Synthesis and Bioactivity Evaluation of N-Arylsulfonylindole Analogs Bearing a Rhodanine Moiety as Antibacterial Agents
Abstract
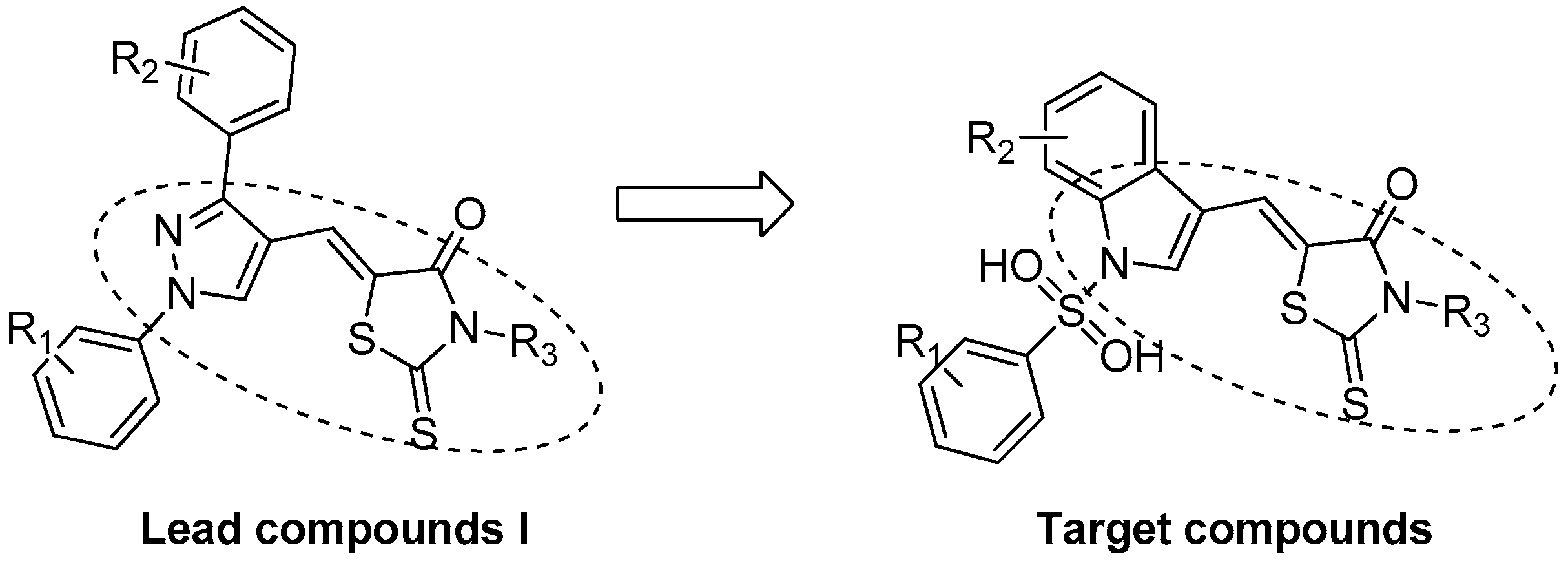
:1. Introduction
2. Results and Discussion
2.1. Chemistry
2.2. Antimicrobial Activity
2.3. Cytotoxicity
2.4. The Structure-Activity Relationships (SARs) Analysis
3. Materials and Methods
3.1. Instruments and Reagents
3.2. Synthesis Method and Spectral Data
3.2.1. General Procedure for the Preparation of Compounds 3a–3b
3.2.2. General Procedure for the Preparation of Compounds 3c–3f
3.2.3. General Procedure for the Preparation of Compounds 5–11
3.2.4. Spectral Data
3.3. Evaluation of Anti-Bacterial Activity In Vitro
3.4. Evaluation of Cytotoxicity In Vitro
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Livermore, D.M. Has the era of untreatable infections arrived? J. Antimicrob. Chemother. 2009, 64 (Suppl. 1), i29–i36. [Google Scholar] [CrossRef] [PubMed]
- Bi, Y.; Liu, X.X.; Zhang, H.Y.; Yang, X.; Liu, Z.Y.; Lu, J.; Lewis, P.J.; Wang, C.Z.; Xu, J.Y.; Meng, Q.G.; et al. Synthesis and Antibacterial Evaluation of Novel 3-Substituted Ocotillol-Type Derivatives as Leads. Molecules 2017, 22, 590. [Google Scholar] [CrossRef] [PubMed]
- Carrel, M.; Perencevich, E.N.; David, M.Z. USA300 Methicillin-Resistant Staphylococcus aureus, United States, 2000–2013. Emerg. Infect. Dis. 2015, 21, 1973–1980. [Google Scholar] [CrossRef] [PubMed]
- Hvistendahl, M. Public health. China takes aim at rampant antibiotic resistance. Science 2012, 336, 795. [Google Scholar] [CrossRef] [PubMed]
- Yezli, S.; Li, H. Antibiotic resistance amongst healthcare-associated pathogens in China. Int. J. Antimicrob. Agents 2012, 40, 389–397. [Google Scholar] [CrossRef] [PubMed]
- Azeredo da Silveira, S.; Perez, A. Liposomes as novel anti-infectives targeting bacterial virulence factors? Expert. Rev. Anti-Infect. Ther. 2015, 13, 531–533. [Google Scholar] [CrossRef] [PubMed]
- Cascioferro, S.; Cusimano, M.G.; Schillaci, D. Antiadhesion agents against Gram-positive pathogens. Future Microbiol. 2014, 9, 1209–1220. [Google Scholar] [CrossRef] [PubMed]
- Tay, S.B.; Yew, W.S. Development of quorum-based anti-virulence therapeutics targeting Gram negative bacterial pathogens. Int. J. Mol. Sci. 2013, 14, 16570–16599. [Google Scholar] [CrossRef] [PubMed]
- Rademacher, J.; Welte, T. New antibiotics - standstill or progress. Med. Klin. Intensivmed. Notfmed. 2017, 112, 206–213. [Google Scholar] [CrossRef] [PubMed]
