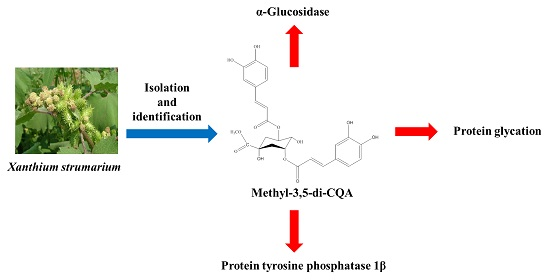
Xanthium strumarium as an Inhibitor of α-Glucosidase, Protein Tyrosine Phosphatase 1β, Protein Glycation and ABTS+ for Diabetic and Its Complication
Abstract
:1. Introduction
2. Results
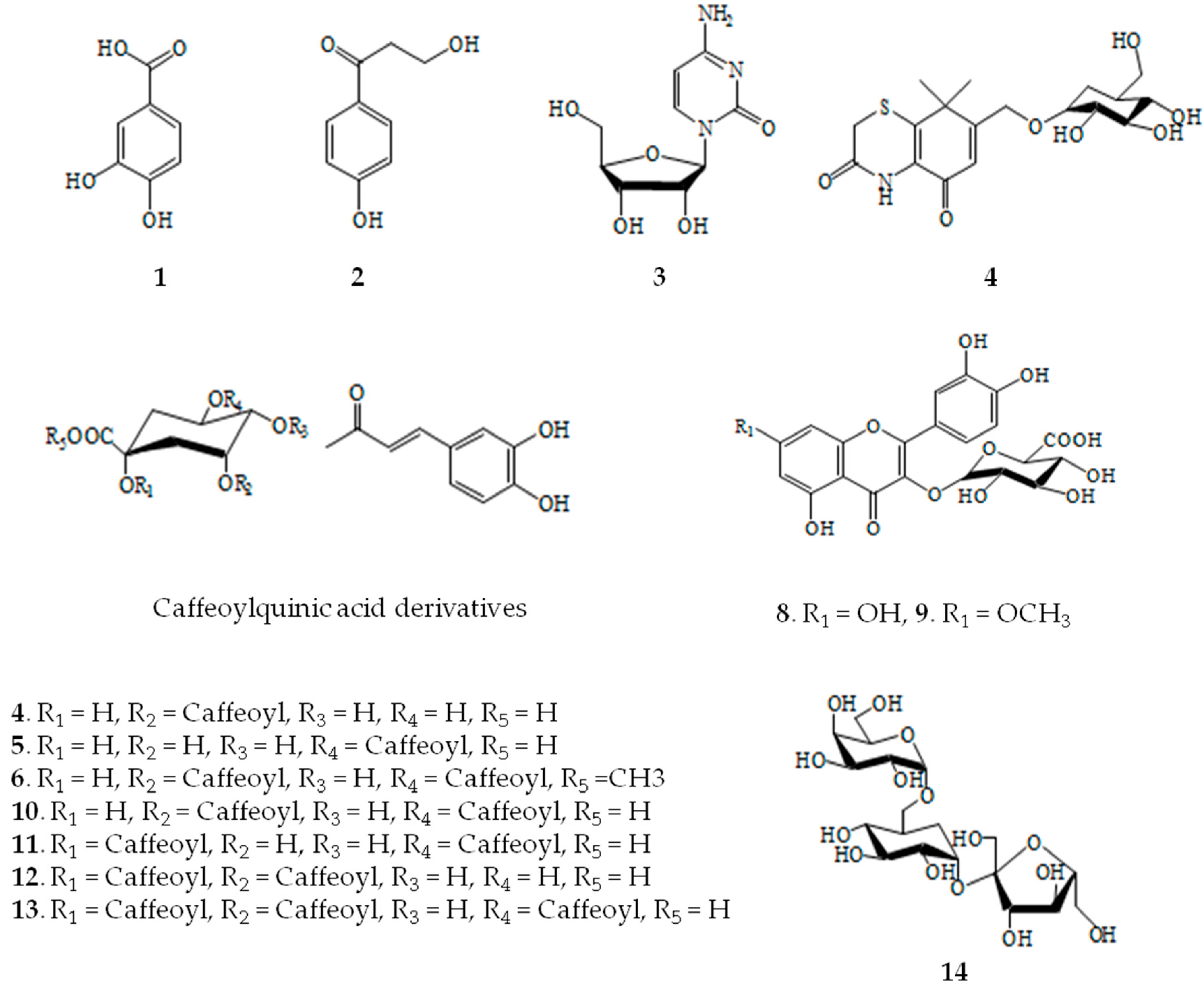
2.1. Structural Determination of Isolate Compounds
2.2. Inhibitory Effect of X. strumarium on α-Glucosidase
2.3. Inhibitory Effects of X. strumarium on PTP1β
2.4. Inhibitory Effects of X. strumarium on AGEs
2.5. Antioxidant Effect of X. strumarium on ABTS+
3. Discussion
4. Experimental Section
4.1. General Experimental Procedures
4.2. Plant Materials
4.3. Extraction and Isolation
4.4. Assay for the α-Glucosidase Inhibitory Activity
4.5. Assay for the PTP1β Inhibitory Activity
4.6. Assay for the AGEs Inhibitory Activity
4.7. Assay for the ABTS+ Inhibitory Activity
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Choi, S.J.; Kim, J.K.; Jang, J.M.; Shin, K.H.; Lim, S.S. Rapid identification of the α-glucosidase inhibitory compounds from Thunberg’s Geranium (Geranium thunbergii Sieb. et Zucc.). Food Sci. Biotechnol. 2012, 2, 987–996. [Google Scholar] [CrossRef]
- Iqbal-Choudhary, M.; Adhiken, A.; Rasheed, S.; Marasini, B.P.; Hussain, N.; Kaleem, W.A. Cyclopeptide alkaloids of Ziziphus oxyphylla Edgw as novel inhibitors of α-glucosidase enzyme and protein glycation. Phytochem. Lett. 2011, 4, 404–406. [Google Scholar] [CrossRef]
- Chougale, A.D.; Ghadyale, V.A.; Panaskar, S.N.; Akalpita, U.; Arvindekar, A.U. Alpha glucosidase inhibition by stem extracts of Tinispora cordifolia. J. Enzyme. Inhib. Med. Chem. 2009, 24, 998–1001. [Google Scholar]
- Schultz, L.D.; Schweitzer, P.A.; Rajan, T.V.; Yi, T.; Ihle, J.N.; Matthews, R.J.; Thomas, M.L.; Beier, D.R. Mutations at the murine motheaten locus are within the hematopoietic cell protein tyrosine Phosphatase (Hcph) gene. Cell 1993, 73, 1445–1454. [Google Scholar] [CrossRef]
- Bialy, L.; Waldmann, H. Inhibitors of protein tyrosine phosphatase: Next-generation drugs? Angew. Chem. Int. Ed. 2005, 44, 3814–3839. [Google Scholar] [CrossRef] [PubMed]
- Asante-Appiah, E.; Kennedy, B.P. Protein tyrosine phosphatase: The quest for negative regulators of insulin action. Am. J. Physiol. 2003, 84, 663–670. [Google Scholar] [CrossRef] [PubMed]
