Dimacrolide Sesquiterpene Pyridine Alkaloids from the Stems of Tripterygium regelii
Abstract
:1. Introduction
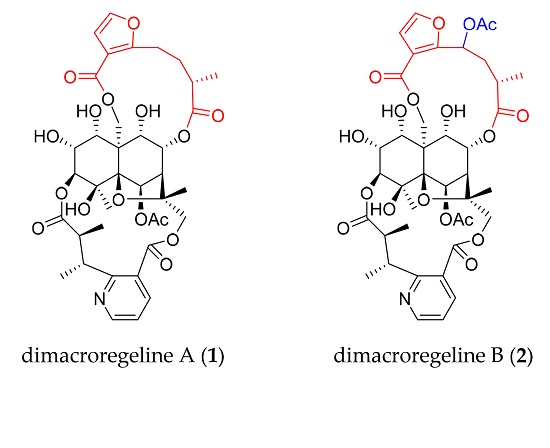
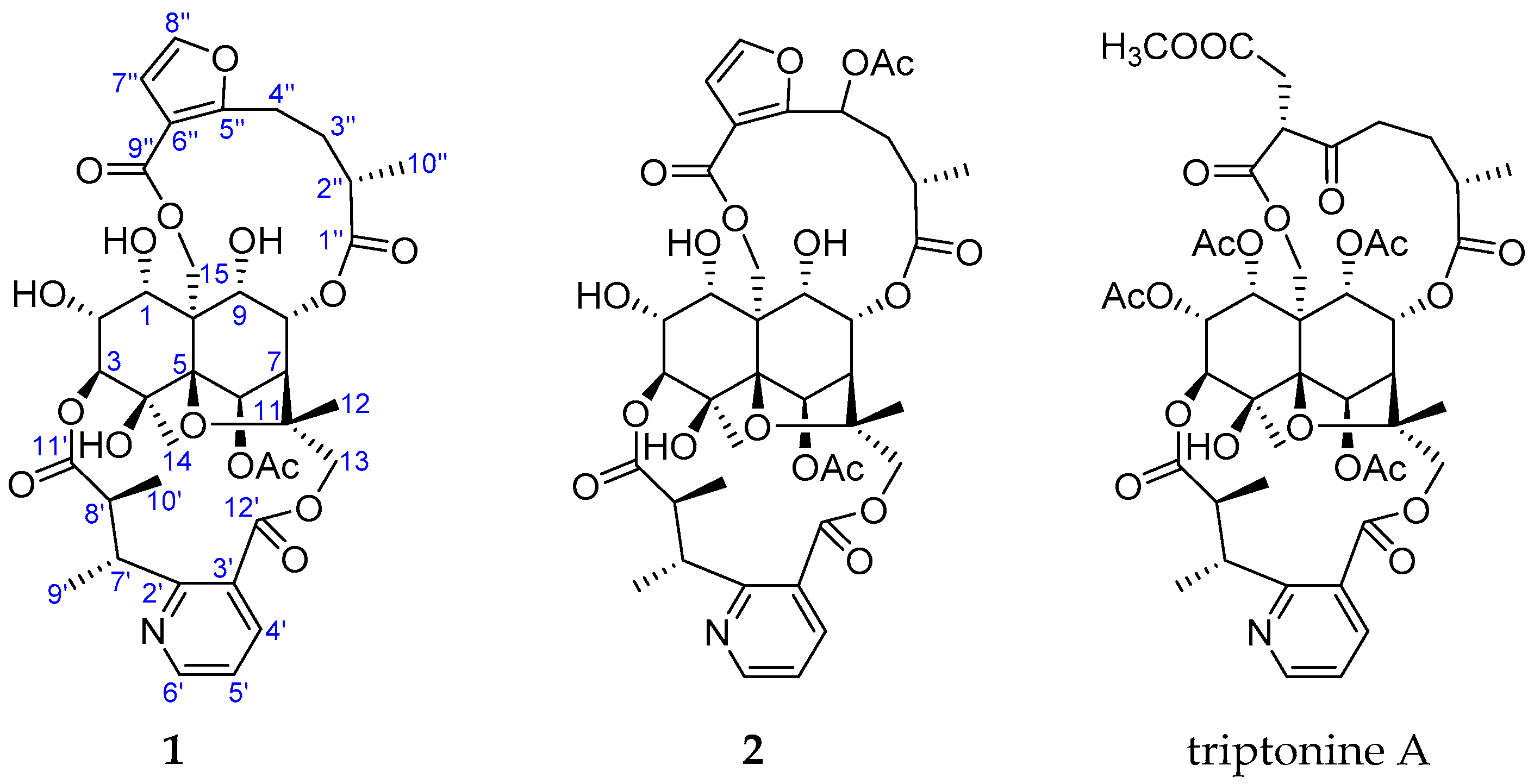
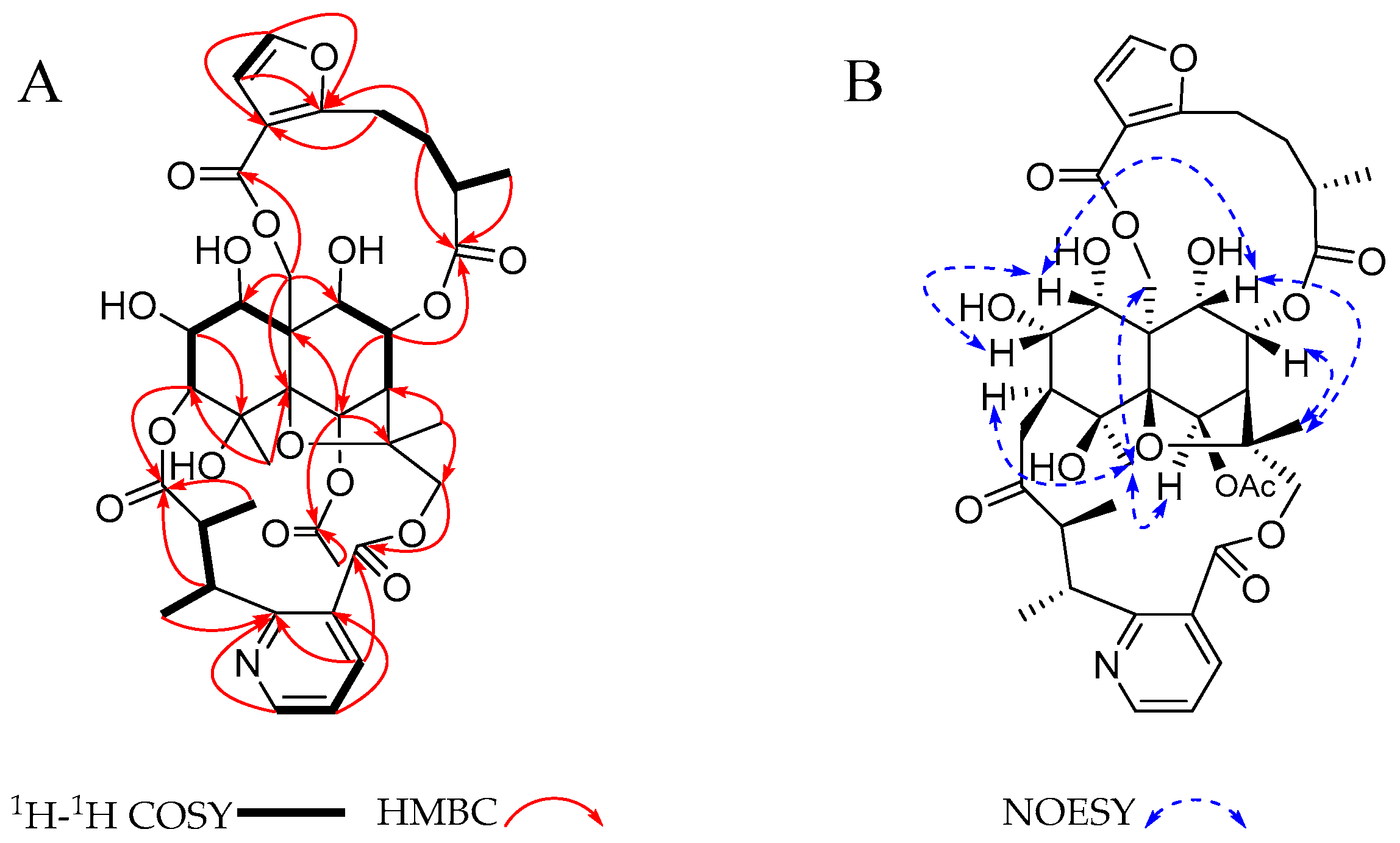
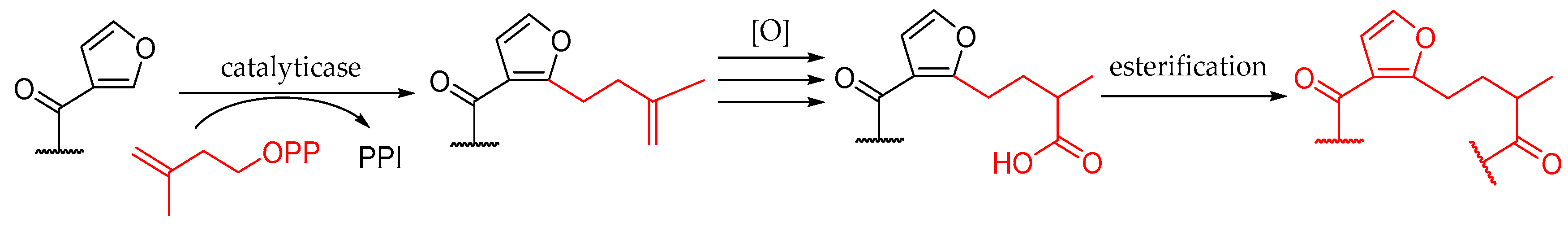
2. Results and Discussion
3. Materials and Methods
3.1. General Procedures
3.2. Plant Material
3.3. Extraction and Isolation
3.4. Inhibition of Proliferation on MH7A Human Synovial Cells
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Editorial Committee of Flora of China. Flora of China (English Revised Version); Science Press and Missouri Botanical Garden Press: Beijing, China; St. Louis, MO, USA, 2013; Volume 11, p. 439. [Google Scholar]
- González, A.G.; Bazzocchi, I.L.; Moujir, L.; Jiménez, I.A. Ethnobotanical uses of celastraceae. Bioactive metabolites. Stud. Nat. Prod. Chem. 2000, 23, 649–738. [Google Scholar]
- Spivey, A.C.; Weston, M.; Woodhead, S. Celastraceae sesquiterpenoids: Biological activity and synthesis. Chem. Soc. Rev. 2002, 31, 43–59. [Google Scholar] [CrossRef] [PubMed]
- Gao, J.M.; Wu, W.J.; Zhang, J.W.; Konishi, Y.S. The dihydro-β-agarofuran sesquiterpenoids. Nat. Prod. Rep. 2007, 24, 1153–1189. [Google Scholar] [CrossRef] [PubMed]
- Baxter, R.L.; Crombie, L.; Simmonds, D.J.; Whiting, D.A. Extractives of Catha edulis (Khat): Occurrence of celastraceaeous alkaloids having mono-and bis-macrolide bridges. J. Chem. Soc. Chem. Commun. 1976, 463–465. [Google Scholar] [CrossRef]
- Baxter, R.L.; Crombie, L.; Simmonds, D.J.; Whiting, D.A.; Braenden, O.J.; Szendrei, K. Alkaloids of catha edulis (khat). Part 1. Isolation and characterisation of eleven new alkaloids with sesquiterpene cores (cathedulins); identification of the quinone–methide root pigments. J. Chem. Soc. Perkin Trans. 1 1979, 2965–2971. [Google Scholar] [CrossRef]
