Time-Dependent Antimicrobial Activity of Filtering Nonwovens with Gemini Surfactant-Based Biocides
Abstract
:1. Introduction
2. Results and Discussion
2.1. Evaluation of the Morphology of the Nonwovens
2.2. Survival of Microorganisms on Filter Materials
3. Materials and Methods
3.1. Nonwovens with Time-Dependent Biocidal Structures
3.2. Manufacturing of Melt-Blown Nonwovens
3.3. Simulation of Use in Organic Dust Pollution Conditions
3.4. Morphology of Filtering Nonwovens
3.5. Microorganisms Tested
3.6. Survival of Microorganisms on Filtering Nonwovens
3.7. Mathematical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Pasanen, A.L.; Keinanen, J.; Kalliokoski, P.; Martikainen, P.I.; Ruuskanen, J. Microbial growth on respirator filters from improper storage. Scand. J. Work Environ. Health 1993, 19, 421–425. [Google Scholar] [CrossRef] [PubMed]
- Brosseau, L.M.; McCullough, N.V.; Vesley, D. Bacterial survival on respirator filters and surgical masks. Appl. BioSaf. 1997, 2, 32–43. [Google Scholar] [CrossRef]
- Reponen, T.A.; Wang, Z.; Willeke, K.; Grinshpun, S.A. Survival of mycobacteria on N95 personal respirators. Infect. Control Hosp. Epidemiol. 1999, 20, 237–241. [Google Scholar] [CrossRef] [PubMed]
- Forthomme, A.; Joubert, A.; Andrès, Y.; Simon, X.; Duquenne, P.; Bemer, D.; LeCoq, L. Microbial aerosol filtration: Growth and release of a bacteria-fungi consortium collected by fibrous filters in different operating conditions. J. Aerosol. Sci. 2014, 72, 32–46. [Google Scholar] [CrossRef]
- Szulc, J.; Otlewska, A.; Okrasa, M.; Majchrzycka, K.; Sulyok, M.; Gutarowska, B. Microbiological contamination at workplaces in a combined heat and power (CHP) station processing plant biomass. Int. J. Environ. Res. Public Health 2017, 14, 99. [Google Scholar] [CrossRef] [PubMed]
- Siedenbiedel, F.; Tiller, J.C. Antimicrobial polymers in solution and on surfaces: Overview and functional principles. Polymers 2012, 4, 46–71. [Google Scholar] [CrossRef]
- Sureshkumar, M.; Siswanto, D.Y.; Chen, Y.C.; Lee, C.K.; Wang, M.J. Antibacterial and biocompatible surfaces based on dopamine autooxidized silver nanoparticles. J. Polym. Sci. Part B Polym. Phys. 2013, 51, 303–310. [Google Scholar] [CrossRef]
- Dastjerdi, R.; Montazer, M.; Shahsavan, S. A novel technique for producing durable multifunctional textiles using nanocomposite coating. Colloids Surf. B Biointerfaces 2010, 81, 32–41. [Google Scholar] [CrossRef] [PubMed]
