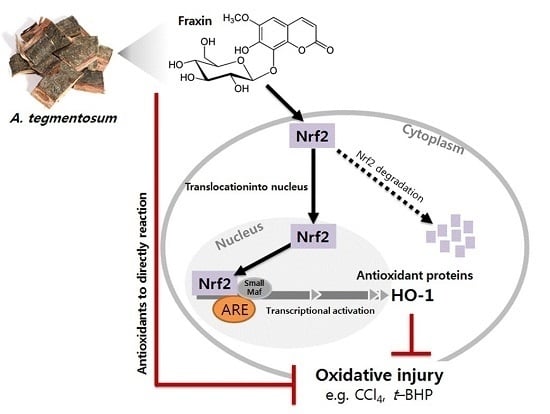
Fraxin Prevents Chemically Induced Hepatotoxicity by Reducing Oxidative Stress
Abstract
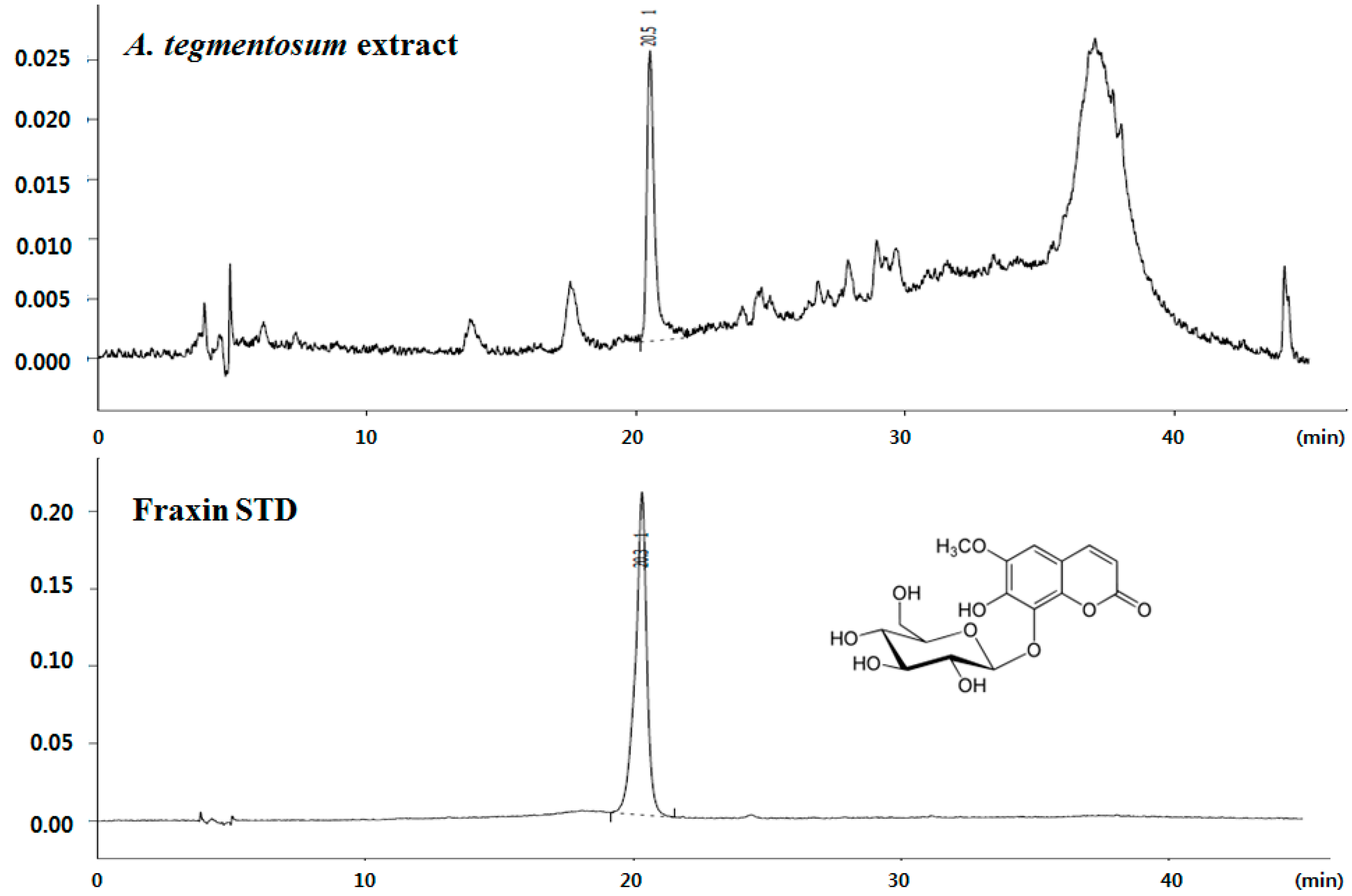
:1. Introduction
2. Materials and Methods
2.1. Experimental Animals and Design
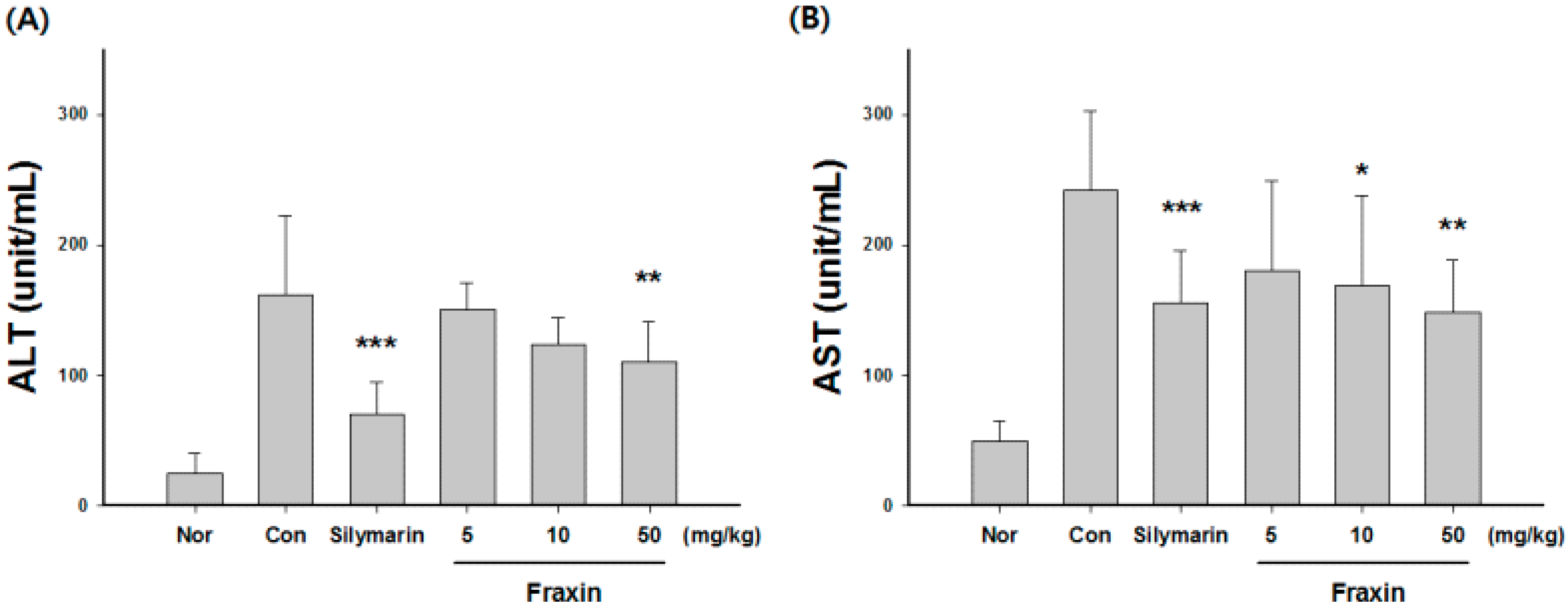
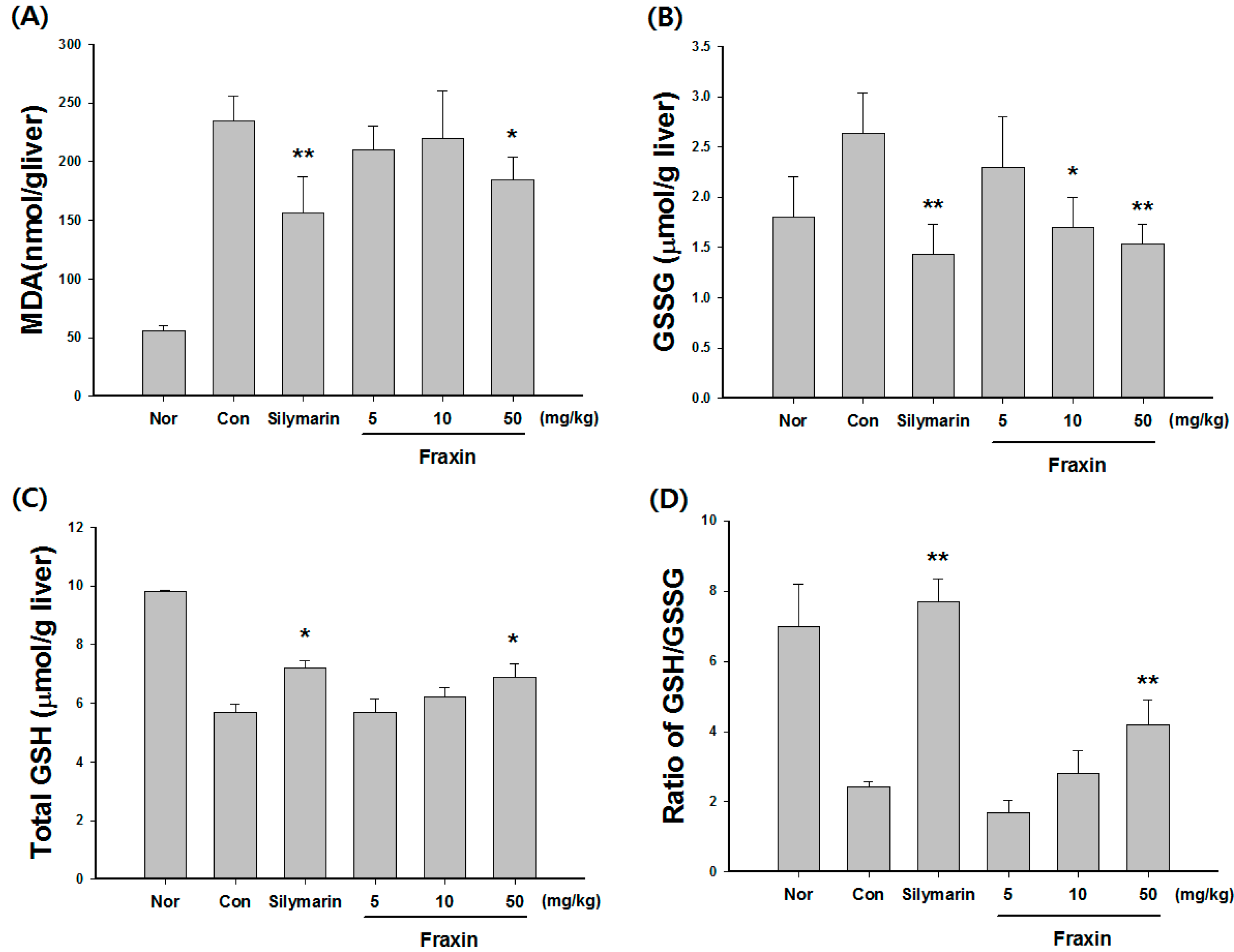
2.2. Estimation of Biochemical Parameters
2.3. Histopathological Procedure
2.4. Cell Culture and Viability Assay
2.5. Measurement of ROS Generation
2.6. ARE Luciferase Assay
2.7. Nuclear Factor Erythroid-Derived 2-Related Factor 2 (Nrf2), Heme Oxygenase-1 (HO-1) Protein Expression Analysis
2.8. HO-1 Gene Expression Analysis
2.9. Statistical Analysis
3. Results
3.1. Protective Effect of Fraxin against CCl4-Induced Hepatic Damage
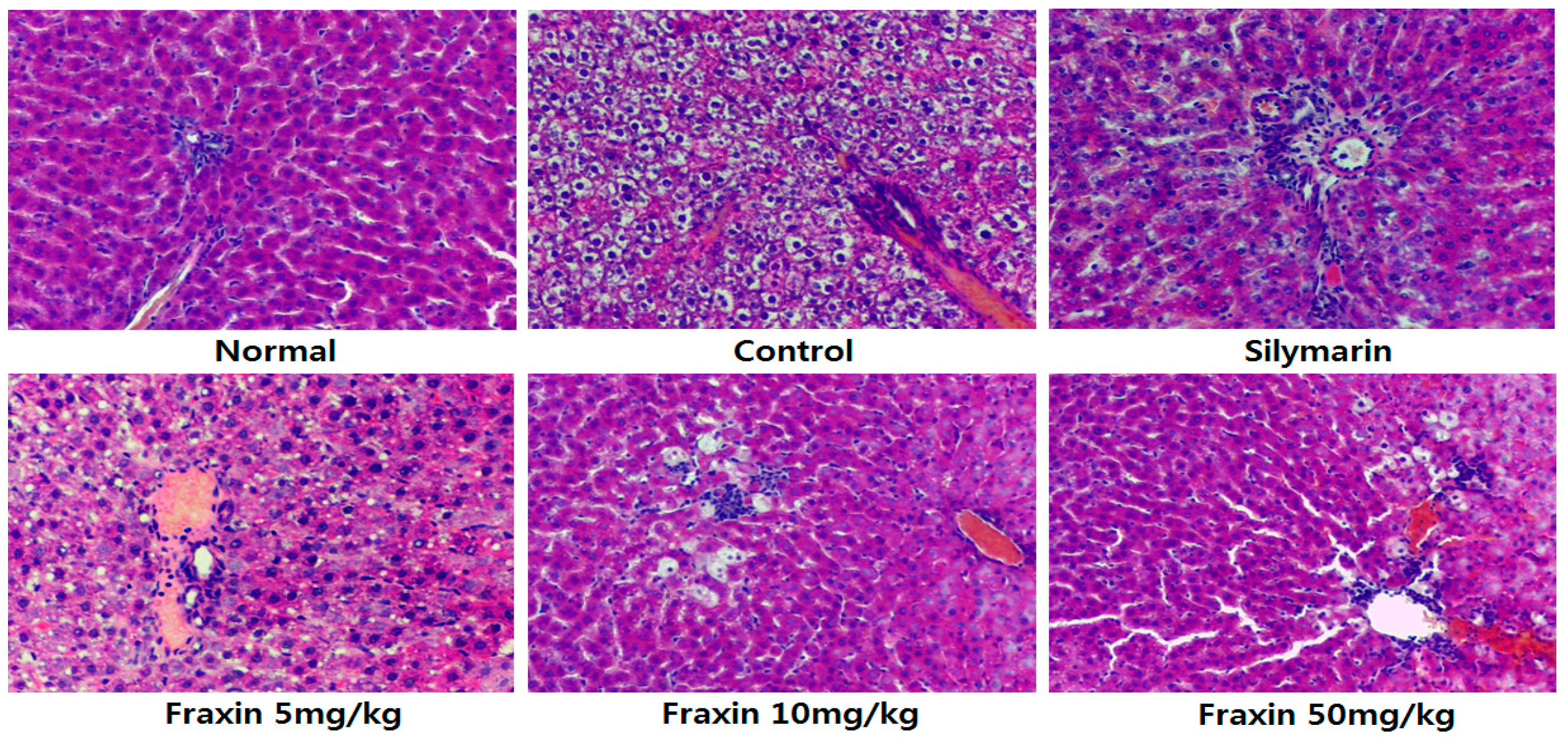
3.2. Histopathology of Hepatic Damage
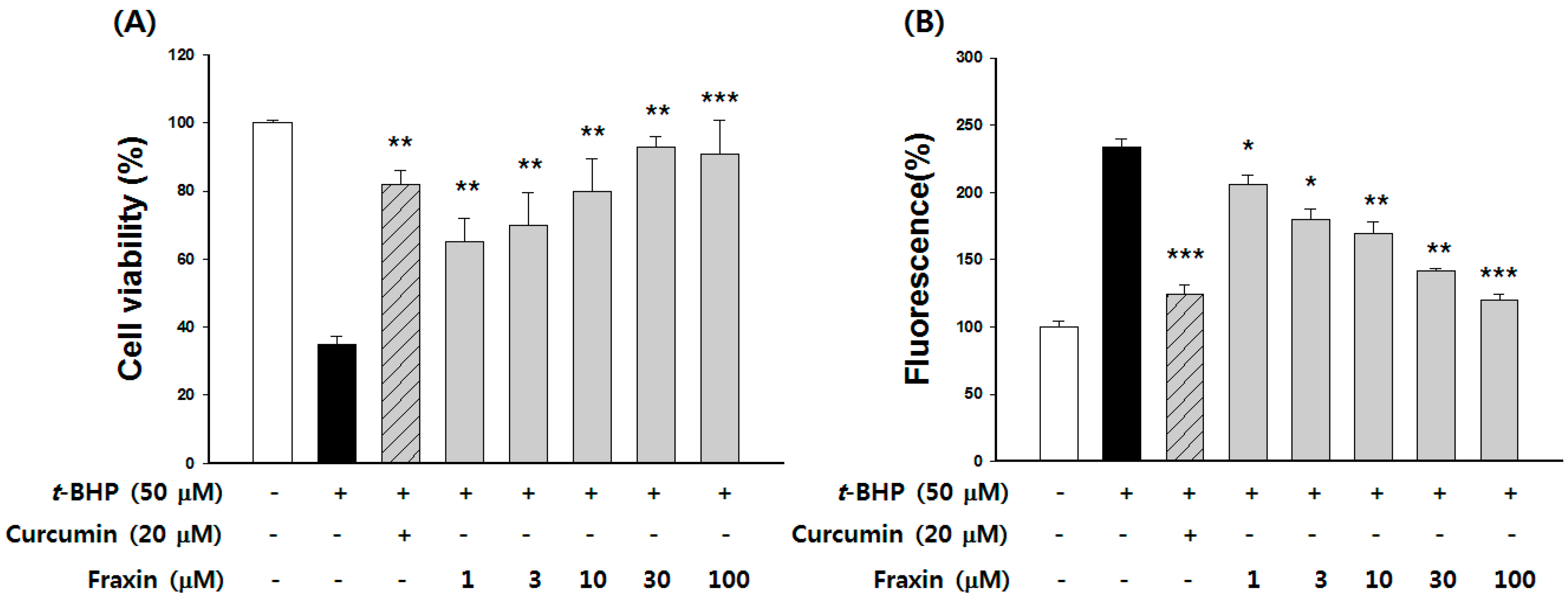
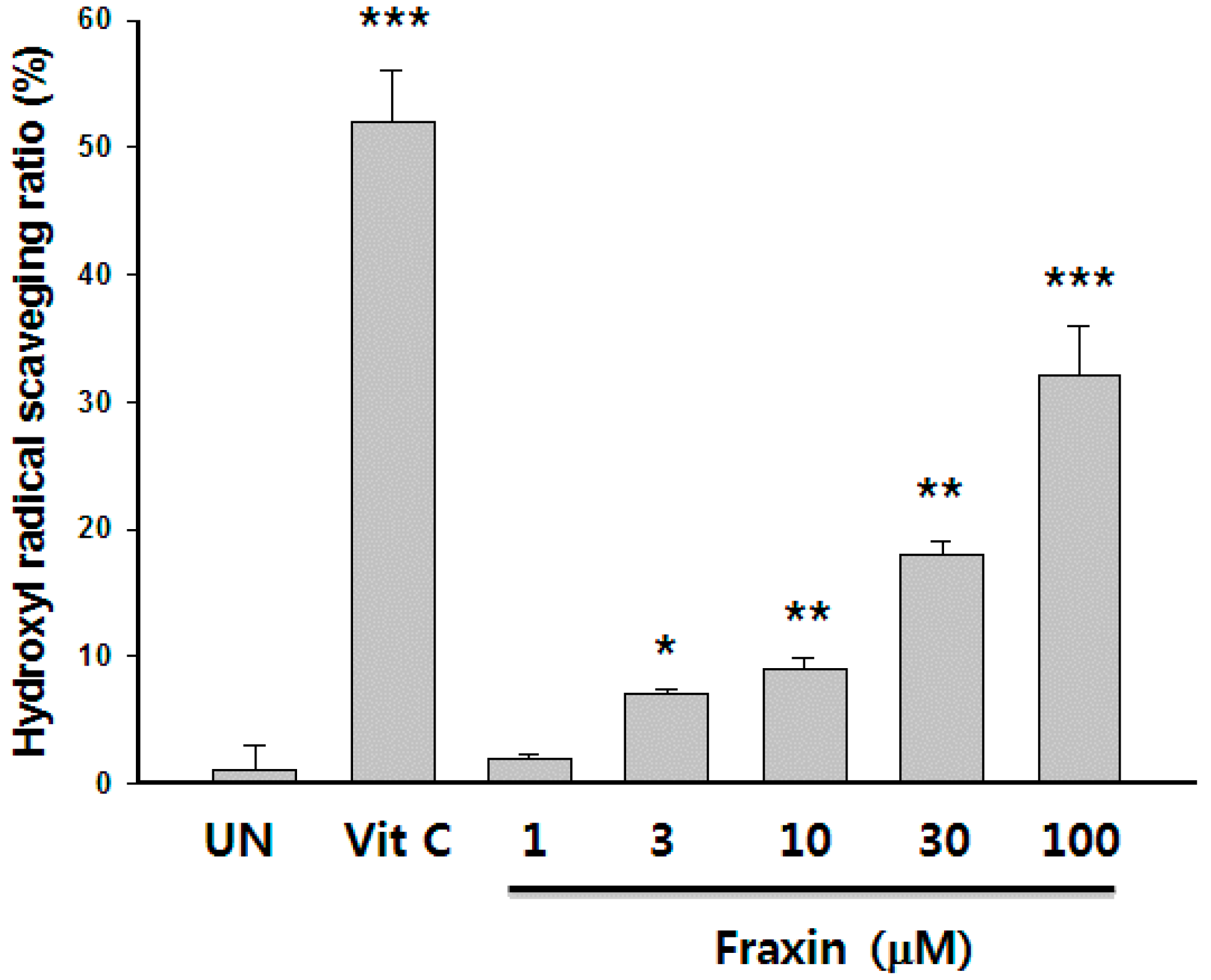
3.3. Protective Effect of Fraxin against t-BHP-Induced Hepatotoxicity
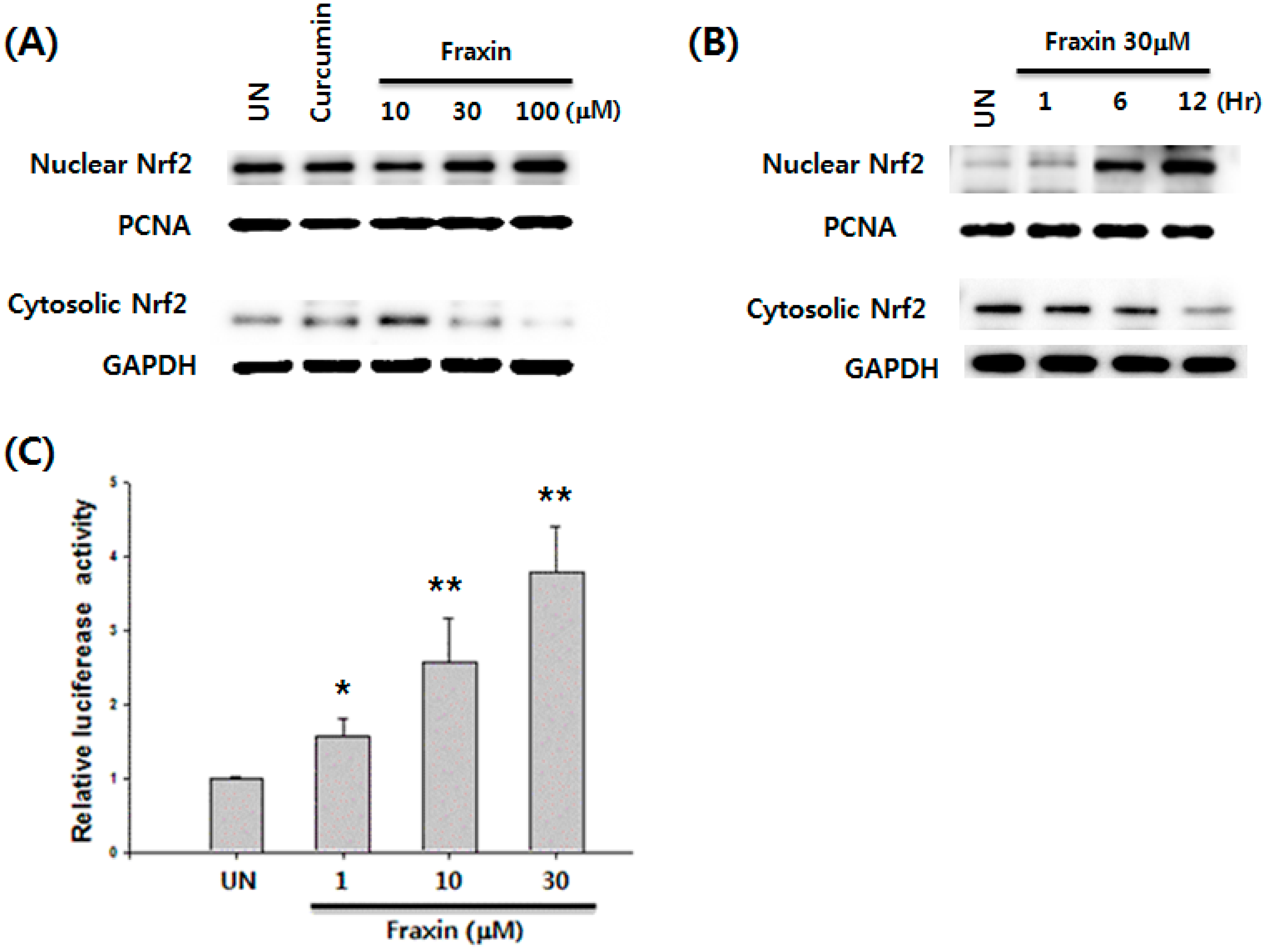
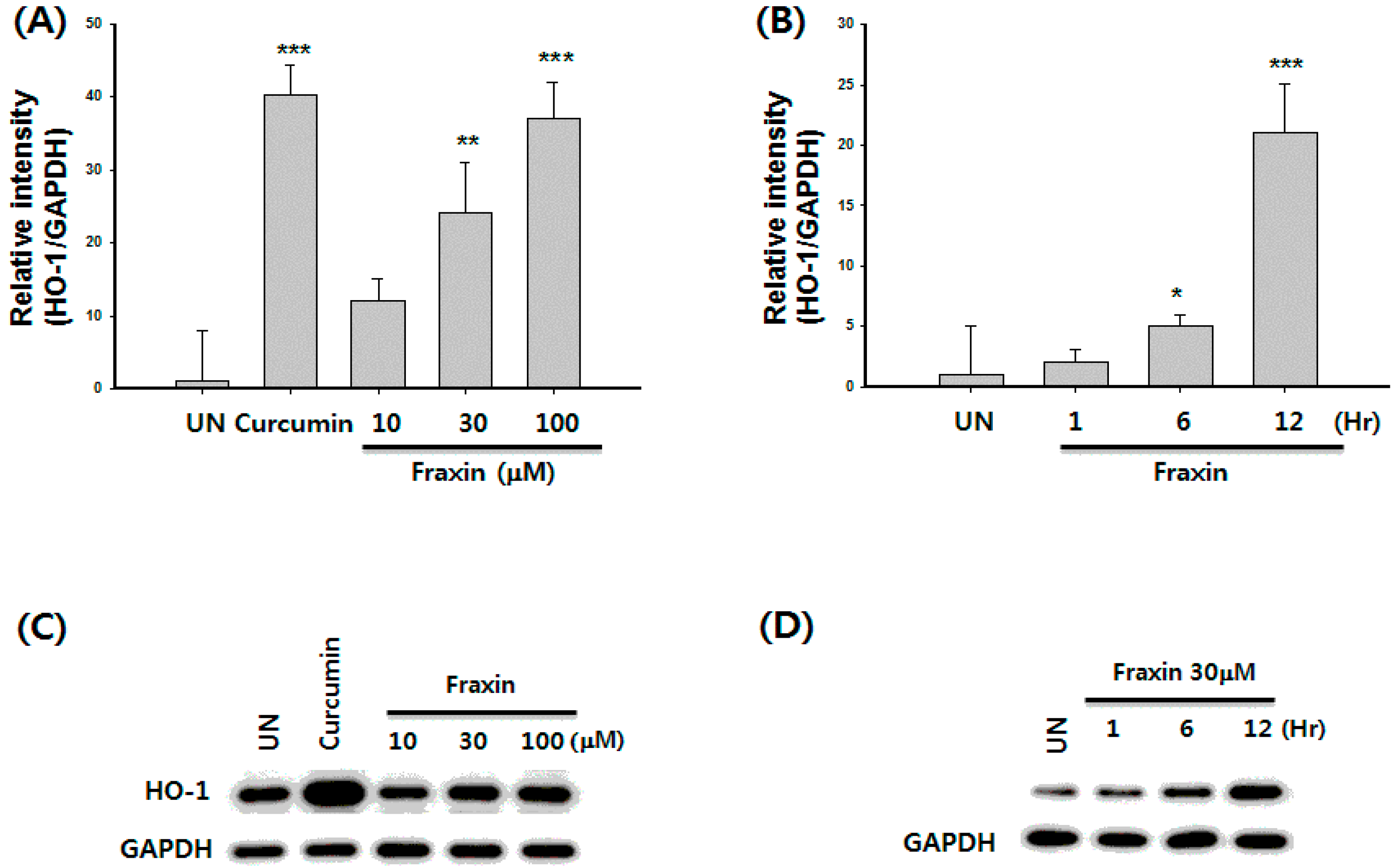
3.4. Effect of Fraxin on the Nrf2 Activation and Target Gene Induction
4. Discussion
