Attenuation of Bleomycin-Induced Pulmonary Fibrosis in Rats with S-Allyl Cysteine
Abstract
:1. Introduction
2. Results
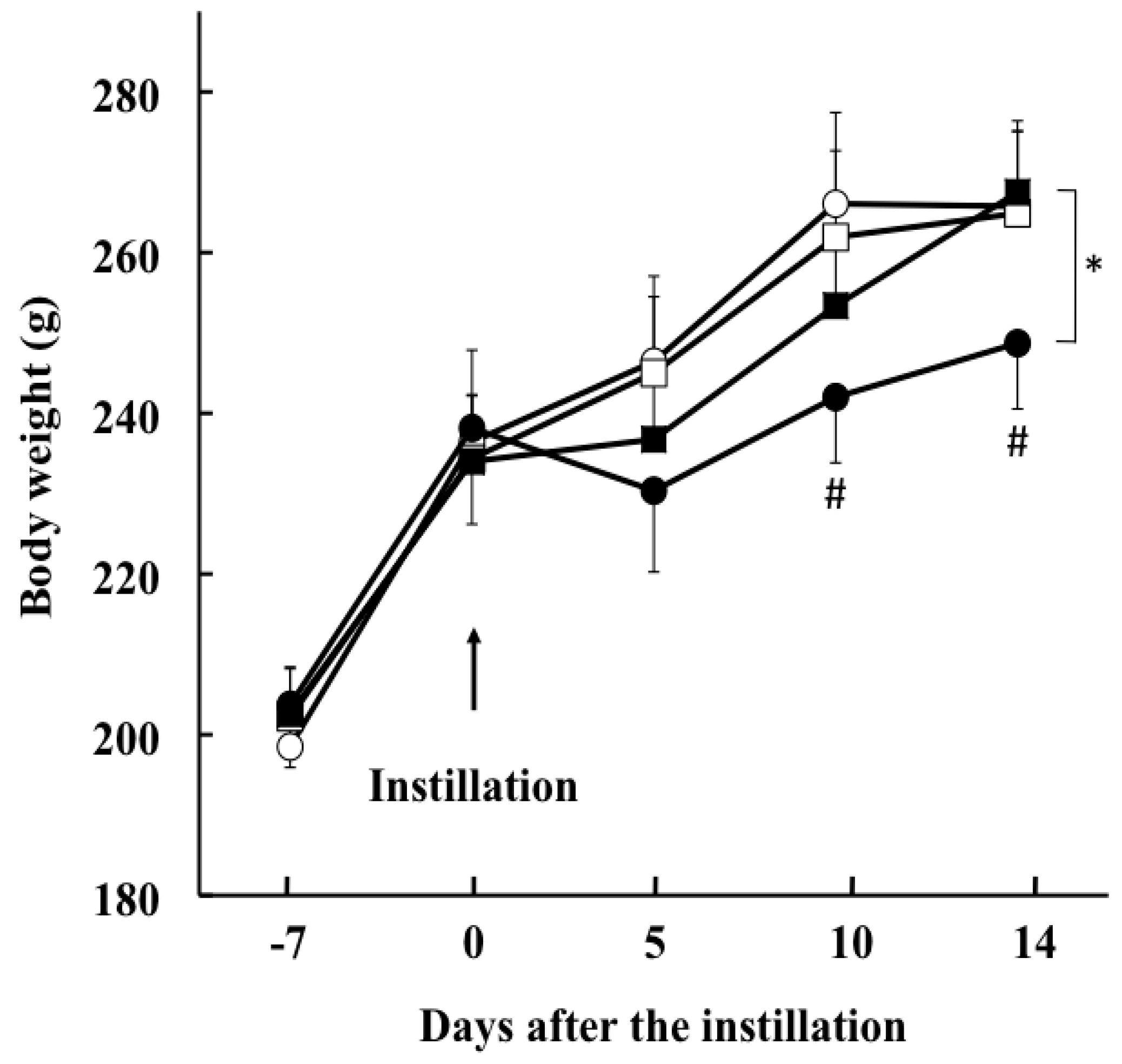
2.1. Changes in Body Weight
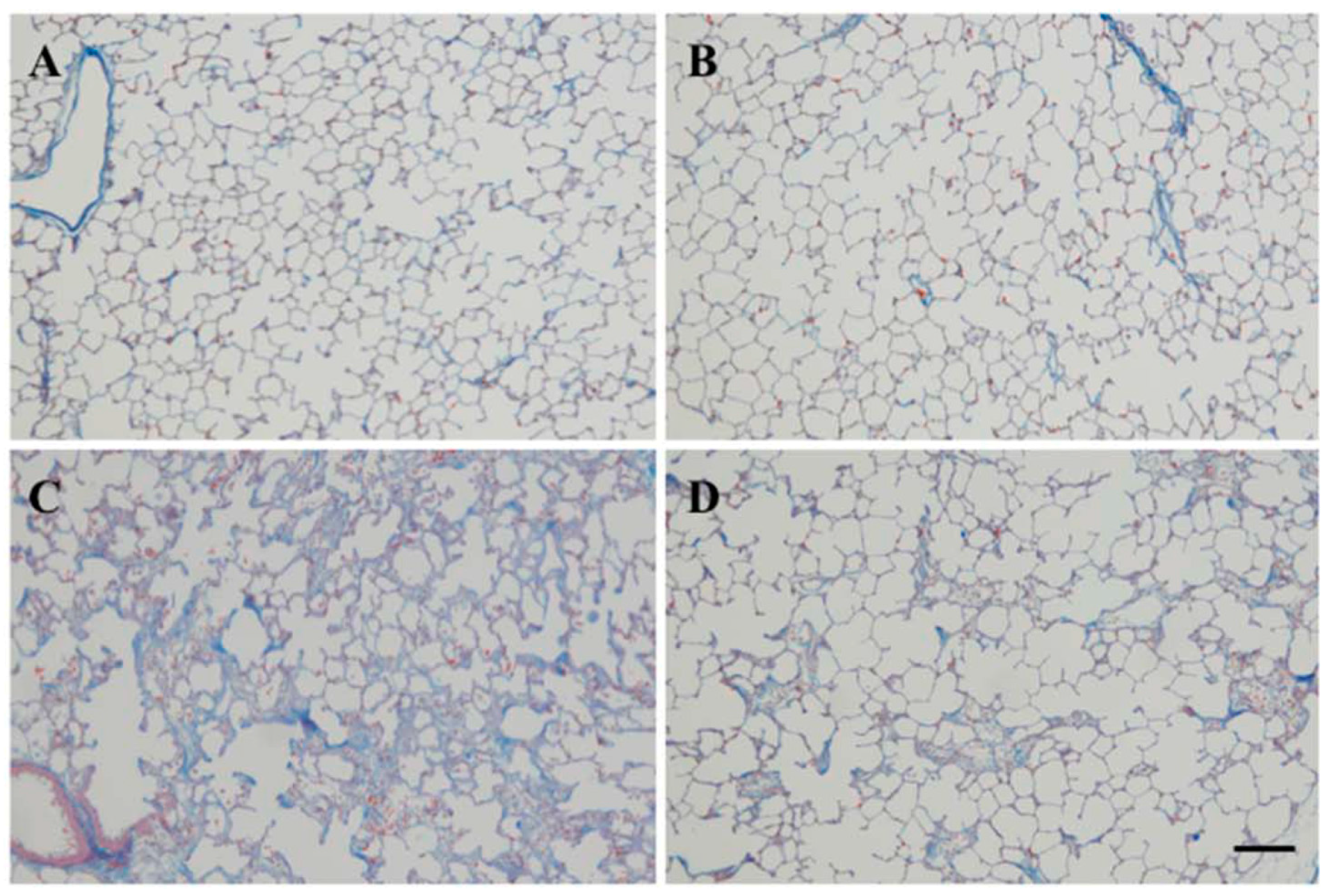
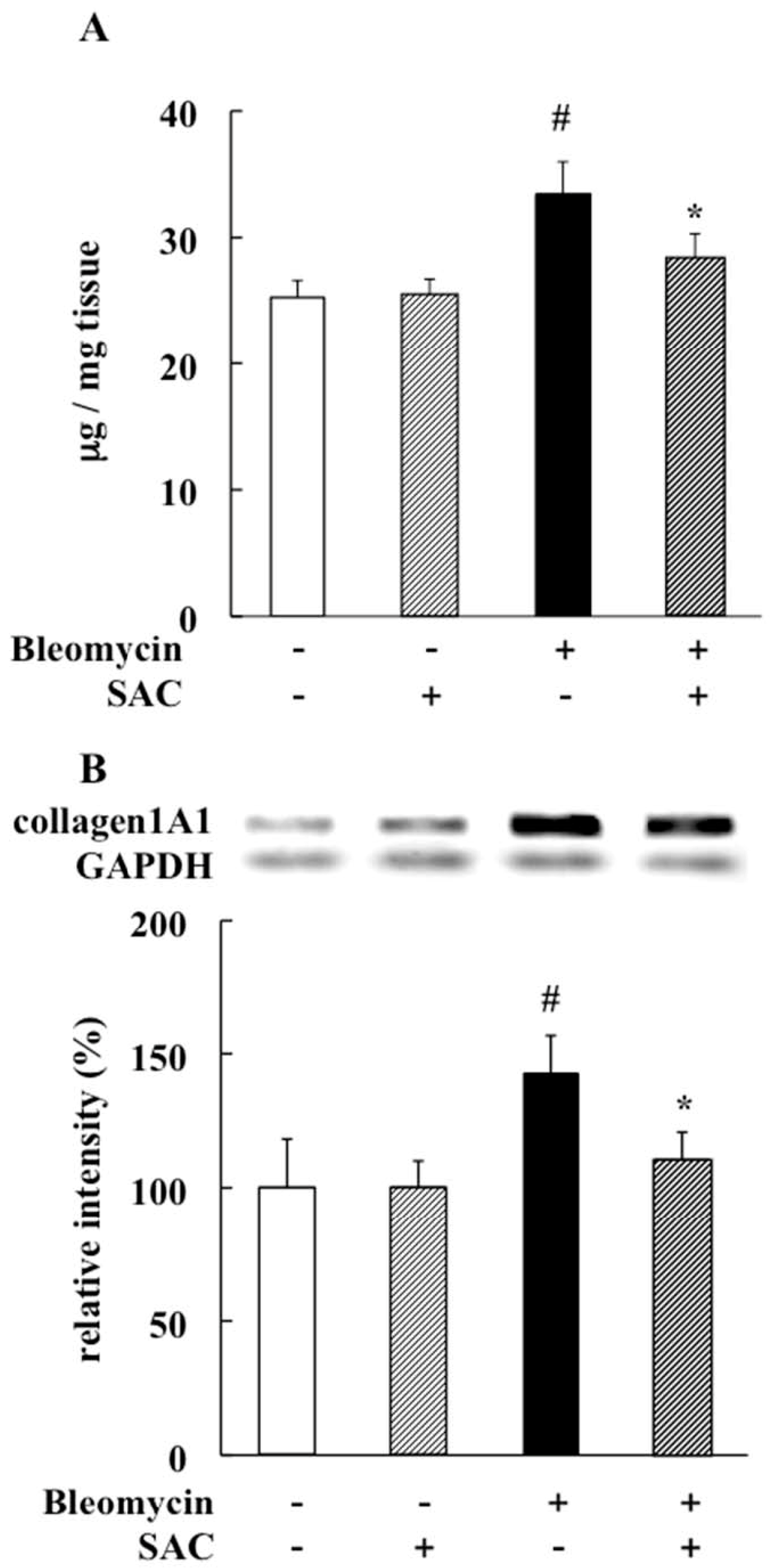
2.2. Effect of SAC on Lung Fibrosis
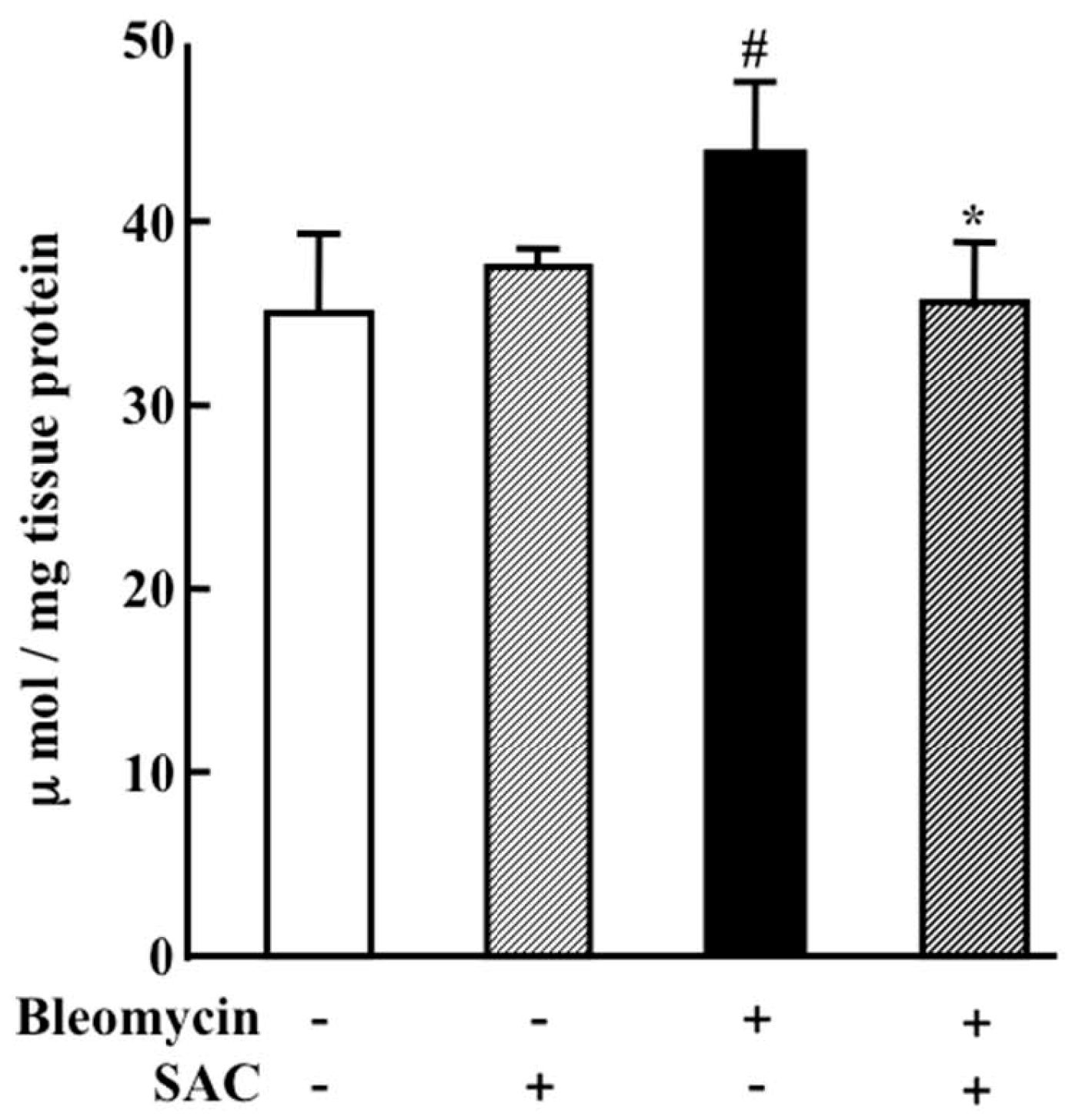
2.3. Effect of SAC on Oxidative Injury to the Lung
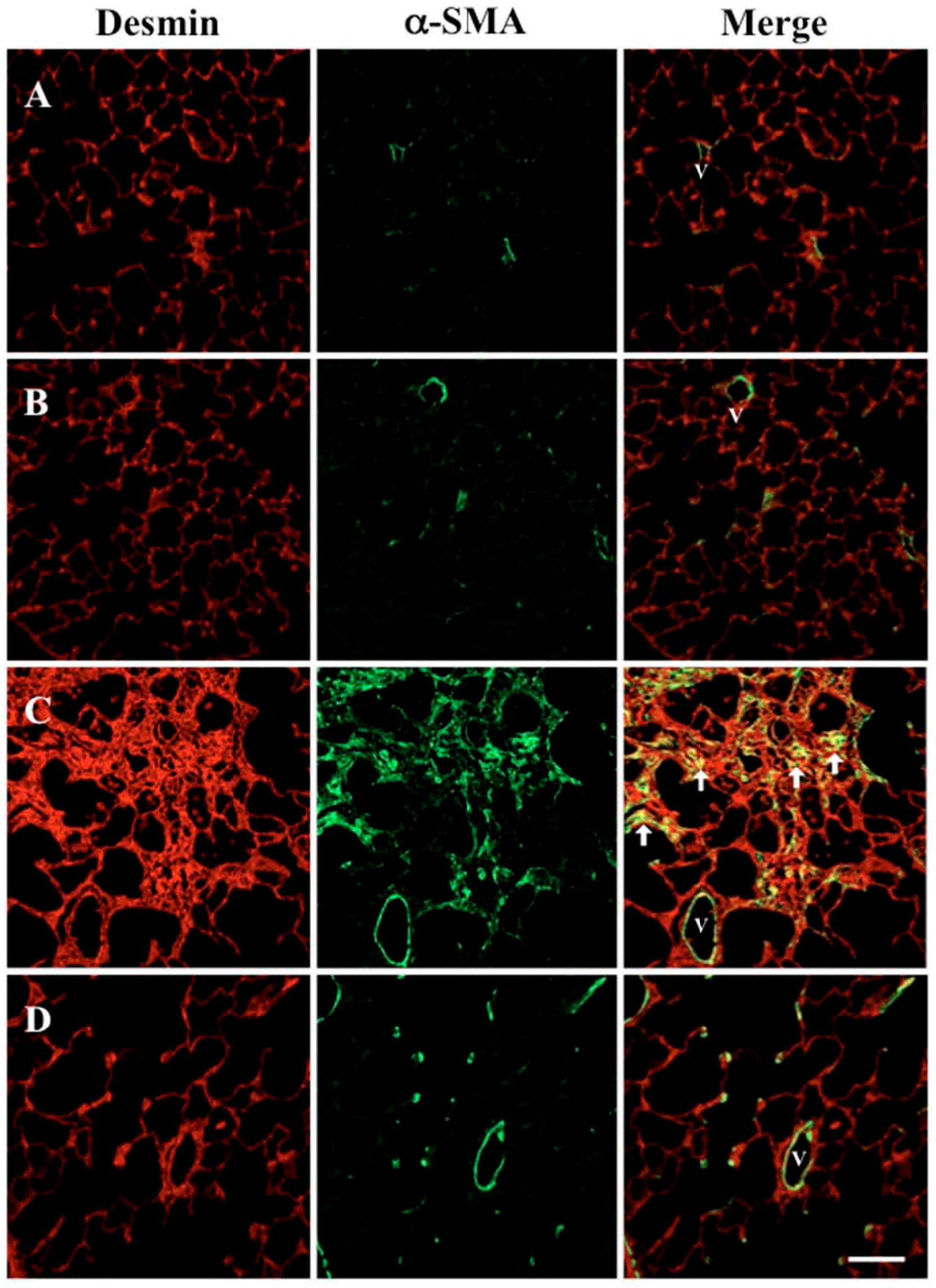
2.4. Effect of SAC on Increased Myofibroblasts in the Lung
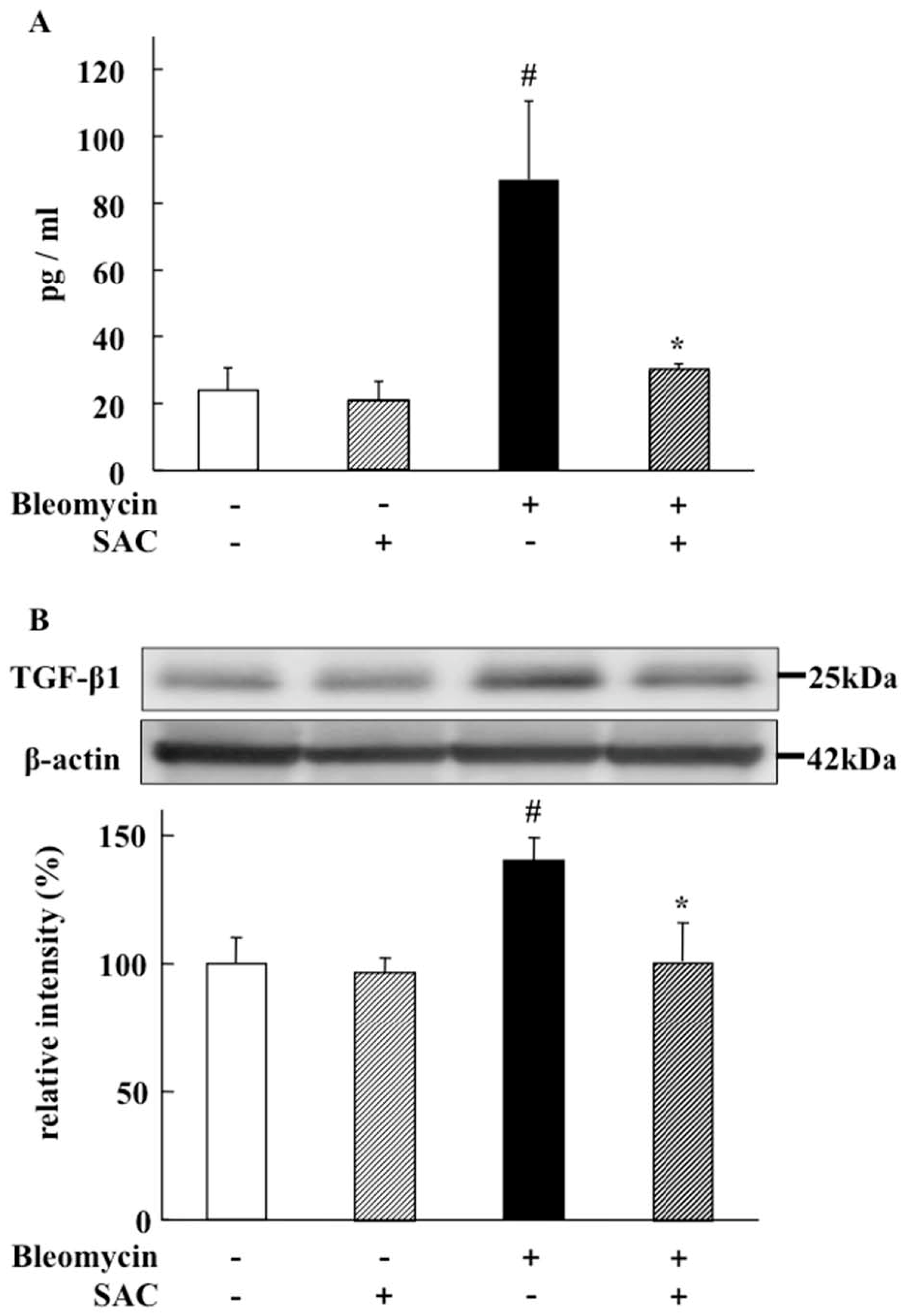
2.5. Effect of SAC on TGF-β1 Levels in Broncho-Alveolar Lavage Fluid and Lung
3. Discussion
4. Materials and Methods
4.1. Reagents
4.2. Animal Treatments
4.3. Collagen Content in Lung
4.4. Collagen mRNA Expression in Lung
4.5. LPO Level in Lung
4.6. Immunohistochemistry for α-SMA and Desmin
4.7. TGF-β1 in BALF
4.8. Western Blot Analysis for TGF-β1 in Lung
4.9. Statistical Analysis
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Mason, R.J.; Schwarz, M.I.; Hunninghake, G.W.; Musson, R.A. NHLBI Workshop Summary. Pharmacological therapy for idiopathic pulmonary fibrosis. Past, present, and future. Am. J. Respir. Crit. Care Med. 1999, 160, 1771–1777. [Google Scholar] [CrossRef] [PubMed]
