Assignment of Absolute Configuration of a New Hepatoprotective Schiartane-Type Nortriterpenoid Using X-Ray Diffraction
Abstract
:1. Introduction
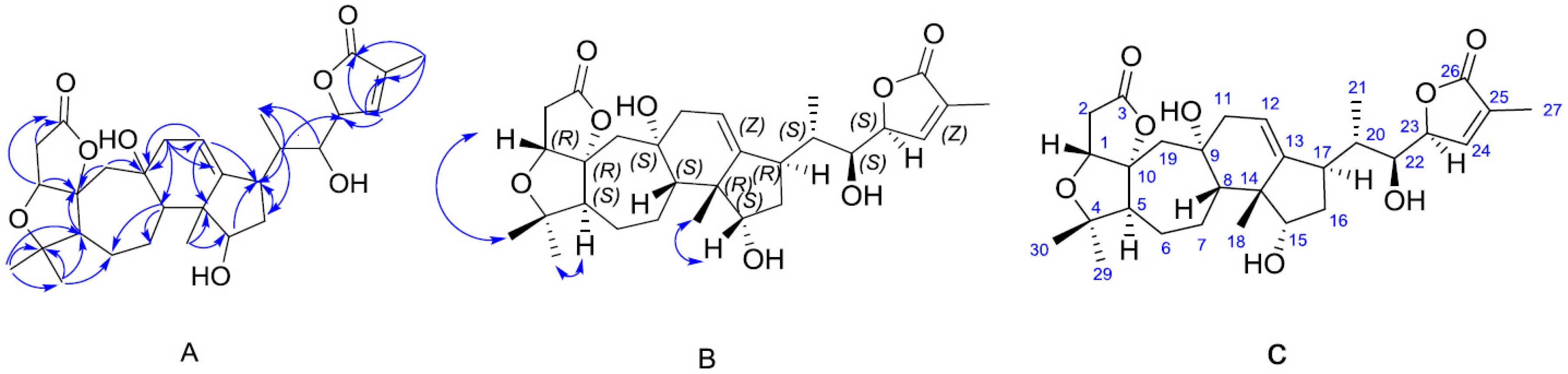
2. Results
3. Materials and Methods
3.1. General Experimental Procedures
3.2. Plant Material
3.3. Extraction and Isolation
3.4. X-Ray Crystallographic Analysis of Micrandilactone H
3.5. Hepatoprotective Activity Assay
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Lei, C.; Huang, S.-X.; Chen, J.-J.; Yang, L.-B.; Xiao, W.-L.; Chang, Y.; Lu, Y.; Huang, H.; Pu, J.-X.; Sun, H.-D. Propindilactones E−J, schiartane nortriterpenoids from Schisandra propinqua var. propinqua. J. Nat. Prod. 2008, 71, 1228–1232. [Google Scholar] [CrossRef] [PubMed]
- Xiao, W.-L.; Li, R.-T.; Huang, S.-X.; Pu, J.-X.; Sun, H.-D. Triterpenoids from the Schisandraceae family. Nat. Prod. Rep. 2008, 25, 871–891. [Google Scholar] [CrossRef] [PubMed]
- Li, R.-T.; Han, Q.-B.; Zheng, Y.-T.; Wang, R.-R.; Yang, L.-M.; Lu, Y.; Sang, S.-Q.; Zheng, Q.-T.; Zhao, Q.-S.; Sun, H.-D. Structure and anti-HIV activity of micrandilactones B and C, new nortriterpenoids possessing a unique skeleton from Schisandra micrantha. Chem. Commun. 2005, 23, 2936–2938. [Google Scholar] [CrossRef] [PubMed]
- Gawronski, J.K.; van Oeveren, A.; van der Deen, H.; Leung, C.W.; Feringa, B.L. Simple circular dichroic method for the determination of absolute configuration of 5-substituted 2(5H)-furanones. J. Org. Chem. 1996, 61, 1513–1515. [Google Scholar] [CrossRef]
- Sheldrick, G.M. Crystal structure refinement with SHELXL. Acta Cryst. 2015, C71, 3–8. [Google Scholar]
- Parsons, S.; Flack, H.D.; Wagner, T. Use of intensity quotients and differences in absolute structure refinement. Acta Cryst. 2013, B69, 249–259. [Google Scholar] [CrossRef] [PubMed]
- Li, C.-J.; Ma, J.; Sun, H.; Zhang, D.; Zhang, D.-M. Guajavadimer A, a dimeric caryophyllene-derived meroterpenoid with a new carbon skeleton from the leaves of Psidium guajava. Org. Lett. 2015, 18, 168–171. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Sample of micrandilactone H is not available.

