Aberrant Expression of Novel Cytokine IL-38 and Regulatory T Lymphocytes in Childhood Asthma
Abstract
:1. Introduction
2. Results and Discussion
2.1. Asthmatic Patients and Control Subjects
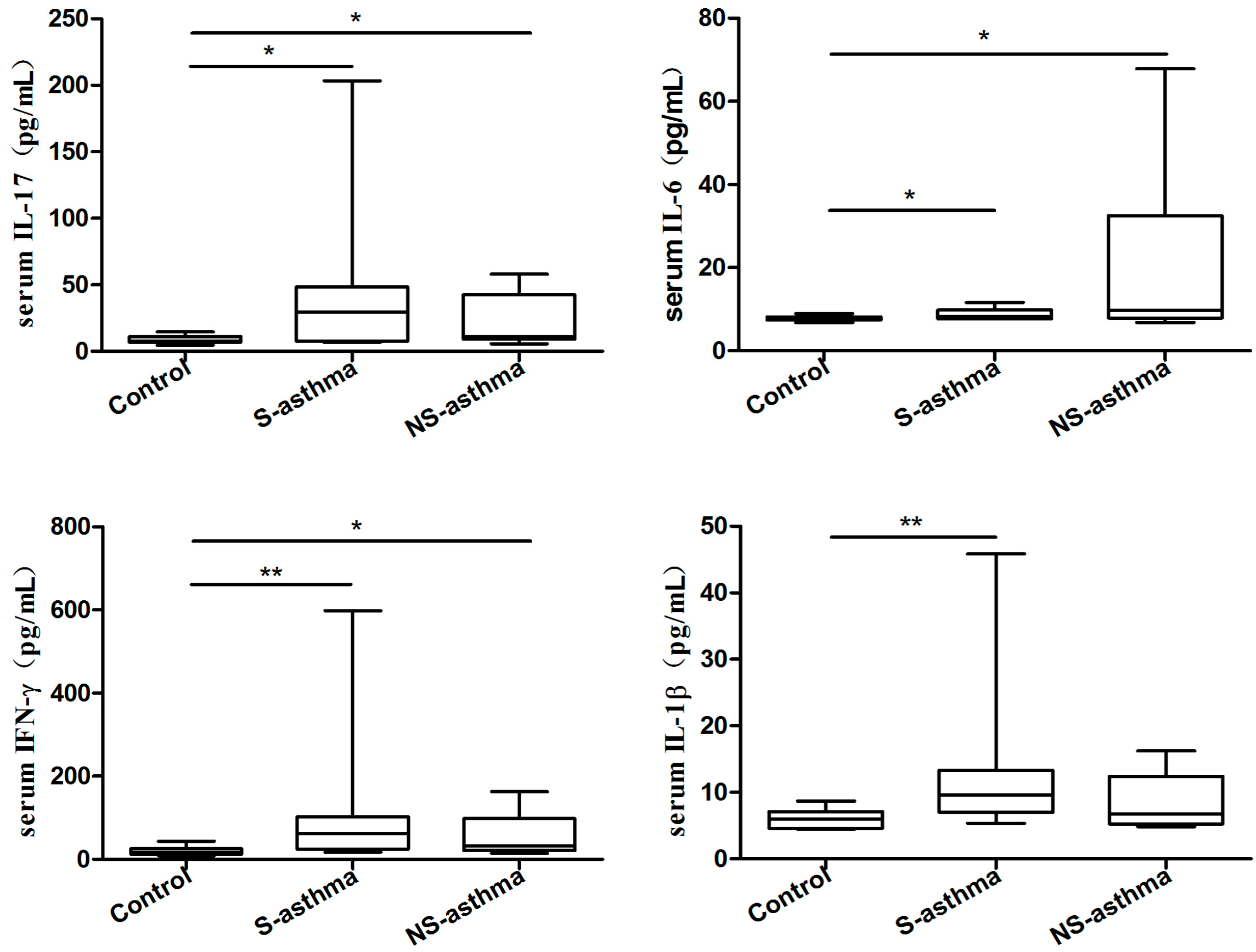
2.2. Serum Levels of IL-38, Periostin and Several Common Cytokines
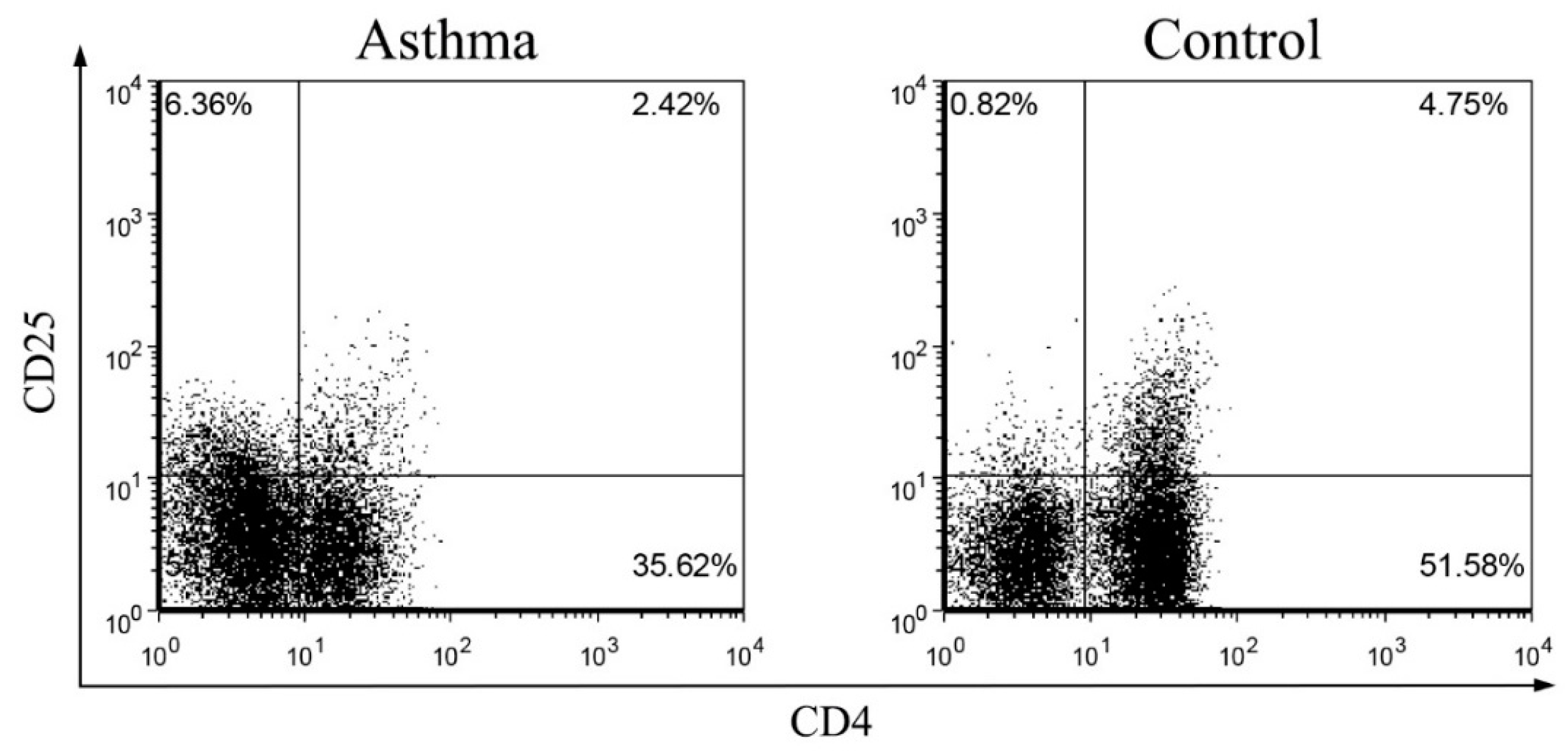
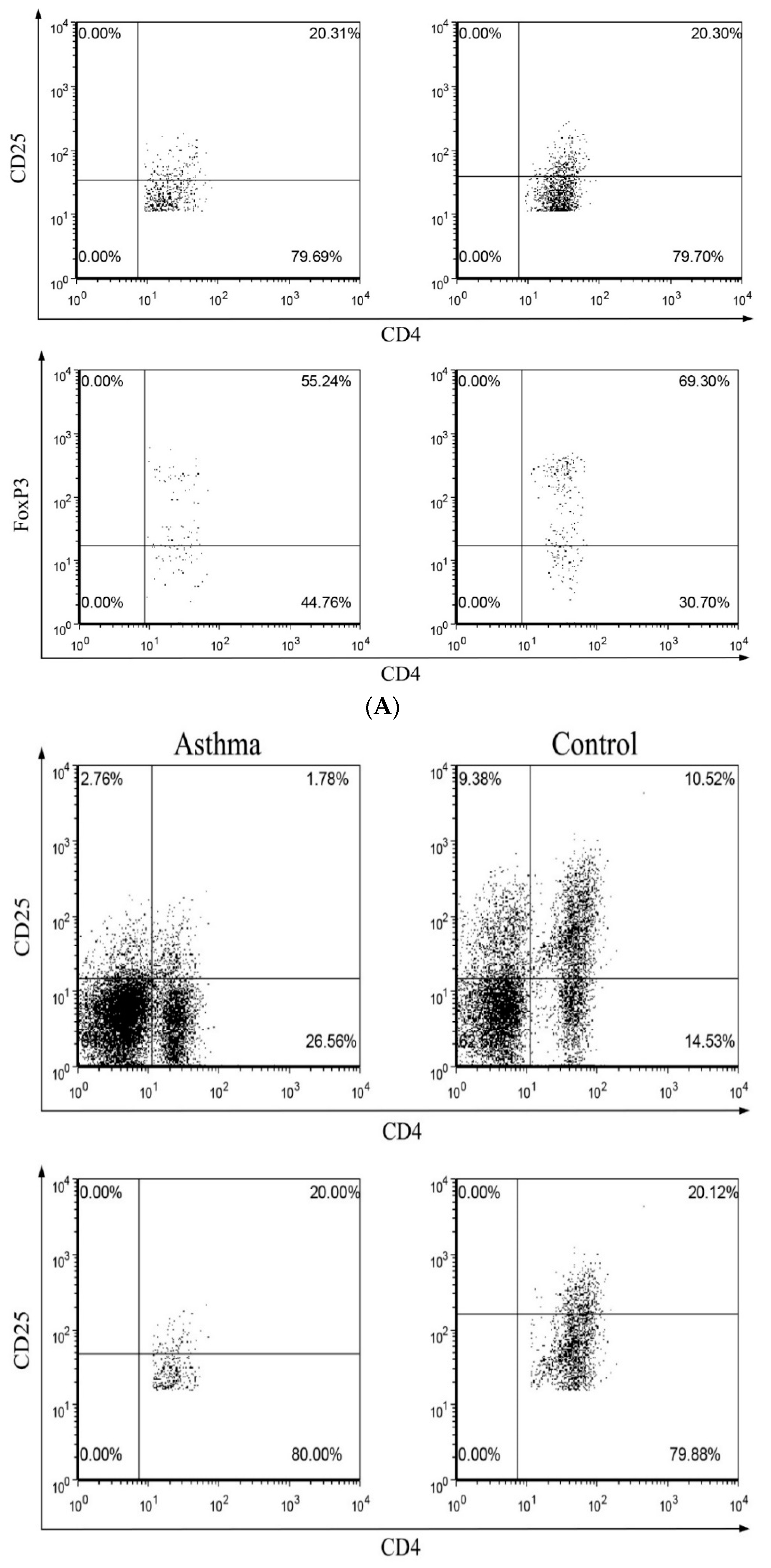
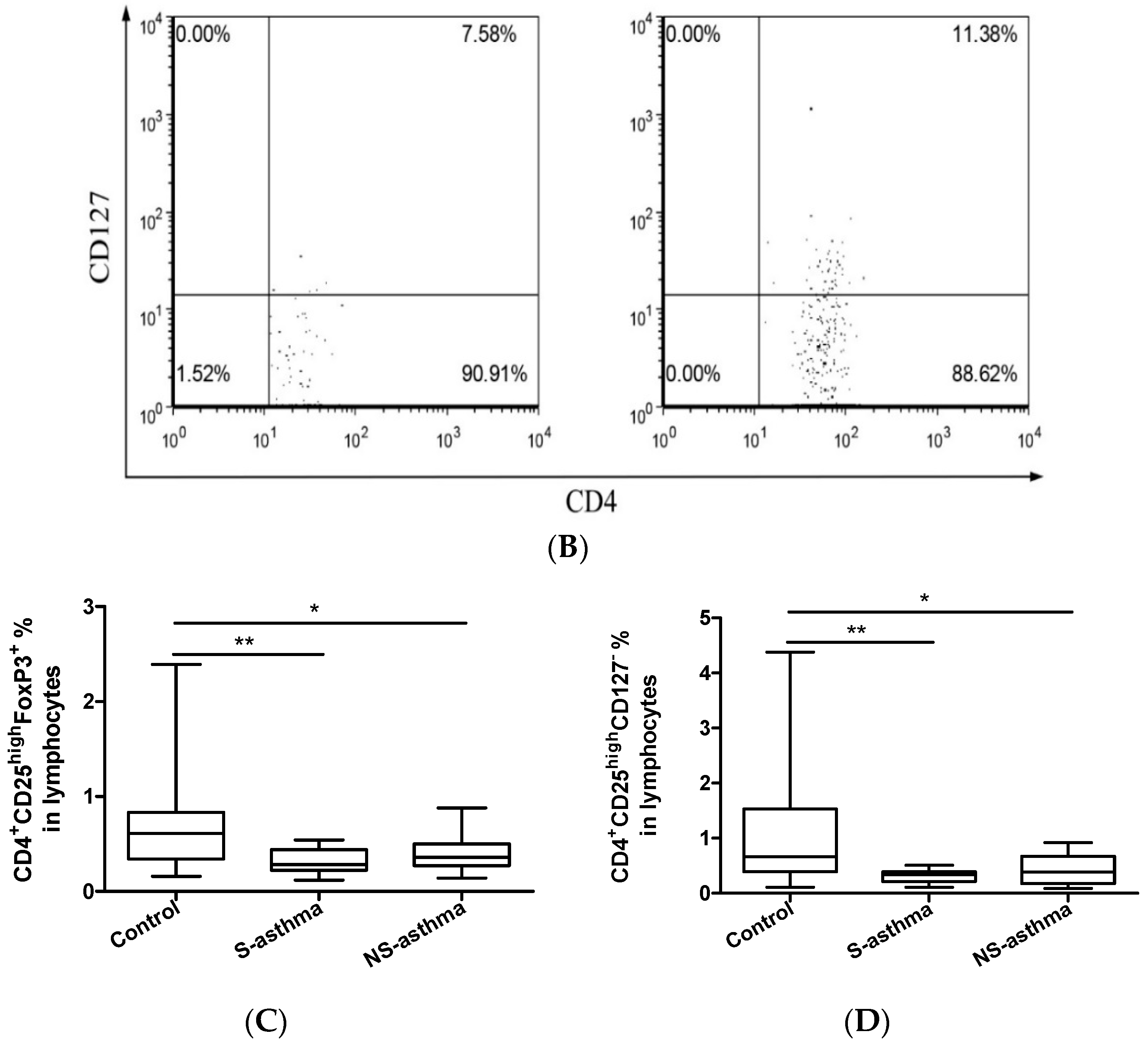
2.3. Circulating Regulatory T Lymphocytes
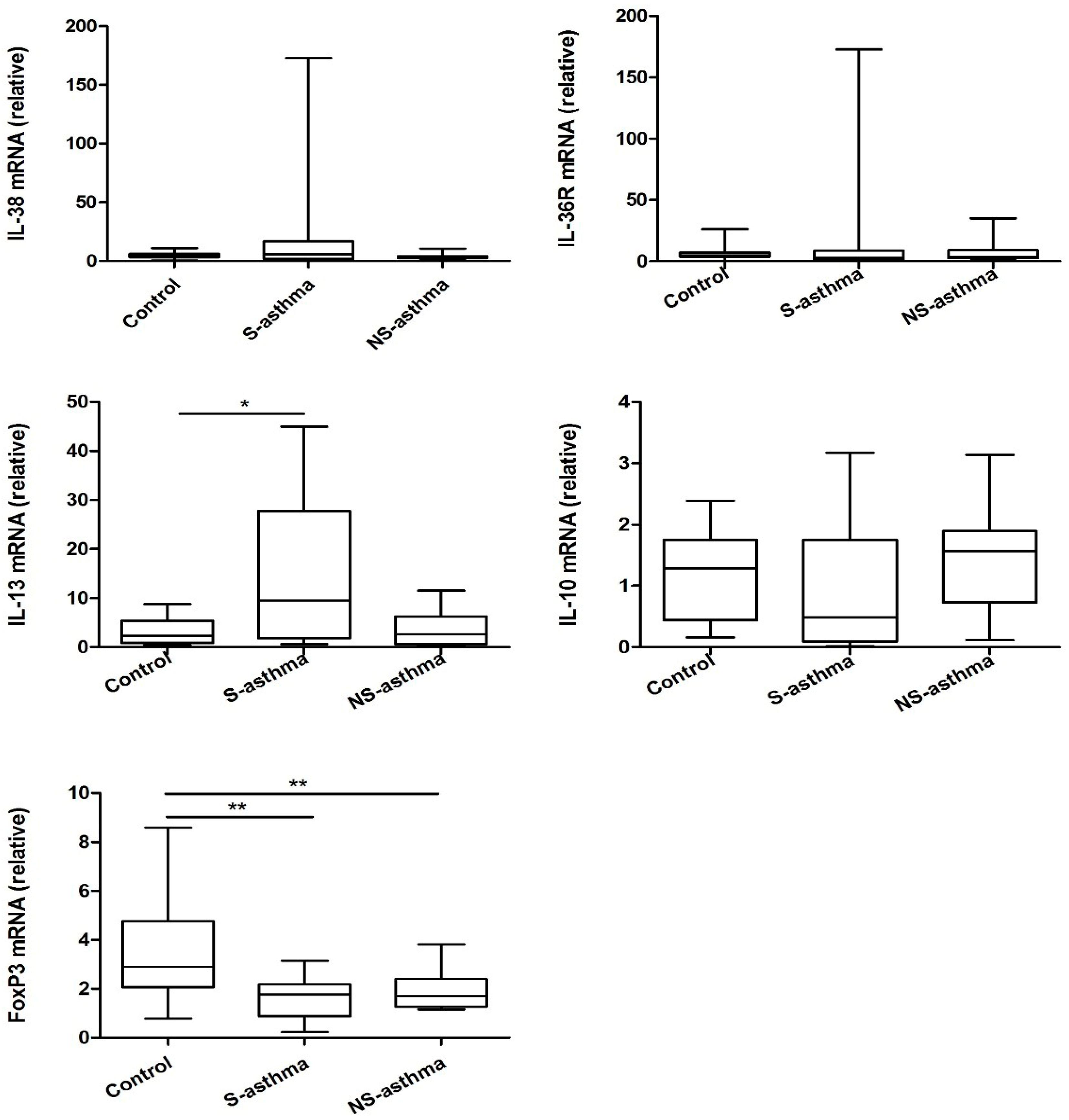
2.4. Quantitative Analysis of the mRNA Expression of IL-38
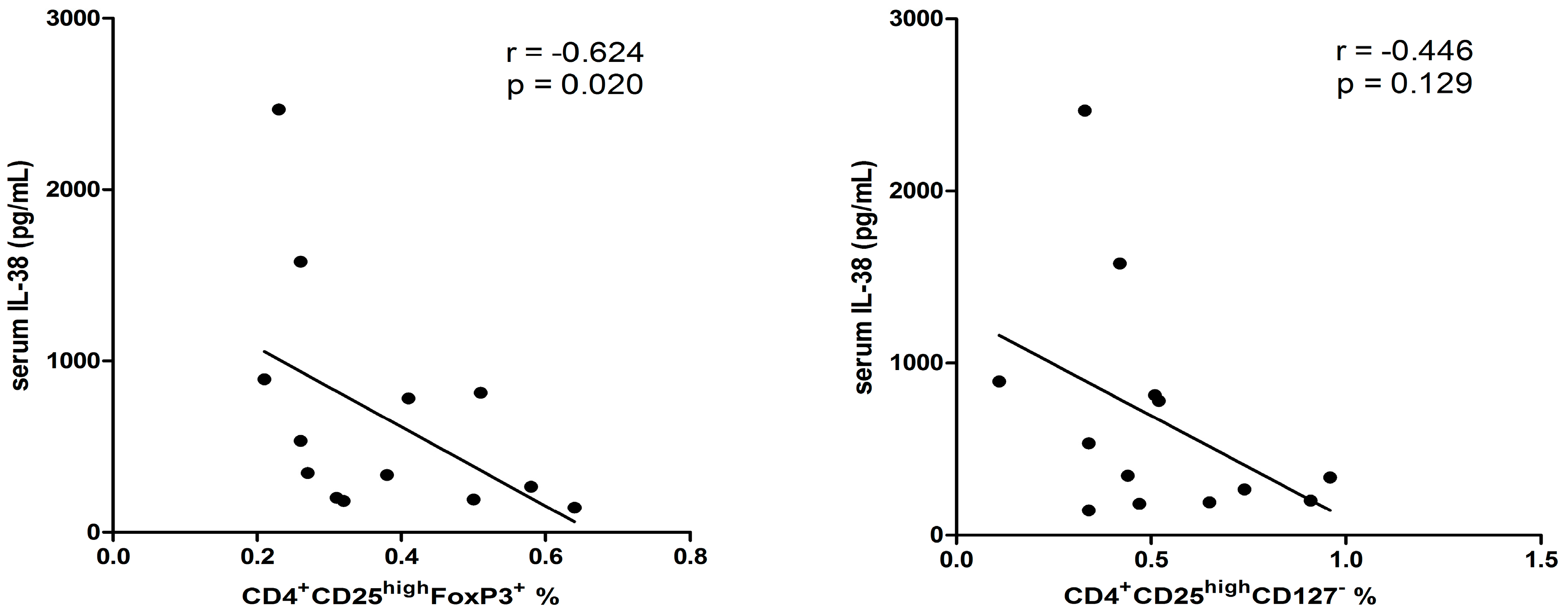
2.5. Correlation among Different Immunological Parameters in Asthmatic Patients
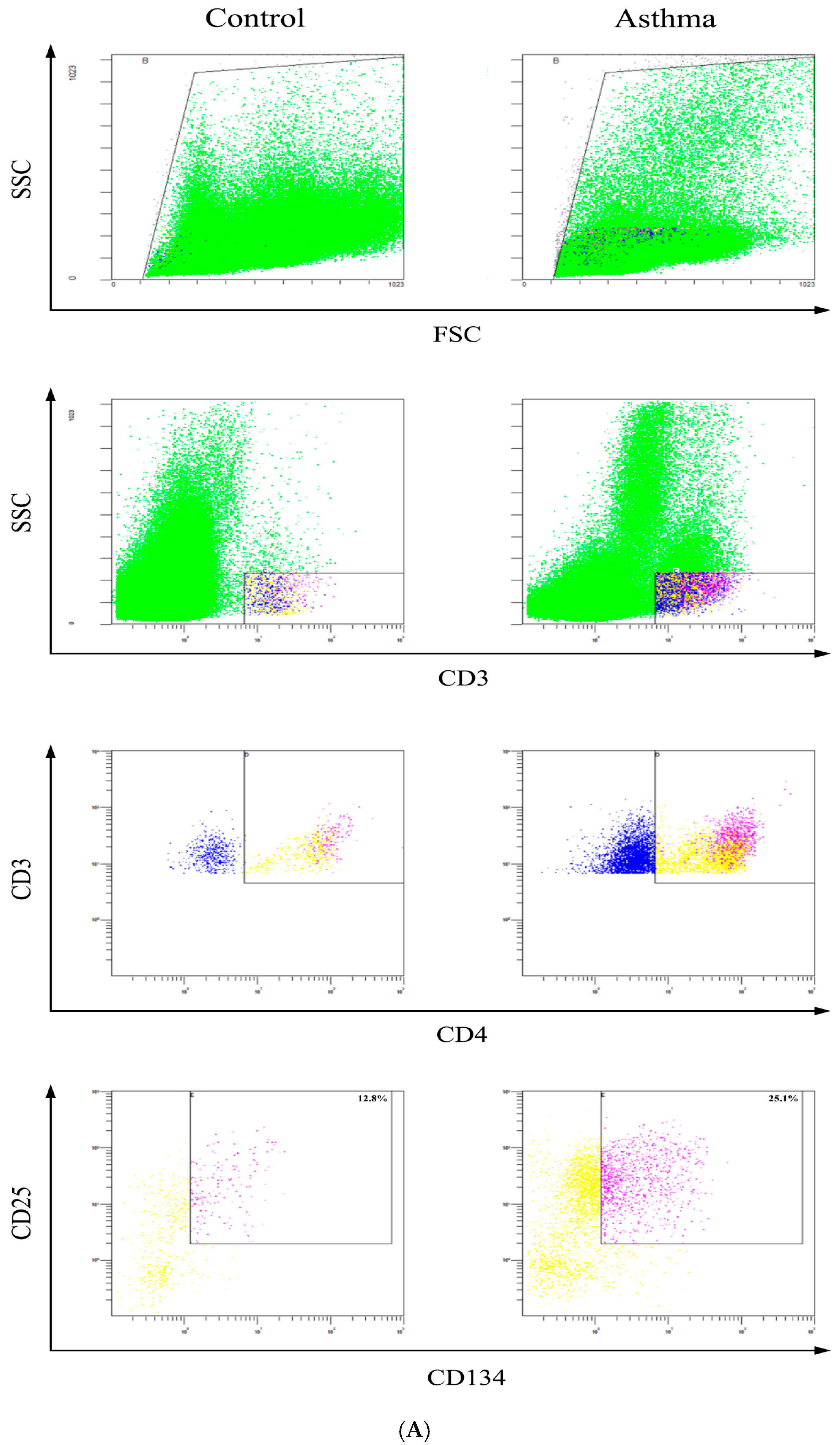
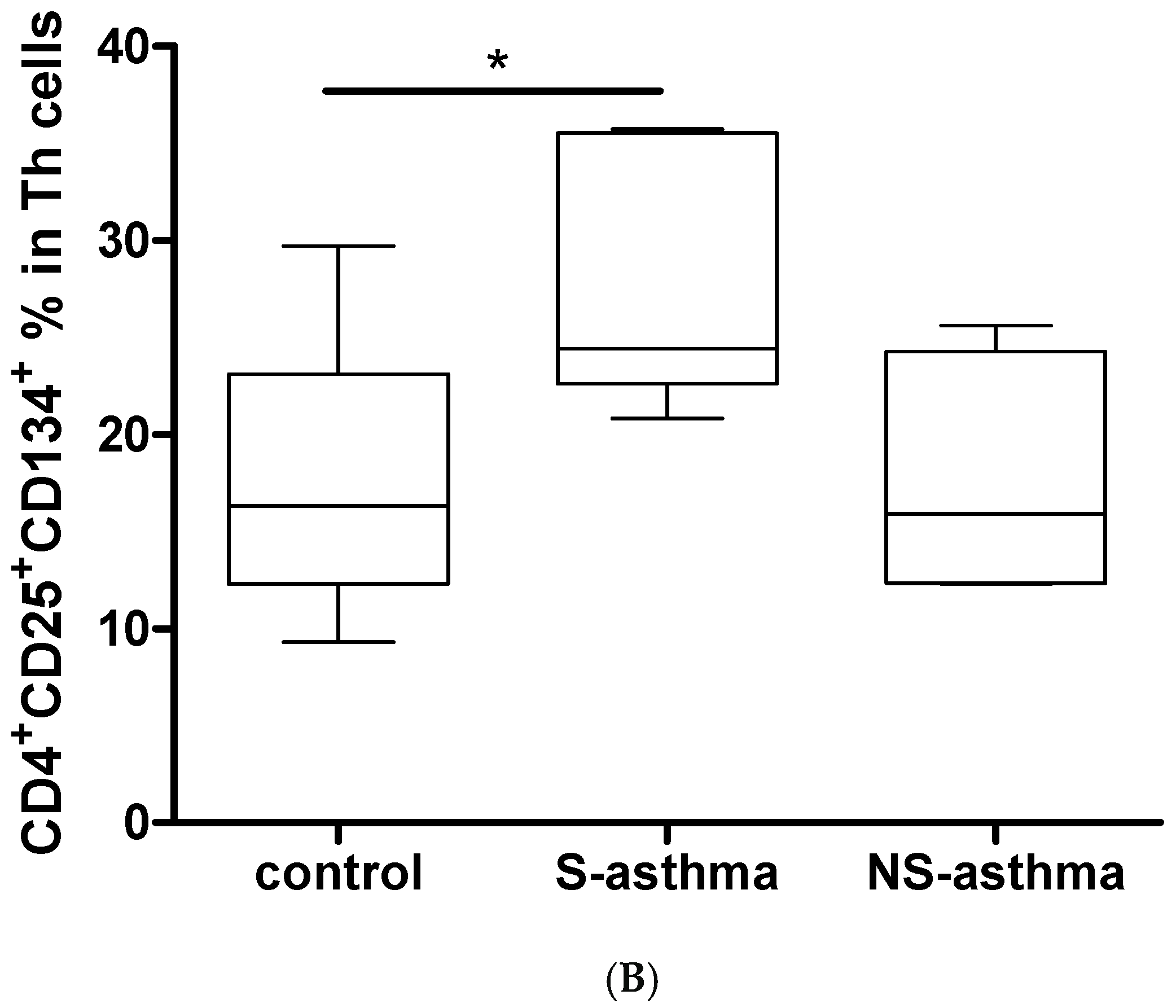
2.6. Determination of Activated CD4+CD25+CD134+ T Cells
3. Experimental Section
3.1. Ethics Statement
3.2. Asthmatic Patients, Non-Allergic Control Subjects and Blood Samples
3.3. Quantitative Analysis of Cytokines and Periostin
3.4. Flow Cytometry of Treg Lymphocytes
3.5. Real time PCR
3.6. Detection of Activated CD4+ T Cells
3.7. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Geering, B.; Stoeckle, C.; Conus, S.; Simon, H.U. Living and dying for inflammation: Neutrophils, eosinophils, basophils. Trends Immunol. 2013, 34, 398–409. [Google Scholar] [CrossRef] [PubMed]
