Five Unprecedented Secondary Metabolites from the Spider Parasitic Fungus Akanthomyces novoguineensis
Abstract
:1. Introduction
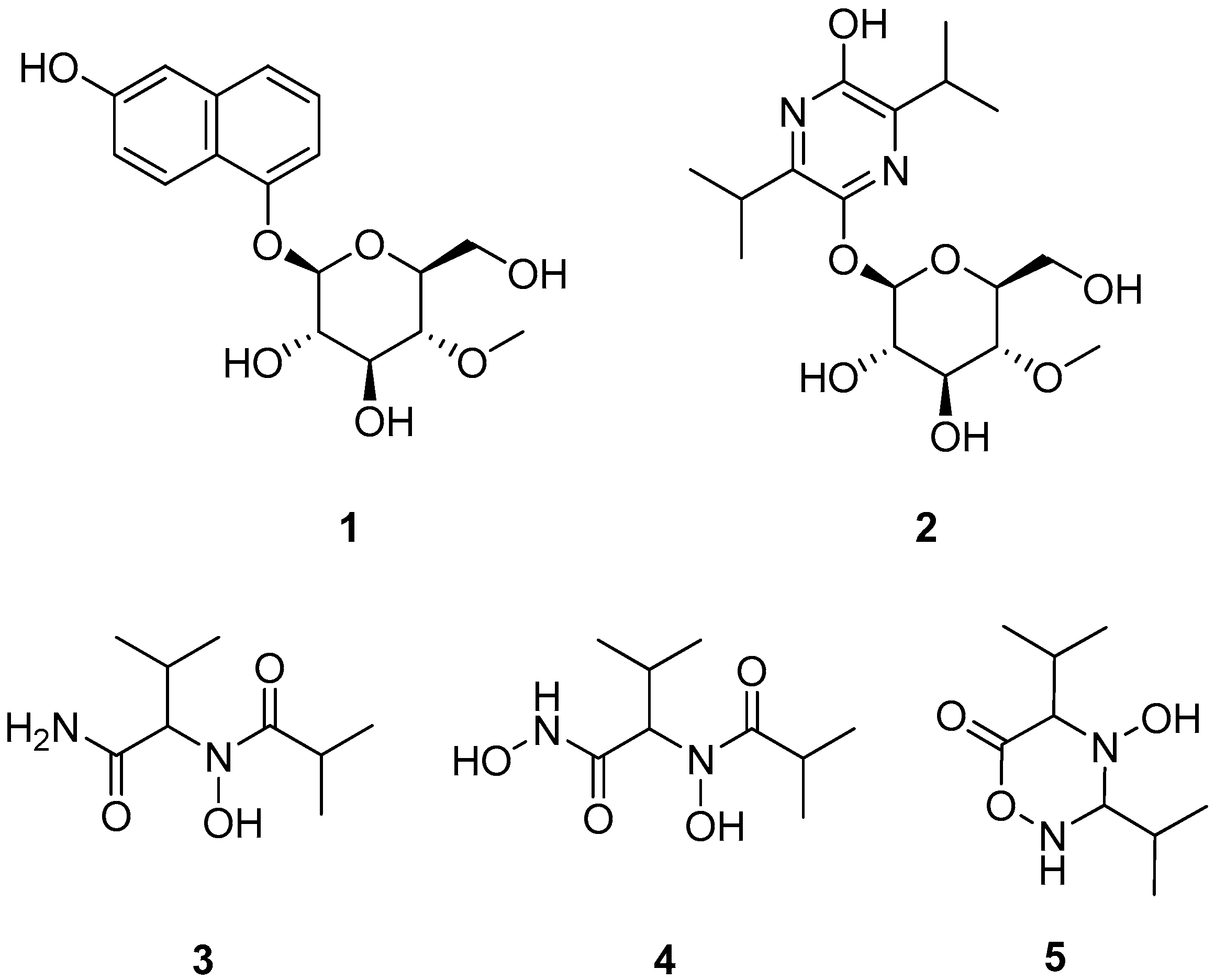
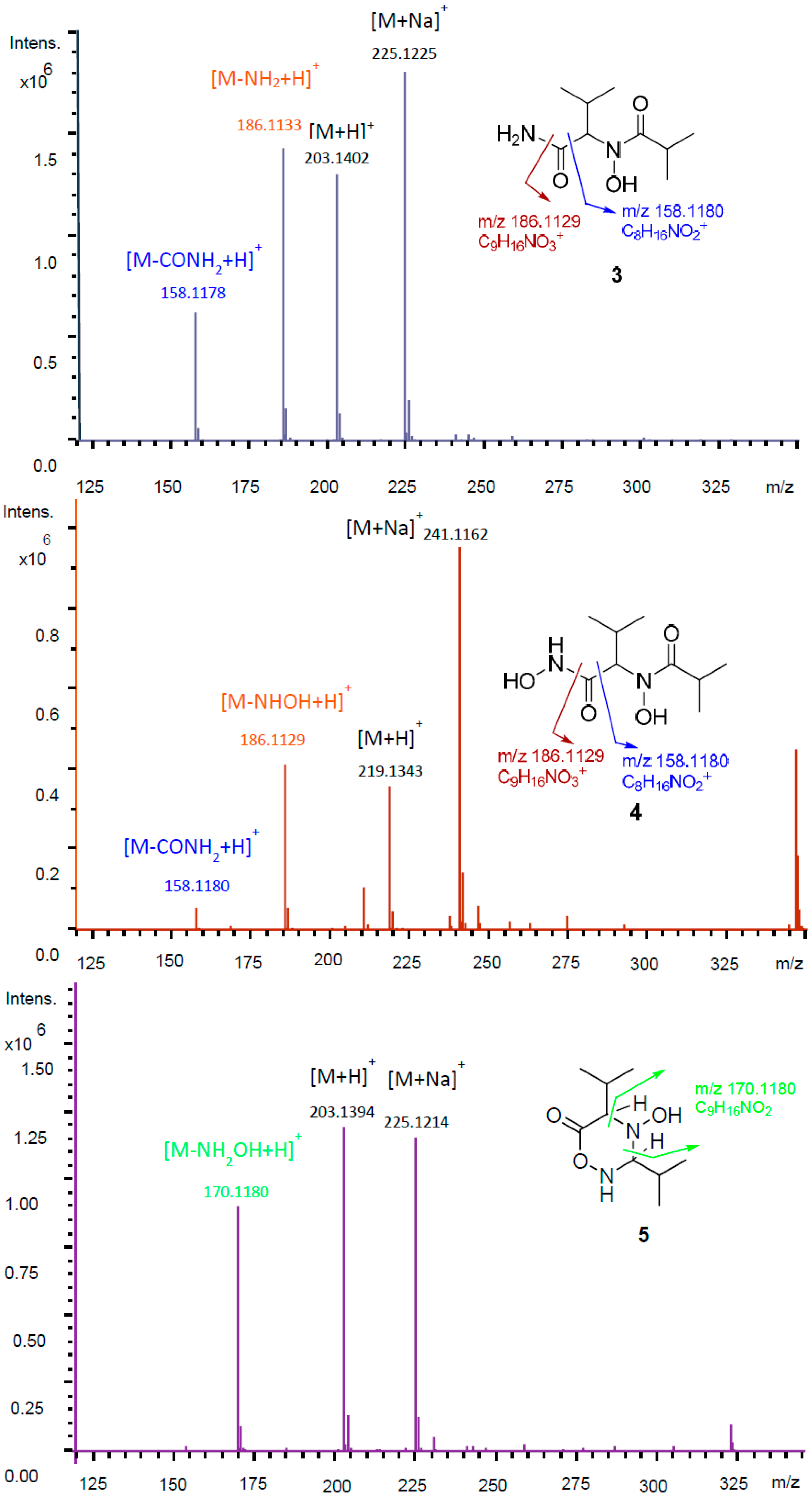
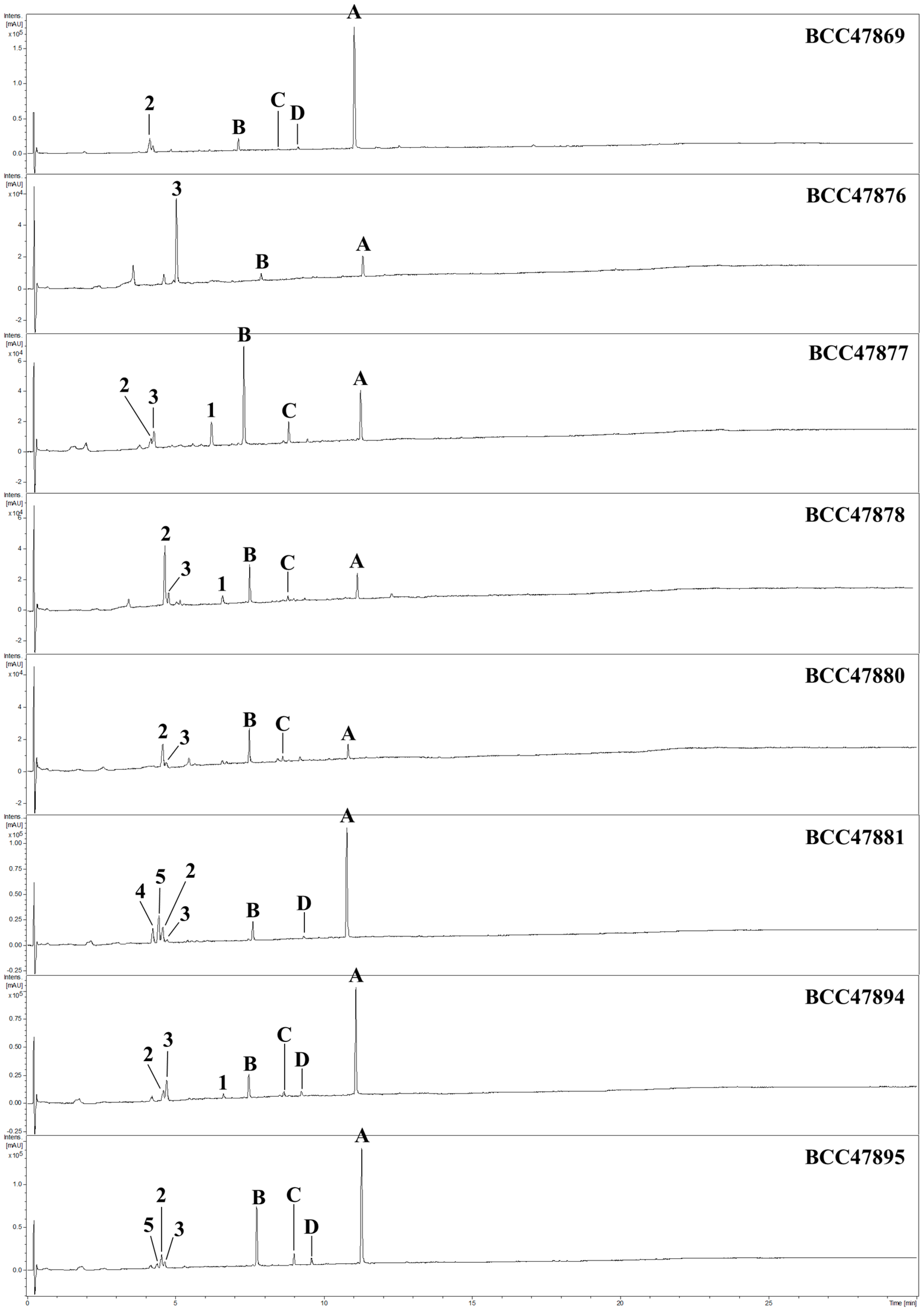
2. Results and Discussion
3. Materials and Methods
3.1. General
3.2. Fungal Material
3.3. Fermentation and Extraction
3.4. Isolation of Compounds 1–5
3.5. Acid Hydrolysis of Akanthol (1)
3.6. Biological Activities
3.6.1. Antimicrobial Activity and Cytotoxicity Assay
3.6.2. Anti-Biofilm Activity Assay
3.6.3. Nematicidal Activity Assay
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Isaka, M.; Kittakoop, P.; Kirtikara, K.; Hywel-Jones, N.L.; Thebtaranonth, Y. Bioactive substances from insect pathogenic fungi. Acc. Chem. Res. 2005, 38, 813–823. [Google Scholar] [CrossRef] [PubMed]
- Molnár, I.; Gibson, D.M.; Krasnoff, S.B. Secondary metabolites from entomopathogenic Hypocrealean fungi. Nat. Prod. Rep. 2010, 27, 1241–1275. [Google Scholar] [CrossRef] [PubMed]
- Hajek, A.E.; Delalibera, I. Fungal pathogens as classical biological control agents against arthropods. BioControl 2010, 55, 147–158. [Google Scholar] [CrossRef]
- Kuephadungphan, W.; Helaly, S.E.; Daengrot, C.; Phongpaichit, S.; Luangsa-ard, J.J.; Rukachaisirikul, V.; Stadler, M. Akanthopyrones A–D, α-pyrones bearing a 4-O-methyl-β-d-glucopyranose moiety from the spider-associated ascomycete Akanthomyces novoguineensis. Concurrent manusript, under review.
