Electrospun Phospholipid Fibers as Micro-Encapsulation and Antioxidant Matrices
Abstract
1. Introduction
2. Results and Discussion
2.1. Morphology
2.2. FTIR Analysis
2.3. Encapsulation Efficiency (EE)
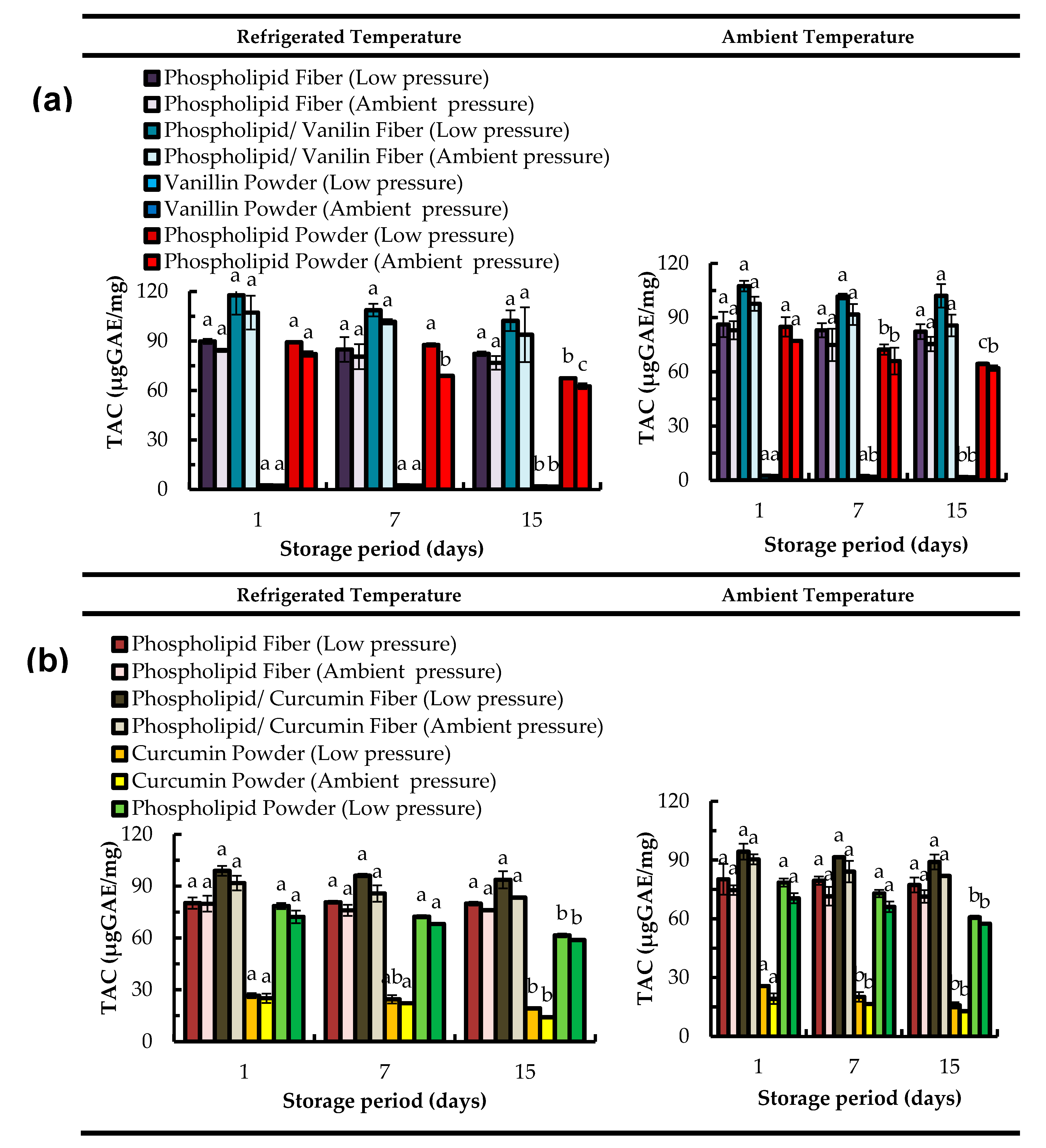
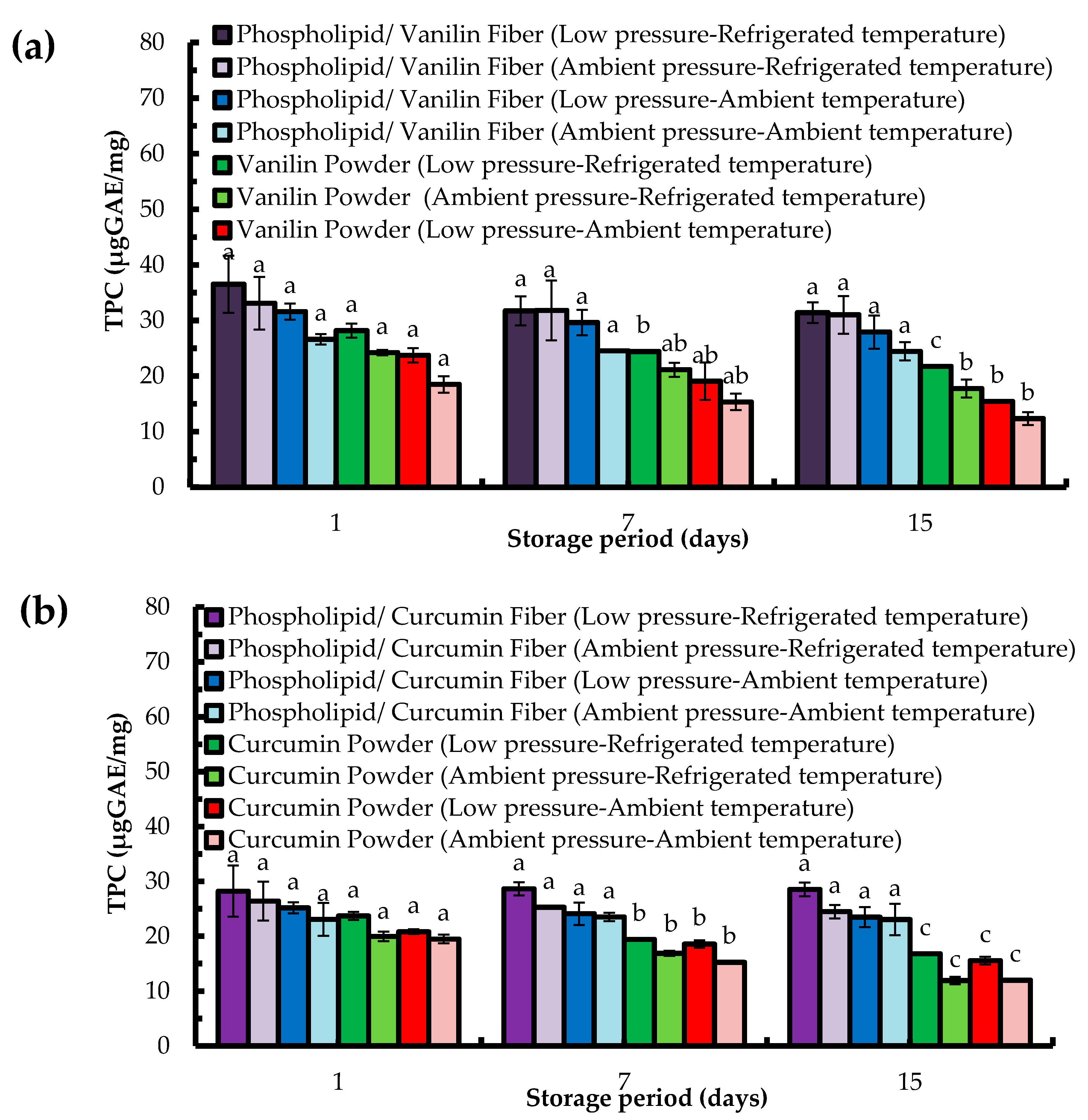
2.4. Total Antioxidant Capacity (TAC) Assay
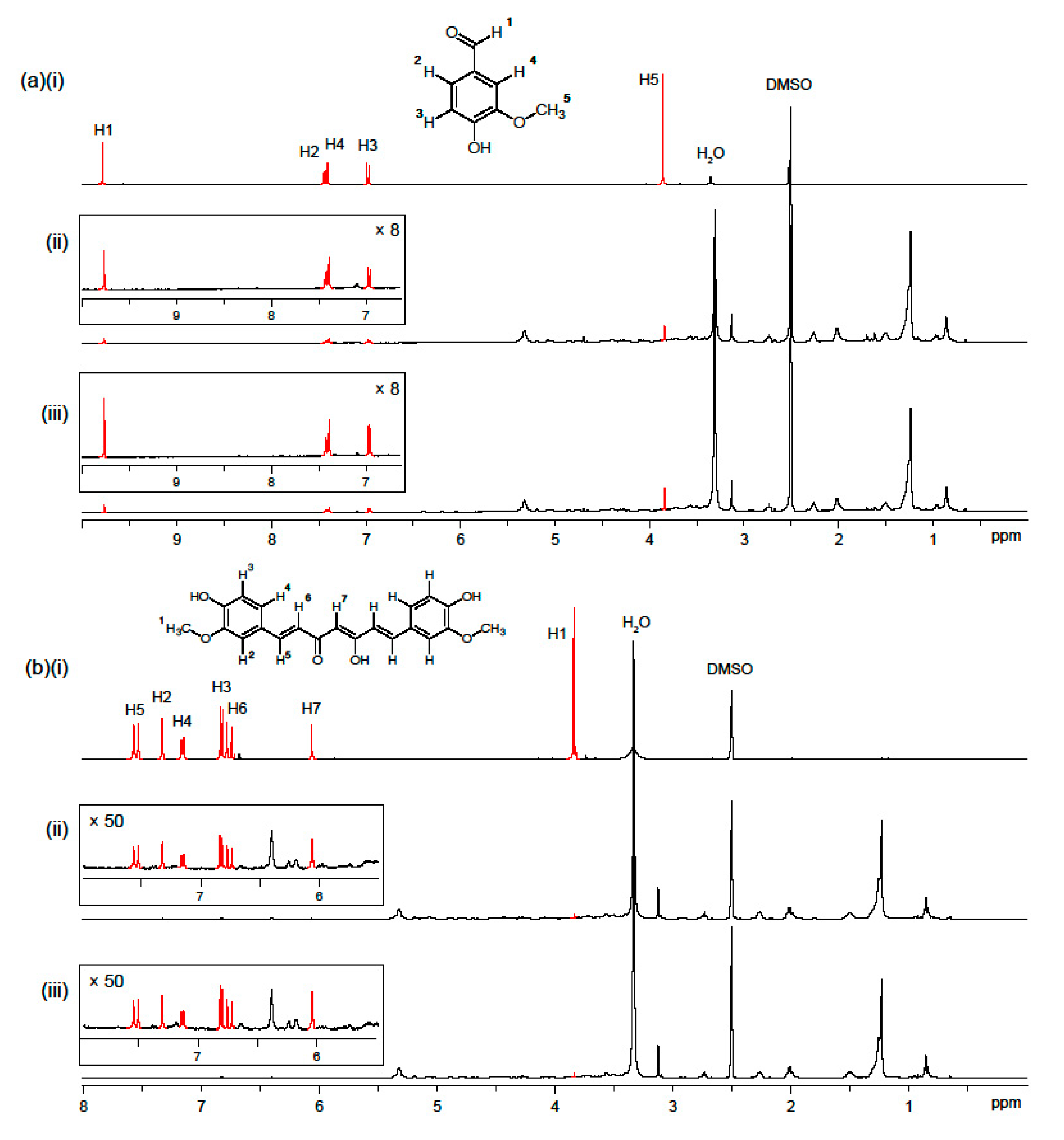
2.5. Stability of Phenolic Compounds Test under Storage by 1H-NMR
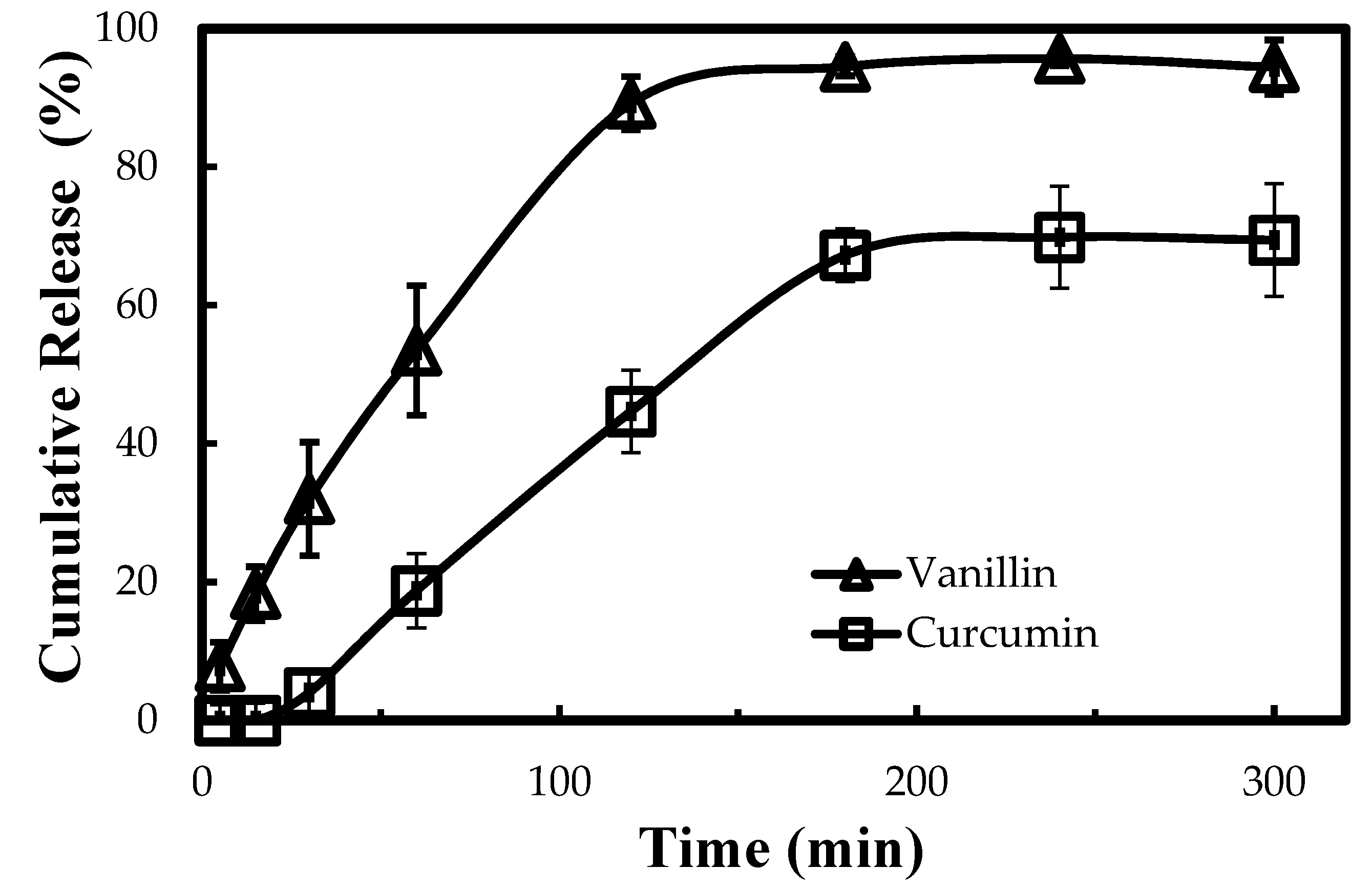
2.6. In Vitro Release Study
3. Materials and Methods
3.1. Materials
3.2. Preparation of Electrospinning Solutions
3.3. Electrospinning Processing
3.4. Morphology
3.5. Fourier Transform Infrared (FTIR) Spectroscopy
3.6. Encapsulation Efficiency
3.7. Total Antioxidant Capacity Assay (TAC)
3.8. Total Phenolic Content (TPC)
3.9. Stability of Phenolic Compounds Test under Storage by 1H-NMR Spectroscopy
3.10. In Vitro Release Study
3.11. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Gosangari, S.L.; Watkin, K.L. Effect of preparation techniques on the properties of curcumin liposomes: Characterization of size, release and cytotoxicity on a squamous oral carcinoma cell line. Pharm. Dev. Technol. 2012, 17, 103–109. [Google Scholar] [CrossRef] [PubMed]
- Maria, A.; Maria, B.; Sinico, C.; Sapienza, L.; Moro, P.A. Phospholipid-detergent systems: Effects of polysorbates on the release of liposomal caffeine. Farmaco 1998, 53, 650–654. [Google Scholar]
- Shazly, G.; Nawroth, T.; Langguth, P. Comparison of dialysis and dispersion methods for in vitro release determination of drugs from multilamellar liposomes. Dissolut. Technol. 2008, 15, 7–10. [Google Scholar] [CrossRef]
- Hühn, E.; Buchholz, H.-G.; Shazly, G.; Maus, S.; Thews, O.; Bausbacher, N.; Rösch, F.; Schreckenberger, M.; Langguth, P. Predicting the in vivo release from a liposomal formulation by IVIVC and non-invasive positron emission tomography imaging. Eur. J. Pharm. Sci. 2010, 41, 71–77. [Google Scholar] [CrossRef] [PubMed]
- Huynh, N.T.; Passirani, C.; Saulnier, P.; Benoit, J.P. Lipid nanocapsules: A new platform for nanomedicine. Int. J. Pharm. 2009, 379, 201–209. [Google Scholar] [CrossRef] [PubMed]
