Hochu-ekki-to Treatment Improves Reproductive and Immune Modulation in the Stress-Induced Rat Model of Polycystic Ovarian Syndrome
Abstract
:1. Introduction
2. Results
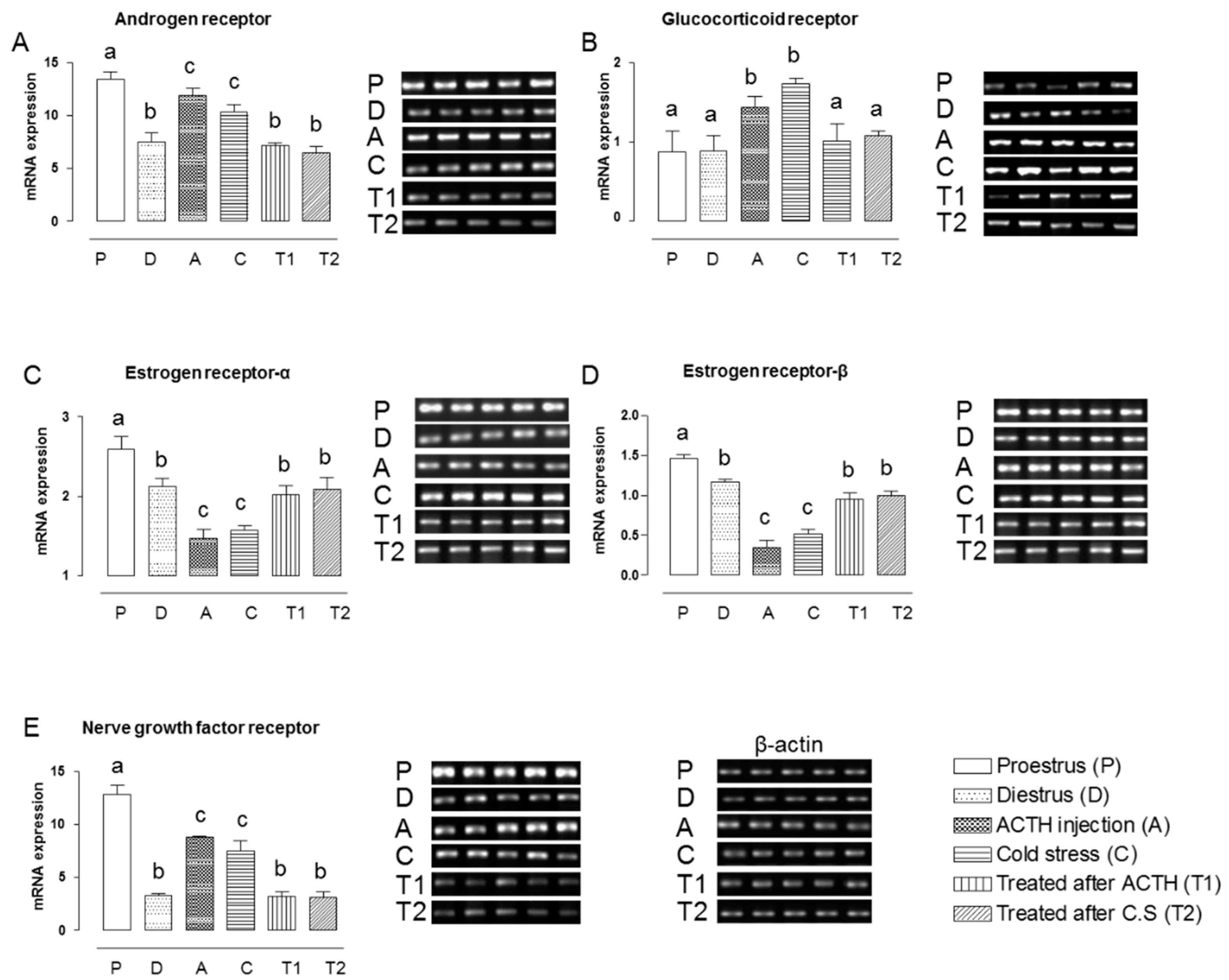
2.1. Effect of Hochu-ekki-to on the mRNA Expression Levels of NGFR and Steroid Hormone Receptors in the Ovaries of Stress-Exposed Rats
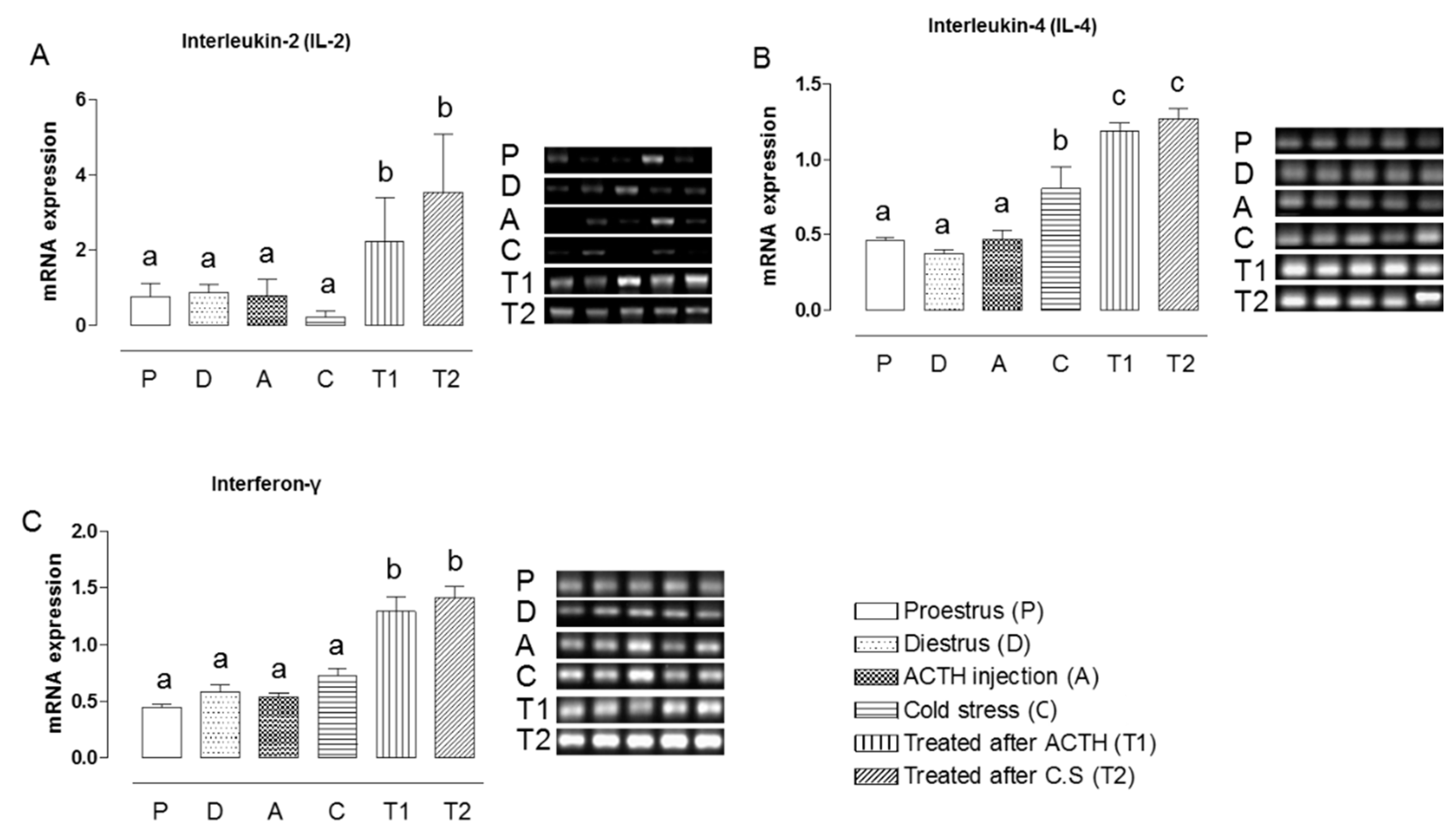
2.2. Effect of Hochu-ekki-to on the mRNA Expression Levels of IL-2, IL-4, and IFN-γ in Stress-Exposed Rats
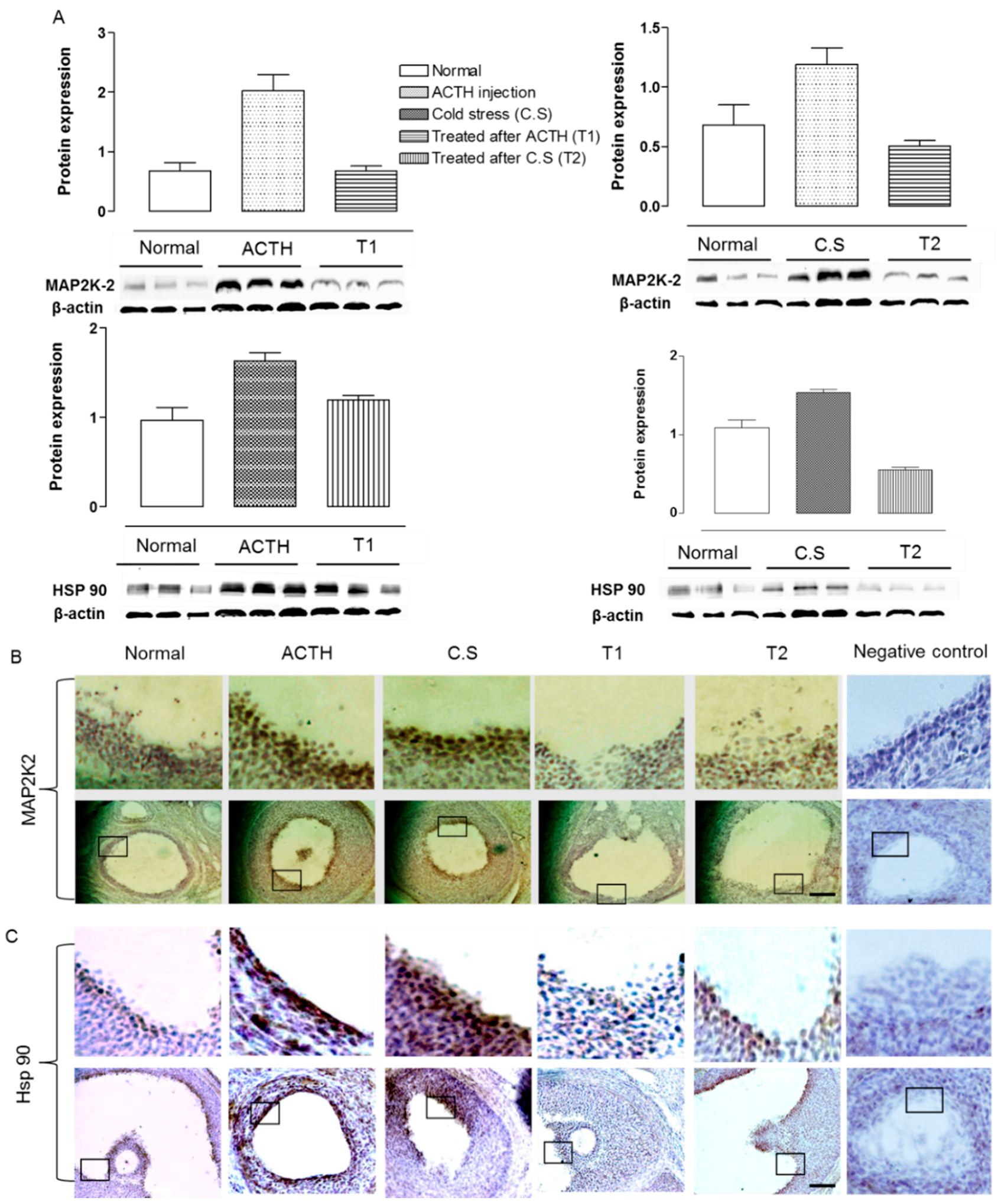
2.3. Effect of Hochu-ekki-to on Hsp 90 and MAP2K2 Expression in the Ovaries of Stress-Exposed Rats
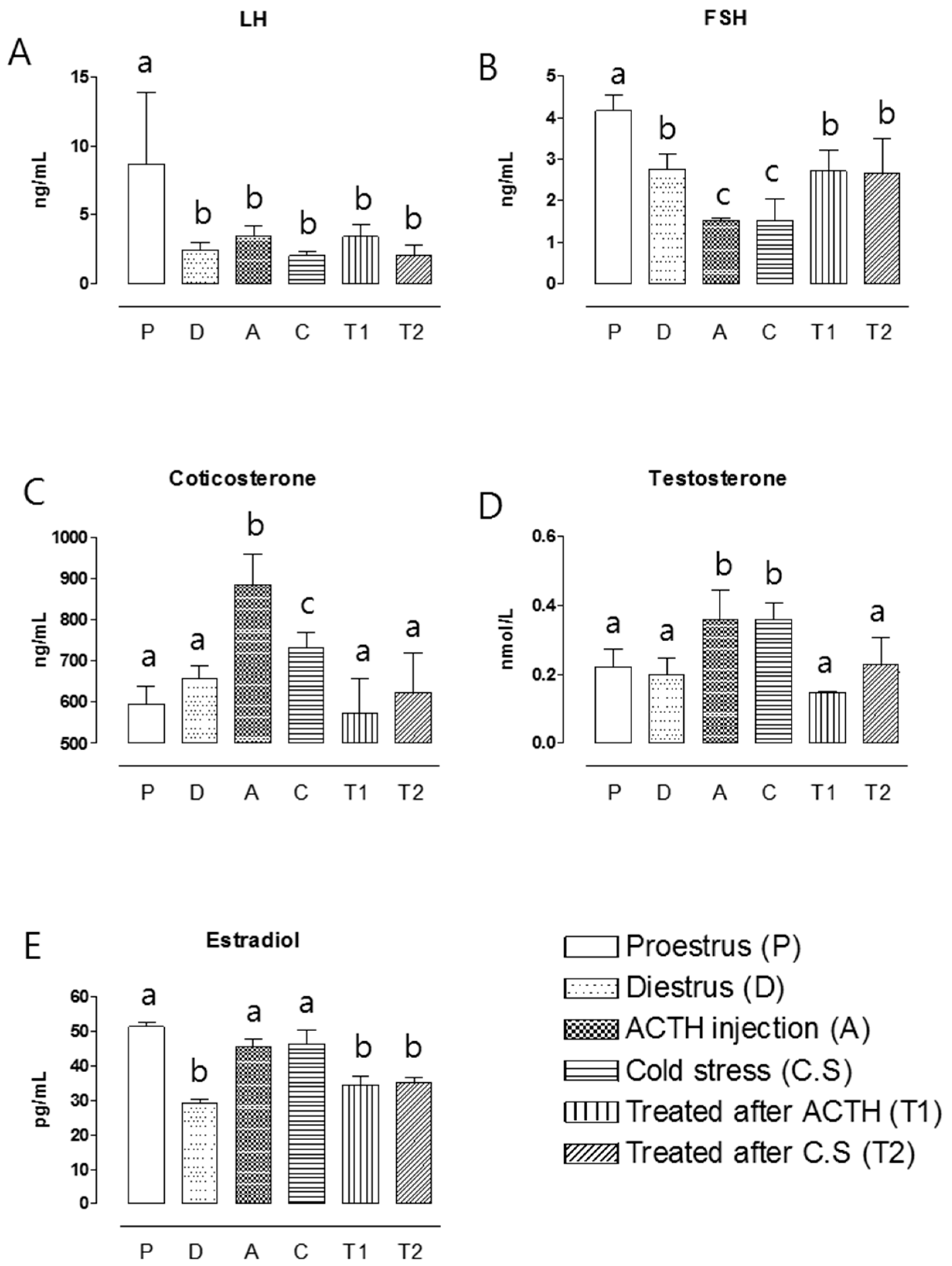
2.4. Hormone Assay
3. Discussion
4. Materials and Methods
4.1. Animals and Treatments
4.2. Tissue Sampling
4.3. RNA Extraction and Reverse Transcription Polymerase Chain Reaction (RT-PCR)
4.4. Western Blotting Analysis
4.5. Immunohistochemistry
4.6. Blood Samples
4.7. ELISA of FSH and LH
4.8. RIAs for Testosterone, Corticosterone and Estradiol
4.9. Statistical Analyses
Supplementary Materials
Acknowledgements
Author Contributions
Conflicts of Interest
References
- Haq, F.; Rizvi, J. Infertility and polycystic ovarian syndrome: A study of association between body mass index and intrafamily marriages. Gynecol. Obstet. Investig. 2008, 65, 269–274. [Google Scholar] [CrossRef] [PubMed]
