2), 2.43 (2H, t, J = 6.8, COCH2), 2.98 (2H, q, J = 6.8, CH2NH), 3.36 (4H, m, 2 × piperazine-CH2), 4.36 (1H, s, CH), 7.12 (4H, t, J = 8.8, Ar-H), 7.41 (4H, mAr-H), 7.97 (1H, t, J = 5.6, exchangeable with D2O, CH2NHSO2), 8.02 (2H, d, J = 9.2, Ar-H), 8.40 (2H, d, J = 9.2, Ar-H); 13C-NMR: δ 32.6, 38.8, 44.6, 51.4, 72.6, 115.9 (d, 2JC–F 21), 124.6, 128.1, 130.3 (d, 3JC–F 8), 138.6, 146.0, 149.5, 162.0 (d, 1JC–F 242), 168.1; 19F-NMR: δ −115.6 (2F, s); MS (ESI) m/z = 545.09 [M + 1]+.
(4-Benzhydrylpiperazin-1-yl)(1-((2-nitrophenyl)sulfonyl)piperidin-3-yl)methanone (4g). Using 3b and 2-nitrobenzenesulfonyl chloride as starting materials compound 4g was obtained in 31% yield; TLC: Rf = 0.40 (ethyl acetate/n-hexane 60% v/v); 1H-NMR: δ 1.30 (1H, m, piperidine-CH), 1.50 (1H, m, piperidine-CH), 1.73 (2H, m, piperidine-CH2), 2.30 (4H, m, 2 × piperazine-CH2), 2.75 (3H, m, piperidine-CH2, COCH), 3.49 (4H, m, 2 × piperazine-CH2), 3.71 (2H, m, piperidine-CH2), 4.30 (1H, s, CH) 7.18 (2H, appt, J = 7.2, Ar-H), 7.29 (4H, appt, J = 7.2, Ar-H), 7.42 (4H, d, J = 7.2, Ar-H) 7.90 (2H, m, Ar-H), 8.02 (2H, m, Ar-H); 13C-NMR: δ 23.9, 26.6, 41.0, 44.7, 45.8, 47.9, 51.9, 74.6, 124.1, 126.9, 127.5, 128.5, 129.6, 130.1, 132.2, 134.6, 142.4, 147.7, 170.2 ; MS (ESI) m/z = 548.9 [M + 1]+.
(4-Benzhydrylpiperazin-1-yl)(1-((3-nitrophenyl)sulfonyl)piperidin-3-yl)methanone (4h). Using 3b and 3-nitrobenzenesulfonyl chloride as starting materials compound 4h was obtained in 47% yield; TLC: Rf = 0.47 (ethyl acetate/n-hexane 60% v/v); 1H-NMR: δ 1.29 (1H, m, piperidine-CH), 1.52 (1H, m, piperidine-CH), 1.73 (2H, m, piperidine-CH2), 2.30 (4H, m, 2 × piperazine-CH2), 2.71 (1H, m, COCH), 2.86 (2H, m, piperidine-CH2), 3.49 (4H, m, 2 × piperazine-CH2), 3.71 (2H, m, piperidine-CH2), 4.31 (1H, s, CH), 7.18 (2H, appt, J = 7.4, Ar-H), 7.30 (4H, appt, J = 7.4, Ar-H), 7.43 (4H, d, J = 7.4, Ar-H), 7.96 (1H, t, J = 8.8, Ar-H), 8.19 (1H, d, J = 8.8, Ar-H), 8.39 (1H, s, Ar-H); 8.55 (1H, d, J = 8.8, Ar-H); 13C-NMR: δ 23.6, 26.6, 41.0, 44.9, 45.9, 48.0, 51.9, 74.7, 121.8, 126.9, 127.5, 127.6, 127.7, 128.6, 131.6, 133.3, 142.5, 148.1, 170.3; MS (ESI) m/z = 548.9 [M + 1]+.
(4-Benzhydrylpiperazin-1-yl)(1-((4-nitrophenyl)sulfonyl)piperidin-3-yl)methanone (4i). Using 3b and 4-nitrobenzenesulfonyl chloride compound 4i was obtained in 45% yield; TLC: Rf = 0.33 (ethyl acetate/n-hexane 40% v/v); 1H-NMR: δ 1.22 (1H, m, piperidine-CH), 1.53 (1H, m, piperidine-CH), 1.69 (2H, m, piperidine-CH2), 2.32 (6H, m, 2 × piperazine-CH2, piperidine-CH2), 2.80 (1H, m, COCH), 3.49 (4H, m, 2 × piperazine-CH2), 3.54 (2H, m, piperidine-CH2), 4.30 (1H, s, CH), 7.18 (2H, appt, J = 7.4, Ar-H), 7.30 (4H, appt, J = 7.4, Ar-H), 7.43 (4H, d, J = 7.4, Ar-H), 7.99 (2H, d, J = 9.0), 8.43 (2H, d, J = 9.0); 13C-NMR: δ 23.6, 26.6, 41.0, 44.8, 45.9, 48.1, 51.9, 74.6, 124.7, 126.9, 127.5, 128.5, 128.8, 141.3, 142.4, 149.9, 170.3; MS (ESI) m/z = 548.9 [M + 1]+.