- Crombie, L.; Crombie, W.M.L.; Whiting, D.A.; Branenden, O.J.; Szendrei, K. Structures of cathedulin alkaloids from Catha edulis (khat) of Kenyan and Ethiopian origin. J. Chem. Soc. Chem. Commun. 1978, 107–108. [Google Scholar] [CrossRef]
- Baxter, R.L.; Crombie, W.M.L.; Crombie, L.; Simmonds, D.J.; Whiting, D.A.; Szendrei, K. Alkaloids of Catha edulis. Part 4. Structures of cathedulins E3, E4, E5, E6, and K12. Novel sesquiterpene alkaloids with mono-and bismacrolide bridges. J. Chem. Soc. Perkin Trans. 1 1979, 2982–2989. [Google Scholar] [CrossRef]
- Crombie, L.; Toplis, D.; Whiting, D.A.; Rozsá, Z.; Hohmann, J.; Szendrei, K. New macrolide sesquiterpene alkaloids of Catha edulis: Examples containing a novel dilactone bridge. J. Chem. Soc. Perkin Trans. 1 1986, 531–534. [Google Scholar] [CrossRef]
- Duan, H.; Takaishi, Y.; Bando, M.; Kido, M.; Imakura, Y.; Lee, K. Novel sesquiterpene esters with alkaloid and monoterpene and related compounds from Tripterygium hypoglaucum: A new class of potent anti-HIV agents. Tetrahedron Lett. 1999, 40, 2969–2972. [Google Scholar] [CrossRef]
- Duan, H.; Takaishi, Y.; Imakura, Y.; Jia, Y.; Li, D.; Cosentino, L.M.; Lee, K.H. Sesquiterpene Alkaloids from Tripterygium hypoglaucum and Tripterygium wilfordii: A new class of potent anti-HIV agents. J. Nat. Prod. 2000, 63, 357–361. [Google Scholar] [CrossRef] [PubMed]
- Luo, Y.; Pu, X.; Luo, G.; Zhou, M.; Ye, Q.; Liu, Y.; Gu, J.; Qi, H.; Li, G.; Zhang, G. Nitrogen-containing dihydro-β-agarofuran derivatives from Tripterygium wilfordii. J. Nat. Prod. 2014, 77, 1650–1657. [Google Scholar] [CrossRef] [PubMed]
