Abstract
2,5-Diformylfuran (DFF) is an important biorenewable building block, namely for the manufacture of new polymers that may replace existing materials derived from limited fossil fuel resources. The current reported methods for the preparation of DFF are mainly derived from the oxidation of 5-hydroxymethylfurfural (HMF) and, to a lesser extent, directly from fructose. 5-Chloromethylfurfural (CMF) has been considered an alternative to HMF as an intermediate building block due to its advantages regarding stability, polarity, and availability from glucose and cellulose. The only reported method for the transformation of CMF to DFF is restricted to the use of DMSO as the solvent and oxidant. We envisioned that the transformation could be performed using more attractive conditions. To that end, we explored the oxidation of CMF to DFF by screening several oxidants such as H2O2, oxone, and pyridine N-oxide (PNO); different heating methods, namely thermal and microwave irradiation (MWI); and also flow conditions. The combination of PNO (4 equiv.) and Cu(OTf)2 (0.5 equiv.) in acetonitrile was identified as the best system, which lead to the formation of DFF in 54% yield under MWI for 5 min at 160 °C. Consequently, a range of different heterogeneous copper catalysts were tested, which allowed for catalyst reuse. Similar results were also observed under flow conditions using copper immobilized on silica under thermal heating at 160 °C for a residence time of 2.7 min. Finally, HMF and 5,5′-oxybis(5-methylene-2-furaldehyde) (OBMF) were the only byproducts identified under the reaction conditions studied.
1. Introduction
The pursuit of new biorenewable building blocks and/or more efficient synthetic routes has intensified during the last few years. To date, several molecules including ethanol, glycerol, carboxylic acids (lactic acid, succinic acid, 3-hydroxypropionic acid, itaconic acid, glutamic acid, levulinic acid, fatty acids), furfural, 5-hydroxymethylfurfural (HMF) and terpenes (α-pinene, limonene, citral, menthol) [1,2,3,4] have been identified as alternatives to petroleum-based building blocks. The discovery of new polymer monomers that may replace the current well established portfolio derived from fossil fuel resources has been recognized as an important issue. 2,5-Furandicarboxylic acid (FDCA), 2,5-dihydroxymethylfuran (DHMF) and 2,5-diformylfuran (DFF), among others, have been identified as promising monomers [5,6]. These furan derivatives are commonly obtained from HMF, which, in turn, is mainly derived from fructose [7,8,9,10,11]. Besides the intensive research and remarkable achievements regarding the production of HMF, there are still limitations that arise from the specific nature of HMF, namely, its thermal and chemical instability, high solubility in water, and low melting point. On the other hand, as highlighted by the Mascal group, 5-chloromethylfurfural (CMF) is an appealing alternative to HMF, due to its higher stability, lower polarity and higher accessibility from glucose and cellulose [12,13,14].
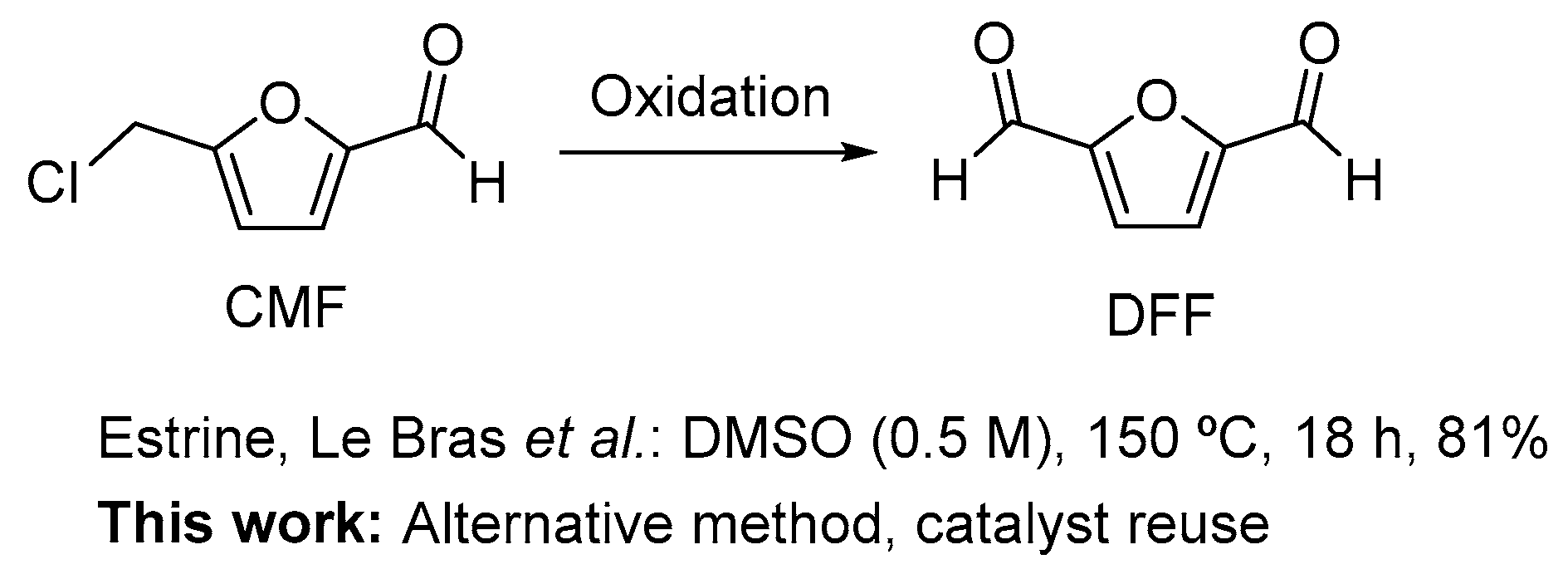
In general, DFF is prepared by the oxidation of HMF [8,11]. The selective oxidation of the hydroxyl group into the aldehyde has been achieved using a variety of different methodologies including homogeneous [8,11,15,16] and heterogeneous [8,11,17,18,19,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37] metal catalysis, a one-step procedure from fructose (DMSO, NaBr (cat.), 150 °C, 50%) [38], using molybdovanadophosphoric heteropolyacids [39,40], using polyaniline-grafted vanadyl acetylacetonate [41], metal free procedures [42,43,44] and enzyme catalysis [45]. The use of CMF as a starting material for the preparation of DFF was previously described in 2014 by Estrine and Le Bras and later used by the Mascal group. However, the use of DMSO as the solvent and oxidant makes this method less attractive (Scheme 1) [38,46]. To that end, we envisioned the possibility of performing such an oxidation without using DMSO (Scheme 1).
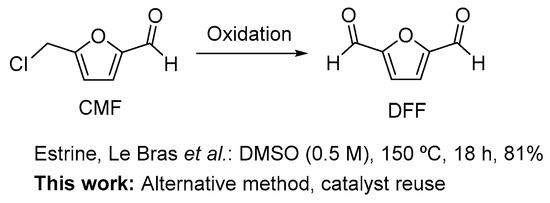
Scheme 1.
Reported methods by Estrine, Le Bras et al. [38] for the oxidation of CMF to DFF and in this study on the search for alternative methods that avoid the use of DMSO and allow catalyst reuse.
2. Results and Discussion
The oxidation of benzylic chlorides to the corresponding aldehydes has been reported using different systems, namely CuCl/Kieselguhr/O2 [47], 2-dimethylamino-N,N-dimethylaniline N-oxide [48], 3,6-bis(triphenylphosphonium)cyclohexene peroxodisulfate [49], NaIO4-DMF [50], H5IO6 in [C12mim][FeCl4] ionic liquid [51], TEMPO (cat)/KNO2 (cat)/O2 [52], NMO/KI (cat) in [emim]Cl ionic liquid, MW [53] and molybdate-based catalyst/H2O2 [54]. Based on that, we started this study by screening several oxidants (1.5 equiv.) for the oxidation of CMF in tetrabutylammonoium chloride (TBAC) as a reaction medium under basic conditions (K2CO3, 2 equiv.) at 110 °C for 5 h. Several oxidants such as urea hydrogen peroxide (UHP), NaBO3, Ca(ClO)2, NaIO4, H2O2, Oxone, KNO2, and NMO yielded full conversion of CMF. However, very low NMR yields of DFF were obtained (<10%, Table S1). In addition, HMF was identified as the single by-product (yields ranging from 3% to 67%), whose formation may derive from the nucleophilic substitution of the chloride atom. Those unsatisfactory results suggest that TBAC salt is not suitable for this transformation under the tested reaction conditions, giving side reactions such as over oxidation and nucleophilic substitution.
From the preliminary screening, pyridine N-oxide (PNO) in combination with CuCl (0.5 equiv.) was identified as the most effective system, providing DFF in 12% yield. Further solvent screening using PNO (2 equiv.) as the oxidant at lower temperature (82 °C) for 24 h produced DFF in up to 9% yield. (Table 1, entries 1–5). Acetonitrile was identified as the most promising solvent, suppressing the undesired formation of HMF (Table 1, entry 1). Furthermore, no conversion of CMF was observed in the absence of the promoter CuCl (Table 1, entry 4). Increasing the amount of PNO to 4 equiv. resulted in an increased yield of DFF to 37% (3% of unreacted CMF was detected). However, HMF was detected in 26% yield (Table 1, entry 6). The use of both anhydrous acetonitrile and PNO was not beneficial for the suppression of HMF formation probably due to trace amounts of water in the PNO (Table 1, entry 7). The use of longer reaction times (55 h, 34%, Table 1, entry 8), higher excess of PNO (8 equiv., 37% yield, Table 1, entry 9), or lower/higher temperatures (60 °C: 13%; 110 °C: 19%; Table 1, entries 10 and 11) were not beneficial for the transformation. In addition, Cu(OTf)2 provided comparable results (35%; Table 1, entry 12). Interestingly, using HMF instead of CMF resulted in no formation of DFF, which suggests that DFF is not formed via HMF oxidation (Table 1, entry 14).