(4-(bis(4-Fluorophenyl)methyl)piperazin-1-yl)(1-((2-nitrophenyl)sulfonyl)piperidin-3-yl)methanone (4j). Using 3d and 2-nitrobenzenesulfonyl chloride as starting materials compound 4j was obtained in 57% yield; TLC: Rf = 0.52 (ethyl acetate/n-hexane 70% v/v); 1H-NMR: δ 1.25 (1H, m, piperidine-CH), 1.50 (1H, m, piperidine-CH), 1.74 (2H, m, piperidine-CH2), 2.26 (4H, m, 2 × piperazine-CH2), 2.63 (1H, m, COCH), 2.75 (2H, m, piperidine-CH2), 3.46 (4H, m, 2 × piperazine-CH2), 3.64 (2H, m, piperidine-CH2), 4.41 (1H, s, CH), 7.12 (4H, t, J = 8.4, Ar-H), 7.43 (4H, m, Ar-H), 7.87 (4H, m, Ar-H); 13C-NMR: δ 23.9, 26.6, 41.0, 44.7, 45.7, 47.8, 51.7, 72.5, 115.2, 116.2 (d, 2JC–F 21), 129.3, 129.4, 129.9, 131.1 (d, 3JC–F 9), 134.6, 138.3, 147.7, 162.1 (d, 1JC–F 242), 170.2; 19F-NMR: δ −115.6 (2F, s); MS (ESI) m/z = 585.24 [M + 1]+.
(4-(bis(4-Fluorophenyl)methyl)piperazin-1-yl)(1-((3-nitrophenyl)sulfonyl)piperidin-3-yl)methanone (4k). Using 3d and 3-nitrobenzenesulfonyl chloride as starting materials compound 4k was obtained in 68% yield; TLC: Rf = 0.53 (ethyl acetate/n-hexane 70% v/v); 1H-NMR: δ 1.17 (1H, m, piperidine-CH), 1.55 (1H, m, piperidine-CH), 1.69 (2H, m, piperidine-CH2), 2.26 (6H, m, 2 × piperazine-CH2, piperidine-CH2), 2.80 (1H, m, COCH), 3.63(6H, m, 2 × piperazine-CH2, piperidine-CH2), 4.41 (1H, s, CH), 7.12 (4H, t, J = 8.4, Ar-H), 7.43 (4H, m, Ar-H), 7.96 (1H, t, J = 8.8, Ar-H), 8.16 (1H, d, J = 8.8, Ar-H), 8.35 (1H, m, Ar-H); 8.55 (1H, d, J = 8.8, Ar-H); 13C-NMR: δ 23.6, 26.6, 41.1, 44., 45.9, 48.0, 51.8, 72.6, 115.3 (d, 2JC–F 21), 121.8, 129.3, 129.4, 129.4, 130.5 (d, 3JC–F 8, 133.3), 137.5, 138.4, 148.1, 161 (d, 1JC–F 242), 170.3; 19F-NMR: δ −115.6 (2F, s) MS (ESI) m/z = 585.14 [M + 1]+.
(4-(bis(4-Fluorophenyl)methyl)piperazin-1-yl)(1-((4-nitrophenyl)sulfonyl)piperidin-3-yl)methanone (4l). Using 3d and 4-nitrobenzenesulfonyl chloride as starting materials compound 4l was obtained in 57% yield; TLC: Rf = 0.84 (ethyl acetate/n-hexane 70% v/v); 1H-NMR: δ 1.15 (1H, m, piperidine-CH), 1.55 (1H, m, piperidine-CH), 1.69 (2H, m, piperidine-CH2), 2.26 (6H, m, 2 × piperazine-CH2, piperidine-CH2), 2.81 (1H, m, COCH), 3.56 (6H, m, 2 × piperazine-CH2, piperidine-CH2), 4.39 (1H, s, CH), 7.12 (4H, t, J = 8.4, Ar-H), 7.43 (4H, m, Ar-H), 7.98 (2H, d J = 9.2, Ar-H) 8.42 (2H, d J = 9.2, Ar-H); 13C-NMR: δ 23.6, 26.6, 41.0, 44.7, 45.8, 48.0, 51.7, 72.5, 115.3 (d, 2JC–F 21), 124 .6, 128.9, 129.3 (d, 3JC–F 9), 138.2, 141.4, 149.9, 162.0 (d, 1JC–F 24), 170.2; MS (ESI) m/z = 585.15 [M + 1]+.
3.1.3. General Procedure for the Synthesis of Amino Benzensulfonamides 5a–l
The appropriate nitrobenzenesulfonamides
4a–
l (1.0 eq) in a solution of H
2O (0.4 mL) and EtOH (0.3 mL) was treated with glacial AcOH (0.05 mL) and Fe (0) (12.0 eq). The reaction mixture was stirred at 75 °C for 1 h (TLC monitoring), then cooled to r.t. and diluted with EtOAc (10.0 mL). The mixture was filtered through Celite 521
®, washed with a saturated NaHCO
3 aqueous solution (3 × 15 mL), brine (3 × 10 mL) and dried over Na
2SO
4. The organic solvent was evaporated
in vacuo to give an oil residue, which was triturated from Et
2O, to afford the titled compounds
5a–
l [
18] as white solids.
