Modified Nucleotides as Substrates of Terminal Deoxynucleotidyl Transferase
Abstract
:1. Introduction
2. Results and Discussion
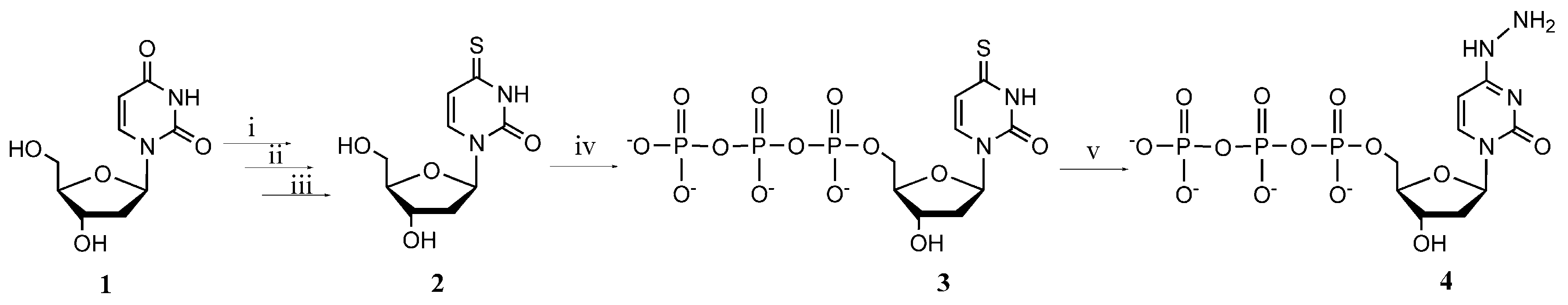
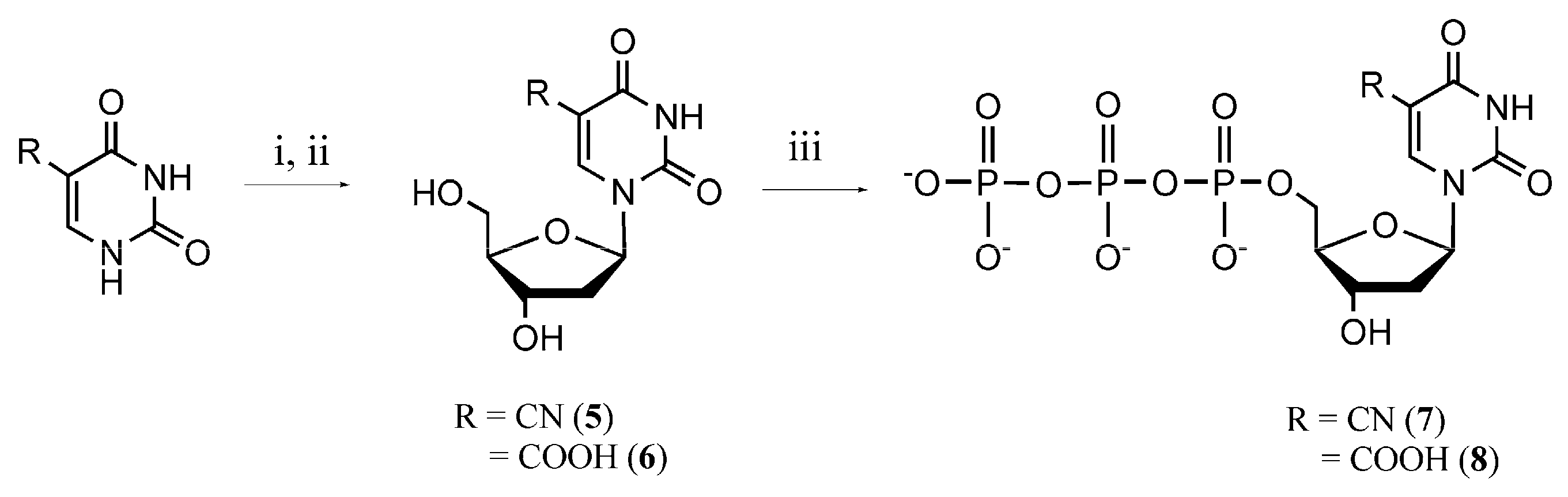
2.1. Synthesis of Modified Nucleotides
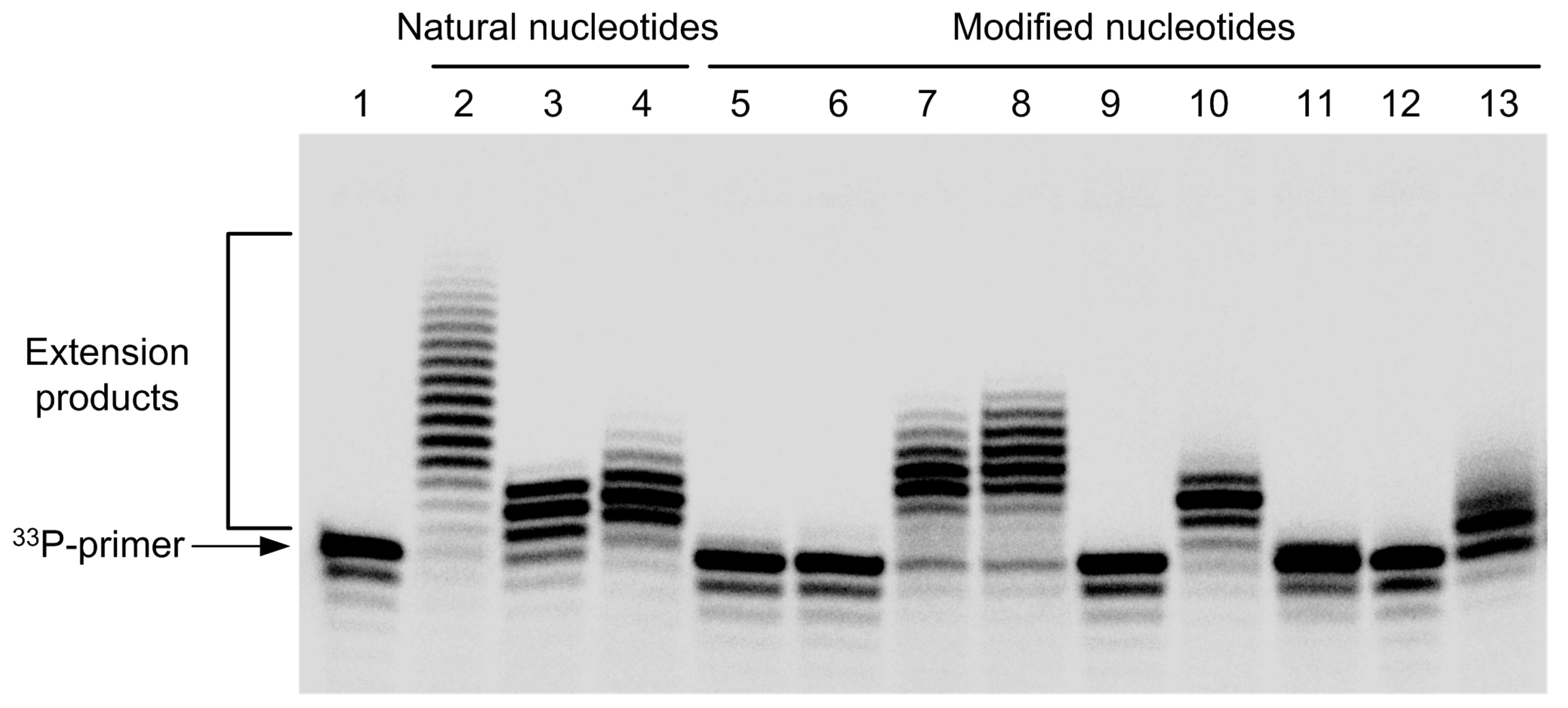
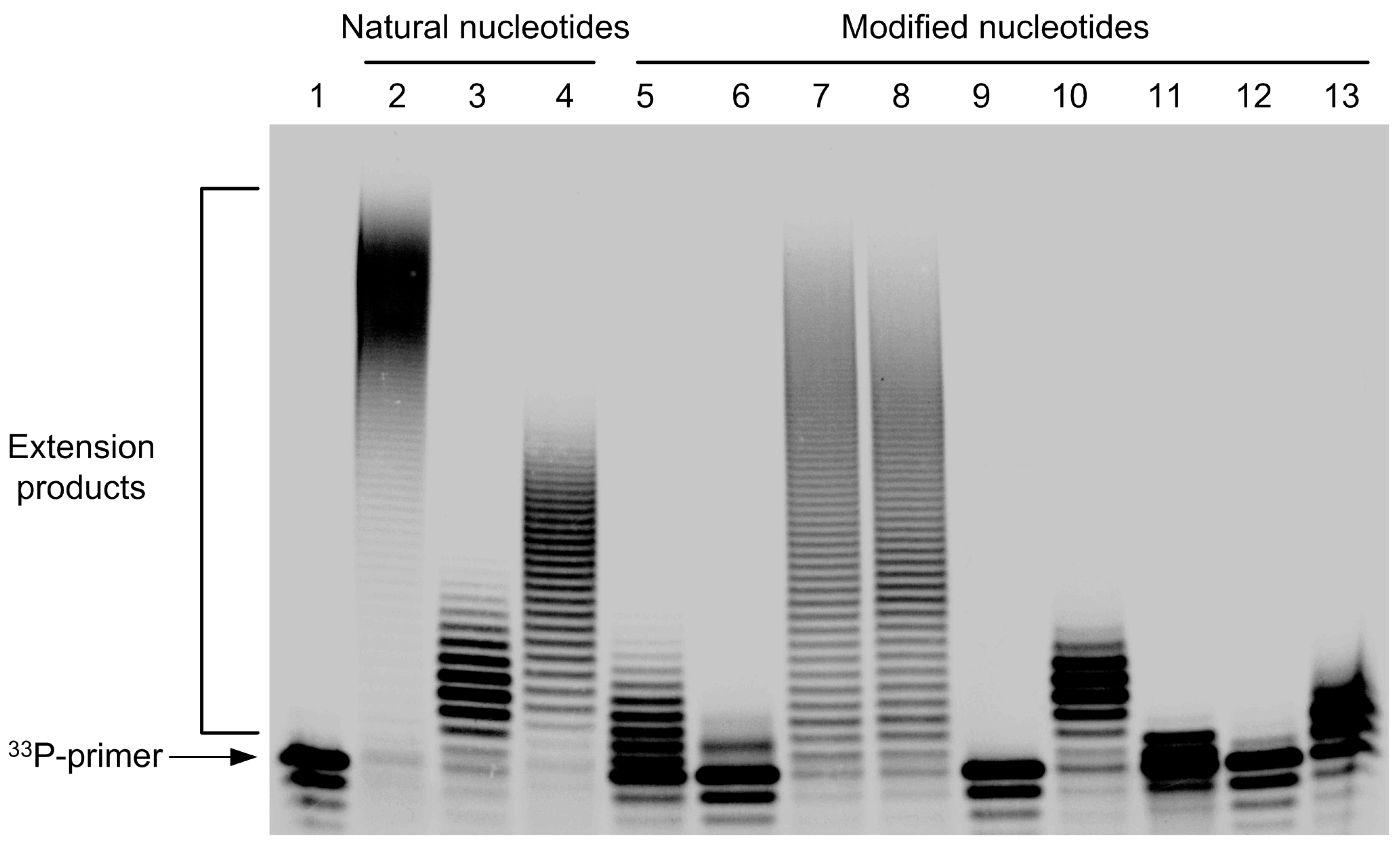
2.2. Incorporation of Modified Nucleotides by Terminal Deoxynucleotidyl Transferase
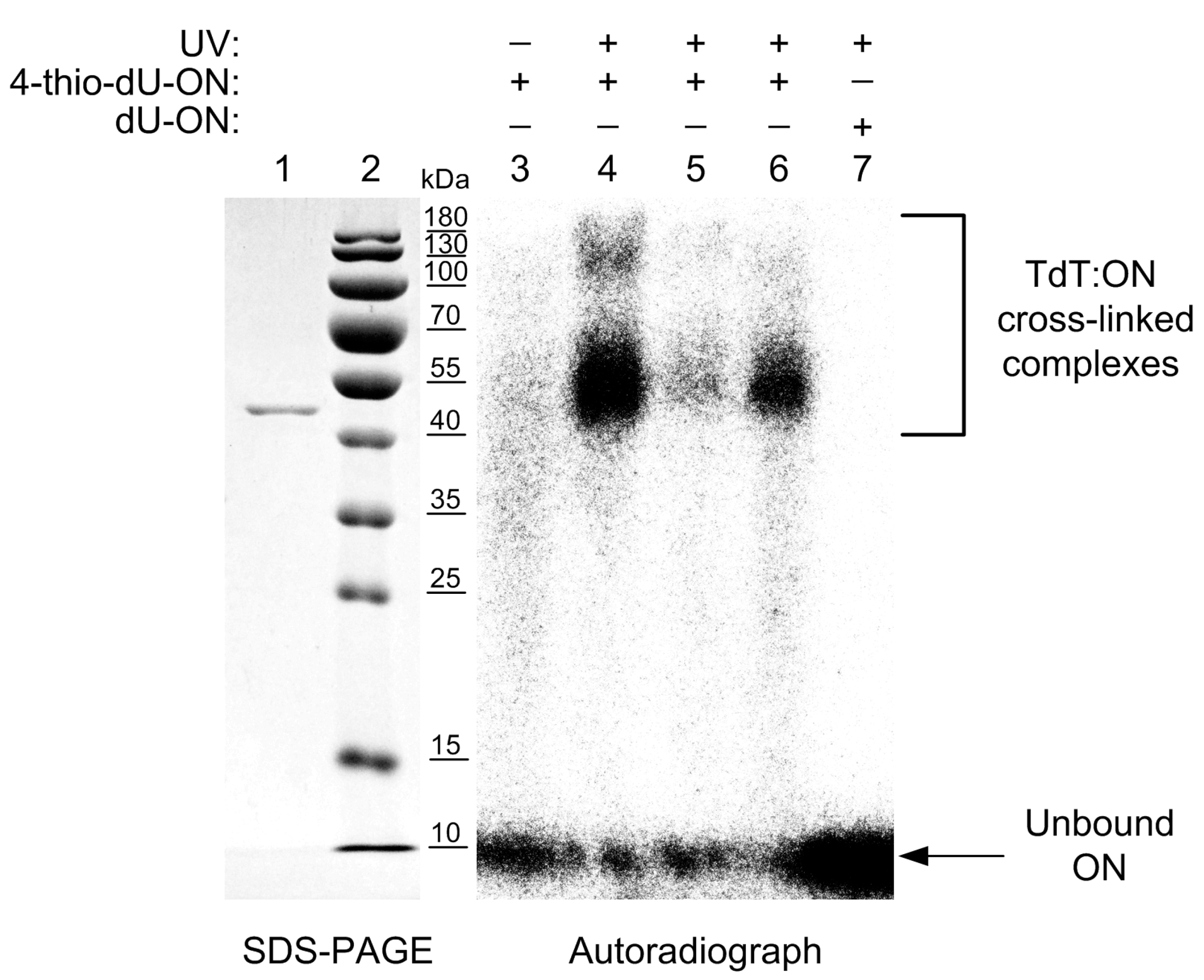
2.3. UV Cross-Linking of Oligonucleotides to Terminal Deoxynucleotidyl Transferase
3. Materials and Methods
3.1. General Information
3.2. Synthesis of 4-Thio-2'-deoxyuridine (2)
3.3. Synthesis of 4-Thio-2'-deoxyuridine-5'-triphosphate (3)
3.4. Synthesis of N4-Amino-2'-deoxycytidine-5'-triphosphate (4)
3.5. Synthesis of 5-Cyano-2ʹ-deoxyuridine (5) and 5-Carboxy-2ʹ-deoxyuridine (6)
3.6. Synthesis of 5-Substituted Deoxyuridine TriphosphateDerivatives 7 and 8
3.7. Synthesis of 2-Pyridone Nucleoside Triphosphates
3.8. Incorporation of Modified Nucleotides by Terminal Deoxynucleotidyl Transferase
3.9. Formation of 4-Thio-dU-ON:Protein Cross-links
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Meek, K.N.; Rangel, A.E.; Heemstra, J.M. Enhancing aptamer function and stability via in vitro selection using modified nucleic acids. Methods 2016, 106, 29–36. [Google Scholar] [CrossRef] [PubMed]
- Lipi, F.; Chen, S.; Chakravarthy, M.; Rakesh, S.; Veedu, R.N. In vitro evolution of chemically-modified nucleic acid aptamers: Pros and cons, and comprehensive selection strategies. RNA Biol. 2016, 13, 1232–1245. [Google Scholar] [CrossRef] [PubMed]
- Kraemer, S.; Vaught, J.D.; Bock, C.; Gold, L.; Katilius, E.; Keeney, T.R.; Kim, N.; Saccomano, N.A.; Wilcox, S.K.; Zichi, D.; et al. From SOMAmer-based biomarker discovery to diagnostic and clinical applications: A SOMAmer-based, streamlined multiplex proteomic assay. PLoS ONE 2011, 6, e26332. [Google Scholar] [CrossRef] [PubMed]
- Tjong, V.; Tang, L.; Zauscher, S.; Chilkoti, A. ‘‘Smart’’ DNA interfaces. Chem. Soc. Rev. 2014, 43, 1612–1626. [Google Scholar] [CrossRef] [PubMed]
- Xiang, B.; He, K.; Zhu, R.; Liu, Z.; Zeng, S.; Huang, Y.; Nie, Z.; Yao, S. Self-assembled DNA hydrogel based on enzymatically polymerized DNA for protein encapsulation and enzyme/DNAzyme hybrid cascade reaction. Appl. Mater. Interfaces 2016, 8, 22801–22807. [Google Scholar] [CrossRef] [PubMed]
- Hong, P.; Li, W.; Li, J. Applications of aptasensors in clinical diagnostics. Sensors 2012, 12, 1181–1193. [Google Scholar] [CrossRef] [PubMed]