- Noumeur, S.R.; Helaly, S.E.; Jansen, R.; Gereke, M.; Stradal, T.E.B.; Harzallah, D.; Stadler, M. Preussilides A–F, Bicyclic polyketides from the endophytic fungus Preussia similis with antiproliferative activity. J. Nat. Prod. 2017, 80, 1531–1540. [Google Scholar] [CrossRef] [PubMed]
- Smith, F. The constitution of mesquite gum. Part III. The structure of the monomethyl glucuronic acid component. J. Chem. Soc. 1951, 2646–2652. [Google Scholar] [CrossRef]
- ACD/Structure Elucidator, version 16.02; Advanced Chemistry Development, Inc.: Toronto, ON, Canada, 2016.
- Pal, D.; Saha, S. Hydroxamic acid—A novel molecule for anticancer therapy. J. Adv. Pharm. Technol. Res. 2012, 3, 92–99. [Google Scholar] [CrossRef] [PubMed]
- Goel, R.; Luxami, V.; Paul, K. Synthesis, in vitro anticancer activity and SAR studies of arylated imidazo[1,2-a]pyrazine-coumarin hybrids. RSC Adv. 2015, 5, 37887–37895. [Google Scholar] [CrossRef]
- Chlupacova, M.K.; Kunes, J.; Buchta, V.; Vejsova, M.; Opletalova, V. Novel pyrazine analogs of chalcones: Synthesis and evaluation of their antifungal and antimycobacterial activity. Molecules 2015, 20, 1104–1117. [Google Scholar] [CrossRef] [PubMed]
- Panda, S.S.; Detistov, O.S.; Girgis, A.S.; Mohapatra, P.P.; Samir, A.; Katritzky, A.R. Synthesis and molecular modeling of antimicrobial active fluoroquinolone-pyrazine conjugates with amino acid linkers. Bioorg. Med. Chem. Lett. 2016, 26, 2198–2205. [Google Scholar] [CrossRef] [PubMed]
- Sebastian, S.H.R.; Al-Alshaikh, M.A.; El-Emam, A.A.; Panicker, C.Y.; Zitko, J.; Dolezal, M.; Vanalsenoy, C. Spectroscopic, quantum chemical studies, Fukui functions, in vitro antiviral activity and molecular docking of 5-chloro-N-(3-nitrophenyl)pyrazine-2-carboxamide. J. Mol. Struct. 2016, 1119, 188–199. [Google Scholar] [CrossRef]
- Rokade, Y.B.; Sayyed, R.Z. Naphthalene derivatives: A new range of antimicrobials with high therapeutic value. Rasayan J. Chem. 2009, 2, 972–980. [Google Scholar]
- Rokade, Y.; Dongare, N. Synthesis and antimicrobial activity of some azetidinone derivatives with the β-naphthol. Rasayan J. Chem. 2010, 3, 641–645. [Google Scholar]
- Isaka, M.; Palasarn, S.; Kocharin, K.; Hywel-Jones, N.L. Comparison of the bioactive secondary metabolites from the scale insect pathogens, anamorph Paecilomyces cinnamomeus, and teleomorph Torrubiella luteorostrata. J. Antibiot. (Tokyo) 2007, 60, 577–581. [Google Scholar] [CrossRef] [PubMed]
- Isaka, M.; Palasarn, S.; Supothina, S.; Komwijit, S.; Luangsa-Ard, J.J. Bioactive compounds from the scale insect pathogenic fungus Conoideocrella tenuis BCC 18627. J. Nat. Prod. 2011, 74, 782–789. [Google Scholar] [CrossRef] [PubMed]