2-Amino-N-(3-(4-benzhydrylpiperazin-1-yl)-3-oxopropyl)benzenesulfonamide (5a). Compound 5a was obtained in 80% yield; m.p. 151–153 °C; TLC: Rf = 0.17 (ethyl acetate/n-hexane 70% v/v); 1H-NMR: δ 2.30 (4H, m, 2 × piperazine-CH2), 2.42 (2H, t, J = 7.2, COCH2), 2.93 (2H, m, CH2NH), 3.33 (4H, m, overlapped with the water peak, 2 × piperazine-CH2), 4.34 (1H, s, CH), 5.92 (2H, s, exchangable with D2O, NH2), 6.63 (1H, t, J = 7.2, Ar-H), 6.83 (1H, m, Ar-H), 7.25 (3H, m, overlapping signals, exchangeable with D2O, SO2NHCH2, 2 × Ar-H), 7.33 (4H, t, J = 7.6, Ar-H), 7.47 (5H, m, 5 × Ar-H); 13C-NMR: δ 33.4, 41.9, 45.6, 52.5, 75.6, 116.1, 117.8, 120.6, 127.9, 128.5, 129.5, 129.9, 134.4, 143.4, 147.2, 169.3; m/z (ESI positive) 479.2 [M + H]+.
3-Amino-N-(3-(4-benzhydrylpiperazin-1-yl)-3-oxopropyl)benzenesulfonamide (5b). Compound 5b was obtained in 85% yield; m.p. 110–112 °C; TLC: Rf = 0.11 (ethyl acetate/n-hexane 50% v/v); 1H-NMR: δ 2.29 (4H, m, 2 × piperazine-CH2), 2.46 (2H, t, m, COCH2), 3.33 (6H, m, NHCH2, 2 X piperazine-CH2), 4.34 (1H, s, CH), 5.59 (2H, s, exchangeable with D2O, NH2), 6.99 (1H, m, Ar-H), 7.02 (1H, m, Ar-H), 7.23 (3H, t, J = 7.6, Ar-H), 7.29 (5H, m, Ar-H), 7.46 (5H, m, overlapping signals, exchangeable with D2O, CH2NHSO2, 4 × Ar-H); 13C-NMR: δ 33.4, 38.4, 45.8, 52.5, 75.6, 116.4, 120.7, 122.5, 126.2, 127.6, 128.2, 129.4, 141.1, 141.6, 148.3, 164.3; m/z (ESI positive) 479.2 [M + H]+.
4-Amino-N-(3-(4-benzhydrylpiperazin-1-yl)-3-oxopropyl)benzenesulfonamide (5c). Compound 5c was obtained in 53% yield; m.p. 146–148 °C; TLC: Rf = 0.25 (ethyl acetate/n-hexane 70% v/v); 1H-NMR: δ 2.26 (4H, m, 2 × piperazine-CH2), 2.48 (2H, t, J = 7.0, COCH2) 3.05 (2H, t, J = 7.0, NHCH2), 3.43 (4H, m, 2 × piperazine-CH2), 4.32 (1H, s, CH), 5.95 (2H, s, exchange with D2O, NH2), 6.63 (1H, d, J = 8.0, Ar-H), 7.23 (3H, m, Ar-H), 7.34 (5H, m, Ar-H), 7.45 (6H, m, overlapping signals, exchangeable with D2O, CH2NHSO2, 5 × Ar-H); 13C-NMR: δ 33.4, 38.4, 45.8, 52.5, 75.6, 117.6, 127.9, 128.5, 128.7, 129.5, 133.4, 143.4, 144.1, 169.3; m/z (ESI positive) 479.2 [M + H]+.
2-Amino-N-(3-(4-(bis(4-fluorophenyl)methyl)piperazin-1-yl)-3-oxopropyl)benzenesulfonamide (5d). Compound 5d was obtained in 65% yield; m.p. 149–152 °C; TLC: Rf = 0.52 (MeOH/DCM 10% v/v); 1H-NMR: δ 2.26 (4H, m, 2 × piperazine-CH2), 2.42 (2H, t, J = 6.6, COCH2), 3.01 (2H, q, J = 6.6, CH2NH), 3.46 (4H, m, 2 × piperazine-CH2), 4.43 (1H, s, CH), 5.92 (2H, s, exchangeable with D2O, NH2), 6.63 (1H, m, Ar-H), 6.84 (1H, m, Ar-H), 7.17 (4H, m, Ar-H), 7.28 (1H, m, Ar-H), 7.37 (1H, m, exchangeable with D2O, SO2NHCH2), 7.48 (5H, m, Ar-H); 13C-NMR: δ 32.3, 38.5, 45.3, 51.3, 72.6, 116.1, 116.2 (d, 2JC–F 21), 116.8, 117.8, 129.9, 130.3 (d, 3JC–F 8), 134.4, 139.3, 147.2, 162.1 (d, 1JC–F 242), 169.3; 19F-NMR: δ −115.6 (2F, s); m/z (ESI positive) 515.2 [M + H]+.
3-Amino-N-(3-(4-(bis(4-fluorophenyl)methyl)piperazin-1-yl)-3-oxopropyl)benzenesulfonamide (5e). Compound 5e was obtained in 45% yield; m.p. 140–142 °C; TLC: Rf = 0.20 (MeOH/DCM 5% v/v); 1H-NMR: δ 2.26 (4H, m, 2 × piperazine-CH2), 2.46 (2H, t, J = 7.0, COCH2), 2.95 (2H, q, J = 7.0, CH2NH), 3.45 (4H, m, piperazine-CH2), 4.43 (1H, s, CH), 5.62 (2H, s, exchange with D2O, NH2), 6.78 (1H, d, J = 7.0, Ar-H), 6.88 (1H, d, J = 7.0, Ar-H), 6.99 (1H, s, Ar-H), 7.19 (5H, m, Ar-H ), 7.33 (1H, s, exchangeable with D2O, SO2NHCH2) 7.47 (4H, m, Ar-H); 13C-NMR: δ 33.3, 40.4 (overlapped with DMSO peak), 44.7, 51.3, 73.5, 112.9, 114.9, 116.2 (d, 2JC–F 21), 119.0, 130.3 (d, 3JC–F 8), 130.8, 140.2, 142.5, 154.2, 162.0 (d, 1JC–F 242), 170.2; 19F-NMR: δ −115.6 (2F, s); m/z (ESI positive) 515.2 [M + H]+.
