Bergamot Essential Oil Attenuates Anxiety-Like Behaviour in Rats
Abstract
:1. Introduction
2. Results
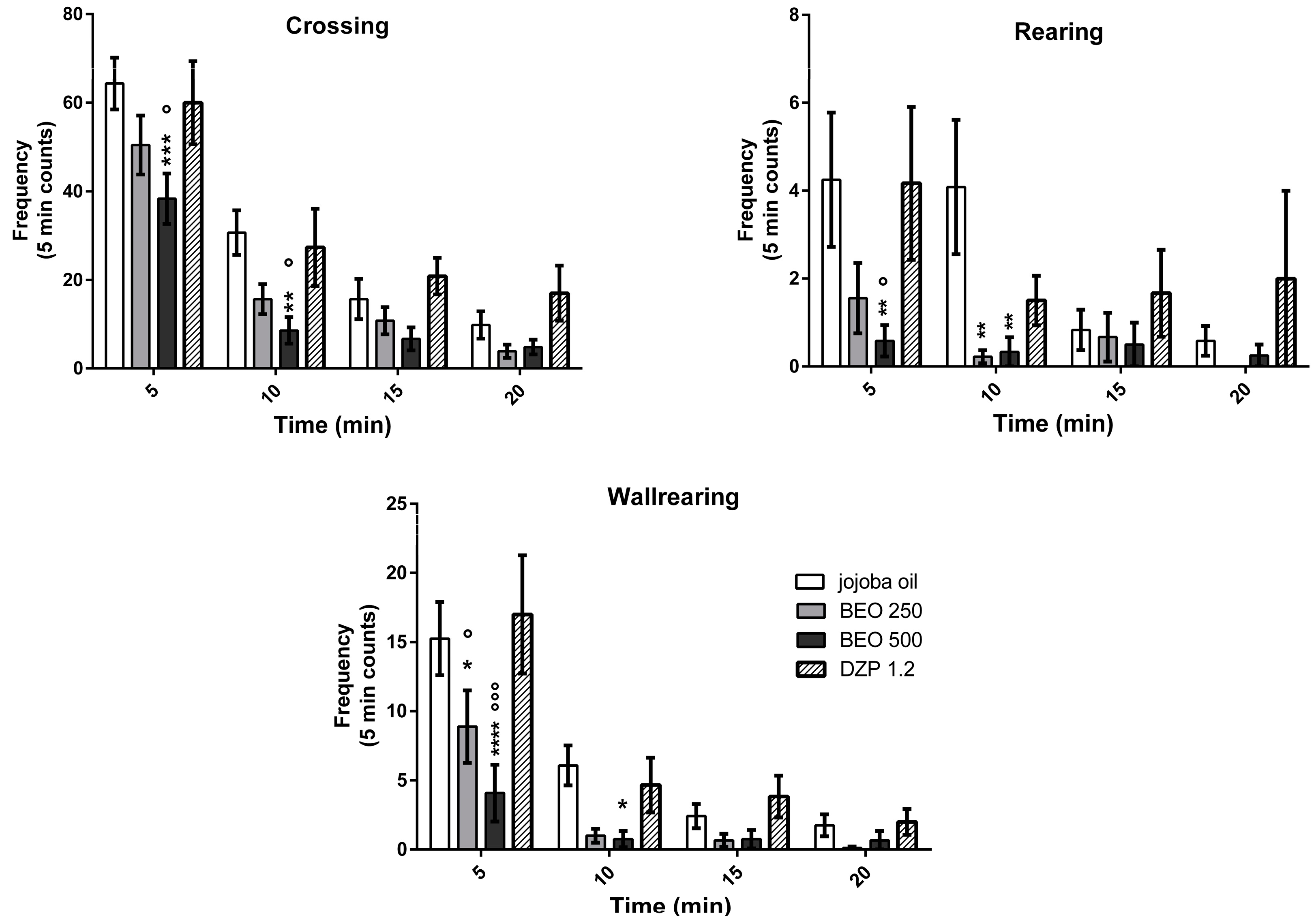
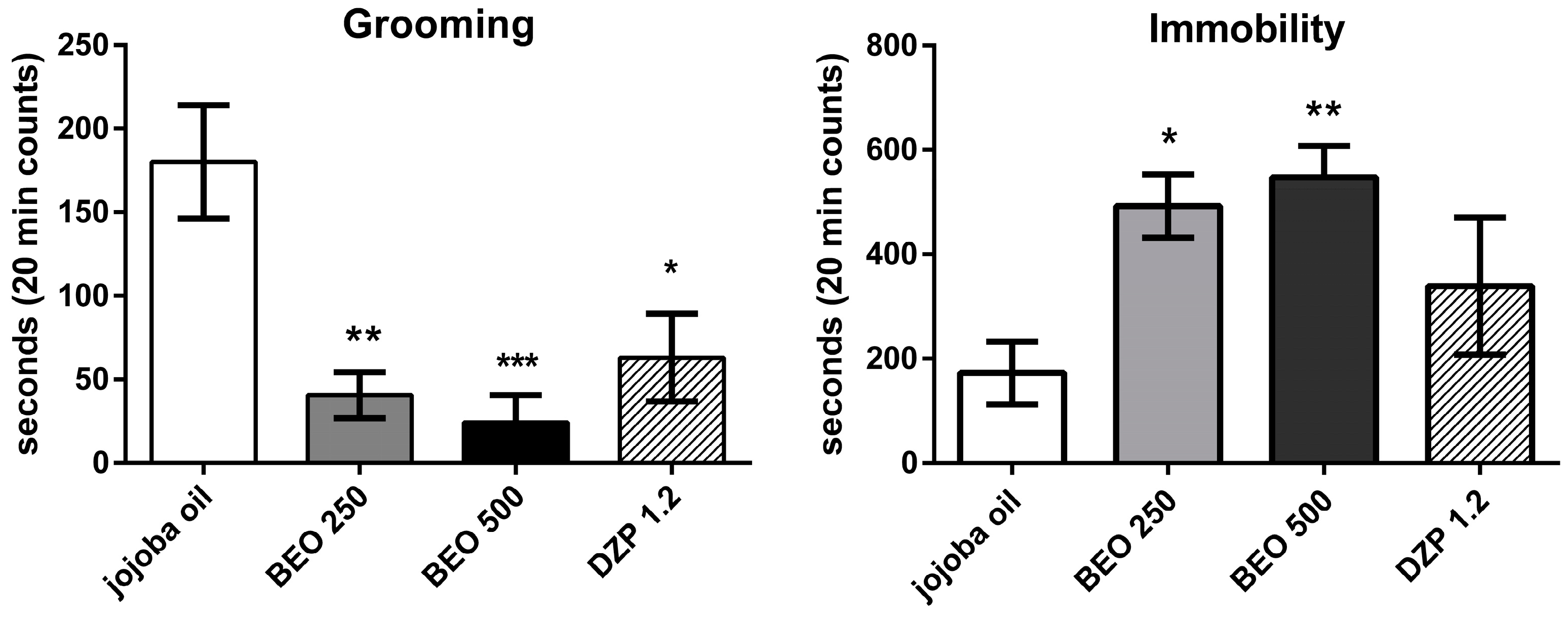
2.1. Effects of BEO on Open Field Test (OFT)
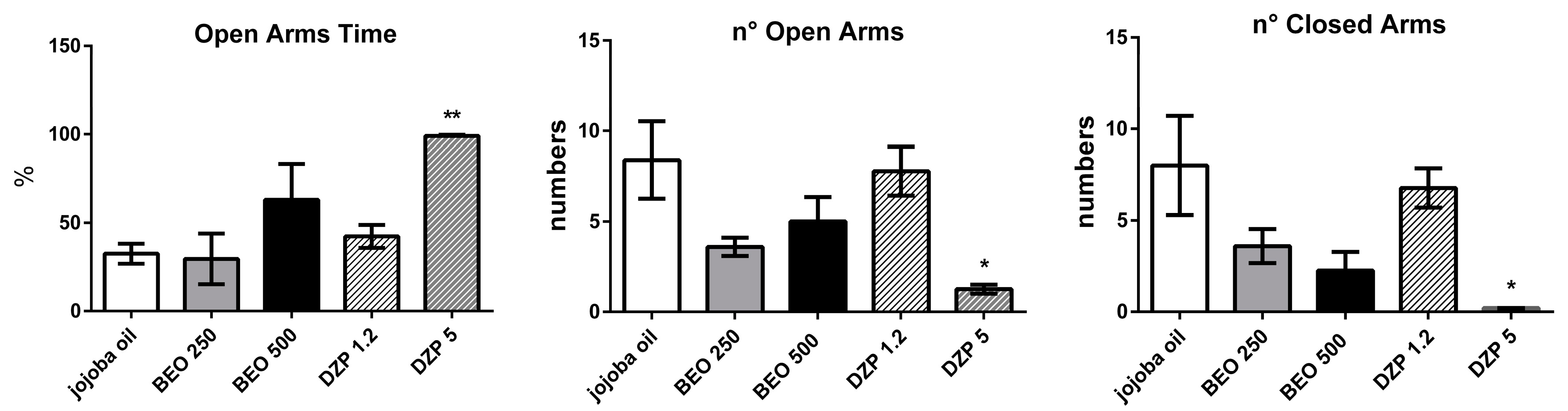
2.2. Effects of BEO on Elevated Plus-Maze Test (EPM)
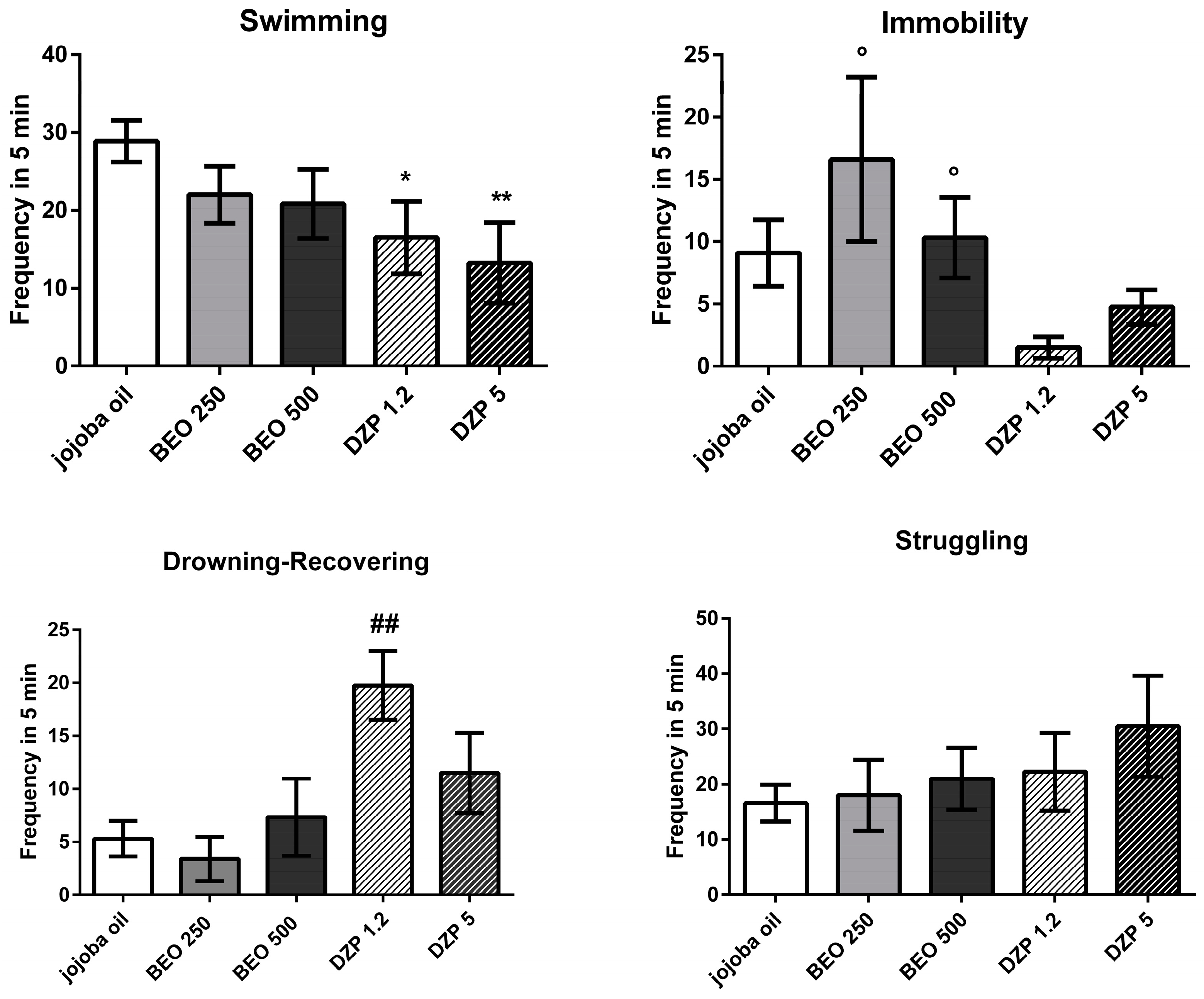
2.3. Effects of BEO on Forced Swimming Test (FST)
3. Discussion
4. Materials and Methods
4.1. Animals
4.2. Bergamot Essential Oil
4.3. Experimental Procedure
4.4. Open Field Test
4.5. Elevated Plus Maze Test
4.6. Forced Swimming Test
4.7. Statistics
5. Conclusions
Acknowledgments
Authors Contributions
Conflicts of Interest
References
- Moufida, S.; Marzouk, B. Biochemical characterization of blood orange, sweet orange, lemon, bergamot and bitter orange. Phytochemistry 2003, 62, 1283–1289. [Google Scholar] [CrossRef]
- Costa, R.; Dugo, P.; Navarra, M.; Raymo, V.; Dugo, G.; Mondelloa, L. Study on the chemical composition variability of some processed bergamot (Citrus bergamia) essential oils. Flavour Fragr. J. 2010, 25, 4–12. [Google Scholar] [CrossRef]
- Donato, P.; Bonaccorsi, I.; Russo, M.; Dugo, P. Determination of new bioflavonoids in bergamot (Citrus bergamia) peel oil by liquid chromatography coupled to tandem ion trap–time-of-flight mass spectrometry. Flavour Fragr. J. 2014, 29, 131–136. [Google Scholar] [CrossRef]
- Melliou, E.; Michaelakis, A.; Koliopoulos, G.; Skaltsounis, A.L.; Magiatis, P. High quality bergamot oil from Greece: Chemical analysis using chiral gas chromatography and larvicidal activity against the West Nile virus vector. Molecules 2009, 14, 839–849. [Google Scholar] [PubMed]
- Laird, K.; Armitage, D.; Phillips, C. Reduction of surface contamination and biofilms of Enterococcus sp. and Staphylococcus aureus using a citrus-based vapour. J. Hosp. Infect. 2012, 80, 61–66. [Google Scholar] [CrossRef] [PubMed]
- Sanguinetti, M.; Posteraro, B.; Romano, L.; Battaglia, F.; Lopizzo, T.; de Caroli, E.; Fadda, G. In vitro activity of Citrus bergamia (bergamot) oil against clinical isolates of dermatophytes. J. Antimicrob. Chemother. 2007, 59, 305–308. [Google Scholar] [CrossRef] [PubMed]
