Recovery of Oil with Unsaturated Fatty Acids and Polyphenols from Chaenomelessinensis (Thouin) Koehne: Process Optimization of Pilot-Scale Subcritical Fluid Assisted Extraction
Abstract
:1. Introduction
2. Results and Discussion
2.1. Optimization of Processing Conditions for Subcritical Fluid Extraction to Recover Oil from C. sinensis Seeds
2.2. RSM Analysis
2.3. Verification Experiment
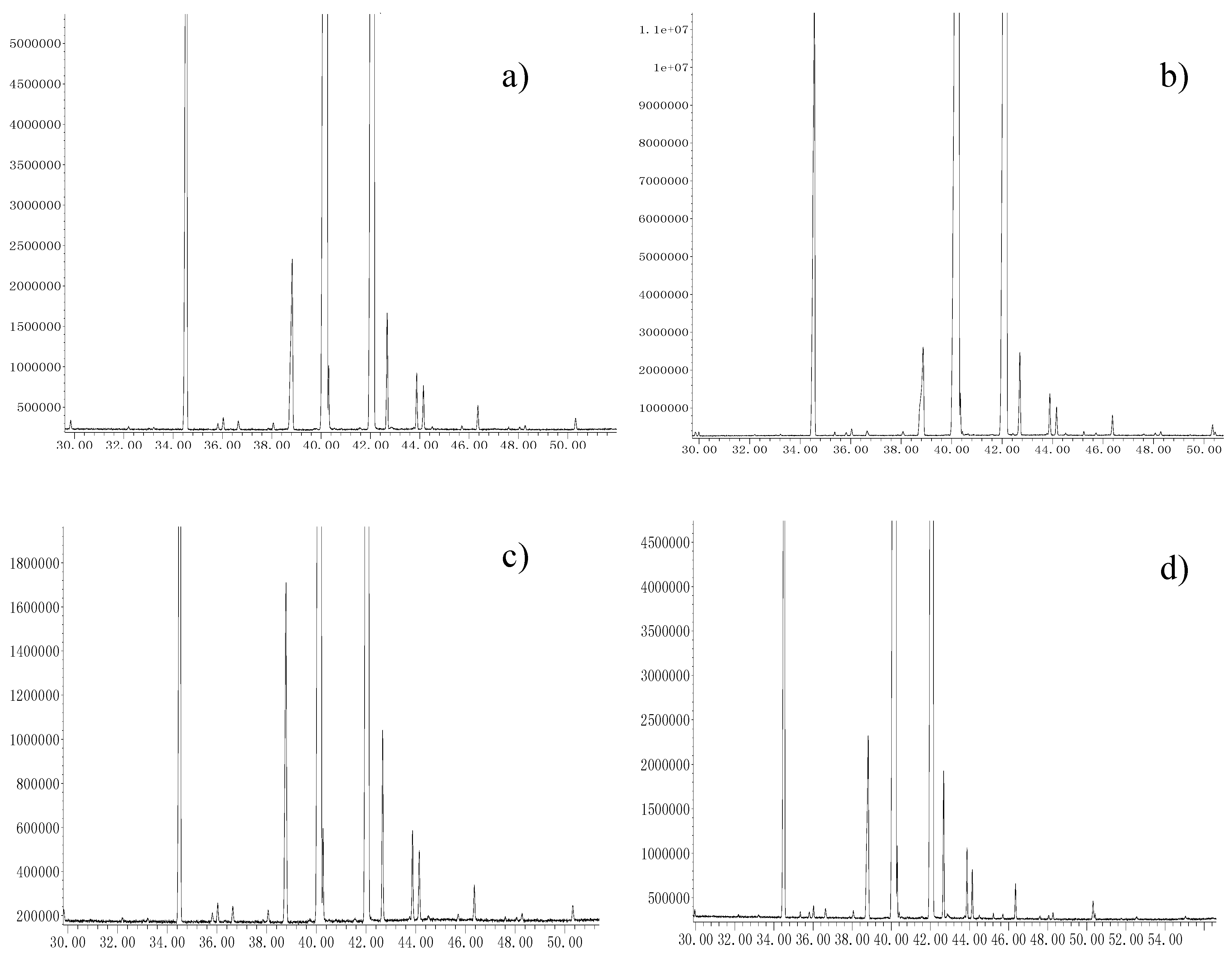
2.4. Oil Extraction Recovery from C. sinensis Seeds after Applying Soxhlet, Cold-Pressing, Microwave and Subcritical Fluid Extraction Methodologies
2.5. Fatty Acid Composition in Oil Recovered from C. sinensis Seeds after Applying Soxhlet, Cold-Pressing, Microwave and Subcritical Fluid Extraction Methodologies
3. Materials and Methods
3.1. Materials and Chemicals
3.2. Oil extraction Methodologies
3.2.1. Soxhlet Extraction (SE)
3.2.2. Cold-Pressed Extraction (CPE)
3.2.3. Microwave-Assisted Extraction (MAE)
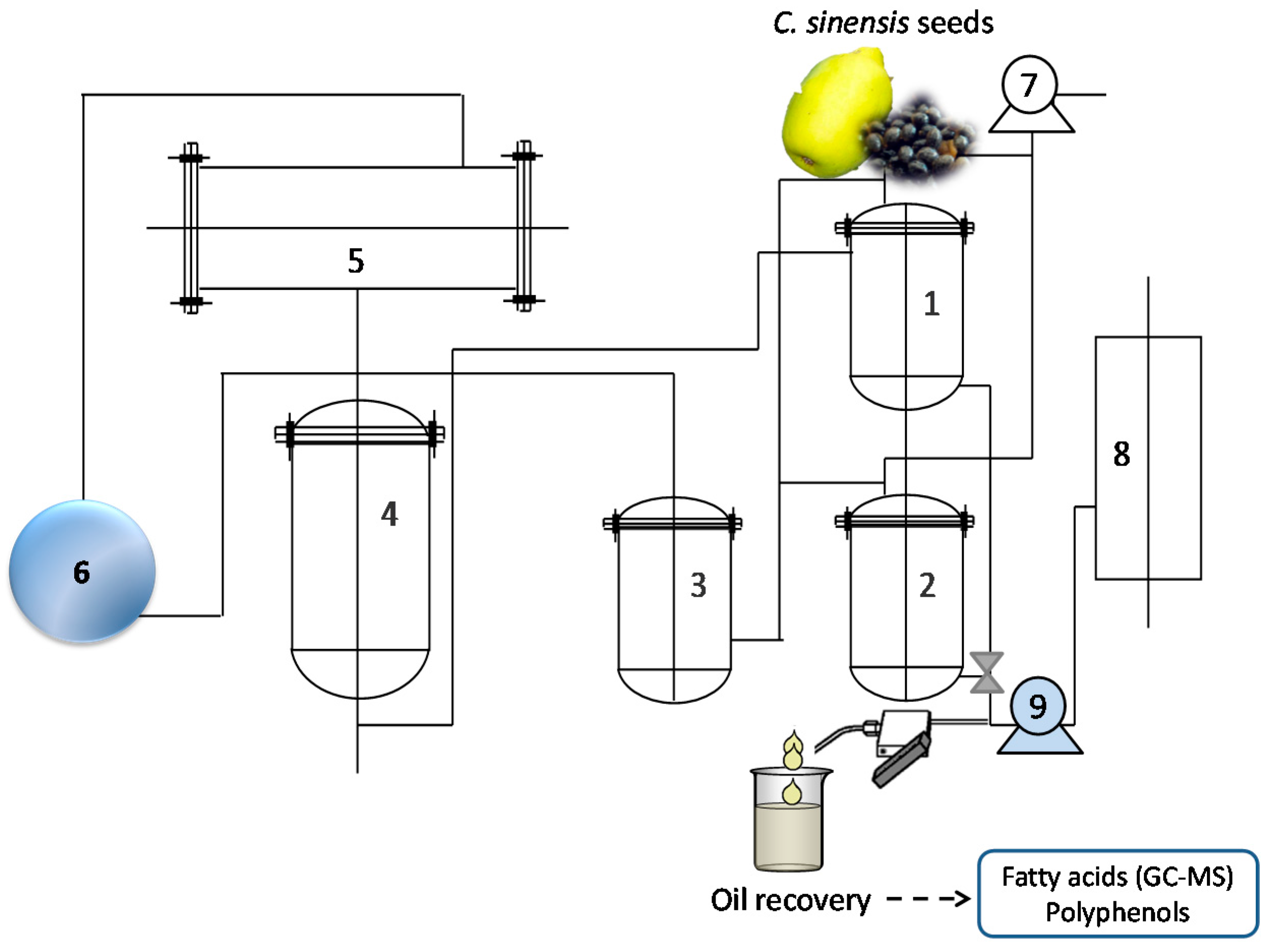
3.2.4. Subcritical Fluid Extraction (SbFE)
Response Surface Methodology Experimental Design to Optimize the Recovery of Oil Using Subcritical Fluid Extraction
3.2.5. Calculation of the Oil Extraction Yield and Recovery Rate
3.3. Fatty Acid Composition Analysis by GC–MS
3.4. Physicochemical Properties and Determination of Polyphenols
3.5. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- McClements, D.J.; Decker, E.A.; Park, Y.; Weiss, J. Structural Design Principles for Delivery of Bioactive Components in Nutraceuticals and Functional Foods. Crit. Rev. Food Sci. 2009, 49, 577–606. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Liu, T.; Chen, X.; Chen, L.; Zhang, W.; Wang, J.; Gao, L.; Chen, Y.; Peng, X. Subcritical co-solvents extraction of lipid from wet microalgae pastes of Nannochloropsis sp. Eur. J. Lipid Sci. Technol. 2012, 114, 205–212. [Google Scholar] [CrossRef] [PubMed]
