Total Flavonoids of Drynariae Rhizoma Prevent Bone Loss Induced by Hindlimb Unloading in Rats
Abstract
:1. Introduction
2. Results
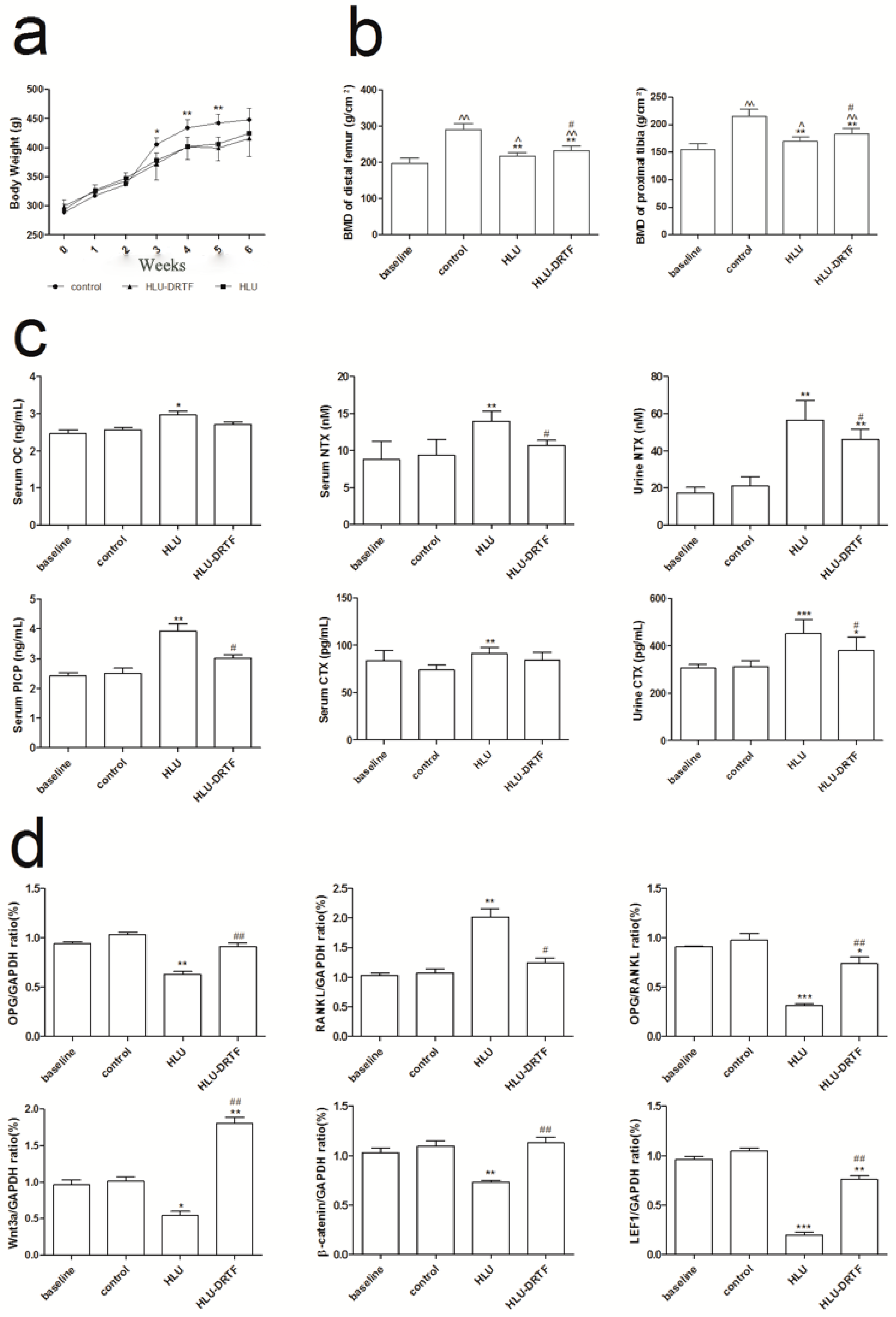
2.1. Body Weights
2.2. BMD Evaluation
2.3. Biochemistry Assay
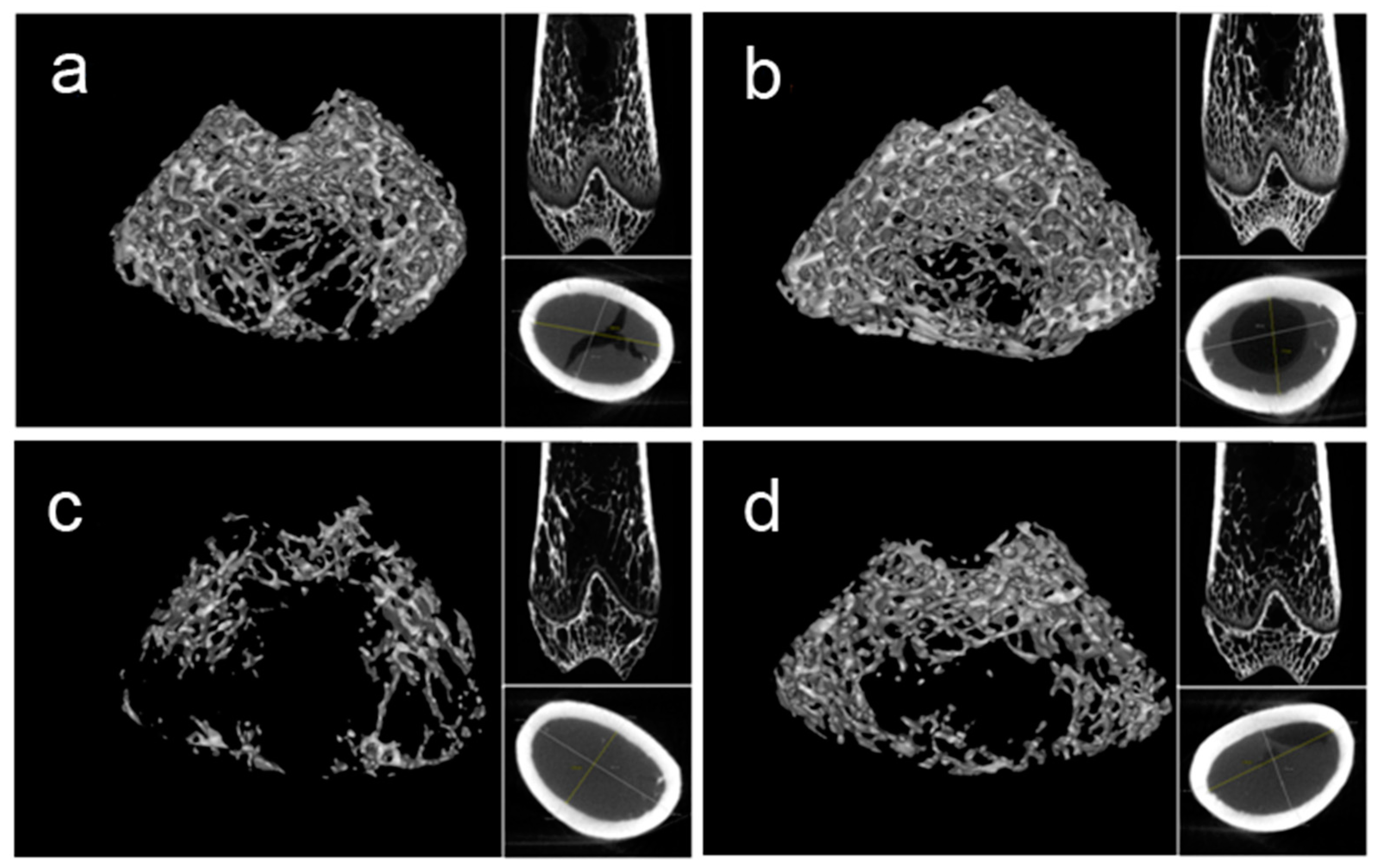
2.4. Bone Microarchitecture
2.5. Biomechanical Test
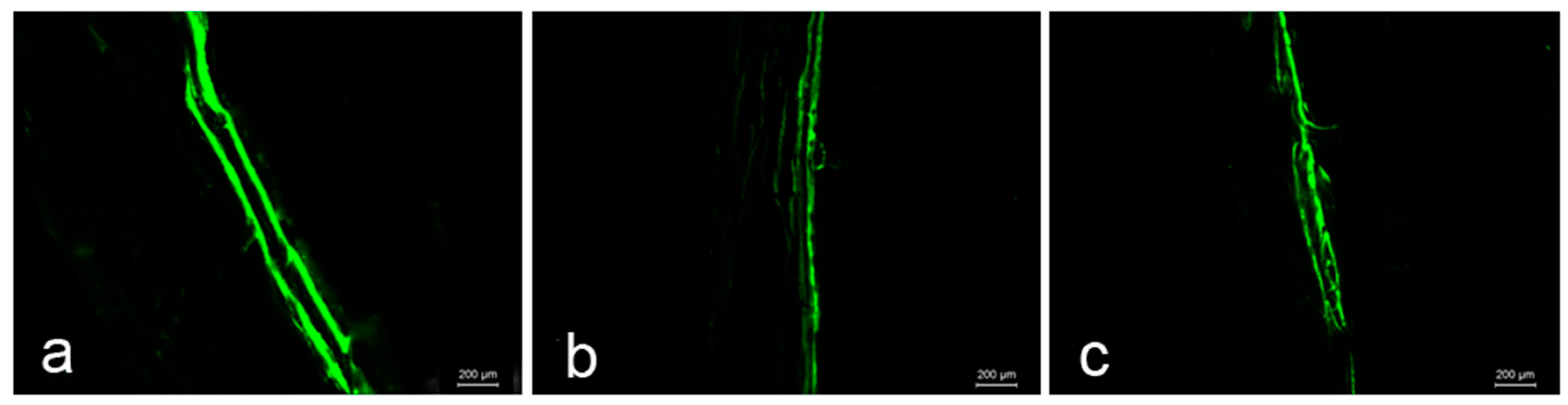
2.6. Bone Histomorphometry Analysis
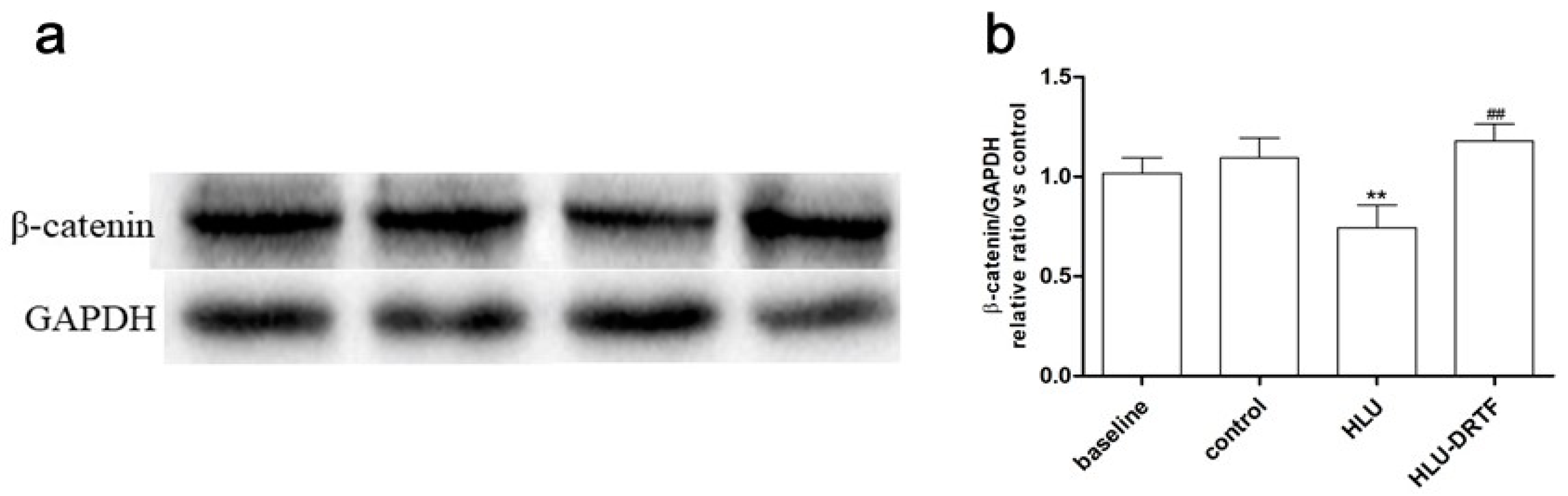
2.7. Results of OPG, RANKL Wnt3a, β-Catenin and LEF1 Gene Expression
2.8. Results of β-Catenin Protein Expression
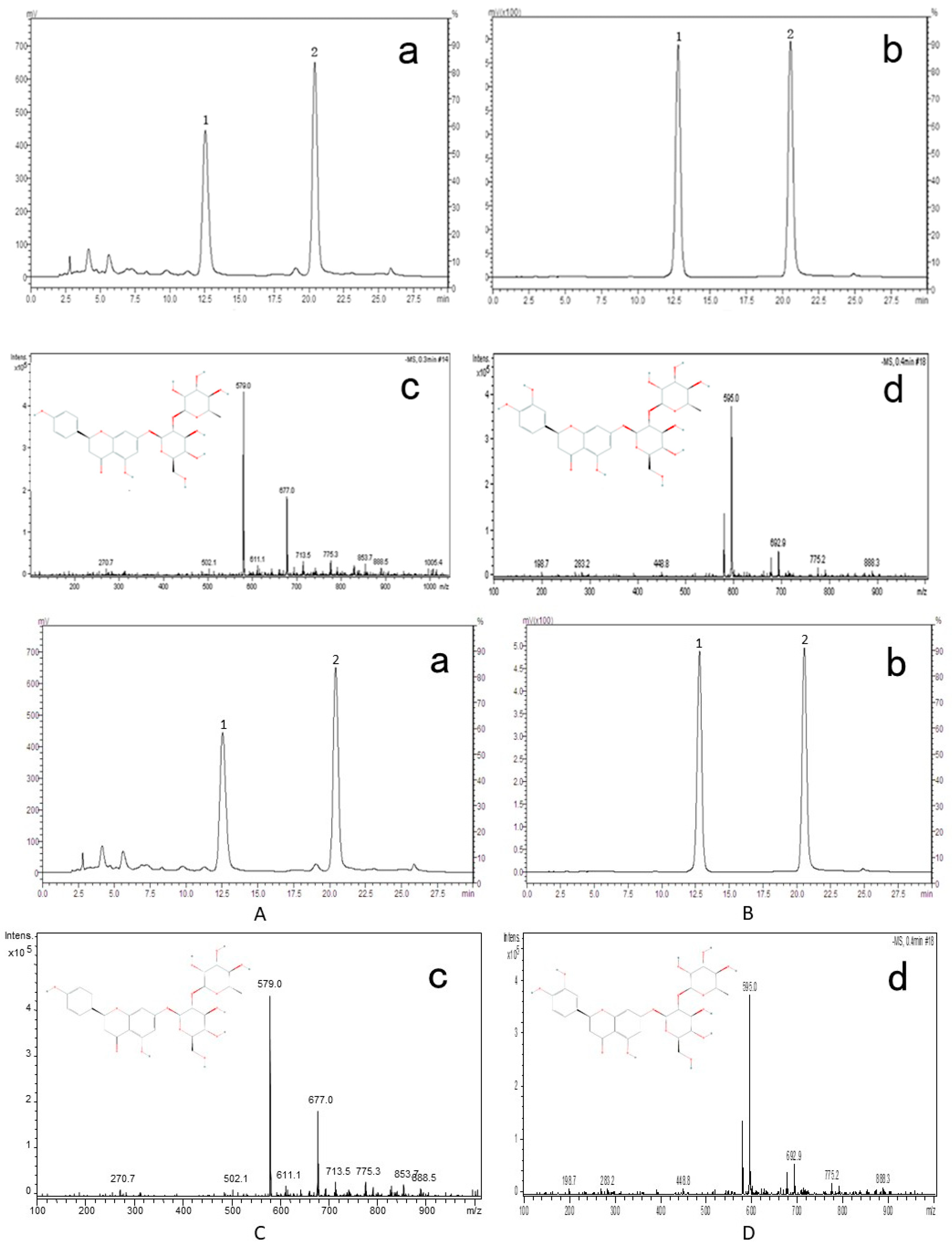
2.9. Identification of Active Compounds in DRTF
3. Discussion
4. Materials and Methods
4.1. Animals and Treatments
4.2. Bone Mineral Density (BMD) Analysis
4.3. Assay for Serum and Urine Chemistry
4.4. Microcomputed Tomography (MicroCT) Analysis
4.5. Biomechanical Test
4.6. Bone Histomorphometry
4.7. Real Time RT-PCR Analysis
4.8. Western Blotting
4.9. Identification of Active Phytochemicals in DRTF
4.10. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| d | day |
| DRTF | total flavonoids of Drynariae Rhizoma |
| OPG | osteocalcin |
| OVX | ovariectomy |
| RANKL | receptor activator of NF-κB ligand |
| BMD | bone mineral density |
| NTX-Ι | N-terminal cross-linked telopeptides of type I collagen |
| CTX-Ι | C-terminal cross-linked telopeptides of type I collagen |
| OC | osteocalcin |
| PICP | C-terminal propeptide of type I procollagen |
| BV/TV | bone volume/tissue volume |
| Tb. N | trabecular number |
| Tb. Th | trabecular thickness |
| Tb. Sp | trabecular separation |
| Cr. Th | cortical thickness |
| NIH | National Institutes of Health |
References
- Blaber, E.A.; Dvorochkin, N.; Lee, C.; Alwood, J.S.; Yousuf, R.; Pianetta, P.; Globus, R.K.; Burns, B.P.; Almeida, E.A. Microgravity induces pelvic bone loss through osteoclastic activity, osteocytic osteolysis, and osteoblastic cell cycle inhibition by cdkn1a/p21. PLoS ONE 2013, 8, e61372. [Google Scholar] [CrossRef] [PubMed]
- Fitts, R.H.; Riley, D.R.; Widrick, J.J. Functional and structural adaptations of skeletal muscle to microgravity. J. Exp. Biol. 2001, 204, 3201–3208. [Google Scholar] [PubMed]
- Chowdhury, B.; Seetharam, A.; Wang, Z.; Liu, Y.; Lossie, A.C.; Thimmapuram, J.; Irudayaraj, J. A study of alterations in DNA epigenetic modifications (5mc and 5hmc) and gene expression influenced by simulated microgravity in human lymphoblastoid cells. PLoS ONE 2016, 11, e0147514. [Google Scholar] [CrossRef] [PubMed]