- Cosentino, M.; Luini, A.; Bombelli, R.; Corasaniti, M.T.; Bagetta, G.; Marino, F. The essential oil of bergamot stimulates reactive oxygen species production in human polymorphonuclear leukocytes. Phytother. Res. 2014, 28, 232–239. [Google Scholar] [CrossRef] [PubMed]
- Bagetta, G.; Morrone, L.A.; Rombolà, L.; Amantea, D.; Russo, R.; Berliocchi, L.; Sakurad, S.; Sakurada, T.; Rotiroti, D.; Corasaniti, M.T. Neuropharmacology of the essential oil of bergamot. Fitoterapia 2010, 81, 453–461. [Google Scholar] [CrossRef] [PubMed]
- Navarra, M.; Mannucci, C.; Delbò, M.; Calapai, G. Citrus bergamia essential oil: From basic research to clinical application. Front. Pharmacol. 2015, 6, 36. [Google Scholar] [CrossRef] [PubMed]
- Morrone, L.A.; Rombola, L.; Pelle, C.; Corasaniti, M.T.; Zappettini, S.; Paudice, P.; Bonanno, G.; Bagetta, G. The essential oil of bergamot enhances the levels of amino acid neurotransmitters in the hippocampus of rat: Implication of monoterpene hydrocarbons. Pharmacol. Res. 2007, 55, 255–262. [Google Scholar] [CrossRef] [PubMed]
- Rombolà, L.; Corasaniti, M.T.; Rotiroti, D.; Tassorelli, C.; Sakurada, S.; Bagetta, G.; Morrone, L.A. Effects of systemic administration of the essential oil of bergamot (BEO) on gross behaviour and EEG power spectra recorded from the rat hippocampus and cerebral cortex. Funct. Neurol. 2009, 24, 107–112. [Google Scholar] [PubMed]
- Amantea, D.; Fratto, V.; Maida, S.; Rotiroti, D.; Ragusa, S.; Nappi, G.; Bagetta, G.; Corasaniti, M.T. Prevention of Glutamate Accumulation and Upregulation of Phospho-Akt may Account for Neuroprotection Afforded by Bergamot Essential Oil against Brain Injury Induced by Focal Cerebral Ischemia in Rat. Int. Rev. Neurobiol. 2009, 85, 389–405. [Google Scholar] [PubMed]
- Sakurada, T.; Kuwahata, H.; Katsuyama, S.; Komatsu, T.; Morrone, L.A.; Corasaniti, M.T.; Bagetta, G.; Sakurada, S. Intraplantar injection of bergamot essential oil into the mouse hindpaw: Effects on capsaicin-induced nociceptive behaviors. Int. Rev. Neurobiol. 2009, 85, 237–248. [Google Scholar] [PubMed]