- Dastjerdi, R.; Montazer, M. A review on the application of inorganic nano-structured materials in the modification of textiles: Focus on anti-microbial properties. Colloids Surf. B Biointerfaces 2010, 79, 5–18. [Google Scholar] [CrossRef] [PubMed]
- Lin, S.; Wang, Z.; Qi, J.; Wu, J.; Tian, T.; Hou, L.; Hao, L.; Yang, J. One-pot fabrication and antimicrobial properties of novel PET nonwoven fabrics. Biomed. Mater. 2011, 6, 45009. [Google Scholar] [CrossRef] [PubMed]
- Mahltig, B.; Fiedler, D.; Fischer, A.; Simon, P. Antimicrobial coatings on textiles-modification of sol-gel layers with organic and inorganic biocides. J. Sol-Gel Sci. Technol. 2010, 55, 269–277. [Google Scholar] [CrossRef]
- Wafa, D.M.; Breidt, F.; Gawish, S.M.; Matthews, S.R.; Donohue, K.V.; Roe, R.M.; Bourham, M.A. Atmospheric plasma-aided biocidal finishes for nonwoven polypropylene fabrics. II. Functionality of synthesized fabrics. Polym. Polym. Compos. 2013, 21, 449–456. [Google Scholar] [CrossRef]
- Nithya, E.; Radhai, R.; Rajendran, R.; Jayakumar, S.; Vaideki, K. Enhancement of the antimicrobial property of cotton fabric using plasma and enzyme pre-treatments. Carbohydr. Polym. 2012, 88, 986–991. [Google Scholar] [CrossRef]
- Yang, M.R.; Chen, K.S.; Tsai, J.C.; Tseng, C.C.; Lin, S.F. The antibacterial activities of hydrophilic-modified nonwoven PET. Mater. Sci. Eng. C 2002, 20, 167–173. [Google Scholar] [CrossRef]
- Majchrzycka, K.; Gutarowska, B.; Brochocka, A.; Brycki, B. New filtering antimicrobial nonwovens with various carriers for biocides as respiratory protective materials against bioaerosol. Int. J. Occup. Saf. Ergon. 2012, 18, 375–385. [Google Scholar] [CrossRef] [PubMed]
- Miaśkiewicz-Peska, E.; Łebkowska, M. Effect of antimicrobial air filter treatment on bacterial survival. Fibres Text. East. Eur. 2011, 19, 73–77. [Google Scholar]
- Gutarowska, B.; Skóra, J.; Nowak, E.; Łysiak, I.; Wdówka, M. Antimicrobial activity and filtration effectiveness of nonwovens with sanitized for respiratory protective equipment. Fibres Text. East. Eur. 2014, 22, 120–125. [Google Scholar]
- Gutarowska, B.; Michalski, A. Antimicrobial activity of filtrating meltblown nonwoven with the addition of silver ions. Fibres Text. East. Eur. 2009, 17, 23–28. [Google Scholar]