- Paulsen, K.; Thiel, C.; Timm, J.; Schmidt, P.M.; Huber, K.; Tauber, S.; Hemmersbach, R.; Seibt, D.; Kroll, H.; Grote, K.-H.; et al. Microgravity-induced alterations in signal transduction in cells of the immune system. Acta Astronaut. 2010, 67, 1116–1125. [Google Scholar] [CrossRef]
- Nicolas, M.; Sandal, G.M.; Weiss, K.; Yusupova, A. Mars-105 study: Time-courses and relationships between coping, defense mechanisms, emotions and depression. J. Environ. Psychol. 2013, 35, 52–58. [Google Scholar] [CrossRef]
- Shackelford, L.C.; LeBlanc, A.D.; Driscoll, T.B.; Evans, H.J.; Rianon, N.J.; Smith, S.M.; Spector, E.; Feeback, D.L.; Lai, D. Resistance exercise as a countermeasure to disuse-induced bone loss. J. Appl. Physiol. (Bethesda, Md.: 1985) 2004, 97, 119–129. [Google Scholar] [CrossRef] [PubMed]
- Sibonga, J.D.; Evans, H.J.; Sung, H.G.; Spector, E.R.; Lang, T.F.; Oganov, V.S.; Bakulin, A.V.; Shackelford, L.C.; Leblanc, A.D. Recovery of spaceflight-induced bone loss: Bone mineral density after long-duration missions as fitted with an exponential function. Bone 2007, 41, 973–978. [Google Scholar] [CrossRef] [PubMed]
- Hargens, A.R.; Bhattacharya, R.; Schneider, S.M. Space physiology vi: Exercise, artificial gravity, and countermeasure development for prolonged space flight. Eur. J. Appl. Physiol. 2013, 113, 2183–2192. [Google Scholar] [CrossRef] [PubMed]
- Chowdhury, P.; Soulsby, M. Lipid peroxidation in rat brain is increased by simulated weightlessness and decreased by a soy-protein diet. Ann. Clin. Lab. Sci. 2002, 32, 188–192. [Google Scholar] [PubMed]
- Peng, S.; Zhang, G.; Zhang, B.T.; Guo, B.; He, Y.; Bakker, A.J.; Pan, X.; Zhen, W.; Hung, L.; Qin, L.; et al. The beneficial effect of icaritin on osteoporotic bone is dependent on the treatment initiation timing in adult ovariectomized rats. Bone 2013, 55, 230–240. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.K.; Li, J.; Liu, J.; Guo, B.; Leung, A.; Zhang, G.; Zhang, B.T. Icaritin requires phosphatidylinositol 3 kinase (pi3k)/akt signaling to counteract skeletal muscle atrophy following mechanical unloading. Sci. Rep. 2016, 6, 20300. [Google Scholar] [CrossRef] [PubMed]
- Durbin, S.M.; Jackson, J.R.; Ryan, M.J.; Gigliotti, J.C.; Alway, S.E.; Tou, J.C. Resveratrol supplementation influences bone properties in the tibia of hindlimb-suspended mature fisher 344 x brown norway male rats. Appl. Physiol. Nutr. Metab. 2012, 37, 1179–1188. [Google Scholar] [CrossRef] [PubMed]
- Habold, C.; Momken, I.; Ouadi, A.; Bekaert, V.; Brasse, D. Effect of prior treatment with resveratrol on density and structure of rat long bones under tail-suspension. J. Bone Miner. Metab. 2011, 29, 15–22. [Google Scholar] [CrossRef] [PubMed]