- Berliocchi, L.; Russo, R.; Levato, A.; Fratto, V.; Bagetta, G.; Sakurada, S.; Sakurada, T.; Mercuri, N.B.; Corasaniti, M.T. (−)-Linalool attenuates allodynia in neuropathic pain induced by spinal nerve ligation in c57/bl6 mice. Int. Rev. Neurobiol. 2009, 85, 221–235. [Google Scholar] [PubMed]
- Sakurada, T.; Mizoguchi, H.; Kuwahata, H.; Katsuyama, S.; Komatsu, T.; Morrone, L.A.; Corasaniti, M.T.; Bagetta, G.; Sakurada, S. Intraplantar injection of bergamot essential oil induces peripheral antinociception mediated by opioid mechanism. Pharmacol. Biochem. Behav. 2011, 97, 436–443. [Google Scholar] [CrossRef] [PubMed]
- Kuwahata, H.; Komatsu, T.; Katsuyama, S.; Corasaniti, M.T.; Bagetta, G.; Sakurada, S.; Sakurada, T.; Takahama, K. Peripherally injected linalool and bergamot essential oil attenuate mechanical allodynia via inhibiting spinal ERK phosphorylation. Pharmacol. Biochem. Behav. 2013, 103, 735–741. [Google Scholar] [CrossRef] [PubMed]
- Katsuyama, S.; Otowa, A.; Kamio, S.; Sato, K.; Yagi, T.; Kishikawa, Y.; Komatsu, T.; Bagetta, G.; Sakurada, T.; Nakamura, H. Effect of plantar subcutaneous administration of bergamot essential oil and linalool on formalin-induced nociceptive behavior in mice. Biomed. Res. 2015, 36, 47–54. [Google Scholar] [CrossRef] [PubMed]
- Rombolà, L.; Amantea, D.; Russo, R.; Adornetto, A.; Berliocchi, L.; Tridico, L.; Corasaniti, M.T.; Sakurada, S.; Sakurada, T.; Bagetta, G.; et al. Rational Basis for the Use of Bergamot Essential Oil in Complementary Medicine to Treat Chronic Pain. Mini Rev. Med. Chem. 2016, 16, 721–728. [Google Scholar] [CrossRef] [PubMed]
- Corasaniti, M.T.; Maiuolo, J.; Maida, S.; Fratto, V.; Navarra, M.; Russo, R.; Amantea, D.; Morrone, L.A.; Bagetta, G. Cell signaling pathways in the mechanisms of neuroprotection afforded by bergamot essential oil against MDA-induced cell death in vitro. Br. J. Pharmacol. 2007, 151, 518–529. [Google Scholar] [CrossRef] [PubMed]
- Berliocchi, L.; Ciociaro, A.; Russo, R.; Cassiano, M.G.; Blandini, F.; Rotiroti, D.; Morrone, L.A.; Corasaniti, M.T. Toxic profile of bergamot essential oil on survival and proliferation of SH-SY5Y neuroblastoma cells. Food Chem. Toxicol. 2011, 49, 2780–2792. [Google Scholar] [CrossRef] [PubMed]