- Jariyapamornkoon, N.; Yibchok-anun, S.; Adisakwattana, S. Inhibition of advanced glycation end products by red grape skin extract and its antioxidant activity. BMC Complement. Altern. Med. 2013, 13, 171. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, P.C.; Huang, G.J.; Ho, Y.L.; Lin, Y.H.; Hung, S.S.; Chiang, Y.C. Activities of antioxidants, α-glucosidase inhibitors and aldose reductase inhibitors of the aqueous extracts of four species in Taiwan. Bot. Stud. 2010, 51, 293–302. [Google Scholar]
- Lee, Y.S.; Kang, Y.H.; Jung, J.Y.; Lee, S.H.; Ohuchi, K.; Shin, K.H. Protein glycation inhibitors from the fruiting body of Phellinus linteus. Biol. Pharm. Bull. 2008, 31, 1968–1972. [Google Scholar] [CrossRef] [PubMed]
- Hsu, F.L.; Chen, Y.C.; Cheng, J.T. Caffeic acid as active principle from the fruit Xanthium strumarium to lower plasma glucose in diabetic rats. Planta Med. 2000, 66, 228–230. [Google Scholar] [CrossRef] [PubMed]
- Han, T.; Li, H.L.; Zhang, Q.Y.; Han, P.; Zheng, H.C.; Rahman, K. Bioactivity-guided fractionation for anti-inflammatory and analgesic properties and constituents of Xanthium strumarium L. Phytomedicine 2007, 14, 825–829. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.T.; Huang, F.L.; Hsu, C.H.F. Thiazinedione from Xanthium strumarium. Phytochemisty 1998, 48, 1083–1085. [Google Scholar] [CrossRef]
- Yoon, H.N.; Lee, M.Y.; Kim, J.K.; Suh, H.W.; Lim, S.S. Aldose reductase inhibitory compounds from Xanthium strumarium. Arch. Pharm. Res. 2013, 36, 1090–1095. [Google Scholar] [CrossRef] [PubMed]
- Ezazul Haque, M.E.; Rahman, S.; Rahmatullah, M.; Jahan, R. Evaluation of antihyperglycemic and antinociceptive activity of Xanthium indicum stem extract in swill albino mice. BMC Complment. Altern. Med. 2013, 13, 296. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Mangelinckx, S.; Ma, L.; Wang, Z.; Li, W.; Kimp, N.D. Caffeoylquinic acid derivatives isolated from the aerial parts of Gynura divaricata and their yeast glucosidase and PTP1β inhibitory activity. Fitoterapia 2014, 99, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Hwang, S.H.; Kwon, S.H.; Wang, Z.Q.; Kim, T.H.; Kang, Y.H.; Lee, J.Y.; Lim, S.S. Optimization of extraction parameters of PTP1 (protein tyrosinase phosphatase 1), inhibitory polyphenols, and anthocyanins from Zea mays L. using response surfact methodology (RSM). BMC Complment. Altern. Med. 2016, 16, 317. [Google Scholar]
- Wan, C.; Yuan, T.; Cirello, A.L.; Seeram, N.P. Antioxidant and α-glucosidase inhibitory phenolics isolated from high bush blueberry flowers. Food. Chem. 2012, 135, 1929–1937. [Google Scholar] [CrossRef] [PubMed]