- Eder, W.; Ege, M.J.; von Mutius, E. The asthma epidemic. N. Engl. J. Med. 2006, 355, 2226–2235. [Google Scholar] [CrossRef] [PubMed]
- Illi, S.; von Mutius, E.; Lau, S.; Niggemann, B.; Grüber, C.; Wahn, U. Perennial allergen sensitisation early in life and chronic asthma in children: A birth cohort study. Lancet 2006, 368, 763–770. [Google Scholar] [CrossRef]
- Lloyd, C.M.; Hessel, E.M. Functions of T cells in asthma: More than just TH2 cells. Nat. Rev. Immunol. 2010, 10, 838–848. [Google Scholar] [CrossRef] [PubMed]
- Strickland, D.H.; Holt, P.G. T regulatory cells in childhood asthma. Trends Immunol. 2011, 32, 420–427. [Google Scholar] [CrossRef] [PubMed]
- Hori, S.; Nomura, T.; Sakaguchi, S. Control of regulatory T cell development by the transcription factor Foxp3. Science 2003, 299, 1057–1061. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Putnam, A.L.; Xu-Yu, Z.; Szot, G.L.; Lee, M.R.; Zhu, S.; Gottlieb, P.A.; Kapranov, P.; Gingeras, T.R.; Fazekas de St Groth, B.; et al. CD127 expression inversely correlates with FoxP3 and suppressive function of human CD4+ T reg cells. J. Exp. Med. 2006, 203, 1701–1711. [Google Scholar] [CrossRef] [PubMed]
- Schaub, B.; Liu, J.; Höppler, S.; Schleich, I.; Huehn, J.; Olek, S.; Wieczorek, G.; Illi, S.; von Mutius, E. Maternal farm exposure modulates neonatal immune mechanisms through regulatory T cells. J. Allergy Clin. Immunol. 2009, 123, 774–782. [Google Scholar] [CrossRef] [PubMed]