- Russo, R.; Ciociaro, A.; Berliocchi, L.; Cassiano, M.G.; Rombolà, L.; Ragusa, S.; Bagetta, G.; Blandini, F.; Corasaniti, M.T. Implication of limonene and linalyl acetate in cytotoxicity induced by bergamot essential oil in human neuroblastoma cells. Fitoterapia 2013, 89, 48–57. [Google Scholar] [CrossRef] [PubMed]
- Russo, R.; Cassiano, M.G.; Ciociaro, A.; Adornetto, A.; Varano, G.P.; Chiappini, C.; Berliocchi, L.; Tassorelli, C.; Bagetta, G.; Corasaniti, M.T. Role of D-Limonene in autophagy induced by bergamot essential oil in SH-SY5Y neuroblastoma cells. PLoS ONE 2014, 9, e113682. [Google Scholar] [CrossRef] [PubMed]
- Jafarzadeh, M.; Arman, S.; Pour, F.F. Effect of aromatherapy with orange essential oil on salivary cortisol and pulse rate in children during dental treatment: A randomized controlled clinical trial. Adv. Biomed. Res. 2013, 2. [Google Scholar] [CrossRef]
- Kasper, S.; Volz, H.P.; Dienel, A.; Schläfke, S. Efficacy of Silexan in mixed anxiety-depression—A randomized, placebo-controlled trial. Eur. Neuropsychopharmacol. 2016, 26, 331–340. [Google Scholar] [CrossRef] [PubMed]
- Ni, C.H.; Hou, W.H.; Kao, C.C.; Chang, M.L.; Yu, L.F.; Wu, C.C.; Chen, C. The anxiolytic effect of aromatherapy on patients awaiting ambulatory surgery, a randomized controlled trial. Evid. Based Complement. Altern. Med. 2013, 2013. [Google Scholar] [CrossRef] [PubMed]
- Saiyudthong, S.; Marsden, C.A. Acute effects of bergamot oil on anxiety-related behaviour and corticosterone level in rats. Phytother. Res. 2011, 25, 858–862. [Google Scholar] [CrossRef] [PubMed]
- Saiyudthong, S.; Mekseepralard, C. Effect of Inhaling Bergamot Oil on Depression-Related Behaviors in Chronic Stressed Rats. Evid. Based Complement. Altern. Med. 2015, 9, S152–S159. [Google Scholar]
- Walsh, R.N.; Cummins, R.A. The Open-Field Test: A critical review. Psychol. Bull. 1976, 83, 482–504. [Google Scholar] [CrossRef] [PubMed]
- Pellow, S.; Chopin, P.; File, S.E.; Briley, M. Validation of open closed arm entries in an elevated plus-maze as a measure of anxiety in the rat. J. Neurosci. Meth. 1985, 14, 149–167. [Google Scholar] [CrossRef]