- Jang, D.S.; Yoo, N.H.; Kim, N.H.; Lee, Y.M.; Kim, C.S.; Kim, J.H.; Kim, J.H.; Kim, J.S. 3,5-Di-O-caffeoyl-epi-quinic acid from the leaves and stems of Erigeron annuus inhibits protein glycation, aldose reductase, and cataractogenesis. Biol. Pharm. Bull. 2010, 33, 329–333. [Google Scholar] [CrossRef] [PubMed]
- Matsui, T.; Ebuchi, S.; Fujise, T.; Abesundara, K.J.M.; Doi, S.; Yamada, H. Strong anti-hyperglycemic effects of water-soluble fraction of Brazilian propolis and its bioactive constituent, 3,4,5-tri-O-caffeoylquinic acid. Biol. Pharm. Bull. 2004, 27, 1797–1803. [Google Scholar] [CrossRef] [PubMed]
- Jung, H.A.; Islam, M.D.N.; Kwon, Y.S.; Jin, S.E.; Son, Y.K.; Park, J.J. Extraction and identification of three major aldose reductase inhibitors from Artemisia montana. Food Chem. Toxicol. 2011, 49, 376–384. [Google Scholar] [CrossRef] [PubMed]
- Chuda, Y.; Ono, H.; Ohnishi-kameyama, M.; Nagata, T.; Tsushida, T. Structural identification of two antioxidant quinic acid derivatives from garland (Chrysanthemum coronarium L.). J. Agric. Food Chem. 1996, 44, 2037–2039. [Google Scholar] [CrossRef]
- Santos, M.D.D.; Gobbo-Neto, L.; Albarella, L.; Souza, G.E.P.; Lopes, N.P. Anaesic activity of di-caffeoylquinic acids from roots of Lychnophora ericoides (Arnica da serra). J. Ethnopharmacol. 2005, 96, 545–549. [Google Scholar] [CrossRef] [PubMed]
- Hu, T.; He, X.W.; Jiang, J.G. Functional analyses on antioxidant, anti-inflammatory, and antiproliferative effects of extracts and compounds from Ilex latifolia Thund., a Chinese bitter tea. J. Agric. Food Chem. 2014, 62, 8608–8615. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.Q.; Hwang, S.H.; Lee, S.Y.; Lim, S.S. Fermentation of purple Jerusalem artichoke extract to improve the α-glucosidase inhibitory effect in vitro and ameliorate blood glucose in db/db mice. Nutr. Res. Pract. 2016, 10, 10–12. [Google Scholar] [CrossRef] [PubMed]
- Huijsduijnen, R.H.V.; Bombrun, A.; Swinnen, D. Selecting protein tyrosine phosphatases as drug targets. Drug Discov. Today 2002, 19, 1013–1019. [Google Scholar] [CrossRef]
- Saltiel, A.R.; Kahn, C.R. Insulin signaling and the regulation of glucose and lipid metabolism. Nature 2001, 414, 799–806. [Google Scholar] [CrossRef] [PubMed]
- Li, H.M.; Hwang, S.H.; Kang, B.G.; Hong, J.S.; Lim, S.S. Inhibitory effects of Colocasia esculenta (L.) schott constituents on aldose reductase. Molecules 2014, 19, 13213–13224. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Samples of the compounds are not available from the authors.