4-Amino-N-(3-(4-(bis(4-fluorophenyl)methyl)piperazin-1-yl)-3-oxopropyl)benzenesulfonamide (5f). Compound 5f was obtained in 53% yield; m.p. 160–162 °C (dec.); TLC: Rf = 0.42 (MeOH/DCM 5% v/v); 1H-NMR: δ 2.26 (4H, m, 2 × piperazine-CH2), 2.45 (2H, t, J = 6.6, COCH2), 2.90 (2H, q, J = 6.6, CH2NH), 3.37 (4H, m, 2 × piperazine-CH2), 4.42 (1H, s, CH), 5.96 (2H, s, exchangeable with D2O, NH2), 6.63 (2H, d, J = 8.8, Ar-H), 7.02 (1H, t, J = 6.6, exchangeable with D2O, SO2NHCH2), 7.18 (4H, m, Ar-H), 7.42 (2H, d, J = 8.8, Ar-H), 7.48 (4H, m, Ar-H); 13C-NMR: δ 33.3, 40.4 (overlap with DMSO peak), 44.7, 52.4, 73.6, 113.6, 116.3 (d, 2JC–F 21), 126.1, 129.3, 130.3 (d, 3JC–F 8), 139.3, 153.4, 162.0 (d, 1JC–F 242), 169.5; 19F-NMR: δ −115.6 (2F, s); m/z (ESI positive) 515.2 [M + H]+.
(1-((2-Aminophenyl)sulfonyl)piperidin-3-yl)(4-benzhydrylpiperazin-1-yl)methanone (5g). Compound 5g was obtained in 94% yield; m.p. 161–183 °C; TLC: Rf = 0.55 (ethyl acetate/n-hexane 60% v/v); 1H-NMR: δ 1.52 (2H, m, piperidine-CH2), 1.71 (2H, m, piperidine-CH2), 2.29 (6H, m, 2 × piperazine-CH2, piperidine-CH2), 2.79 (1H, m, COCH), 3.48 (4H, m, 2 X piperazine-CH2) 3.57 (2H, m, piperidine-CH2), 4.35 (1H, s, CH), 6.06 (2H, s, exchangeable with D2O, NH2), 6.65 (1H, t, J = 7.2, Ar-H), 6.87 (1H, d, J = 8.4, Ar-H), 7.22 (2H, m, Ar-H), 7.33 (5H, m, Ar-H), 7.36 (1H, m, Ar-H), 7.41 (4H, m, Ar-H); 13C-NMR: δ 24.6, 27.8, 42.0, 45.8, 46.7, 48.9, 52.9, 75.7, 116.2, 118.3, 127.9, 128.6, 129.5, 130.6, 132.5, 135.0, 143.4, 148.2, 171.5; m/z (ESI positive) 519.2 [M + H]+.
(1-((3-Aminophenyl)sulfonyl)piperidin-3-yl)(4-benzhydrylpiperazin-1-yl)methanone (5h). Compound 5h was obtained in 42% yield; m.p. 157–159 °C; TLC Rf = 0.26 (ethyl acetate/n-hexane 60% v/v); 1H-NMR: δ 1.57 (2H, m, piperidine-CH2), 1.72 (2H, m, piperidine-CH2), 2.29 (6H, m, 2 × piperazine-CH2, piperidine-CH2) 2.82 (1H, m, COCH), 2.49 (6H, m, 2 × piperazine-CH2, piperidine-CH2), 4.36 (1H, s, CH), 5.67 (2H, s, exchange with D2O, NH2), 6.82 (2H, m, Ar-H), 6.91 (1H, m, Ar-H), 7.26 (3H, m, Ar-H), 7.36 (4H, t, J = 7.6, Ar-H), 7.48 (4H, m, Ar-H); 13C-NMR: δ 24.6, 27.7, 35.6, 38.2, 47.0, 49.2, 52.5, 75.6, 114.2, 117.1, 118.7, 126.2, 127.8, 128.5, 129.5, 132.5, 143.4, 148.7, 175.2; m/z (ESI positive) 519.2 [M + H]+.
(1-((4-Aminophenyl)sulfonyl)piperidin-3-yl)(4-benzhydrylpiperazin-1-yl)methanone (5i). Compound 5i was obtained in 70% yield; m.p. 132–135 °C; TLC Rf = 0.38 (ethyl acetate/n-hexane 70% v/v); 1H-NMR: δ 1.60 (2H, m, piperidine-CH2), 1.73 (2H, m, piperidine-CH2), 2.35 (6H, m, 2 × piperazine-CH2, piperidine-CH2), 2.86 (1H, m, COCH), 3.62 (6H, m, 2 × piperazine-CH2, piperidine-CH2), 4.36 (1H, s, CH), 6.10 (2H, s, exchangeable with D2O peak, NH2), 6.67 (2H, d, J = 8.4, Ar-H), 7.23 (2H, t, J = 7.4, Ar-H), 7.36 (6H, m, Ar-H), 7.49 (4H, d, J = 7.4, Ar-H); 13C-NMR: δ) 24.5, 27.7, 42.0, 45.7, 47.3, 49.2, 52.5, 75.6, 113.6, 125.6, 127.8, 128.4, 129.4, 130.2, 143.3, 154.0, 171.3; m/z (ESI positive) 519.2 [M + H]+.