- Shivalingam, A.; Brown, T. Synthesis of chemically modified DNA. Biochem. Soc. Trans. 2016, 44, 709–715. [Google Scholar] [CrossRef] [PubMed]
- Brody, E.N.; Gold, L. Aptamers as therapeutic and diagnostic agents. Rev. Mol. Biotechnol. 2000, 74, 5–13. [Google Scholar] [CrossRef]
- Kuwahara, M.; Nagashima, J.; Hasegawa, M.; Tamura, T.; Kitagata, R.; Hanawa, K.; Hososhima, S.; Kasamatsu, T.; Ozaki, H.; Sawai, H. Systematic characterization of 2’-deoxynucleoside- 5’-triphosphate analogs as substrates for DNA polymerases by polymerase chain reaction and kinetic studies on enzymatic production of modified DNA. Nucleic Acids Res. 2006, 34, 5383–5394. [Google Scholar] [CrossRef] [PubMed]
- Capek, P.; Cahova, H.; Pohl, R.; Hocek, M.; Gloeckner, C.; Marx, A. An efficient method for the construction of functionalized DNA bearing amino acid groups through cross-coupling reactions of nucleoside triphosphates followed by primer extension or PCR. Chem. Eur. J. 2007, 13, 6196–6203. [Google Scholar] [CrossRef] [PubMed]
- Berdis, A.J.; McCutcheon, D. The use of non-natural nucleotides to probe template-independent DNA synthesis. Chembiochem 2007, 8, 1399–1408. [Google Scholar] [CrossRef] [PubMed]
- Gutsmiedl, K.; Fazio, D.; Carell, T. High-density DNA functionalization by a combination of Cu-catalyzed and Cu-free click chemistry. Chem. Eur. J. 2010, 16, 6877–6883. [Google Scholar] [CrossRef] [PubMed]
- Schoch, J.; Staudt, M.; Samanta, A.; Wiessler, M.; Jaschke, A. Site-specific one-pot dual labeling of DNA by orthogonal cycloaddition chemistry. Bioconjug. Chem. 2012, 23, 1382–1386. [Google Scholar] [CrossRef] [PubMed]
- Kong, D.; Lei, Y.; Yeung, W.; Hili, R. Enzymatic synthesis of sequence-defined synthetic nucleic acid polymers with diverse functional groups. Angew. Chem. 2016, 128, 13358–13362. [Google Scholar] [CrossRef]
- Hocek, M. Synthesis of base-modified 2′-deoxyribonucleoside triphosphates and their use in enzymatic synthesis of modified DNA for applications in bioanalysis and chemical biology. J. Org. Chem. 2014, 79, 9914–9921. [Google Scholar] [CrossRef] [PubMed]
- Kasahara, Y.; Kuwahara, M. Artificial specific binders directly recovered from chemically modified nucleic acid libraries. J. Nucleic Acids 2012, 156482. [Google Scholar] [CrossRef] [PubMed]