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Torres, L.R.; Santana, F.C.; Torres-Leal, F.L.; Melo, I.L.; Yoshime, L.T.; Matos-Neto, E.M.; Seelaender, M.C.; Araujo, C.M.; Cogliati, B.; Mancini-Filho, J. Pequi (Caryocar brasiliense Camb.) almond oil attenuates carbon tetrachloride-induced acute hepatic injury in rats: Antioxidant and anti-inflammatory effects. Food Chem. Toxicol. 2016, 97, 205–216. [Google Scholar] [CrossRef] [PubMed]
- Stickel, F.; Egerer, G.; Seitz, H.K. Hepatotoxicity of botanicals. Public Health Nutr. 2000, 3, 113–124. [Google Scholar] [CrossRef] [PubMed]
- Galicia-Moreno, M.; Gutierrez-Reyes, G. The role of oxidative stress in the development of alcoholic liver disease. Rev. Gastroenterol. Mex. 2014, 79, 135–144. [Google Scholar] [CrossRef] [PubMed]
- Chang, B.Y.; Lee, D.S.; Lee, J.K.; Kim, Y.C.; Cho, H.K.; Kim, S.Y. Protective activity of kudzu (Pueraria thunbergiana) vine on chemically-induced hepatotoxicity: In vitro and in vivo studies. BMC Complement. Altern. Med. 2016, 16, 39. [Google Scholar] [CrossRef] [PubMed]
- Surai, P.F. Silymarin as a natural antioxidant: An overview of the current evidence and perspectives. Antioxidants 2015, 4, 204–247. [Google Scholar] [CrossRef] [PubMed]
- Mollazadeh, H.; Hosseinzadeh, H. The protective effect of nigella sativa against liver injury: A review. Iran. J. Basic Med. Sci. 2014, 17, 958–966. [Google Scholar] [PubMed]
- Li, S.; Tan, H.Y.; Wang, N.; Zhang, Z.J.; Lao, L.; Wong, C.W.; Feng, Y. The role of oxidative stress and antioxidants in liver diseases. Int. J. Mol. Sci. 2015, 16, 26087–26124. [Google Scholar] [CrossRef] [PubMed]