(1-((2-Aminophenyl)sulfonyl)piperidin-3-yl)(4-(bis(4-fluorophenyl)methyl)piperazin-1-yl)methanone (5j). Compound 5j was obtained in 62% yield; m.p. 160–162 °C (dec.); TLC: Rf = 0.72 (ethyl acetate/n-hexane 70% v/v); 1H-NMR: δ 1.56 (2H, m, piperidine-CH2), 1.73 (2H, m, piperidine-CH2), 2.24 (4H, m, 2 × piperazine-CH2), 2.37 (2H, m, piperidine-CH2), 2.80 (1H, m, COCH), 3.56 (6H, m, 2 × piperazine-CH2, piperidine-CH2), 4.41 (1H, s, CH), 6.05 (2H, s, exchangeable with D2O, NH2) 6.66 (1H, t, J = 7.8, Ar-H), 6.88 (1H, d, J = 7.8, Ar-H), 7.18 (4H, m, Ar-H), 7.31 (1H, t, J = 7.8, Ar-H), 7.41 (1H, d, J = 7.8, Ar-H), 7.48 (4H, m, Ar-H); 13C-NMR: δ 24.5, 27.6, 42.0, 44.6, 45.8, 48.9, 52.0, 73.6, 116.1, 116.2 (d, 2JC–F 21), 116.7, 118.2, 130.2, (d, 3JC–F 9), 130.8, 135.6, 139.2, 148.1, 162.1 (d, 1JC–F 242), 171.3; 19F-NMR: δ −115.6 (2F, s); m/z (ESI positive) 555.2 [M + H]+.
(1-((2-Aminophenyl)sulfonyl)piperidin-3-yl)(4-(bis(4-fluorophenyl)methyl)piperazin-1-yl)methanone (5k). Compound 5k was obtained in 72% yield; m.p. 160–162 °C (dec.); TLC: Rf = 0.38 (ethyl acetate/n-hexane 70% v/v); 1H-NMR: δ 1.57 (2H, m, piperidine-CH2), 1.73 (2H, m, piperidine-CH2), 2.29 (6H, m, 2 × piperazine-CH2, piperidine-CH2), 2.82 (1H, m, COCH), 3.55 (4H, m, 2 × piperazine-CH2), 3.63 (2H, m, piperidine-CH2), 4.45 (1H, s, CH), 5.68 (2H, s, exchangeable with D2O, NH2), 6.77 (2H, m, Ar-H), 6.92 (1H, s, Ar-H), 7.18 (4H, m, Ar-H ), 7.28 (1H, m, Ar-H), 7.48 (4H, m, Ar-H ); 13C-NMR: δ 24.6, 27.7, 41.9, 45.7, 47.0, 49.2, 52.3, 73.5, 112.4, 114.8, 116.3 (d, 2JC–F 21), 118.7, 130.3 (d, 3JC–F 8), 130.6, 136.8, 139.2, 150.4, 162.0 (d, 1JC–F 242), 171.4; 19F-NMR: δ −115.6 (2F, s); m/z (ESI positive) 555.2 [M + H]+.
(1-((4-Aminophenyl)sulfonyl)piperidin-3-yl)(4-(bis(4-fluorophenyl)methyl)piperazin-1-yl)methanone (5l). Compound 5l was obtained in 60% yield; m.p. 160–162 °C (dec.); TLC: Rf = 0.42 (ethyl acetate/n-hexane 70% v/v); 1H-NMR: δ 1.56 (2H, m, piperidine-CH2), 1.72 (2H, m, piperidine-CH2), 2.26 (6H, m, 2 × piperazine-CH2, piperidine-CH2), 2.83 (1H, m, COCH), 3.54 (4H, m, 2 × piperazine-CH2), 3.61 (2H, m, piperidine-CH2), 4.45 (1H, s, CH), 6.10 (2H, s, exchangeable with D2O, NH2), 6.68 (2H, d, J = 8.8, Ar-H), 7.18 (4H, m, Ar-H), 7.35 (2H, d, J = 8.8, Ar-H), 7.48 (4H, m, Ar-H); 13C-NMR: δ 24.5, 27.8, 42.3, 45.7, 47.0, 49.2, 52.4, 73.5, 113.6, 116.3 (d, 2JC–F 21), 119.4, 130.3 (d, 3JC–F 8), 130.5, 139.2, 154.1, 162.0 (d, 1JC–F 242), 171.4; 19F-NMR: δ −115.6 (2F, s); m/z (ESI positive) 555.2 [M + H]+.
3.1.4. General Procedure for the Synthesis of Sulfamides 6a–l
The appropriate aminobenzensulfonamides
5a–
l (1.0 eq) dissolved in dry DMA (5.0 mL) at 0 °C were treated with Et
3N (1.3 eq) and freshly prepared sulfamoyl chloride until consumption of starting material was confirmed (TLC monitoring). Then the solution was quenched with slush and extracted with EtOAc (3 × 20 mL). The combined organic layers were washed with NaHCO
3 aqueous solution. (3 × 10 mL), HCl aqueous solution 1.0 M (1 × 10 mL), brine (3 × 10 mL), dried over Na
2SO
4, filtered-off and concentrated under
vacuo. The obtained residue was purified by trituration from Et
2O to afford the titled sulfamides
6a–
l [
19] as white solids.