- Sawai, H.; Nagashima, J.; Kuwahara, M.; Kitagata, R.; Tamura, T.; Matsui, I. Differences in substrate specificity of C(5)-substituted or C(5)-unsubstituted pyrimidine nucleotides by DNA polymerases from thermophilic bacteria, archaea, and phages. Chem. Biodivers. 2007, 4, 1979–1995. [Google Scholar] [CrossRef] [PubMed]
- Hottin, A.; Marx, A. Structural insights into the processing of nucleobase-modified nucleotides by DNA polymerases. Acc. Chem. Res. 2016, 49, 418–427. [Google Scholar] [CrossRef] [PubMed]
- Hollenstein, M. Nucleoside triphosphates—Building blocks for the modification of nucleic acids. Molecules 2012, 17, 13569–13591. [Google Scholar] [CrossRef] [PubMed]
- Fowler, J.D.; Suo, Z. Biochemical, structural, and physiological characterization of terminal deoxynucleotidyl transferase. Chem. Rev. 2006, 106, 2092–2110. [Google Scholar] [CrossRef] [PubMed]
- Motea, E.A.; Berdis, A.J. Terminal deoxynucleotidyl transferase: The story of a misguided DNA polymerase. Biochim. Biophys. Acta 2010, 1804, 1151–1166. [Google Scholar] [CrossRef] [PubMed]
- Arzumanov, A.A.; Victorova, L.S.; Jasko, M.V.; Yesipov, D.S.; Krayevsky, A.A. Terminal deoxynucleotidyl transferase catalyzes the reaction of DNA phosphorylation. Nucleic Acids Res. 2000, 28, 1276–1281. [Google Scholar] [CrossRef] [PubMed]
- Sosunov, V.V.; Santamaria, F.; Victorova, L.S.; Gosselin, G.; Rayner, B.; Krayevsky, A.A. Stereochemical control of DNA biosynthesis. Nucleic Acids Res. 2000, 28, 1170–1175. [Google Scholar] [CrossRef] [PubMed]
- Krayevsky, A.A.; Victorova, L.S.; Arzumanov, A.A.; Jasko, M.V. Terminal deoxynucleotidyl transferase: Catalysis of DNA (oligodeoxynucleotide) phosphorylation. Pharmacol. Ther. 2000, 85, 165–173. [Google Scholar] [CrossRef]
- Cho, Y.; Kool, E.T. Enzymatic synthesis of fluorescent oligomers assembled on a DNA backbone. ChemBioChem. 2006, 7, 669–672. [Google Scholar] [CrossRef] [PubMed]
- Hollenstein, M.; Wojciechowski, F.; Leumann, C.J. Polymerase incorporation of pyrene-nucleoside triphosphates. Bioorg. Med. Chem. Lett. 2012, 22, 4428–4430. [Google Scholar] [CrossRef] [PubMed]
- Jarchow-Choy, S.K.; Krueger, A.T.; Liu, H.; Gao, J.; Kool, E.T. Fluorescent xDNA nucleotides as efficient substrates for a template-independent polymerase. Nucleic Acids Res. 2011, 39, 1586–1594. [Google Scholar] [CrossRef] [PubMed]
- Kuwahara, M.; Obika, S.; Takeshima, H.; Hagiwara, Y.; Nagashima, J.I.; Ozaki, H.; Sawai, H.; Imanishi, T. Smart conferring of nuclease resistance to DNA by 3′-end protection using 2′,4′-bridged nucleoside-5′-triphosphates. Bioorg. Med. Chem. Lett. 2009, 19, 2941–2943. [Google Scholar] [CrossRef] [PubMed]