- Schaub, B.; Liu, J.; Höppler, S.; Haug, S.; Sattler, C.; Lluis, A.; Illi, S.; von Mutius, E. Impairment of T-regulatory cells in cord blood of atopic mothers. J. Allergy Clin. Immunol. 2008, 121, 1491–1499. [Google Scholar] [CrossRef] [PubMed]
- Ling, E.M.; Smith, T.; Nguyen, X.D.; Pridgeon, C.; Dallman, M.; Arbery, J.; Carr, V.A.; Robinson, D.S. Relation of CD4+ CD25+ regulatory T-cell suppression of allergen-driven T-cell activation to atopic status and expression of allergic disease. Lancet 2004, 363, 608–615. [Google Scholar] [CrossRef]
- Ngoc, L.P.; Gold, D.R.; Tzianabos, A.O.; Weiss, S.T.; Celedon, J.C. Cytokines, allergy, and asthma. Curr. Opin. Allergy Clin. Immunol. 2005, 5, 161–166. [Google Scholar] [CrossRef] [PubMed]
- Al-Shami, A.; Spolski, R.; Kelly, J.; Keane-Myers, A.; Leonard, W.J. A role for TSLP in the development of inflammation in an asthma model. J. Exp. Med. 2005, 202, 829–839. [Google Scholar] [CrossRef] [PubMed]
- Cheung, P.F.; Wong, C.K.; Lam, C.W. Molecular mechanisms of cytokine and chemokine release from eosinophils activated by IL-17A, IL-17F, and IL-23: Implication for Th17 lymphocytes-mediated allergic inflammation. J. Immunol. 2008, 180, 5625–5635. [Google Scholar] [CrossRef] [PubMed]