- Barba, F.J.; Esteve, M.J.; Frigola, A. Impact of high-pressure processing on vitamin E (alpha-, gamma-, and delta-tocopherol), vitamin D (cholecalciferol and ergocalciferol), and fatty acid profiles in liquid foods. J. Agric. Food. Chem. 2012, 60, 3763–3768. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Chen, P.; He, J.; Deng, L.; Wang, L.; Lei, J.; Rong, L. Extraction of oil from Jatropha curcas seeds by subcritical fluid extraction. Ind. Crop. Prod. 2014, 62, 235–241. [Google Scholar] [CrossRef]
- Venturi, F.; Sanmartin, C.; Taglieri, I.; Nari, A.; Andrich, G.; Terzuoli, E.; Donnini, S.; Nicolella, C.; Zinnai, A. Development of Phenol-Enriched Olive Oil with Phenolic Compounds Extracted from Wastewater Produced by Physical Refining. Soc. Sci. Electron. Publ. 2017, 9, 916. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Liu, X. Fatty acid composition and physiochemical properties of Decaisnea fargesii Franch seed oil. China Oils Fats 2011, 36, 78–79. [Google Scholar]
- Giacoppo, S.; Galuppo, M.; Lombardo, G.E.; Ulaszewska, M.M.; Mattivi, F.; Bramanti, P.; Mazzon, E.; Navarra, M. Neuroprotective effects of a polyphenolic white grape juice extract in a mouse model of experimental autoimmune encephalomyelitis. Fitoterapia 2015, 103, 171–186. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Mei, L.; Wang, Q.; Shao, Y.; Tao, Y. Optimization of subcritical fluid extraction of seed oil from Nitraria tangutorum using response surface methodology. LWT-Food Sci. Technol. 2014, 56, 168–174. [Google Scholar] [CrossRef]
- Ly, C.; Yockell-Lelievre, J.; Ferraro, Z.M.; Arnason, J.T.; Ferrier, J.; Gruslin, A. The effects of dietary polyphenols on reproductive health and early development. Hum. Reprod. Update 2015, 21, 228–248. [Google Scholar] [CrossRef] [PubMed]
- Zinnai, A.; Sanmartin, C.; Taglieri, I.; Andrich, G.; Venturi, F. Supercritical fluid extraction from microalgae with high content of LC-PUFAs. A case of study: Sc-CO2 oil extraction from Schizochytrium sp. J. Supercrit. Fluids 2016, 116, 126–131. [Google Scholar] [CrossRef]