- Mendes, A.C.; Baran, E.T.; Reis, R.L.; Azevedo, H.S. Fabrication of phospholipid-xanthan microcapsules by combining microfluidics with self-assembly. Acta Biomater. 2013, 9, 6675–6685. [Google Scholar] [CrossRef] [PubMed]
- Taylor, T.M.; Weiss, J.; Davidson, P.M.; Bruce, B.D. Liposomal nanocapsules in food science and agriculture. Crit. Rev. Food Sci. Nutr. 2005, 45, 587–605. [Google Scholar] [CrossRef] [PubMed]
- Mouritsen, O.G. Life—As A Matter of Fat: The Emerging Science of Lipidomics; Springer: Heidelberg, Germany, 2005; ISBN 3540232486. [Google Scholar]
- Mouritsen, O.G. Lipids, curvature, and nano-medicine. Eur. J. Lipid Sci. Technol. 2011, 113, 1174–1187. [Google Scholar] [CrossRef] [PubMed]
- McKee, M.G.; Layman, J.M.; Cashion, M.P.; Long, T.E. Phospholipid nonwoven electrospun membranes. Science 2006, 311, 353–355. [Google Scholar] [CrossRef] [PubMed]
- Jørgensen, L.; Qvortrup, K.; Chronakis, I.S. Phospholipid electrospun nanofibers: Effect of solvents and co-axial processing on morphology and fiber diameter. RSC Adv. 2015, 5, 53644–53652. [Google Scholar] [CrossRef]
- Mendes, A.C.; Nikogeorgos, N.; Lee, S.; Chronakis, I.S. Nanomechanics of Electrospun Phospholipid Fiber. Appl. Phys. Lett. 2015, 106. [Google Scholar] [CrossRef]
- Mendes, A.C.; Gorzelanny, C.; Halter, N.; Schneider, S.W.; Chronakis, I.S. Hybrid electrospun chitosan-phospholipids nanofibers for transdermal drug delivery. Int. J. Pharm. 2016, 510, 48–56. [Google Scholar] [CrossRef] [PubMed]
- Mendes, A.C.; Shekarforoush, E.; Engwer, C.; Beeren, S.R.; Gorzelanny, C.; Goycoolea, F.M.; Chronakis, I.S. Co-assembly of chitosan and phospholipids into hybrid hydrogels. Pure Appl. Chem. 2016, 88, 905–916. [Google Scholar] [CrossRef]
- De Sousa, R.S.; de Moraes Nogueira, A.O.; Marques, V.G.; Clementin, R.M.; de Lima, V.R. Effects of α-eleostearic acid on asolectin liposomes dynamics: Relevance to its antioxidant activity. Bioorg. Chem. 2013, 51, 8–15. [Google Scholar] [CrossRef] [PubMed]