| Position | δH | δC |
|---|---|---|
| 1 | 4.27 (d, 4.5) | 82.5 |
| 2α | 2.72 (d, 18.0) | 36.9 |
| 2β | 2.94 (dd, 18.0, 4.5) | |
| 3 | 175.9 | |
| 4 | 85.2 | |
| 5 | 2.57 (dd, 13.0, 4.0) | 59.6 |
| 6α | 1.69 (overlapped) | 28.7 |
| 6β | 1.31 (overlapped) | |
| 7α | 2.37 (m) | 25.3 |
| 7β | 2.07 (overlapped) | |
| 8 | 1.72 (overlapped) | 58.8 |
| 9 | 70.8 | |
| 10 | 99.7 | |
| 11α | 2.35 (m) | 42.7 |
| 11β | 2.26 (m) | |
| 12α | 5.76 (d, 7.0) | 120.9 |
| 12β | ||
| 13 | 147.7 | |
| 14 | 52.8 | |
| 15 | 4.00 (s) | 77.1 |
| 16α | 2.30 (m) | 40.4 |
| 16β | 2.11 (dd, 8.5, 3.0) | |
| 17 | 3.31 (m) | 43.3 |
| 18 | 1.12 (s) | 26.6 |
| 19α | 2.15 (d, 15.0) | 46.0 |
| 19β | 2.02 (d, 15.0) | |
| 20 | 2.21 (m) | 44.3 |
| 21 | 1.34 (d, 7.0) | 16.1 |
| 22 | 4.00 (s) | 74.9 |
| 23 | 5.27 (br, s) | 83.0 |
| 24 | 7.23 (s) | 148.9 |
| 25 | 130.5 | |
| 26 | 175.1 | |
| 27 | 1.83(s) | 10.9 |
| 29 | 1.27(s) | 29.9 |
| 30 | 23.5 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, X.; Fronczek, F.R.; Chen, J.; Liu, J.; Ferreira, D.; Li, S.; Hamann, M.T. Assignment of Absolute Configuration of a New Hepatoprotective Schiartane-Type Nortriterpenoid Using X-Ray Diffraction. Molecules 2017, 22, 65. https://doi.org/10.3390/molecules22010065
Wang X, Fronczek FR, Chen J, Liu J, Ferreira D, Li S, Hamann MT. Assignment of Absolute Configuration of a New Hepatoprotective Schiartane-Type Nortriterpenoid Using X-Ray Diffraction. Molecules. 2017; 22(1):65. https://doi.org/10.3390/molecules22010065
Chicago/Turabian StyleWang, Xiaojuan, Frank R. Fronczek, Jiabao Chen, Jiabao Liu, Daneel Ferreira, Shuai Li, and Mark T. Hamann. 2017. "Assignment of Absolute Configuration of a New Hepatoprotective Schiartane-Type Nortriterpenoid Using X-Ray Diffraction" Molecules 22, no. 1: 65. https://doi.org/10.3390/molecules22010065
APA StyleWang, X., Fronczek, F. R., Chen, J., Liu, J., Ferreira, D., Li, S., & Hamann, M. T. (2017). Assignment of Absolute Configuration of a New Hepatoprotective Schiartane-Type Nortriterpenoid Using X-Ray Diffraction. Molecules, 22(1), 65. https://doi.org/10.3390/molecules22010065