| Peaks | Rt (min) | UV/Vis λMax (nm) | LC-ESI-MS | Compounds | |
|---|---|---|---|---|---|
| Mass [M]+ (m/z) | Fragment [M + H]+ (m/z) | ||||
| 1 | 7.20 | 294, 258, 227 | 154.12 | 137.58 | Protocatechuic acid |
| 2 | 10.28 | 290, 254, 229 | 166.17 | 108.14 | 3-Hydroxy-1-(4-hydroxy phenyl)propan-1-one |
| 3 | 10.94 | 266, 219 | 243.22 | 111.10 | Cytidine |
| 4 | 12.18 | 328, 243 | 368.34 | 180.16 | Neochlorogenic acid methyl ester |
| 5 | 14.86 | 327, 242, 224 | 353.99 | 209.22 | Chlorogenic acid |
| 6 | 17.55 | 330, 244 | 530.48 | 368.34 | Methyl-3,5-di-O-caffeoylquinic acid |
| 7 | 17.90 | 346, 242 | 401.67 | 239.29 | Thiazine-3,5-dione-11-O-glucopyranoside |
| 8 | 19.34 | 352, 257, 223 | 508.71 | 332.26 | Patuletin-3-glucuronide |
| 9 | 20.13 | 354, 256, 202 | 478.36 | 302.24 | Quercetin-3-O-glucuronide |
| 10 | 20.59 | 328, 243, 223 | 516.17 | 354.20 | 3,5-Di-O-caffeoylquinic acid |
| 11 | 21.51 | 330, 244, 223 | 516.17 | 354.20 | 1,5-Di-O-caffeoylquinic acid |
| 12 | 24.18 | 328, 245, 223 | 516.17 | 354.20 | 1,3-Di-O-caffeoylquinic acid |
| 13 | 24.78 | 327, 244, 224 | 678.27 | 516.45 | 1,3,5-Tri-O-caffeoylquinic acid |
| Entry | Acarbose 1 | Suramin 2 | Aminoguanidine 3 | Trolox 4 | MeOH | CH2Cl2 | EtOAc | n-BuOH | Water |
|---|---|---|---|---|---|---|---|---|---|
| IC50 (μg/mL) 5 | |||||||||
| Positive Control | Extract | Fraction | |||||||
| α-Glucosidase | 377.19 ± 38.17 | - | - | - | >500 | - | 399.66 ± 37.51 | - | - |
| PTP1β | - | 8.96 + 0.83 | - | - | 12.88 ± 1.18 | 20.81 ± 2.09 | 9.80 ± 0.89 | 15.08 ± 1.46 | 28.44 ± 2.79 |
| AGEs | - | - | 166.22 ± 16.29 | - | >200 | >200 | 132.95 ± 12.34 | 168.75 ± 17.87 | - |
| ABTS+ | - | - | - | 7.51 ± 0.58 | 78.32 ± 0.71 | 8.52 ± 0.86 | 9.34 ± 0.92 | 84.47 ± 8.51 | 4.68 ± 4.61 |
| Entry | Products | Concentration (μg/mL) | Inhibition (%) | IC50 1 (μg/mL) |
|---|---|---|---|---|
| Acarbose 2 | 1000 | 106.14 | 377.19 ± 37.8 (584.79 μM) | |
| 500 | 60.29 | |||
| 250 | 41.06 | |||
| 50 | 16.98 | |||
| 1 | Protocatechuic acid | 50 | 17.45 | - |
| 2 | 3-Hydroxy-1-(4-hydroxy phenyl) propan-1-one | 50 | 8.60 | - |
| 3 | Cytidine | 50 | 4.70 | - |
| 4 | Neochlorogenic acid methyl ester | 50 | - | - |
| 5 | Chlorogenic acid | 50 | 6.60 | - |
| 6 | Methyl-3,5-di-O-caffeoylquinic acid | 25 | 96.50 | 9.78 ± 0.79 (18.42 μM) |
| 12.5 | 59.11 | |||
| 5 | 40.00 | |||
| 2.5 | 22.63 | |||
| 7 | Thiazine-3,5-dione-11-O-glucopyranoside | 50 | 10.18 | - |