- Kikuchi, H.; Takahashi, N.; Oshima, Y. Novel aromatics bearing 4-O-methylglucose unit isolated from the oriental crude drug Bombyx batryticatus. Tetrahedron Lett. 2004, 45, 367–370. [Google Scholar] [CrossRef]
- Kornsakulkarn, J.; Thongpanchang, C.; Lapanun, S.; Srichomthong, K. Isocoumarin glucosides from the scale insect fungus Torrubiella tenuis BCC 12732. J. Nat. Prod. 2009, 72, 1341–1343. [Google Scholar] [CrossRef] [PubMed]
- Kornsakulkarn, J.; Saepua, S.; Srichomthong, K.; Supothina, S.; Thongpanchang, C. New mycotoxins from the scale insect fungus Aschersonia coffeae Henn. BCC 28712. Tetrahedron 2012, 68, 8480–8486. [Google Scholar] [CrossRef]
- Kornsakulkarn, J.; Saepua, S.; Laksanacharoen, P.; Rachtawee, P.; Thongpanchang, C. Xanthone and anthraquinone-type mycotoxins from the scale insect fungus Aschersonia marginata BCC 28721. Tetrahedron Lett. 2013, 54, 3813–3815. [Google Scholar] [CrossRef]
- Saepua, S.; Kornsakulkarn, J.; Choowong, W.; Supothina, S.; Thongpanchang, C. Bioxanthracenes and monomeric analogues from insect pathogenic fungus Conoideocrella luteorostrata Zimm. BCC 31648. Tetrahedron 2015, 71, 2400–2408. [Google Scholar] [CrossRef]
- Helaly, S.E.; Richter, C.; Thongbai, B.; Hyde, K.D.; Stadler, M. Lentinulactam, a hirsutane sesquiterpene with an unprecedented lactam modification. Tetrahedron Lett. 2016, 57, 5911–5913. [Google Scholar] [CrossRef]
- Luangsa-ard, J.J.; Mongkolsamrit, S.; Thanakitpipattana, D.; Khonsanit, A.; Tasanathai, K.; Noisripoom, W.; Humber, R.A. Clavicipitaceous entomopathogens: New species in Metarhizium and a new genus Nigelia. Mycol. Prog. 2017, 16, 369–391. [Google Scholar] [CrossRef]
- Phainuphong, P.; Rukachaisirikul, V.; Saithong, S.; Phongpaichit, S.; Bowornwiriyapan, K.; Muanprasat, C.; Srimaroeng, C.; Duangjai, A.; Sakayaroj, J. Lovastatin analogues from the soil-derived fungus Aspergillus sclerotiorum PSU-RSPG178. J. Nat. Prod. 2016, 79, 1500–1507. [Google Scholar] [CrossRef] [PubMed]
- Richter, C.; Helaly, S.E.; Thongbai, B.; Hyde, K.D.; Stadler, M. Pyristriatins A and B: Pyridino-cyathane antibiotics from the Basidiomycete Cyathus cf. striatus. J. Nat. Prod. 2016, 79, 1684–1688. [Google Scholar] [CrossRef] [PubMed]
- Rahme, L.G.; Stevens, E.J.; Wolfort, S.F.; Shao, J.; Tompkins, R.G.; Ausubel, F.M. Common virulence factors for bacterial pathogenicity in plants and animals. Science 1995, 268, 1899–1902. [Google Scholar] [CrossRef] [PubMed]
- O’Toole, G.A. Microtiter dish biofilm formation assay. J. Vis. Exp. 2011, 47, 2437. [Google Scholar] [CrossRef] [PubMed]