- Tang, C.C.; Huang, H.P.; Lee, Y.J.; Tang, Y.H.; Wang, C.J. Hepatoprotective effect of mulberry water extracts on ethanol-induced liver injury via anti-inflammation and inhibition of lipogenesis in C57BL/6J mice. Food Chem. Toxicol. 2013, 62, 786–796. [Google Scholar] [CrossRef] [PubMed]
- Yu, T.; Lee, J.; Lee, Y.G.; Byeon, S.E.; Kim, M.H.; Sohn, E.H.; Lee, Y.J.; Lee, S.G.; Cho, J.Y. In vitro and in vivo anti-inflammatory effects of ethanol extract from acer tegmentosum. J. Ethnopharmacol. 2010, 128, 139–147. [Google Scholar] [CrossRef] [PubMed]
- Hwang, Y.H.; Park, H.; Ma, J.Y. In vitro and in vivo safety evaluation of acer tegmentosum. J. Ethnopharmacol. 2013, 148, 99–105. [Google Scholar] [CrossRef] [PubMed]
- Tung, N.H.; Ding, Y.; Kim, S.K.; Bae, K.; Kim, Y.H. Total peroxyl radical-scavenging capacity of the chemical components from the stems of acer tegmentosum maxim. J. Agric. Food Chem. 2008, 56, 10510–10514. [Google Scholar] [CrossRef] [PubMed]
- Sagar, T.; Rantlha, M.; Kruger, M.C.; Coetzee, M.; Deepak, V. Ferulic acid impairs osteoclast fusion and exacerbates survival of mature osteoclasts. Cytotechnology 2016, 68, 1963–1972. [Google Scholar] [CrossRef] [PubMed]