N-(3-(4-Benzhydrylpiperazin-1-yl)-3-oxopropyl)-2-(sulfamoylamino)benzenesulfonamide (6a). Compound 6a was obtained in 57% yield; m.p. 218–220 °C (dec.); TLC: Rf = 0.50 (MeOH/DCM 10% v/v); 1H-NMR: δ 2.28 (4H, m, 2 × piperazine-CH2), 2.45 (2H, t, J = 7.0, COCH2), 3.02 (2H, q, J = 7.0, CH2NH), 3.37 (4H, m, overlap with H2O, 2 × piperazine-CH2), 4.35 (1H, s, CH), 7.23 (3H, m, Ar-H), 7.33 (4H, t, J = 7.4, Ar-H), 7.46 (4H, d, J = 7.4, Ar-H), 7.58 (2H, s, exchangeable with D2O, NHSO2NH2), 7.50 (2H, m, Ar-H), 7.79 (1H, m, Ar-H), 8.02 (1H, m, exchangeable with D2O, SO2NHCH2) 8.81 (1H, s, exchangeable with D2O, NHSO2NH2); 13C-NMR: δ 33.1, 40.4, 45.5, 52.6, 75.5, 119.5, 123.0, 127.8, 128.5, 129.4, 130.0, 131.7, 136.4, 139.0, 143.9, 170.1; m/z (ESI positive) 558.0 [M + H]+.
N-(3-(4-Benzhydrylpiperazin-1-yl)-3-oxopropyl)-3-(sulfamoylamino)benzenesulfonamide (6b). Compound 6b was obtained in 54% yield; m.p. 165–167 °C; TLC: Rf = 0.42 (MeOH/DCM 10% v/v); 1H-NMR: δ 2.29 (4H, m, 2 × piperazine-CH2), 2.46 (2H, t, J = 7.0, COCH2), 2.97 (2H, m, CH2NH), 3.44 (4H, m, 2 × piperazine-CH2), 4.34 (1H, s, CH), 7.24 (2H, m, Ar-H), 7.28 (2H, s, exchangeable with D2O, NHSO2NH2), 7.33 (5H, m, Ar-H), 7.42 (1H, m, exchangeable with D2O, SO2NHCH2), 7.49 (5H, m, Ar-H), 7.62 (2H, m, Ar-H), 9.96 (1H, s, exchangeable with D2O, NHSO2NH2); 13C-NMR: δ 33.4, 40.4 (overlap with DMSO peak), 45.6, 52.5, 75.6, 116.4, 120.7, 122.5, 127.8, 128.5, 129.5, 130.6, 141.1, 143.4, 145.9, 169.2; m/z (ESI positive) 558.0 [M + H]+.
N-(3-(4-Benzhydrylpiperazin-1-yl)-3-oxopropyl)-4-(sulfamoylamino)benzenesulfonamide (6c). Compound 6c was obtained in 64% yield; m.p. 122–124 °C; TLC: Rf = 0.30 (ethyl acetate/n-hexane 80% v/v); 1H-NMR: δ 2.29 (4H, m, 2 × piperazine-CH2), 2.46 (2H, t, J = 7.0, COCH2), 2.92 (2H, q, J = 7.0, NHCH2), 3.43 (4H, m, 2 × piperazine-CH2), 4.34 (1H, s, CH), 7.22 (2H, m, Ar-H), 7.31 (6H, m, Ar-H), 7.40 (3H, m, exchangeable with D2O, NHSO2NH2, SO2NHCH2), 7.46 (4H, d, J = 7.6, Ar-H), 7.70 (2H, d, J = 8.4, Ar-H), 10.18 (1H, s, exchangeable with D2O, NHSO2NH2); 13C-NMR: δ 33.4, 38.4, 45.8, 52.5, 75.6, 117.6, 127.9, 128.5, 128.7, 129.5, 133.4, 143.4, 144.1, 169.3; m/z (ESI positive) 558.0 [M + H]+.
N-(3-(4-(bis(4-Fluorophenyl)methyl)piperazin-1-yl)-3-oxopropyl)-2(sulfamoylamino)benzenesulfonamide (6d). Compound 6d was obtained in 52% yield; m.p. 162–164 °C; TLC: Rf = 0.42 (MeOH/DCM 10% v/v); 1H-NMR: δ 2.26 (4H, m, 2 × piperazine-CH2), 2.45 (2H, t, J = 6.6, COCH2), 3.01 (2H, q, J = 6.6, CH2NH), 3.37 (4H, m, overlap with water peak, 2 × piperazine-CH2), 4.43 (1H, s, CH), 7.22 (5H, m, Ar-H), 7.43 (4H, m, Ar-H), 7.60 (2H, s, exchangeable with D2O, NHSO2NH2), 7.65 (2H, m, Ar-H), 7.75 (1H, m, Ar-H), 8,03 (1H, m, exchangeable with D2O, SO2NHCH2), 8.81 (1H, s, exchangeable with D2O, NHSO2NH2); 13C-NMR: δ 32.3, 38.5, 45.3, 51.3, 72.6, 115.1, 115.4 (d, 2JC–F 21), 118.4, 123.0, 130.0, 130.3 (d, 3JC–F 8), 135.9, 138.2, 139.6, 161.1 (d, 1JC–F 242), 169.3; 19F-NMR: δ −115.6 (2F, s); m/z (ESI positive) 594.0 [M + H]+.