- Slavíčková, M.; Pohl, R.; Hocek, M. Additions of thiols to 7-vinyl-7-deazaadenine nucleosides and nucleotides. Synthesis of hydrophobic derivatives of 2′-deoxyadenosine, dATP and DNA. J. Org. Chem. 2016, 81, 11115–11125. [Google Scholar] [CrossRef] [PubMed]
- Winz, M.L.; Linder, E.C.; André, T.; Becker, J.; Jäschke, A. Nucleotidyl transferase assisted DNA labeling with different click chemistries. Nucleic Acids Res. 2015, 43, e110. [Google Scholar] [CrossRef] [PubMed]
- Delarue, M.; Boulé, J.B.; Lescar, J.; Expert-Bezançon, N.; Jourdan, N.; Sukumar, N.; Rougeon, F.; Papanicolaou, C. Crystal structures of a template-independent DNA polymerase: Murine terminal deoxynucleotidyltransferase. EMBO J. 2002, 21, 427–439. [Google Scholar] [CrossRef] [PubMed]
- Meisenheimer, K.M.; Koch, T.H. Photocross-linking of nucleic acids to associated proteins. Crit. Rev. Biochem. Mol. Biol. 1997, 32, 101–140. [Google Scholar] [CrossRef] [PubMed]
- Golden, M.C.; Collins, B.D.; Willis, M.C.; Koch, T.H. Diagnostic potential of PhotoSELEX-evolved ssDNA aptamers. J. Biotechnol. 2000, 81, 167–178. [Google Scholar] [CrossRef]
- Webb, C.F.; Jones, G.D.D.; Ward, J.F.; Moyer, D.J.; Aguilera, J.A.; Ling, L.L. Mechanisms of radiosensitization in bromodeoxyuridine-substituted cells. Int. J. Radiat. Biol. 1993, 64, 695–705. [Google Scholar] [CrossRef] [PubMed]
- Dextraze, M.-E.; Gantchev, T.; Girouard, S.; Hunting, D. DNA interstrand cross-links induced by ionizing radiation: An unsung lesion. Mutat. Res. 2010, 704, 101–107. [Google Scholar] [CrossRef] [PubMed]
- Limoli, C.L.; Ward, J.F. Response of bromodeoxyuridine-substituted Chinese hamster cells to UVA light exposure in the presence of Hoechst dye 33258: survival and DNA repair studies. Radiat. Res. 1994, 138, 312–319. [Google Scholar] [CrossRef] [PubMed]
- Brem, R.; Zhang, X.; Xu, Y.Z.; Karran, P. UVA photoactivation of DNA containing halogenated thiopyrimidines induces cytotoxic DNA lesions. J. Photochem. Photobiol. B 2015, 145, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Rudra, A.; Hou, D.; Zhang, Y.; Coulter, J.; Zhou, H.; DeWeese, T.L.; Greenberg, M.M. Bromopyridone nucleotide analogues, anoxic selective radiosensitizing agents that are incorporated in DNA by polymerases. J. Org. Chem. 2015, 80, 10675–10685. [Google Scholar] [CrossRef] [PubMed]
- Shaughnessy, K.H. Palladium-catalyzed modification of unprotected nucleosides, nucleotides, and oligonucleotides. Molecules 2015, 20, 9419–9454. [Google Scholar] [CrossRef] [PubMed]