- Wong, C.K.; Ho, C.Y.; Ko, F.W.; Chan, C.H.; Ho, A.S.; Hui, D.S.; Lam, C.W. Proinflammatory cytokines (IL-17, IL-6, IL-18 and IL-12) and Th cytokines (IFN-γ, IL-4, IL-10 and IL-13) in patients with allergic asthma. Clin. Exp. Immunol. 2001, 125, 177–183. [Google Scholar] [CrossRef] [PubMed]
- Dinarello, C.; Arend, W.; Sims, J.; Smith, D.; Blumberg, H.; O’Neill, L.; Goldbach-Mansky, R.; Pizarro, T.; Hoffman, H.; Bufler, P. IL-1 family nomenclature. Nat. Immunol. 2010, 11, 973. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.; Ho, A.S.; Haley-Vicente, D.; Zhang, J.; Bernal-Fussell, J.; Pace, A.M.; Hansen, D.; Schweighofer, K.; Mize, N.K.; Ford, J.E. Cloning and characterization of IL-1HY2, a novel interleukin-1 family member. J. Biol. Chem. 2001, 276, 20597–20602. [Google Scholar] [CrossRef] [PubMed]
- Van de Veerdonk, F.L.; Stoeckman, A.K.; Wu, G.; Boeckermann, A.N.; Azam, T.; Netea, M.G.; Joosten, L.A.; van der Meer, J.W.; Hao, R.; Kalabokis, V. IL-38 binds to the IL-36 receptor and has biological effects on immune cells similar to IL-36 receptor antagonist. Proc. Natl. Acad. Sci. USA 2012, 109, 3001–3005. [Google Scholar] [CrossRef] [PubMed]
- Rudloff, I.; Godsell, J.; Nold-Petry, C.A.; Harris, J.; Hoi, A.; Morand, E.F.; Nold, M.F. Brief Report: Interleukin-38 Exerts Antiinflammatory Functions and Is Associated With Disease Activity in Systemic Lupus Erythematosus. Arthritis Rheumatol. 2015, 67, 3219–3225. [Google Scholar] [CrossRef] [PubMed]