- Du, H.; Wu, J.; Li, H.; Zhong, P.X.; Xu, Y.J.; Li, C.H.; Ji, K.X.; Wang, L.S. Polyphenols and triterpenes from Chaenomeles fruits: Chemical analysis and antioxidant activities assessment. Food Chem. 2013, 141, 4260–4268. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.-D.; Li, H.-P.; Liu, L.-Q.; Li, Q.; Li, Y.-F. Analysis of Nutritional Components in the Seed of Chinese Flowering Quince Fruit. Acta Nutr. Sin. 2008, 1, 111–112. [Google Scholar]
- Tsukui, A.; Santos Junior, H.M.; Oigman, S.S.; de Souza, R.O.; Bizzo, H.R.; Rezende, C.M. Microwave-assisted extraction of green coffee oil and quantification of diterpenes by HPLC. Food Chem. 2014, 164, 266–271. [Google Scholar] [CrossRef] [PubMed]
- Ivanov, D.S.; Čolović, R.R.; Lević, J.D.; Sredanović, S.A. Optimization of supercritical fluid extraction of linseed oil using RSM. Eur. J. Lipid Sci. Technol. 2012, 114, 807–815. [Google Scholar] [CrossRef]
- Rapinel, V.; Rombaut, N.; Rakotomanomana, N.; Vallageas, A.; Cravotto, G.; Chemat, F. An original approach for lipophilic natural products extraction: Use of liquefied n-butane as alternative solvent to n-hexane. LWT-Food Sci. Tech. 2016, 85, 524–533. [Google Scholar] [CrossRef]
- Wang, L.; Wu, M.; Liu, H.M.; Ma, Y.X.; Wang, X.D.; Qin, G.Y. Subcritical Fluid Extraction of Chinese Quince Seed: Optimization and Product Characterization. Molecules 2017, 22, 528. [Google Scholar] [CrossRef] [PubMed]
- Khuri, A.I.; Cornell, J.A. Response surfaces: Designs and analyses. Technometrics 1987, 31, 214–273. [Google Scholar]
- Zeković, Z.; Vidović, S.; Vladić, J.; Radosavljević, R.; Cvejin, A.; Elgndi, M.A.; Pavlić, B. Optimization of subcritical water extraction of antioxidants from Coriandrum sativum seeds by response surface methodology. J. Supercri. Fluid. 2014, 95, 560–566. [Google Scholar] [CrossRef]
- Wu, J.; Yu, D.; Sun, H.; Zhang, Y.; Zhang, W.; Meng, F.; Du, X. Optimizing the extraction of anti-tumor alkaloids from the stem of Berberis amurensis by response surface methodology. Ind. Crop. Prod. 2015, 69, 68–75. [Google Scholar] [CrossRef]