- Favre, A.; Saintomé, C.; Fourrey, J.-L.; Clivio, P.; Laugâa, P. Thionucleobases as intrinsic photoaffinity probes of nucleic acid structure and nucleic acid-protein interactions. J. Photochem. Photobiol. B 1998, 42, 109–124. [Google Scholar] [CrossRef]
- Ascano, M.; Hafner, M.; Cekan, P.; Gerstberger, S.; Tuschl, T. Identification of RNA–protein interaction networks using PAR-CLIP. WIREs RNA 2012, 3, 159–177. [Google Scholar] [CrossRef] [PubMed]
- Guven, M.; Barnouin, K.; Snijders, A.P.; Karran, P. Photosensitized UVA-induced crosslinking between human DNA repair and replication proteins and DNA revealed by proteomic analysis. J. Proteome Res. 2016, 15, 4612–4623. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Xiong, X.; Yi, C. Epitranscriptome sequencing technologies: Decoding RNA modifications. Nat. Methods 2017, 1, 23–31. [Google Scholar] [CrossRef]
- Chiou, C.-H.; Lee, G.-B. A micromachined DNA manipulation platform for the stretching and rotation of a single DNA molecule. J. Micromech. Microeng. 2005, 15, 109–117. [Google Scholar] [CrossRef]
- Massey, A.; Xu, Y.Z.; Karran, P. Photoactivation of DNA thiobases as a potential novel therapeutic option. Curr. Biol. 2001, 11, 1142–1146. [Google Scholar] [CrossRef]
- Massey, A.; Xu, Y.Z.; Karran, P. Ambiguous coding is required for the lethal interaction between methylated DNA bases and DNA mismatch repair. DNA Repair 2002, 1, 275–286. [Google Scholar] [CrossRef]
- Xu, Y.Z.; Zhang, X.; Wu, H.C.; Massey, A. Karran, 4-Thio-5-bromo-20-deoxyuridine: Chemical synthesis and therapeutic potential of UVA-induced DNA damage. Bioorg. Med. Chem. Lett. 2004, 14, 995–997. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, S.S.; Kao, P.M.; Kwoh, D.Y. Synthesis of 5’ oligonucleotide hydrazide derivatives and their use in preparation of enzyme-nucleic acid hybridization probes. Anal. Biochem. 1989, 178, 43–51. [Google Scholar] [CrossRef]
- Grimm, G.N.; Boutorine, A.S.; Helene, C. Rapid routes of synthesis of oligonucleotide conjugates from non-protected oligonucleotides and ligands possessing different nucleophilic or electrophilic functional groups. Nucleosides Nucleotides Nucleic Acids 2000, 19, 1943–1965. [Google Scholar] [CrossRef] [PubMed]
- Raddatz, S.; Mueller-Ibeler, J.; Kluge, J.; Wäß, L.; Burdinski, G.; Havens, J.R.; Onofrey, T.J.; Wang, D.; Schweitzer, M. Hydrazide oligonucleotides: New chemical modification for chip array attachment and conjugation. Nucleic Acids Res. 2002, 30, 4793–4802. [Google Scholar] [CrossRef] [PubMed]