- Flora of China Editorial Committee of Chinese Academy of Sciences. Flora of China; Beijing Science and Technology Press: Beijing, China, 1999; Volume 45, p. 181. [Google Scholar]
- Editorial Board of Zhonghua Bencao. Zhonghua Bencao; Shanghai Science and Technology Press: Shanghai, China, 1999; Volume 5, pp. 205–206. [Google Scholar]
- Shen, J.H.; Zhou, B.N. Studies on diterpene-quinones of Tripterygium regelii Sprague. Chin. Chem. Lett. 1992, 3, 113–116. [Google Scholar]
- Hori, H.; Pang, G.M.; Harimaya, K.; Iitaka, Y.; Inayama, S. Isolation and structure of regelin and regelinol, new antitumor ursene-type triterpenoids from Tripterygium regelii. Chem. Pharm. Bull. 1987, 35, 2125–2128. [Google Scholar] [CrossRef] [PubMed]
- Harada, R.; Kakisawa, H.; Kobayashi, S.; Musya, M.; Nakanishi, K.; Takahashi, Y. Structure of pristimerin, a quinonoid triterpene. Tetrahedron Lett. 1962, 3, 603–607. [Google Scholar] [CrossRef]
- Pang, G.M.; Zhao, C.J.; Hori, H.; Inayama, S. Studies on new triterpenoids of Tripterygium regelii. Acta Pharm. Sin. 1989, 24, 75–79. [Google Scholar]
- Shen, J.H.; Zhou, B.N. Study on the triterpenoids of Tripterygium regelii. Acta Bot. Sin. 1992, 34, 475–475. [Google Scholar]
- Lee, B.W.; Seo, W.D.; Gal, S.W.; Yang, M.S.; Park, K.H. Quinone methide triterpenes from Tripterygium regelii. Agric. Chem. Biotechnol. 2004, 47, 77–80. [Google Scholar]
- Hori, H.; Pang, G.M.; Harimaya, K.; Iitaka, Y.; Inayama, S. The absolute configuration of regelidine, a novel 6-nicotinoyl dihydroagarofuran sesquiterpene alkaloid from Tripterygium regelii. Chem. Pharm. Bull. 1987, 35, 4683–4686. [Google Scholar] [CrossRef]