- Whang, W.K.; Park, H.S.; Ham, I.; Oh, M.; Namkoong, H.; Kim, H.K.; Hwang, D.W.; Hur, S.Y.; Kim, T.E.; Park, Y.G.; et al. Natural compounds,fraxin and chemicals structurally related to fraxin protect cells from oxidative stress. Exp. Mol. Med. 2005, 37, 436–446. [Google Scholar] [CrossRef] [PubMed]
- Syed, S.N.; Rizvi, W.; Kumar, A.; Khan, A.A.; Moin, S.; Ahsan, A. In vitro antioxidant and in vivo hepatoprotective activity of leave extract of raphanus sativus in rats using CCL4 model. Afr. J. Tradit. Complement. Altern. Med. 2014, 11, 102–106. [Google Scholar] [CrossRef] [PubMed]
- Sajid, M.; Khan, M.R.; Shah, N.A.; Shah, S.A.; Ismail, H.; Younis, T.; Zahra, Z. Phytochemical, antioxidant and hepatoprotective effects of alnus nitida bark in carbon tetrachloride challenged sprague dawley rats. BMC Complement. Altern. Med. 2016, 16, 268. [Google Scholar] [CrossRef] [PubMed]
- Reitman, S.; Frankel, S. A colorimetric method for the determination of serum glutamic oxalacetic and glutamic pyruvic transaminases. Am. J. Clin. Pathol. 1957, 28, 56–63. [Google Scholar] [CrossRef] [PubMed]
- Draper, H.H.; Hadley, M. Malondialdehyde determination as index of lipid peroxidation. Methods Enzymol. 1990, 186, 421–431. [Google Scholar] [PubMed]
- Griffith, O.W.; Meister, A. Potent and specific inhibition of glutathione synthesis by buthionine sulfoximine (s-n-butyl homocysteine sulfoximine). J. Biol. Chem. 1979, 254, 7558–7560. [Google Scholar] [PubMed]