- Jia, K.; Feng, X.; Liu, K.; Han, Y.; Xue, Y.; Xue, C. Development of a subcritical fluid extraction and GC-MS validation method for polychlorinated biphenyls (PCBs) in marine samples. J. Chromatogr. B 2013, 923–924, 37–42. [Google Scholar] [CrossRef] [PubMed]
- Dong, X.Y.; Zhong, J.; Wei, F.; Lv, X.; Wu, L.; Lei, Y.; Liao, B.S.; Quek, S.Y.; Chen, H. Triacylglycerol Composition Profiling and Comparison of High-Oleic and Normal Peanut Oils. J. Am. Oil Chem. Soc. 2015, 92, 233–242. [Google Scholar] [CrossRef]
- Harhar, H.; Gharby, S.; Kartah, B.; Pioch, D.; Guillaume, D.; Charrouf, Z. Effect of harvest date of Argania spinosa fruits on Argan oil quality. Ind. Crop. Prod. 2014, 56, 156–159. [Google Scholar] [CrossRef]
- Akintunde, A.M.; Ajala, S.O.; Betiku, E. Optimization of Bauhinia monandra seed oil extraction via artificial neural network and response surface methodology: A potential biofuel candidate. Ind. Crop. Prod. 2015, 67, 387–394. [Google Scholar] [CrossRef]
- Cerchiara, T.; Chidichimo, G.; Ragusa, M.I.; Belsito, E.L.; Liguori, A.; Arioli, A. Characterization and utilization of Spanish Broom (Spartium junceum L.) seed oil. Ind. Crop. Prod. 2010, 31, 423–426. [Google Scholar] [CrossRef]
- Yang, J.; Huang, Q.; Huang, F.; Deng, Q.; Zheng, C. Adsorption refining technology of cold-pressed linseed oil. China Oils Fats 2012, 9, 19–22. [Google Scholar]
- Li, M.; Ngadi, M.O.; Ma, Y. Optimisation of pulsed ultrasonic and microwave-assisted extraction for curcuminoids by response surface methodology and kinetic study. Food Chem. 2014, 165, 29–34. [Google Scholar] [CrossRef] [PubMed]
- Apolinario, A.C.; de Lima Damasceno, B.P.; de Macedo Beltrao, N.E.; Pessoa, A.; Converti, A.; da Silva, J.A. Inulin-type fructans: A review on different aspects of biochemical and pharmaceutical technology. Carbohydr. Polym. 2014, 101, 368–378. [Google Scholar] [CrossRef] [PubMed]