- Porsolt, R.D.; Bertin, A.; Jalfre, M. “Behavioural despair” in rats and mice: Strain differences and the effects of imipramine. Eur. J. Pharmacol. 1978, 51, 291–294. [Google Scholar] [CrossRef]
- Lim, W.C.; Seo, J.M.; Lee, C.I.; Pyo, H.B.; Lee, B.C. Stimulative and sedative effects of essential oils upon inhalation in mice. Arch. Pharm. Res. 2005, 28, 770–774. [Google Scholar] [CrossRef] [PubMed]
- Shaw, D.; Annett, J.M.; Doherty, B.; Leslie, J.C. Anxiolytic effects of lavander oil inhalation on open-field behaviour in rats. Phytomedicine 2007, 4, 613–620. [Google Scholar] [CrossRef] [PubMed]
- Chioca, L.R.; Antunes, V.D.; Ferro, M.M.; Losso, E.M.; Andreatini, R. Anosmia does not impair the anxiolytic-like effect of lavender essential oil inhalation in mice. Life Sci. 2013, 92, 971–975. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, M.; Yamanaka, A.; Asanuma, C.; Asano, H.; Satou, T.; Koike, K. Anxiolytic-like effect of inhalation of essential oil from Lavandula officinalis: Investigation of changes in 5-HT turnover and involvement of olfactory stimulation. Nat. Prod. Commun. 2014, 9, 1023–1026. [Google Scholar] [PubMed]
- Komiya, M.; Takeuchi, T.; Harada, E. Lemon oil vapor causes an anti-stress effect via modulating the 5-HT and DA activities in mice. Behav. Brain Res. 2006, 172, 240–249. [Google Scholar] [CrossRef] [PubMed]
- Faturi, C.B.; Leite, J.R.; Alves, P.B.; Canton, A.C.; Teixeira-Silva, F. Anxiolytic-like effect of sweet orange aroma in Wistar rats. Prog. Neuropsychopharmacol. Biol. Psychiatry 2010, 34, 605–609. [Google Scholar] [CrossRef] [PubMed]
- Pultrini Ade, M.; Galindo, L.A.; Costa, M. Effects of the essential oil from Citrus aurantium L. in experimental anxiety models in mice. Life Sci. 2006, 78, 1720–1725. [Google Scholar] [CrossRef] [PubMed]
- Kalueff, A.V.; Tuohimaa, P. Mouse grooming microstructure is a reliable anxiety marker bidirectionally sensitive to GABAergic drugs. Eur. J. Pharmacol. 2005, 508, 147–153. [Google Scholar] [CrossRef] [PubMed]
- Moody, T.W.; Merali, Z.; Crawley, J.N. The effects of anxiolytics and other agents on rat grooming behavior. Ann. N. Y. Acad. Sci. 1993, 90, 281–290. [Google Scholar] [CrossRef]