Table 1.
Optimization of reaction conditions for the oxidation of CMF using PNO as an oxidant under thermal heating.
The use of microwave irradiation (MWI) using 4 equiv. of PNO and 0.5 equiv. of Cu(OTf)2 in acetonitrile (0.5 M) at 82 °C lead to the formation of DFF in 49% yield after 6 h, together with 24% of unreacted CMF (Table 2, entry 1). Remarkably, the formation of HMF was not observed under these conditions, which is considerably better than the best results achieved under thermal heating (37% of DFF after 24 h, Table 1, entry 6). Further optimization of the reaction conditions to 160 °C and only 5 min of reaction time resulted in the formation of DFF in up to 51% yield (isolated yield of 50%, Table 2, entries 9 and 10, see complete data in Table S3). In addition, the reaction performed in the absence of the promoter Cu(OTf)2 yielded only 6% of DFF (Table 2, entry 11). Finally, no formation of DFF was observed in the absence of the PNO oxidant, resulting in the complete decomposition of the CMF substrate instead (Table 2, entry 15).

Table 2.
Selected reaction conditions and yields of DFF, CMF, and HMF for the oxidation reactions of CMF performed under microwave irradiation (MWI).
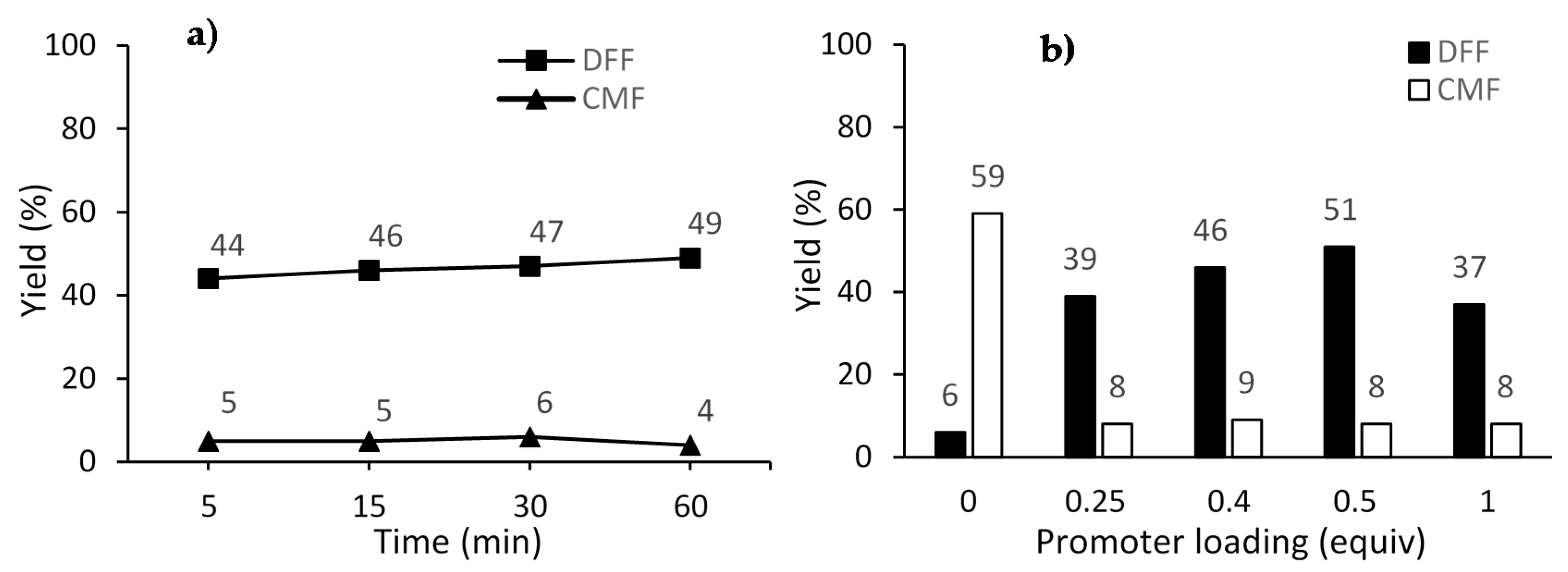
The study of the reaction time was carried out following the general procedure at 140 °C with 0.5 equiv. of Cu(OTf)2. The results shown in Figure 1a indicate that there is no pronounced advantage for both the DFF yield and CMF conversion by running the reaction for more than 5 min. Regarding the promoter loading, it was observed that 0.5 equiv. of Cu(OTf)2 is the optimal amount, yielding the product DFF in 51% yield.
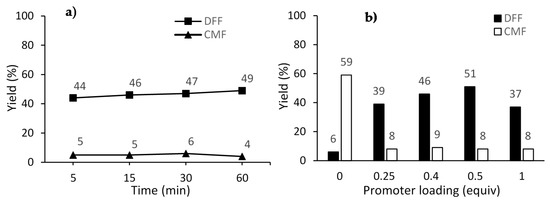
Figure 1.
Oxidation of CMF for (a) different reaction times at 140 °C using 0.5 equiv. of Cu(OTf)2; (b) different promoter loading at 160 °C for 5 min. Reaction conditions: microwave irradiation (MWI), CMF (0.35 mmol, 50 mg), 4 equiv. of pyridine N-oxide, in CH3CN (0.5 M). Yields were determined by 1H-NMR analysis of the crude reaction mixture using 1,3,5-trimethoxybenzene as internal standard.
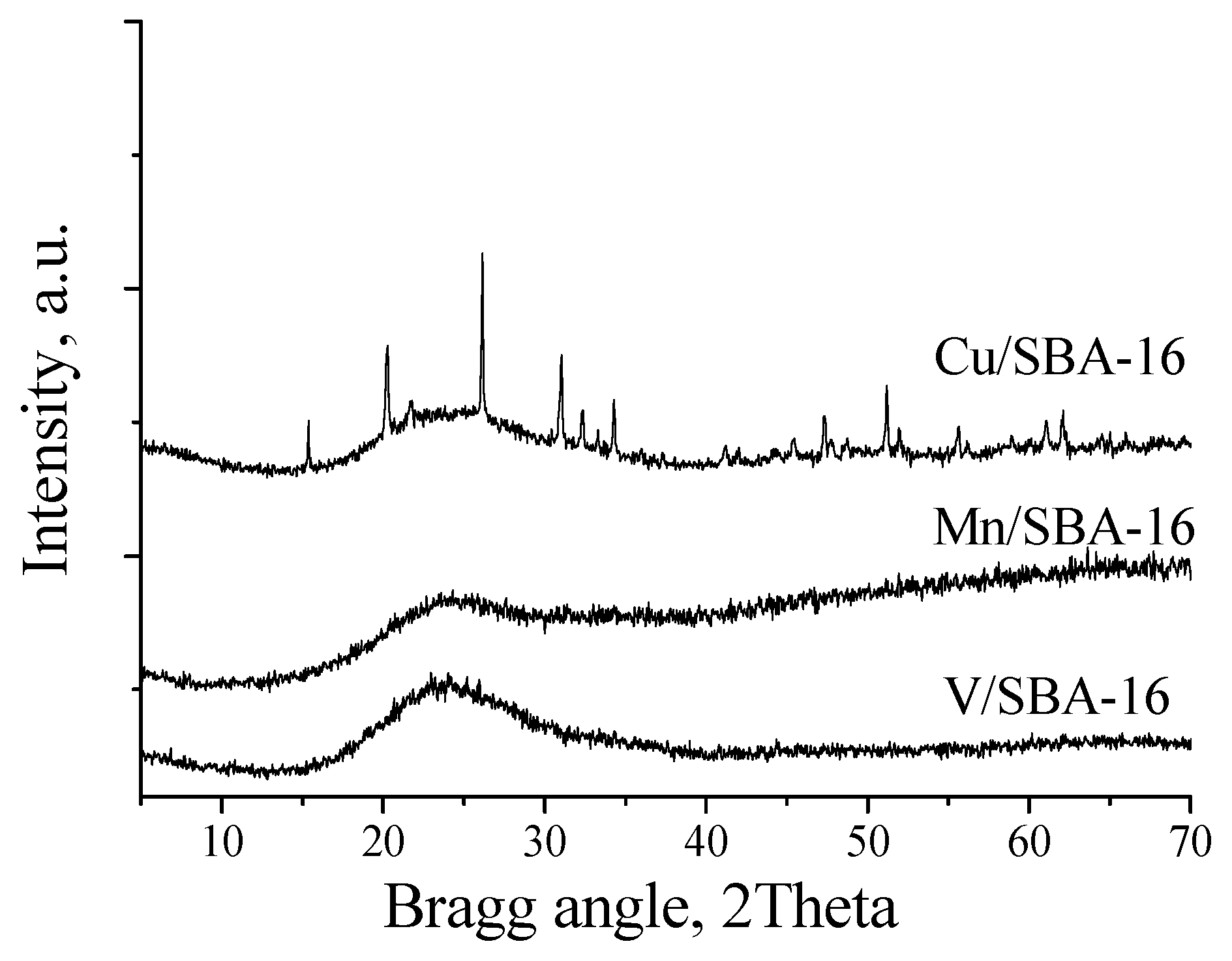
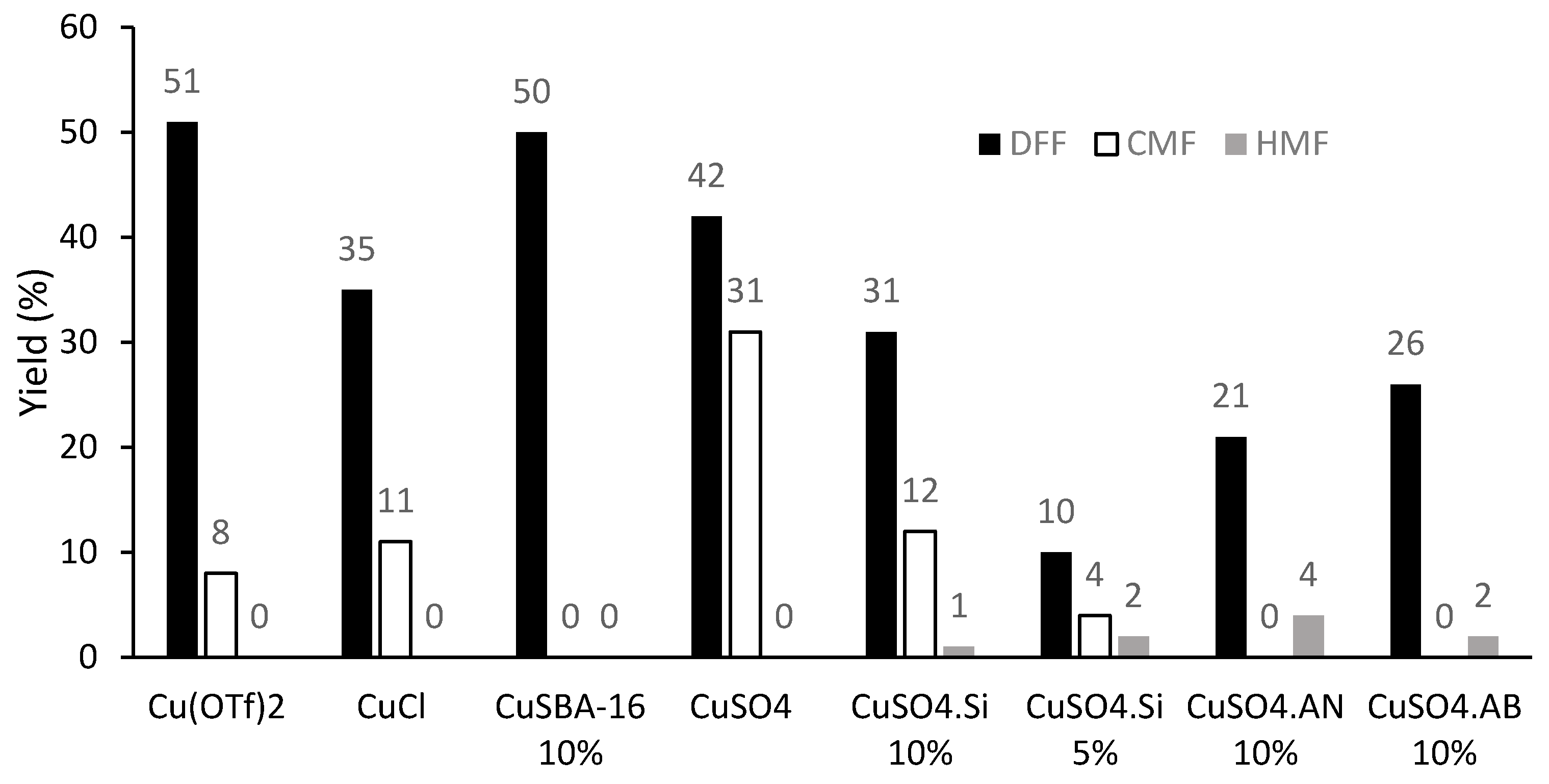
