Function Oriented Molecular Design: Dendrimers as Novel Antimicrobials
Abstract
:1. Introduction
2. Structure of Dendrimers and Methods for Dendrimers Preparation
2.1. Synthesis of Dendrimers
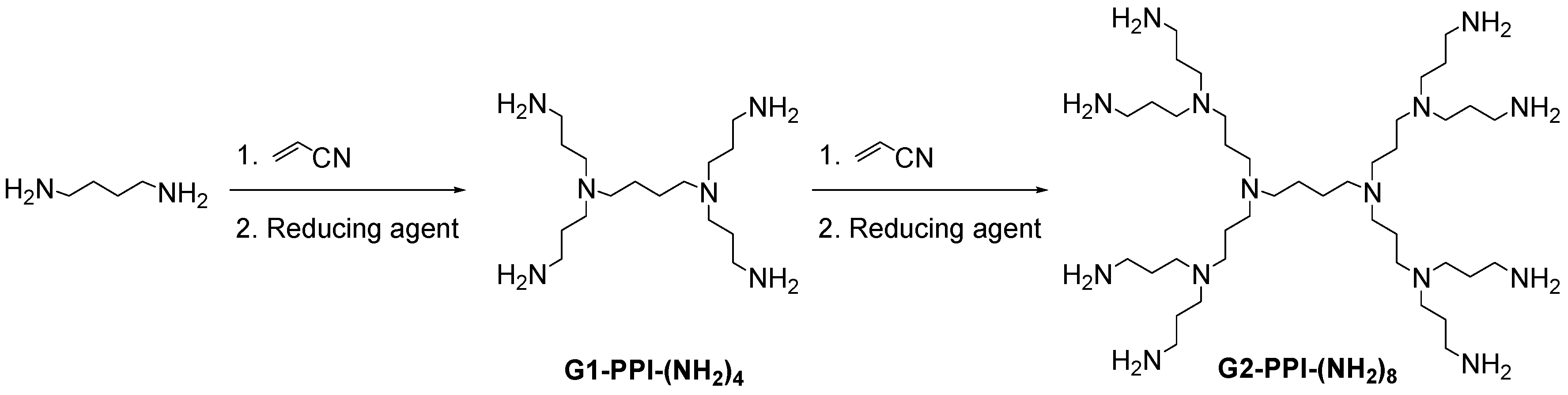
2.2. Poly(propylene imine) Dendrimers (PPI)
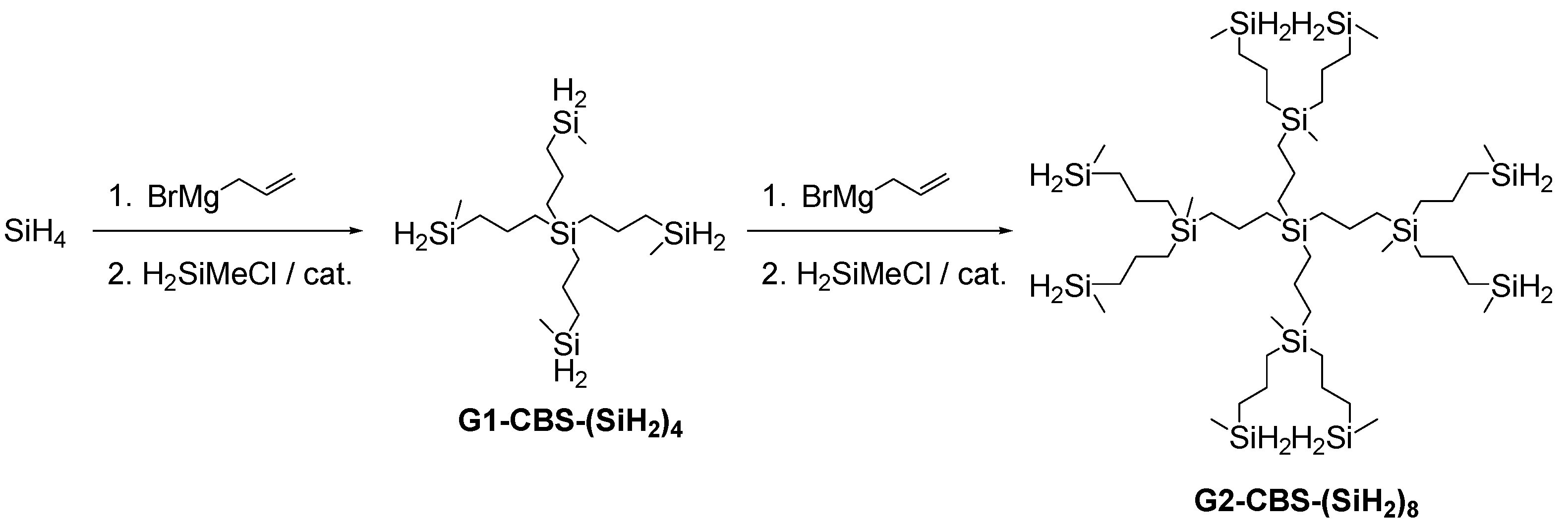
2.3. Carbosilane Dendrimers (CBS)
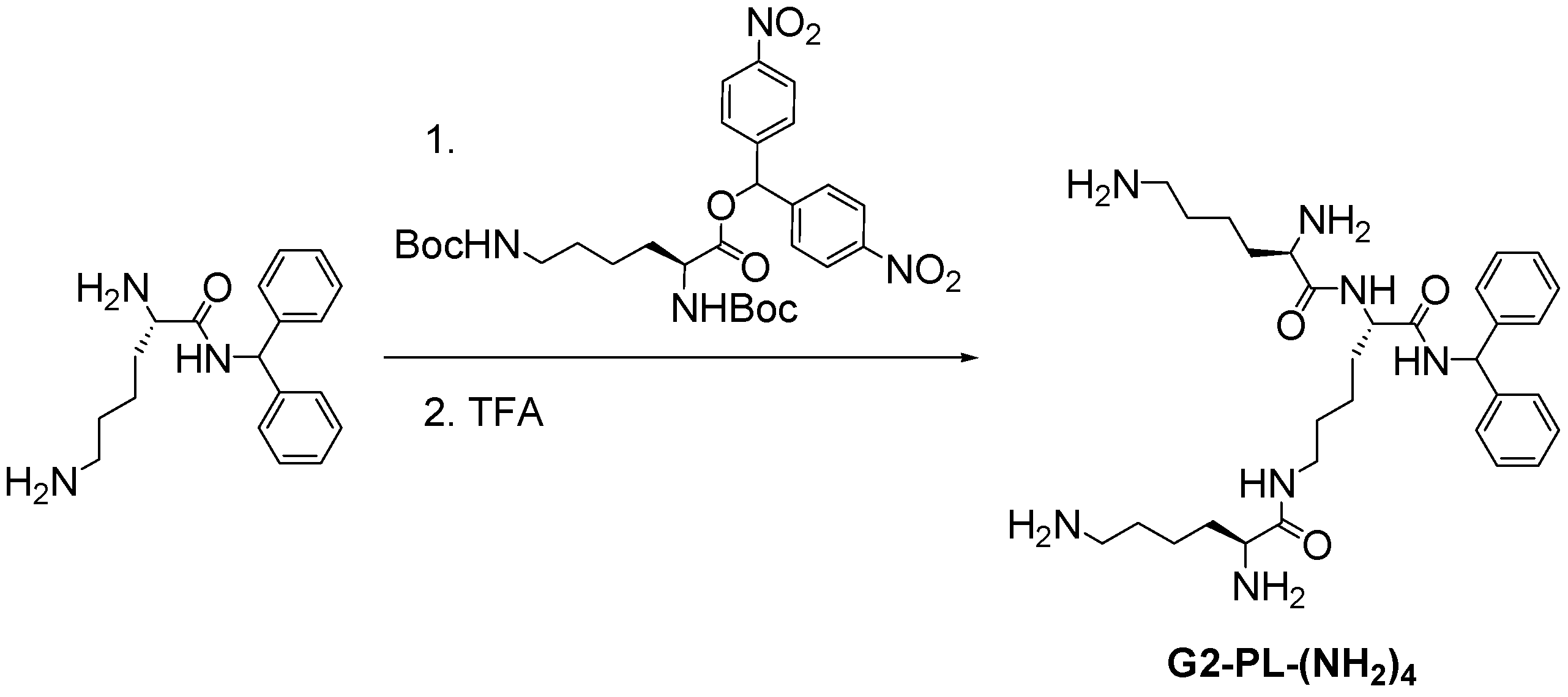
2.4. Peptide Dendrimers
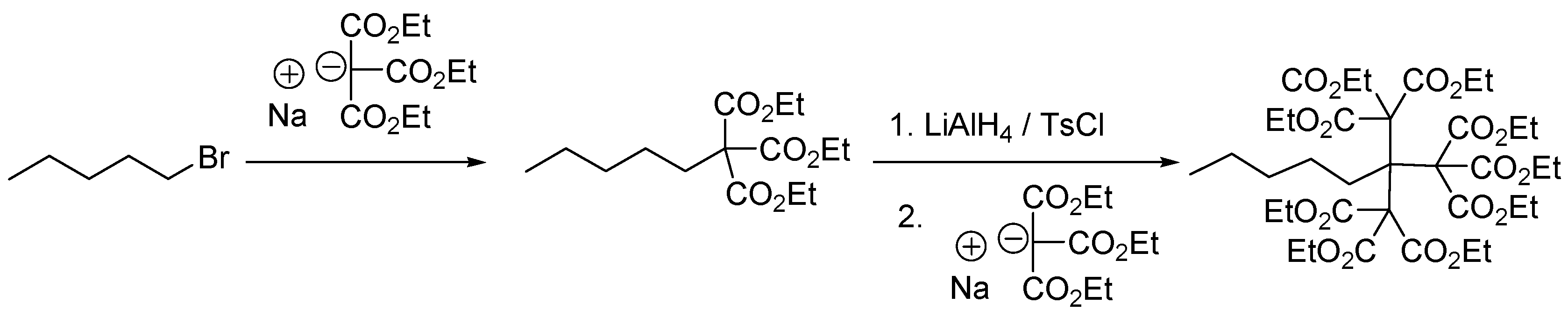
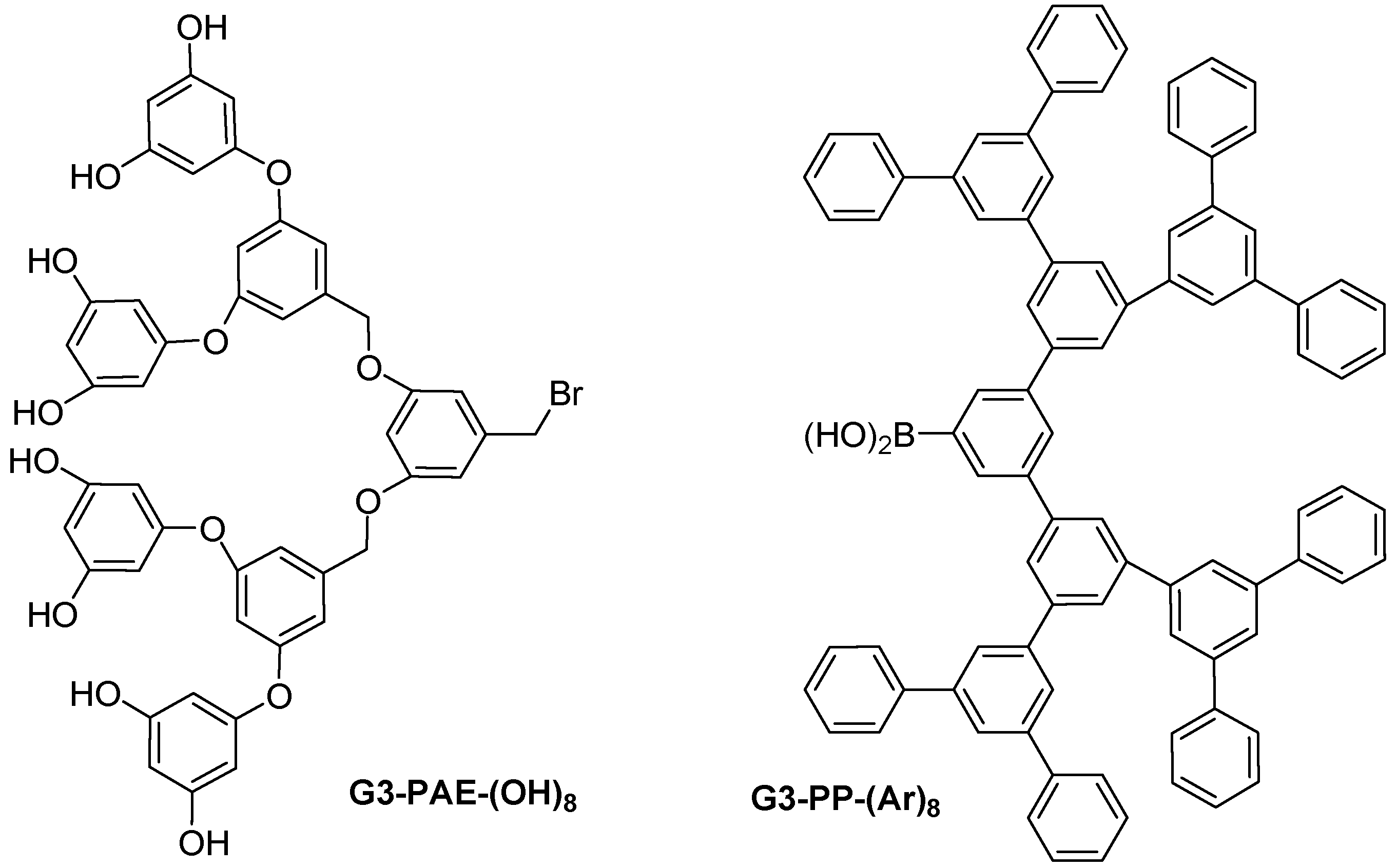
2.5. Newkome-Type Dendrimers or Arborols
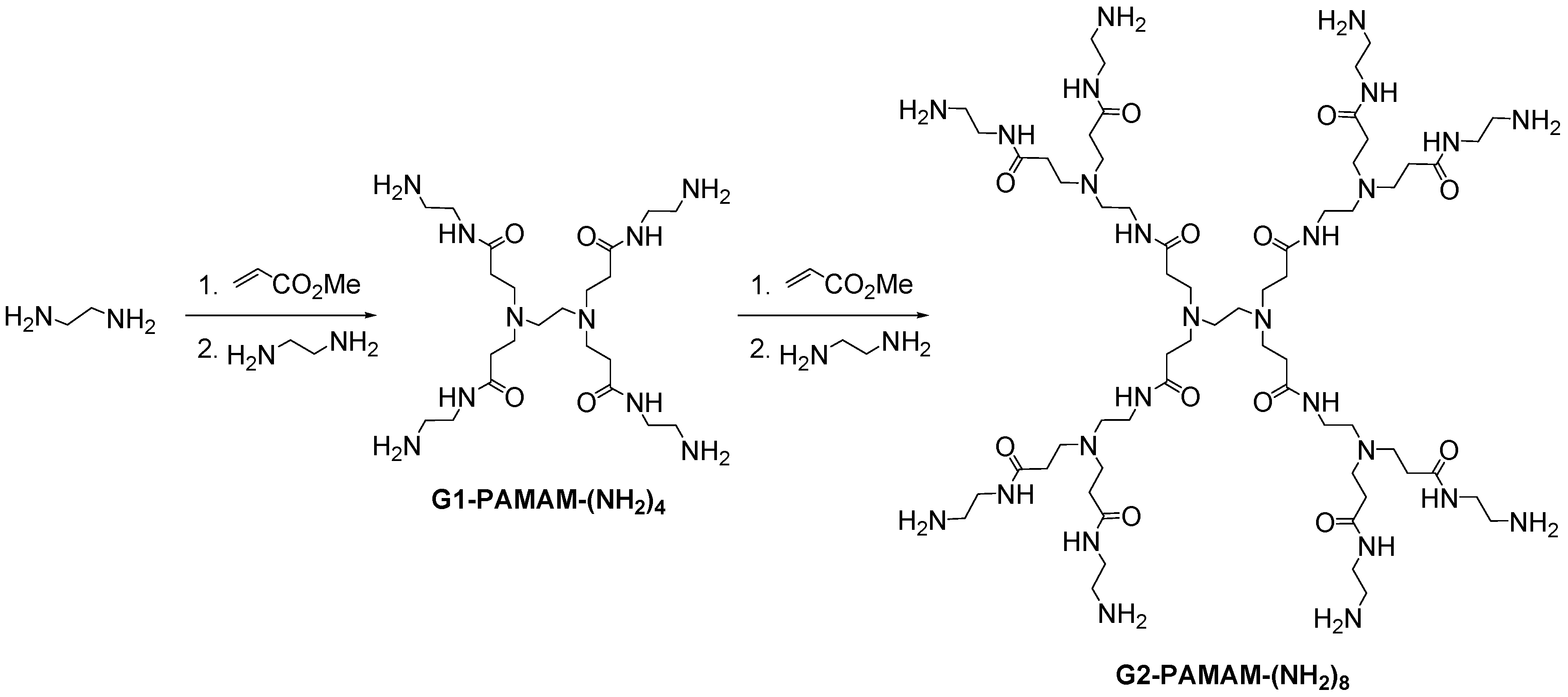
2.6. Polyamido(amine) (PAMAM) Dendrimers
2.7. Phosphorus Dendrimers
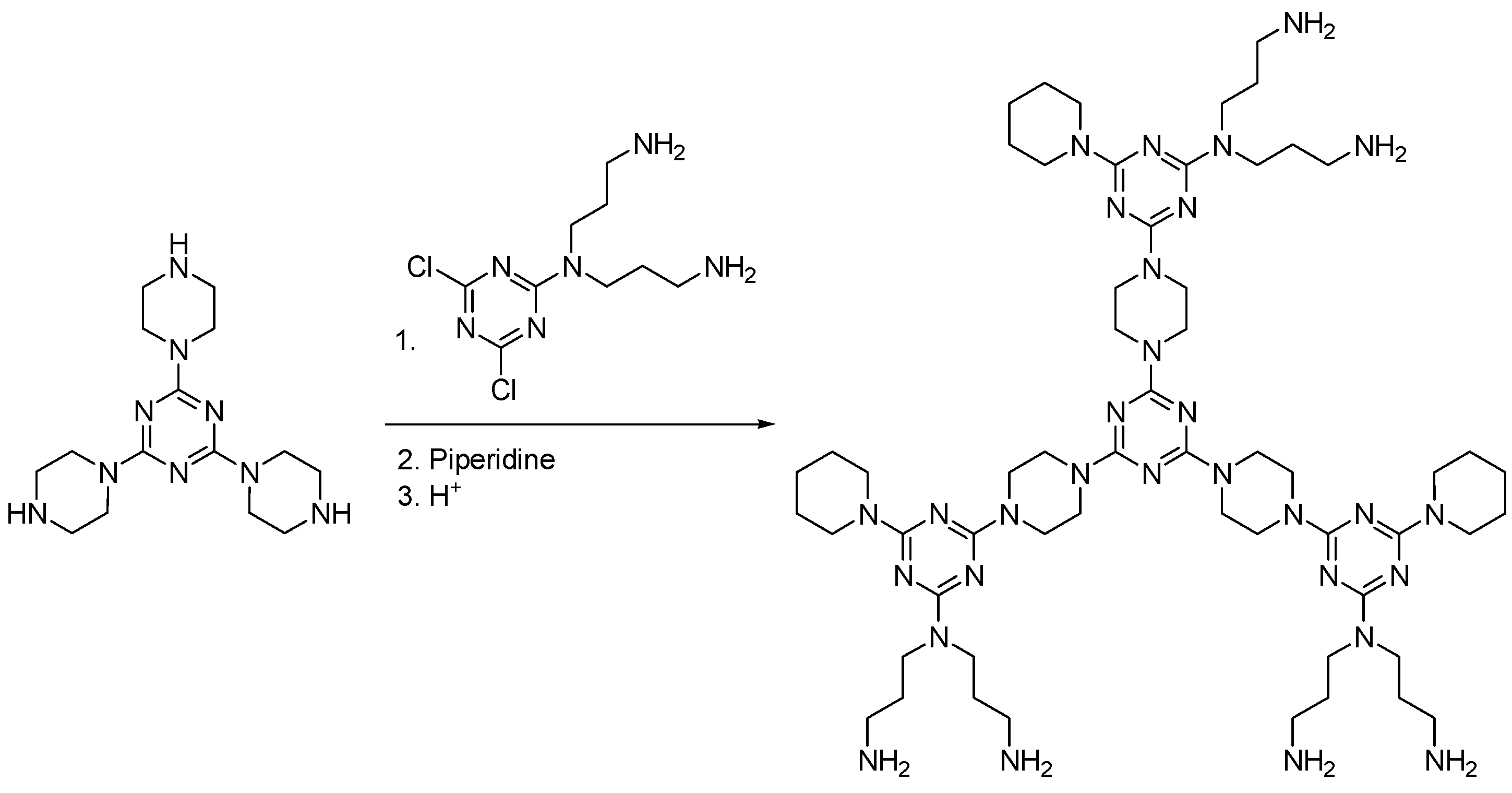
2.8. Other Types of Dendrimers
3. Characterization of Dendrimers
3.1. Spectroscopic and Spectrometric Methods
3.1.1. Mass Spectrometry (MS)
3.1.2. Nuclear Magnetic Resonance (NMR) Spectroscopy
3.1.3. Electron Paramagnetic Resonance (EPR)
3.1.4. Fluorescence Spectroscopy
3.1.5. Infrared and Raman Spectroscopy
3.1.6. UV-Vis Spectroscopy
3.2. Chromatographic Techniques
3.2.1. Gel Permeation Chromatography (GPC)
3.2.2. High-Performance Liquid Chromatography (HPLC)
3.3. Scattering Techniques
3.3.1. Small Angle Neutron Scattering (SANS)
3.3.2. Quasi-Elastic Neutron Scattering (QENS)
3.3.3. Small-Angle X-ray Scattering (SAXS)
3.3.4. Dynamic Light Scattering (DLS)
3.4. Microscopy
3.4.1. Atomic Force Microscopy (AFM)
3.4.2. Scanning Tunnelling Microscopy (STM)
3.5. Electrophoretic Techniques
3.6. Other Types of Techniques
3.6.1. X-ray Diffraction
3.6.2. Acid-Base Titration
4. Applications of Dendrimers
4.1. Drug Delivery and Cell Transfection
4.2. Antimicrobial Activity
4.2.1. Antibacterial Activity
4.2.2. Antiviral Activity
4.2.3. Antiparasitic Activity
5. Conclusions
Acknowledgments
Conflicts of Interest
References
- McCarthy, T.D.; Karellas, P.; Henderson, S.A.; Giannis, M.; O’Keefe, D.F.; Heery, G.; Paull, J.R.; Matthews, B.R.; Holan, G. Dendrimers as drugs: Discovery and preclinical and clinical development of dendrimer-based microbicides for HIV and STI prevention. Mol. Pharm. 2005, 2, 312–318. [Google Scholar] [CrossRef] [PubMed]
- Tomalia, D.A.; Fréchet, J.M.J. Discovery of dendrimers and dendritic polymers: A brief historical perspective. J. Polym. Sci. Part A 2002, 40, 2719–2728. [Google Scholar] [CrossRef]
- Buhleier, E.; Wehner, W.; Vöegtle, F. “Cascade”- and “nonskid-chain-like” syntheses of molecular cavity topologies. Synthesis 1978, 9, 155–158. [Google Scholar] [CrossRef]
- Denkewalter, R.G.; Kolc, J.; Lukasavage, W.J. Macromolecular Highly Branched Homogeneous Compound Based on Lysine Units. U.S. Patent 4,289,872, 15 September 1981. [Google Scholar]
- Tomalia, D.A.; Baker, H.; Dewald, J.; Hall, M.; Kallos, G.; Martin, S.; Roeck, J.; Ryder, J.; Smith, P. A new class of polymers: Starburst-dendritic macromolecules. Polym. J. 1985, 17, 117–132. [Google Scholar] [CrossRef]
- Newkome, G.R.; Yao, Z.; Baker, G.R.; Gupta, V.K. Micelles. Part 1. Cascade molecules: A new approach to micelles. A [27]-arborol. J. Org. Chem. 1985, 50, 2003–2004. [Google Scholar] [CrossRef]
- Wörner, C.; Mülhaupt, R. Polynitril- und polyaminfunktionalisierte poly(trimethylenimin)-dendrimere. Angew. Chem. 1993, 105, 1367–1370. [Google Scholar] [CrossRef]
- De Brabander-van den Berg, E.; Meijer, E.W. Poly(propylene imine) dendrimers: Large-scale synthesis by hetereogeneously catalyzed hydrogenations. Angew. Chem. Int. Ed. 1993, 32, 1308–1311. [Google Scholar] [CrossRef]
- Tomalia, D.A.; Naylor, A.M.; Goddard, W.A. Starburst dendrimers: Molecular-level control of size, shape, surface chemistry, topology, and flexibility from atoms to macroscopic matter. Angew. Chem. Int. Ed. 1990, 29, 138–175. [Google Scholar] [CrossRef]
- Sadler, K.; Tam, J.P. Peptide dendrimers: Applications and synthesis. J. Biotechnol. 2002, 90, 195–229. [Google Scholar] [CrossRef]
- Hawker, C.J.; Frechet, J.M.J. Preparation of polymers with controlled molecular architecture. A new convergent approach to dendritic macromolecules. J. Am. Chem. Soc. 1990, 112, 7638–7647. [Google Scholar] [CrossRef]
- Grinstaff, M.W. Biodendrimers: New polymeric biomaterials for tissue engineering. Chemistry 2002, 8, 2839–2846. [Google Scholar] [CrossRef]
- Ihre, H.; Hult, A.; Söderlind, E. Synthesis, characterization, and 1 h nmr self-diffusion studies of dendritic aliphatic polyesters based on 2,2-bis(hydroxymethyl)propionic acid and 1,1,1-tris(hydroxyphenyl)ethane. J. Am. Chem. Soc. 1996, 118, 6388–6395. [Google Scholar] [CrossRef]
- Turnbull, W.B.; Stoddart, J.F. Design and synthesis of glycodendrimers. J. Biotechnol. 2002, 90, 231–255. [Google Scholar] [CrossRef]
- Nilsen, T.W.; Grayzel, J.; Prensky, W. Dendritic nucleic acid structures. J. Theor. Biol. 1997, 187, 273–284. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Tseng, Y.D.; Kwon, S.Y.; D’Espaux, L.; Bunch, J.S.; McEuen, P.L.; Luo, D. Controlled assembly of dendrimer-like DNA. Nat. Mater. 2004, 3, 38–42. [Google Scholar] [CrossRef] [PubMed]
- Lundquist, J.J.; Toone, E.J. The cluster glycoside effect. Chem. Rev. 2002, 102, 555–578. [Google Scholar] [CrossRef] [PubMed]
- Mammen, M.; Choi, S.-K.; Whitesides, G.M. Polyvalent interactions in biological systems: Implications for design and use of multivalent ligands and inhibitors. Angew. Chem. Int. Ed. 1998, 37, 2754–2794. [Google Scholar] [CrossRef]
- Boas, U.; Heegaard, P.M.H. Dendrimers in drug research. Chem. Soc. Rev. 2004, 33, 43–63. [Google Scholar] [CrossRef] [PubMed]