- Boucher, H.W.; Talbot, G.H.; Benjamin, D.K.; Bradley, J.; Guidos, R.J.; Jones, R.N.; Murray, B.E.; Bonomo, R.A.; Gilbert, D. Infectious Diseases Society of America. 10 x '20 Progress--development of new drugs active against gram-negative bacilli: an update from the Infectious Diseases Society of America. Clin. Infect. Dis. 2013, 56, 1685–1694. [Google Scholar] [CrossRef] [PubMed]
- Bhale, P.S.; Chavan, H.V.; Dongare, S.B.; Shringare, S.N.; Mule, Y.B.; Nagane, S.S.; Bandgar, B.P. Synthesis of extended conjugated indolyl chalcones as potent anti-breast cancer, anti-inflammatory and antioxidant agents. Bioorg. Med. Chem. Lett. 2017, 27, 1502–1507. [Google Scholar] [CrossRef] [PubMed]
- Diao, P.C.; Li, Q.; Hu, M.J.; Ma, Y.F.; You, W.W.; Hong, K.H.; Zhao, P.L. Synthesis and biological evaluation of novel indole-pyrimidine hybrids bearing morpholine and thiomorpholine moieties. Eur. J. Med. Chem. 2017, 13, 110–118. [Google Scholar] [CrossRef] [PubMed]
- Manuel-Manresa, P.; Korrodi-Gregório, L.; Hernando, E.; Villanueva, A.; Martínez-García, D.; Rodilla, A.M.; Ramos, R.; Fardilha, M.; Moya, J.; Quesada, R.; et al. Novel indole-based tambjamine-analogues induce apoptotic lung cancer cell death through p38 mitogen-activated protein kinase activation. Mol. Cancer. Ther. 2017. (In press) [Google Scholar] [CrossRef] [PubMed]
- Barbosa, V.A.; Baréa, P.; Mazia, R.S.; Ueda-Nakamura, T.; Costa, W.F.; Foglio, M.A.; Goes Ruiz, A.L.; Carvalho, J.E.; Vendramini-Costa, D.B.; Nakamura, C.V.; et al. Synthesis and evaluation of novel hybrids β-carboline-4-thiazolidinones as potential antitumor and antiviral agents. Eur. J. Med. Chem. 2016, 124, 1093–1104. [Google Scholar] [CrossRef] [PubMed]
- Kumar, V.P.; Renjitha, J.; Salfeena, C.F.; Ashitha, K.T.; Keri, R.S.; Varughese, S.; Sasidhar, B.S. Antibacterial and anti-tubercular evaluation of Dihydronaphthalenone-indole hybrid analogues. Chem. Biol. Drug. Des. 2017. (In press) [Google Scholar] [CrossRef]
- Patil, S.A.; Patil, S.A.; Patil, R. Medicinal applications of (benz)imidazole- and indole-based macrocycles. Chem. Biol. Drug. Des. 2017, 89, 639–649. [Google Scholar] [CrossRef] [PubMed]
- Tan, M.C.; Wong, W.Y.; Ng, W.L.; Yeo, K.S.; Mohidin, T.B.; Lim, Y.Y.; Lafta, F.; Mohd Ali, H.; Ea, C.K. Identification of 5-Methoxy-2-(Diformylmethylidene)-3,3-Dimethylindole as an Anti-Influenza A Virus Agent. PLoS ONE 2017, 12, e0170352. [Google Scholar] [CrossRef] [PubMed]
- Scuotto, M.; Abdelnabi, R.; Collarile, S.; Schiraldi, C.; Delang, L.; Massa, A.; Ferla, S.; Brancale, A.; Leyssen, P.; Neyts, J.; et al. Discovery of novel multi-target indole-based derivatives as potent and selective inhibitors of chikungunya virus replication. Bioorg. Med. Chem. 2017, 25, 327–337. [Google Scholar] [CrossRef] [PubMed]