We envisioned that this procedure would be more effective under heterogeneous conditions. Based on that, several different catalysts, namely commercially available CuCl or CuSO4 as well as supported Cu catalysts, were tested under the conditions optimized using the promoter Cu(OTf)2 (0.5 mol %, PNO (4 equiv.), acetonitrile (0.5 M), MWI, 160 °C, 5 min). The adsorption of CuSO4 in silica (CuSO4·Si) and neutral (CuSO4·AN) or basic alumina (CuSO4·AB) was performed by simple evaporation of the solid support suspension in an aqueous solution of CuSO4. TheCu/SiO2 catalysts were prepared according to the reported procedure [55] by dispersion of the copper complex in silica followed by water evaporation and calcination (400 °C, 4 h). The SBA-16 catalysts were synthesized according to the procedure of Hu et al. [56]. The XRD data of the Cu, Mn, and V modified SBA-16 silica materials in the lower two theta regions (not shown) confirm the preservation of the mesoporous structure after the impregnation process. In the higher two theta regions, reflections with low intensity, characteristic of CuO, were detected in the Cu/SBA-16 material. No reflections typical of Mn and V oxide species could be observed by XRD (Figure 2), which is due to the formation of finely dispersed metal oxide species in them. The crystallite size of the copper oxide particles was determined by the Scherrer method to be around 20 nm. The calculated textural parameters from the nitrogen physisorption measurements of the metal-containing silica materials are listed in Table 3. No significant pore size reduction can be observed in SBA-16 supported catalysts either in the spherical pores (~8 nm) or in the interconnecting channels (2.5 nm). These data support that a portion of the metal oxides penetrated into the channels and cavities of the mesoporous supports, but no significant pore blocking occurred.
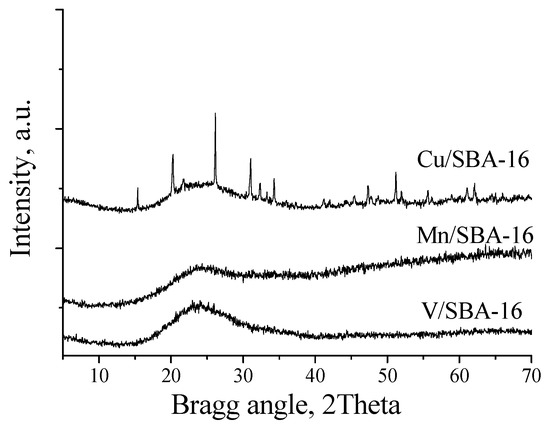
Figure 2.
XRD temperature-programmed reduction of the Cu/SBA-16, V/SBA-16 and Mn/SBA-16 catalysts.

Table 3.
Textural properties of the studied SBA-16 samples.
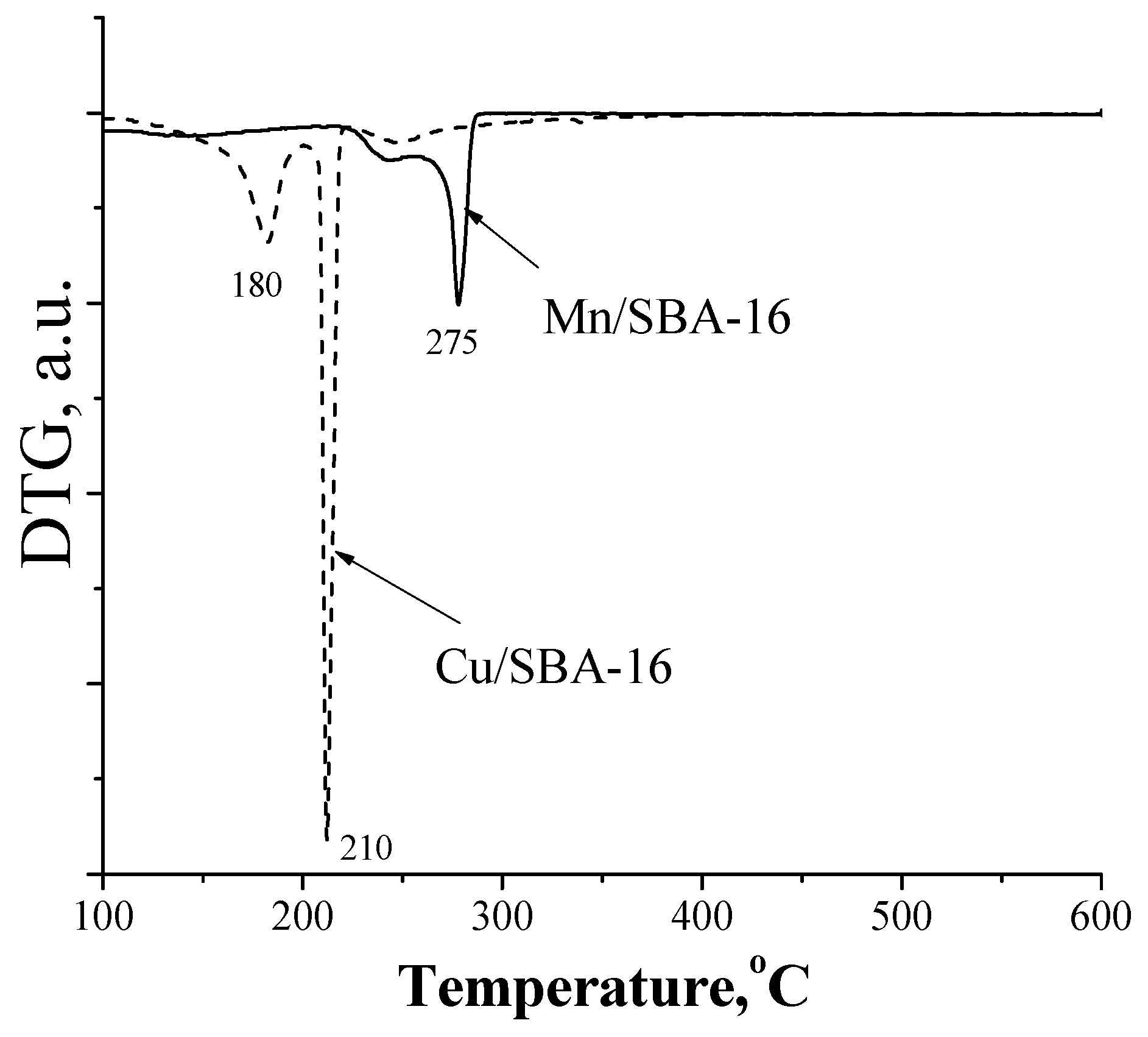
The temperature-programmed reduction (TPR) data (Figure 3) show the total reduction of CuO to Cu(o) in the Cu/SBA-16 sample which occurred at low temperature (around 210 °C). Partial reduction of the Mn oxide species was detected in the Mn/SBA-16 sample at higher temperature (275 °C).