- Han, B.H.; Yang, H.O.; Ryu, J.H. Tripterregeline A, B and C, sesquiterpene alkaloids from Tripterygium regelii. Arch. Pharm. Res. 1989, 12, 310–312. [Google Scholar] [CrossRef]
- Fan, D.; Zhu, G.Y.; Chen, M.; Xie, L.M.; Jiang, Z.H.; Xu, L.; Bai, L.P. Dihydro-β-agarofuran sesquiterpene polyesters isolated from the stems of Tripterygium regelii. Fitoterapia 2016, 112, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Fan, D.; Parhira, S.; Zhu, G.Y.; Jiang, Z.H.; Bai, L.-P. Triterpenoids from the stems of Tripterygium regelii. Fitoterapia 2016, 113, 69–73. [Google Scholar] [CrossRef] [PubMed]
- Schaneberg, B.T.; Green, D.K.; Sneden, A.T. Dihydroagarofuran sesquiterpene alkaloids from Maytenus putterlickoides. J. Nat. Prod. 2001, 64, 624–626. [Google Scholar] [CrossRef] [PubMed]
- Duan, H.Q.; Takaishi, Y.; Momota, H.; Ohmoto, Y.; Taki, T.; Jia, Y.F.; Li, D. Immunosuppressive sesquiterpene alkaloids from Tripterygium wilfordii. J. Nat. Prod. 2001, 64, 582–587. [Google Scholar] [CrossRef] [PubMed]
- Lhinhatrakool, T.; Prabpai, S.; Kongsaeree, P.; Sutthivaiyakit, S. Antiplasmodial sesquiterpene alkaloids from the roots of Maytenus mekongensis. J. Nat. Prod. 2011, 74, 1386–1391. [Google Scholar] [CrossRef] [PubMed]
- Santos, V.A.F.F.M.; Regasini, L.O.; Nogueira, C.R.; Passerini, G.D.; Martinez, I.; Bolzani, V.S.; Graminha, M.A.S.; Cicarelli, R.M.B.; Furlan, M. Antiprotozoal sesquiterpene pyridine alkaloids from Maytenus ilicifolia. J. Nat. Prod. 2012, 75, 991–995. [Google Scholar] [CrossRef] [PubMed]
- Duan, H.Q.; Kawazoe, K.; Takaishi, Y. Sesquiterpene alkaloids from Tripterygium hypoglaucum. Phytochemistry 1997, 45, 617–621. [Google Scholar] [CrossRef]