- Norman, R.J.; Dewailly, D.; Legro, R.S.; Hickey, T.E. Polycystic ovary syndrome. Lancet 2007, 370, 685–697. [Google Scholar] [CrossRef]
- Kimura, M.; Sasada, T.; Kanai, M.; Kawai, Y.; Yoshida, Y.; Hayashi, E.; Iwata, S.; Takabayashi, A. Preventive effect of a traditional herbal medicine, hochu-ekki-to, on immunosuppression induced by surgical stress. Surg. Today 2008, 38, 316–322. [Google Scholar] [CrossRef] [PubMed]
- Ohgitani, E.; Kita, M.; Mazda, O.; Imanishi, J. Combined administration of oseltamivir and hochu-ekki-to (tj-41) dramatically decreases the viral load in lungs of senescence-accelerated mice during influenza virus infection. Arch. Virol. 2014, 159, 267–275. [Google Scholar] [CrossRef] [PubMed]
- Cho, S.; Hong, T.; Kaneko, A.; Yoshino, G.; Sato, N.; Kikuchi, K.; Aikawa, Y.; Yasuno, F.; Inoue, K.; Cyong, J.C. Evaluation of immunological effects of hochu-ekki-to (tj-41) prophylactic administration in mice. Am. J. Chin. Med. 2004, 32, 235–243. [Google Scholar] [CrossRef] [PubMed]
- Kuroiwa, A.; Liou, S.; Yan, H.; Eshita, A.; Naitoh, S.; Nagayama, A. Effect of a traditional Japanese herbal medicine, hochu-ekki-to (bu-zhong-yi-qi tang), on immunity in elderly persons. Int. Immunopharmacol. 2004, 4, 317–324. [Google Scholar] [CrossRef] [PubMed]