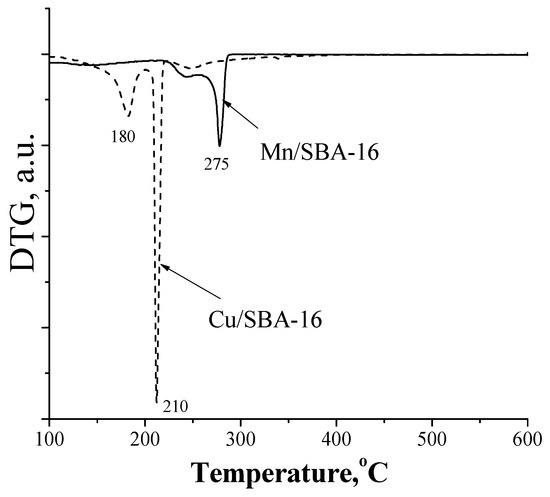
Figure 3.
TPR of the Cu/SBA-16 and Mn/SBA-16 catalysts.
Oxidation of CMF in the presence of different heterogeneous catalysts indicated that CuSBA-16 (10%) is the most efficient promoter, leading to a similar yield of DFF as the homogeneous promoter Cu(OTf)2. Remarkably, CuSO4, which is the cheapest catalyst tested, gave 42% yield of DFF (Figure 4). Using a higher amount of CuSO4 (1%), longer reaction time (up to 30 min), or higher temperature (up to 170 °C) gave lower yields of DFF (38%, 41%, and 35%, respectively), together with the formation of small amounts of HMF (up to 7%, see full results in Table S4).
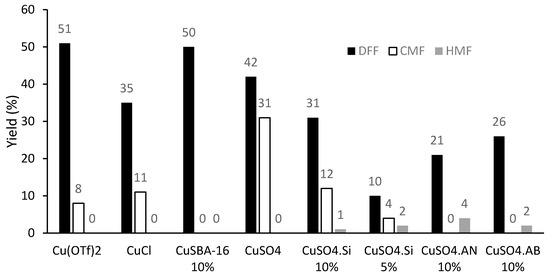
Figure 4.
Effect of the promoter nature on the oxidation of CMF under microwave irradiation (MWI). Reaction conditions: CMF (0.35 mmol, 50 mg), catalyst, PNO (4 equiv.) in CH3CN (0.5 M) and 160 °C for 5 min under MWI; yields were determined by 1H-NMR analysis of the crude reaction mixture using 1,3,5-trimethoxybenzene as an internal standard.
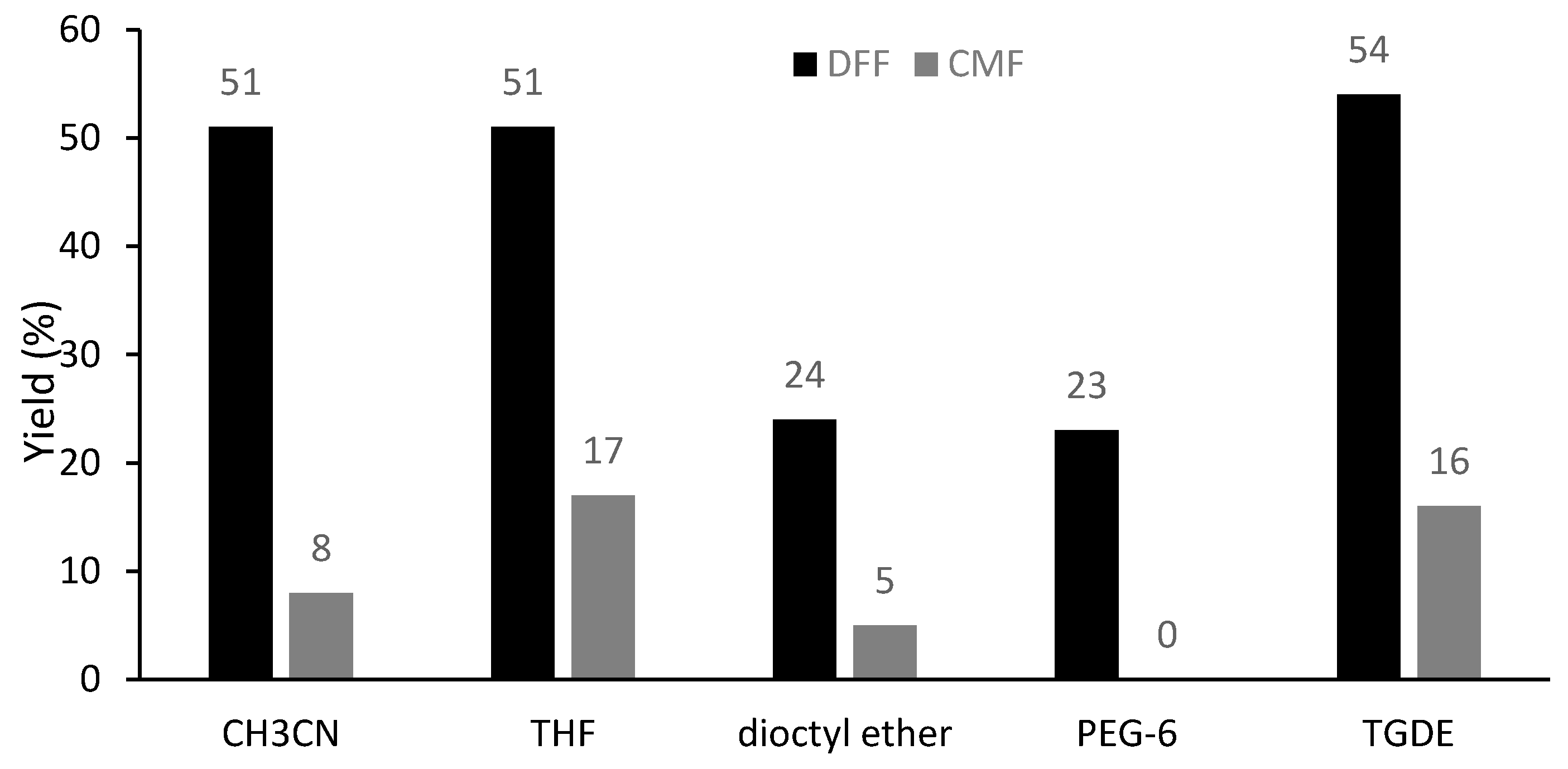
In order to facilitate the flow process, several ethers were tested as solvents for the oxidation of CMF using Cu(OTf)2 as the promoter, including those with high boiling points (b.p. higher than 160 °C) such as dioctyl ether, PEG-6 and triethylene glycol dimethyl ether (TGDE) (Figure 5). Remarkably, TGDE gave a better yield of DFF (54%) than acetonitrile, together with 16% of unreacted CMF. In addition, the oxidation is also feasible using CuSO4 or supported CuSO4 on silica (CuSO4·Si (10%)) in TGDE, yielding DFF in 33% and 44%, respectively (Table S5).
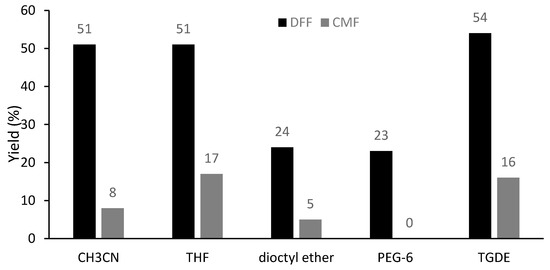
Figure 5.