- Antsypovich, S.I.; von Kiedrowski, G. A novel versatile phosphoramidite building block for the synthesis of 5′- and 3′-hydrazide modified oligonucleotides. Nucleosides Nucleotides Nucleic Acids 2005, 24, 211–226. [Google Scholar] [PubMed]
- Zatsepin, T.S.; Gait, M.J.; Oretskaya, T.S.; Stetsenko, D.A. Synthesis of 2’-hydrazine oligonucleotides and their efficient conjugation with aldehydes and 1,3-diketones. Tetrahedron Lett. 2006, 47, 5515–5518. [Google Scholar] [CrossRef]
- Gamboa Varela, J.; Gates, K.S. A simple, high-yield synthesis of DNA duplexes containing a covalent, thermally cleavable interstrand cross-link at a defined location. Angew. Chem. 2015, 54, 7666–7669. [Google Scholar] [CrossRef] [PubMed]
- Negishi, K.; Takahashi, M.; Yamashita, Y.; Nishizawa, M.; Hayatsu, H. Mutagenesis by N4-aminocytidine: Induction of AT to GC transition and its molecular mechanism. Biochemistry 1985, 24, 7273–7278. [Google Scholar] [CrossRef] [PubMed]
- Stankevičiūtė, J.; Vaitekūnas, J.; Petkevičius, V.; Gasparavičiūtė, R.; Tauraitė, D.; Meškys, R. Oxyfunctionalization of pyridine derivatives using whole cells of Burkholderia sp. MAK1. Sci. Rep. 2016, 6, 39129. [Google Scholar] [CrossRef] [PubMed]
- McKay, C.S.; Finn, M.G. Click chemistry in complex mixtures: Bioorthogonal bioconjugation. Chem. Biol. 2014, 21, 1075–1101. [Google Scholar] [CrossRef] [PubMed]
- Tauraitė, D.; Dabužinskaite, J.; Ražanas, R.; Urbonavičius, J.; Stankevičiūtė, J.; Serva, S.; Meškys, R. Synthesis of novel derivatives of 5-carboxyuracil. Chemija 2015, 26, 120–125. [Google Scholar]
- Aldhoun, M.; Massi, A.; Dondoni, A. Click azide-nitrile cycloaddition as a new ligation tool for the synthesis of tetrazole-tethered C-glycosyl α-amino acids. J. Org. Chem. 2008, 73, 9565–9575. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Wang, J.; Xu, Y.-Z. Systematic assignment of NMR spectra of 5-substituted-4-thiopyrimidine nucleosides. Magn. Reson. Chem. 2013, 51, 523–529. [Google Scholar] [CrossRef] [PubMed]
- El-Tayeb, A.; Qi, A.; Nicholas, R.A.; Müller, C.E. Structural modifications of UMP, UDP, and UTP leading to subtype-selective agonists for P2Y2, P2Y4, and P2Y6 receptors. J. Med. Chem. 2011, 54, 2878–2890. [Google Scholar] [CrossRef] [PubMed]