- Yan, X.; Kita, M.; Minami, M.; Yamamoto, T.; Kuriyama, H.; Ohno, T.; Iwakura, Y.; Imanishi, J. Antibacterial effect of kampo herbal formulation hochu-ekki-to (bu-zhong-yi-qi-tang) on helicobacter pylori infection in mice. Microbiol. Immunol. 2002, 46, 475–482. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, H.; Mizuno, N.; Teramae, H.; Kutsuna, H.; Ueoku, S.; Onoyama, J.; Yamanaka, K.; Fujita, N.; Ishii, M. The effects of hochu-ekki-to in patients with atopic dermatitis resistant to conventional treatment. Int. J. Tissue React. 2004, 26, 113–117. [Google Scholar] [PubMed]
- Wang, X.Q.; Takahashi, T.; Zhu, S.J.; Moriya, J.; Saegusa, S.; Yamakawa, J.; Kusaka, K.; Itoh, T.; Kanda, T. Effect of hochu-ekki-to (tj-41), a Japanese herbal medicine, on daily activity in a murine model of chronic fatigue syndrome. eCAM 2004, 1, 203–206. [Google Scholar] [CrossRef] [PubMed]
- Yoshioka, H.; Fukaya, S.; Onosaka, S.; Nonogaki, T.; Nagatsu, A. Kampo formula “hochu-ekki-to” suppressed carbon tetrachloride-induced hepatotoxicity in mice. Environ. Health Prev. Med. 2016, 21, 579–584. [Google Scholar] [CrossRef] [PubMed]
- Beato, M.; Klug, J. Steroid hormone receptors: An update. Hum. Reprod. Update 2000, 6, 225–236. [Google Scholar] [CrossRef] [PubMed]
- Nakada, T.; Watanabe, K.; Matsumoto, T.; Santa, K.; Triizuka, K.; Hanawa, T. Effect of orally administered hochu-ekki-to, a Japanese herbal medicine, on contact hypersensitivity caused by repeated application of antigen. Int. Immunopharmacol. 2002, 2, 901–911. [Google Scholar] [CrossRef]