- Gross, T.J.; Hunninghake, G.W. Idiopathic pulmonary fibrosis. N. Engl. J. Med. 2001, 345, 517–525. [Google Scholar] [CrossRef] [PubMed]
- Chua, F.; Gauldie, J.; Laurent, G.J. Pulmonary fibrosis: Searching for model answers. Am. J. Respir. Cell Mol. Biol. 2005, 33, 9–13. [Google Scholar] [CrossRef] [PubMed]
- Bouros, D.; Antoniou, K.M. Current and future therapeutic approaches in idiopathic pulmonary fibrosis. Eur. Respir. J. 2005, 26, 693–702. [Google Scholar] [CrossRef] [PubMed]
- Takeda, Y.; Tsujino, K.; Kijima, T.; Kumanogoh, A. Efficacy and safety of pirfenidone for idiopathic pulmonary fibrosis. Patient Prefer. Adherence 2014, 8, 361–370. [Google Scholar] [CrossRef] [PubMed]
- Amagase, H.; Petesch, B.L.; Matsuura, H.; Kasuga, S.; Itakura, Y. Intake of garlic and its bioactive components. J. Nutr. 2001, 131, 955S–962S. [Google Scholar] [PubMed]
- Numagami, Y.; Sato, S.; Ohnishi, S.T. Attenuation of rat ischemic brain damage by aged garlic extracts: A possible protecting mechanism as antioxidants. Neurochem. Int. 1996, 29, 135–143. [Google Scholar] [CrossRef]