- Che, Z.; Tian, Y.; Hu, Z.; Chen, Y.; Liu, S.; Chen, G. Synthesis and in vitro anti-HIV-1 activity of a series of N-arylsulfonyl-3-propionylindoles. Z. Naturforsch. C 2016, 71, 105–109. [Google Scholar] [CrossRef] [PubMed]
- Silvestri, R.; De Martino, G.; La Regina, G.; Artico, M.; Massa, S.; Vargiu, L.; Mura, M.; Loi, A.G.; Marceddu, T.; La Colla, P. Novel indolyl aryl sulfones active against HIV-1 carrying NNRTI resistance mutations: Synthesis and SAR studies. J. Med. Chem. 2003, 46, 2482–2493. [Google Scholar] [CrossRef] [PubMed]
- Vera, G.; Lagos, C.F.; Almendras, S.; Hebel, D.; Flores, F.; Valle-Corvalán, G.; Pessoa-Mahana, C.D.; Mella-Raipán, J.; Montecinos, R.; Recabarren-Gajardo, G. Extended N-Arylsulfonylindoles as 5-HT6 Receptor Antagonists: Design, Synthesis & amp; Biological Evaluation. Molecules 2016, 21, 1070. [Google Scholar]
- Fan, L.L.; Liu, W.Q.; Xu, H.; Yang, L.M.; Lv, M.; Zheng, Y.T. Anti human immunodeficiency virus-1 (HIV-1) agents 3. synthesis and in vitro anti-HIV-1 activity of some N-arylsulfonylindoles. Chem. Pharm. Bull. (Tokyo) 2009, 57, 797–800. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Wang, Y.Y. Antifungal agents. Part 5: Synthesis and antifungal activities of aminoguanidine derivatives of N-arylsulfonyl-3-acylindoles. Bioorg. Med. Chem. Lett. 2010, 20, 7274–7277. [Google Scholar] [CrossRef] [PubMed]
- Song, M.X.; Deng, X.Q.; Li, Y.R.; Zheng, C.J.; Hong, L.; Piao, H.R. Synthesis And biological evaluation of (E)-1-(substituted)-3-phenylprop-2-en-1-ones bearing rhodanines as potent antimicrobial agents. J. Enzym. Inhib. Med. Chem. 2014, 29, 647–653. [Google Scholar] [CrossRef] [PubMed]
- Song, M.X.; Zheng, C.J.; Deng, X.Q.; Wei, Z.Y.; Piao, H.R. The Synthesis and Anti-Bacterial Activities of N-carboxymethyl Rhodanines. Med. Chem. 2014, 4, 441–448. [Google Scholar]
- Song, M.X.; Zheng, C.J.; Sun, L.P.; Wu, Y.; Hong, L.; Li, Y.J.; Liu, Y.; Wei, Z.Y.; Jin, M.J.; Piao, H.R. Synthesis and antibacterial evaluation of rhodanine-based 5-aryloxy pyrazoles against selected methicillin resistant and quinolone-resistant Staphylococcus aureus (MRSA and QRSA). Eur. J. Med. Chem. 2013, 60, 376–385. [Google Scholar] [CrossRef] [PubMed]
- Song, M.X.; Zheng, C.J.; Deng, X.Q.; Wang, Q.; Hou, S.P.; Liu, T.T.; Xing, X.L.; Piao, H.R. Synthesis and bioactivity evaluation of rhodanine derivatives as potential anti-bacterial agents. Eur. J. Med. Chem. 2012, 54, 403–412. [Google Scholar] [CrossRef] [PubMed]