- Pan, Y.; Tikekar, R.V.; Nitin, N. Effect of antioxidant properties of lecithin emulsifier on oxidative stability of encapsulated bioactive compounds. Int. J. Pharm. 2013, 450, 129–137. [Google Scholar] [CrossRef] [PubMed]
- Judde, A.; Villeneuve, P.; Rossignol-Castera, A.; Guillou, A. Antioxidant effect of soy lecithins on vegetable oil stability and their synergism with tocopherols. J. Am. Oil Chem. Soc. 2003, 80, 1209–1215. [Google Scholar] [CrossRef]
- Cui, L.; Decker, E.A. Phospholipids in foods: Prooxidants or antioxidants? J. Sci. Food Agric. 2016, 96, 18–31. [Google Scholar] [CrossRef] [PubMed]
- Choe, E.; Min, D.B. Mechanisms of Antioxidants in the Oxidation of Foods. Compr. Rev. Food Sci. Food Saf. 2009, 8, 345–358. [Google Scholar] [CrossRef]
- Doert, M.; Jaworska, K.; Moersel, J.T.; Kroh, L.W. Synergistic effect of lecithins for tocopherols: Lecithin-based regeneration of α-tocopherol. Eur. Food Res. Technol. 2012, 235, 915–928. [Google Scholar] [CrossRef]
- Brewer, M.S. Natural Antioxidants: Sources, Compounds, Mechanisms of Action, and Potential Applications. Compr. Rev. Food Sci. Food Saf. 2011, 10, 221–247. [Google Scholar] [CrossRef]
- Celebioglu, A.; Kayaci-Senirmak, F.; Kusku, S.İ.; Durgun, E.; Uyar, T. Polymer-free nanofibers from vanillin/cyclodextrin inclusion complexes: High thermal stability, enhanced solubility and antioxidant property. Food Funct. 2016. [Google Scholar] [CrossRef] [PubMed]
- Hundre, S.Y.; Karthik, P.; Anandharamakrishnan, C. Effect of whey protein isolate and β-cyclodextrin wall systems on stability of microencapsulated vanillin by spray-freeze drying method. Food Chem. 2015, 174, 16–24. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.Z.; Williams, G.R.; Hou, X.X.; Zhu, L.M. Electrospun curcumin-loaded fibers with potential biomedical applications. Carbohydr. Polym. 2013, 94, 147–153. [Google Scholar] [CrossRef] [PubMed]
- Kharat, M.; Du, Z.; Zhang, G.; McClements, D.J. Physical and chemical stability of curcumin in aqueous solutions and emulsions: Impact of pH, temperature, and molecular environment. J. Agric. Food Chem. 2017. [Google Scholar] [CrossRef] [PubMed]