- Utsuyama, M.; Seidlar, H.; Kitagawa, M.; Hirokawa, K. Immunological restoration and anti-tumor effect by Japanese herbal medicine in aged mice. Mech. Ageing Dev. 2001, 122, 341–352. [Google Scholar] [CrossRef]
- Yang, S.H.; Kao, T.I.; Chiang, B.L.; Chen, H.Y.; Chen, K.H.; Chen, J.L. Immune-modulatory effects of bu-zhong-yi-qi-tang in ovalbumin-induced murine model of allergic asthma. PLoS ONE 2015, 10, e0127636. [Google Scholar] [CrossRef] [PubMed]
- Engmann, L.; DiLuigi, A.; Schmidt, D.; Nulsen, J.; Maier, D.; Benadiva, C. The use of gonadotropin-releasing hormone (GnRH) agonist to induce oocyte maturation after cotreatment with GnRH antagonist in high-risk patients undergoing in vitro fertilization prevents the risk of ovarian hyperstimulation syndrome: A prospective randomized controlled study. Fertil. Steril. 2008, 89, 84–91. [Google Scholar] [PubMed]
- Carriere, P.D.; Brawer, J.R.; Farookhi, R. Alterations in gonadotropin-releasing hormone-dependent gonadotropin secretion in rats with polycystic ovaries. Biol. Reprod. 1991, 45, 685–690. [Google Scholar] [CrossRef] [PubMed]
- Hemmings, R.; Farookhi, R.; Brawer, J.R. Pituitary and ovarian responses to luteinizing hormone releasing hormone in a rat with polycystic ovaries. Biol. Reprod. 1983, 29, 239–248. [Google Scholar] [CrossRef] [PubMed]