- Zhong, Y.; Yu, K.; Wang, X.; Wang, X.; Ji, Q.; Zeng, Q. Elevated Plasma IL-38 Concentrations in Patients with Acute ST-Segment Elevation Myocardial Infarction and Their Dynamics after Reperfusion Treatment. Mediat. Inflamm. 2015, 2015. [Google Scholar] [CrossRef] [PubMed]
- Hayashi, N.; Yoshimoto, T.; Izuhara, K.; Matsui, K.; Tanaka, T.; Nakanishi, K. T helper 1 cells stimulated with ovalbumin and IL-18 induce airway hyperresponsiveness and lung fibrosis by IFN-γ and IL-13 production. Proc. Natl. Acad. Sci. USA 2007, 104, 14765–14770. [Google Scholar] [CrossRef] [PubMed]
- Woodruff, P.G.; Boushey, H.A.; Dolganov, G.M.; Barker, C.S.; Yang, Y.H.; Donnelly, S.; Ellwanger, A.; Sidhu, S.S.; Dao-Pick, T.P.; Pantoja, C. Genome-wide profiling identifies epithelial cell genes associated with asthma and with treatment response to corticosteroids. Proc. Natl. Acad. Sci. USA 2007, 104, 15858–15863. [Google Scholar] [CrossRef] [PubMed]
- Takayama, G.; Arima, K.; Kanaji, T.; Toda, S.; Tanaka, H.; Shoji, S.; McKenzie, A.N.; Nagai, H.; Hotokebuchi, T.; Izuhara, K. Periostin: A novel component of subepithelial fibrosis of bronchial asthma downstream of IL-4 and IL-13 signals. J. Allergy Clin. Immunol. 2006, 118, 98–104. [Google Scholar] [CrossRef] [PubMed]