- Mostafa, M.G.; Mima, T.; Ohnishi, S.T.; Mori, K. S-allylcysteine ameliorates doxorubicin toxicity in the heart and liver in mice. Planta Med. 2000, 66, 148–151. [Google Scholar] [CrossRef]
- Kodai, S.; Takemura, S.; Minamiyama, Y.; Hai, S.; Yamamoto, S.; Kubo, S.; Yoshida, Y.; Niki, E.; Okada, S.; Hirohashi, K.; et al. S-allyl cysteine prevents CCl4-induced acute liver injury in rats. Free Radic. Res. 2007, 41, 489–497. [Google Scholar] [CrossRef] [PubMed]
- Kodai, S.; Takemura, S.; Kubo, S.; Azuma, H.; Minamiyama, Y. Therapeutic administration of an ingredient of aged-garlic extracts, S-allyl cysteine resolves liver fibrosis established by carbon tetrachloride in rats. J. Clin. Biochem. Nutr. 2015, 56, 179–185. [Google Scholar] [CrossRef] [PubMed]
- Kinnula, V.L.; Fattman, C.L.; Tan, R.J.; Oury, T.D. Oxidative stress in pulmonary fibrosis: A possible role for redox modulatory therapy. Am. J. Respir. Crit. Care Med. 2005, 172, 417–422. [Google Scholar] [CrossRef] [PubMed]
- Thannickal, V.J.; Lee, D.Y.; White, E.S.; Cui, Z.; Larios, J.M.; Chacon, R.; Horowitz, J.C.; Day, R.M.; Thomas, P.E. Myofibroblast differentiation by transforming growth factor-β1 is dependent on cell adhesion and integrin signaling via focal adhesion kinase. J. Biol. Chem. 2003, 278, 12384–12389. [Google Scholar] [CrossRef] [PubMed]