- Liu, W.; Li, J.; Cai, Y.; Wu, Q.; Pan, Y.; Chen, Y.; Chen, Y.; Zheng, X.; Li, W.; Zhang, X.; et al. Hepatic IGF-1R overexpression combined with the activation of GSK-3beta and FOXO3a in the development of liver cirrhosis. Life Sci. 2016, 147, 97–102. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Cao, L.; Zhao, Q.; Zhang, L.; Chen, J.; Liu, B.; Zhao, B. Preliminary characterizations, antioxidant and hepatoprotective activity of polysaccharide from cistanche deserticola. Int. J. Biol. Macromol. 2016, 93, 678–685. [Google Scholar] [CrossRef] [PubMed]
- Perez-Gutierrez, R.M.; Garcia-Campoy, A.H.; Muniz-Ramirez, A. Properties of flavonoids isolated from the bark of eysenhardtia polystachya and their effect on oxidative stress in streptozotocin-induced diabetes mellitus in mice. Oxid. Med. Cell. Longev. 2016, 2016, 9156510. [Google Scholar] [CrossRef] [PubMed]
- Panat, N.A.; Maurya, D.K.; Ghaskadbi, S.S.; Sandur, S.K. Troxerutin, a plant flavonoid, protects cells against oxidative stress-induced cell death through radical scavenging mechanism. Food Chem. 2016, 194, 32–45. [Google Scholar] [CrossRef] [PubMed]
- Chang, B.Y.; Kim, S.B.; Lee, M.K.; Park, H.; Kim, S.Y. Improved chemotherapeutic activity by morus alba fruits through immune response of toll-like receptor 4. Int. J. Mol. Sci. 2015, 16, 24139–24158. [Google Scholar] [CrossRef] [PubMed]