- Kong, X.H.; Niu, Y.B.; Song, X.M.; Zhao, D.D.; Wang, J.; Wu, X.L.; Zhang, R.; Mei, Q.B. Astragaloside II induces osteogenic activities of osteoblasts through the bone morphogenetic protein-2/mapk and smad1/5/8 pathways. Int. J. Mol. Med. 2012, 29, 1090–1098. [Google Scholar] [PubMed]
- Xu, Y.X.; Wu, C.L.; Wu, Y.; Tong, P.J.; Jin, H.T.; Yu, N.Z.; Xiao, L.W. Epimedium-derived flavonoids modulate the balance between osteogenic differentiation and adipogenic differentiation in bone marrow stromal cells of ovariectomized rats via wnt/beta-catenin signal pathway activation. Chin. J. Integr. Med. 2012, 18, 909–917. [Google Scholar] [CrossRef] [PubMed]
- Niu, Y.B.; Li, Y.H.; Kong, X.H.; Zhang, R.; Sun, Y.; Li, Q.; Li, C.; Liu, L.; Wang, J.; Mei, Q.B. The beneficial effect of radix dipsaci total saponins on bone metabolism in vitro and in vivo and the possible mechanisms of action. Osteoporos. Int. 2012, 23, 2649–2660. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; Liu, Z.G.; Li, C.; Hu, S.J.; Liu, L.; Wang, J.P.; Mei, Q.B. Du-zhong (Eucommia ulmoides oliv.) cortex extract prevent ovx-induced osteoporosis in rats. Bone 2009, 45, 553–559. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.Q. Ben Cao Shi Yi; Anhui Science and Technology Press: Anhui, China, 2004. [Google Scholar]
- Huang, Y.; Liu, X.; Zhao, L.; Li, F.; Xiong, Z. Kidney tissue targeted metabolic profiling of glucocorticoid-induced osteoporosis and the proposed therapeutic effects of rhizoma drynariae studied using uhplc/ms/ms. Biomed. Chromatogr. 2014, 28, 878–884. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.L.; Lei, L.H.; Ding, P.H.; Tang, Q.; Wu, Y.M. Osteogenic effect of drynariae rhizoma extracts and naringin on mc3t3-e1 cells and an induced rat alveolar bone resorption model. Arch. Oral Biol. 2011, 56, 1655–1662. [Google Scholar] [CrossRef] [PubMed]
- Jeong, J.C.; Kang, S.C.; Jeong, C.W.; Kim, H.M.; Lee, Y.C.; Chang, Y.C.; Kim, C.H. Inhibition of drynariae rhizoma extracts on bone resorption mediated by processing of cathepsin k in cultured mouse osteoclasts. Int. Immunopharmacol. 2003, 3, 1685–1697. [Google Scholar] [CrossRef] [PubMed]
- Wong, R.W.; Rabie, B.; Bendeus, M.; Hagg, U. The effects of rhizoma curculiginis and rhizoma drynariae extracts on bones. Chin. Med. 2007, 2, 13. [Google Scholar] [CrossRef] [PubMed]
- An, H.J.; Lee, G.G.; Lee, K.T. Drynariae rhizoma increases immune response in mice. Nat. Prod. Commun. 2012, 7, 905–908. [Google Scholar] [PubMed]