- Tomasek, J.J.; Gabbiani, G.; Hinz, B.; Chaponnier, C.; Brown, R.A. Myofibroblasts and mechano-regulation of connective tissue remodelling. Nat. Rev. Mol. Cell Biol. 2002, 3, 349–363. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Shi, W.; Wang, Y.L.; Chen, H.; Bringas, P., Jr.; Datto, M.B.; Frederick, J.P.; Wang, X.F.; Warburton, D. Smad3 deficiency attenuates bleomycin-induced pulmonary fibrosis in mice. Am. J. Physiol. Lung Cell. Mol. Physiol. 2002, 282, L585–L593. [Google Scholar] [CrossRef] [PubMed]
- Mizuguchi, S.; Takemura, S.; Minamiyama, Y.; Kodai, S.; Tsukioka, T.; Inoue, K.; Okada, S.; Suehiro, S. S-allyl cysteine attenuated CCl4-induced oxidative stress and pulmonary fibrosis in rats. BioFactors 2006, 26, 81–92. [Google Scholar] [CrossRef] [PubMed]
- Moore, M.D.; Fu, W.; Soheilian, F.; Nagashima, K.; Ptak, R.G.; Pathak, V.K.; Hu, W.S. Suboptimal inhibition of protease activity in human immunodeficiency virus type 1: Effects on virion morphogenesis and RNA maturation. Virology 2008, 379, 152–160. [Google Scholar] [CrossRef] [PubMed]