- Boas, U.; Christensen, J.B.; Heegaard, P.M.H. Dendrimers in Medicine and Biotechnology: New Molecular Tools; Royal Society of Chemistry: London, UK, 2006. [Google Scholar]
- Grayson, S.M.; Frèchet, J.M.J. Convergent dendrons and dendrimers: From synthesis to applications. Chem. Rev. 2001, 101, 3819–3868. [Google Scholar] [CrossRef] [PubMed]
- Hawker, C.; Frechet, M.J. A new convergent approach to monodisperse dendritic macromolecules. J. Chem. Soc. Chem. Commun. 1990, 15, 1010–1013. [Google Scholar] [CrossRef]
- Caminade, A.-M.; Laurent, R.; Delavaux-Nicot, B.; Majoral, J.-P. “Janus” dendrimers: Syntheses and properties. New J. Chem. 2012, 36, 217–226. [Google Scholar] [CrossRef]
- Walter, M.V.; Malkoch, M. Simplifying the synthesis of dendrimers: Accelerated approaches. Chem. Soc. Rev. 2012, 41, 4593–4609. [Google Scholar] [CrossRef] [PubMed]
- Such, G.K.; Johnston, A.P.R.; Liang, K.; Caruso, F. Synthesis and functionalization of nanoengineered materials using click chemistry. Prog. Polym. Sci. 2012, 37, 985–1003. [Google Scholar] [CrossRef]
- Valério, C.; Ruiz, J.; Alonso, E.; Boussaguet, P.; Guittard, J.; Blais, J.-C.; Astruc, D. Syntheses of polyamine-and polynitrile dendrimers from a nona-arm core up to 144-nitrile using vogtle’s iteration. Bull. Soc. Chim. Fr. 1997, 134, 907–914. [Google Scholar]
- Wörner, C.; Mülhaupt, R. Polynitrile- and Polyamine-Functional Poly(trimetilene imine) Dendrimers. Angew. Chem. Int. Ed. 1993, 105, 1306–1308. [Google Scholar] [CrossRef]
- De Brabander-van den Berg, E.M.M.; Nijenhuis, A.; Mure, M.; Keulen, J.; Reintjens, R.; Vandenbooren, F.; Bosman, B.; de Raat, R.; Frijns, T. Large-scale production of polypropylenimine dendrimers. Macromol. Symp. 1994, 77, 51–62. [Google Scholar] [CrossRef]
- Hadjichristidis, N.; Guyot, A.; Fetters, L.J. Star-branched polymers. 1. The synthesis of star polyisoprenes using octa- and dodecachlorosilanes as linking agents. Macromolecules 1978, 11, 668–672. [Google Scholar] [CrossRef]
- Van der Made, A.W.; Van Leeuwen, P.W. Silane dendrimers. J. Chem. Soc. Chem. Commun. 1992, 19, 1400–1401. [Google Scholar] [CrossRef]
- Muzafarov, A.M.; Rebrov, E.A. From the discovery of sodiumoxyorganoalkoxysilanes to the organosilicon dendrimers and back. J. Polym. Sci. Part A 2008, 46, 4935–4948. [Google Scholar] [CrossRef]
- Zhou, L.L.; Roovers, J. Synthesis of novel carbosilane dendritic macromolecules. Macromolecules 1993, 26, 963–968. [Google Scholar] [CrossRef]
- Alonso, B.; Cuadrado, I.; Moran, M.; Losada, J. Organometallic silicon dendrimers. J. Chem. Soc. Chem. Commun. 1994, 2575–2576. [Google Scholar] [CrossRef]
- Lobete, F.; Cuadrado, I.; Casado, C.M.; Alonso, B.; Moran, M.; Losada, J. Silicon-based organometallic dendritic macromolecules containing {η6-(organosilyl)arene}chromium tricarbonyl moieties. J. Organomet. Chem. 1996, 509, 109–113. [Google Scholar] [CrossRef]
- Arnaiz, E.; Doucede, L.I.; Garcia-Gallego, S.; Urbiola, K.; Gomez, R.; Tros de Ilarduya, C.; de la Mata, F.J. Synthesis of cationic carbosilane dendrimers via click chemistry and their use as effective carriers for DNA transfection into cancerous cells. Mol. Pharm. 2012, 9, 433–447. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Nieves, J.; Ortega, P.; Muñoz-Fernandez, M.A.; Gomez, R.; de la Mata, F.J. Synthesis of carbosilane dendrons and dendrimers derived from 1,3,5-trihydroxybenzene. Tetrahedron 2010, 66, 9203–9213. [Google Scholar] [CrossRef]
- Denkewalter, R.G.; Kolc, J.F.; Lukasavage, W.J. Macromolecular Highly Branched Diamino Carboxylic Acids. U.S. Patent 4,410,688, 18 October 1983. [Google Scholar]
- Crespo, L.; Sanclimens, G.; Pons, M.; Giralt, E.; Royo, M.; Albericio, F. Peptide and amide bond-containing dendrimers. Chem. Rev. 2005, 105, 1663–1681. [Google Scholar] [CrossRef] [PubMed]
- Heegaard, P.M.H.; Boas, U.; Sorensen, N.S. Dendrimers for vaccine and immunostimulatory uses. A review. Bioconjug. Chem. 2010, 21, 405–418. [Google Scholar] [CrossRef] [PubMed]
- Rengan, K.; Engel, R. Phosphonium cascade molecules. J. Chem. Soc. Chem. Commun. 1990, 16, 1084–1085. [Google Scholar] [CrossRef]
- Caminade, A.-M.; Majoral, J.-P. Phosphorus-Containing Dendritic Architectures: Synthesis and Applications; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2012. [Google Scholar]
- Caminade, A.-M.; Majoral, J.-P. Phosphorus-containing dendrimers: Uses as catalysts, for materials, and in biology. In Catalysis by Metal Complexes; Springer: Dordrecht, The Netherlands, 2011; Volume 37, pp. 265–303. [Google Scholar]
- Caminade, A.-M.; Turrin, C.-O.; Laurent, R.; Maraval, A.; Majoral, J.-P. Synthetic pathways towards phosphorus dendrimers and dendritic architectures. Curr. Org. Chem. 2006, 10, 2333–2355. [Google Scholar] [CrossRef]
- Caminade, A.-M.; Maraval, V.; Laurent, R.; Turrin, C.-O.; Sutra, P.; Leclaire, J.; Griffe, L.; Marchand, P.; Baudoin-Dehoux, C.; Rebout, C.; et al. Phosphorus dendrimers: From synthesis to applications. C. R. Chim. 2003, 6, 791–801. [Google Scholar] [CrossRef]
- Miller, T.M.; Neenan, T.X. Convergent synthesis of monodisperse dendrimers based upon 1,3,5-trisubstituted benzenes. Chem. Mater. 1990, 2, 346–349. [Google Scholar] [CrossRef]
- Lim, J.; Simanek, E.E. Triazine dendrimers as drug delivery systems: From synthesis to therapy. Adv. Drug Deliv. Rev. 2012, 64, 826–835. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Simanek, E.E. Dendrimers based on melamine. Divergent and orthogonal, convergent syntheses of a G3 dendrimer. Org. Lett. 2000, 2, 843–845. [Google Scholar] [CrossRef] [PubMed]