- Lin, S.; Liu, M.T.; Wang, S.J.; Li, S.; Yang, Y.C.; Shi, J.G. Coumarins from branch of fraxinus sieboldiana and their antioxidative activity. Zhongguo Zhong Yao Za Zhi 2008, 33, 1708–1710. [Google Scholar] [PubMed]
- Park, K.M.; Yang, M.C.; Lee, K.H.; Kim, K.R.; Choi, S.U.; Lee, K.R. Cytotoxic phenolic constituents of acer tegmentosum maxim. Arch. Pharm. Res. 2006, 29, 1086–1090. [Google Scholar] [CrossRef] [PubMed]
- Schempp, H.; Weiser, D.; Elstner, E.F. Biochemical model reactions indicative of inflammatory processes. Activities of extracts from fraxinus excelsior and populus tremula. Arzneim. Forsch. 2000, 50, 362–372. [Google Scholar]
- Klein-Galczinsky, C. Pharmacological and clinical effectiveness of a fixed phytogenic combination trembling poplar (populus tremula), true goldenrod (solidago virgaurea) and ash (fraxinus excelsior) in mild to moderate rheumatic complaints. Wien. Med. Wochenschr. 1999, 149, 248–253. [Google Scholar] [PubMed]
- Ivanovska, N.; Yossifova, T.; Vassileva, E.; Kostova, I. Effect of some hydroxycoumarins on complement-mediated hemolysis in human serum. Methods Find. Exp. Clin. Pharm. 1994, 16, 557–562. [Google Scholar]