- Price, L.C.; Buescher, R.W. Decomposition of turmeric curcuminoids as affected by light, solvent and oxygen. J. Food Biochem. 1996, 20, 125–133. [Google Scholar] [CrossRef]
- Mendes, A.C.; Stephansen, K.; Chronakis, I.S. Electrospinning of food proteins and polysaccharides. Food Hydrocoll. 2017, 68, 53–68. [Google Scholar] [CrossRef]
- Yu, D.-G.; Branford-White, C.; Williams, G.R.; Bligh, S.W.A.; White, K.; Zhu, L.-M.; Chatterton, N.P. Self-assembled liposomes from amphiphilic electrospun nanofibers. Soft Matter 2011, 7, 8239. [Google Scholar] [CrossRef]
- Zhang, J.; Cohn, C.; Qiu, W.; Zha, Z.; Dai, Z.; Wu, X. Atomic force microscopy of electrospun organic-inorganic lipid nanofibers. Appl. Phys. Lett. 2011, 99, 103702. [Google Scholar] [CrossRef] [PubMed]
- Mai, T.T.T.; Nguyen, T.T.T.; Le, Q.D.; Nguyen, T.N.; Ba, T.C.; Nguyen, H.B.; Phan, T.B.H.; Tran, D.L.; Nguyen, X.P.; Park, J.S. A novel nanofiber Cur-loaded polylactic acid constructed by electrospinning. Adv. Nat. Sci. Nanosci. Nanotechnol. 2012, 3, 25014. [Google Scholar] [CrossRef]
- Dhurai, B.; Saraswathy, N.; Maheswaran, R.; Sethupathi, P.; Vanitha, P.; Vigneshwaran, S.; Rameshbabu, V. Electrospinning of curcumin loaded chitosan/poly (lactic acid) nanofilm and evaluation of its medicinal characteristics. Front. Mater. Sci. 2013, 7, 350–361. [Google Scholar] [CrossRef]
- Wang, C.; Ma, C.; Wu, Z.; Liang, H.; Yan, P.; Song, J.; Ma, N.; Zhao, Q. Enhanced Bioavailability and Anticancer Effect of Curcumin-Loaded Electrospun Nanofiber: In Vitro and In Vivo Study. Nanoscale Res. Lett. 2015, 10, 439. [Google Scholar] [CrossRef] [PubMed]
- Blanco-Padilla, A.; Lopez-Rubio, A.; Loarca-Pina, G.; Gomez-Mascaraque, L.G.; Mendoza, S. Characterization, release and antioxidant activity of curcumin-loaded amaranth-pullulan electrospun fibers. LWT Food Sci. Technol. 2015, 63, 1137–1144. [Google Scholar] [CrossRef]