- Ramalinga, U.; Clogston, J.D.; Patri, A.K.; Simpson, J.T. Characterization of nanoparticles by matrix assisted laser desorption ionization time-of-flight mass spectrometry. Methods Mol. Biol. 2011, 697, 53–61. [Google Scholar] [PubMed]
- Baytekin, B.; Werner, N.; Luppertz, F.; Engeser, M.; Brueggemann, J.; Bitter, S.; Henkel, R.; Felder, T.; Schalley, C.A. How useful is mass spectrometry for the characterization of dendrimers? “Fake defects” in the ESI and MALDI mass spectra of dendritic compounds. Int. J. Mass Spectrom. 2006, 249, 138–148. [Google Scholar] [CrossRef]
- Bosman, A.W.; Janssen, H.M.; Meijer, E.W. About dendrimers: Structure, physical properties, and applications. Chem. Rev. 1999, 99, 1665–1688. [Google Scholar] [CrossRef] [PubMed]
- Qi, Z.; Schalley, C.A. Host-guest chemistry of dendrimers in the gas phase. Supramol. Chem. 2010, 22, 672–682. [Google Scholar] [CrossRef]
- Chai, M.; Niu, Y.; Youngs, W.J.; Rinaldi, P.L. Structure and conformation of dab dendrimers in solution via multidimensional NMR techniques. J. Am. Chem. Soc. 2001, 123, 4670–4678. [Google Scholar] [CrossRef] [PubMed]
- Ortega, P.; Moreno, S.; Tarazona, M.P.; de la Mata, F.J.; Gomez, R.R. New hyperbranched carbosiloxane-carbosilane polymers with aromatic units in the backbone. Eur. Polym. J. 2012, 48, 1413–1421. [Google Scholar] [CrossRef]
- Lartigue, M.-L.; Donnadieu, B.; Galliot, C.; Caminade, A.-M.; Majoral, J.-P.; Fayet, J.-P. Large dipole moments of phosphorus-containing dendrimers. Macromolecules 1997, 30, 7335–7337. [Google Scholar] [CrossRef]
- Koper, G.J.M.; van Genderen, M.H.P.; Elissen-Román, C.; Baars, M.W.P.L.; Meijer, E.W.; Borkovec, M. Protonation mechanism of poly(propylene imine) dendrimers and some associated oligo amines. J. Am. Chem. Soc. 1997, 119, 6512–6521. [Google Scholar] [CrossRef]
- Hu, J.; Xu, T.; Cheng, Y. NMR insights into dendrimer-based host-guest systems. Chem. Rev. 2012, 112, 3856–3891. [Google Scholar] [CrossRef] [PubMed]
- Ottaviani, M.F.; Turro, N.J. Characterization of dendrimer structures by ESR techniques. In Advanced ESR Methods in Polymer Research; Schlick, S., Ed.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2006. [Google Scholar]
- Galán, M.; Sánchez Rodríguez, J.; Cangiotti, M.; García-Gallego, S.; Jiménez, J.L.; Gómez, R.; Ottaviani, M.F.; Muñoz-Fernández, M.A.; de la Mata, F.J. Antiviral properties against HIV of water soluble copper carbosilane dendrimers and their EPR characterization. Curr. Med. Chem. 2012, 19, 4984–4994. [Google Scholar] [CrossRef] [PubMed]
- García-Gallego, S.; Cangiotti, M.; Fiorani, L.; Fattori, A.; Muñoz-Fernández, M.Á.; Gómez, R.; Ottaviani, M.F.; de la Mata, F.J. Anionic sulfonated and carboxylated PPI dendrimers with EDA core: Synthesis and characterization as selective metal complexing agents. Dalton Trans. 2013, 42, 5874–5889. [Google Scholar] [CrossRef] [PubMed]
- Ottaviani, M.F.; Cangiotti, M.; Fiorani, L.; Fattori, A.; Wasiak, T.; Appelhans, D.; Klajnert, B. Kinetics of amyloid and prion fibril formation in the absence and presence of dense shell sugar-decorated dendrimers. Curr. Med. Chem. 2012, 19, 5907–5921. [Google Scholar] [CrossRef] [PubMed]
- Yuan, W.; Yuan, J.; Zhou, M.; Pan, C. Synthesis, characterization, and fluorescence of pyrene-containing eight-arm star-shaped dendrimer-like copolymer with pentaerythritol core. J. Polym. Sci. Part A 2008, 46, 2788–2798. [Google Scholar] [CrossRef]
- Kitabatake, M.; Sisido, M.; Kitamatsu, M. Synthesis of peptide dendrimers consisting of fluorescent groups surrounding with dendritic amino acids. In Proceedings of the 47th Japanese Peptide Symposium, Kyoto, Japan, 4–9 December 2010; p. 227. [Google Scholar]
- Paulo, P.M.R.; Costa, S.M.B. Single-molecule fluorescence of a phthalocyanine in PAMAM dendrimers reveals intensity-lifetime fluctuations from quenching dynamics. J. Phys. Chem. C 2010, 114, 19035–19043. [Google Scholar] [CrossRef]
- Rasines, B.; Sánchez-Nieves, J.; Molina, I.T.; Guzmán, M.; Muñoz-Fernández, M.Á.; Gómez, R.; de la Mata, F.J. Synthesis and fluorescent properties of cationic carbosilane dendrimers containing eugenol linkers for their use in biomedical applications. New J. Chem. 2012, 36, 360–370. [Google Scholar] [CrossRef]
- Miller, L.L.; Duan, R.G.; Tully, D.C.; Tomalia, D.A. Electrically conducting dendrimers. J. Am. Chem. Soc. 1997, 119, 1005–1010. [Google Scholar] [CrossRef]
- Furer, V.L.; Vandukova, I.I.; Majoral, J.P.; Caminade, A.M.; Kovalenko, V.I. Spectral additive properties of phosphorus-containing dendrimers. Vib. Spectrosc. 2007, 43, 351–357. [Google Scholar] [CrossRef]
- Archut, A.; Vogtle, F.; De, C.L.; Azzellini, G.C.; Balzani, V.; Ramanujam, P.S.; Berg, R.H. Azobenzene-functionalized cascade molecules: Photoswitchable supramolecular systems. Chem. Eur. J. 1998, 4, 699–706. [Google Scholar] [CrossRef]
- Kim, C.; Son, S. Preparation of double-layered dendritic carbosilanes. J. Organomet. Chem. 2000, 599, 123–127. [Google Scholar] [CrossRef]
- Hawker, C.J.; Wooley, K.L.; Frechet, J.M.J. Solvatochromism as a probe of the microenvironment in dendritic polyethers: Transition from an extended to a globular structure. J. Am. Chem. Soc. 1993, 115, 4375–4376. [Google Scholar] [CrossRef]
- Akiyama, H.; Miyashita, K.; Hari, Y.; Obika, S.; Imanishi, T. Synthesis of novel polyesteramine dendrimers by divergent and convergent methods. Tetrahedron 2013, 69, 6810–6820. [Google Scholar] [CrossRef]