- Iossifova, T.; Kujumgiev, A.; Ignatova, A.; Vassileva, E.; Kostova, I. Antimicrobial effects of some hydroxycoumarins and secoiridoids from fraxinus ornus bark. Die Pharm. 1994, 49, 298–299. [Google Scholar]
- Furfaro, A.L.; Macay, J.R.; Marengo, B.; Nitti, M.; Parodi, A.; Fenoglio, D.; Marinari, U.M.; Pronzato, M.A.; Domenicotti, C.; Traverso, N. Resistance of neuroblastoma GI-ME-N cell line to glutathione depletion involves Nrf2 and heme oxygenase-1. Free Radic. Biol. Med. 2012, 52, 488–496. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Nino, W.R.; Pedraza-Chaverri, J. Protective effect of curcumin against heavy metals-induced liver damage. Food Chem. Toxicol. 2014, 69, 182–201. [Google Scholar] [CrossRef] [PubMed]
- Satoh, T.; McKercher, S.R.; Lipton, S.A. Nrf2/are-mediated antioxidant actions of pro-electrophilic drugs. Free Radic. Biol. Med. 2013, 65, 645–657. [Google Scholar] [CrossRef] [PubMed]
- Ihara, H. Regulatory mechanism of cytotoxicity by reactive sulfur species. Nihon Yakurigaku Zasshi 2016, 147, 290–293. [Google Scholar] [CrossRef] [PubMed]
- Vobornikova, I.; Pohanka, M. Smartphone-based colorimetric detection of glutathione. Neuro Endocrinol. Lett. 2016, 37, 139–143. [Google Scholar] [PubMed]