- Suwantong, O.; Opanasopit, P.; Ruktanonchai, U.; Supaphol, P. Electrospun cellulose acetate fiber mats containing curcumin and release characteristic of the herbal substance. Polymer 2007, 48, 7546–7557. [Google Scholar] [CrossRef]
- Kayaci, F.; Uyar, T. Encapsulation of vanillin/cyclodextrin inclusion complex in electrospun polyvinyl alcohol (PVA) nanowebs: Prolonged shelf-life and high temperature stability of vanillin. Food Chem. 2012, 133, 641–649. [Google Scholar] [CrossRef]
- Kanawung, K.; Panitchanapan, K.; Puangmalee, S.; Utok, W.; Kreua-ongarjnukool, N.; Rangkupan, R.; Meechaisue, C.; Supaphol, P. Preparation and Characterization of Polycaprolactone/Diclofenac Sodium and Poly(vinyl alcohol)/Tetracycline Hydrochloride Fiber Mats and Their Release of the Model Drugs. Polym. J. 2007, 39, 369–378. [Google Scholar] [CrossRef]
- Lopes De Azambuja, C.R.; Dos Santos, L.G.; Rodrigues, M.R.; Rodrigues, R.F.M.; Da Silveira, E.F.; Azambuja, J.H.; Flores, A.F.C.; Horn, A.P.; Dora, C.L.; Muccillo-Baisch, A.L.; et al. Physico-chemical characterization of asolectin-genistein liposomal system: An approach to analyze its in vitro antioxidant potential and effect in glioma cells viability. Chem. Phys. Lipids 2015, 193, 24–35. [Google Scholar] [CrossRef] [PubMed]
- Peng, H.; Xiong, H.; Li, J.; Xie, M.; Liu, Y.; Bai, C.; Chen, L. Vanillin cross-linked chitosan microspheres for controlled release of resveratrol. Food Chem. 2010, 121, 23–28. [Google Scholar] [CrossRef]
- Rezaei, A.; Tavanai, H.; Nasirpour, A. Fabrication of electrospun almond gum/PVA nanofibers as a thermostable delivery system for vanillin. Int. J. Biol. Macromol. 2016, 91, 536–543. [Google Scholar] [CrossRef] [PubMed]
- Rezaei, A.; Nasirpour, A.; Tavanai, H.; Fathi, M. A study on the release kinetics and mechanisms of vanillin incorporated in almond gum/polyvinyl alcohol composite nanofibers in different aqueous food simulants and simulated saliva. Flavour Fragr. J. 2016, 31, 442–447. [Google Scholar] [CrossRef]
- Noshad, M.; Mohebbi, M.; Koocheki, A.; Shahidi, F. Microencapsulation of vanillin by spray drying using soy protein isolate-maltodextrin as wall material. Flavour Fragr. J. 2015, 30, 387–391. [Google Scholar] [CrossRef]