- Voronina, N.V.; Tatarinova, E.A.; Bystrova, A.V.; Ignat’eva, G.M.; Myakushev, V.D.; Muzafarov, A.M. Gel permeation chromatography of the dendritic molecules: Applicability of the universal calibration method. Polym. Prepr. 2009, 50, 487–488. [Google Scholar]
- Van Dongen Mallory, A.; Desai, A.; Orr, B.G.; Baker, J.R.; Banaszak, H.M.M. Quantitative analysis of generation and branch defects in g5 poly(amidoamine) dendrimer. Polymer 2013, 54, 4126–4133. [Google Scholar] [CrossRef] [PubMed]
- Frechet, J.M.J.; Lochman, L.; Smigol, V.; Svec, F. Reversed-phase high-performance liquid chromatography of functionalized dendritic macromolecules. J. Chromatogr. A 1994, 667, 284–289. [Google Scholar] [CrossRef]
- Wang, X.; Guerrand, L.; Wu, B.; Li, X.; Boldon, L.; Chen, W.-R.; Liu, L. Characterizations of polyamidoamine dendrimers with scattering techniques. Polymers 2012, 4, 600–616. [Google Scholar] [CrossRef]
- Poetschke, D.; Ballauff, M.; Lindner, P.; Fischer, M.; Voegtle, F. Analysis of the structure of dendrimers in solution by small-angle neutron scattering including contrast variation. Macromolecules 1999, 32, 4079–4087. [Google Scholar] [CrossRef]
- Topp, A.; Bauer, B.J.; Tomalia, D.A.; Amis, E.J. Effect of solvent quality on the molecular dimensions of pamam dendrimers. Macromolecules 1999, 32, 7232–7237. [Google Scholar] [CrossRef]
- Topp, A.; Bauer, B.J.; Klimash, J.W.; Spindler, R.; Tomalia, D.A.; Amis, E.J. Probing the location of the terminal groups of dendrimers in dilute solution. Macromolecules 1999, 32, 7226–7231. [Google Scholar] [CrossRef]
- Rosenfeldt, S.; Dingenouts, N.; Ballauff, M.; Werner, N.; Voegtle, F.; Lindner, P. Distribution of end groups within a dendritic structure: A sans study including contrast variation. Macromolecules 2002, 35, 8098–8105. [Google Scholar] [CrossRef]
- Li, X.; Zamponi, M.; Hong, K.; Porcar, L.; Shew, C.-Y.; Jenkins, T.; Liu, E.; Smith, G.S.; Herwig, K.W.; Liu, Y.; et al. pH responsiveness of polyelectrolyte dendrimers: A dynamical perspective. Soft Matter 2011, 7, 618–622. [Google Scholar] [CrossRef]
- Prosa, T.J.; Bauer, B.J.; Amis, E.J.; Tomalia, D.A.; Scherrenberg, R. A saxs study of the internal structure of dendritic polymer systems. J. Polym. Sci. Part B 1997, 35, 2913–2924. [Google Scholar] [CrossRef]
- Prosa, T.J.; Bauer, B.J.; Amis, E.J. From stars to spheres: A saxs analysis of dilute dendrimer solutions. Macromolecules 2001, 34, 4897–4906. [Google Scholar] [CrossRef]
- Jachimska, B.; Lapczynska, M.; Zapotoczny, S. Reversible swelling process of sixth-generation poly(amido amine) dendrimers molecule as determined by quartz crystal microbalance technique. J. Phys. Chem. C 2013, 117, 1136–1145. [Google Scholar] [CrossRef]
- Zhang, J.; Hu, J.; Feng, X.; Li, Y.; Zhao, L.; Xu, T.; Cheng, Y. Interactions between oppositely charged dendrimers. Soft Matter 2012, 8, 9800–9806. [Google Scholar] [CrossRef]
- Betley, T.A.; Banaszak, H.M.M.; Orr, B.G.; Swanson, D.R.; Tomalia, D.A.; Baker, J.R., Jr. Tapping mode atomic force microscopy investigation of poly(amidoamine) dendrimers: Effects of substrate and pH on dendrimer deformation. Langmuir 2001, 17, 2768–2773. [Google Scholar] [CrossRef]
- Pedziwiatr-Werbicka, E.; Shcharbin, D.; Maly, J.; Maly, M.; Zaborski, M.; Gabara, B.; Ortega, P.; de la Mata, F.J.; Gomez, R.; Munoz-Fernandez, M.A.; et al. Carbosilane dendrimers are a non-viral delivery system for antisense oligonucleotides: Characterization of dendriplexes. J. Biomed. Nanotechnol. 2012, 8, 57–73. [Google Scholar] [CrossRef] [PubMed]
- Pu, Y.; Chang, S.; Yuan, H.; Wang, G.; He, B.; Gu, Z. The anti-tumor efficiency of poly(l-glutamic acid) dendrimers with polyhedral oligomeric silsesquioxane cores. Biomaterials 2013, 34, 3658–3666. [Google Scholar] [CrossRef] [PubMed]
- Shi, L.; Fleming, C.J.; Riechers, S.L.; Yin, N.-N.; Luo, J.; Lam, K.S.; Liu, G.-Y. High-resolution imaging of dendrimers used in drug delivery via scanning probe microscopy. J. Drug Deliv. 2011, 2011, 254095. [Google Scholar] [CrossRef] [PubMed]
- Biricova, V.; Laznickova, A. Dendrimers: Analytical characterization and applications. Bioorg. Chem. 2009, 37, 185–192. [Google Scholar] [CrossRef] [PubMed]
- Shi, X.; Patri, A.K.; Lesniak, W.; Islam, M.T.; Zhang, C.; Baker, J.R., Jr.; Balogh, L.P. Analysis of poly(amidoamine)-succinamic acid dendrimers by slab-gel electrophoresis and capillary zone electrophoresis. Electrophoresis 2005, 26, 2960–2967. [Google Scholar] [CrossRef] [PubMed]
- Castagnola, M.; Zuppi, C.; Rossetti, D.V.; Vincenzoni, F.; Lupi, A.; Vitali, A.; Meucci, E.; Messana, I. Characterization of dendrimer properties by capillary electrophoresis and their use as pseudostationary phases. Electrophoresis 2002, 23, 1769–1778. [Google Scholar] [CrossRef]
- Montealegre, C.; Rasines, B.; Gomez, R.; de la Mata, F.J.; Garcia-Ruiz, C.; Marina, M.L. Characterization of carboxylate-terminated carbosilane dendrimers and their evaluation as nanoadditives in capillary electrophoresis for vegetable protein profiling. J. Chromatogr. A 2012, 1234, 16–21. [Google Scholar] [CrossRef] [PubMed]
- Stoeckigt, D.; Lohmer, G.; Belder, D. Separation and identification of basic dendrimers using capillary electrophoresis online coupled to a sector mass spectrometer. Rapid Commun. Mass Spectrom. 1996, 10, 521–526. [Google Scholar] [CrossRef]
- Shcharbin, D.; Pedziwiatr, E.; Bryszewska, M. How to study dendriplexes I: Characterization. J. Control. Release 2009, 135, 186–197. [Google Scholar] [CrossRef] [PubMed]