- Anuja, G.I.; Latha, P.G.; Suja, S.R.; Shyamal, S.; Shine, V.J.; Sini, S.; Pradeep, S.; Shikha, P.; Rajasekharan, S. Anti-inflammatory and analgesic properties of Drynaria quercifolia (L.) j. Smith. J. Ethnopharmacol. 2010, 132, 456–460. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Li, H.; Yu, J.; Hong, M.; Zhou, J.; Zhu, L.; Wang, Y.; Luo, M.; Xia, Z.; Yang, Z.J.; et al. Protective effects of chinese herbal medicine rhizoma drynariae in rats after traumatic brain injury and identification of active compound. Mol. Neurobiol. 2016, 53, 4809–4820. [Google Scholar] [CrossRef] [PubMed]
- Sung, Y.Y.; Kim, D.S.; Yang, W.K.; Nho, K.J.; Seo, H.S.; Kim, Y.S.; Kim, H.K. Inhibitory effects of drynaria fortunei extract on house dust mite antigen-induced atopic dermatitis in nc/nga mice. J. Ethnopharmacol. 2012, 144, 94–100. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.E.; Liu, H.C.; Lin, Y.L.; Liu, S.H.; Yang, R.S.; Chen, R.M. Drynaria fortunei j. Sm. Improves the bone mass of ovariectomized rats through osteocalcin-involved endochondral ossification. J. Ethnopharmacol. 2014, 158, 94–101. [Google Scholar] [CrossRef] [PubMed]
- Morey-Holton, E.R.; Globus, R.K. Hindlimb unloading of growing rats: A model for predicting skeletal changes during space flight. Bone 1998, 22, 83S–88S. [Google Scholar] [CrossRef]
- Vico, L.; Hinsenkamp, M.; Jones, D.; Marie, P.J.; Zallone, A.; Cancedda, R. Osteobiology, strain, and microgravity. Part ii: Studies at the tissue level. Calcif. Tissue Int. 2001, 68, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.-B.; Meng, F.-H.; Pan, X.-F.; Xiong, Z.-L.; Li, F.-M. Hplc determination of neoeriocitrin and naringin in rhizoma drynariae. Chin. J. Pharm. Anal. 2006, 26, 808–810. [Google Scholar]
- Wimalawansa, S.M.; Wimalawansa, S.J. Simulated weightlessness-induced attenuation of testosterone production may be responsible for bone loss. Endocrine 1999, 10, 253–260. [Google Scholar] [CrossRef] [PubMed]
- Felix, K.; Wise, K.; Manna, S.; Yamauchi, K.; Wilson, B.L.; Thomas, R.L.; Kulkarni, A.; Pellis, N.R.; Ramesh, G.T. Altered cytokine expression in tissues of mice subjected to simulated microgravity. Mol. Cell. Biochem. 2004, 266, 79–85. [Google Scholar] [CrossRef] [PubMed]
- Morey-Holton, E.; Globus, R.K.; Kaplansky, A.; Durnova, G. The hindlimb unloading rat model: Literature overview, technique update and comparison with space flight data. Adv. Space Biol. Med. 2005, 10, 7–40. [Google Scholar] [PubMed]
- Sonnenfeld, G.; Morey, E.R.; Williams, J.A.; Mandel, A.D. Effect of a simulated weightlessness model on the production of rat interferon. J. Interferon Res. 1982, 2, 467–470. [Google Scholar] [CrossRef] [PubMed]