Effect of the solvent on the oxidation of CMF under microwave irradiation (MWI). Reaction conditions: CMF (0.35 mmol, 50 mg), Cu(OTf)2 (0.5 equiv.), PNO (4 equiv.) in a concentration of 0.5 M at 160 °C for 5 min under MWI; yields were determined by 1H-NMR analysis of the crude reaction mixture using 1,3,5-trimethoxybenzene as an internal standard.
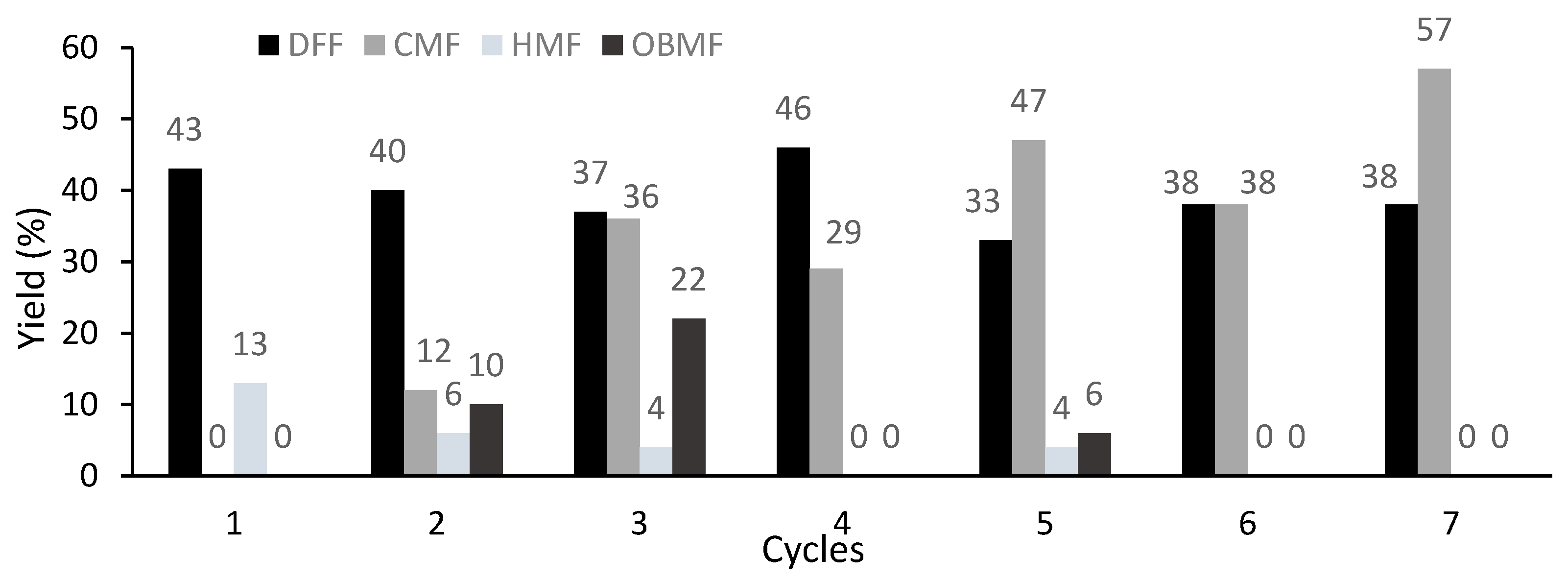
The heterogeneous catalyst CuSO4·Si (10%) was selected to explore catalyst reuse (Figure 6). Remarkably, only a slight erosion of the DFF yield was observed over seven cycles. (1st cycle: 43%; 7th cycle: 38%). In addition, the amount of unreacted CMF increased from cycle to cycle (1st cycle: none; 7th cycle: 57%). Interestingly, for the cycles in which the formation of HMF was observed, formation of the dimer OBMF was also observed, which could arise from the reaction of HMF with CMF.
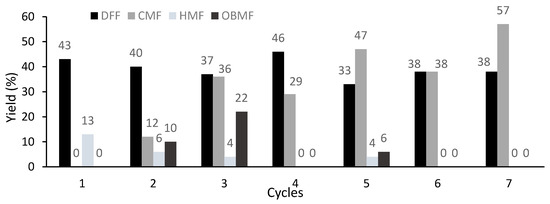
Figure 6.
Catalyst reuse for the oxidation of CMF. Reaction conditions for each cycle: CMF (0.35 mmol, 50 mg), 0.5 equiv. of CuSO4·Si 10%, 4 equiv. of PNO in TGDE (0.5 M) at 160 °C for 5 min under microwave irradiation; yields were determined by 1H-NMR analysis of the crude reaction mixture using 1,3,5-trimethoxybenzene as an internal standard. OBMF: 5,5′-oxybis(5-methylene-2-furaldehyde).
Finally, the oxidation of CMF was explored under flow conditions. Initially, the flow study started by testing the efficiency of the catalyst by performing four oxidation reactions in continuous flow conditions. The selected conditions to perform the flow experiments were based on the best conditions obtained under microwave irradiation and using TGDE (b.p. = 216 °C) as the solvent because it allows the reaction to be performed at 160 °C without causing pressure issues. The other reaction parameters used were: 0.5 mL/min flow rate and 10% (wt/wt) CuSO4 adsorbed on silica catalyst. The yields obtained for each identified product or starting material are presented in Table 4 for four consecutive flow experiments using the same catalyst. It is worth mentioning that in contrast to the results obtained under batch conditions (thermal and MWI), the mass balance is almost complete for all of the experiments, indicating that there are no side products besides the formation of HMF and OBMF. For the first reaction, the yields of DFF and CMF are similar to the ones obtained under MWI or batch conditions using the same solvent, temperature and catalyst, 43% vs. 44% and 15% vs. 22%, respectively (Table 4 vs. Table S5). Under flow conditions, the formation of HMF leading to an almost complete mass balance was observed, while under MWI conditions, the mass balance was only 66%, which suggests that the flow may minimize further transformations such as over oxidation. These polar products are probably absorbed on the silica when the reaction solution is filtered to separate the catalyst from the crude and this could explain the incomplete mass balance for the microwave reactions. Through the consecutive reactions, the yields of DFF, CMF and HMF decrease while the formation of a new product was observed in up to 77%—the dimer OBMF. These results indicate that decreasing the activity of the catalyst results in the formation of HMF that consequently leads to the formation of OBMF.

Table 4.
Oxidation of CMF for four consecutive flow reactions using CuSO4·Si 10%.
Since CMF was not totally consumed, we tried to improve the yield of DFF by using the same flow conditions as previously described, but at 180 °C. However, a lower performance was observed providing yields of 20%, 16%, 43% and 20% of DFF, CMF, HMF and OBMF, respectively, indicating that these conditions are detrimental for the formation of DFF (Table 4 in brackets).
In order to minimize the side reaction of HMF formation, which appeared to be promoted under flow conditions and was probably due to the presence of water, the catalyst was dried for 24 h at 130 °C. However, there was no improvement in the results (Table 5, entry 2 vs. 1). Since the TGDE solvent is highly hygroscopic, the solvent was changed to tetraethylene glycol dimethyl ether (TEGDME, b.p. = 275 °C), that is less hygroscopic, providing only a slightly higher yield of DFF (40% vs. 36% for TGDE, Table 5, entry 3 vs. 2) and only a 17% yield reduction of HMF, although with the formation of OBMF in a 10% yield. Milled hydrated CuSO4 was also tested, resulting in a lower yield of DFF (17%), together with a higher yield of HMF (71%). In addition, an increase in the pressure was observed, which was due to column blocking during operation. In order to circumvent this limitation, a lower amount (5%) of CuSO4 immobilized in silica was tested, resulting in comparable results to the ones observed for 10% CuSO4 in silica (Table 5, entry 5 vs. entry 1). A 1:1 mixture of Cu2O/silica gave only a 37% yield of DFF as the only detected product (Table 5, entry 6). The use of Cu/SiO2 prepared according to a reported procedure [55], gave a 29% yield of DFF and a 49% yield of undesired HMF, which was less efficient then CuSO4 supported in silica. In order to reduce subsequent over oxidation, lower residence times were tested (Table 5, entries 9–10) by increasing the flow rate from 0.5 to 1 mL/min. This condition allowed for the formation of DFF with 42% yield. In addition, increasing the amount of promoter to 1 equiv. gave a 54% yield of DFF (Table 5 entry 10). The CMF loading was also increased by passing 1 g through the column containing the immobilized catalyst Cu/SiO2 and all CMF was consumed, yielding 27% of DFF and 54% of HMF (Table S10).

Table 5.
Oxidation of CMF under flow conditions using different heterogeneous catalysts.