- Weber, N.; Ortega, P.; Clemente, M.I.; Shcharbin, D.; Bryszewska, M.; de la Mata, F.J.; Gomez, R.; Munoz-Fernandez, M.A. Characterization of carbosilane dendrimers as effective carriers of siRNA to HIV-infected lymphocytes. J. Control. Release 2008, 132, 55–64. [Google Scholar] [CrossRef] [PubMed]
- Brewis, M.; Clarkson, G.J.; Goddard, V.; Helliwell, M.; Holder, A.M.; McKeown, N.B. Silicon phthalocyanines with axial dendritic substituents. Angew. Chem. Int. Ed. 1998, 37, 1092–1094. [Google Scholar] [CrossRef]
- Peterca, M.; Percec, V.; Imam, M.R.; Leowanawat, P.; Morimitsu, K.; Heiney, P.A. Molecular structure of helical supramolecular dendrimers. J. Am. Chem. Soc. 2008, 130, 14840–14852. [Google Scholar] [CrossRef] [PubMed]
- Percec, V.; Imam, M.R.; Peterca, M.; Leowanawat, P. Self-organizable vesicular columns assembled from polymers dendronized with semifluorinated janus dendrimers act as reverse thermal actuators. J. Am. Chem. Soc. 2012, 134, 4408–4420. [Google Scholar] [CrossRef] [PubMed]
- Cakara, D.; Kleimann, J.; Borkovec, M. Microscopic protonation equilibria of poly(amidoamine) dendrimers from macroscopic titrations. Macromolecules 2003, 36, 4201–4207. [Google Scholar] [CrossRef]
- Malik, N.; Evagorou, E.G.; Duncan, R. Dendrimer-platinate: A novel approach to cancer chemotherapy. Anticancer Drugs 1999, 10, 767–776. [Google Scholar] [CrossRef] [PubMed]
- Kukowska-Latallo, J.F.; Candido, K.A.; Cao, Z.; Nigavekar, S.S.; Majoros, I.J.; Thomas, T.P.; Balogh, L.P.; Khan, M.K.; Baker, J.R., Jr. Nanoparticle targeting of anticancer drug improves therapeutic response in animal model of human epithelial cancer. Cancer Res. 2005, 65, 5317–5324. [Google Scholar] [CrossRef] [PubMed]
- Bhadra, D.; Bhadra, S.; Jain, S.; Jain, N.K. A pegylated dendritic nanoparticulate carrier of fluorouracil. Int. J. Pharm. 2003, 257, 111–124. [Google Scholar] [CrossRef]
- Kolhe, P.; Misra, E.; Kannan, R.M.; Kannan, S.; Lieh-Lai, M. Drug complexation, in vitro release and cellular entry of dendrimers and hyperbranched polymers. Int. J. Pharm. 2003, 259, 143–160. [Google Scholar] [CrossRef]
- El-Sayed, M.; Rhodes, C.A.; Ginski, M.; Ghandehari, H. Transport mechanism(s) of poly (amidoamine) dendrimers across caco-2 cell monolayers. Int. J. Pharm. 2003, 265, 151–157. [Google Scholar] [CrossRef]
- Yao, W.; Sun, K.; Mu, H.; Liang, N.; Liu, Y.; Yao, C.; Liang, R.; Wang, A. Preparation and characterization of puerarin-dendrimer complexes as an ocular drug delivery system. Drug Dev. Ind. Pharm. 2010, 36, 1027–1035. [Google Scholar] [CrossRef] [PubMed]
- Carberry, T.P.; Tarallo, R.; Falanga, A.; Finamore, E.; Galdiero, M.; Weck, M.; Galdiero, S. Dendrimer functionalization with a membrane-interacting domain of herpes simplex virus type 1: Towards intracellular delivery. Chemistry 2012, 18, 13678–13685. [Google Scholar] [CrossRef] [PubMed]
- Falanga, A.; Tarallo, R.; Carberry, T.; Galdiero, M.; Weck, M.; Galdiero, S. Elucidation of the interaction mechanism with liposomes of gh625-peptide functionalized dendrimers. PLoS ONE 2014, 9, e112128. [Google Scholar] [CrossRef] [PubMed]
- Galdiero, S.; Falanga, A.; Morelli, G.; Galdiero, M. Gh625: A milestone in understanding the many roles of membranotropic peptides. Biochim. Biophys. Acta 2015, 1848, 16–25. [Google Scholar] [CrossRef] [PubMed]
- Dhanikula, R.S.; Hammady, T.; Hildgen, P. On the mechanism and dynamics of uptake and permeation of polyether-copolyester dendrimers across an in vitro blood-brain barrier model. J. Pharm. Sci. 2009, 98, 3748–3760. [Google Scholar] [CrossRef] [PubMed]
- Beg, S.; Samad, A.; Alam, M.I.; Nazish, I. Dendrimers as novel systems for delivery of neuropharmaceuticals to the brain. CNS Neurol. Disord. Drug Targets 2011, 10, 576–588. [Google Scholar] [CrossRef] [PubMed]
- Jevprasesphant, R.; Penny, J.; Attwood, D.; McKeown, N.B.; D’Emanuele, A. Engineering of dendrimer surfaces to enhance transepithelial transport and reduce cytotoxicity. Pharm. Res. 2003, 20, 1543–1550. [Google Scholar] [CrossRef] [PubMed]
- Kannan, S.; Dai, H.; Navath, R.S.; Balakrishnan, B.; Jyoti, A.; Janisse, J.; Romero, R.; Kannan, R.M. Dendrimer-based postnatal therapy for neuroinflammation and cerebral palsy in a rabbit model. Sci. Transl. Med. 2012, 4, 130ra46. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; He, H.; Jia, X.; Lu, W.L.; Lou, J.; Wei, Y. A dual-targeting nanocarrier based on poly(amidoamine) dendrimers conjugated with transferrin and tamoxifen for treating brain gliomas. Biomaterials 2012, 33, 3899–3908. [Google Scholar] [CrossRef] [PubMed]
- Shcharbin, D.; Shakhbazau, A.; Bryszewska, M. Poly(amidoamine) dendrimer complexes as a platform for gene delivery. Expert Opin. Drug Deliv. 2013, 10, 1687–1698. [Google Scholar] [CrossRef] [PubMed]
- Merkel, O.M.; Mintzer, M.A.; Sitterberg, J.; Bakowsky, U.; Simanek, E.E.; Kissel, T. Triazine dendrimers as nonviral gene delivery systems: Effects of molecular structure on biological activity. Bioconjug. Chem. 2009, 20, 1799–1806. [Google Scholar] [CrossRef] [PubMed]
- Agrawal, A.; Min, D.H.; Singh, N.; Zhu, H.; Birjiniuk, A.; von Maltzahn, G.; Harris, T.J.; Xing, D.; Woolfenden, S.D.; Sharp, P.A.; et al. Functional delivery of siRNA in mice using dendriworms. ACS Nano 2009, 3, 2495–2504. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.I.; Seo, H.J.; Choi, J.S.; Jang, H.S.; Baek, J.U.; Kim, K.; Park, J.S. PAMAM-PEG-PAMAM: Novel triblock copolymer as a biocompatible and efficient gene delivery carrier. Biomacromolecules 2004, 5, 2487–2492. [Google Scholar] [CrossRef] [PubMed]