- Dobson, H.; Ribadu, A.Y.; Noble, K.M.; Tebble, J.E.; Ward, W.R. Ultrasonography and hormone profiles of adrenocorticotrophic hormone (ACTH)-induced persistent ovarian follicles (cysts) in cattle. J. Reprod. Fertil. 2000, 120, 405–410. [Google Scholar] [CrossRef] [PubMed]
- Csermely, P.; Schnaider, T.; Soti, C.; Prohaszka, Z.; Nardai, G. The 90-kda molecular chaperone family: Structure, function, and clinical applications. A comprehensive review. Pharmacol. Ther. 1998, 79, 129–168. [Google Scholar] [CrossRef]
- Park, E.; Cockrem, J.F.; Han, K.H.; Kim, D.H.; Jung, M.H.; Chu, J.P. Stress-induced activation of ovarian heat shock protein 90 in a rat model of polycystic ovary syndrome. J. Obstet. Gynaecol. Res. 2012, 38, 396–407. [Google Scholar] [CrossRef] [PubMed]
- Wadekar, S.A.; Li, D.; Periyasamy, S.; Sanchez, E.R. Inhibition of heat shock transcription factor by GR. Mol. Endocrinol. 2001, 15, 1396–1410. [Google Scholar] [CrossRef] [PubMed]
- Jaiswal, R.K.; Weissinger, E.; Kolch, W.; Landreth, G.E. Nerve growth factor-mediated activation of the mitogen-activated protein (map) kinase cascade involves a signaling complex containing b-raf and hsp90. J. Boil. Chem. 1996, 271, 23626–23629. [Google Scholar]
- Lan, C.W.; Chen, M.J.; Tai, K.Y.; Yu, D.C.; Yang, Y.C.; Jan, P.S.; Yang, Y.S.; Chen, H.F.; Ho, H.N. Functional microarray analysis of differentially expressed genes in granulosa cells from women with polycystic ovary syndrome related to mapk/erk signaling. Sci. Rep. 2015, 5, 14994. [Google Scholar] [CrossRef] [PubMed]