- Chen, Y.; Wu, Q.; Zhang, Z.; Yuan, L.; Liu, X.; Zhou, L. Preparation of curcumin-loaded liposomes and evaluation of their skin permeation and pharmacodynamics. Molecules 2012, 17, 5972–5987. [Google Scholar] [CrossRef] [PubMed]
- Belyagoubi-Benhammou, N.; Belyagoubi, L.; El Zerey-Belaskri, A.; Atik-Bekkara, F. In vitro antioxidant properties of flavonoid fractions from Pistacia atlantica Desf. subsp. atlantica fruit using five techniques. J. Mater. Environ. Sci. 2015, 6, 1118–1125. [Google Scholar]
- Ali, S.S.; Kasoju, N.; Luthra, A.; Singh, A.; Sharanabasava, H.; Sahu, A.; Bora, U. Indian medicinal herbs as sources of antioxidants. Food Res. Int. 2008, 41, 1–15. [Google Scholar] [CrossRef]
- Prieto, P.; Pineda, M.; Aguilar, M. Spectrophotometric quantitation of antioxidant capacity through the formation of a phosphomolybdenum complex: Specific application to the determination of vitamin E. Anal. Biochem. 1999, 269, 337–341. [Google Scholar] [CrossRef] [PubMed]
- Tusevski, O.; Kostovska, A.; Iloska, A.; Trajkovska, L.; Simic, S.G. Phenolic production and antioxidant properties of some Macedonian medicinal plants. Cent. Eur. J. Biol. 2014, 9, 888–900. [Google Scholar] [CrossRef]
- Chevolleau, S.; Mallet, J.F.; Ucciani, E.; Gamisans, J.; Gruber, M. Antioxidant activity in leaves of some mediterranean plants. J. Am. Oil Chem. Soc. 1992, 69, 1269–1271. [Google Scholar] [CrossRef]
- Jayaprakasha, G.K.; Jaganmohan Rao, L.; Sakariah, K.K. Antioxidant activities of curcumin, demethoxycurcumin and bisdemethoxycurcumin. Food Chem. 2006, 98, 720–724. [Google Scholar] [CrossRef]
- Saito, H.; Ishihara, K. Antioxidant activity and active sites of phospholipids as antioxidants. J. Am. Oil Chem. Soc. 1997, 74, 1531–1536. [Google Scholar] [CrossRef]
- Bandarra, N.M.; Campos, R.M.; Batista, I.; Nunes, M.L.; Empis, J.M. Antioxidant synergy of alpha-tocopherol and phospholipids. J. Am. Oil Chem. Soc. 1999, 76, 905–913. [Google Scholar] [CrossRef]