- Siu, W.S.; Wong, H.L.; Lau, C.P.; Shum, W.T.; Wong, C.W.; Gao, S.; Fung, K.P.; Lau, C.B.; Hung, L.K.; Ko, C.H.; et al. The effects of an antiosteoporosis herbal formula containing epimedii herba, ligustri lucidi fructus and psoraleae fructus on density and structure of rat long bones under tail-suspension, and its mechanisms of action. Phytother. Res. 2013, 27, 484–492. [Google Scholar] [CrossRef] [PubMed]
- Bloomfield, S.A.; Allen, M.R.; Hogan, H.A.; Delp, M.D. Site- and compartment-specific changes in bone with hindlimb unloading in mature adult rats. Bone 2002, 31, 149–157. [Google Scholar] [CrossRef]
- Oganov, V.S. Modern analysis of bone loss mechanisms in microgravity. J. Gravitat. Physiol. 2004, 11, 143–146. [Google Scholar]
- Smith, B.J.; Lucas, E.A.; Turner, R.T.; Evans, G.L.; Lerner, M.R.; Brackett, D.J.; Stoecker, B.J.; Arjmandi, B.H. Vitamin e provides protection for bone in mature hindlimb unloaded male rats. Calcif. Tissue Int. 2005, 76, 272–279. [Google Scholar] [CrossRef] [PubMed]
- Aaron, J.E.; Shore, P.A.; Itoda, M.; Morrison, R.J.; Hartopp, A.; Hensor, E.M.; Hordon, L.D. Mapping trabecular disconnection “hotspots” in aged human spine and hip. Bone 2015, 78, 71–80. [Google Scholar] [CrossRef] [PubMed]
- Devareddy, L.; Khalil, D.A.; Smith, B.J.; Lucas, E.A.; Soung do, Y.; Marlow, D.D.; Arjmandi, B.H. Soy moderately improves microstructural properties without affecting bone mass in an ovariectomized rat model of osteoporosis. Bone 2006, 38, 686–693. [Google Scholar] [CrossRef] [PubMed]
- Laib, A.; Kumer, J.L.; Majumdar, S.; Lane, N.E. The temporal changes of trabecular architecture in ovariectomized rats assessed by microct. Osteoporos. Int. 2001, 12, 936–941. [Google Scholar] [CrossRef] [PubMed]
- Turner, C.H.; Burr, D.B. Basic biomechanical measurements of bone: A tutorial. Bone 1993, 14, 595–608. [Google Scholar] [CrossRef]
- Zhang, Z.; Song, C.; Fu, X.; Liu, M.; Li, Y.; Pan, J.; Liu, H.; Wang, S.; Xiang, L.; Xiao, G.G.; et al. High-dose diosgenin reduces bone loss in ovariectomized rats via attenuation of the rankl/opg ratio. Int. J. Mol. Sci. 2014, 15, 17130–17147. [Google Scholar] [CrossRef] [PubMed]
- Bord, S.; Ireland, D.C.; Beavan, S.R.; Compston, J.E. The effects of estrogen on osteoprotegerin, rankl, and estrogen receptor expression in human osteoblasts. Bone 2003, 32, 136–141. [Google Scholar] [CrossRef]
- Vaananen, H.K.; Laitala-Leinonen, T. Osteoclast lineage and function. Arch. Biochem. Biophys. 2008, 473, 132–138. [Google Scholar] [CrossRef] [PubMed]
- Boyce, B.F.; Xing, L. Functions of rankl/rank/opg in bone modeling and remodeling. Arch. Biochem. Biophys. 2008, 473, 139–146. [Google Scholar] [CrossRef] [PubMed]