- Brochocka, A.; Majchrzycka, K. Technology for the production of bioactive melt-blown filtration materials applied to respiratory protective devices. Fibres Text. East. Eur. 2009, 17, 92–98. [Google Scholar]
- Woo, M.H.; Grippin, A.; Wu, C.Y.; Baney, R.H. Use of dialdehyde starch treated filters for protection against airborne viruses. J. Aerosol Sci. 2012, 46, 77–82. [Google Scholar] [CrossRef]
- Tiliket, G.; LeSage, D.; Moules, V.; Rosa-Calatrava, M.; Lina, B.; Valleton, J.M.; Nguyen, Q.T.; Lebrun, L. A new material for airborne virus filtration. Chem. Eng. J. 2011, 173, 341–351. [Google Scholar] [CrossRef]
- Gliścińska, E.; Gutarowska, B.; Brycki, B.; Krucińska, I. Electrospun polyacrylonitrile nanofibers modified by quaternary ammonium salts. J. Appl. Polym. Sci. 2013, 128, 767–775. [Google Scholar] [CrossRef]
- Heimbuch, B.K.; Wander, J.D. Bioaerosol Challenges to Antimicrobial Surface Treatments: Enhanced Efficacy Against MS2 coli Phage of Air Filter Media Coated with Polystyrene-4-methyltrimethylammonium Triiodide; Defense Technical Information Center: Fort Belvoir, VA, USA, 2006.
- Majchrzycka, K.; Okrasa, M.; Brochocka, A.; Urbaniak-Domagała, W. Influence of low-temperature plasma treatment on the liquid filtration efficiency of melt-blown PP nonwovens in the conditions of simulated use of respiratory protective equipment. Chem. Process Eng. 2017, 38, 195–207. [Google Scholar] [CrossRef]
- Brycki, B.; Kowalczyk, I.; Kozirog, A. Synthesis, molecular structure, spectral properties and antifungal activity of polymethylene-α,ω-bis(N,N-dimethyl-N-dodecyloammonium bromides). Molecules 2011, 16, 319–335. [Google Scholar] [CrossRef] [PubMed]
- Zana, R.; Xia, J. Gemini Surfactants: Synthesis, Interfacial and Solution-Phase Behavior, and Applications; Marcel Dekker: New York, NY, USA, 2004; p. 331. ISBN 978-0-8247-4705-3. [Google Scholar]
- Koziróg, A.; Brycki, B. Monomeric and gemini surfactants as antimicrobial agents—influence on environmental and reference strains. Acta Biochim. Pol. 2015, 62, 879–883. [Google Scholar] [CrossRef] [PubMed]
- Brycki, B.; Drgas, M.; Bielawska, M.; Zdziennicka, A.; Jańczuk, B. Synthesis, spectroscopic studies, aggregation and surface behavior of hexamethylene-1,6-bis(N,N-dimethyl-N-dodecylammonium bromide). J. Mol. Liq. 2016, 221, 1086–1096. [Google Scholar] [CrossRef]