Availability: Samples of the extracts are available from the authors. |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Park, E.; Choi, C.W.; Kim, S.J.; Kim, Y.-I.; Sin, S.; Chu, J.-P.; Heo, J.Y. Hochu-ekki-to Treatment Improves Reproductive and Immune Modulation in the Stress-Induced Rat Model of Polycystic Ovarian Syndrome. Molecules 2017, 22, 978. https://doi.org/10.3390/molecules22060978
Park E, Choi CW, Kim SJ, Kim Y-I, Sin S, Chu J-P, Heo JY. Hochu-ekki-to Treatment Improves Reproductive and Immune Modulation in the Stress-Induced Rat Model of Polycystic Ovarian Syndrome. Molecules. 2017; 22(6):978. https://doi.org/10.3390/molecules22060978
Chicago/Turabian StylePark, Eunkuk, Chun Whan Choi, Soo Jeong Kim, Yong-In Kim, Samkee Sin, Jong-Phil Chu, and Jun Young Heo. 2017. "Hochu-ekki-to Treatment Improves Reproductive and Immune Modulation in the Stress-Induced Rat Model of Polycystic Ovarian Syndrome" Molecules 22, no. 6: 978. https://doi.org/10.3390/molecules22060978
APA StylePark, E., Choi, C. W., Kim, S. J., Kim, Y.-I., Sin, S., Chu, J.-P., & Heo, J. Y. (2017). Hochu-ekki-to Treatment Improves Reproductive and Immune Modulation in the Stress-Induced Rat Model of Polycystic Ovarian Syndrome. Molecules, 22(6), 978. https://doi.org/10.3390/molecules22060978