- Ask, K.; Martin, G.E.; Kolb, M.; Gauldie, J. Targeting genes for treatment in idiopathic pulmonary fibrosis: Challenges and opportunities, promises and pitfalls. Proc. Am. Thorac. Soc. 2006, 3, 389–393. [Google Scholar] [CrossRef] [PubMed]
- Sime, P.J.; Xing, Z.; Graham, F.L.; Csaky, K.G.; Gauldie, J. Adenovector-mediated gene transfer of active transforming growth factor-beta1 induces prolonged severe fibrosis in rat lung. J. Clin. Investig. 1997, 100, 768–776. [Google Scholar] [CrossRef] [PubMed]
- Khalil, N.; Corne, S.; Whitman, C.; Yacyshyn, H. Plasmin regulates the activation of cell-associated latent TGF-beta 1 secreted by rat alveolar macrophages after in vivo bleomycin injury. Am. J. Respir. Cell Mol. Biol. 1996, 15, 252–259. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.; Konigshoff, M.; Jayachandran, A.; Handley, D.; Seeger, W.; Kaminski, N.; Eickelberg, O. Transgelin is a direct target of TGF-β/Smad3-dependent epithelial cell migration in lung fibrosis. FASEB J. 2008, 22, 1778–1789. [Google Scholar] [CrossRef] [PubMed]
- Kum, Y.S.; Kim, K.H.; Park, T.I.; Suh, I.S.; Oh, H.K.; Cho, C.H.; Park, J.B.; Chang, Y.C.; Park, J.H.; Lee, K.G.; et al. Antifibrotic effect via the regulation of transcription factor Sp1 in lung fibrosis. Biochem. Biophys. Res. Commun. 2007, 363, 368–374. [Google Scholar] [CrossRef] [PubMed]