- Safafar, H.; van Wagenen, J.; Møller, P.; Jacobsen, C. Carotenoids, phenolic compounds and tocopherols contribute to the antioxidative properties of some microalgae species grown on industrial wastewater. Mar. Drugs 2015, 13, 7339–7356. [Google Scholar] [CrossRef] [PubMed]
- Tavassoli-Kafrani, E.; Goli, S.A.H.; Fathi, M. Fabrication and characterization of electrospun gelatin nanofibers crosslinked with oxidized phenolic compounds. Int. J. Biol. Macromol. 2017, 103, 1062–1068. [Google Scholar] [CrossRef] [PubMed]
- Yakub, G.; Toncheva, A.; Manolova, N.; Rashkov, I.; Kussovski, V.; Danchev, D. Curcumin-loaded poly(l-lactide-co-d,l-lactide) electrospun fibers: Preparation and antioxidant, anticoagulant, and antibacterial properties. J. Bioact. Compat. Polym. 2014, 29, 607–627. [Google Scholar] [CrossRef]
- Neo, Y.P.; Ray, S.; Jin, J.; Gizdavic-Nikolaidis, M.; Nieuwoudt, M.K.; Liu, D.; Quek, S.Y. Encapsulation of food grade antioxidant in natural biopolymer by electrospinning technique: A physicochemical study based on zein-gallic acid system. Food Chem. 2013, 136, 1013–1021. [Google Scholar] [CrossRef] [PubMed]
- Aytac, Z.; Kusku, S.I.; Durgun, E.; Uyar, T. Encapsulation of gallic acid/cyclodextrin inclusion complex in electrospun polylactic acid nanofibers: Release behavior and antioxidant activity of gallic acid. Mater. Sci. Eng. C 2016, 63, 231–239. [Google Scholar] [CrossRef] [PubMed]
- Sampath, M.; Lakra, R.; Korrapati, P.; Sengottuvelan, B. Curcumin loaded poly (lactic-co-glycolic) acid nanofiber for the treatment of carcinoma. Colloids Surf. B Biointerfaces 2014, 117, 128–134. [Google Scholar] [CrossRef] [PubMed]
- Korsmeyer, R.W.; Gurny, R.; Doelker, E.; Buri, P.; Peppas, N.A. Mechanisms of solute release from porous hydrophilic polymers. Int. J. Pharm. 1983, 15, 25–35. [Google Scholar] [CrossRef]
- Wongsasulak, S.; Pathumban, S.; Yoovidhya, T. Effect of entrapped α-tocopherol on mucoadhesivity and evaluation of the release, degradation, and swelling characteristics of zein-chitosan composite electrospun fibers. J. Food Eng. 2014, 120, 110–117. [Google Scholar] [CrossRef]