3. Materials and Methods
3.1. General Materials and Methods
Tetrabutylammonium chloride (TBAC), 1,3,5-trimethoxybenzene, urea hydrogen peroxide (UHP), sodium perborate tetrahydrate (NaBO3·4H2O), calcium hypochlorite (Ca(ClO)2), sodium periodate (NaIO4), hydrogen peroxide (H2O2), oxone (2KHSO5·KHSO4·K2SO4), potassium nitrite (KNO2), N-methylmorpholine-N-oxide (NMO), potassium carbonate (K2CO3), potassium bromide (KBr), pyridine N-oxide (PNO), (2,2,6,6-tetramethylpiperidin-1-yl)oxyl (TEMPO), sodium nitrite (NaNO2), p-tolyl sulfoxide, chlorotrimethylsilane, copper(I) chloride (CuCl), copper(II) triflate (Cu(OTf)2), copper(I) triflate toluene complex (Cu(OTf)), copper oxide (Cu2O), and copper(II) sulphate (CuSO4) were purchased from Sigma-Aldrich (Lisboa, Portugal) and used as received unless otherwise stated. Column chromatography was performed using silica gel 60 GF254 Merck Ref. 1.07730.1000 (Darmstadt, Germany). Catalysts were adsorbed or immobilized in silica gel 60 GF254 MercK Ref. 1.07730.1000, aluminium oxide 90 active basic MercK Ref. 101076 (AB), or aluminium oxide 90 active neutral MercK Ref. 101077 (AN). The reagents dioctyl ether (Ref. 249599), tetraethylene glycol dimethyl ether (Ref. 172405) (TEGDME), and triethylene glycol dimethyl ether (Ref. 8.08249.0250) (TGDE) were purchased from Sigma Aldrich and polyglycol 300 (PEG-6) was obtained from Clariant (Artziniega, Spain). All solvents, tetrahydrofuran (THF), acetonitrile, tert-butyl alcohol (t-BuOH), chloroform, ethyl acetate, dichloromethane, and n-hexane were used without further purification.
5-(Hydroxymethyl)furfural (HMF) was synthetized from fructose using the procedure previously reported by our group [57,58]. 5-(chloromethyl)furan-2-carbaldehyde (CMF) was prepared from HMF following a reported procedure [59].
Nuclear magnetic resonance (NMR) spectra were recorded using a Bruker MX300 spectrometer (Rheinstetten, Germany). The microwave experiments were performed on a CEM Discover Benchmat apparatus (CEM Corporation, Matthews, NC, USA) using a glass pressure vessel (10 mL).
3.2. Preparation of Heterogeneous Catalysts
General procedure for the adsorption of CuSO4 in silica, basic alumina or neutral alumina is as follows: the desired solid support (10 g) was added to CuSO4 (1 g) in water (50 mL). After stirring for 10 min, water was removed under vacuum in a rotary evaporator and left in an oven at 130 °C for 24 h. The resulting solid was used without further treatment.
General procedure for the preparation of the Cu/SiO2 immobilized catalysts: the Cu/SiO2 catalysts were prepared according to a reported procedure [55]. Briefly, to prepare a catalyst with x%, NH4OH was added to a solution of Cu(NO3)2·2.5H2O until pH 9. The silica was added and stirred for 20 min. The solution was submerged in an ice bath at 0 °C and diluted with water in order to hydrolyze the copper complex and precipitate the dispersed product. The solid was filtered and washed with water, dried overnight at 110 °C and calcined at 400 °C for 4 h.
Synthesis of SBA-16 and its Mn-, Cu- and V-modified analogs: the parent silica SBA-16 material was synthesized according to the procedure of Hu et al. [56]. Pluronic F127 triblock copolymer and cetyl trimethyl ammonium bromide (CTAB) were used as templates and tetraethylortosilicate (TEOS) was used as a silica source. In a typical synthesis, F127 (1.0 g) and CTAB (0.12 g) were completely dissolved into a solution of 130 mL water and 10 mL concentrated HCl, followed by the addition of 4.0 mL of TEOS under stirring. After 1 h of stirring at 40 °C, the mixture was heated at 80 °C for 24 h under static conditions. The solid product was filtered, washed with water three times and dried at 50 °C. The template was removed by calcination in air at 550 °C for 5 h with a heating rate of 1 °C/min.
Mn, Cu, V-containing (5–20 wt %) SBA-16 materials were prepared by incipient wetness impregnation with manganese (II) acetylacetonate, copper (II) acetylacetonate and vanadyl(VI) sulphate as salt precursors, respectively.
Characterization of the initial and modified SBA-16 materials: the X-ray powder diffraction (XRD) patterns were recorded on a PANalytical X´Pert PRO (HTK) high-resolution diffractometer (The Netherlands) using Cu Kα1 radiation (1.5406 Å) in the 2 θ range from 5° to 80° (100 s per 0.016° step) for the samples and from 10° to 70° (100 s per 0.016° step) for the sample holder using a fully opened X´Celerator detector.
Nitrogen physisorption measurements were carried out at −196 °C using a Tristar 3000 Micromeritics volumetric adsorption analyzer (Ense, Germany). Before the adsorption analysis, the samples were outgassed under vacuum for 2 h at 200 °C in the port of the adsorption analyzer. The Brunauer–Emmett–Teller (BET) specific surface area was calculated from adsorption data in the relative pressure range from 0.05 to 0.21. The pore size distributions (PSDs) were calculated from the nitrogen adsorption data using an algorithm based on the ideas of Barrett, Joyner and Halenda (BJH).
The reducibility of the Cu, Mn, and V-modified samples were investigated by the temperature-programmed reduction (TPR) technique in H2/Ar flow (10:90, 20 mL/min) using a conventional TPR apparatus equipped with a heat conductivity cell and a trap for the removal of released water. Before TPR, run samples were pretreated in oxygen at 350 °C for 1 h.
General procedure for the oxidation of CMF under microwave irradiation (MWI): CMF (0.35 mmol, 50 mg), pyridine N-oxide (4 equiv., 1.4 mmol), Cu(OTf)2 (0.5 equiv.) and CH3CN (0.5 M) were added to a microwave pressure vessel (10 mL). The resulting mixture was allowed to stir at the desired temperature and time. The mixture was filtered through a pad of silica gel using a mixture of hexane/ethyl acetate (1:1) and the solvents were evaporated. Quantification of CMF, DFF, and HMF was performed by 1H-NMR analysis of the crude reaction mixture by using 1,3,5-trimethoxybenzene as an internal standard. DFF and CMF were isolated by column chromatography purification using hexane:ethyl acetate (2:1).
Procedure for the oxidation of CMF and catalyst reuse: CMF (0.35 mmol, 50 mg), pyridine N-oxide (4 equiv., 1.4 mmol), CuSO4·Si (10% w/w, 0.5 equiv.) and TGDE (0.5 M) was added to a microwave pressure vessel (10 mL). The resulting mixture was stirred for 5 min at 160 °C under microwave irradiation. The mixture was filtered first through cotton using a Pasteur pipette (to collect the catalyst) followed by filtration through silica using a mixture of hexane/ethyl acetate 1:1 and the solvents were removed under vacuum. Quantification of CMF, DFF, HMF and (5,5′-(oxybis(methylene))bis(furan-5,2-diyl))dimethanol (OBMF) was performed by 1H-NMR analysis of the crude mixture using 1,3,5-trimethoxybenzene as an internal standard. The collected catalyst was then reused in the next cycle using the same reaction conditions.
General procedure for CMF oxidation using flow conditions: an empty HPLC column (i.d. = 3.9 mm; L = 300 mm) was filled with the respective modified silica and/or promoter and placed inside a gas chromatography (GC) oven. The desired solvent was passed through the column at room temperature using an HPLC pump until the column was completely filled. The temperature of the GC oven was then raised to the desired value and stabilized for 30 min. A solution of CMF (1.56 mmol, 225 mg) and pyridine N-oxide (4 equiv.) in the respective solvent (0.5 M in CMF) was passed through the column under constant flow and the output was sequentially collected in different vials.
Note 1: Quantification by 1H-NMR was performed taking into account the weight of the collected fraction and by adding a specific amount of the internal standard (1,3,5-trimethoxybenzene).
Note 2: The promoter amount was calculated by weighing the column before and after filling with the modified silica. When necessary, the resulting empty volume of the column was filled with silica.
For the cases in which the column was blocked during the operation, the experiment was repeated by filling the end of the column with silica followed by the supported catalyst.
4. Conclusions
This study describes the oxidation of CMF to DFF by exploring a range of oxidants, promoters and reaction conditions. Pyridine N-oxide (PNO) in combination with the Cu based promoter under homogeneous and heterogeneous conditions was identified as the best system. In addition, the heterogeneous catalyst can be reused and applied under flow conditions providing similar performance as obtained under homogeneous conditions.
Supplementary Materials
Supplementary materials are available online.
Acknowledgments
The authors acknowledge the Fundação para a Ciência e a Tecnologia (FCT) (ref. SFRH/BPD/100433/2014, SFRH/BPD/109476/2015, and UID/DTP/04138/2013), and the European Research Area Network; ERANet LAC (ref. ELAC2014/BEE-0341) for financial support; REDE/1518/REM/2005 (FF-UL) for the mass service; Jorge M. C. Sousa e Silva for performing preliminary non-conclusive experiments using gas chromatography; Ágnes Szegedi from the Institute of Materials and Environmental Chemistry, Research Centre for Natural Sciences, Hungarian Academy of Sciences for the XRD and N2 physisorption analysis.