- Selman, M.; Pardo, A. Idiopathic pulmonary fibrosis: An epithelial/fibroblastic cross-talk disorder. Respir. Res. 2002, 3, 3. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K.; Rekhter, M.D.; Gordon, D.; Phan, S.H. Myofibroblasts and their role in lung collagen gene expression during pulmonary fibrosis. A combined immunohistochemical and in situ hybridization study. Am. J. Pathol. 1994, 145, 114–125. [Google Scholar] [PubMed]
- Wu, Z.; Yang, L.; Cai, L.; Zhang, M.; Cheng, X.; Yang, X.; Xu, J. Detection of epithelial to mesenchymal transition in airways of a bleomycin induced pulmonary fibrosis model derived from an α-smooth muscle actin-Cre transgenic mouse. Respir. Res. 2007, 8, 1. [Google Scholar] [CrossRef] [PubMed]
- Collard, H.R.; King, T.E., Jr. Treatment of idiopathic pulmonary fibrosis: The rise and fall of corticosteroids. Am. J. Med. 2001, 110, 326–328. [Google Scholar] [CrossRef]
- American Thoracic Society (ATS); European Respiratory Society (ERS). Idiopathic pulmonary fibrosis: Diagnosis and treatment. International consensus statement. Am. J. Respir. Crit. Care Med. 2000, 161, 646–664. [Google Scholar]