- Kramer, I.; Halleux, C.; Keller, H.; Pegurri, M.; Gooi, J.H.; Weber, P.B.; Feng, J.Q.; Bonewald, L.F.; Kneissel, M. Osteocyte wnt/beta-catenin signaling is required for normal bone homeostasis. Mol. Cell. Biol. 2010, 30, 3071–3085. [Google Scholar] [CrossRef] [PubMed]
- Lara-Castillo, N.; Kim-Weroha, N.A.; Kamel, M.A.; Javaheri, B.; Ellies, D.L.; Krumlauf, R.E.; Thiagarajan, G.; Johnson, M.L. In vivo mechanical loading rapidly activates beta-catenin signaling in osteocytes through a prostaglandin mediated mechanism. Bone 2015, 76, 58–66. [Google Scholar] [CrossRef] [PubMed]
- Zahoor, M.; Cha, P.H.; Min do, S.; Choi, K.Y. Indirubin-3′-oxime reverses bone loss in ovariectomized and hindlimb-unloaded mice via activation of the wnt/beta-catenin signaling. J. Bone Miner. Res. 2014, 29, 1196–1205. [Google Scholar] [CrossRef] [PubMed]
- Bonewald, L.F.; Johnson, M.L. Osteocytes, mechanosensing and wnt signaling. Bone 2008, 42, 606–615. [Google Scholar] [CrossRef] [PubMed]
- Robinson, J.A.; Chatterjee-Kishore, M.; Yaworsky, P.J.; Cullen, D.M.; Zhao, W.; Li, C.; Kharode, Y.; Sauter, L.; Babij, P.; Brown, E.L.; et al. Wnt/beta-catenin signaling is a normal physiological response to mechanical loading in bone. J. Biol. Chem. 2006, 281, 31720–31728. [Google Scholar] [CrossRef] [PubMed]
- Krishnan, V.; Bryant, H.U.; Macdougald, O.A. Regulation of bone mass by wnt signaling. J. Clin. Investig. 2006, 116, 1202–1209. [Google Scholar] [CrossRef] [PubMed]
- Shang, Z.P.; Zhao, Q.C.; Tan, J.J.; Yang, L.; Yan, M.; Shi, G.B. Chemical constituents from rhizomes of drynaria fortunei. Pract. Pharm. Clin. Rem. 2010, 13, 262–272. [Google Scholar]
- Gao, Y.; Wang, X.L.; Wang, N.L.; Yao, X.S. Chemical constituents from drynaria fortunei. Chin. J. Med. Chem. 2008, 8, 284–287. [Google Scholar]
- National Pharmacopoeia Committee. Pharmacopoeia of the People’s Republic of China; People’s Medical Publishing House: Beijing, China, 2015; p. 239. [Google Scholar]
- Pang, W.Y.; Wang, X.L.; Mok, S.K.; Lai, W.P.; Chow, H.K.; Leung, P.C.; Yao, X.S.; Wong, M.S. Naringin improves bone properties in ovariectomized mice and exerts oestrogen-like activities in rat osteoblast-like (umr-106) cells. Br. J. Pharmacol. 2010, 159, 1693–1703. [Google Scholar] [CrossRef] [PubMed]
- Wong, K.C.; Pang, W.Y.; Wang, X.L.; Mok, S.K.; Lai, W.P.; Chow, H.K.; Leung, P.C.; Yao, X.S.; Wong, M.S. Drynaria fortunei-derived total flavonoid fraction and isolated compounds exert oestrogen-like protective effects in bone. Br. J. Nutr. 2013, 110, 475–485. [Google Scholar] [CrossRef] [PubMed]