- Roselló-Soto, E.; Galanakis, C.M.; Brnčić, M.; Orlien, V.; Trujillo, F.J.; Mawson, R.; Knoerzer, K.; Tiwari, B.K.; Barba, F.J. Clean recovery of antioxidant compounds from plant foods, by-products and algae assisted by ultrasounds processing. Modeling approaches to optimize processing conditions. Trends Food Sci. Technol. 2015, 42, 134–149. [Google Scholar] [CrossRef]
- Xu, X.; Mu, H.; Høy, C.E.; Adler-Nissen, J. Production of specifically structured lipids by enzymatic interesterification in a pilot enzyme bed reactor: Process optimization by response surface methodology. Eur. J. Lipid Sci. Technol. 1999, 101, 207–214. [Google Scholar] [CrossRef]
- Zhao, S.; Zhang, D. A parametric study of supercritical carbon dioxide extraction of oil from Moringa oleifera seeds using a response surface methodology. Sep. Purif. Technol. 2013, 113, 9–17. [Google Scholar] [CrossRef]
- Ma, Q.; Fan, X.-D.; Liu, X.-C.; Qiu, T.-Q.; Jiang, J.-G. Ultrasound-enhanced subcritical water extraction of essential oils from Kaempferia galangal L. and their comparative antioxidant activities. Sep. Purif. Technol. 2015, 150, 73–79. [Google Scholar] [CrossRef]
- Singleton, V.L.; Orthofer, R.; Lamuela-Raventós, R.M. Analysis of total phenols and other oxidation substrates and antioxidants by means of folin-ciocalteu reagent. Methods Enzymol. 1999, 299, 152–178. [Google Scholar]
- Zheng, Y.-Y.; Li, C.; Huang, X.-Y. Study on the content determination of total flavonoids in Olea europaea L. leaves. Spectrosc. Spect. Anal. 2011, 31, 547–550. [Google Scholar]
Sample Availability: Samples of the compounds are available from the authors. |
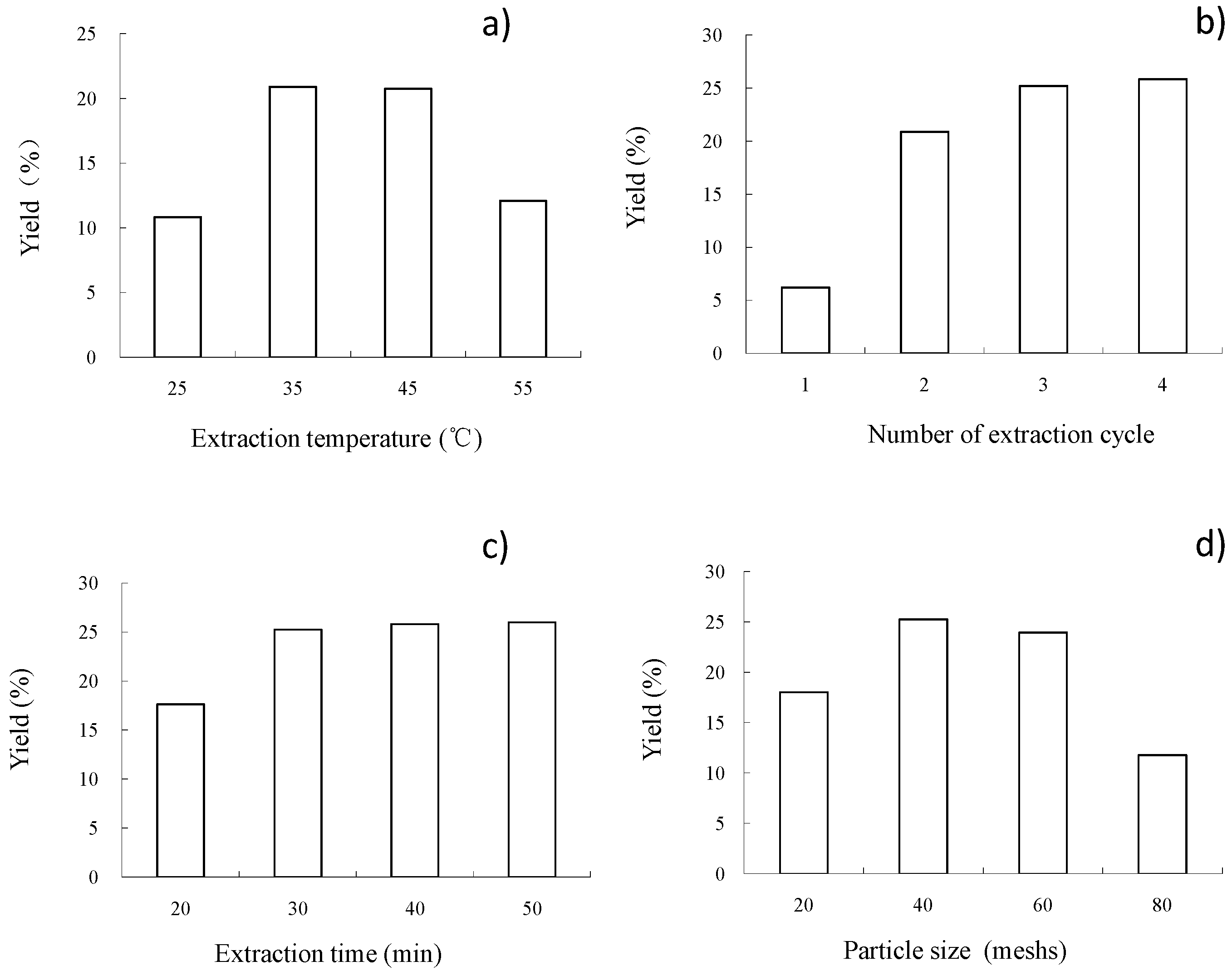
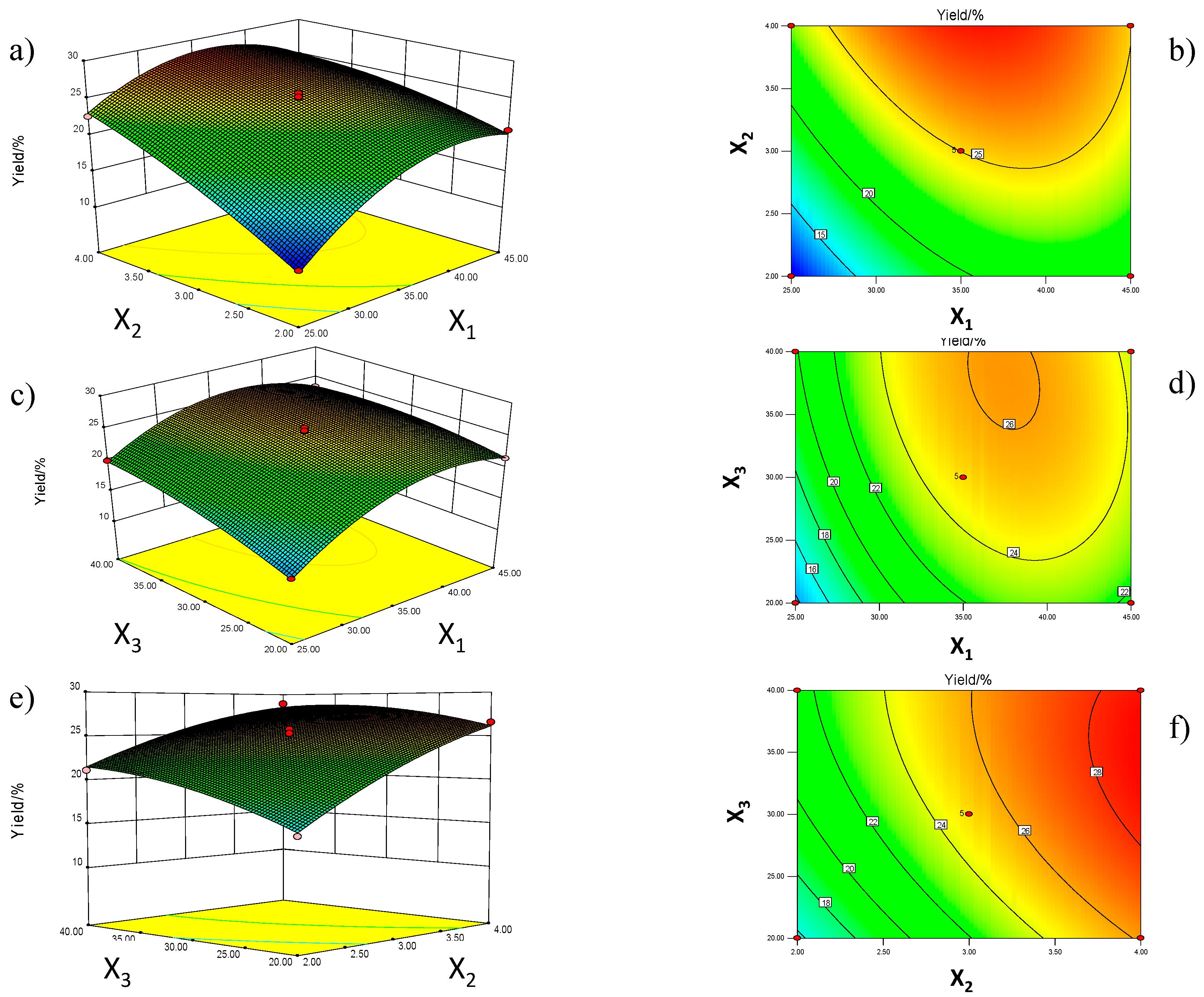
| Run | Coded Variables | Decoded Variables | Extraction Yield (%) | |||||
|---|---|---|---|---|---|---|---|---|
| X1 | X2 | X3 | X1 a | X2 b | X3 c | Experimental Value | Predicted Value | |
| 1 | −1 | −1 | 0 | 25 | 2 | 30 | 10.84 | 10.54 |
| 2 | 1 | −1 | 0 | 45 | 2 | 30 | 20.75 | 20.26 |