- Yuyama, N.; Davies, D.E.; Akaiwa, M.; Matsui, K.; Hamasaki, Y.; Suminami, Y.; Yoshida, N.L.; Maeda, M.; Pandit, A.; Lordan, J.L. Analysis of novel disease-related genes in bronchial asthma. Cytokine 2002, 19, 287–296. [Google Scholar] [CrossRef] [PubMed]
- Croft, M. Co-stimulatory members of the TNFR family: Keys to effective T-cell immunity? Nat. Rev. Immunol. 2003, 3, 609–620. [Google Scholar] [CrossRef] [PubMed]
- Streeter, P.R.; Zhang, X.; Tittle, T.V.; Schön, C.N.; Weinberg, A.D.; Maziarz, R.T. CD25 expression distinguishes functionally distinct alloreactive CD4+ CD134+(OX40+) T-cell subsets in acute graft-versus-host disease. Biol. Blood Marrow Transplant. 2004, 10, 298–309. [Google Scholar] [CrossRef] [PubMed]
- Wong, C.K.; Leung, T.F.; Chu, I.M.T.; Dong, J.; Lam, Y.Y.O.; Lam, C.W.K. Aberrant expression of regulatory cytokine IL-35 and pattern recognition receptor NOD2 in patients with allergic asthma. Inflammation 2015, 38, 348–360. [Google Scholar] [CrossRef] [PubMed]
- Hamelmann, E.; Gelfand, E.W. IL-5-induced airway eosinophilia–the key to asthma? Immunol. Rev. 2001, 179, 182–191. [Google Scholar] [CrossRef] [PubMed]
- Wills-Karp, M.; Luyimbazi, J.; Xu, X.; Schofield, B.; Neben, T.Y.; Karp, C.L.; Donaldson, D.D. Interleukin-13: Central mediator of allergic asthma. Science 1998, 282, 2258–2261. [Google Scholar] [CrossRef] [PubMed]