- Lefever, M.; Kosmeder, J.W., II; Farrell, M.; Bieniarz, C. Microwave-mediated synthesis of labeled nucleotides with utility in the synthesis of DNA probes. Bioconjug. Chem. 2010, 21, 1773–1778. [Google Scholar] [CrossRef] [PubMed]
- Markley, J.C.; Chirakul, P.; Sologub, D.; Sigurdsson, S.T. Incorporation of 2’-deoxy-5-(trifluoromethyl)uridine and 5-cyano-2’-deoxyuridine into DNA. Bioorg. Med. Chem. Lett. 2001, 11, 2453–2455. [Google Scholar] [CrossRef]
- Berthod, T.; Pétillot, Y.; Guy, A.; Cadet, J.; Molko, D. Synthesis of oligonucleotides containing 5-carboxy-2′-deoxyuridine at defined sites. J. Org. Chem. 1996, 61, 6075–6078. [Google Scholar] [CrossRef]
- Guerniou, V.; Gasparutto, D.; Sauvaigo, S.; Favier, A.; Cadet, J. New synthesis of 5-carboxy-2’-deoxyuridine and its incorporation into synthetic oligonucleotides. Nucleosides Nucleotides Nucleic Acids 2003, 22, 1073–1075. [Google Scholar] [CrossRef] [PubMed]
- Johnson, D.; Morgan, A.R. The isolation of a high molecular weight terminal deoxynucleotidyl transferase from calf thymus. Biochem. Biophys. Res. Commun. 1976, 72, 840–849. [Google Scholar] [CrossRef]
- Chang, L.M.; Bollum, F.J. Multiple roles of divalent cation in the terminal deoxynucleotidyltransferase reaction. J. Biol. Chem. 1990, 265, 17436–17440. [Google Scholar] [PubMed]
- Chirpich, T.P. Factors affecting terminal deoxynucleotidyl transferase activity in cacodylate buffer. Biochem. Biophys. Res. Commun. 1977, 78, 1219–1226. [Google Scholar] [CrossRef]
- Gouge, J.; Rosario, S.; Romain, F.; Beguin, P.; Delarue, M. Structures of intermediates along the catalytic cycle of terminal deoxynucleotidyltransferase: Dynamical aspects of the two-metal ion mechanism. J. Mol. Biol. 2013, 425, 4334–4352. [Google Scholar] [CrossRef] [PubMed]
- Tauraitė, D.; Ražanas, R.; Mikalkėnas, A.; Serva, S.; Meškys, R. Synthesis of pyridone-based nucleoside analogues as substrates or inhibitors of DNA polymerases. Nucleosides Nucleotides Nucleic Acids 2016, 35, 163–177. [Google Scholar] [CrossRef] [PubMed]
- Takezawa, Y.; Kobayashi, T.; Shionoya, M. The effects of magnesium ions on the enzymatic synthesis of ligand-bearing artificial DNA by template-independent polymerase. Int. J. Mol. Sci. 2016, 17, 906. [Google Scholar] [CrossRef] [PubMed]
- Konig, J.; Zarnack, K.; Luscombe, N.M.; Ule, J. Protein–RNA interactions: New genomic technologies and perspectives. Nat. Rev. Genet. 2012, 13, 77–83. [Google Scholar] [CrossRef] [PubMed]