- Munger, J.S.; Huang, X.; Kawakatsu, H.; Griffiths, M.J.; Dalton, S.L.; Wu, J.; Pittet, J.F.; Kaminski, N.; Garat, C.; Matthay, M.A.; et al. The integrin αvβ6 binds and activates latent TGF β1: A mechanism for regulating pulmonary inflammation and fibrosis. Cell 1999, 96, 319–328. [Google Scholar] [CrossRef]
- Bonniaud, P.; Margetts, P.J.; Kolb, M.; Schroeder, J.A.; Kapoun, A.M.; Damm, D.; Murphy, A.; Chakravarty, S.; Dugar, S.; Higgins, L.; et al. Progressive transforming growth factor β1-induced lung fibrosis is blocked by an orally active ALK5 kinase inhibitor. Am. J. Respir. Crit. Care Med. 2005, 171, 889–898. [Google Scholar] [CrossRef] [PubMed]
- Scotton, C.J.; Chambers, R.C. Molecular targets in pulmonary fibrosis: The myofibroblast in focus. Chest 2007, 132, 1311–1321. [Google Scholar] [CrossRef] [PubMed]
- Sheppard, D. Transforming growth factor beta: A central modulator of pulmonary and airway inflammation and fibrosis. Proc. Am. Thorac. Soc. 2006, 3, 413–417. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Not available. |
| Collagen1A1 | sense | 5′-CCAAAGGATCTCCTGGTGAA-3′ |