- Choleris, E.; Thomas, A.W.; Kavaliers, M.; Prato, F.S. A detailed ethological analysis of the mouse open field test: Effects of diazepam, chlordiazepoxide and an extremely low frequency pulsed magnetic field. Neurosci. Biobehav. Rev. 2001, 25, 235–260. [Google Scholar] [CrossRef]
- Detke, M.J.; Lucki, I. Detection of serotonergic and noradrenergic antidepressants in the rat forced swimming test: The effects of water depth. Behav. Brain Res. 1996, 73, 43–46. [Google Scholar] [CrossRef]
- Calil, C.M.; Marcondes, F.K. The comparison of immobility time in experimental rat swimming models. Life Sci. 2006, 79, 1712–1719. [Google Scholar] [CrossRef] [PubMed]
- Binik, Y.M.; Sullivan, M.J.L. Sudden swimming deaths: A psychomotor reinterpretation. Psychophysiology 1983, 20, 670–681. [Google Scholar] [CrossRef] [PubMed]
- Vanderwolf, C.H. Hippocampal electrical activity and voluntary movement in the rat. Electroencephalogr. Clin. Neurophysiol. 1969, 26, 407–418. [Google Scholar] [CrossRef]
- Van Lier, H.; Drinkenburg, W.H.; van Eeten, Y.J.; Coenen, A.M. Effects of diazepam and zolpidem on EEG beta frequencies are behavior-specific in rats. Neuropharmacology 2004, 47, 163–174. [Google Scholar] [CrossRef] [PubMed]
- Kopp, C.; Rudolph, U.; Löw, K.; Tobler, I. Modulation of rhythmic brain activity by diazepam: GABA(A) receptor subtype and state specificity. Proc. Natl. Acad. Sci. USA 2004, 101, 3674–3679. [Google Scholar] [CrossRef] [PubMed]
- Siok, C.J.; Cogan, S.M.; Shifflett, L.B.; Doran, A.C.; Kocsis, B.; Hajós, M. Comparative analysis of the neurophysiological profile of group II metabotropic glutamate receptor activators and diazepam: Effects on hippocampal and cortical EEG patterns in rats. Neuropharmacology 2012, 62, 226–236. [Google Scholar] [CrossRef] [PubMed]
- Chioca, L.R.; Ferro, M.M.; Baretta, I.P.; Oliveira, S.M.; Silva, C.R.; Ferreira, J.; Losso, E.M.; Andreatini, R. Anxiolytic-like effect of lavender essential oil inhalation in mice: Participation of serotonergic but not GABAA/benzodiazepine neurotransmission. J. Ethnopharmacol. 2013, 147, 412–418. [Google Scholar] [CrossRef] [PubMed]
- Umezu, T.; Nagano, K.; Ito, H.; Kosakai, K.; Sakaniwa, M.; Morita, M. Anticonflict effects of lavender oil and identification of its active constituents. Pharmacol. Biochem. Behav. 2006, 85, 713–721. [Google Scholar] [CrossRef] [PubMed]
