Antiplasmodial Activity, Cytotoxicity and Structure-Activity Relationship Study of Cyclopeptide Alkaloids
Abstract
:1. Introduction
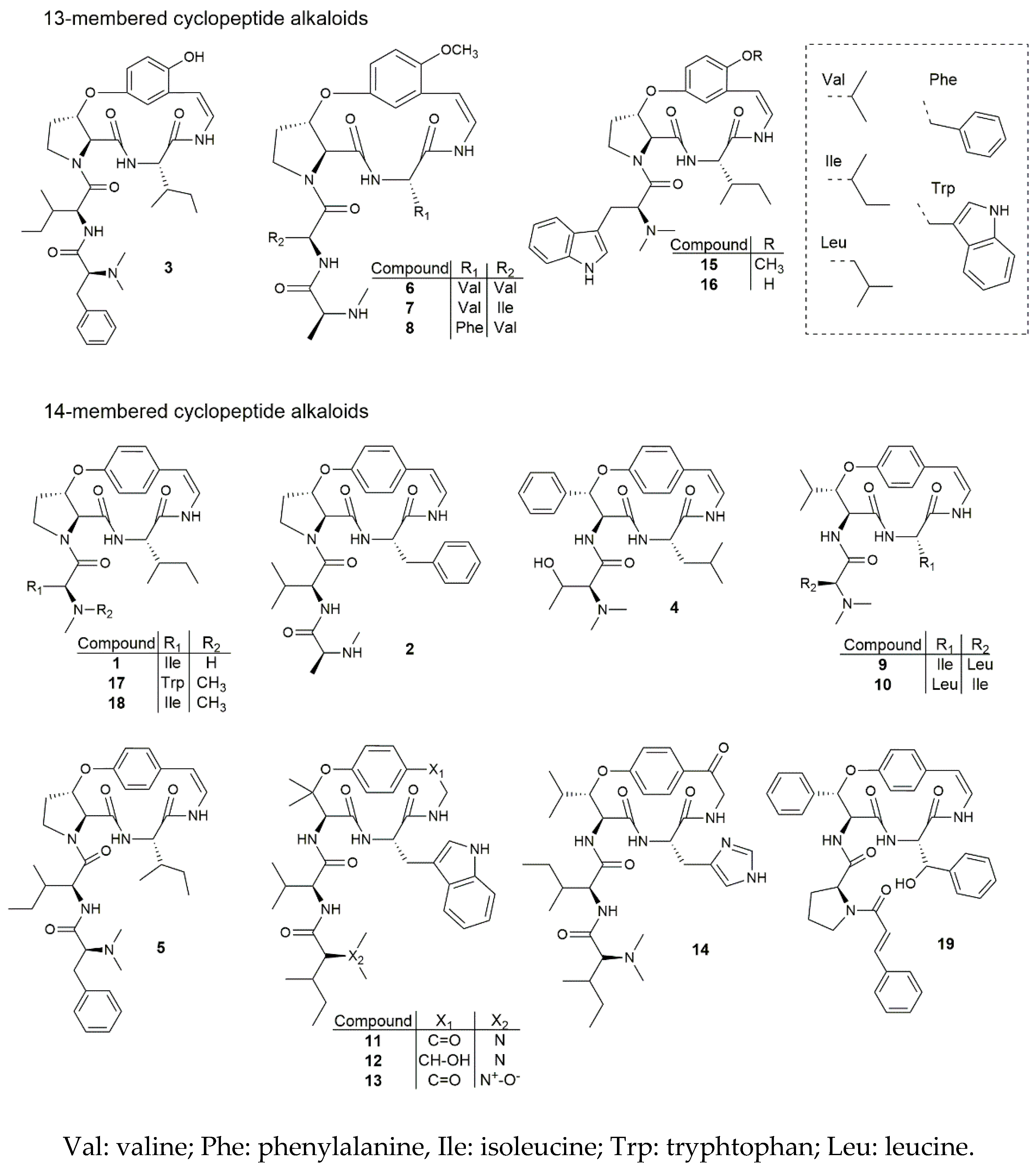
2. Results and Discussion
2.1. Antiplasmodial and Cytotoxic Activities
2.2. Qualitative Structure-Activity Relationship Study
2.3. Quantitative Structure-Activity Relationship Study
3. Materials and Methods
3.1. General Experimental Procedures
3.2. Plant Material, Extraction, and Isolation
3.3. Antiplasmodial and Cytotoxic Activities
3.4. QSAR
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- WHO. World Malaria Report 2015; WHO Press: Geneva, Switzerland, 2015. [Google Scholar]
- Harvey, A.L. Natural products in drug discovery. Drug Discov. Today 2008, 13, 894–901. [Google Scholar] [CrossRef] [PubMed]
- Vonthron-Senecheau, C.; Weniger, B.; Ouattara, M.; Bi, F.T.; Kamenan, A.; Lobstein, A.; Brun, R.; Anton, R. In vitro antiplasmodial activity and cytotoxicity of ethnobotanically selected Ivorian plants. J. Ethnopharmacol. 2003, 87, 221–225. [Google Scholar] [CrossRef]
- Suksamrarn, S.; Suwannapoch, N.; Aunchai, N.; Kuno, M.; Ratananukul, P.; Haritakun, R.; Jansakul, C.; Ruchirawat, S. Ziziphine N, O, P and Q, new antiplasmodial cyclopeptide alkaloids from Ziziphus oenoplia var. Brunoniana. Tetrahedron 2005, 61, 1175–1180. [Google Scholar] [CrossRef]
- Panseeta, P.; Lomchoey, K.; Prabpai, S.; Kongsaeree, P.; Suksamrarn, A.; Ruchirawat, S.; Suksamrarn, S. Antiplasmodial and antimycobacterial cyclopeptide alkaloids from the root of Ziziphus mauritiana. Phytochemistry 2011, 72, 909–915. [Google Scholar] [CrossRef] [PubMed]
- Tuenter, E.; Exarchou, V.; Balde, A.; Cos, P.; Maes, L.; Apers, S.; Pieters, L. Cyclopeptide alkaloids from Hymenocardia acida. J. Nat. Prod. 2016, 79, 1746–1751. [Google Scholar] [CrossRef] [PubMed]
- Tuenter, E.; Ahmad, R.; Foubert, K.; Amin, A.; Cos, P.; Maes, L.; Apers, S.; Pieters, L.; Exarchou, V. Isolation and structure elucidation by LC-DAD-MS and LC-DAD-SPE-NMR of cyclopeptide alkaloids from the roots of Ziziphus oxyphylla and evaluation of their antiplasmodial activity. J. Nat. Prod. 2016, 79, 2865–2872. [Google Scholar] [CrossRef] [PubMed]