- Watters, J.L.; Satia, J.A.; da Costa, K.A.; Boysen, G.; Collins, L.B.; Morrow, J.D.; Milne, G.L.; Swenberg, J.A. Comparison of three oxidative stress biomarkers in a sample of healthy adults. Biomarkers 2009, 14, 587–595. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Wang, C.Z.; Ye, J.Z.; Li, H.T. Hepatoprotective effects of polyprenols from ginkgo biloba l. Leaves on ccl4-induced hepatotoxicity in rats. Fitoterapia 2011, 82, 834–840. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Not available. |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chang, B.Y.; Jung, Y.S.; Yoon, C.-S.; Oh, J.S.; Hong, J.H.; Kim, Y.-C.; Kim, S.Y. Fraxin Prevents Chemically Induced Hepatotoxicity by Reducing Oxidative Stress. Molecules 2017, 22, 587. https://doi.org/10.3390/molecules22040587
Chang BY, Jung YS, Yoon C-S, Oh JS, Hong JH, Kim Y-C, Kim SY. Fraxin Prevents Chemically Induced Hepatotoxicity by Reducing Oxidative Stress. Molecules. 2017; 22(4):587. https://doi.org/10.3390/molecules22040587
Chicago/Turabian StyleChang, Bo Yoon, Young Suk Jung, Chi-Su Yoon, Jun Seok Oh, Jae Heoi Hong, Youn-Chul Kim, and Sung Yeon Kim. 2017. "Fraxin Prevents Chemically Induced Hepatotoxicity by Reducing Oxidative Stress" Molecules 22, no. 4: 587. https://doi.org/10.3390/molecules22040587
APA StyleChang, B. Y., Jung, Y. S., Yoon, C.-S., Oh, J. S., Hong, J. H., Kim, Y.-C., & Kim, S. Y. (2017). Fraxin Prevents Chemically Induced Hepatotoxicity by Reducing Oxidative Stress. Molecules, 22(4), 587. https://doi.org/10.3390/molecules22040587