Author Contributions
C.A.M. Afonso, J.A.S. Coelho, and S.P. Simeonov conceived and designed the experiments; J.A.S. Coelho and S.P. Simeonov performed the preliminary experiments; A.I. Vicente performed the main experiments; H.I. Lazarova and M.D. Popova synthesized and characterized the Cu, V, and Mn SBA-16 catalysts. All authors contributed to the manuscript preparation.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Corma, A.; Iborra, S.; Velty, A. Chemical Routes for the Transformation of Biomass into Chemicals. Chem. Rev. 2007, 107, 2411–2502. [Google Scholar] [CrossRef] [PubMed]
- Vispute, T.P.; Zhang, H.; Sanna, A.; Xiao, R.; Huber, G.W. Renewable Chemical Commodity Feedstocks from Integrated Catalytic Processing of Pyrolysis Oils. Science 2010, 330, 1222–1227. [Google Scholar] [CrossRef] [PubMed]
- Takagaki, A.; Nishimura, S.; Ebitani, K. Catalytic Transformations of Biomass-Derived Materials into Value-Added Chemicals. Catal. Surv. Asia 2012, 16, 164–182. [Google Scholar] [CrossRef]
- Christensen, C.H.; Rass-Hansen, J.; Marsden, C.C.; Taarning, E.; Egeblad, K. The Renewable Chemicals Industry. ChemSusChem 2008, 1, 283–289. [Google Scholar] [CrossRef] [PubMed]
- Dutta, S.; De, S.; Saha, B. A Brief Summary of the Synthesis of Polyester Building-Block Chemicals and Biofuels from 5-Hydroxymethylfurfural. ChemPlusChem 2012, 77, 259–272. [Google Scholar] [CrossRef]
- Gandini, A. The irruption of polymers from renewable resources on the scene of macromolecular science and technology. Green Chem. 2011, 13, 1061–1083. [Google Scholar] [CrossRef]
- Lewkowski, J. Synthesis, chemistry and applications of 5-hydroxymethyl-furfural and its derivatives. ARKIVOC 2001, 17–54. [Google Scholar] [CrossRef]
- Rosatella, A.A.; Simeonov, S.P.; Frade, R.F.M.; Afonso, C.A.M. 5-Hydroxymethylfurfural (HMF) as a building block platform: Biological properties, synthesis and synthetic applications. Green Chem. 2011, 13, 754–793. [Google Scholar] [CrossRef]
- Teong, S.P.; Yi, G.; Zhang, Y. Hydroxymethylfurfural production from bioresources: Past, present and future. Green Chem. 2014, 16, 2015–2026. [Google Scholar] [CrossRef]
- Mukherjee, A.; Dumont, M.-J.; Raghavan, V. Review: Sustainable production of hydroxymethylfurfural and levulinic acid: Challenges and opportunities. Biomass Bioenergy 2015, 72, 143–183. [Google Scholar] [CrossRef]
- van Putten, R.-J.; van der Waal, J.C.; de Jong, E.; Rasrendra, C.B.; Heeres, H.J.; de Vries, J.G. Hydroxymethylfurfural, A Versatile Platform Chemical Made from Renewable Resources. Chem. Rev. 2013, 113, 1499–1597. [Google Scholar] [CrossRef] [PubMed]
- Mascal, M.; Nikitin, E.B. Direct, High-Yield Conversion of Cellulose into Biofuel. Angew. Chem. Int. Ed. 2008, 47, 7924–7926. [Google Scholar] [CrossRef] [PubMed]
- Mascal, M.; Nikitin, E.B. Dramatic Advancements in the Saccharide to 5-(Chloromethyl)furfural Conversion Reaction. ChemSusChem 2009, 2, 859–861. [Google Scholar] [CrossRef] [PubMed]
- Mascal, M. 5-(Chloromethyl)furfural is the New HMF: Functionally Equiv.alent But More Practical in Terms of its Production From Biomass. ChemSusChem 2015, 8, 3391–3395. [Google Scholar] [CrossRef] [PubMed]
- Hansen, T.S.; Sádaba, I.; García-Suárez, E.J.; Riisager, A. Cu catalyzed oxidation of 5-hydroxymethylfurfural to 2,5-diformylfuran and 2,5-furandicarboxylic acid under benign reaction conditions. Appl. Catal. A 2013, 456, 44–50. [Google Scholar] [CrossRef]
- Kompanets, M.O.; Kushch, O.V.; Litvinov, Y.E.; Pliekhov, O.L.; Novikova, K.V.; Novokhatko, A.O.; Shendrik, A.N.; Vasilyev, A.V.; Opeida, I.O. Oxidation of 5-hydroxymethylfurfural to 2,5-diformylfuran with molecular oxygen in the presence of N-hydroxyphthalimide. Catal. Commun. 2014, 57, 60–63. [Google Scholar] [CrossRef]
- Le, N.-T.; Lakshmanan, P.; Cho, K.; Han, Y.; Kim, H. Selective oxidation of 5-hydroxymethyl-2-furfural into 2,5-diformylfuran over VO2+ and Cu2+ ions immobilized on sulfonated carbon catalysts. Appl. Catal. A 2013, 464–465, 305–312. [Google Scholar] [CrossRef]
- Saha, B.; Gupta, D.; Abu-Omar, M.M.; Modak, A.; Bhaumik, A. Porphyrin-based porous organic polymer-supported iron(III) catalyst for efficient aerobic oxidation of 5-hydroxymethyl-furfural into 2,5-furandicarboxylic acid. J. Catal. 2013, 299, 316–320. [Google Scholar] [CrossRef]
- Nie, J.; Xie, J.; Liu, H. Efficient aerobic oxidation of 5-hydroxymethylfurfural to 2,5-diformylfuran on supported Ru catalysts. J. Catal. 2013, 301, 83–91. [Google Scholar] [CrossRef]
- Jia, X.; Ma, J.; Wang, M.; Du, Z.; Lu, F.; Wang, F.; Xu, J. Promoted role of Cu(NO3)2 on aerobic oxidation of 5-hydroxymethylfurfural to 2,5-diformylfuran over VOSO4. Appl. Catal. A 2014, 482, 231–236. [Google Scholar] [CrossRef]
- Yadav, G.D.; Sharma, R.V. Biomass derived chemicals: Environmentally benign process for oxidation of 5-hydroxymethylfurfural to 2,5-diformylfuran by using nano-fibrous Ag-OMS-2-catalyst. Appl. Catal. B Environ. 2014, 147, 293–301. [Google Scholar] [CrossRef]
- Zhang, Z.; Yuan, Z.; Tang, D.; Ren, Y.; Lv, K.; Liu, B. Iron Oxide Encapsulated by Ruthenium Hydroxyapatite as Heterogeneous Catalyst for the Synthesis of 2,5-Diformylfuran. ChemSusChem 2014, 7, 3496–3504. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Liu, B.; Huang, K.; Zhang, Z. Aerobic Oxidation of Biomass-Derived 5-(Hydroxymethyl)furfural into 2,5-Diformylfuran Catalyzed by the Trimetallic Mixed Oxide (Co–Ce–Ru). Ind. Eng. Chem. Res. 2014, 53, 1313–1319. [Google Scholar] [CrossRef]
- Wang, S.; Zhang, Z.; Liu, B.; Li, J. Environmentally Friendly Oxidation of Biomass Derived 5-Hydroxymethylfurfural into 2,5-Diformylfuran Catalyzed by Magnetic Separation of Ruthenium Catalyst. Ind. Eng. Chem. Res. 2014, 53, 5820–5827. [Google Scholar] [CrossRef]
- Artz, J.; Palkovits, R. Base-Free Aqueous-Phase Oxidation of 5-Hydroxymethylfurfural over Ruthenium Catalysts Supported on Covalent Triazine Frameworks. ChemSusChem 2015, 8, 3832–3838. [Google Scholar] [CrossRef] [PubMed]
- Han, X.; Geng, L.; Guo, Y.; Jia, R.; Liu, X.; Zhang, Y.; Wang, Y. Base-free aerobic oxidation of 5-hydroxymethylfurfural to 2,5-furandicarboxylic acid over a Pt/C-O-Mg catalyst. Green Chem. 2016, 18, 1597–1604. [Google Scholar] [CrossRef]
- Guo, Y.; Chen, J. Bicomponent Assembly of VO2 and Polyaniline-Functionalized Carbon Nanotubes for the Selective Oxidation of Biomass-Based 5-Hydroxymethylfurfural to 2,5-Diformylfuran. ChemPlusChem 2015, 80, 1760–1768. [Google Scholar] [CrossRef]
- Neațu, F.; Marin, R.S.; Florea, M.; Petrea, N.; Pavel, O.D.; Pârvulescu, V.I. Selective oxidation of 5-hydroxymethyl furfural over non-precious metal heterogeneous catalysts. Appl. Catal. B Environ. 2016, 180, 751–757. [Google Scholar] [CrossRef]
- Mittal, N.; Nisola, G.M.; Malihan, L.B.; Seo, J.G.; Kim, H.; Lee, S.-P.; Chung, W.-J. One-pot synthesis of 2,5-diformylfuran from fructose using a magnetic bi-functional catalyst. RSC Adv. 2016, 6, 25678–25688. [Google Scholar] [CrossRef]
- Li, S.; Su, K.; Li, Z.; Cheng, B. Selective oxidation of 5-hydroxymethylfurfural with H2O2 catalyzed by a molybdenum complex. Green Chem. 2016, 18, 2122–2128. [Google Scholar] [CrossRef]