- Zheng, C.J.; Song, M.X.; Sun, L.P.; Wu, Y.; Hong, L.; Piao, H.R. Synthesis and biological evaluation of 5-aryloxypyrazole derivatives bearing a rhodanine-3-aromatic acid as potential antimicrobial agents. Bioorg. Med. Chem. Lett. 2012, 22, 7024–7028. [Google Scholar] [CrossRef] [PubMed]
- Jin, X.; Zheng, C.J.; Song, M.X.; Wu, Y.; Sun, L.P.; Li, Y.J.; Yu, L.J.; Piao, H.R. Synthesis and antimicrobial evaluation of l-phenylalanine-derived C5-substituted rhodanine and chalcone derivatives containing thiobarbituric acid or 2-thioxo-4-thiazolidinone. Eur. J. Med. Chem. 2012, 56, 203–209. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.H.; Zheng, C.J.; Sun, L.P.; Piao, H.R. Synthesis of new chalcone derivatives containing a rhodanine-3-acetic acid moiety with potential anti-bacterial activity. Eur. J. Med. Chem. 2010, 45, 5739–5743. [Google Scholar] [CrossRef] [PubMed]
- Clinical and Laboratory Standards Institute. Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria that Grow Aerobically, Approved Standard M7-A6; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2003. [Google Scholar]
Sample Availability: Samples of the compounds are available from the authors for a short period of time. |


| Compd. | R1- | R2- | R3- | Gram-Positive Strains | Gram-Negative Strains | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 26003 a | 25923 b | 32067 c | 29212 d | 63501 e | 25922 f | 44568 g | 27853 h | 10104 i | ||||
| 5a | H | 5-Br | CH2COOH | 8 | 8 | >128 | 32 | >128 | >128 | >128 | >128 | 32 |
| 5b | H | 6-Cl | CH2COOH | >128 | >128 | >128 | >128 | >128 | >128 | >128 | >128 | >128 |
| 5c | CH3 | 5-Br | CH2COOH | 8 | 16 | >128 | >128 | >128 | >128 | >128 | >128 | >128 |
| 5d | CH3 | 6-Cl | CH2COOH | 4 | 4 | >128 | 32 | >128 | >128 | >128 | >128 | 32 |
| 6a | H | H |  | 2 | 1 | 16 | 8 | >128 | >128 | >128 | >128 | 8 |
| 6b | H | 6-Cl |  | 4 | 2 | >128 | 16 | >128 | >128 | >128 | >128 | 16 |
| 6c | CH3 | H |  | 2 | 1 | >128 | 4 | >128 | >128 | >128 | >128 | 4 |
| 6d | CH3 | 5-Br |  | 2 | 2 | >128 | 4 | >128 | >128 | >128 | >128 | 4 |
| 6e | CH3 | 6-Cl |  | 64 | >128 | >128 | >128 | >128 | >128 | >128 | >128 | >128 |
| 7a | H | H |  | 2 | 2 | 128 | 4 | >128 | >128 | >128 | >128 | 4 |
| 7b | H | 6-Cl |  | 32 | 16 | >128 | 8 | >128 | >128 | >128 | >128 | 8 |
| 7c | CH3 | H |  | 2 | 16 | >128 | >128 | >128 | >128 | >128 | >128 | >128 |
| 7d | CH3 | 5-Br |  | 2 | 2 | >128 | 16 | >128 | >128 | >128 | >128 | 16 |
| 8a | H | H |  | 2 | 1 | 4 | 16 | >128 | >128 | >128 | >128 | 16 |
| 8b | H | 6-Cl |  | 1 | 0.5 | 2 | 4 | >128 | >128 | >128 | >128 | 4 |
| 8c | CH3 | H |  | 1 | 1 | 128 | >128 | >128 | >128 | >128 | >128 | >128 |