- Linck, V.M.; da Silva, A.L.; Figueiró, M.; Caramão, E.B.; Moreno, P.R.; Elisabetsky, E. Effects of inhaled Linalool in anxiety, social interaction and aggressive behavior in mice. Phytomedicine 2010, 17, 679–683. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, M.; Satou, T.; Ohashi, M.; Hayashi, S.; Sadamoto, K.; Koike, K. Interspecies comparison of chemical composition and anxiolytic-like effects of lavender oils upon inhalation. Nat. Prod. Commun. 2011, 6, 1769–1774. [Google Scholar] [PubMed]
- Tampi, R.R.; Tampi, D.J. Efficacy and Tolerability of Benzodiazepines for the Treatment of Behavioral and Psychological Symptoms of Dementia: A Systematic Review of Randomized Controlled Trials. Am. J. Alzheimer Dis. Other Dement. 2014, 29, 565–574. [Google Scholar] [CrossRef] [PubMed]
- Wells, R.E.; Phillips, R.S.; Schachter, S.C.; McCarthy, E.P. Complementary and alternative medicine use among US adults with common neurological conditions. J. Neurol. 2010, 257, 1822–1831. [Google Scholar] [CrossRef] [PubMed]
- Crawford, C.; Lee, C.; Buckenmaier, C.; Schoomaker, E.; Petri, R.; Jonas, W. Active Self-Care Therapies for Pain (PACT) Working Group. The current state of the science for active self-care complementary and integrative medicine therapies in the management of chronic pain symptoms: Lessons learned, directions for the future. Pain Med. 2014, 15 (Suppl. 1), S104–S113. [Google Scholar] [CrossRef] [PubMed]
- Purohit, M.P.; Zafonte, R.D.; Sherman, L.M.; Davis, R.B.; Giwerc, M.Y.; Shenton, M.E.; Yeh, G.Y. Neuropsychiatric symptoms and expenditure on complementary and alternative medicine. J. Clin. Psychiatry 2015, 76, 870–876. [Google Scholar] [CrossRef] [PubMed]
- Defrancesco, M.; Marksteiner, J.; Fleischhackeret, W.W.; Blasko, I. Use of Benzodiazepines in Alzheimer’s Disease: A Systematic Review of Literature. Int. J. Neuropsychopharmacol. 2015, 18. [Google Scholar] [CrossRef] [PubMed]
- Forrester, L.T.; Maayan, N.; Orrell, M.; Spector, A.E.; Buchan, L.D.; Soares-Weiser, K. Aromatherapy for dementia. Cochrane Database Syst. Rev. 2014, 2, CD003150. [Google Scholar]