- Horáková, P.; Macíčková-Cahová, H.; Pivoňková, H.; Špaček, J.; Havran, L.; Hocek, M.; Fojta, M. Tail-labelling of DNA probes using modified deoxynucleotide triphosphates and terminal deoxynucleotidyl tranferase. Application in electrochemical DNA hybridization and protein-DNA binding assays. Org. Biomol. Chem. 2011, 9, 1366–1371. [Google Scholar] [CrossRef] [PubMed]
- Smith, C.L.; Simmonds, A.C.; Hamilton, A.L.; Martin, D.L.; Lashford, A.G.; Loakes, D.; Hill, F.; Brown, D.M. Use of 5-nitroindole-2′-deoxyribose-5′-triphosphate for labelling and detection of oligonucleotides. Nucleosides Nucleotides Nucleic Acids 1998, 17, 555–564. [Google Scholar] [CrossRef]
- Kobayashi, T.; Takezawa, Y.; Sakamoto, A.; Shionoya, M. Enzymatic synthesis of ligand-bearing DNAs for metal-mediated base pairing utilising a template-independent polymerase. Chem. Commun. 2016, 52, 3762–3765. [Google Scholar] [CrossRef] [PubMed]
- Loc’h, J.; Rosario, S.; Delarue, M. Structural basis for a new templated activity by terminal deoxynucleotidyl transferase: Implications for V(D)J recombination. Structure 2016, 24, 1452–1463. [Google Scholar] [CrossRef] [PubMed]
- Gong, S.-S.; Sun, J.; You, Y.-H.; Chen, J.-Z.; Liu, G.-D.; Sun, Q. Efficient synthesis of 5-carboxy-2′- deoxypyrimidine nucleoside 5′-triphosphates. Nucleosides Nucleotides Nucleic Acids 2016, 35, 295–304. [Google Scholar] [CrossRef] [PubMed]
- Sontheimer, E.J. Site-specific RNA crosslinking with 4-thiouridine. Mol. Biol. Rep. 1994, 20, 35–44. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Not available. |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tauraitė, D.; Jakubovska, J.; Dabužinskaitė, J.; Bratchikov, M.; Meškys, R. Modified Nucleotides as Substrates of Terminal Deoxynucleotidyl Transferase. Molecules 2017, 22, 672. https://doi.org/10.3390/molecules22040672
Tauraitė D, Jakubovska J, Dabužinskaitė J, Bratchikov M, Meškys R. Modified Nucleotides as Substrates of Terminal Deoxynucleotidyl Transferase. Molecules. 2017; 22(4):672. https://doi.org/10.3390/molecules22040672
Chicago/Turabian StyleTauraitė, Daiva, Jevgenija Jakubovska, Julija Dabužinskaitė, Maksim Bratchikov, and Rolandas Meškys. 2017. "Modified Nucleotides as Substrates of Terminal Deoxynucleotidyl Transferase" Molecules 22, no. 4: 672. https://doi.org/10.3390/molecules22040672
APA StyleTauraitė, D., Jakubovska, J., Dabužinskaitė, J., Bratchikov, M., & Meškys, R. (2017). Modified Nucleotides as Substrates of Terminal Deoxynucleotidyl Transferase. Molecules, 22(4), 672. https://doi.org/10.3390/molecules22040672