| 3 | −1 | 1 | 0 | 25 | 4 | 30 | 22.61 | 23.10 |
| 4 | 1 | 1 | 0 | 45 | 4 | 30 | 24.78 | 25.09 |
| 5 | −1 | 0 | −1 | 25 | 3 | 20 | 13.74 | 13.65 |
| 6 | 1 | 0 | −1 | 45 | 3 | 20 | 21.57 | 21.67 |
| 7 | −1 | 0 | 1 | 25 | 3 | 40 | 19.93 | 19.83 |
| 8 | 1 | 0 | 1 | 45 | 3 | 40 | 23.44 | 23.53 |
| 9 | 0 | −1 | −1 | 35 | 2 | 20 | 15.28 | 15.67 |
| 10 | 0 | 1 | −1 | 35 | 4 | 20 | 26.56 | 26.16 |
| 11 | 0 | −1 | 1 | 35 | 2 | 40 | 21.08 | 21.48 |
| 12 | 0 | 1 | 1 | 35 | 4 | 40 | 28.78 | 28.39 |
| 13 | 0 | 0 | 0 | 35 | 3 | 30 | 25.73 | 25.02 |
| 14 | 0 | 0 | 0 | 35 | 3 | 30 | 23.70 | 25.02 |
| 15 | 0 | 0 | 0 | 35 | 3 | 30 | 25.23 | 25.02 |
| 16 | 0 | 0 | 0 | 35 | 3 | 30 | 25.18 | 25.02 |
| 17 | 0 | 0 | 0 | 35 | 3 | 30 | 25.26 | 25.02 |
| Parameter | Coefficient Estimate | Standard Error | Sum of Squares | Degree of Freedom | Mean Square | F-Value | p-Value * |
|---|---|---|---|---|---|---|---|
| Model | 366.00 | 9 | 40.67 | 76.80 | <0.0001 | ||
| Intercept | 25.02 | 0.33 | 1 | ||||
| X1 | 2.93 | 0.33 | 68.56 | 1 | 68.56 | 129.48 | <0.0001 |
| X2 | 4.35 | 0.26 | 151.21 | 1 | 151.21 | 285.56 | <0.0001 |
| X3 | 2.01 | 0.26 | 32.32 | 1 | 32.32 | 61.04 | 0.0001 |
| X1X2 | −1.94 | 0.36 | 14.98 | 1 | 14.98 | 28.28 | 0.0011 |
| X1X3 | −1.08 | 0.36 | 4.67 | 1 | 4.67 | 8.81 | 0.0209 |
| X2X3 | −0.89 | 0.36 | 3.20 | 1 | 3.20 | 6.05 | 0.0435 |
| X12 | −4.27 | 0.35 | 76.59 | 1 | 76.59 | 144.65 | <0.0001 |
| X22 | −1.01 | 0.35 | 4.30 | 1 | 4.30 | 8.11 | 0.0248 |
| X32 | −1.08 | 0.35 | 4.96 | 1 | 4.96 | 9.36 | 0.0183 |
| Residual | 3.71 | 7 | 0.53 | ||||
| Lack of fit | 1.33 | 3 | 0.44 | 0.75 | 0.5773 | ||
| Pure error | 2.37 | 4 | 0.59 | ||||
| Cor Total | 369.71 | 16 | |||||
| R2 = 0.9900 | Adj R2 = 0.9771 | ||||||
| C.V.% = 3.30 | Pred R2 = 0.9323 | ||||||
| PRESS = 25.03 | Adeq precision = 31.988 | ||||||
| Method | Extraction Yield (%) | Recovery Rate (%) |
|---|---|---|
| SE | 29.0 ± 0.78 a | 100.00 |
| SbFE | 25.79 ± 0.06 b | 88.93 |