N-(3-(4-(bis(4-Fluorophenyl)methyl)piperazin-1-yl)-3-oxopropyl)-3(sulfamoylamino)benzenesulfonamide (6e). Compound 6e was obtained in 87% yield; m.p. 182–184 °C; TLC: Rf = 0.37 (MeOH/DCM 10% v/v); 1H-NMR: δ 2.26 (4H, m, 2 × piperazine-CH2), 2.46 (2H, t, J = 7.0, COCH2), 2.95 (2H, q, J = 7.0, CH2NH), 3.40 (4H, m, piperazine-CH2), 4.43 (1H, s, CH), 7.17 (5H, m, Ar-H), 7.29 (2H, s, exchangeable with D2O, NHSO2NH2), 7.38 (1H, m, exchangeable with D2O, SO2NHCH2), 7.47 (6H, m, Ar-H), 7.60 (1H, s, Ar-H), 9.96 (1H, s, exchangeable with D2O, NHSO2NH2); 13C-NMR: δ 33.3, 40.4 (overlap with DMSO peak), 44.7, 51.3, 73.5, 116.2 (d, 2JC–F 21), 117.0, 121.5, 122.7, 130.3 (d, 3JC–F 8), 130.8, 136.1, 139.2, 141.3, 162.0 (d, 1JC–F 242), 169.3; 19F-NMR: δ −115.6 (2F, s); m/z (ESI positive) 594.0 [M + H]+.
N-(3-(4-(bis(4-Fluorophenyl)methyl)piperazin-1-yl)-3-oxopropyl)-4-(sulfamoylamino)benzenesulfonamide (6f). Compound 6f was obtained in 35% yield; m.p. 149–152 °C; TLC Rf = 0.21 (MeOH/DCM 5% v/v); 1H-NMR: δ 2.26 (4H, m, 2 × piperazine-CH2), 2.45 (2H, t, J = 6.6, COCH2), 2.92 (2H, q, J = 6.6, CH2NH), 3.37 (4H, m, overlapped with water peak, 2 × piperazine-CH2), 4.43 (1H, s, CH), 7.16 (4H, m, Ar-H), 7.22 (2H, d, J = 8.8, Ar-H), 7.40 (2H, s, exchangeable with D2O, NHSO2NH2), 7.47 (5H, m, 4 × Ar-H, exchangeable with D2O, SO2NHCH2), 7.76 (2H, d, J = 8.8, Ar-H), 10.20 (1H, s, exchangeable with D2O, NHSO2NH2); 13C-NMR: δ 33.3, 40.4 (overlapped with DMSO peak), 44.7, 52.4, 73.6, 116.3, 116.6 (d, 2JC–F 21), 129.4, 130.1, 130.3 (d, 3JC–F 8), 139.3, 142.9, 162.0 (d, 1JC–F 242), 169.5; 19F-NMR: δ −115.6 (2F, s); m/z (ESI positive) 594.0 [M + H]+.
(1-((2-Sulfamoylaminophenyl)sulfonyl)piperidin-3-yl)(4-benzhydrylpiperazin-1-yl)methanone (6g). Compound 6g was obtained in 73% yield; m.p. 143–145 °C (dec.); TLC: Rf = 0.25 (MeOH/DCM 5% v/v); 1H-NMR: δ 1.56 (2H, m, piperidine-CH2), 1.76 (2H, m, piperidine-CH2), 2.40 (4H, m, 2 × piperazine-CH2), 2.84 (1H, m, COCH), 3.49 (4H, m, 2 × piperazine-CH2), 3.62 (4H, m, 2 × piperidine-CH2), 4.35 (1H, s, CH), 7.25 (3H, m, Ar-H), 7.34 (4H, m, Ar-H), 7.46 (4H, m, Ar-H), 7.71 (5H, m, overlapping signals, exchangeable with D2O, NHSO2NH2, 3 × Ar-H), 8.16 (1H, s, exchangeable with D2O, NHSO2NH2); 13C-NMR: δ 24.5, 27.7, 42.0, 45.8, 46.7, 48.9, 52.5, 75.6, 119.0, 123.2, 127.8, 128.5, 129.4, 129.5, 132.4, 135.5, 138.2, 143.4, 171.3; m/z (ESI positive) 598.0 [M + H]+.
4(1-((3-Sulfamoylaminophenyl)sulfonyl)piperidin-3-yl)(4-benzhydrylpiperazin-1-yl)methanone (6h). Compound 6h was obtained in 44% yield; m.p. 150–152 °C; TLC: Rf = 0.39 (ethyl acetate/n-hexane 70% v/v); 1H-NMR: δ 1.59 (2H, m, piperidine-CH2), 1.73 (2H, m, piperidine-CH2), 2.27 (6H, m, 2 × piperazine-CH2, piperidine-CH2), 2.82 (1H, m, COCH), 3.56 (6H, m, 2 × piperazine-CH2, piperidine-CH2), 4.36 (1H, s, CH), 7.23 (2H, m, Ar-H), 7.34 (6H, m, overlapping signals, exchangeable with D2O, NHSO2NH2, 4 × Ar-H), 7.48 (6H, m, Ar-H), 7.57 (2H, Ar-H), 10.58 (1H, s, exchangeable with D2O, NHSO2NH2); 13C-NMR: δ 24.6, 27.7, 42.0, 45.8, 47.0, 49.2, 52.5, 75.6, 117.1, 121.3, 122.7, 127.9, 128.5, 129.5, 130.9, 132.6, 137.2, 141.4, 171.5; m/z (ESI positive) 598.0 [M + H]+.