- Kabesch, M.; Schedel, M.; Carr, D.; Woitsch, B.; Fritzsch, C.; Weiland, S.K.; von Mutius, E. IL-4/IL-13 pathway genetics strongly influence serum IgE levels and childhood asthma. J. Allergy Clin. Immunol. 2006, 117, 269–274. [Google Scholar] [CrossRef] [PubMed]
- Ramadas, R.A.; Ewart, S.L.; Medoff, B.D.; LeVine, A.M. Interleukin-1 family member 9 stimulates chemokine production and neutrophil influx in mouse lungs. Am. J. Respir. Cell Mol. Biol. 2011, 44, 134–145. [Google Scholar] [CrossRef] [PubMed]
- Ramadas, R.A.; Ewart, S.L.; Iwakura, Y.; Medoff, B.D.; LeVine, A.M. IL-36α exerts pro-inflammatory effects in the lungs of mice. PLoS ONE 2012, 7, e45784. [Google Scholar] [CrossRef] [PubMed]
- Wong, C.K.; Lun, S.W.; Ko, F.W.; Wong, P.T.; Hu, S.Q.; Chan, I.H.; Hui, D.S.; Lam, C.W. Activation of peripheral Th17 lymphocytes in patients with asthma. Immunol. Investig. 2009, 38, 652–664. [Google Scholar] [CrossRef] [PubMed]
- Jia, G.; Erickson, R.W.; Choy, D.F.; Mosesova, S.; Wu, L.C.; Solberg, O.D.; Shikotra, A.; Carter, R.; Audusseau, S.; Hamid, Q. Periostin is a systemic biomarker of eosinophilic airway inflammation in asthmatic patients. J. Allergy Clin. Immunol. 2012, 130, 647–654. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Sample of the IL-38 is not available from the authors.