- Lv, G.; Wang, H.; Yang, Y.; Li, X.; Deng, T.; Chen, C.; Zhu, Y.; Hou, X. Aerobic selective oxidation of 5-hydroxymethyl-furfural over nitrogen-doped graphene materials with 2,2,6,6-tetramethylpiperidin-oxyl as co-catalyst. Catal. Sci. Tech. 2016, 6, 2377–2386. [Google Scholar] [CrossRef]
- Neaţu, F.; Petrea, N.; Petre, R.; Somoghi, V.; Florea, M.; Parvulescu, V.I. Oxidation of 5-hydroxymethyl furfural to 2,5-diformylfuran in aqueous media over heterogeneous manganese based catalysts. Catal. Today 2016, 278, 66–73. [Google Scholar] [CrossRef]
- Liao, L.; Liu, Y.; Li, Z.; Zhuang, J.; Zhou, Y.; Chen, S. Catalytic aerobic oxidation of 5-hydroxymethylfurfural into 2,5-diformylfuran over VO2+ and Cu2+ immobilized on amino-functionalized core-shell magnetic Fe3O4@SiO2. RSC Adv. 2016, 6, 94976–94988. [Google Scholar] [CrossRef]
- Baruah, D.; Hussain, F.L.; Suri, M.; Saikia, U.P.; Sengupta, P.; Dutta, D.K.; Konwar, D. Bi(NO3)3·5H2O and cellulose mediated Cu-NPs—A highly efficient and novel catalytic system for aerobic oxidation of alcohols to carbonyls and synthesis of DFF from HMF. Catal. Commun. 2016, 77, 9–12. [Google Scholar] [CrossRef]
- Ghosh, K.; Molla, R.A.; Iqubal, M.A.; Islam, S.S.; Islam, S.M. Ruthenium nanoparticles supported on N-containing mesoporous polymer catalyzed aerobic oxidation of biomass-derived 5-hydroxymethylfurfural (HMF) to 2,5-diformylfuran (DFF). Appl. Catal. A 2016, 520, 44–52. [Google Scholar] [CrossRef]
- Wang, F.; Jiang, L.; Wang, J.; Zhang, Z. Catalytic Conversion of Fructose and 5-Hydroxymethylfurfural into 2,5-Diformylfuran over SBA-15 Supported Ruthenium Catalysts. Energy Fuels 2016, 30, 5885–5892. [Google Scholar] [CrossRef]
- Zhang, W.; Xie, J.; Hou, W.; Liu, Y.; Zhou, Y.; Wang, J. One-Pot Template-Free Synthesis of Cu–MOR Zeolite toward Efficient Catalyst Support for Aerobic Oxidation of 5-Hydroxymethylfurfural under Ambient Pressure. ACS Appl. Mater. Interfaces 2016, 8, 23122–23132. [Google Scholar] [CrossRef] [PubMed]
- Laugel, C.; Estrine, B.; Le Bras, J.; Hoffmann, N.; Marinkovic, S.; Muzart, J. NaBr/DMSO-Induced Synthesis of 2,5-Diformylfuran from Fructose or 5-(Hydroxymethyl)furfural. ChemCatChem 2014, 6, 1195–1198. [Google Scholar] [CrossRef]
- Liu, R.; Chen, J.; Chen, L.; Guo, Y.; Zhong, J. One-Step Approach to 2,5-Diformylfuran from Fructose by Using a Bifunctional and Recyclable Acidic Polyoxometalate Catalyst. ChemPlusChem 2014, 79, 1448–1454. [Google Scholar] [CrossRef]
- Liu, Y.; Zhu, L.; Tang, J.; Liu, M.; Cheng, R.; Hu, C. One-pot, One-step Synthesis of 2,5-Diformylfuran from Carbohydrates over Mo-Containing Keggin Heteropolyacids. ChemSusChem 2014, 7, 3541–3547. [Google Scholar] [CrossRef] [PubMed]
- Xu, F.; Zhang, Z. Polyaniline-Grafted VO(acac)2: An Effective Catalyst for the Synthesis of 2,5-Diformylfuran from 5-Hydroxymethylfurfural and Fructose. ChemCatChem 2015, 7, 1470–1477. [Google Scholar] [CrossRef]
- Mittal, N.; Nisola, G.M.; Malihan, L.B.; Seo, J.G.; Lee, S.-P.; Chung, W.-J. Metal-free mild oxidation of 5-hydroxymethylfurfural to 2,5-diformylfuran. Korean J. Chem. Eng. 2014, 31, 1362–1367. [Google Scholar] [CrossRef]
- Mittal, N.; Nisola, G.M.; Seo, J.G.; Lee, S.-P.; Chung, W.-J. Organic radical functionalized SBA-15 as a heterogeneous catalyst for facile oxidation of 5-hydroxymethylfurfural to 2,5-diformylfuran. J. Mol. Catal. A Chem. 2015, 404–405, 106–114. [Google Scholar] [CrossRef]
- Lv, G.; Wang, H.; Yang, Y.; Deng, T.; Chen, C.; Zhu, Y.; Hou, X. Graphene Oxide: A Convenient Metal-Free Carbocatalyst for Facilitating Aerobic Oxidation of 5-Hydroxymethylfurfural into 2, 5-Diformylfuran. ACS Catal. 2015, 5, 5636–5646. [Google Scholar] [CrossRef]
- Qin, Y.-Z.; Li, Y.-M.; Zong, M.-H.; Wu, H.; Li, N. Enzyme-catalyzed selective oxidation of 5-hydroxymethylfurfural (HMF) and separation of HMF and 2,5-diformylfuran using deep eutectic solvents. Green Chem. 2015, 17, 3718–3722. [Google Scholar] [CrossRef]
- Dutta, S.; Wu, L.; Mascal, M. Production of 5-(chloromethyl)furan-2-carbonyl chloride and furan-2,5-dicarbonyl chloride from biomass-derived 5-(chloromethyl)furfural (CMF). Green Chem. 2015, 17, 3737–3739. [Google Scholar] [CrossRef]
- Hashemi, M.M.; Beni, Y.A. Copper(I) Chloride/Kieselguhr: A Versatile Catalyst for Oxidation of Alkyl Halides and Alkyl Tosylates to the Carbonyl Compounds. J. Chem. Res. Synop. 1999, 434–435. [Google Scholar] [CrossRef]
- Chandrasekhar, S.; Sridhar, M. A bifunctional approach towards the mild oxidation of organic halides: 2-Dimethylamino-N,N-dimethylaniline N-oxide. Tetrahedron Lett. 2000, 41, 5423–5425. [Google Scholar] [CrossRef]
- Badri, R.; Soleymani, M. Selective Oxidation of Benzylic Substrates to Their Corresponding Carbonyl Compounds with 3,6-Bis(Triphenylphosphonium)cyclohexene Peroxodisulfate. Synth. Commun. 2003, 33, 1325–1332. [Google Scholar] [CrossRef]
- Das, S.; Panigrahi, A.K.; Maikap, G.C. NaIO4–DMF: A novel reagent for the oxidation of organic halides to carbonyl compounds. Tetrahedron Lett. 2003, 44, 1375–1377. [Google Scholar] [CrossRef]
- Hu, Y.L.; Liu, Q.F.; Lu, T.T.; Lu, M. Highly efficient oxidation of organic halides to aldehydes and ketones with H5IO6 in ionic liquid [C12mim][FeCl4]. Catal. Commun. 2010, 11, 923–927. [Google Scholar] [CrossRef]
- Liu, Q.; Lu, M.; Yang, F.; Wei, W.; Sun, F.; Yang, Z.; Huang, S. Aerobic Oxidation of Benzylic Halides to Carbonyl Compounds with Molecular Oxygen Catalyzed by TEMPO/KNO2 in Aqueous Media. Synth. Commun. 2010, 40, 1106–1114. [Google Scholar] [CrossRef]
- Khumraksa, B.; Phakhodee, W.; Pattarawarapan, M. Rapid oxidation of organic halides with N-methylmorpholine N-oxide in an ionic liquid under microwave irradiation. Tetrahedron Lett. 2013, 54, 1983–1986. [Google Scholar] [CrossRef]
- Bayat, A.; Shakourian-Fard, M.; Ramezanpour, S.; Hashemi, M.M. A green procedure for direct oxidation of organic halides to aldehydes and ketones catalyzed by a molybdate-based catalyst. New J. Chem. 2015, 39, 3845–3851. [Google Scholar] [CrossRef]
- Carniti, P.; Gervasini, A.; Modica, V.H.; Ravasio, N. Catalytic selective reduction of NO with ethylene over a series of copper catalysts on amorphous silicas. Appl. Catal. B Environ. 2000, 28, 175–185. [Google Scholar] [CrossRef]
- Hu, Y.; Zhi, Z.; Zhao, Q.; Wu, C.; Zhao, P.; Jiang, H.; Jiang, T.; Wang, S. 3D cubic mesoporous silica microsphere as a carrier for poorly soluble drug carvedilol. Microporous Mesoporous Mater. 2012, 147, 94–101. [Google Scholar] [CrossRef]
- Simeonov, S.P.; Coelho, J.A.S.; Afonso, C.A.M. An Integrated Approach for the Production and Isolation of 5-Hydroxymethylfurfural from Carbohydrates. ChemSusChem 2012, 5, 1388–1391. [Google Scholar] [CrossRef] [PubMed]
- Svilen, P.; Simeonov, J.A.S.C.; Afonso, C.A.M. Synthesis of 5-(Hydroxymethyl)furfural (HMF). Org. Synth. 2016, 93, 29–36. [Google Scholar]
- Sanda, K.; Rigal, L.; Gaset, A. Synthèse du 5-bromométhyl-et du 5-chlorométhyl-2-furannecarboxaldéhyde. Carbohydr. Res. 1989, 187, 15–23. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds HMF, CMF, DFF and OBMF are available from the authors.
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).