| 8d | CH3 | 5-Br |  | 2 | 2 | >128 | 64 | >128 | >128 | >128 | >128 | 64 |
| 8e | CH3 | 6-Cl |  | 2 | 2 | >128 | 64 | >128 | >128 | >128 | >128 | 64 |
| 9 | H | H |  | 4 | 4 | >128 | 32 | >128 | >128 | >128 | >128 | 32 |
| 10 | H | H |  | 2 | 2 | 16 | 16 | >128 | >128 | >128 | >128 | 16 |
| 11 | H | H |  | >128 | 8 | >128 | 32 | >128 | >128 | >128 | >128 | 32 |
| Gatifloxacin | — | — | — | 0.125 | 0.125 | >128 | >128 | 1 | 2 | 0.125 | 0.125 | 2 |
| Moxifloxacin | — | — | — | 0.125 | 0.125 | >128 | >128 | 1 | 2 | 0.125 | 0.125 | 2 |
| Norfloxacin | — | — | — | 0.125 | 0.125 | >128 | >128 | 1 | 2 | 0.125 | 0.125 | 2 |
| Oxacillin | — | — | — | 0.125 | 0.125 | >128 | >128 | 128 | >128 | 128 | >128 | >128 |
| penicillin | — | — | — | 0.125 | 0.125 | >128 | >128 | 128 | 128 | 128 | >128 | >128 |
| Compound | R1- | R2- | R3- | Multidrug-Resistant Gram-Positive Strains | |
|---|---|---|---|---|---|
| 43300 a | 33591 b | ||||
| 6a | H | H |  | 0.5 | 1 |
| 6c | CH3 | H |  | 0.5 | 1 |
| 8a | H | H |  | 1 | 2 |
| 8b | H | 6-Cl |  | 2 | 2 |
| 8c | CH3 | H |  | 1 | 2 |
| Gatifloxacin | — | — | — | 0.5 | 0.25 |
| Moxifloxacin | — | — | — | 0.5 | 0.25 |
| Norfloxacin | — | — | — | 0.5 | 0.25 |
| Oxacillin | — | — | — | 64 | 8 |
| Penicillin | — | — | — | 32 | >32 |
| Compound | IC50 (µg/mL) a |
|---|---|
| 6a | 36.90 |
| 8b | 54.09 |
| 8c | 32.28 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Song, M.-X.; Li, S.-H.; Peng, J.-Y.; Guo, T.-T.; Xu, W.-H.; Xiong, S.-F.; Deng, X.-Q. Synthesis and Bioactivity Evaluation of N-Arylsulfonylindole Analogs Bearing a Rhodanine Moiety as Antibacterial Agents. Molecules 2017, 22, 970. https://doi.org/10.3390/molecules22060970
Song M-X, Li S-H, Peng J-Y, Guo T-T, Xu W-H, Xiong S-F, Deng X-Q. Synthesis and Bioactivity Evaluation of N-Arylsulfonylindole Analogs Bearing a Rhodanine Moiety as Antibacterial Agents. Molecules. 2017; 22(6):970. https://doi.org/10.3390/molecules22060970
Chicago/Turabian StyleSong, Ming-Xia, Song-Hui Li, Jiao-Yang Peng, Ting-Ting Guo, Wen-Hui Xu, Shao-Feng Xiong, and Xian-Qing Deng. 2017. "Synthesis and Bioactivity Evaluation of N-Arylsulfonylindole Analogs Bearing a Rhodanine Moiety as Antibacterial Agents" Molecules 22, no. 6: 970. https://doi.org/10.3390/molecules22060970
APA StyleSong, M.-X., Li, S.-H., Peng, J.-Y., Guo, T.-T., Xu, W.-H., Xiong, S.-F., & Deng, X.-Q. (2017). Synthesis and Bioactivity Evaluation of N-Arylsulfonylindole Analogs Bearing a Rhodanine Moiety as Antibacterial Agents. Molecules, 22(6), 970. https://doi.org/10.3390/molecules22060970