- Hunley, M.T.; McKee, M.G.; Long, T.E. Submicron functional fibrous scaffolds based on electrospun phospholipids. J. Mater. Chem. 2007, 17, 605. [Google Scholar] [CrossRef]
Sample Availability: Not available. |



| Peak Number | Group Frequency (cm−1) | Assignment | ||||||
|---|---|---|---|---|---|---|---|---|
| Vanillin Powder | Curcumin Powder | Phos Powder | Phos (Limonene) Fiber | Phos (Isooctane) Fiber | Phos/Vanillin Fiber | Phos/CurcuminFiber | ||
| 1 | - | 3085–3552 | 3001 | 3000 | 3000 | 3000 | 3000 | phenolic O-H stretching |
| 2 | - | - | 2920 2850 | 2922 2852 | 2930 2859 | 2908 2847 | 2923 2853 | C-H stretching of CH2 groups |
| 3 | 1660 | - | 1730 | 1735 | 1746 | 1725 | 1740 | C=O stretching of carbonyl groups |
| 4 | 1590 1510 | 1601 | - | - | - | - | - | stretching vibrations of the benzene ring |
| 5 | 1273 | 1240 | 1230 | 1243 | 1205 | 1229 | PO2− groups; aromatic C-O stretching | |
| 6 | 1150 | 1152 | - | - | - | - | - | C-O-C stretching |
| 7 | 731 | - | - | - | - | - | - | stretching vibrations of the benzene ring |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shekarforoush, E.; Mendes, A.C.; Baj, V.; Beeren, S.R.; Chronakis, I.S. Electrospun Phospholipid Fibers as Micro-Encapsulation and Antioxidant Matrices. Molecules 2017, 22, 1708. https://doi.org/10.3390/molecules22101708
Shekarforoush E, Mendes AC, Baj V, Beeren SR, Chronakis IS. Electrospun Phospholipid Fibers as Micro-Encapsulation and Antioxidant Matrices. Molecules. 2017; 22(10):1708. https://doi.org/10.3390/molecules22101708
Chicago/Turabian StyleShekarforoush, Elhamalsadat, Ana C. Mendes, Vanessa Baj, Sophie R. Beeren, and Ioannis S. Chronakis. 2017. "Electrospun Phospholipid Fibers as Micro-Encapsulation and Antioxidant Matrices" Molecules 22, no. 10: 1708. https://doi.org/10.3390/molecules22101708
APA StyleShekarforoush, E., Mendes, A. C., Baj, V., Beeren, S. R., & Chronakis, I. S. (2017). Electrospun Phospholipid Fibers as Micro-Encapsulation and Antioxidant Matrices. Molecules, 22(10), 1708. https://doi.org/10.3390/molecules22101708