(1-((4-Sulfamoylaminophenyl)sulfonyl)piperidin-3-yl)(4-benzhydrylpiperazin-1-yl)methanone (6i). Compound 6i was obtained in 39% yield; m.p. 183–185 °C (dec.); silica gel TLC Rf = 0.27 (ethyl acetate/n-hexane 70% v/v); 1H-NMR: δ 1.60 (2H, m, piperidine-CH2), 1.73 (2H, m, piperidine-CH2), 2.35 (6H, m, 2 × piperazine-CH2, piperidine-CH2), 2.86 (1H, m, COCH), 3.62 (6H, m, 2 × piperazine-CH2, piperidine-CH2), 4.36 (1H, s, CH), 7.23 (2H, t, J = 7.4, Ar-H), 7.36 (6H, m, Ar-H), 7.49 (6H, m, overlapping signals, exchangeable with D2O, NHSO2NH2, 4 × Ar-H), 7.64 (2H, d, J = 8.4, Ar-H), 10.49 (1H, s, exchange with D2O, NHSO2NH2); 13C-NMR: δ 24.5, 27.7, 42.0, 45.7, 47.3, 49.2, 52.5, 75.6, 113.6, 127.8, 128.5, 129.4, 129.8, 130.3, 140.9, 142.2, 171.5; m/z (ESI positive) 598.0 [M + H]+.
(1-((2-Sulfamoylaminophenyl)sulfonyl)piperidin-3-yl)(4-(bis(4-fluorophenyl)methyl)piperazin-1-yl)methanone (6j). Compound 6j was obtained in 25% yield; m.p. 142–144 °C; TLC: Rf = 0.58 (MeOH/DCM 10% v/v); 1H-NMR: δ 1.56 (2H, m, piperidine-CH2), 1.73 (2H, m, piperidine-CH2), 2.24 (4H, m, 2 × piperazine-CH2), 2.37 (2H, m, piperidine-CH2), 2.80 (1H, m, COCH), 3.56 (6H, m, 2 × piperazine-CH2, piperidine-CH2), 4.41 (1H, s, CH), 7.15 (3H, t, J = 8.4, Ar-H), 7.23 (2H, m, Ar-H), 7.45 (4H, m, Ar-H), 7.69 (5H, m, overlapping signals, exchangeable with D2O, NHSO2NH2, 3 × Ar-H), 8.82 (1H, s, exchangeable with D2O, NHSO2NH2); 13C-NMR: δ 24.5, 27.6, 42.0, 44.6, 45.8, 48.9, 52.0, 73.6, 115.9, 116.3 (d, 2JC–F 21), 119.1, 123.1, 130.2, (d, 3JC–F 9), 130.8, 135.6, 138.2, 139.3, 162.1 (d, 1JC–F 242), 171.3; 19F-NMR: δ −115.6 (2F, s); m/z (ESI positive) 634 [M + H]+.
(1-((3-Sulfamoylaminophenyl)sulfonyl)piperidin-3-yl)(4-(bis(4-fluorophenyl)methyl)piperazin-1-yl)methanone (6k). Compound 6k was obtained in 44% yield; m.p. 162–164 °C (dec.); TLC: Rf = 0.48 (MeOH/DCM 10% v/v); 1H-NMR: δ 1.57 (2H, m, piperidine-CH2), 1.73 (2H, m, piperidine-CH2), 2.29 (6H, m, 2 × piperazine-CH2, piperidine-CH2), 2.82 (1H, m, COCH), 3.55 (4H, m, 2 × piperazine-CH2), 3.63 (2H, m, piperidine-CH2), 4.45 (1H, s, CH), 7.18 (4H, m, Ar-H), 7.33 (3H, m, overlapping signals, exchangeable with D2O, NHSO2NH2, Ar-H), 7.48 (6H, m, Ar-H), 7.57 (1H, m, Ar-H), 9.97 (1H, s, exchangeable with D2O, NHSO2NH2); 13C-NMR: δ 24.6, 27.7, 41.9, 45.7, 47.0, 49.2, 52.3, 73.5, 116.3 (d, 2JC–F 21), 118.7, 121.5, 122.7, 130.3 (d, 3JC–F 8), 130.6, 136.8, 138.3, 139.3, 162.0 (d, 1JC–F 242), 171.4; 19F-NMR: δ −115.6 (2F, s); m/z (ESI positive) 634.0 [M + H]+.
(1-((4-Sulfamoylaminophenyl)sulfonyl)piperidin-3-yl)(4-(bis(4-fluorophenyl)methyl)piperazin-1-yl)methanone (6l). Compound 6l was obtained in 73% yield; m.p. 160–162 °C (dec.); TLC: Rf = 0.60 (MeOH/DCM 10% v/v); 1H-NMR: δ 1.56 (2H, m, piperidine-CH2), 1.72 (2H, m, piperidine-CH2), 2.26 (6H, m, 2 × piperazine-CH2, piperidine-CH2), 2.83 (1H, m, COCH), 3.54 (4H, m, 2 × piperazine-CH2), 3.61 (2H, m, piperidine-CH2), 4.45 (1H, s, CH), 7.18 (4H, m, Ar-H), 7.34 (2H, d, J = 8.4, Ar-H), 7.48 (6H, m, overlapping signals, exchangeable with D2O, NHSO2NH2, 4 × Ar-H), 7.65 (2H, d, J = 8.4, Ar-H), 10.29 (1H, s, exchangeable with D2O, NHSO2NH2); 13C-NMR: δ 24.5, 27.8, 42.3, 45.7, 47.0, 49.2, 52.4, 73.5, 116.3 (d, 2JC–F 21), 117.5, 127.1, 129.6, 130.3 (d, 3JC–F 8), 139.2, 144.7, 162.0 (d, 1JC–F 242), 171.4; 19F-NMR: δ −115.6 (2F, s); m/z (ESI positive) 634.0 [M + H]+.