- Brüning, R.; Wagner, H. Übersicht über die celastraceen-inhaltsstoffe: Chemie, chemotaxonomie, biosynthese, pharmakologie. Phytochemistry 1978, 17, 1821–1858. [Google Scholar] [CrossRef]
- Lee, H.J.; Waller, G.R. Biosynthesis of pyridine alkaloids from Tripterygium wilfordii. Phytochemistry 1972, 11, 2233–2240. [Google Scholar] [CrossRef]
- Wang, R.; Chen, R.; Li, J.; Liu, X.; Xie, K.; Chen, D.; Peng, Y.; Dai, J. Regiospecific Prenylation of Hydroxyxanthones by a Plant Flavonoid Prenyltransferase. J. Nat. Prod. 2016. [Google Scholar] [CrossRef] [PubMed]
- Bartok, B.; Firestein, G.S. Fibroblast-like synoviocytes: Key effector cells in rheumatoid arthritis. Immunol. Rev. 2010, 233, 233–255. [Google Scholar] [CrossRef] [PubMed]
- Pap, T.; Müller-Ladner, U.; Gay, R.E.; Gay, S. Fibroblast biology: Role of synovial fibroblasts in the pathogenesis of rheumatoid arthritis. Arthritis Res. Ther. 2000, 2, 1. [Google Scholar]
- Noss, E.H.; Brenner, M.B. The role and therapeutic implications of fibroblast-like synoviocytes in inflammation and cartilage erosion in rheumatoid arthritis. Immunol. Rev. 2008, 223, 252–270. [Google Scholar] [CrossRef] [PubMed]
- Nakayama, H.; Yaguchi, T.; Yoshiya, S.; Nishizaki, T. Resveratrol induces apoptosis MH7A human rheumatoid arthritis synovial cells in a sirtuin 1-dependent manner. Rheumatol. Int. 2012, 32, 151–157. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Jia, X.; Sui, C.; Zhao, Y.; Mei, Y.; Zheng, Y.; Zhang, Z. Effects of thapsigargin on the proliferation and survival of human rheumatoid arthritis synovial cells. Sci. World J. 2014, 2014, 605416. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Samples of the compounds 1 and 2 are available from the authors.
| Position | 1 | 2 | ||
|---|---|---|---|---|