- Yuan, Q.; Yeudall, W.A.; Yang, H. Pegylated polyamidoamine dendrimers with bis-aryl hydrazone linkages for enhanced gene delivery. Biomacromolecules 2010, 11, 1940–1947. [Google Scholar] [CrossRef] [PubMed]
- Santos, J.L.; Oliveira, H.; Pandita, D.; Rodrigues, J.; Pego, A.P.; Granja, P.L.; Tomas, H. Functionalization of poly(amidoamine) dendrimers with hydrophobic chains for improved gene delivery in mesenchymal stem cells. J. Control. Release 2010, 144, 55–64. [Google Scholar] [CrossRef] [PubMed]
- Pandita, D.; Santos, J.L.; Rodrigues, J.; Pego, A.P.; Granja, P.L.; Tomas, H. Gene delivery into mesenchymal stem cells: A biomimetic approach using RGD nanoclusters based on poly(amidoamine) dendrimers. Biomacromolecules 2011, 12, 472–481. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.S.; Nam, K.; Park, J.Y.; Kim, J.B.; Lee, J.K.; Park, J.S. Enhanced transfection efficiency of PAMAM dendrimer by surface modification with l-arginine. J. Control. Release 2004, 99, 445–456. [Google Scholar] [CrossRef] [PubMed]
- Wada, K.; Arima, H.; Tsutsumi, T.; Chihara, Y.; Hattori, K.; Hirayama, F.; Uekama, K. Improvement of gene delivery mediated by mannosylated dendrimer/alpha-cyclodextrin conjugates. J. Control. Release 2005, 104, 397–413. [Google Scholar] [CrossRef] [PubMed]
- Wood, K.C.; Azarin, S.M.; Arap, W.; Pasqualini, R.; Langer, R.; Hammond, P.T. Tumor-targeted gene delivery using molecularly engineered hybrid polymers functionalized with a tumor-homing peptide. Bioconjug. Chem. 2008, 19, 403–405. [Google Scholar] [CrossRef] [PubMed]
- Waite, C.L.; Roth, C.M. PAMAM-RGD conjugates enhance siRNA delivery through a multicellular spheroid model of malignant glioma. Bioconjug. Chem. 2009, 20, 1908–1916. [Google Scholar] [CrossRef] [PubMed]
- Ghilardi, A.; Pezzoli, D.; Bellucci, M.C.; Malloggi, C.; Negri, A.; Sganappa, A.; Tedeschi, G.; Candiani, G.; Volonterio, A. Synthesis of multifunctional PAMAM-aminoglycoside conjugates with enhanced transfection efficiency. Bioconjug. Chem. 2013, 24, 1928–1936. [Google Scholar] [CrossRef] [PubMed]
- Luo, K.; Li, C.; Wang, G.; Nie, Y.; He, B.; Wu, Y.; Gu, Z. Peptide dendrimers as efficient and biocompatible gene delivery vectors: Synthesis and In Vitro characterization. J. Control. Release 2011, 155, 77–87. [Google Scholar] [CrossRef] [PubMed]
- Kaneshiro, T.L.; Lu, Z.R. Targeted intracellular codelivery of chemotherapeutics and nucleic acid with a well-defined dendrimer-based nanoglobular carrier. Biomaterials 2009, 30, 5660–5666. [Google Scholar] [CrossRef] [PubMed]
- Mintzer, M.A.; Simanek, E.E. Nonviral vectors for gene delivery. Chem. Rev. 2009, 109, 259–302. [Google Scholar] [CrossRef] [PubMed]
- Lakshminarayanan, A.; Ravi, V.K.; Tatineni, R.; Rajesh, Y.B.; Maingi, V.; Vasu, K.S.; Madhusudhan, N.; Maiti, P.K.; Sood, A.K.; Das, S.; et al. Efficient dendrimer-DNA complexation and gene delivery vector properties of nitrogen-core poly(propyl ether imine) dendrimer in mammalian cells. Bioconjug. Chem. 2013, 24, 1612–1623. [Google Scholar] [CrossRef] [PubMed]
- Nierengarten, I.; Nothisen, M.; Sigwalt, D.; Biellmann, T.; Holler, M.; Remy, J.S.; Nierengarten, J.F. Polycationic pillar[5]arene derivatives: Interaction with DNA and biological applications. Chemistry 2013, 19, 17552–17558. [Google Scholar] [CrossRef] [PubMed]
- Ma, D.; Zhao, Y.; Zhou, X.Y.; Lin, Q.M.; Zhang, Y.; Lin, J.T.; Xue, W. Photoenhanced gene transfection by a star-shaped polymer consisting of a porphyrin core and poly(l-lysine) dendron arms. Macromol. Biosci. 2013, 13, 1221–1227. [Google Scholar] [CrossRef] [PubMed]
- Ma, D.; Zhang, H.B.; Chen, Y.Y.; Lin, J.T.; Zhang, L.M. New cyclodextrin derivative containing poly(l-lysine) dendrons for gene and drug co-delivery. J. Colloid Interface Sci. 2013, 405, 305–311. [Google Scholar] [CrossRef] [PubMed]
- Abd-El-Aziza, A.S.; El-Sadanya, S.K.; Overya, D.P.; Kerra, R.G. Antimicrobial and anticancer activities of organoiron melamine dendrimers capped with piperazine moieties. Eur. Polym. J. 2016, 82, 307–323. [Google Scholar] [CrossRef]
- Rasines, B.; Hernandez-Ros, J.M.; de las Cuevas, N.; Copa-Patino, J.L.; Soliveri, J.; Munoz-Fernandez, M.A.; Gomez, R.; de la Mata, F.J. Water-stable ammonium-terminated carbosilane dendrimers as efficient antibacterial agents. Dalton Trans. 2009, 40, 8704–8713. [Google Scholar] [CrossRef] [PubMed]
- Matos, J.S.; White, D.G.; Harmon, R.J.; Langlois, B.E. Isolation of staphylococcus aureus from sites other than the lactating mammary gland. J. Dairy Sci. 1991, 74, 1544–1549. [Google Scholar] [CrossRef]
- Tanne, J.H. Data collection system needed for gun injuries. BMJ 2000, 321, 1370. [Google Scholar] [CrossRef] [PubMed]
- Ladd, E.; Sheikhi, A.; Li, N.; van de Ven, T.G.M.; Kakkar, A. Design and synthesis of dendrimers with facile surface group functionalization, and an evaluation of their bactericidal efficacy. Molecules 2017, 22, 868. [Google Scholar] [CrossRef] [PubMed]
- Jevprasesphant, R.; Penny, J.; Jalal, R.; Attwood, D.; McKeown, N.B.; D’Emanuele, A. The influence of surface modification on the cytotoxicity of PAMAM dendrimers. Int. J. Pharm. 2003, 252, 263–266. [Google Scholar] [CrossRef]