| 8 | Patuletin-3-glucuronide | 50 | 6.11 | - |
| 9 | Quercetin-3-O-glucuronide | 50 | 15.40 | - |
| 10 | 3,5-di-O-caffeoylquinic acid | 100 | 99.44 | 45.48 ± 4.52 (88.14 μM) |
| 50 | 52.85 | |||
| 25 | 33.72 | |||
| 5 | 9.34 | |||
| 11 | 1,5-di-O-caffeoylquinic acid | 50 | 46.20 | - |
| 12 | 1,3-di-O-caffeoylquinic acid | 50 | 38.37 | - |
| 13 | 1,3,5-tri-O-caffeoylquinic acid | 50 | 22.05 | - |
| 14 | Raffinose | 50 | 5.82 | - |
| Entry | Products | Concentration (μg/mL) | Inhibition (%) | IC50 1 (μg/mL) |
|---|---|---|---|---|
| Sumarin 2 | 14.29 | 83.58 | 7.84 ± 0.75 (5.51 μM) | |
| 7.15 | 52.24 | |||
| 3.57 | 26.44 | |||
| 1.43 | 10.45 | |||
| 1 | Protocatechuic acid | 10 | 28.58 | - |
| 2 | 3-Hydroxy-1-(4-hydroxy phenyl) propan-1-one | 10 | 5.88 | - |
| 3 | Cytidine | 10 | - | - |
| 4 | Neochlorogenic acid methyl ester | 10 | 18.79 | - |
| 5 | Chlorogenic acid | 10 | 31.34 | - |
| 6 | Methyl-3,5-di-O-caffeoylquinic acid | 10 | 82.47 | 5.09 ± 0.50 (1.88 μM) |
| 5 | 50.01 | |||
| 2.5 | 30.66 | |||
| 1 | 24.00 | |||
| 7 | Thiazine-3,5-dione-11-O-glucopyranoside | 10 | - | - |
| 8 | Patuletin-3-glucuronide | 10 | - | - |
| 9 | Quercetin-3-O-glucuronide | 10 | 10.71 | - |
| 10 | 3,5-di-O-caffeoylquinic acid | 10 | 18.40 | - |
| 11 | 1,5-di-O-caffeoylquinic acid | 10 | 10.62 | - |
| 12 | 1,3-di-O-caffeoylquinic acid | 10 | 22.34 | - |
| 13 | 1,3,5-tri-O-caffeoylquinic acid | 10 | 39.17 | - |
| 14 | Raffinose | 10 | - | - |
| Entry | Products | Concentration (μg/mL) | Inhibition (%) | IC50 1 (μg/mL) |
|---|---|---|---|---|
| Aminoguanidine 2 | 400 | 98.17 | 155.88 ± 15.45 (1410.00 μM) | |
| 200 | 61.01 | |||
| 100 | 37.84 | |||
| 50 | 27.97 | |||
| 1 | Protocatechuic acid | 100 | 9.49 | - |
| 2 | 3-Hydroxy-1-(4-hydroxy phenyl) propan-1-one | 100 | 8.50 | - |
| 3 | Cytidine | 100 | 33.16 | - |
| 4 | Neochlorogenic acid methyl ester | 100 | 11.40 | - |
| 5 | Chlorogenic acid | 100 | 7.47 | - |
| 6 | Methyl-3,5-di-O-caffeoylquinic acid | 100 | 89.47 | 43.96 ± 3.89 (82.79 μM) |
| 50 | 54.77 | |||
| 20 | 34.59 | |||
| 10 | 24.24 | |||
| 7 | Thiazine-3,5-dione-11-O-glucopyranoside | 100 | 22.24 | - |
| 8 | Patuletin-3-glucuronide | 100 | 6.11 | - |
| 9 | Quercetin-3-O-glucuronide | 100 | 23.96 | - |
| 10 | 3,5-Di-O-caffeoylquinic acid | 100 | 42.91 | - |
| 11 | 1,5-Di-O-caffeoylquinic acid | 100 | 25.04 | - |
| 12 | 1,3-Di-O-caffeoylquinic acid | 100 | 40.19 | - |