| δH (J in Hz) | δC, Type | δH (J in Hz) | δC, Type | |
| 1 | 4.19, d (3.6) | 75.4, CH | 4.24, d (3.6) | 74.9, CH |
| 2 | 3.84, dd (3.6, 3.0) | 73.0, CH | 3.83, dd (3.6, 2.4) | 73.0, CH |
| 3 | 4.75, d, (3.0) a | 80.4, CH | 4.79, d (2.4) | 80.2, CH |
| 4 | 72.2, C | 72.3, C | ||
| 5 | 95.3, C | 94.9, C | ||
| 6 | 6.61, s | 76.6, CH | 6.20, s | 78.5, CH |
| 7 | 2.62, d (3.6) | 51.1, CH | 2.94, d (4.2) | 50.0, CH |
| 8 | 5.50, dd (6.0, 3.6) | 72.9, CH | 5.44, dd (6.0, 4.2) | 73.4, CH |
| 9 | 4.42, d (6.0) | 73.6, CH | 4.43, d (6.0) | 73.7, CH |
| 10 | 54.8, C | 55.2, C | ||
| 11 | 85.1, C | 84.7, C | ||
| 12 | 1.61, s | 18.4, CH3 | 1.59, s | 18.2, CH3 |
| 13 a | 5.96, d (11.4) | 71.6, CH2 | 5.78, d (11.4) | 71.4, CH2 |
| 13 b | 3.84, d (11.4) a | 3.81, d (11.4) | ||
| 14 | 1.58, s | 24.1, CH3 | 1.77, d (0.6) | 24.6, CH3 |
| 15 a | 5.62, d (14.4) | 63.2, CH2 | 5.21, d (14.4) | 64.1, CH2 |
| 15 b | 4.76, d (14.4) a | 5.01, d (14.4) | ||
| 2′ | 165.7, C | 165.3, C | ||
| 3′ | 127.2, C | 127.4, C | ||
| 4′ | 8.16, dd (7.8, 1.8) | 139.2, CH | 8.13, dd (7.8, 1.8) | 139.0, CH |
| 5′ | 7.39, dd (7.8, 4.8) | 122.8, CH | 7.39, dd (7.8, 4.8) | 122.8, CH |
| 6′ | 8.66, dd (4.8, 1.8) | 152.4, CH | 8.66, dd (4.8, 1.8) | 152.4, CH |
| 7′ | 4.62, qd (6.6, 1.2) a | 37.6, CH | 4.57, qd (6.6, 1.2) a | 37.7, CH |
| 8′ | 2.44, br q (6.6) | 46.3, CH | 2.46, qd (6.6, 1.2) | 46.2, CH |
| 9′ | 1.36, d (6.6) | 12.0, CH3 | 1.36, d (6.6) | 12.2, CH3 |
| 10′ | 1.13, d (6.6) | 9.8, CH3 | 1.16, d (6.6) | 9.9, CH3 |
| 11′ | 175.9, C | 175.9, C | ||
| 12′ | 170.1, C | 170.0, C | ||
| 1″ | 177.1, C | 176.5, C | ||
| 2″ | 2.50, m | 38.0, CH | 2.16, m | 35.5, CH |
| 3″ a | 2.06, m | 33.2, CH2 | 2.36, td (12.6, 4.8) | 37.9, CH2 |