| Characteristics of Subjects | Non-Steroid-Treated Patients (n = 20) | Steroid-Treated Patients (n = 20) | Control Subjects (n = 20) |
|---|---|---|---|
| Sex (male/female) | 9/11 | 8/12 | 10/10 |
| Age, year (mean ± SD, range) | 7.3 ± 0.6, 6–9 | 7.5 ± 0.6, 6–9 | 7.8 ± 0.9 |
| Eosinophil % in PB | 5.0 (4.0–6.0) *** | 5.5 (5.0–6.0) *** | 2.0 (1.0–2.0) |
| Basophil % in PB | 1.0 (0.0–1.0) | 1.0 (1.0–1.0) | 1.0 (1.0–1.0) |
| Presence of at least one | |||
| Positive allergen-specific IgE, n (%) | 18 (90%) | 19 (95%) | 0 (0) |
| House dust mite Dp1, n (%) | 12 (60%) | 13 (65%) | 0 (0) |
| House dust mite Df1, n (%) | 11 (55%) | 10 (50%) | 0 (0) |
| Cockroaches, n (%) | 8 (40%) | 7 (35%) | 0 (0) |
| Cat dander, n (%) | 4 (20%) | 5 (25%) | 0 (0) |
| mRNA Expression | IL-38 mRNA |
|---|---|
| IL-36R mRNA | r = 0.407, p = 0.006 ** |
| IL-13 mRNA | r = 0.156, p = 0.714 |
| IL-10 mRNA | r = −0.348, p = 0.019 * |
| FoxP3 mRNA | r = −0.093, p = 0.566 |
| Primer | Sequence of Primers | |
|---|---|---|
| IL-10 | Forward | 5′-GCCTAACATGCTTCGAGATC-3′ |
| Reverse | 5′-TGATGTCTGGGTCTTGGTTC-3′ | |
| IL-36R | Forward | 5′-GCTGGAGTGTCCACAGCATA-3′ |
| Reverse | 5′-GCGATAAGCCCTCCTATCAA-3′ | |
| IL-38 | Forward | 5′-TTATCCTTGTGGGCTCAGTT-3′ |
| Reverse | 5′-AATCCGTTCCCTTGGCTTTT-3′ | |
| IL-13 | Forward | 5′-TGAGGAGCTGGTCAACATCA-3′ |
| Reverse | 5′-CAGGTTGATGCTCCATACCAT-3′ | |
| FoxP3 | Forward | 5′-GCACATTCCCAGAGTTCCT-3′ |
| Reverse | 5′-TTGAGTGTCCGCTGCTTC-3′ | |
| GAPDH | Forward | 5′-ATGGGGAAGGTGAAGGTCG-3′ |
| Reverse | 5′-GGGGTCATTGATGGCAACAATA-3′ |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chu, M.; Chu, I.M.T.; Yung, E.C.M.; Lam, C.W.K.; Leung, T.F.; Wong, G.W.K.; Wong, C.K. Aberrant Expression of Novel Cytokine IL-38 and Regulatory T Lymphocytes in Childhood Asthma. Molecules 2016, 21, 933. https://doi.org/10.3390/molecules21070933
Chu M, Chu IMT, Yung ECM, Lam CWK, Leung TF, Wong GWK, Wong CK. Aberrant Expression of Novel Cytokine IL-38 and Regulatory T Lymphocytes in Childhood Asthma. Molecules. 2016; 21(7):933. https://doi.org/10.3390/molecules21070933
Chicago/Turabian StyleChu, Man, Ida M.T. Chu, Edmund C.M. Yung, Christopher W.K. Lam, Ting F. Leung, Gary W.K. Wong, and Chun K. Wong. 2016. "Aberrant Expression of Novel Cytokine IL-38 and Regulatory T Lymphocytes in Childhood Asthma" Molecules 21, no. 7: 933. https://doi.org/10.3390/molecules21070933
APA StyleChu, M., Chu, I. M. T., Yung, E. C. M., Lam, C. W. K., Leung, T. F., Wong, G. W. K., & Wong, C. K. (2016). Aberrant Expression of Novel Cytokine IL-38 and Regulatory T Lymphocytes in Childhood Asthma. Molecules, 21(7), 933. https://doi.org/10.3390/molecules21070933