- Ballard, C.G.; Gauthier, S.; Cummings, J.L.; Brodaty, H.; Grosberg, G.T.; Robert, P.; Lyketsos, C.G. Management of agitation and aggression associated with Alzheimer disease. Nat. Rev. Neurol. 2009, 5, 245–255. [Google Scholar] [CrossRef] [PubMed]
- Corbett, A.; Husebo, B.; Malcangio, M.; Staniland, A.; Cohen-Mansfield, J.; Aarsland, D.; Ballard, C. Assessment and treatment of pain in people with dementia. Nat. Rev. Neurol. 2012, 8, 264–274. [Google Scholar] [CrossRef] [PubMed]
- Husebo, B.S.; Achterberg, W.; Flo, E. Identifying and Managing Pain in People with Alzheimer’s Disease and Other Types of Dementia: A Systematic Review. CNS Drugs 2016, 30, 481–497. [Google Scholar] [CrossRef] [PubMed]
- Prut, L.; Belzung, C. The open field as a paradigm to measure the effects of drugs on anxiety-like behaviors: A review. Eur. J. Pharmacol. 2003, 463, 3–33. [Google Scholar] [CrossRef]
Sample Availability: Not available. |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rombolà, L.; Tridico, L.; Scuteri, D.; Sakurada, T.; Sakurada, S.; Mizoguchi, H.; Avato, P.; Corasaniti, M.T.; Bagetta, G.; Morrone, L.A. Bergamot Essential Oil Attenuates Anxiety-Like Behaviour in Rats. Molecules 2017, 22, 614. https://doi.org/10.3390/molecules22040614
Rombolà L, Tridico L, Scuteri D, Sakurada T, Sakurada S, Mizoguchi H, Avato P, Corasaniti MT, Bagetta G, Morrone LA. Bergamot Essential Oil Attenuates Anxiety-Like Behaviour in Rats. Molecules. 2017; 22(4):614. https://doi.org/10.3390/molecules22040614
Chicago/Turabian StyleRombolà, Laura, Laura Tridico, Damiana Scuteri, Tsukasa Sakurada, Shinobu Sakurada, Hirokazu Mizoguchi, Pinarosa Avato, Maria Tiziana Corasaniti, Giacinto Bagetta, and Luigi Antonio Morrone. 2017. "Bergamot Essential Oil Attenuates Anxiety-Like Behaviour in Rats" Molecules 22, no. 4: 614. https://doi.org/10.3390/molecules22040614
APA StyleRombolà, L., Tridico, L., Scuteri, D., Sakurada, T., Sakurada, S., Mizoguchi, H., Avato, P., Corasaniti, M. T., Bagetta, G., & Morrone, L. A. (2017). Bergamot Essential Oil Attenuates Anxiety-Like Behaviour in Rats. Molecules, 22(4), 614. https://doi.org/10.3390/molecules22040614