| 3″ b | 1.91, m | 2.25, td (12.6, 1.8) | ||
| 4″ a | 3.77, m | 26.8, CH2 | 6.89, dd (12.6, 4.8) | 68.0, CH |
| 4″ b | 2.96, ddd (14.4, 7.2, 4.2) | |||
| 5″ | 161.3, C | 154.3, C | ||
| 6″ | 116.0, C | 119.9, C | ||
| 7″ | 6.73, d (1.8) | 113.4, CH | 6.80, d (1.8) | 113.5, CH |
| 8″ | 7.42, d (1.8) | 142.3, CH | 7.58, d (1.8) | 144.1, CH |
| 9″ | 166.2, C | 165.6, C | ||
| 10″ | 1.14, d (7.2) | 17.4, CH3 | 1.10, d (6.6) | 17.7, CH3 |
| OH-4 | 4.64, s a | 4.57, s a | ||
| OAc-6 | 2.12, s | 21.5, CH3 | 2.09, s | 21.7, CH3 |
| 171.3, C | 171.9, C | |||
| OAc-4″ | 1.96, s | 20.9, CH3 | ||
| 171.6, C | ||||
| Compounds # | Cell Viability (%) (n = 3) | Inhibition Rate (%) |
|---|---|---|
| Vehicle control | 100 ± 5.9 | 0 |
| 1 | 94.6 ± 5.4 | 5.4 ± 5.4 |
| 2 | 86.7 ± 5.2 * | 13.3 ± 5.2 * |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fan, D.; Zhu, G.-Y.; Li, T.; Jiang, Z.-H.; Bai, L.-P. Dimacrolide Sesquiterpene Pyridine Alkaloids from the Stems of Tripterygium regelii. Molecules 2016, 21, 1146. https://doi.org/10.3390/molecules21091146
Fan D, Zhu G-Y, Li T, Jiang Z-H, Bai L-P. Dimacrolide Sesquiterpene Pyridine Alkaloids from the Stems of Tripterygium regelii. Molecules. 2016; 21(9):1146. https://doi.org/10.3390/molecules21091146
Chicago/Turabian StyleFan, Dongsheng, Guo-Yuan Zhu, Ting Li, Zhi-Hong Jiang, and Li-Ping Bai. 2016. "Dimacrolide Sesquiterpene Pyridine Alkaloids from the Stems of Tripterygium regelii" Molecules 21, no. 9: 1146. https://doi.org/10.3390/molecules21091146
APA StyleFan, D., Zhu, G.-Y., Li, T., Jiang, Z.-H., & Bai, L.-P. (2016). Dimacrolide Sesquiterpene Pyridine Alkaloids from the Stems of Tripterygium regelii. Molecules, 21(9), 1146. https://doi.org/10.3390/molecules21091146