- Stadler, M.; Anke, H.; Arendholz, W.-R.; Hansske, F.; Anders, U.; Sterner, O.; Bergquist, K.-E. Lachnumon and lachnumol A, new metabolites with nematicidal and antimicrobial activities from the Ascomycete Lachnum papyraceum (Karst.) Karst. J. Antibiot. (Tokyo) 1993, 46, 961–967. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds 1–5 are available from the authors. |

| Pos | 1 | Pos | 2 | ||
|---|---|---|---|---|---|
| δH (J in Hz) | δC, Type | δH (J in Hz) | δC, Type | ||
| 1 | 7.35, s | 118.8, CH | 2 | - | 154.9, C |
| 2 | - | 153.5, C | 3 | - | 139.5, C |
| 3 | 6.81, dd, 3.4, 5.2 | 110.6, CH | 5 | - | 147.6, C |
| 4 | 7.35, dd, 6.5, 3.4 | 127.4, CH | 6 | - | 144.7, C |
| 4a | - | 115.1, C | 7 | 3.24, m | 30.6, CH |
| 5 | - | 154.1, C | 8 | 1.20, d, 3.7 | 21.1, CH3 |
| 6 | 7.30, brdd, 0.9, 7.7 | 110.5, CH | 9 | 1.19, d, 3.4 | 21.0, CH3 |
| 7 | 7.37, dd,8.0, 7.7 | 126.2, CH | 10 | 3.31, m | 28.7, CH |
| 8 | 7.55, dd, 0.9, 8.2 | 123.0, CH | 11 | 1.23, d, 4.0 | 21.2, CH3 |
| 8a | - | 136.3, C | 12 | 1.22, d, 4.6 | 21.2, CH3 |
| OH | 9.29 | - | |||
| 4-O-methyl-β-d-glucopyranose | |||||
| 1′ | 5.07, d, 8.2 | 102.5, CH | 1′ | 5.63, d, 7.9 | 98.8, CH |
| 2′ | 3.40, dd, 8.2, 8.6 | 73.7, CH | 2′ | 3.51, dd, 8.1, 8.2 | 74.9, CH |
| 3′ | 3.48, overlapping | 76.0, CH | 3′ | 3.58, dd, 8.9, 9.5 | 78.9, CH |
| 4′ | 3.10, dd, 9.0, 9.5 | 79.1, CH | 4′ | 3.21, dd, 8.9, 8.2 | 80.8, CH |
| 5′ | 3.50, overlapping | 76.2, CH | 5′ | 3.32, m | 77.7, CH |
| 6′ | 3.72, ddd, 11.9, 4.8, 1.7 | 60.3, CH2 | 6′ | 3.76, dd, 2.1, 12.2 | 62.2, CH2 |
| 3.56, dd, 11.6, 5.1 | 3.67, dd, 4.4, 12.1 | ||||
| OMe | 3.48, s | 59.7, CH3 | OMe | 3.59, s | 61.0, CH3 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Helaly, S.E.; Kuephadungphan, W.; Phongpaichit, S.; Luangsa-ard, J.J.; Rukachaisirikul, V.; Stadler, M. Five Unprecedented Secondary Metabolites from the Spider Parasitic Fungus Akanthomyces novoguineensis. Molecules 2017, 22, 991. https://doi.org/10.3390/molecules22060991
Helaly SE, Kuephadungphan W, Phongpaichit S, Luangsa-ard JJ, Rukachaisirikul V, Stadler M. Five Unprecedented Secondary Metabolites from the Spider Parasitic Fungus Akanthomyces novoguineensis. Molecules. 2017; 22(6):991. https://doi.org/10.3390/molecules22060991
Chicago/Turabian StyleHelaly, Soleiman E., Wilawan Kuephadungphan, Souwalak Phongpaichit, Janet Jennifer Luangsa-ard, Vatcharin Rukachaisirikul, and Marc Stadler. 2017. "Five Unprecedented Secondary Metabolites from the Spider Parasitic Fungus Akanthomyces novoguineensis" Molecules 22, no. 6: 991. https://doi.org/10.3390/molecules22060991
APA StyleHelaly, S. E., Kuephadungphan, W., Phongpaichit, S., Luangsa-ard, J. J., Rukachaisirikul, V., & Stadler, M. (2017). Five Unprecedented Secondary Metabolites from the Spider Parasitic Fungus Akanthomyces novoguineensis. Molecules, 22(6), 991. https://doi.org/10.3390/molecules22060991