- Tuenter, E.; Exarchou, V.; Apers, S.; Pieters, L. Cyclopeptide alkaloids. Phytochem. Rev. 2016, 1–15. [Google Scholar] [CrossRef]
- Westerhuis, J.A.; de Jong, S.; Smilde, A.K. Direct orthogonal signal correction. Chemometr. Intell. Lab. 2001, 56, 13–25. [Google Scholar] [CrossRef]
- Kang, K.B.; Ming, G.; Kim, G.J.; Ha, T.K.Q.; Choi, H.; Oh, W.K.; Sung, S.H. Jubanines F-J, cyclopeptide alkaloids from the roots of Ziziphus jujuba. Phytochemistry 2015, 119, 90–95. [Google Scholar] [CrossRef] [PubMed]
- Tuenter, E.; Foubert, K.; Staerk, D.; Apers, S.; Pieters, L. Isolation and structure elucidation of cyclopeptide alkaloids from Ziziphus nummularia and Ziziphus spina-christi by HPLC-DAD-MS and HPLC-PDA-(HRMS)-SPE-NMR. Phytochemistry. submitted for publication.
- Trevisan, G.; Maldaner, G.; Velloso, N.A.; Sant'Anna, G.D.; Ilha, V.; Gewehr, C.D.V.; Rubin, M.A.; Morel, A.F.; Ferreira, J. Antinociceptive effects of 14-membered cyclopeptide alkaloids. J. Nat. Prod. 2009, 72, 608–612. [Google Scholar] [CrossRef] [PubMed]
- Medina, R.P.; Schuquel, I.T.A.; Pomini, A.M.; Silva, C.C.; Oliveira, C.M.A.; Kato, L.; Nakamura, C.V.; Santin, S.M.O. Ixorine, a new cyclopeptide alkaloid from the branches of Ixora brevifolia. J. Braz. Chem. Soc. 2016, 27, 753–758. [Google Scholar]
- Cos, P.; Vlietinck, A.J.; Vanden Berghe, D.; Maes, L. Anti-infective potential of natural products: How to develop a stronger in vitro “proof-of-concept”. J. Ethnopharmacol. 2006, 106, 290–302. [Google Scholar] [CrossRef] [PubMed]
- Mesia, K.; Cimanga, R.K.; Dhooghe, L.; Cos, P.; Apers, S.; Totte, J.; Tona, G.L.; Pieters, L.; Vlietinck, A.J.; Maes, L. Antimalarial activity and toxicity evaluation of a quantified Nauclea pobeguinii extract. J. Ethnopharmacol. 2010, 131, 10–16. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Samples of the compounds are not available from the authors.