| MAE (ethyl acetate) | 24.6 ± 0.52 c | 84.83 |
| CPE | 19.0 ± 1.47 d | 65.52 |
| Fatty Acids, Methyl Ester | Abbreviated Formula | Characteristic Ions | Molecular Weight | Relative Percentage (%) | |||
|---|---|---|---|---|---|---|---|
| SE | MAE | CPE | SbFE | ||||
| Dodecanoic acid | C12:0 | 74, 171, 214 | 200 | - | - | - | 0.01 |
| Tetradecanoic acid | C14:0 | 74, 199, 242 | 228 | - | - | - | 0.02 |
| Hexadecanoic acid | C16:0 | 74, 227, 270 | 256 | 9.76 | 8.36 | 9.61 | 11.21 |
| Octadecanoic acid | C18:0 | 74, 255, 298 | 284 | 2.22 | 2.24 | 2.22 | - |
| 9-Octadecenoic acid | C18:1 | 55, 264, 296 | 282 | 43.81 | 44.65 | 44.38 | 46.09 |
| 9,12-Octadecadienoic acid | C18:2 | 67, 263, 294 | 280 | 42.45 | 42.7 | 42.53 | 41.79 |
| 9,12,15-Octadecatrienoic acid | C18:3 | 69, 261, 292 | 278 | 0.33 | 0.33 | - | 0.35 |
| Eicosanoic acid | C20:0 | 74, 283, 326 | 312 | 0.85 | 1.02 | 0.85 | - |
| 11-Eicosenoic acid | C20:1 | 55, 292, 324 | 310 | 0.39 | 0.46 | 0.41 | 0.52 |
| Docosanoic acid | C22:0 | 74, 143, 354 | 340 | 0.19 | 0.23 | - | - |
| Monounsaturated fatty acids (MUFA) | 44.20 | 45.11 | 44.79 | 46.61 | |||
| Polyunsaturated fatty acids (PUFA) | 42.78 | 43.03 | 42.53 | 42.14 | |||
| Unsaturated fatty acids | 86.99 | 88.14 | 87.32 | 88.75 | |||
| Symbol | Independent Variable | Coded Levels | ||
|---|---|---|---|---|
| −1 | 0 | 1 | ||
| X1 | Extraction temperature (°C) | 25 | 35 | 45 |
| X2 | Number of extraction cycle | 2 | 3 | 4 |
| X3 | Extraction time (min) | 20 | 30 | 40 |
| Run | Coded Variables | Decoded Variables | Extraction Yield (%) | |||||
|---|---|---|---|---|---|---|---|---|
| X1 | X2 | X3 | X1 a | X2 b | X3 c | Experimental Value | Predicted Value | |
| 1 | −1 | −1 | 0 | 25 | 2 | 30 | 10.84 | 10.54 |
| 2 | 1 | −1 | 0 | 45 | 2 | 30 | 20.75 | 20.26 |
| 3 | −1 | 1 | 0 | 25 | 4 | 30 | 22.61 | 23.10 |
| 4 | 1 | 1 | 0 | 45 | 4 | 30 | 24.78 | 25.09 |
| 5 | −1 | 0 | −1 | 25 | 3 | 20 | 13.74 | 13.65 |
| 6 | 1 | 0 | −1 | 45 | 3 | 20 | 21.57 | 21.67 |