- Alencar de Queiroz, A.A.; Abraham, G.A.; Pires Camillo, M.A.; Higa, O.Z.; Silva, G.S.; del Mar Fernandez, M.; San Roman, J. Physicochemical and antimicrobial properties of boron-complexed polyglycerol-chitosan dendrimers. J. Biomater. Sci. Polym. Ed. 2006, 17, 689–707. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.Z.; Beck-Tan, N.C.; Dhurjati, P.; van Dyk, T.K.; LaRossa, R.A.; Cooper, S.L. Quaternary ammonium functionalized poly(propylene imine) dendrimers as effective antimicrobials: Structure-activity studies. Biomacromolecules 2000, 1, 473–480. [Google Scholar] [CrossRef] [PubMed]
- Bernstein, D.I.; Stanberry, L.R.; Sacks, S.; Ayisi, N.K.; Gong, Y.H.; Ireland, J.; Mumper, R.J.; Holan, G.; Matthews, B.; McCarthy, T.; et al. Evaluations of unformulated and formulated dendrimer-based microbicide candidates in mouse and guinea pig models of genital herpes. Antimicrob. Agents Chemother. 2003, 47, 3784–3788. [Google Scholar] [CrossRef] [PubMed]
- Pini, A.; Giuliani, A.; Falciani, C.; Runci, Y.; Ricci, C.; Lelli, B.; Malossi, M.; Neri, P.; Rossolini, G.M.; Bracci, L. Antimicrobial activity of novel dendrimeric peptides obtained by phage display selection and rational modification. Antimicrob. Agents Chemother. 2005, 49, 2665–2672. [Google Scholar] [CrossRef] [PubMed]
- Percec, V.; Dulcey, A.E.; Balagurusamy, V.S.; Miura, Y.; Smidrkal, J.; Peterca, M.; Nummelin, S.; Edlund, U.; Hudson, S.D.; Heiney, P.A.; et al. Self-assembly of amphiphilic dendritic dipeptides into helical pores. Nature 2004, 430, 764–768. [Google Scholar] [CrossRef] [PubMed]
- Mecke, A.; Lee, I.; Baker, J.R., Jr.; Holl, M.M.; Orr, B.G. Deformability of poly(amidoamine) dendrimers. Eur. Phys. J. E 2004, 14, 7–16. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Young, A.W.; Hu, P.; Rice, A.J.; Zhou, C.; Zhang, Y.; Kallenbach, N.R. Tuning the membrane selectivity of antimicrobial peptides by using multivalent design. ChemBioChem 2007, 8, 2063–2065. [Google Scholar] [CrossRef] [PubMed]
- Scorciapino, M.A.; Serra, I.; Manzo, G.; Rinaldi, A.C. Antimicrobial dendrimeric peptides: Structure, activity and new therapeutic applications. Int. J. Mol. Sci. 2017, 18, 542. [Google Scholar] [CrossRef] [PubMed]
- Pires, J.; Siriwardena, T.N.; Stach, M.; Tinguely, R.; Kasraian, S.; Luzzaro, F.; Leib, S.L.; Darbre, T.; Reymond, J.L.; Endimiani, A. In Vitro activity of the novel antimicrobial peptide dendrimer g3kl against multidrug-resistant acinetobacter baumannii and pseudomonas aeruginosa. Antimicrob. Agents Chemother. 2015, 59, 7915–7918. [Google Scholar] [CrossRef] [PubMed]
- Hou, S.; Zhou, C.; Liu, Z.; Young, A.W.; Shi, Z.; Ren, D.; Kallenbach, N.R. Antimicrobial dendrimer active against escherichia coli biofilms. Bioorg. Med. Chem. Lett. 2009, 19, 5478–5481. [Google Scholar] [CrossRef] [PubMed]
- Lopez, A.I.; Kumar, A.; Planas, M.R.; Li, Y.; Nguyen, T.V.; Cai, C. Biofunctionalization of silicone polymers using poly(amidoamine) dendrimers and a mannose derivative for prolonged interference against pathogen colonization. Biomaterials 2011, 32, 4336–4346. [Google Scholar] [CrossRef] [PubMed]
- Johansson, E.M.; Crusz, S.A.; Kolomiets, E.; Buts, L.; Kadam, R.U.; Cacciarini, M.; Bartels, K.M.; Diggle, S.P.; Camara, M.; Williams, P.; et al. Inhibition and dispersion of pseudomonas aeruginosa biofilms by glycopeptide dendrimers targeting the fucose-specific lectin lecb. Chem. Boil. 2008, 15, 1249–1257. [Google Scholar] [CrossRef] [PubMed]
- Bahar, A.A.; Liu, Z.; Totsingan, F.; Buitrago, C.; Kallenbach, N.; Ren, D. Synthetic dendrimeric peptide active against biofilm and persister cells of pseudomonas aeruginosa. Appl. Microbiol. Biotechnol. 2015, 99, 8125–8135. [Google Scholar] [CrossRef] [PubMed]
- Thompson, J.P.; Schengrund, C.L. Inhibition of the adherence of cholera toxin and the heat-labile enterotoxin of Escherichia coli to cell-surface gm1 by oligosaccharide-derivatized dendrimers. Biochem. Pharmacol. 1998, 56, 591–597. [Google Scholar] [CrossRef]
- Thompson, J.P.; Schengrund, C.L. Oligosaccharide-derivatized dendrimers: Defined multivalent inhibitors of the adherence of the cholera toxin b subunit and the heat labile enterotoxin of E. coli to gm1. Glycoconj. J. 1997, 14, 837–845. [Google Scholar] [CrossRef] [PubMed]
- Rahal, E.A.; Fadlallah, S.M.; Nassar, F.J.; Kazzi, N.; Matar, G.M. Approaches to treatment of emerging Shiga toxin-producing Escherichia coli infections highlighting the o104:H4 serotype. Front. Cell. Infect. Microb. 2015, 5, 24. [Google Scholar] [CrossRef] [PubMed]
- Nagahori, N.; Lee, R.T.; Nishimura, S.; Page, D.; Roy, R.; Lee, Y.C. Inhibition of adhesion of type 1 fimbriated escherichia coli to highly mannosylated ligands. ChemBioChem 2002, 3, 836–844. [Google Scholar] [CrossRef]
- Cheng, Y.; Qu, H.; Ma, M.; Xu, Z.; Xu, P.; Fang, Y.; Xu, T. Polyamidoamine (PAMAM) dendrimers as biocompatible carriers of quinolone antimicrobials: An In Vitro study. Eur. J. Med. Chem. 2007, 42, 1032–1038. [Google Scholar] [CrossRef] [PubMed]
- Agnihotri, S.A.; Mallikarjuna, N.N.; Aminabhavi, T.M. Recent advances on chitosan-based micro- and nanoparticles in drug delivery. J. Control. Release 2004, 100, 5–28. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.K.; Myc, A.; Silpe, J.E.; Sumit, M.; Wong, P.T.; McCarthy, K.; Desai, A.M.; Thomas, T.P.; Kotlyar, A.; Holl, M.M.; et al. Dendrimer-based multivalent vancomycin nanoplatform for targeting the drug-resistant bacterial surface. ACS Nano 2013, 7, 214–228. [Google Scholar] [CrossRef] [PubMed]
- Abd-El-Aziz, A.S.; Agatemor, C.; Etkin, N.; Overy, D.P.; Lanteigne, M.; McQuillan, K.; Kerr, R.G. Antimicrobial organometallic dendrimers with tunable activity against multidrug-resistant bacteria. Biomacromolecules 2015, 16, 3694–3703. [Google Scholar] [CrossRef] [PubMed]