| Compound | Plant Source | P. falciparum K1 IC50 (μM) | MRC-5 IC50 (μM) | |
|---|---|---|---|---|
| 1 | Nummularine-U | Z. nummularia | 23.0 ± 8.5 | >64.0 |
| 2 | Mauritine-F | 34.2 ± 9.14 | >64.0 | |
| 3 | Spinanine-B | Z. spina-christi | 2.1 ± 0.3 | >64.0 |
| 4 | Nummularine-E | >64.0 | >64.0 | |
| 5 | Amphibine-D | 8.9 ± 1.5 | >64.0 | |
| 6 | Jubanine-F | Z. jujuba | 12.8 ± 2.9 | >64.0 |
| 7 | Jubanine-G | 4.7 ± 2.4 | >64.0 | |
| 8 | Nummularine-B | 3.6 ± 1.3 | >64.0 | |
| 9 | Adouetine-X | 7.5 ± 1.8 | 19.1 ± 11.9 | |
| 10 | Frangulanine | H. dulcis | 14.9 ± 5.2 | 30.6 ± 6.5 |
| 11 | Hymenocardine | H. acida | 16.4 ± 6.8 | 51.1 ± 17.2 |
| 12 | Hymenocardinol | 17.5 ± 8.7 | >64.0 | |
| 13 | Hymenocardine N-oxide | 12.2 ± 6.6 | >64.0 | |
| 14 | Hymenocardine-H | 27.9 ± 16.5 | >64.0 | |
| 15 | Nummularine-R | Z. oxyphylla | 3.2 ± 2.6 | 30.6 ± 4.0 |
| 16 | O-desmethylnummularine-R | 7.1 ± 1.6 | >64.0 | |
| 17 | Hemsine-A | 13.6 ± 9.3 | >64.0 | |
| 18 | Ramosine-A | >32.0 | >64.0 | |
| 19 | Oxyphylline-F | 7.4 ± 3.0 | 31.2 ± 1.4 | |
| Control 1 | Chloroquine | 0.15 | nt | |
| Control 2 | Tamoxifen | nt | 10.0 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tuenter, E.; Segers, K.; Kang, K.B.; Viaene, J.; Sung, S.H.; Cos, P.; Maes, L.; Heyden, Y.V.; Pieters, L. Antiplasmodial Activity, Cytotoxicity and Structure-Activity Relationship Study of Cyclopeptide Alkaloids. Molecules 2017, 22, 224. https://doi.org/10.3390/molecules22020224
Tuenter E, Segers K, Kang KB, Viaene J, Sung SH, Cos P, Maes L, Heyden YV, Pieters L. Antiplasmodial Activity, Cytotoxicity and Structure-Activity Relationship Study of Cyclopeptide Alkaloids. Molecules. 2017; 22(2):224. https://doi.org/10.3390/molecules22020224
Chicago/Turabian StyleTuenter, Emmy, Karen Segers, Kyo Bin Kang, Johan Viaene, Sang Hyun Sung, Paul Cos, Louis Maes, Yvan Vander Heyden, and Luc Pieters. 2017. "Antiplasmodial Activity, Cytotoxicity and Structure-Activity Relationship Study of Cyclopeptide Alkaloids" Molecules 22, no. 2: 224. https://doi.org/10.3390/molecules22020224
APA StyleTuenter, E., Segers, K., Kang, K. B., Viaene, J., Sung, S. H., Cos, P., Maes, L., Heyden, Y. V., & Pieters, L. (2017). Antiplasmodial Activity, Cytotoxicity and Structure-Activity Relationship Study of Cyclopeptide Alkaloids. Molecules, 22(2), 224. https://doi.org/10.3390/molecules22020224