- Xu, T.; Wang, L.; Tao, Y.; Ji, Y.; Deng, F.; Wu, X.H. The function of naringin in inducing secretion of osteoprotegerin and inhibiting formation of osteoclasts. Evid. Based Complement. Altern. Med. 2016, 2016, 8981650. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Zeng, Z.; Cai, G. Comparison of neoeriocitrin and naringin on proliferation and osteogenic differentiation in mc3t3-e1. Phytomedicine 2011, 18, 985–989. [Google Scholar] [CrossRef] [PubMed]
- Pastoureau, P.; Chomel, A.; Bonnet, J. Specific evaluation of localized bone mass and bone loss in the rat using dual-energy X-ray absorptiometry subregional analysis. Osteoporos. Int. 1995, 5, 143–149. [Google Scholar] [CrossRef] [PubMed]
- Delmas, P.D.; Eastell, R.; Garnero, P.; Seibel, M.J.; Stepan, J. The use of biochemical markers of bone turnover in osteoporosis. Committee of scientific advisors of the international osteoporosis foundation. Osteoporos. Int. 2000, 11, S2–S17. [Google Scholar] [CrossRef] [PubMed]
- Laib, A.; Barou, O.; Vico, L.; Lafage-Proust, M.H.; Alexandre, C.; Rugsegger, P. 3d micro-computed tomography of trabecular and cortical bone architecture with application to a rat model of immobilisation osteoporosis. Med. Biol. Eng. Compt. 2000, 38, 326–332. [Google Scholar] [CrossRef]
- Marinozzi, F.; Bini, F.; Marinozzi, A.; Zuppante, F.; De Paolis, A.; Pecci, R.; Bedini, R. Technique for bone volume measurement from human femur head samples by classification of micro-ct image histograms. Annali dell’Istituto Superiore di Sanita 2013, 49, 300–305. [Google Scholar] [PubMed]
- Ederveen, A.G.; Spanjers, C.P.; Quaijtaal, J.H.; Kloosterboer, H.J. Effect of 16 months of treatment with tibolone on bone mass, turnover, and biomechanical quality in mature ovariectomized rats. J. Bone Miner. Res. 2001, 16, 1674–1681. [Google Scholar] [CrossRef] [PubMed]
- Dempster, D.W.; Compston, J.E.; Drezner, M.K.; Glorieux, F.H.; Kanis, J.A.; Malluche, H.; Meunier, P.J.; Ott, S.M.; Recker, R.R.; Parfitt, A.M. Standardized nomenclature, symbols, and units for bone histomorphometry: A 2012 update of the report of the asbmr histomorphometry nomenclature committee. J. Bone Miner. Res. 2013, 28, 2–17. [Google Scholar] [CrossRef] [PubMed]
- Lau, W.S.; Chan, R.Y.; Guo, D.A.; Wong, M.S. Ginsenoside rg1 exerts estrogen-like activities via ligand-independent activation of eralpha pathway. J. Steroid Biochem. Mol. Biol. 2008, 108, 64–71. [Google Scholar] [CrossRef] [PubMed]
- Chang, S.; Chen, W.; Yang, J. Another formula for calculating the gene change rate in real-time rt-pcr. Mol. Biol. Rep. 2009, 36, 2165–2168. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are available from the authors. |