- Brycki, B.; Gutarowska, B.; Majchrzycka, K.; Brochocka, A.; Orlikowski, W.; Krucińska, I.; Gliścińska, E.; Krzyzanowski, J.; Łysiak, I. Środek Biobójczy do Wytwarzania Włóknin Filtracyjnych oraz Sposób Otrzymywania Środka Biobójczego do Wytwarzania Włóknin Filtracyjnych. PL 211878 B1, 31 July 2012. [Google Scholar]
- Majchrzycka, K.; Brochocka, A.; Brycki, B. Biocidal agent for modification of poly(lactic acid) high-efficiency filtering nonwovens. Fibres Text. East. Eur. 2015, 23, 88–95. [Google Scholar]
- Majchrzycka, K. Evaluation of a new bioactive nonwoven fabric for respiratory protection. Fibres Text. East. Eur. 2014, 22, 81–88. [Google Scholar]
- Majchrzycka, K.; Okrasa, M.; Szulc, J.; Gutarowska, B. The impact of dust in filter materials of respiratory protective devices on the microorganisms viability. Int. J. Ind. Ergon. 2017, 58, 109–116. [Google Scholar] [CrossRef]
- Maus, R.; Goppelsröder, A.; Umhauer, H. Viability of bacteria in unused air filter media. Atmos. Environ. 1997, 31, 2305–2310. [Google Scholar] [CrossRef]
- Majchrzycka, K.; Okrasa, M.; Skóra, J.; Gutarowska, B. Evaluation of the survivability of microorganisms deposited on filtering respiratory protective devices under varying conditions of humidity. Int. J. Environ. Res. Public Health 2016, 13, 98. [Google Scholar] [CrossRef] [PubMed]
- Beumer, R.; Bloomfield, S.F.; Exner, M.; Fara, G.M.; Nath, K.J.; Scott, E. Biocide Usage and Antimicrobial Resistance in Home Settings: An Update; International Scientific Forum on Home Hygiene: Montacute, UK, 2003. [Google Scholar]
- Majchrzycka, K.; Brycki, B.; Okrasa, M. Zestaw Struktur Porowatych o Działaniu Biobójczym do Modyfikacji Włóknin Filtracyjnych Długotrwałego Użycia. P-416228, 22 February 2016. [Google Scholar]
- Majchrzycka, K.; Okrasa, M.; Brycki, B.; Szulc, J.; Gutarowska, B. Efficiency study of bioactive porous structures with time-dependent activity in filtering melt-blown nonwovens. Przem. Chem. 2017, 96, 534–538. [Google Scholar] [CrossRef]
- British Standards Institution. BS EN 13274-7:2008: Respiratory Protective Devices. Methods of Test. Determination of Particle Filter Penetration; British Standards Institution: Englewood, CO, USA, 2008. [Google Scholar]
- British Standards Institution. BS EN 13274-3:2001: Respiratory Protective Devices. Methods of Test. Determination of Breathing Resistance; British Standards Institution: Englewood, CO, USA, 2001. [Google Scholar]