| antisense | 5′-GGAAACCTCTCTCGCCTCTT-3′ | |
| GAPDH | sense | 5′-CCTGCACCACCAACTGC-3′ |
| antisense | 5′-CCATGCCAGTGAGCTTC-3′ |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tsukioka, T.; Takemura, S.; Minamiyama, Y.; Mizuguchi, S.; Toda, M.; Okada, S. Attenuation of Bleomycin-Induced Pulmonary Fibrosis in Rats with S-Allyl Cysteine. Molecules 2017, 22, 543. https://doi.org/10.3390/molecules22040543
Tsukioka T, Takemura S, Minamiyama Y, Mizuguchi S, Toda M, Okada S. Attenuation of Bleomycin-Induced Pulmonary Fibrosis in Rats with S-Allyl Cysteine. Molecules. 2017; 22(4):543. https://doi.org/10.3390/molecules22040543
Chicago/Turabian StyleTsukioka, Takuma, Shigekazu Takemura, Yukiko Minamiyama, Shinjiro Mizuguchi, Michihito Toda, and Shigeru Okada. 2017. "Attenuation of Bleomycin-Induced Pulmonary Fibrosis in Rats with S-Allyl Cysteine" Molecules 22, no. 4: 543. https://doi.org/10.3390/molecules22040543
APA StyleTsukioka, T., Takemura, S., Minamiyama, Y., Mizuguchi, S., Toda, M., & Okada, S. (2017). Attenuation of Bleomycin-Induced Pulmonary Fibrosis in Rats with S-Allyl Cysteine. Molecules, 22(4), 543. https://doi.org/10.3390/molecules22040543