- Sepulveda-Crespo, D.; Cena-Diez, R.; Jimenez, J.L.; Angeles Munoz-Fernandez, M. Mechanistic studies of viral entry: An overview of dendrimer-based microbicides as entry inhibitors against both HIV and HSV-2 overlapped infections. Med. Res. Rev. 2017, 37, 149–179. [Google Scholar] [CrossRef] [PubMed]
- Galdiero, S.; Falanga, A.; Tarallo, R.; Russo, L.; Galdiero, E.; Cantisani, M.; Morelli, G.; Galdiero, M. Peptide inhibitors against herpes simplex virus infections. J. Pept. Sci. 2013, 19, 148–158. [Google Scholar] [CrossRef] [PubMed]
- Sepulveda-Crespo, D.; Gomez, R.; De La Mata, F.J.; Jimenez, J.L.; Munoz-Fernandez, M.A. Polyanionic carbosilane dendrimer-conjugated antiviral drugs as efficient microbicides: Recent trends and developments in HIV treatment/therapy. Nanomedicine 2015, 11, 1481–1498. [Google Scholar] [CrossRef] [PubMed]
- Abd-El-Aziz, A.S.; Agatemor, C.; Etkin, N. Antimicrobial resistance challenged with metal-based antimicrobial macromolecules. Biomaterials 2017, 118, 27–50. [Google Scholar] [CrossRef] [PubMed]
- Schillerstrom, J.E.; Sanchez-Reilly, S.; O’Donnell, L. Improving student comfort with death and dying discussions through facilitated family encounters. Acad. Psychiatry 2012, 36, 188–190. [Google Scholar] [CrossRef] [PubMed]
- Sepulveda-Crespo, D.; Lorente, R.; Leal, M.; Gomez, R.; De la Mata, F.J.; Jimenez, J.L.; Munoz-Fernandez, M.A. Synergistic activity profile of carbosilane dendrimer G2-STE16 in combination with other dendrimers and antiretrovirals as topical anti-HIV-1 microbicide. Nanomedicine 2014, 10, 609–618. [Google Scholar] [CrossRef] [PubMed]
- Parboosing, R.; Chonco, L.; de la Mata, F.J.; Govender, T.; Maguire, G.E.; Kruger, H.G. Potential inhibition of HIV-1 encapsidation by oligoribonucleotide-dendrimer nanoparticle complexes. Int. J. Nanomed. 2017, 12, 317–325. [Google Scholar] [CrossRef] [PubMed]
- Sepulveda-Crespo, D.; Jimenez, J.L.; Gomez, R.; De La Mata, F.J.; Majano, P.L.; Munoz-Fernandez, M.A.; Gastaminza, P. Polyanionic carbosilane dendrimers prevent hepatitis C virus infection in cell culture. Nanomedicine 2017, 13, 49–58. [Google Scholar] [CrossRef] [PubMed]
- Gorzkiewicz, M.; Klajnert-Maculewicz, B. Dendrimers as nanocarriers for nucleoside analogues. Eur. J. Pharm. Biopharm. 2017, 114, 43–56. [Google Scholar] [CrossRef] [PubMed]
- Angel, S.O.; Matrajt, M.; Echeverria, P.C. A review of recent patents on the protozoan parasite HSP90 as a drug target. Recent Pat. Biotechnol. 2013, 7, 2–8. [Google Scholar] [CrossRef] [PubMed]
- Dye, C. After 2015: Infectious diseases in a new era of health and development. Philos. Trans. R. Soc. Lond. B 2014, 369, 20130426. [Google Scholar] [CrossRef] [PubMed]
- Ouellette, M. Biochemical and molecular mechanisms of drug resistance in parasites. Trop. Med. Int. Health 2001, 6, 874–882. [Google Scholar] [CrossRef] [PubMed]
- Leekha, S.; Terrell, C.L.; Edson, R.S. General principles of antimicrobial therapy. Mayo Clin. Proc. 2011, 86, 156–167. [Google Scholar] [CrossRef] [PubMed]
- Wan, J.; Alewood, P.F. Peptide-decorated dendrimers and their bioapplications. Angew. Chem. Int. Ed. 2016, 55, 5124–5134. [Google Scholar] [CrossRef] [PubMed]
- Daftarian, P.M.; Stone, G.W.; Kovalski, L.; Kumar, M.; Vosoughi, A.; Urbieta, M.; Blackwelder, P.; Dikici, E.; Serafini, P.; Duffort, S.; et al. A targeted and adjuvanted nanocarrier lowers the effective dose of liposomal amphotericin b and enhances adaptive immunity in murine cutaneous leishmaniasis. J. Infect. Dis. 2013, 208, 1914–1922. [Google Scholar] [CrossRef] [PubMed]
- Jain, K.; Verma, A.K.; Mishra, P.R.; Jain, N.K. Surface-engineered dendrimeric nanoconjugates for macrophage-targeted delivery of amphotericin B: Formulation development and In Vitro and In Vivo evaluation. Antimicrob. Agents Chemother. 2015, 59, 2479–2487. [Google Scholar] [CrossRef] [PubMed]
- Sinha, S.; Medhi, B.; Sehgal, R. Challenges of drug-resistant malaria. Parasite 2014, 21, 61. [Google Scholar] [CrossRef] [PubMed]
- Kaushik, N.K.; Sharma, J.; Sahal, D. Anti-plasmodial action of de novo-designed, cationic, lysine-branched, amphipathic, helical peptides. Malar. J. 2012, 11, 256. [Google Scholar] [CrossRef] [PubMed]
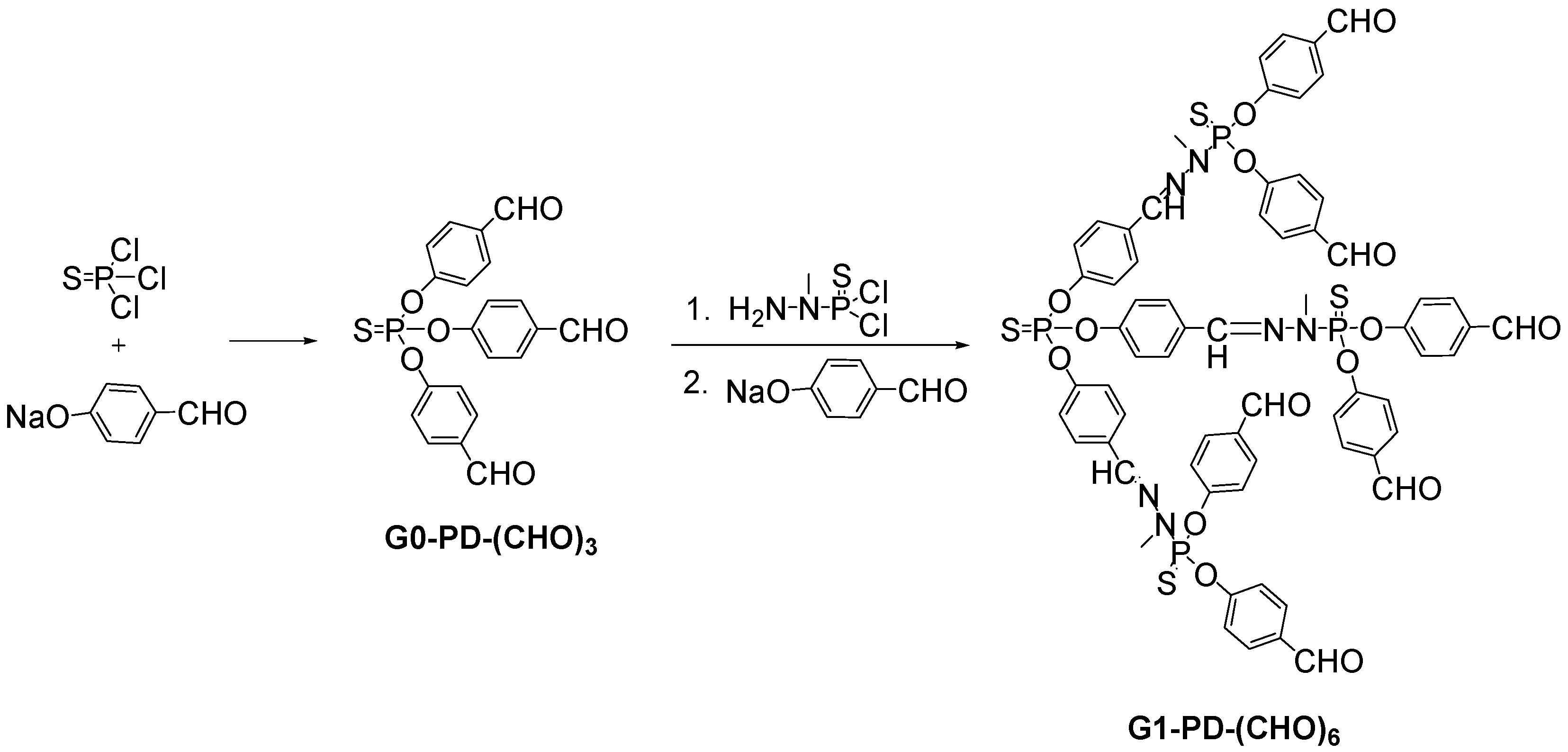
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
García-Gallego, S.; Franci, G.; Falanga, A.; Gómez, R.; Folliero, V.; Galdiero, S.; De la Mata, F.J.; Galdiero, M. Function Oriented Molecular Design: Dendrimers as Novel Antimicrobials. Molecules 2017, 22, 1581. https://doi.org/10.3390/molecules22101581
García-Gallego S, Franci G, Falanga A, Gómez R, Folliero V, Galdiero S, De la Mata FJ, Galdiero M. Function Oriented Molecular Design: Dendrimers as Novel Antimicrobials. Molecules. 2017; 22(10):1581. https://doi.org/10.3390/molecules22101581
Chicago/Turabian StyleGarcía-Gallego, Sandra, Gianluigi Franci, Annarita Falanga, Rafael Gómez, Veronica Folliero, Stefania Galdiero, Francisco Javier De la Mata, and Massimiliano Galdiero. 2017. "Function Oriented Molecular Design: Dendrimers as Novel Antimicrobials" Molecules 22, no. 10: 1581. https://doi.org/10.3390/molecules22101581
APA StyleGarcía-Gallego, S., Franci, G., Falanga, A., Gómez, R., Folliero, V., Galdiero, S., De la Mata, F. J., & Galdiero, M. (2017). Function Oriented Molecular Design: Dendrimers as Novel Antimicrobials. Molecules, 22(10), 1581. https://doi.org/10.3390/molecules22101581