- Dutkiewicz, J.; Śpiewak, R.; Jabłoński, L.; Szymańska, J. Biological Occupational Risk Factors. Classification, Exposed Occupational Groups, Measurement, Prevention; Ad Punctum: Lublin, Poland, 2007. [Google Scholar]
- American Association of Textile Chemists and Colorists (AATCC). AATCC Test Method 100-2004: Antibacterial Finishes on Textile Materials: Assessment of AATCC Technical Manual/2010; AATCC: Research Triangle Park, NC, USA, 2004. [Google Scholar]
Sample Availability: Samples of the SPBS and bioactive nonwoven with SPBS could be available from the authors. |


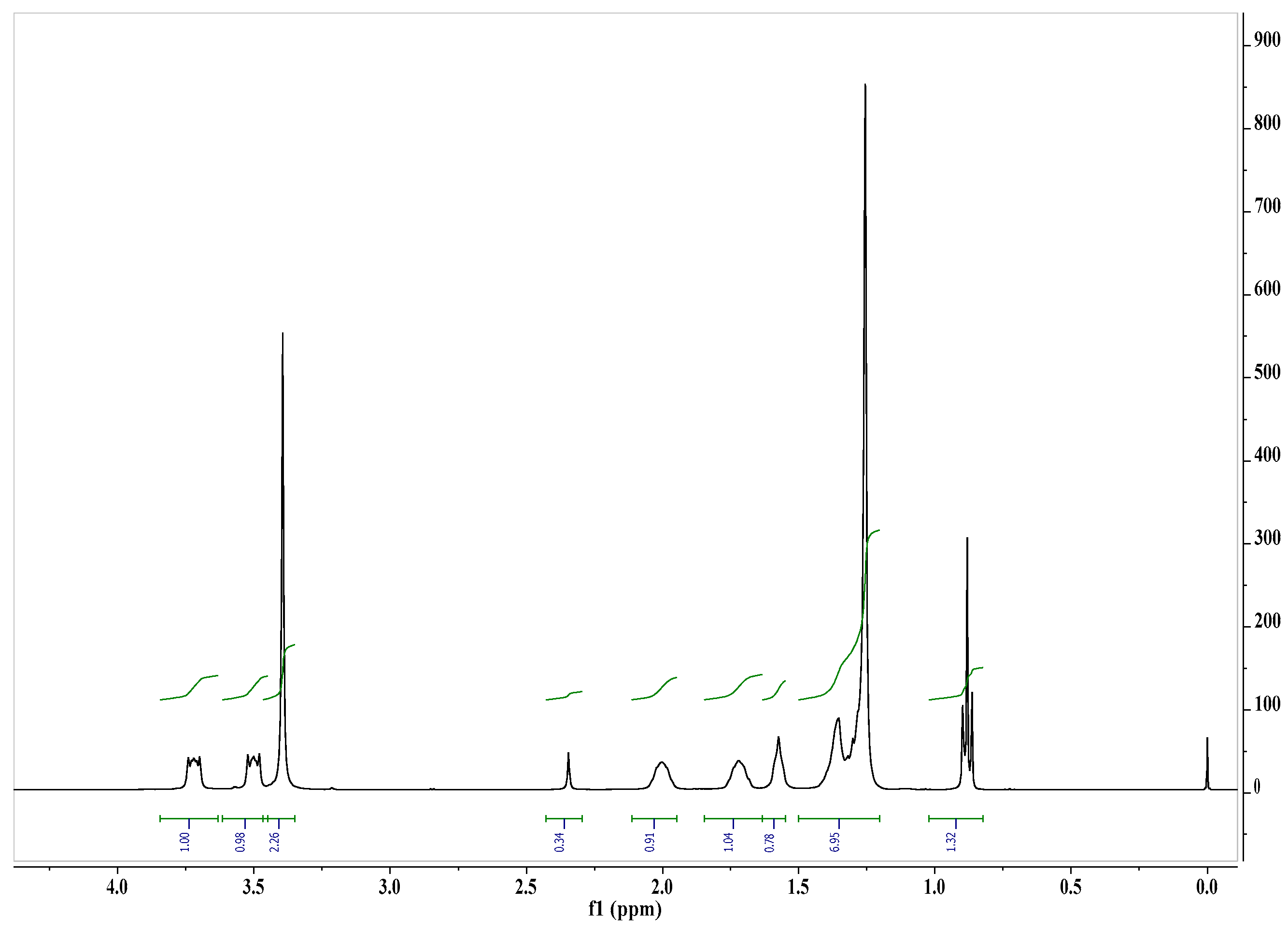
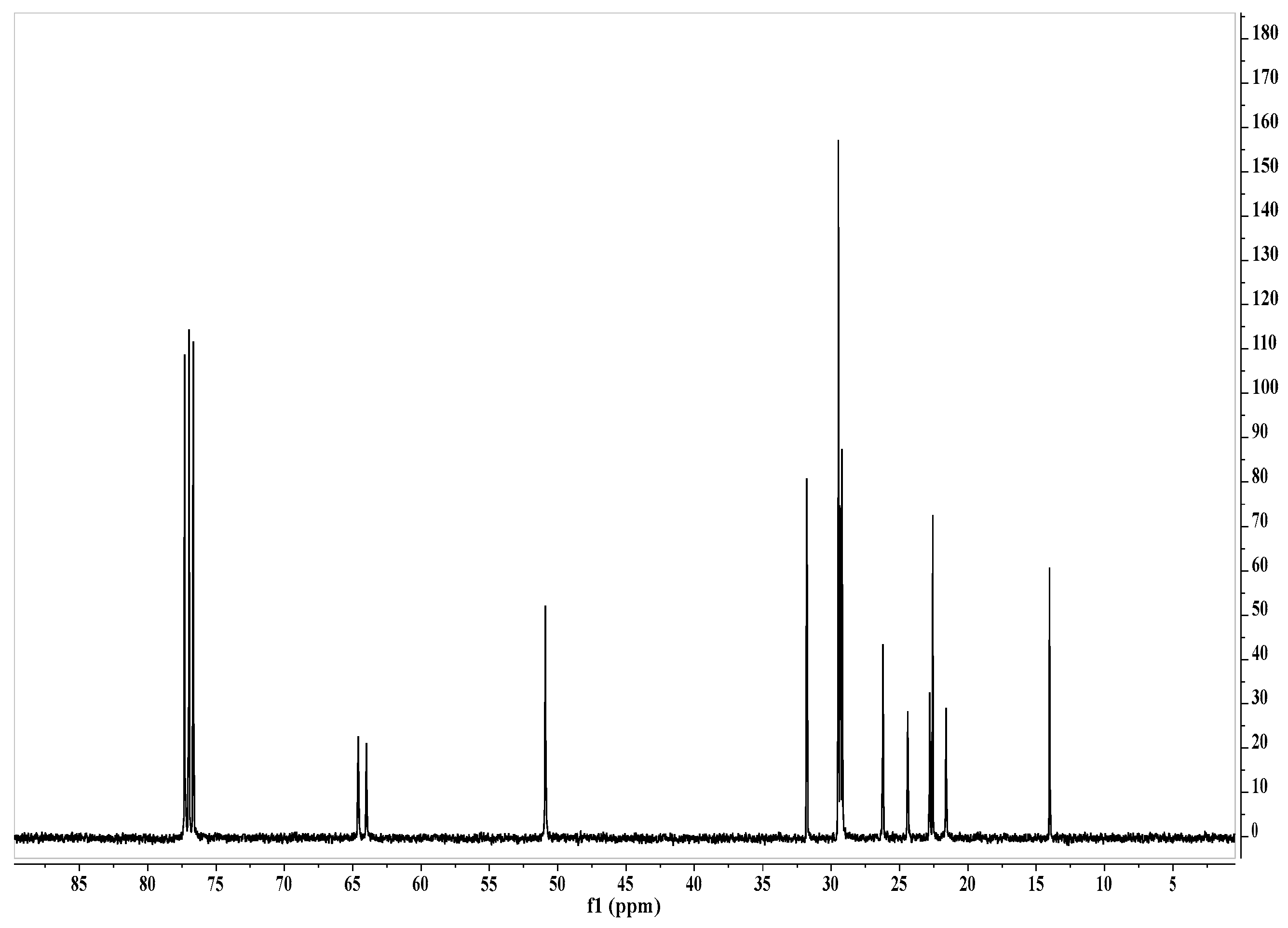
| Microorganisms | Reduction of Microorganisms Number, R (%) | Survival Index, N (%) | ||||
|---|---|---|---|---|---|---|
| 8 h | 24 h | 32 h | 8 h | 24 h | 32 h | |
| Escherichia coli | 12.23 | 77.15 | 97.46 | 94.96 | 1.06 | 0.12 |
| Pseudomonas fluorescens | 3.80 | 87.50 | 87.85 | 45.90 | 7.82 | 1.24 |
| Aspergillus niger | - 1 | 80.11 | 94.53 | 22.07 | 1.35 | 3.78 |
| Penicillium chrysogenum | - 1 | 80.09 | 46.78 | 14.43 | 9.93 | 6.85 |
| No. | Type of Biocidal Structures | Concentration of GS-12-6-12, % | Concentration of 1,2-Propanediol, % |
|---|---|---|---|
| 1 | HA-2L G5 | 2 | 5 |
| 2 | HA-5L G4 | 5 | 4 |
| 3 | HA-5L G3 | 5 | 3 |
| 4 | HA-2L G1 | 2 | 1 |
| 5 | HA-10L | 10 | 0 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Majchrzycka, K.; Okrasa, M.; Szulc, J.; Brycki, B.; Gutarowska, B. Time-Dependent Antimicrobial Activity of Filtering Nonwovens with Gemini Surfactant-Based Biocides. Molecules 2017, 22, 1620. https://doi.org/10.3390/molecules22101620
Majchrzycka K, Okrasa M, Szulc J, Brycki B, Gutarowska B. Time-Dependent Antimicrobial Activity of Filtering Nonwovens with Gemini Surfactant-Based Biocides. Molecules. 2017; 22(10):1620. https://doi.org/10.3390/molecules22101620
Chicago/Turabian StyleMajchrzycka, Katarzyna, Małgorzata Okrasa, Justyna Szulc, Bogumił Brycki, and Beata Gutarowska. 2017. "Time-Dependent Antimicrobial Activity of Filtering Nonwovens with Gemini Surfactant-Based Biocides" Molecules 22, no. 10: 1620. https://doi.org/10.3390/molecules22101620
APA StyleMajchrzycka, K., Okrasa, M., Szulc, J., Brycki, B., & Gutarowska, B. (2017). Time-Dependent Antimicrobial Activity of Filtering Nonwovens with Gemini Surfactant-Based Biocides. Molecules, 22(10), 1620. https://doi.org/10.3390/molecules22101620