| 7 | −1 | 0 | 1 | 25 | 3 | 40 | 19.93 | 19.83 |
| 8 | 1 | 0 | 1 | 45 | 3 | 40 | 23.44 | 23.53 |
| 9 | 0 | −1 | −1 | 35 | 2 | 20 | 15.28 | 15.67 |
| 10 | 0 | 1 | −1 | 35 | 4 | 20 | 26.56 | 26.16 |
| 11 | 0 | −1 | 1 | 35 | 2 | 40 | 21.08 | 21.48 |
| 12 | 0 | 1 | 1 | 35 | 4 | 40 | 28.78 | 28.39 |
| 13 | 0 | 0 | 0 | 35 | 3 | 30 | 25.73 | 25.02 |
| 14 | 0 | 0 | 0 | 35 | 3 | 30 | 23.70 | 25.02 |
| 15 | 0 | 0 | 0 | 35 | 3 | 30 | 25.23 | 25.02 |
| 16 | 0 | 0 | 0 | 35 | 3 | 30 | 25.18 | 25.02 |
| 17 | 0 | 0 | 0 | 35 | 3 | 30 | 25.26 | 25.02 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhu, Z.; Zhang, R.; Zhan, S.; He, J.; Barba, F.J.; Cravotto, G.; Wu, W.; Li, S. Recovery of Oil with Unsaturated Fatty Acids and Polyphenols from Chaenomelessinensis (Thouin) Koehne: Process Optimization of Pilot-Scale Subcritical Fluid Assisted Extraction. Molecules 2017, 22, 1788. https://doi.org/10.3390/molecules22101788
Zhu Z, Zhang R, Zhan S, He J, Barba FJ, Cravotto G, Wu W, Li S. Recovery of Oil with Unsaturated Fatty Acids and Polyphenols from Chaenomelessinensis (Thouin) Koehne: Process Optimization of Pilot-Scale Subcritical Fluid Assisted Extraction. Molecules. 2017; 22(10):1788. https://doi.org/10.3390/molecules22101788
Chicago/Turabian StyleZhu, Zhenzhou, Rui Zhang, Shaoying Zhan, Jingren He, Francisco J. Barba, Giancarlo Cravotto, Weizhong Wu, and Shuyi Li. 2017. "Recovery of Oil with Unsaturated Fatty Acids and Polyphenols from Chaenomelessinensis (Thouin) Koehne: Process Optimization of Pilot-Scale Subcritical Fluid Assisted Extraction" Molecules 22, no. 10: 1788. https://doi.org/10.3390/molecules22101788
APA StyleZhu, Z., Zhang, R., Zhan, S., He, J., Barba, F. J., Cravotto, G., Wu, W., & Li, S. (2017). Recovery of Oil with Unsaturated Fatty Acids and Polyphenols from Chaenomelessinensis (Thouin) Koehne: Process Optimization of Pilot-Scale Subcritical Fluid Assisted Extraction. Molecules, 22(10), 1788. https://doi.org/10.3390/molecules22101788