| 13 | 1,3,5-Tri-O-caffeoylquinic acid | 100 | 40.22 | - |
| 14 | Raffinose | 100 | 14.99 | - |
| Entry | Products | Concentration (μg/mL) | Inhibition (%) | IC50 1 (μg/mL) |
|---|---|---|---|---|
| Trolox 2 | 16.66 | 95.06 | 7.43 ± 0.71 (29.72 μM) | |
| 8.33 | 54.14 | |||
| 3.33 | 34.52 | |||
| 1.66 | 17.27 | |||
| 1 | Protocatechuic acid | 16.66 | 97.22 | 4.94 ± 0.38 (32.93 μM) |
| 8.33 | 68.72 | |||
| 3.33 | 52.46 | |||
| 1.66 | 25.17 | |||
| 2 | 3-Hydroxy-1-(4-hydroxy phenyl) propan-1-one | 3.33 | - | - |
| 3 | Cytidine | 3.33 | - | - |
| 4 | Neochlorogenic acid methyl ester | 3.33 | 8.66 | - |
| 5 | Chlorogenic acid | 3.33 | 23.59 | - |
| 6 | Methyl-3,5-di-O-caffeoylquinic acid | 8.33 | 98.31 | 3.20 ± 0.28 (6.03 μM) |
| 3.33 | 69.25 | |||
| 1.66 | 33.69 | |||
| 0.33 | 7.49 | |||
| 7 | Thiazine-3,5-dione-11-O-glucopyranoside | 3.33 | - | - |
| 8 | Patuletin-3-glucuronide | 3.33 | 30.53 | - |
| 9 | Quercetin-3-O-glucuronide | 3.33 | 12.32 | - |
| 10 | 3,5-Di-O-caffeoylquinic acid | 3.33 | 40.74 | - |
| 11 | 1,5-Di-O-caffeoylquinic acid | 3.33 | 30.48 | - |
| 12 | 1,3-Di-O-caffeoylquinic acid | 3.33 | 22.12 | - |
| 13 | 1,3,5-Tri-O-caffeoylquinic acid | 3.33 | 38.06 | - |
| 14 | Raffinose | 3.33 | - | - |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hwang, S.H.; Wang, Z.; Yoon, H.N.; Lim, S.S. Xanthium strumarium as an Inhibitor of α-Glucosidase, Protein Tyrosine Phosphatase 1β, Protein Glycation and ABTS+ for Diabetic and Its Complication. Molecules 2016, 21, 1241. https://doi.org/10.3390/molecules21091241
Hwang SH, Wang Z, Yoon HN, Lim SS. Xanthium strumarium as an Inhibitor of α-Glucosidase, Protein Tyrosine Phosphatase 1β, Protein Glycation and ABTS+ for Diabetic and Its Complication. Molecules. 2016; 21(9):1241. https://doi.org/10.3390/molecules21091241
Chicago/Turabian StyleHwang, Seung Hwan, Zhiqiang Wang, Ha Na Yoon, and Soon Sung Lim. 2016. "Xanthium strumarium as an Inhibitor of α-Glucosidase, Protein Tyrosine Phosphatase 1β, Protein Glycation and ABTS+ for Diabetic and Its Complication" Molecules 21, no. 9: 1241. https://doi.org/10.3390/molecules21091241
APA StyleHwang, S. H., Wang, Z., Yoon, H. N., & Lim, S. S. (2016). Xanthium strumarium as an Inhibitor of α-Glucosidase, Protein Tyrosine Phosphatase 1β, Protein Glycation and ABTS+ for Diabetic and Its Complication. Molecules, 21(9), 1241. https://doi.org/10.3390/molecules21091241