| Parameters | Baseline | Control | HLU | HLU–DRTF |
|---|---|---|---|---|
| BV/TV (%) | 0.288 ± 0.025 | 0.316 ± 0.010 | 0.183 ± 0.016 ** | 0.252 ± 0.007 * # |
| Tb. Th (mm) | 0.060 ± 0.004 | 0.072 ± 0.002 ^ | 0.056 ± 0.006 ** | 0.068 ± 0.003 # |
| Tb. N (1/mm) | 4.243 ± 0.579 | 4.950 ± 0.250 | 1.719 ± 0.439 ** | 2.817 ± 0.334 ** # |
| Tb. Sp (mm) | 0.146 ± 0.024 | 0.128 ± 0.011 | 0.373 ± 0.084 ** | 0.252 ± 0.023 * # |
| Cr. Th (mm) | 0.447 ± 0.021 | 0.569 ± 0.028 ^ | 0.496 ± 0.054 | 0.508 ± 0.032 |
| Parameters | Baseline | Control | HLU | HLU–DRTF | |
|---|---|---|---|---|---|
| Femur | Max-Stress (Mpa) | 74.05 ± 13.25 | 98.86 ± 9.08 ^ | 70.12 ± 7.31 ** | 90.96 ± 9.49 # |
| Young’s modulus (Mpa) | 1673.24 ± 120.9 | 1891.80 ± 179.25 | 1593.82 ± 103.16 * | 1911.27 ± 67.80 # | |
| Max-Load (N) | 105.38 ± 10.73 | 145.92 ± 17.08 ^^ | 107.61 ± 7.05 ** | 128.42 ± 7.16 # | |
| Stiffness (N/mm) | 111.14 ± 15.23 | 168.37 ± 11.32 ^^ | 125.94 ± 11.12 ** | 159.89 ± 15.54 ^^ # | |
| Energy (N.mm) | 77.15 ± 13.15 | 112.42 ± 19.32 ^^ | 61.69 ± 3.04 *** | 78.08 ± 6.85 ** | |
| Tibia | Max-Stress (Mpa) | 98.38 ± 7.59 | 111.92 ± 6.67 ^ | 85.89 ± 4.56 ** | 98.95 ± 4.43 * # |
| Young’s modulus (Mpa) | 1548.24 ± 54.61 | 1791.80 ± 97.91 ^ | 1418.82 ± 87.75 ** | 1736.27 ± 173.61 ## | |
| Max-Load (N) | 55.03 ± 5.95 | 72.45 ± 8.27 ^^ | 54.85 ± 3.24 ** | 69.43 ± 2.78 # | |
| Stiffness (N/mm) | 77.50 ± 10.12 | 97.21 ± 14.20 ^ | 72.99 ± 4.44 * | 88.48 ± 3.43 # | |
| Energy (N.mm) | 61.32 ± 3.24 | 78.55 ± 7.49 ^^ | 61.22 ± 3.78 ** | 66.82 ± 2.59 * |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Song, S.; Gao, Z.; Lei, X.; Niu, Y.; Zhang, Y.; Li, C.; Lu, Y.; Wang, Z.; Shang, P. Total Flavonoids of Drynariae Rhizoma Prevent Bone Loss Induced by Hindlimb Unloading in Rats. Molecules 2017, 22, 1033. https://doi.org/10.3390/molecules22071033
Song S, Gao Z, Lei X, Niu Y, Zhang Y, Li C, Lu Y, Wang Z, Shang P. Total Flavonoids of Drynariae Rhizoma Prevent Bone Loss Induced by Hindlimb Unloading in Rats. Molecules. 2017; 22(7):1033. https://doi.org/10.3390/molecules22071033
Chicago/Turabian StyleSong, Shuanghong, Ziyang Gao, Xujun Lei, Yinbo Niu, Yuan Zhang, Cuiqin Li, Yi Lu, Zhezhi Wang, and Peng Shang. 2017. "Total Flavonoids of Drynariae Rhizoma Prevent Bone Loss Induced by Hindlimb Unloading in Rats" Molecules 22, no. 7: 1033. https://doi.org/10.3390/molecules22071033
APA StyleSong, S., Gao, Z., Lei, X., Niu, Y., Zhang, Y., Li, C., Lu, Y., Wang, Z., & Shang, P. (2017). Total Flavonoids of Drynariae Rhizoma Prevent Bone Loss Induced by Hindlimb Unloading in Rats. Molecules, 22(7), 1033. https://doi.org/10.3390/molecules22071033